Introduction
Invasive breast carcinoma of no special type
(IBC-NST) comprises a large and heterogenous group of invasive
breast carcinomas lacking the morphological hallmarks of special
histologic types (1). IBC-NST is
the most common histological subtype, accounting for 80% of all
invasive breast carcinomas (2). In
recent years, several morphologic patterns of IBC-NST have been
recognized and most of them have also been acknowledged in the
latest classification of the World Health Organization (WHO). These
include: Carcinomas with oncocytic, sebaceous, lipid-rich,
glycogen-rich/clear cell, medullary, neuroendocrine, pleomorphic,
osteoclast-like giant cells, choriocarcinomatous and melanocytic
features/patterns (2).
Glycogen-rich/clear cell carcinoma (GRCCC) of the
breast has some common features with clear cell carcinomas of other
organs. It was first described by Hull et al in
1981(3) and its incidence has been
estimated as less than 3%. According to the WHO classification of
tumors, GRCCC of the breast is defined as a rare subtype of IBC-NST
in which more than 90% of the tumor cells have abundant clear
cytoplasm containing glycogen. To date, the clinicopathologic and
biologic features of GRCCC are yet to be described and clinical
outcome is still unclear. Some authors hypothesized that it may be
similar to other breast carcinomas when compared on a stage-matched
basis (4), while others indicated a
less favorable outcome for patients with GRCCC (5,6).
In this article, we report two cases of GRCCC of the
breast encountered in our clinical practice over a period of 5
years (2015-2020), featuring a similar immunophenotype, but
completely different morphological patterns and patient
outcome.
Case report 1
We encountered the first case of GRCCC in our daily
clinical practice in September 2015, when a 64-year-old female
patient with no family history of breast cancer, presented with a
lump in the left breast. Initial physical examination revealed a
palpable, tender and irregular mass with retraction of the
overlying epidermis, located in the upper-outer quadrant of the
left breast. Ultrasonography revealed an irregular, hypoechoic
breast tumor with heterogeneous internal echoes, qualifying as
BI-RADS 5. The core needle biopsy was signed out as infiltrative
breast carcinoma with clear cells, defer to excision for final
classification. The patient underwent total mastectomy with
axillary lymph node dissection and the specimen was sent to our
Department of Pathology for histopathological examination. A
white-tan, spiculated tumor mass measuring 46/32/28 mm was
identified in the upper-outer quadrant of the breast.
Histologically, the tumor was composed of polygonal
epithelial cells with well-defined borders and strikingly clear or
finely-granular cytoplasm, arranged in invasive nests, cords and/or
trabecular structures, separated by thin fibrous septa (Fig. 1A). Tumor cell nuclei had irregular
shapes and sizes, featuring granular chromatin pattern and
prominent mitotic figures (Fig.
1B). The average mitotic index was 17 mitoses per 10 HPF.
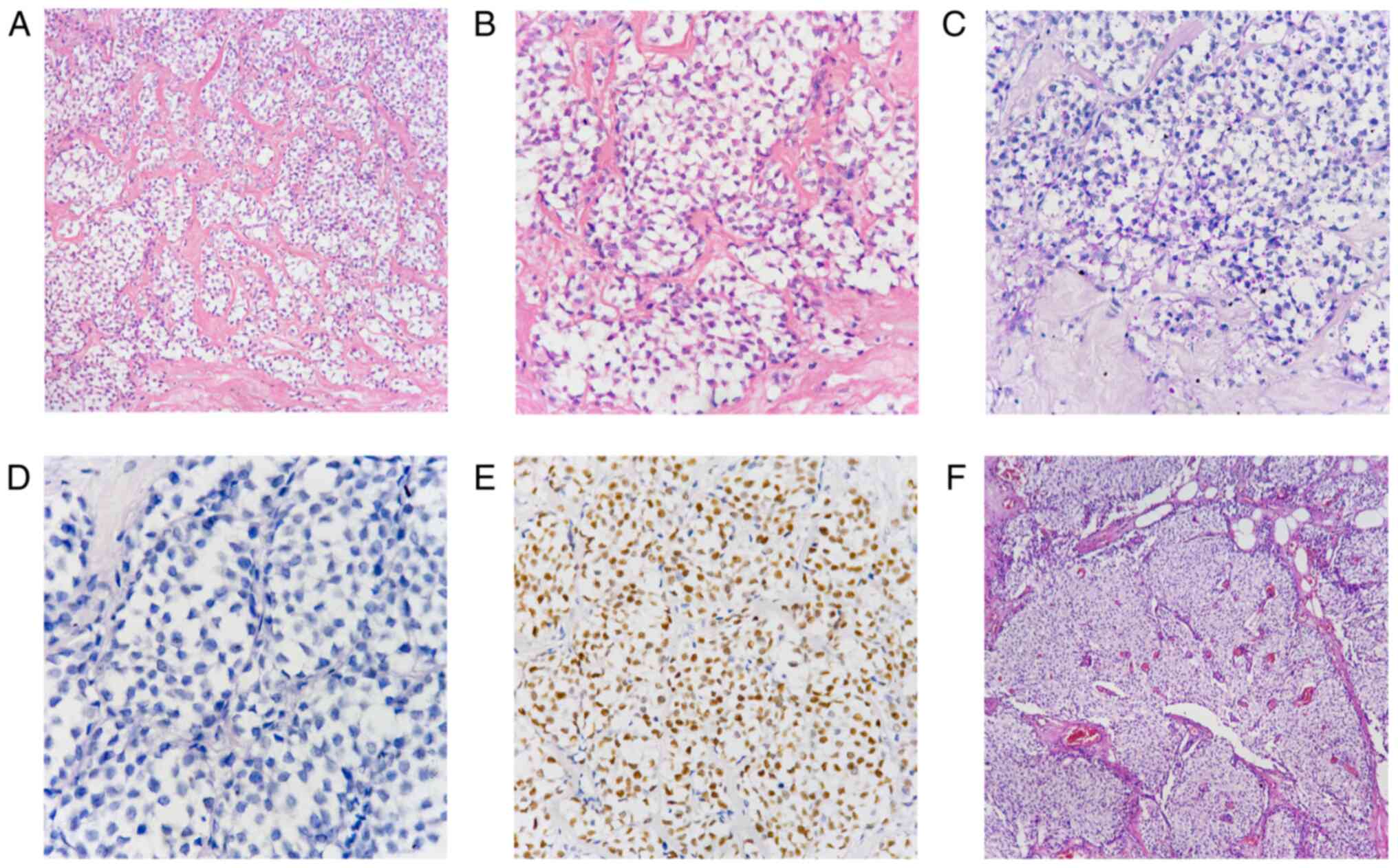 | Figure 1(A) Infiltrative nests, cords or
trabecular structures composed of polygonal epithelial cells with
well-defined borders and strikingly clear or finely-granular
cytoplasm (H&E stain, magnification, x200). (B) Tumor cell
nuclei had irregular shapes and sizes, featuring granular chromatin
pattern and prominent mitotic figures (H&E stain,
magnification, x400). (C) Tumor cells showed diffuse
finely-granular cytoplasmic positivity in periodic acid-Schiff
staining (PAS stain, magnification x400). (D) Finely-granular
cytoplasmic positivity of the tumor cells was completely absent in
periodic acid-Schiff staining with diastase digestion (PAS-D stain,
magnification, x400). (E) Diffuse nuclear positivity for ER in
91-100% of tumor cells (IHC staining with DAB chromogen,
magnification, x400). (F) Bulk of the tumor was composed of nodular
structures with thin fibrovascular septa and dilated blood spaces
(H&E stain, magnification, x100). H&E, hematoxylin and
eosin; ER, estrogen receptor; IHC, immunohistochemistry. |
Focal lympho-vascular infiltration was noted and
macrometastases were identified in four of the 12 ipsilateral
axillary lymph nodes examined. Periodic acid-Schiff staining method
employing diastase confirmed the presence of cytoplasmic glycogen
granules (Fig. 1C and D).
Immunostaining with myoepithelial markers revealed
no intraductal component within or at the periphery of the tumor.
Across the entire proliferation, the tumor cells were estrogen
receptor (ER)-positive (Fig. 1E),
progesterone (PR)-negative and HER2-negative (score 0). Ki67
proliferation index was 25%. E-cadherin revealed crisp membranous
positivity in all tumor cells and p53 was normal, showing
‘wild-type’ pattern of expression. The cytokeratin panel revealed
positivity for CK7 and CK8/18.
Based on these findings, the morphological features
and immunohistochemical phenotype supported the diagnosis of
glycogen-rich clear cell carcinoma of the breast, pT1c pN2a.
A postsurgical full-body CT revealed sclerotic
lesions located in the liver and third and fourth lumbar spine
vertebrae (L3-L4). The patient underwent eight cycles of
chemotherapy, but unfortunately succumbed to the disease after 7
months.
Case report 2
The second case we encountered was diagnosed in
January 2020, in a 54-year-old woman who underwent simple
subcutaneous mastectomy accompanied by sentinel lymph node excision
for a clinically palpable left/right breast mass. The lesion was
previously diagnosed as IBC-NST on core needle biopsy. A
subcutaneous simple mastectomy specimen, measuring 230x190x70 mm,
with coded surgical markings was received in buffered formalin. The
posterior resection margin was inked black and the specimen was
serially sectioned at 5-mm intervals. Upon gross examination, the
tumor mass was located in the outer-upper quadrant, measured
18/16/12 mm and appeared to be well-circumscribed, with
heterogeneous appearance, featuring tan and hemorrhagic areas. The
whole tumor was embedded, including surrounding fibrous areas and
resection margins. Complete submission of the sentinel lymph node
for routine histology and immunohistochemistry was performed,
according to standard protocols. All sections were stained with
hematoxylin and eosin (H&E). Periodic acid Schiff stain (PAS)
with and without prior diastase digestion, Mucicarmine and Alcian
Blue stains were performed on representative histological
sections.
Microscopic examination revealed a relatively
homogenous proliferation of average-sized polygonal cells with
sharply-defined borders, clear or finely granular cytoplasm and
round to oval nuclei with clumped chromatin and prominent nucleoli.
Cytoplasmic clearing was consistent across the entire tumor.
Nuclear atypia was mild to moderate and the highest mitotic index
was 6 mitoses per 10 HPF. The neoplastic cells were arranged in
compact nodular structures featuring thin and inconspicuous
fibro-vascular septa, with focally dilated blood spaces (Fig. 1F). The tumor nests lacked peripheral
myoepithelial lining and were closely packed together, forming a
well-circumscribed but unencapsulated multinodular structure
(Fig. 2A). Focal stromal invasion
was noted in the form of solid nests (Fig. 2B). No lympho-vascular invasion or
necrosis was noted. There were no proliferative epithelial changes
in the surrounding breast tissue, except for non-proliferative
fibrocystic changes.
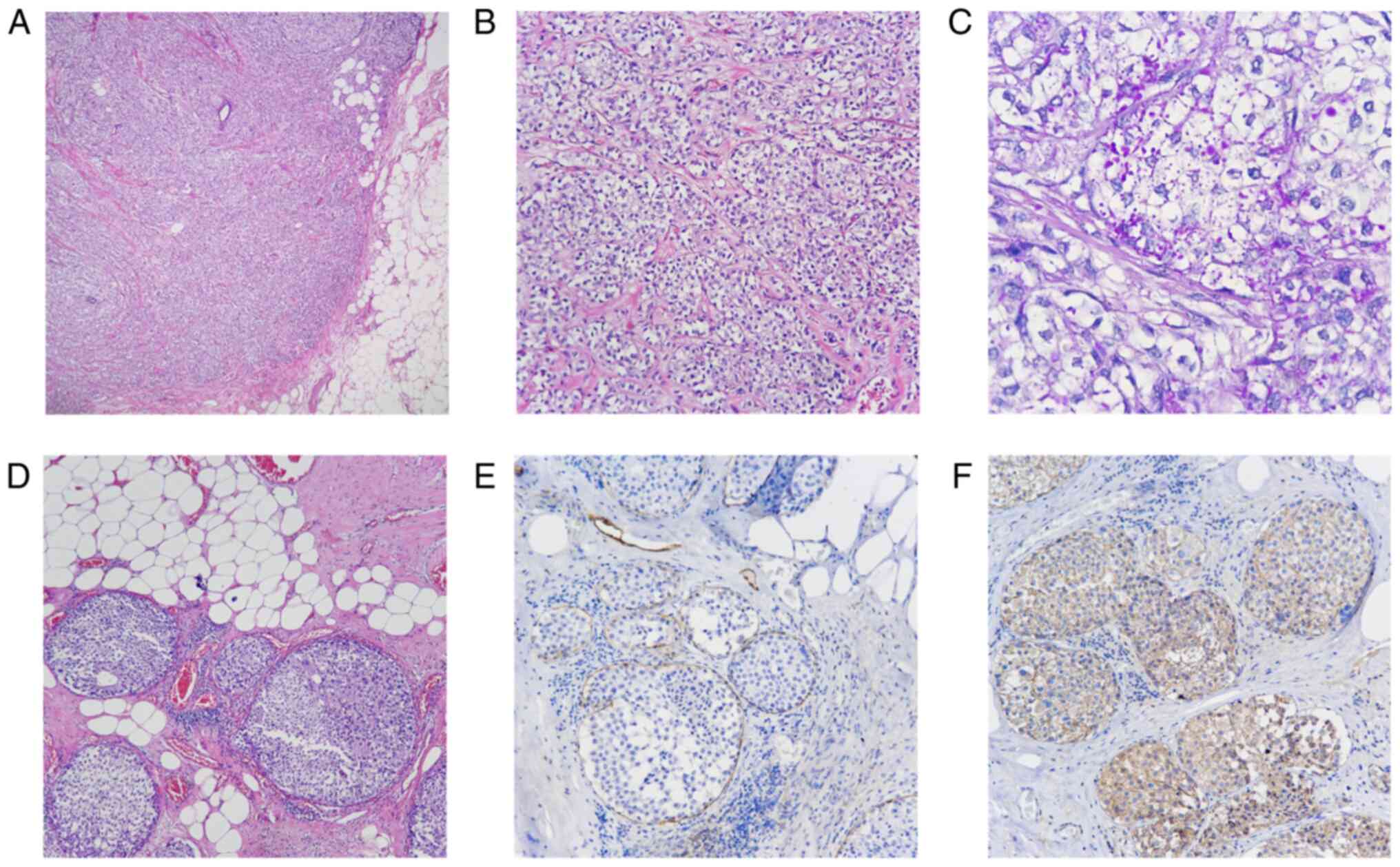 | Figure 2(A) The periphery of the tumor mass
well-defined, with pushing borders infiltrating the surrounding fat
(H&E stain, magnification, x40). (B) Stromal invasion was noted
in the form of solid nests, similar to the ones observed in the
first case (H&E stain, magnification, x100). (C) Tumor cells
showed diffuse finely-granular cytoplasmic positivity in periodic
acid-Schiff staining (PAS stain, magnification, x400). (D)
Periphery of the tumor showed areas of in situ glycogen-rich
clear cell carcinoma (H&E stain, magnification, x200). (E) The
in situ component revealed a continuous layer of
myoepithelial cells at the periphery, demonstrated by smooth muscle
myosin (SMM) staining (IHC with DAB chromogen, magnification,
x200). (F) Both the in situ and invasive areas revealed
crisp membranous positivity for E-cadherin (IHC with DAB chromogen,
magnification, x200). H&E, hematoxylin and eosin; IHC,
immunohistochemistry. |
Upon histochemical examination, the vast majority of
cells revealed variable granular cytoplasmic positivity in PAS
staining, which vanished upon diastase pre-treatment (Fig. 2C). Both Mucicarmine and Alcian Blue
stains were negative.
Immunostaining for p63 and smooth muscle myosin
revealed limited areas of intraductal component with clear cell
features and solid papillary pattern at the periphery of the tumor
(Fig. 2D and E). The bulk of the tumor lacked
myoepithelial cells both within and at the periphery of the
nodules. Tumor cells were strongly and diffusely positive for ER,
but completely negative for PR. HER2 was negative, revealing
incomplete membranous staining in 25% of the tumor cells (score 1+)
and Ki67 proliferation index was 5%. GATA binding protein 3 (GATA3)
was also diffusely positive, in compliance with a breast primary
tumor. E-cadherin revealed crisp membranous positivity both within
the intraductal and invasive components (Fig. 2F). Synaptophysin and chromogranin
did not show any neuroendocrine differentiation.
The sentinel lymph node examined in 35 different
sections, including routine H&E staining as well as AE1/AE3 and
CK7 immunolabeling, revealed reactive follicular hyperplasia, sinus
histiocytosis and lipomatous atrophy, with no tumor cells
whatsoever.
Based on these findings, the morphological features
and immunohistochemical phenotype supported the diagnosis of
glycogen-rich clear cell carcinoma of the breast with solid
papillary pattern, pT1c pN0 (sn).
A thorough radiological and clinical assessment did
not reveal any metastatic foci or other primary tumors. To date,
the patient has been followed for 6 months with no evidence of
recurrence or metastasis.
Discussion
Invasive breast carcinoma comprises a heterogenous
group of diseases with various pathologic features, genomic
alterations, response to treatment and clinical outcome. Most
tumors are thought to derive from the terminal ductal lobular unit
and up to 75% of invasive breast carcinomas are classified as
ductal carcinomas. The remaining 25% of breast carcinomas are
divided into special histologic subtypes, based on the presence of
various characteristic morphologic features (2).
Glycogen-rich clear cell carcinoma (GRCCC) is an
extremely rare subtype of breast carcinoma, accounting for anywhere
between 0.1 and 3% of all invasive breast cancers (7). The actual incidence varies from study
to study, but most authors agree that it represents a very uncommon
morphologic pattern of invasive breast carcinoma of no special
type.
According to the World Health Organization
definition, GRCCC is a morphological subtype of breast carcinoma in
which more than 90% of the tumor cells feature clear or
finely-granular cytoplasm with diastase-sensitive glycogen deposits
(2). Tumor cells have moderate to
marked nuclear atypia, well-defined cell borders and may be
arranged in sheets, nests, cords, solid, papillary, micropapillary,
cribriform, alveolar or tubular structures. Some authors also
report neuroendocrine, apocrine and mucinous differentiation
(4,8-10).
Most cases of GRCCC are invasive tumors. Pure intraductal GRCCCs
are exceptionally rare (11,12).
GRCCC usually affects women in the 5th decade of
life (13). Patients may present
with tumor mass, skin dimpling, nipple retraction or pain. Tumor
diameter usually ranges between 1 and 6.5 cm (14), but can reach up to 15 cm (14) or more. Eun et al recently
published a review of radiologic features of GRCCC (15). Based on their findings, GRCCC
usually presents as an oval-shaped spiculated tumor mass with
irregular margins and fine calcifications (8,16-19).
However, mammography may sometimes be inconclusive due to dense
breast tissue (20) or may feature
benign characteristics (21).
The first case of GRCCC was described by Hull et
al in 1981(3). To the best of
our knowledge, only three glycogen-rich breast carcinomas with
solid papillary pattern have been reported to date (3,4,22).
Case 2 encountered in our practice was extremely similar to the
case reported by Hull et al featuring separate areas of
solid papillary growth and invasive breast carcinoma (3).
An article published by Gürbüz and Özkara in 2003
was the first publication to detail the immunophenotypical profile
of GRCCC, with focus on the differential diagnosis with clear cell
renal cell carcinoma (CCRCC). Both tumors may show positivity for
CK7, CK8/18 and CK19. Coexpression of vimentin and cytokeratin is a
classic feature in CCRCC, but it can also be identified in high
grade invasive ductal carcinomas. The absence of ER and PR
expression may be interpreted as a feature of non-breast
carcinomas, but can also be seen in high grade breast carcinomas.
HMWCK 34β12E (pan-epithelial cytokeratin including CK1, CK5, CK10
and CK14) is expressed by all epithelial cells of the breast and
has been reported to be rarely positive in CCRCC (22). Currently, we benefit from several
immunohistochemical markers which can accurately differentiate
between a GRCCC primary to the breast and a CCRCC. Mammaglobin,
gross cystic disease fluid protein 15 (GCDFP-15) and GATA3 are
positive in GRCCC and negative in CCRCC. On the other hand, renal
cell carcinoma (RCC), paired-box gene 8 (PAX8) and cluster of
differentiation 10 (CD10) are positive in CCRCC and negative in
GRCCC. However, it is important to emphasize that from a
morphological standpoint, GRCCC of the breast retains the
fundamental characteristics of clear cell tumors from other
organs.
Normal breast histology may feature clear cell areas
as a consequence of physiological changes occurring during
pregnancy (14). In regards to
breast tumors with clear cell features, the differential diagnosis
of GRCCC includes secretory carcinoma, lipid-rich carcinoma,
apocrine carcinoma and mucinous carcinoma (23). Cutaneous tumors with clear cell
morphology such as clear cell hidradenoma, sebaceous neoplasms and
malignant melanoma may involve the breast primarily or secondarily
and may be considered in the differential diagnosis. On the other
hand, glycogen rich clear cell tumors can also arise and
metastasize from other organs such as lung, endometrium, salivary
gland, ovary and uterine cervix (13,20,22,24,25).
In most cases, an appropriate immunohistochemical panel can
identify the cell lineage and special stains such as PAS and PAS-D
can confirm the presence of intracytoplasmic glycogen. Molecular
techniques are not required in order to establish this diagnosis,
but may also play an important role, based on the morphological
features of the tumor. For example, detection of t(12;15)(p13;q25)
chromosomal translocation results in the ETV6-NTRK3 fusion gene,
which is specific for secretory carcinoma.
Differential diagnosis with other mammary tumors
with clear cells is also challenging. For example, clear cell
‘sugar’ tumor, a tumor with perivascular epithelioid
differentiation (PEComa), is characterized by a solid proliferation
of epithelioid tumor cells with glycogen-rich clear cytoplasm. This
neoplasm has originally been described in the lung, but has
recently been reported in extra-pulmonary sites as well, including
the breast. Unlike our two cases of GRCCC, the immunophenotype of
PEComa is characterized by intense positivity for HMB45, variable
expression of muscle markers and complete absence of cytokeratins
(26).
Many authors agree that the immunophenotype of GRCCC
tends to be ER-positive, PR-negative and HER2-negative (27). Apart from steroid receptors and
HER2, the molecular features of GRCCC are largely unknown. However,
multiple authors have reported ER-negative, PR-negative, HER2
positive cases (28) as well as
triple-negative cases (27), which
suggests that GRCCC is almost as heterogenous as IBC-NST. Kuroda
et al published a series of 20 cases in which 56% were
luminal A, 12% were luminal B and 32% were triple-negative
(13). The largest study to date,
based on the Surveillance, Epidemiology and End Results (SEER)
database, including a total of 155 cases, identified 45% grade 3
carcinomas with triple-negative phenotype (7).
Due to the rarity of GRCCC of the breast (less than
160 cases to date, mainly as isolated reports or short series), the
overall prognosis of this tumor is still controversial.
Conventional wisdom holds that the outcome of patients with GRCCC
of the breast is not favorable and is probably at least the same or
worse than that of IBC-NST when compared on a stage-matched basis
(13,20). The SEER database study including
data from 1973 to 2015 concluded that GRCCC is an aggressive tumor
with poorer prognosis irrespective of AJCC stage, tumor grade,
patient age, treatment and ER/PR/HER2 status (7). Hull and Warfel reported that 5 of 10
patients with GRCCC died of metastatic disease (24). Toikkanen and Joensuu reported that 5
of 6 patients presented with axillary lymph node metastases at
diagnosis and died of the disease within 7 years (5). Fisher et al reported a poorer
outcome among 45 patients with GRCCC, greater frequency of nodal
metastasis, and higher histologic grade when compared to 1,510
cases of non-clear cell breast carcinoma (6). On the other hand, Ma et al
reported 24 cases with no significant difference in overall
survival between GRCCC and control invasive breast carcinomas when
matched by age, tumor size, nodal status, and immune-phenotype
(29). Hayes et al reported
21 cases of GRCCC and also found that prognosis was not different
from non-GRCCC when the tumor was matched by size, grade, and lymph
node status (4). Our two cases
revealed divergent patient outcome as well. The first case showed a
highly infiltrative tumor, with lymph node and distant metastases.
The patient died within 16 months of metastatic disease. The second
case showed a well-defined breast mass with solid papillary
architecture and no sentinel lymph node metastasis. The patient is
well today and has had no clinical recurrence of the disease after
6 months of follow-up without radiotherapy or chemotherapy. The
prognosis is probably better if the tumor is detected early and if
there are no lymph node metastases.
In conclusion, GRCCC is an extremely rare breast
cancer subtype with controversial prognosis. While some authors
report no significant difference in survival when stage-matched
with IBC-NST, others observed poorer outcome in patients with
GRCCC. After correlating contradictory literature data with our
clinical practice findings, we conclude that GRCCC is a
heterogenous subtype of breast cancer with a wide morphological
spectrum and variable clinical outcome which should be managed
similarly to IBC-NST. While some morphological features such as
solid papillary pattern may hint at a more favorable outcome,
similar to IBC-NST, these are simply speculations which warrant
further study. We believe that to date, the molecular phenotype
remains the main variable with clinical outcome significance and
further molecular studies would not only enhance the knowledge on
GRCCC but would also pave the way for novel treatment modalities
for this peculiar mammary malignancy.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
Further information regarding the case reports is
available from the corresponding author on reasonable request.
Authors' contribution
TAG, OM and ACL had substantial contributions to the
conception of the article and the acquisition of clinical data. TT,
DC, OT, NS and REB had substantial contributions to the analysis
and interpretation of data. All the authors were involved in
writing the manuscript and revised the manuscript critically for
important intellectual content, approved the final version to be
published and agreed to be accountable for all aspects of the work
in ensuring that questions related to the accuracy or integrity of
any part of the work are resolved.
Ethics approval and consent to
participate
The present report was conducted in accordance with
the Declaration of Helsinki, and written informed consent was
obtained from all patients prior to enrollment in the analysis.
Patient consent for publication
Patient consent for the publication of the images
was obtained before publication.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Thondavadi SR, Krishnamurthy J and
Gubbanna VM: A case report of glycogen-rich clear cell carcinoma of
breast. Indian J Pathol Microbiol. 53:374–375. 2010.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Allison KH, Brogi E and Ellis IO: WHO
Classification of Tumours: Breast Tumours: 5th edition.
International Agency for Research on Cancer, Lyon, pp102-109,
2019.
|
|
3
|
Hull MT, Priest JB, Broadie TA, Ransburg
RC and McCarthy LJ: Glycogen-rich clear cell carcinoma of the
breast: A light and electron microscopic study. Cancer.
48:2003–2009. 1981.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hayes MM, Seidman JD and Ashton MA:
Glycogen-rich clear cell carcinoma of the breast: A
clinicopathologic study of 21 cases. Am J Surg Pathol. 19:904–911.
1995.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Toikkanen S and Joensuu H: Glycogen-rich
clear-cell carcinoma of the breast: A clinicopathologic and flow
cytometric study. Hum Pathol. 22:81–83. 1991.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fisher ER, Tavares J, Bulatao IS, Sass R
and Fisher B: Glycogen-rich, clear cell breast cancer: With
comments concering other clear cell variants. Hum Pathol.
16:1085–1090. 1985.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zhou Z, Kinslow CJ, Hibshoosh H, Guo H,
Cheng SK, He C, Gentry MS and Sun RC: Clinical features, survival
and prognostic factors of glycogen-rich clear cell carcinoma (GRCC)
of the breast in the U.S. Population. J Clin Med.
8(246)2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Sørensen FB and Paulsen SM: Glycogen-rich
clear cell carcinoma of the breast: A solid variant with mucus. A
light microscopic, immunohistochemical and ultrastructural study of
a case. Histopathology. 11:857–869. 1987.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Di Tommaso L, Pasquinelli G, Portincasa G
and Santini D: Glycogen-rich clear-cell breast carcinoma with
neuroendocrine differentiation features. Pathologica. 93:676–680.
2001.PubMed/NCBI(In Italian).
|
|
10
|
Ionescu CA, Matei A, Navolan D, Dimitriu
M, Bohâltea R, Neacsu A, Ilinca C and Ples L: Correlation of
ultrasound features and the Risk of Ovarian Malignancy Algorithm
score for different histopathological subtypes of benign adnexal
masses. Medicine (Baltimore). 97(e11762)2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Salemis NS: Intraductal glycogen-rich
clear cell carcinoma of the breast: A rare presentation and review
of the literature. Breast Care (Basel). 7:319–321. 2012.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Seki H, Sasaki K, Morinaga S, Asanuma F,
Yanaihara H, Kaneda M, Suzuki K, Ishii Y, Kamiya N, Osaku M and
Ikeda T: A case of glycogen-rich clear cell carcinoma of the breast
with extensive intraductal components and micrometastases to the
axillary lymph node. Gan To Kagaku Ryoho. 43:239–241.
2016.PubMed/NCBI(In Japanese).
|
|
13
|
Kuroda H, Sakamoto G, Ohnisi K and Itoyama
S: Clinical and pathological features of glycogen-rich clear cell
carcinoma of the breast. Breast Cancer. 12:189–195. 2005.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ratti V and Pagani O: Clear cell carcinoma
of the breast: A rare breast cancer subtype-case report and
literature review. Case Rep Oncol. 8:472–477. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Eun NL, Cha YJ, Son EJ, Gweon HM, Kim JA
and Youk JH: Clinical imaging of glycogen-rich clear cell carcinoma
of the breast: A case series with literature review. Magn Reson Med
Sci. 18:238–242. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Takekawa Y, Kubo A, Morita T, Kameda K,
Kimura M, Sakakibara M, Yoshii R and Yamashita Y: Histopathological
and immunohistochemical findings in a case of glycogen-rich clear
cell carcinoma of the breast. Rinsho Byori. 54:27–30.
2006.PubMed/NCBI
|
|
17
|
Pak I, Kutun S, Celik A, Alyanak A, Ardic
F and Cetin A: Glycogen-rich ‘clear cell’ carcinoma of the breast.
Breast J. 11(288)2005.
|
|
18
|
Trupiano JK, Ogrodowczyk E and Bergman S:
Pathologic quiz case: Mass in the right breast. Glycogen-rich clear
cell carcinoma of the breast. Arch Pathol Lab Med. 127:1629–1630.
2003.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Satoh F, Umemura S, Itoh H, Miyajima Y,
Tokuda Y, Tajima T and Osamura RY: Fine needle aspiration cytology
of glycogen-rich clear cell carcinoma of the breast: A case report.
Acta Cytol. 42:413–418. 1998.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Mizukami Y, Takayama T, Takemura A,
Ichikawa K, Onoguchi M and Taniya T: Glycogen-rich clear cell
carcinoma of the breast: A case report. J Med Ultrason. 36:39–43.
2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Markopoulos C, Mantas D, Philipidis T,
Kouskos E, Antonopoulou Z, Hatzinikolaou ML and Gogas H:
Glycogen-rich clear cell carcinoma of the breast. World J Surg
Oncol. 6(44)2008.
|
|
22
|
Gürbüz Y and Ozkara SK: Clear cell
carcinoma of the breast with solid papillary pattern: A case report
with immunohistochemical profile. J Clin Pathol. 56:552–554.
2003.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yerushalmi R, Hayes MM and Gelmon KA:
Breast carcinoma-Rare types: Review of the literature. Ann Oncol.
20:1763–1770. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Hull MT and Warfel KA: Glycogen-rich clear
cell carcinomas of the breast. A clinicopathologic and
ultrastructural study. Am J Surg Pathol. 10:553–559.
1986.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Badea M, Baroş A, Bohîlţea RE, Julea IE,
Furtunescu FL, Istrate-Ofiţeru AM, Iovan L, Cîrstoiu MM, Burcin MR,
Turcan N, et al: Modern interdisciplinary monitoring of cervical
cancer risk. Rom J Morphol Embryol. 60:469–478. 2019.PubMed/NCBI
|
|
26
|
Govender D, Sabaratnam RM and Essa AS:
Clear cell ‘sugar’ tumor of the breast: Another extrapulmonary site
and review of the literature. Am J Surg Pathol. 26:670–675.
2002.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Vranic S, Skenderi F, Beslagic V and
Gatalica Z: Glycogen-rich clear cell carcinoma of the breast: A
comprehensive review. Appl Immunohistochem Mol Morphol. 28:655–660.
2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Sinha KP and Bharati P: Glycogen rich
clear cell carcinoma of the breast: a rare case report. IOSR J
Dental Medical Sci. 15:85–87. 2016.e-ISSN: 2279-0853, p-ISSN:
2279-0861. Ver. VIII.
|
|
29
|
Ma X, Han Y, Fan Y, Cao X and Wang X:
Clinicopathologic characteristics and prognosis of glycogen-rich
clear cell carcinoma of the breast. Breast J. 20:166–173.
2014.PubMed/NCBI View Article : Google Scholar
|
















