Introduction
In December 2019, a cluster of pneumonia cases of an
unknown cause was reported in Wuhan, China. The pathogen causing
this condition was subsequently identified to be the novel
coronavirus, named severe acute respiratory syndrome coronavirus 2
(SARS-CoV-2), with the pneumonia it causes named as coronavirus
disease 2019 (COVID-19) (1).
COVID-19 is highly infectious, such that the number of patients has
increased sharply since its outbreak in 2019. COVID-19 has caused
mortality and morbidity on an enormous scale, directly affecting
societies across the world (2-8).
Although the entire general population is considered to be
susceptible to this disease, the elderly are among those who are
the most vulnerable to succumbing to COVID-19 (9-11).
Cross-infection in hospitals is one of the key routes of COVID-19
transmission (12), where elderly
patients subsequently become more susceptible to nosocomial
infection because of low immunity (13,14).
In February 2020, the Chinese Center for Disease Control and
Prevention reported that the fatality rate of confirmed COVID-19
cases was 2.3% in the general population, 3.6% in individuals aged
60-69 years, 8.0% in those aged 70-79 years and 14.8% in those aged
80 years (2). Mortality among
patients infected with COVID-19 occurred mainly in those aged
>80 years with underlying diseases, such as hypertension,
cardiovascular disease and diabetes (12,15).
In addition, the susceptibility of elderly patients to nosocomial
infection, especially to nosocomial pulmonary infection, is
particularly evident during the COVID-19 pandemic (11).
Nosocomial infections are also called
hospital-acquired infections and can be acquired during a patient's
stay at the hospital, where the symptoms are manifested either
during hospitalisation or after discharge. Nosocomial infections
are of particular concern, since they increase the disease burden
to add to the risk of high morbidity and mortality (16). According to a definition proposed
by the United States Center for Disease Control and Prevention in
1988, a nosocomial infection is an infection acquired during
hospital stay that did not exist or was at an incubation stage upon
admission (17). Clinically,
infections that occur 48 or 72 h after hospital admission are
typically referred to as hospital-acquired infections (18). Given the deterioration of physical
and immune functions and the development of primary diseases,
elderly patients are at high risk of nosocomial infection with poor
prognosis (18). Nosocomial
infections in elderly inpatients lead to prolonged hospital stays
and increased risk of more serious conditions, including sepsis,
organ dysfunction and multi-organ dysfunction (19). Specifically, nosocomial pneumonia
is the most common hospital-related infection that has high rates
of morbidity and mortality in addition to high levels of healthcare
resource consumption (20-24).
Nosocomial infections also pose serious challenge to
healthcare professionals globally during the COVID-19 pandemic.
Nosocomial infection of COVID-19 directly impacts the quality of
life of patients and also results in extra expenditure to hospitals
(12,25-27).
Considering the high risk of human-to-human transmission of
SARS-CoV-2 infection, the majority of hospitals, especially those
in China, have established a series of systems to control
nosocomial SARS-CoV-2 infection. During the COVID-19 pandemic, a
series of special measures were developed for elderly inpatients
seeking medical treatment and hospitalisation at the Geriatric
Department of The First Affiliated Hospital with Nanjing Medical
University. As a result, no hospitalised patients or medical staff
developed SARS-CoV-2 infection and the incidence of nosocomial
infections was significantly reduced. In the present study,
measures used to control nosocomial infections in elderly patients
during the COVID-19 pandemic were analysed. The objective was to
compare the incidence of nosocomial infection in the previous
routine medical setting with that following the implementation of
epidemic prevention and control measures. These results show that
epidemic prevention and control measures can improve the medical
environment, in addition to reducing the burden and harm caused by
nosocomial infection.
Patients and methods
Ethics
The present retrospective observational cohort study
analysed elderly patients hospitalised in the Geriatric Department
of The First Affiliated Hospital with Nanjing Medical University
(Nanjing, China). The institutional review board of The First
Affiliated Hospital with Nanjing Medical University approved this
study and granted a waiver of informed consent from study
participants because of the retrospective design. In the present
retrospective study, patient data were obtained through the
electronic medical record system.
Data collection
A total of 4,066 elderly patients admitted from
January to March in 2019 and 2020 who were hospitalised at The
First Affiliated Hospital with Nanjing Medical University for
>48 h were selected. The inclusion criteria were as follows: i)
Patients aged ≥65 years; and ii) hospital stay >48 h. The
exclusion criteria were as follows: i) Patients aged <65 years;
ii) hospital stay ≤48 h. Among them, 3,073 (75.58%) patients
hospitalised from January 2019 to March 2019 were designated into
the non-intervention group, whereas the remaining 933 (24.42%)
patients hospitalised from January 2020 to March 2020 after the
COVID-19 outbreak were designated into the intervention group. To
rule out any changes caused by the community floating population
and the social environment during this 2-year study, 93 long-stay
patients in stable condition were selected as a subgroup based on
the 4,066 patients for longitudinal analysis. The community
floating population refers to patients who have been hospitalised
in the Geriatric Department of The First Affiliated Hospital with
Nanjing Medical University for <2 years. The age-adjusted
Charlson Comorbidity Index (aCCI) is a more widely used comorbidity
scoring system than CCI, which quantifies comorbidities based on
the types and severity of a patients' diseases and can be used to
predict the risk of death from a disease. The higher the aCCI
score, the higher the risk of mortality. By contrast, the lower the
aCCI score, the lower the risk (28-30).
Long-stay patients refer to those who have been
hospitalised in the Geriatric Department of The First Affiliated
Hospital with Nanjing Medical University for ≥2 years. Because this
group of patients include those with multiple chronic diseases,
homes or nursing homes cannot stabilise their physical condition
and they therefore require long-term hospitalisation. The patients
in stable conditions do not include those in acute phases of the
disease or those that suffer from malignancies. Patients aged
>65 years were included into the geriatrics program. The
incidence of nosocomial infections during the epidemic prevention
and control period (24 January 2020 to 24 March 2020) and the
previous period of hospitalisation (24 January 2019 to 24 March
2019) was analysed. Of these 93 long-stay patients, 93 were all
enrolled according to the inclusion criteria. The inclusion
criteria were as follows: i) Patients aged ≥65 years; ii) hospital
stay ≥2 years; and iii) patients in a stable condition. The
exclusion criteria were as follows: i) Patients aged <65 years;
ii) hospital stay <2 years; iii) patients in the acute phase of
a disease; iv) patients with malignant tumours who were undergoing
chemotherapy, radiotherapy, immunotherapy; or v) patients that had
a terminal disease. The objective was to compare the incidence of
nosocomial infection in the previous routine medical setting with
that following the implementation of the epidemic prevention and
control measures.
Interventions
On 24 January 2020, The First Affiliated Hospital
with Nanjing Medical University officially launched its first-level
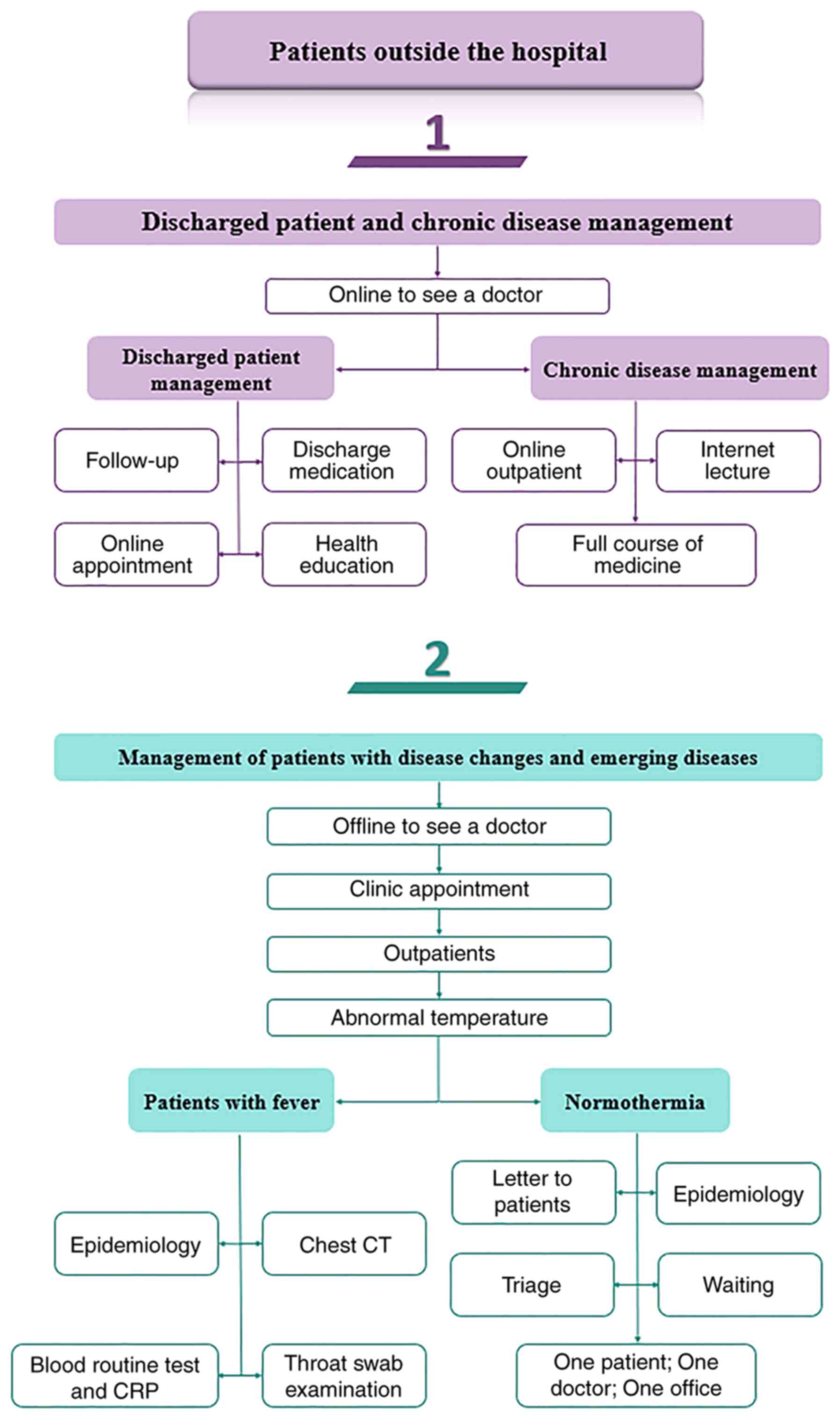
response to the COVID-19 pandemic (31). Elderly patients in the geriatric
department were mainly divided into the following groups:
Inpatients; outpatients; discharged patients; and patients with
chronic disease. These patients were followed up for personalised
management according to their conditions.
During hospital visits, all patients were required
to wear masks, following which their body temperatures were checked
and epidemiological history was recorded. If any of the patients
showed symptoms of fever and pneumonia, they were transferred to a
special unit, namely the ‘fever unit’, which was designed to
observe and isolate patients suspected of infection. For patients
with suspected symptoms, novel coronavirus nucleic acid tests on
oropharyngeal swabs, chest CT and blood tests, including routine
blood test and C-reactive protein test, were performed in a
negative-pressure isolation room (32,33).
Novel coronavirus nucleic acid test was performed using reverse
transcription-quantitative PCR (RT-qPCR). The clinical samples of
patients with PCR-confirmed COVID-19 were obtained in the form of
throat swabs. Different conserved SARS-CoV-2 gene sequences can be
targeted for RT-qPCR detection. Proposals from previous reports
suggest using RT-qPCR as the molecular assay for detecting
SARS-CoV-2 (4,34). Throughout all processes, all
medical staff must wear masks, isolation suits and wash their
hands. A professional online service for assessment, diagnosis and
treatment was also set up for the management of patients with
chronic disease and follow-up of discharged patients to avoid
unnecessary hospital visits, reduce the risk of nosocomial
infections and burden of hospital outpatient service to facilitate
the reasonable allocation of medical resources. A nosocomial
infection is an infection acquired during hospital stay that did
not previously occur or was at the incubation stage upon admission
(17).
For pre-diagnosis, an outpatient waiting area was
set up according to the 1-m social distance rule to avoid
cross-infection in the hospital. Patients with mild illness were
treated in a separate room both during the early outbreak (2019)
and under the current management protocols (2020). In the
outpatient geriatric department, due to the limited ability of
self-care and communication difficulties displayed by the elderly
patients, only one individual was allowed to accompany the patients
into the consultation room. Doctors, patients and accompanying
staff were all required to wear masks at all times.
For outpatients with chronic diseases, the
prescription dosage was extended to 3 months depending on the
condition. In situations where the drug was urgently required by
the patients but could not be procured, solutions included
temporary procurement, logistics and delivery, door-to-door
delivery and other professional online services. In addition, to
guarantee the pharmacological needs of the patients during the
epidemic, patients who are in urgent need of drugs (anticoagulants,
insulin, anti-rejection drugs for organ transplantation and
emergency drugs for sudden diseases) can contact the nearest drug
handling enterprises in their residential areas for consultation
and purchase drugs in cases of drug shortages in hospitals.
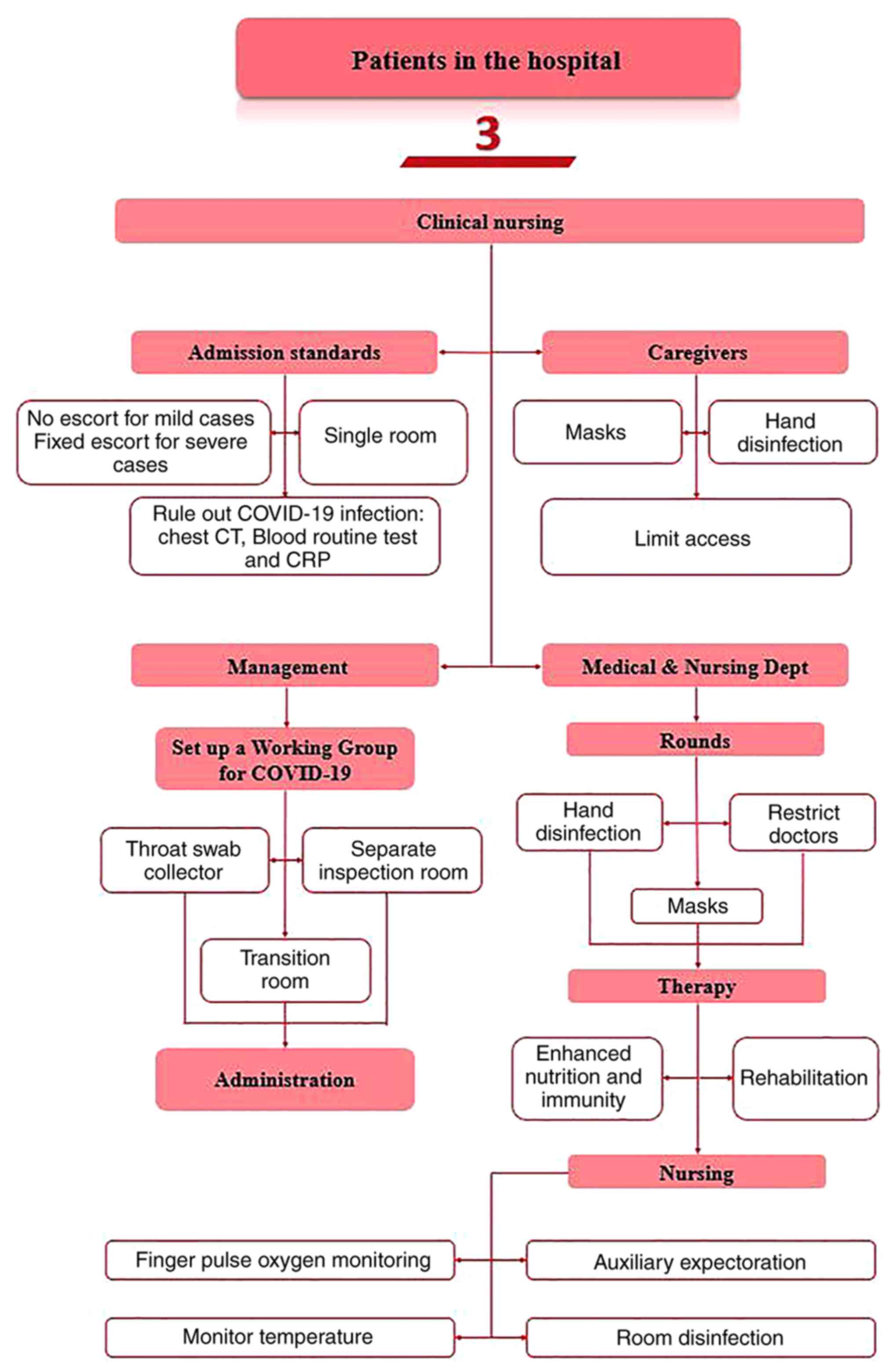
Preventive management
The First Affiliated Hospital with Nanjing Medical
University has successfully formulated seven editions of
hospitalisation standards for inpatients, of which the seventh
version is quoted here (35). At
the beginning of the outbreak, patients who had to be admitted to
hospital due to their condition required examination before
admission. Chest CT, C-reactive protein estimation, blood routine
test and novel coronavirus nucleic acid tests were performed before
hospitalisation. Patients without epidemiological exposure to
COVID-19 cases were admitted to the hospital within 14 days of
examination. At the epidemic control stage, the hospital ward was
arranged into single rooms, where temporary isolation wards were
allocated for nucleic acid testing, chest CT and blood sample
analysis before the patients were admitted. If the results of
nucleic acid detection, chest CT and blood sample analysis, along
with the epidemiological history assessment, were negative,
hospital-acquired COVID-19 infection would be ruled out. It was
necessary to conduct nucleic acid detection tests twice for
high-risk patients, such as those with a recent history of fever
and characteristic signs of COVID-19 according to CT images.
Multiple patchy ‘ground glass’ opacities in the bilateral multiple
lobular regions with periphery distribution are typical chest CT
features of COVID-19 pneumonia, where interlobular thickening and
adjacent pleura can also occur (36). If the nucleic acid test results
were positive, the patient would then be immediately isolated and
sent to the negative pressure isolation room through a special
channel. In addition, any medical staff members exposed to a
patient suspected of COVID-19 were isolated under observation for
14 days.
The principle of ‘no companion for mild cases’ and
‘one companion for severe cases’ was implemented in the geriatrics
ward. The accompanying personnel was required to undergo a nucleic
acid test before entering the ward. In addition, patients and
caregivers were not allowed to enter or leave the ward
unauthorised.
During the hospitalisation period, family members
were not allowed to visit the patients. Therefore, the elderly
inpatients were encouraged to communicate with their families
through ‘cloud visitation’. If the elderly inpatient did not know
how to use the Internet for video chat, the nurses would provide
assistance.
The COVID-19 outbreak has affected not only the
lifestyle of elderly patients but also their mental health. Elderly
patients, especially inpatients, tend to be more prone to
depression, anxiety and insomnia (37,38).
In particular, 10-15% elderly patients suffer from clinical
depression, which requires serious expert intervention (37,38).
Behaviour driven by negative emotions not only aggravate the
disease course but can also adversely affect ward management
(39). Therefore, psychological
protection for elderly patients forms an important part of the
clinical treatment strategy (40,41).
To address this, a ‘mind comfort room’ managed by clinical
psychologists and medical staff was set up at the hospital. This
‘mind comfort room’ is a spacious apartment for elderly inpatients,
where they can read magazines, watch movies, listen to music and
play chess. In addition, sufficient space was provided for elderly
inpatients to perform exercises, such as Tai chi and aerobic
gymnastics.
Publicity and education on epidemic prevention were
provided for the discharged patients. Follow-up telephone calls
were made and online lectures on diseases were conducted regularly.
Management procedures for both outpatients and inpatients are shown
in Figs. 1 and 2, respectively.
Statistical analyses
All categorical data were described as frequency (N,
%) and calculated with the Wilson score using the 95% confidence
interval (CI). Comparisons were performed using the non-parametric
Mann-Whitney or Kruskal-Wallis tests for two- and
multiple-independent samples, respectively. Univariate and
multivariate logistic regression models were used to compare
clinical outcomes between the groups, where the association between
the outcome and each variable/research factor, including age, sex,
nervous system, respiratory system, endocrine system, digestive
system, cardiovascular system, urinary system, ELSE, malignant
tumor, trauma and nosocomial infection, were first analysed.
Although these factors can be used as independent risk factors,
there may be correlations among different factors, where part of
this correlation among these factors can be masked within the
univariate analysis. As a result, multivariate logistic regression
was then used to detect the interactions of the correlation among
these factors with the results.
Propensity score matching analysis was performed to
mitigate the effect of selection bias and potential confounding
factors between two groups. Propensity scores were calculated using
all variables except for interventions, namely age, sex and
underlying disease. For propensity score matching, a
nearest-neighbour 1:2 matching scheme (42) with a calliper size of 0.1 was used.
Inverse probability treatment weighting (IPTW) (43,44)
and standardised mortality ratio weight (SMRW) (45-47)
were calculated based on the logistic regression model and used to
evaluate the risk of nosocomial infections. Results were stratified
for age, which is known to be associated with variations in the
incidence of nosocomial infections (11,13).
By contrast, the generalised estimating equation (GEE) model
(48) with a negative binomial
distribution was generated using an unstructured working
correlation matrix to investigate the risk of nosocomial
infections. This utilises a related structure describing the
different measured outcomes without any assumptions being made
about its structure:
In this case, Yik represents the
kth response of the ith patient, whereas
ajk represents the correlation coefficient between the
jth response and the kth response. In
particular, Yij represents the outcome of the
jth hospitalisation of the ith patient,
whilst Yik represents the outcome of the kth
hospitalisation of the ith patient. Infection is the
specific outcome.
Specifically, model 0 represents the unadjusted base
model, whereas model I, represents that adjusted for sex and age at
baseline. Model II represents model I that was adjusted further for
chronicity disease, malignant tumor and CCI. The variables included
in the GEE model are similar to those included in logistic
regression. Since the outcome variables were derived from a
patient's multiple hospitalisations, the GEE models took into
account the clustering of multiple hospitalisations of the same
patient to minimise bias when producing estimates (49). Nomogram construction and validation
were performed in accordance with a previously reported guideline
(50). A nomogram was constructed
according to the independent prognostic factors of survival. Based
on the logistic, dichotomous and sequential screening analyses, a
nomogram incorporating the risk factors was created for predicting
infection outcomes using the EmpowerStats statistical software 2.0
(http://www.empowerstats.net/), which was
calculated using the R packages ‘Survival’ and ‘Rms’ (R version
3.4.3) (51-53).
Comparisons among categorical variables were
analysed by the χ2 test. All statistical tests were performed using
SPSS 22.0 software (IBM Corp.) and the EmpowerStats statistical
software 2.0 (www.empowerstats.com; X&Y Solutions Inc.).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Baseline characteristics of the
participants
Among the 4,066 patients, >50% were elderly aged
>70 years, where 66.3% were male. The three dominant chronic
diseases in the cohort were disorders in the nervous system
(n=1,080, 26.56%), digestive system (n=941, 23.14%) and
cardiovascular system (n=466, 11.46%). Parameters (age, sex,
nervous system, respiratory system, endocrine system, digestive
system, cardiovascular system, malignant tumor, trauma, and
nosocomial infection) between the intervention and non-intervention
groups were different except for urinary system (Table I). Specifically, there was a higher
proportion of patients in the nervous system, respiratory system,
digestive system, cardiovascular system, trauma and nosocomial
infection categories in the unintervened group, whilst a higher
proportion of patients was observed in the endocrine system and
malignant tumor categories in the intervened group. The proportion
of patients aged <70 years old was higher, whereas that of
patients aged ≥70 years was lower, in the unintervened group
compared with that in the intervened group. There was difference in
the intervention and non-intervention groups at the sex ratios of
the patients recruited in study setting with unweighted or
propensity 1:2 matching, where patients included in the population
analysis predominantly male. After adjusting for all covariates by
Propensity score matching (PSM) analysis, a similar distribution in
the different covariates was observed except for age, nervous
system, digestive system, urinary system and malignant tumor. After
the IPTW and SMRW adjustment, the association between intervening
measures and nosocomial infection remained significant. However,
none of the other parameters had their significance preserved,
although a difference was observed in age distribution after SMRW,
which is similar to those after unadjusted analysis (Table I). Comprehensively, the nosocomial
infection rate between the intervention and non-intervention groups
was found to be significantly different regardless of the
calculation methods used, where there was a lower proportion in the
intervened group (non-intervention vs. intervention: Unweighted,
4.72 vs. 1.91%, P<0.001; propensity 1:2 matching, 4.94 vs.
1.73%, P<0.001; IPTW, 4.79 vs. 1.7%, P<0.001; SMRW, 5 vs.
1.91%, P<0.001).
 | Table IBaseline characteristics of the
elderly patients in the present study.a |
Table I
Baseline characteristics of the
elderly patients in the present study.a
| A, Unweighted
sample (n=4,066) |
|---|
|
Characteristics | Total [n=4,066; n
(%)] | Unintervened
[n=3,073; n (%)] | Intervened [n=993;
n (%)] | P-value |
|---|
| Age, years | | | | <0.001 |
|
<70 | 1,987 (48.87) | 1,567 (50.99) | 420 (42.30) | |
|
70-90 | 1,404 (34.53) | 1,040 (33.84) | 364 (36.66) | |
|
≥90 | 675 (16.60) | 466 (15.16) | 209 (21.05) | |
| Sex | | | | 0.005 |
|
Male | 2,696 (66.31) | 2,001 (65.12) | 695 (69.99) | |
|
Female | 1,370 (33.69) | 1,072 (34.88) | 298 (30.01) | |
| Nervous system | 1,080 (26.56) | 857 (27.89) | 223 (22.46) | <0.001 |
| Respiratory
system | 329 (8.09) | 280 (9.11) | 49 (4.93) | <0.001 |
| Endocrine
system | 393 (9.67) | 224 (7.29) | 169 (17.02) | <0.001 |
| Digestive
system | 941 (23.14) | 766 (24.93) | 175 (17.62) | <0.001 |
| Cardiovascular
system | 466 (11.46) | 388 (12.63) | 78 (7.85) | <0.001 |
| Urinary system | 141 (3.47) | 107 (3.48) | 34 (3.42) | 0.931 |
| ELSE | 59 (1.45) | 51 (1.66) | 8 (0.81) | 0.05 |
| Malignant
tumor | 597 (14.68) | 376 (12.24) | 221 (22.26) | <0.001 |
| Trauma | 47 (1.16) | 42 (1.37) | 5 (0.50) | 0.027 |
| Nosocomial
infection | 164 (4.03) | 145 (4.72) | 19 (1.91) | <0.001 |
| B, Propensity 1:2
matching (n=2,466) |
|
Characteristics | Total [n=2,466; n
(%)] | Unintervened
[n=1,598; n (%)] | Intervened [n=868;
n (%)] | P-value |
| Age, years | | | | 0.793 |
|
<70 | 1,094 (44.36) | 702 (43.93) | 392 (45.16) | |
|
70-90 | 899 (36.46) | 590 (36.92) | 309 (35.60) | |
|
≥90 | 473 (19.18) | 306 (19.15) | 167 (19.24) | |
| Sex | | | | 0.047 |
|
Male | 1,656 (67.15) | 1,051 (65.77) | 605 (69.70) | |
|
Female | 810 (32.85) | 547 (34.23) | 263 (30.30) | |
| Nervous system | 471 (19.10) | 306 (19.15) | 165 (19.01) | 0.933 |
| Respiratory
system | 181 (7.34) | 136 (8.51) | 45 (5.18) | 0.002 |
| Endocrine
system | 274 (11.11) | 128 (8.01) | 146 (16.82) | <0.001 |
| Digestive
system | 492 (19.95) | 322 (20.15) | 170 (19.59) | 0.737 |
| Cardiovascular
system | 284 (11.52) | 217 (13.58) | 67 (7.72) | <0.001 |
| Urinary system | 127 (5.15) | 95 (5.94) | 32 (3.69) | 0.015 |
| ELSE | 57 (2.31) | 26 (1.63) | 31 (3.57) | 0.002 |
| Malignant
tumor | 548 (22.22) | 341 (21.34) | 207 (23.85) | 0.152 |
| Trauma | 32 (1.30) | 27 (1.69) | 5 (0.58) | 0.02 |
| Nosocomial
infection | 94 (3.81) | 79 (4.94) | 15 (1.73) | <0.001 |
| C, IPTW
(n=4,066) |
|
Characteristics | Total [n=4,066; n
(%)] | Unintervened
[n=3,073; n (%)] | Intervened [n=993;
n (%)] | P-value |
| Age, years | | | | 0.058 |
|
<70 | 1,987 (48.87) | 1,510 (49.15) | 477 (48.04) | |
|
70-90 | 1,415 (34.80) | 1,082 (35.2) | 333 (33.53) | |
|
≥90 | 664 (16.33) | 481 (15.65) | 183 (18.43) | |
| Sex | | | | 0.947 |
|
Male | 2,694 (66.26) | 2,037 (66.28) | 657 (66.18) | |
|
Female | 1,372 (33.74) | 1,036 (33.72) | 336 (33.82) | |
| Nervous system | 1,069 (26.29) | 814 (26.49) | 255 (25.72) | 0.578 |
| Respiratory
system | 336 (8.26) | 249 (8.11) | 87 (8.8) | 0.429 |
| Endocrine
system | 387 (9.52) | 293 (9.54) | 94 (9.44) | 0.909 |
| Digestive
system | 943 (23.19) | 711 (23.14) | 232 (23.34) | 0.875 |
| Cardiovascular
system | 459 (11.29) | 351 (11.43) | 108 (10.83) | 0.538 |
| Urinary system | 140 (3.44) | 106 (3.46) | 34 (3.41) | 0.933 |
| ELSE | 58 (1.43) | 44 (1.44) | 14 (1.37) | 0.848 |
| Malignant
tumor | 609 (14.98) | 457 (14.86) | 152 (15.34) | 0.669 |
| Trauma | 49 (1.21) | 36 (1.16) | 13 (1.33) | 0.624 |
| Nosocomial
Infection | 164 (4.03) | 147 (4.79) | 17 (1.7) | <0.0001 |
| D, SMRW
(n=4066) |
|
Characteristics | Total [n=4,066; n
(%)] | Unintervened
[n=3,073; n (%)] | Intervened [n=993;
n (%)] | P-value |
| Age, years | | | | 0.006 |
|
<70 | 1,755 (43.16) | 1,335 (43.44) | 420 (42.3) | |
|
70-90 | 1,574 (38.71) | 1,210 (39.39) | 364 (36.66) | |
|
≥90 | 737 (18.13) | 528 (17.17) | 209 (21.04) | |
| Sex | | | | 0.943 |
|
Male | 2,843 (69.92) | 2,148 (69.89) | 695 (69.99) | |
|
Female | 1,223 (30.08) | 925 (30.11) | 298 (30.01) | |
| Nervous system | 904 (22.23) | 681 (22.17) | 223 (22.46) | 0.826 |
| Respiratory
system | 203 (4.99) | 154 (5.01) | 49 (4.93) | 0.917 |
| Endocrine
system | 676 (16.63) | 507 (16.51) | 169 (17.02) | 0.663 |
| Digestive
system | 716 (17.61) | 541 (17.6) | 175 (17.62) | 0.985 |
| Cardiovascular
system | 316 (7.77) | 238 (7.75) | 78 (7.85) | 0.899 |
| Urinary system | 138 (3.39) | 104 (3.4) | 34 (3.42) | 0.968 |
| ELSE | 32 (0.79) | 24 (0.78) | 8 (0.81) | 0.927 |
| Malignant
tumor | 927 (22.80) | 706 (22.97) | 221 (22.26) | 0.587 |
| Trauma | 21 (0.52) | 16 (0.51) | 5 (0.50) | 0.972 |
| Nosocomial
Infection | 173 (4.25) | 154(5) | 19 (1.91) | <0.0001 |
Multivariate logistic regression and
stratified analysis of interventions
In the multivariate logistic regression model, the
risk of nosocomial infections was lower in the intervention group
compared with that in the non-intervention group [odds ratio
(OR)=0.36; 95% CI=0.22-0.59 and P<0.001; Table II]. Even after IPTW and SMRW
adjustment, the differences remained significant (IPTW: OR=0.33;
95% CI=0.25-0.44 and P<0.001; SMRW: OR=0.35; 95% CI=0.21-0.61;
P=0.002; Table II). Because it
was found in previous clinical studies that nosocomial infection in
the elderly is more common, age was considered to be a risk factor
of nosocomial infection (11,13),
it was necessary to stratify the analysis by age (aged 70-90 years:
Unadjusted, OR=2.46; 95% CI=1.64-3.68 and P<0.001; IPTW,
OR=2.35, 95% CI=1.71-3.24 and P<0.001; SMRW, OR=2.35, 95%
CI=1.23-4.49 and P=0.0098; aged ≥90 years: Unadjusted, OR=3.39, 95%
CI=2.14-5.39 and P<0.001; IPTW, OR=3.47, 95% CI=2.42-4.98 and
P<0.001; SMRW, OR=3.34; 95% CI=1.61-6.96; P=0.0012; Table II). In addition, a stratified
analysis of gastric catheterisation was performed to exclude this
confounding factor. After the age stratification and adjustment for
sex, chronic disease, malignant tumour and trauma, the results of
both IPTW and SMRW showed a lower risk of nosocomial infection in
the intervention group compared with that in the non-intervention
group (aged <70 years: IPTW, OR=0.39; 95% CI=0.23-0.68 and
P=0.0008; SMRW, OR=0.47; 95% CI=0.16-1.44 and P=0.1887; aged 70-90
years: IPTW, OR=0.28, 95% CI=0.18-0.44; P<0.001; SMRW, OR=0.34;
95% CI=0.15-0.76 and P=0.0091; age ≥90 years: IPTW, OR=0.28; 95%
CI=0.17-0.46 and P<0.001; SMRW, OR=0.26; 95% CI=0.10-0.68 and
P=0.0064; Table III).
 | Table IIMultivariate logistic regression
analyses for nosocomial infection in IPTW- and SMRW-matched
cohort.a |
Table II
Multivariate logistic regression
analyses for nosocomial infection in IPTW- and SMRW-matched
cohort.a
| A, Unadjusted |
|---|
| Parameter | OR (95% CI) | P-value |
|---|
| Intervention (yes
vs. no) | 0.36
(0.22-0.59) | <0.0001 |
| Sex (female vs.
male) | 0.96
(0.68-1.34) | 0.7951 |
| Age (vs.
<70) | | |
|
≥70,
<90 | 2.46
(1.64-3.68) | <0.001 |
|
≥90 | 3.39
(2.14-5.39) | <0.001 |
| Nervous system (yes
vs. no) | 2.66
(0.36-19.71) | 0.339 |
| Respiratory system
(yes vs. no) | 1.34
(0.17-10.58) | 0.783 |
| Endocrine system
(yes vs. no) | 1.22
(0.15-9.84) | 0.849 |
| Digestive system
(yes vs. no) | 0.94
(0.12-7.27) | 0.95 |
| Cardiovascular
system (yes vs. no) | 1.47
(0.19-11.38) | 0.71 |
| Urinary system (yes
vs. no) | 2.40
(0.29-19.70) | 0.415 |
| ELSE (yes vs.
no) | 1.18
(0.41-3.37) | 0.756 |
| Malignant tumor
(yes vs. no) | 1.98
(0.26-15.08) | 0.508 |
| Trauma (yes vs.
no) | 2.50
(0.25-25.26) | 0.437 |
| B, Adjusted with
IPTW |
| Parameter | OR (95% CI) | P-value |
| Intervention (yes
vs. no) | 0.33
(0.25-0.44) | <0.0001 |
| Sex (female vs.
male) | 0.89
(0.68-1.17) | 0.4009 |
| Age (vs.
<70) | | |
|
≥70,
<90 | 2.35
(1.71-3.24) | <0.001 |
|
≥90 | 3.47
(2.42-4.98) | <0.001 |
| Nervous system (yes
vs. no) | 3.22
(0.66-15.84) | 0.15 |
| Respiratory system
(yes vs. no) | 1.39
(0.26-7.28) | 0.7 |
| Endocrine system
(yes vs. no) | 1.66
(0.32-8.61) | 0.545 |
| Digestive system
(yes vs. no) | 1.39
(0.27-7.05) | 0.692 |
| Cardiovascular
system (yes vs. no) | 2.57
(0.51-12.91) | 0.253 |
| Urinary system (yes
vs. no) | 3.56
(0.67-18.77) | 0.135 |
| ELSE (yes vs.
no) | 0.95
(0.36-2.48) | 0.914 |
| Malignant tumor
(yes vs. no) | 2.94
(0.59-14.64) | 0.189 |
| Trauma (yes vs.
no) | 2.72
(0.40-18.60) | 0.307 |
| C, Adjusted with
SMRW |
| Parameter | OR (95% CI) | P-value |
| Intervention (yes
vs. no) | 0.35
(0.21-0.61) | <0.0001 |
| Sex (female vs.
male) | 0.81
(0.46-1.42) | 0.463 |
| Age (vs.
<70) | | |
|
≥70,
<90 | 2.35
(1.23-4.49) | 0.01 |
|
≥90 | 3.34
(1.61-6.96) | <0.001 |
| Nervous system (yes
vs. no) | 2.89
(0.38-22.21) | 0.308 |
| Respiratory system
(yes vs. no) | 1.03
(0.09-11.63) | 0.983 |
| Endocrine system
(yes vs. no) | 1.55
(0.19-12.66) | 0.684 |
| Digestive system
(yes vs. no) | 1.36
(0.16-11.70) | 0.778 |
| Cardiovascular
system (yes vs. no) | 2.51
(0.30-21.22) | 0.398 |
| Urinary system (yes
vs. no) | 3.08
(0.32-29.14) | 0.327 |
| ELSE (yes vs.
no) | 0.83
(0.05-12.82) | 0.895 |
| Malignant tumor
(yes vs. no) | 2.55
(0.33-19.63) | 0.37 |
| Trauma (yes vs.
no) | 2.38
(0.05-112.79) | 0.659 |
 | Table IIIEffect of intervening measures in
IPTW-adjusted and SMRW-adjusted logistic regression analyses for
the risk of nosocomial infection, stratified according to the
age.a |
Table III
Effect of intervening measures in
IPTW-adjusted and SMRW-adjusted logistic regression analyses for
the risk of nosocomial infection, stratified according to the
age.a
| | Intervened | Unadjusted | Adjusted with
IPTWb | Adjusted with
SMRWb |
|---|
| Age group
(years) | No (n, %) | Yes (n, %) | OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value |
|---|
| <70 | 1,567 (50.99) | 420 (42.30) | 0.48
(0.18-1.27) | 0.141 | 0.39
(0.23-0.68) | <0.001 | 0.47
(0.16-1.44) | 0.189 |
| ≥70, <90 | 1,040 (33.84) | 364 (36.66) | 0.33
(0.15-0.70) | 0.004 | 0.28
(0.18-0.44) | <0.001 | 0.34
(0.15-0.76) | 0.009 |
| ≥90 | 466 (15.16) | 209 (21.05) | 0.27
(0.11-0.66) | 0.004 | 0.28
(0.17-0.46) | <0.001 | 0.26
(0.10-0.68) | 0.006 |
| Total | 3,039 (75.58) | 993 (24.42) | 0.36
(0.22-0.59) | <0.001 | 0.33
(0.25-0.44) | <0.001 | 0.35
(0.21-0.61) | <0.001 |
Baseline characteristics of the
long-stay subgroup population
To rule out any changes owing to the community
floating population and social environment during the present
1-year study, 93 long-stay patients in stable conditions were
selected as the study population for longitudinal analysis.
Charlson comorbidity index (CCI) was included as a new variable,
which is commonly used to assess the impact of co-comorbidities on
patient survival over 10 years in addition to the underlying
disease for which the patient is currently being treated (28-30).
The age-adjusted CCI (aCCI) is a more widely used Comorbidity
scoring system than CCI (28). It
quantifies comorbidities based on the patients' age, types and
severity of a patient's diseases and can be used to predict the
risk of mortality from a disease (28-30).
By contrast, the generalised estimating equation (GEE) model with a
negative binomial distribution and an unstructured working
correlation matrix were used to investigate the longitudinal
relationship between interventions and the risk of nosocomial
infections. The working correlation matrix constitute the
covariance of linear predictor of the generalised linear model
which reflect the clustering of the multiple hospitalisations of
the same patient. The unstructured working correlation matrix means
that no relation assumption of the element in the matrix is
assigned, so each element of the correlation matrix should be
estimated (54). The baseline
characteristics of the 93 patients are listed in Table IV. In total, >90% of the
patients were elderly aged >70 years, where 68.82% were male.
Nervous system disease was the most common chronic malady in this
population (75.27%). aCCI was used to score comorbidities, where it
was found that 67.74% of the patients had higher scores (≥6). Among
these inpatients, the risk of nosocomial infections was lower in
the intervention group compared with that in the non-intervention
group (22.58 vs. 37.63%, respectively; P=0.025), especially the
risk of pulmonary infection (13.98 vs. 41.94%, respectively,
P<0.001; Table IV).
 | Table IVBaseline characteristics for the
longitudinal study of long-stay patients.a |
Table IV
Baseline characteristics for the
longitudinal study of long-stay patients.a
|
Characteristics | Total (n=186), n
(%) | Unintervened
(n=93), n (%) | Intervened (n=93),
n (%) | P-value |
|---|
| Age, years | | | | - |
|
<70 | 18 (9.68) | 9 (9.68) | 9 (9.68) | |
|
≥70,
<90 | 76 (40.86) | 38 (40.86) | 38 (40.86) | |
|
≥90 | 92 (49.46) | 46 (49.46) | 46 (49.46) | |
| Sex | | | | - |
|
Male | 128 (68.82) | 64 (68.82) | 64 (68.82) | |
|
Female | 58 (31.18) | 29 (31.18) | 29 (31.18) | |
| Nervous system | 140 (75.27) | 69 (74.19) | 71 (76.34) | 0.734 |
| Respiratory
system | 24 (12.9) | 14 (15.05) | 10 (10.75) | 0.382 |
| Endocrine
system | 72 (38.71) | 41 (44.09) | 31 (33.33) | 0.132 |
| Cardiovascular
system | 155 (83.33) | 78 (83.87) | 77 (82.80) | 0.844 |
| Malignant
tumor | 9 (4.84) | 3 (3.23) | 6 (6.45) | 0.305 |
| aCCI | | | | 0.754 |
|
<6 | 60 (32.26) | 31 (33.33) | 29 (31.18) | |
|
≥6 | 126 (67.74) | 62 (66.67) | 64 (68.82) | |
| Gastric
catheterisation | 66 (35.48) | 35 (37.63) | 31 (33.33) | 0.54 |
| Nosocomial
infection | 56 (30.11) | 35 (37.63) | 21 (22.58) | 0.025 |
| Pulmonary
infection | 52 (27.96) | 39 (41.94) | 13 (13.98) | <0.001 |
| Other
infections | 16 (8.60) | 11 (11.83) | 5 (5.38) | 0.117 |
GEE estimation of the risk of
nosocomial infection associated with the interventions
The rates of nosocomial and pulmonary infections
were lower in the intervention group compared with those in the
non-intervention group based on the univariate analysis (nosocomial
infection with GEE adjustment: OR=0.5165, 95% CI=0.2763-0.965 and
P=0.0384; pulmonary infection with GEE adjustment: OR=0.2186, 95%
CI=0.1129-0.4231 and P<0.0001; Table V). In addition, a multivariate
logistics regression model was constructed to evaluate the
influence of the intervention on the risk of nosocomial and
pulmonary infections. Compared with that in the non-intervention
group, the intervention group had a lower risk of nosocomial and
pulmonary infections (nosocomial infection with GEE adjustment:
OR=0.50, 95% CI=0.27-0.90 and P=0.0217; pulmonary infection with
GEE adjustment: OR=0.23, 95% CI=0.12-0.42 and P<0.0001)
(Table VI). Furthermore, in both
the univariate analysis and multiple logistic regression models,
gastric catheterisation was found to be a high-risk factor for
infection (nosocomial infection with GEE adjustment: OR=2.33, 95%
CI=1.12-4.83 and P=0.0229; pulmonary infection with GEE adjustment:
OR=3.59, 95% CI=1.69-7.61; P=0.0009; Tables V and VI). Therefore, a stratified analysis for
gastric catheterisation was adopted. Among the inpatients without
gastric catheterisation, the risk of nosocomial and pulmonary
infections was significantly lower in the intervention group
compared with that in the non-intervention group after adjusting
with GEE in the multiple logistic regression model (nosocomial
infection: OR=0.28, 95% CI=0.11-0.61 and P=0.001; pulmonary
infection: OR=0.10, 95% CI=0.03-0.32; P<0.001; Table VII).
 | Table VUnivariate analysis of risk factors
for infection based on the GEE model. |
Table V
Univariate analysis of risk factors
for infection based on the GEE model.
| A, Nosocomial
infection |
|---|
| | Unadjusted | Adjusted With
GEE |
|---|
| Parameters | OR (95% CI) | P-value | OR (95% CI) | P-value |
|---|
| Intervention (yes
vs. no) | 0.48
(0.25-0.92) | 0.027 | 0.5165
(0.2763-0.9653) | 0.038 |
| Sex (female vs.
male) | 1.07
(0.54-2.09) | 0.853 | 1.2652
(0.5568-2.8747) | 0.574 |
| Age (vs.
<70) | | | | |
|
≥70,
<90 | 2.04
(0.54-7.74) | 0.296 | 1.1845
(0.2574-5.451) | 0.828 |
|
≥90 | 2.54
(0.68-9.44) | 0.164 | 1.0932
(0.2251-5.3079) | 0.912 |
| Nervous system (yes
vs. no) | 2.48
(1.07-5.73) | 0.034 | 1.6831
(0.6924-4.0913) | 0.251 |
| Respiratory system
(yes vs. no) | 1.47
(0.60-3.59) | 0.4 | 1.3962
(0.5328-3.6584) | 0.497 |
| Endocrine system
(yes vs. no) | 1.42
(0.75-2.69) | 0.277 | 1.3131
(0.6176-2.7919) | 0.479 |
| Cardiovascular
system (yes vs. no) | 2.55
(0.92-7.03) | 0.07 | 2.1568
(0.8579-5.4225) | 0.102 |
| aCCI (≥6 vs.
<6) | 1.45
(0.72-2.89) | 0.296 | 1.0225
(0.9371-1.1157) | 0.617 |
| Gastric
catheterisation (yes vs. no) | 2.42
(1.27-4.62) | 0.007 | 2.3313
(1.1242-4.8348) | 0.023 |
| B, Pulmonary
infection |
| | Unadjusted | Adjusted With
GEE |
| Parameters | OR (95% CI) | P-value | OR (95% CI) | P-value |
| Intervention (yes
vs. no) | 0.23
(0.11-0.46) | <0.001 | 0.2186
(0.1129-0.4231) | <0.001 |
| Sex (female vs.
male) | 0.97
(0.49-1.95) | 0.94 | 1.271
(0.5084-3.1775) | 0.608 |
| Age (vs.
<70) | | | | |
|
≥70,
<90 | 2.86
(0.60-13.54) | 0.186 | 2.3157
(0.5904-9.0833) | 0.229 |
|
≥90 | 3.87
(0.84-17.93) | 0.084 | 2.1334
(0.5572-8.1693) | 0.269 |
| Nervous system (yes
vs. no) | 2.64
(1.10-6.36) | 0.031 | 1.8451
(0.7142-4.7665) | 0.206 |
| Respiratory system
(yes vs. no) | 1.66
(0.68-4.07) | 0.268 | 1.6081
(0.4203-6.1518) | 0.488 |
| Endocrine system
(yes vs. no) | 1.71
(0.90-3.28) | 0.104 | 1.7201
(0.7416-3.9897) | 0.206 |
| Cardiovascular
system (yes vs. no) | 1.76
(0.68-4.57) | 0.247 | 1.3119
(0.4318-3.9863) | 0.632 |
| Malignant tumor
(yes vs. no) | N/A | N/A | N/A | N/A |
| aCCI (≥6 vs.
<6) | 1.62
(0.79-3.33) | 0.189 | 1.0359
(0.9442-1.1364) | 0.456 |
| Gastric
catheterisation (yes vs. no) | 3.31
(1.70-6.43) | <0.001 | 3.5871
(1.6915-7.607) | <0.001 |
| C, Other
infections |
| | Unadjusted | Adjusted With
GEE |
| Parameters | OR (95% CI) | P-value | OR (95% CI) | P-value |
| Intervention (yes
vs. no) | 0.42
(0.14-1.27) | 0.126 | 0.4421
(0.1489-1.3124) | 0.142 |
| Sex (female vs.
male) | 3.17
(1.12-9.00) | 0.03 | 3.0069
(1.1468-7.8838) | 0.025 |
| Age (vs.
<70) | | | | |
|
≥70,
<90 | 0.44
(0.07-2.64) | 0.372 | 0.2084
(0.0036-11.9256) | 0.448 |
|
≥90 | 0.98
(0.20-4.88) | 0.976 | 0.5239
(0.0066-41.8813) | 0.772 |
| Nervous system (yes
vs. no) | 0.70
(0.23-2.13) | 0.529 | 0.6582
(0.1559-2.7785) | 0.569 |
| Respiratory system
(yes vs. no) | 1.64
(0.43-6.23) | 0.469 | 1.5633
(0.3953-6.1825) | 0.524 |
| Endocrine system
(yes vs. no) | 1.66
(0.59-4.63) | 0.336 | 1.7703
(0.5055-6.2001) | 0.372 |
| Cardiovascular
system (yes vs. no) | 3.21
(0.41-25.28) | 0.267 | 2.4038
(0.2467-23.4249) | 0.45 |
| Malignant tumor
(yes vs. no) | 1.35
(0.16-11.53) | 0.784 | 0.601
(0.01-36.2662) | 0.808 |
| aCCI (≥6 vs.
<6) | 2.19
(0.60-7.98) | 0.237 | 1.0812
(0.9113-1.2828) | 0.371 |
| Gastric
catheterisation (yes vs. no) | 0.39
(0.11-1.43) | 0.156 | 0.3795
(0.0721-1.9981) | 0.253 |
 | Table VIMultivariate analysis of risk factors
of infection based on the GEE model.a |
Table VI
Multivariate analysis of risk factors
of infection based on the GEE model.a
| A, Nosocomial
infection |
|---|
| | Unadjusted | Adjusted With
GEE |
|---|
| Parameters | OR (95% CI) | P-value | OR (95% CI) | P-value |
|---|
| Intervention (yes
vs. no) | 0.50
(0.25-0.97) | 0.042 | 0.50
(0.27-0.90) | 0.022 |
| Sex (female vs.
male) | 1.11
(0.52-2.34) | 0.788 | 1.11
(0.50-2.48) | 0.803 |
| Age (vs.
<70) | | | | |
|
≥70,
<90 | 1.08
(0.24-4.78) | 0.923 | 1.08
(0.19-5.96) | 0.933 |
|
≥90 | 1.17
(0.26-5.33) | 0.842 | 1.17
(0.21-6.59) | 0.862 |
| Nervous system (yes
vs. no) | 1.92
(0.76-4.85) | 0.168 | 1.92
(0.81-4.57) | 0.14 |
| Respiratory system
(yes vs. no) | 1.30
(0.49-3.40) | 0.598 | 1.30
(0.50-3.35) | 0.593 |
| Endocrine system
(yes vs. no) | 1.21
(0.59-2.48) | 0.609 | 1.21
(0.55-2.63) | 0.636 |
| Cardiovascular
system (yes vs. no) | 2.10
(0.71-6.25) | 0.183 | 2.10
(0.81-5.43) | 0.126 |
| aCCI (≥6 vs.
<6) | 1.21
(0.56-2.61) | 0.633 | 1.21
(0.49-3.00) | 0.686 |
| Gastric
catheterisation (yes vs. no) | 2.33
(1.15-4.73) | 0.019 | 2.33
(1.12-4.83) | 0.023 |
| B, Pulmonary
infection |
| | Unadjusted | Adjusted With
GEE |
| Parameters | OR (95% CI) | P-value | OR (95% CI) | P-value |
| Intervention (yes
vs. no) | 0.23
(0.11-0.48) | <0.001 | 0.23
(0.12-0.42) | <0.001 |
| Sex (female vs.
male) | 1.02
(0.46-2.28) | 0.954 | 1.02
(0.43-2.42) | 0.957 |
| Age (vs.
<70) | | | | |
|
≥70,
<90 | 1.78
(0.31-10.14) | 0.516 | 1.78
(0.39-8.11) | 0.456 |
|
≥90 | 2.09
(0.36-12.27) | 0.414 | 2.09
(0.47-9.20) | 0.329 |
| Nervous system (yes
vs. no) | 2.16
(0.80-5.82) | 0.129 | 2.16
(0.92-5.04) | 0.076 |
| Respiratory system
(yes vs. no) | 1.43
(0.52-3.94) | 0.488 | 1.43
(0.39-5.21) | 0.586 |
| Endocrine system
(yes vs. no) | 1.46
(0.68-3.15) | 0.335 | 1.46
(0.62-3.43) | 0.387 |
| Cardiovascular
system (yes vs. no) | 1.34
(0.45-3.96) | 0.598 | 1.34
(0.43-4.20) | 0.617 |
| CCI (≥6 vs.
<6) | 1.31
(0.57-3.00) | 0.522 | 1.31
(0.50-3.43) | 0.581 |
| Gastric
catheterisation (yes vs. no) | 3.59
(1.65-7.80) | <0.005 | 3.59
(1.69-7.61) | <0.001 |
| C, Other
infections |
| | Unadjusted | Adjusted With
GEE |
| Parameters | OR (95% CI) | P-value | OR (95% CI) | P-value |
| Intervention (yes
vs. no) | 0.46
(0.14-1.48) | 0.192 | 0.46
(0.16-1.36) | 0.16 |
| Sex (female vs.
male) | 3.30
(1.00-10.86) | 0.049 | 3.30
(1.17-9.29) | 0.024 |
| Age (vs.
<70) | | | | |
|
≥70,
<90 | 0.23
(0.03-1.97) | 0.181 | 0.23
(0.01-8.54) | 0.427 |
|
≥90 | 0.51
(0.06-4.30) | 0.532 | 0.51
(0.01-21.63) | 0.722 |
| Nervous system (yes
vs. no) | 0.57
(0.15-2.13) | 0.4 | 0.57
(0.14-2.34) | 0.432 |
| Respiratory system
(yes vs. no) | 1.54
(0.33-7.22) | 0.585 | 1.54
(0.38-6.25) | 0.547 |
| Endocrine system
(yes vs. no) | 1.87
(0.57-6.20) | 0.303 | 1.87
(0.53-6.63) | 0.33 |
| Cardiovascular
system (yes vs. no) | 2.42
(0.28-21.26) | 0.425 | 2.42
(0.24-24.77) | 0.456 |
| Malignant tumor
(yes vs. no) | 0.63
(0.04-9.19) | 0.737 | 0.63
(0.02-25.37) | 0.807 |
| CCI (≥6 vs.
<6) | 2.13
(0.50-9.12) | 0.31 | 2.13
(0.43-10.54) | 0.356 |
| Gastric
catheterisation (yes vs. no) | 0.38
(0.10-1.51) | 0.169 | 0.38
(0.07-2.00) | 0.253 |
 | Table VIILogistic regression model for the
association between intervening measure and infection stratified by
gastric catheterisation.a |
Table VII
Logistic regression model for the
association between intervening measure and infection stratified by
gastric catheterisation.a
| A, Model 0 |
|---|
| | Not catherised | Catherterised | Total |
|---|
| Type of
infection | OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value |
|---|
| Nosocomial
infection (yes vs. no) | | | | | | |
|
Unadjusted | 0.28
(0.11-0.71) | 0.007 | 0.96
(0.36-2.56) | 0.94 | 0.49
(0.25-0.94) | 0.032 |
|
Adjusted
with GEE | 0.28
(0.13-0.60) | <0.001 | 0.96
(0.35-2.62) | 0.941 | 0.49
(0.27-0.88) | 0.016 |
| Pulmonary infection
(yes vs. no) | | | | | | |
|
Unadjusted | 0.10
(0.03-0.35) | <0.001 | 0.40
(0.15-1.10) | 0.075 | 0.21
(0.10-0.45) | <0.001 |
|
Adjusted
with GEE | 0.10
(0.03-0.31) | <0.001 | 0.40
(0.15-1.11) | 0.078 | 0.21
(0.11-0.40) | <0.001 |
| Other infections
(yes vs. no) | | | | | | |
|
Unadjusted | 0.38
(0.11-1.29) | 0.121 | 0.55
(0.05-6.38) | 0.633 | 0.40
(0.13-1.22) | 0.109 |
|
Adjusted
with GEE | 0.38
(0.12-1.22) | 0.104 | 0.55
(0.13-2.31) | 0.414 | 0.40
(0.15-1.10) | 0.076 |
| B, Model I |
| | Not catherised | Catherterised | Total |
| Type of
infection | OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value |
| Nosocomial
infection (yes vs. no) | | | | | | |
|
Unadjusted | 0.25
(0.10-0.66) | 0.005 | 1.00
(0.35-2.82) | 0.999 | 0.49
(0.25-0.94) | 0.032 |
|
Adjusted
with GEE | 0.25
(0.11-0.58) | <0.001 | 1.00
(0.33-3.02) | 0.999 | 0.49
(0.27-0.88) | 0.016 |
| Pulmonary infection
(yes vs. no) | | | | | | |
|
Unadjusted | 0.09
(0.02-0.32) | <0.001 | 0.40
(0.14-1.11) | 0.077 | 0.21
(0.10-0.44) | <0.001 |
|
Adjusted
with GEE | 0.09
(0.03-0.30) | <0.005 | 0.40
(0.14-1.14) | 0.086 | 0.21
(0.11-0.40) | <0.001 |
| Other infections
(yes vs. no) | | | | | | |
|
Unadjusted | 0.34
(0.09-1.25) | 0.104 | N/A | N/A | 0.43
(0.14-1.37) | 0.154 |
|
Adjusted
with GEE | 0.34
(0.09-1.26) | 0.107 | 0.48
(0.07-3.11) | 0.438 | 0.40
(0.14-1.13) | 0.083 |
| C, Model II |
| | Not catherised | Catherterised | Total |
| Type of
infection | OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value |
| Nosocomial
infection (yes vs. no) | | | | | | |
|
Unadjusted | 0.27
(0.10-0.73) | 0.01 | 1.01
(0.29-3.52) | 0.985 | 0.52
(0.26-1.02) | 0.058 |
|
Adjusted
with GEE | 0.27
(0.11-0.61) | 0.002 | N/A | N/A | 0.52
(0.28-0.96) | 0.038 |
| Pulmonary infection
(yes vs. no) | | | | | | |
|
Unadjusted | 0.10
(0.02-0.37) | <0.001 | 0.39
(0.12-1.20) | 0.099 | 0.22
(0.10-0.48) | <0.001 |
|
Adjusted
with GEE | 0.10
(0.03-0.32) | 0 | 0.39
(0.12-1.24) | 0.111 | 0.22
(0.11-0.42) | <0.001 |
| Other infections
(yes vs. no) | | | | | | |
|
Unadjusted | 0.29
(0.07-1.24) | 0.094 | N/A | N/A | 0.43
(0.14-1.37) | 0.154 |
|
Adjusted
with GEE | 0.29
(0.08-1.02) | 0.053 | N/A | N/A | 0.43
(0.15-1.25) | 0.122 |
Predictive model for infection
outcomes
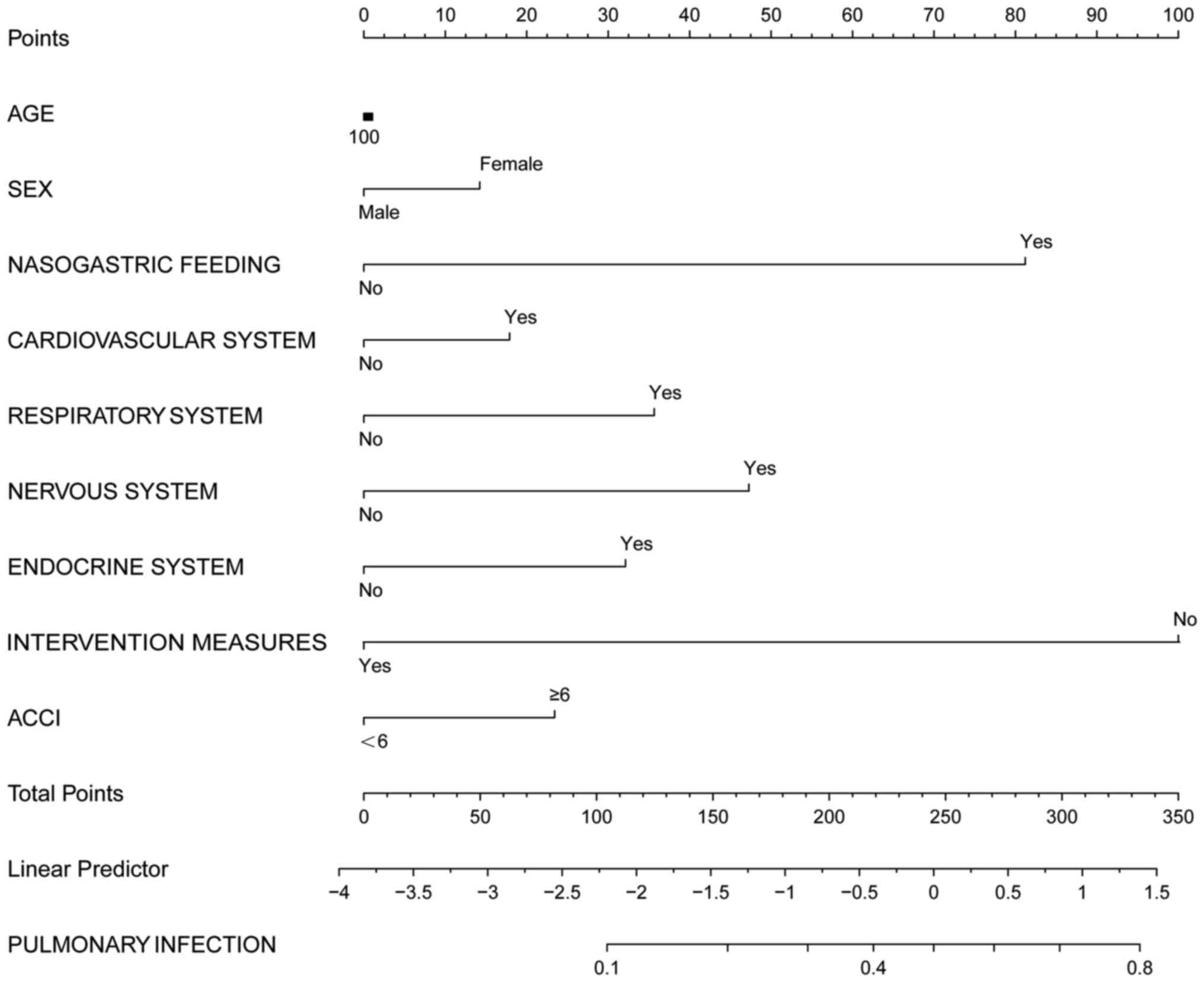
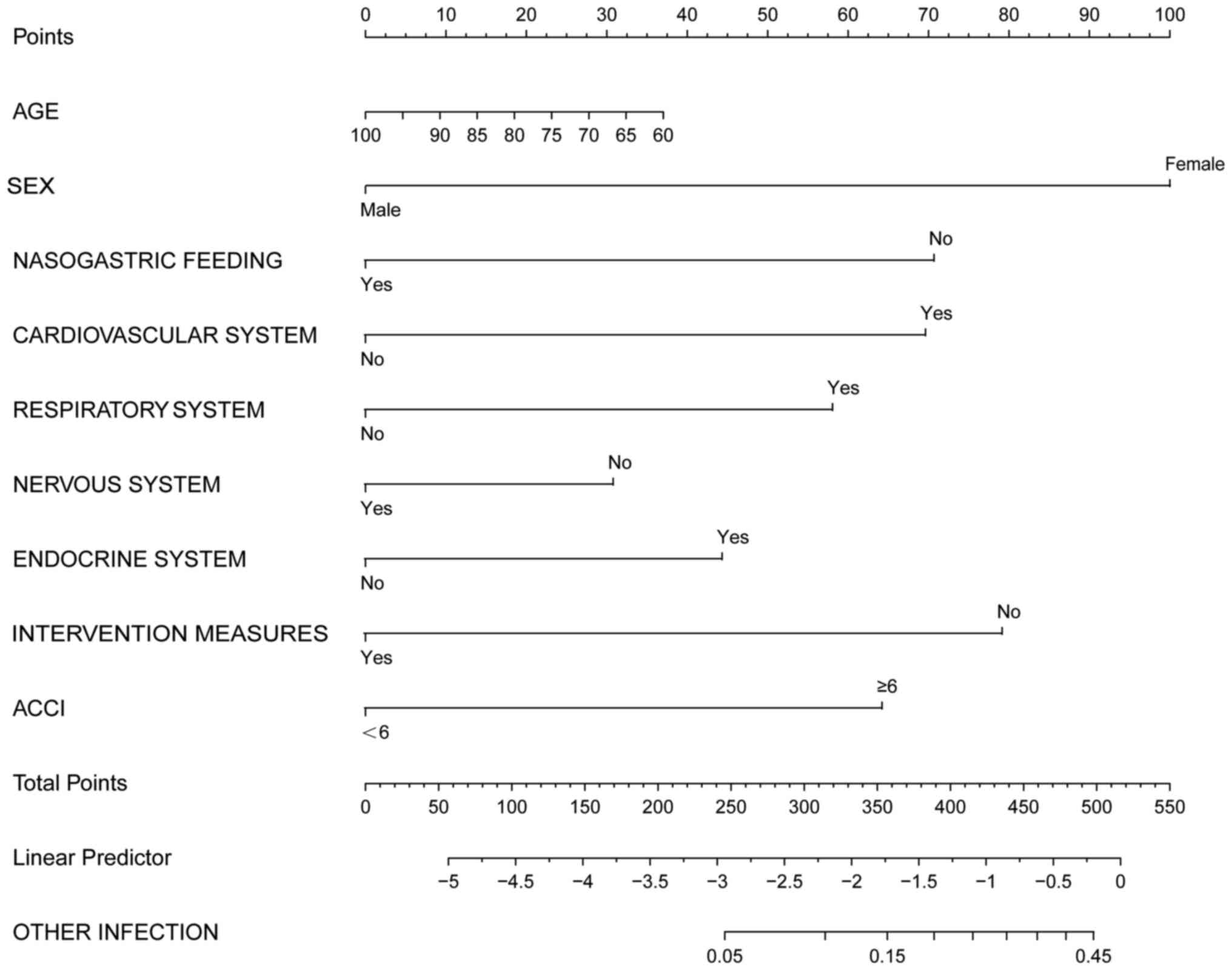
Based on the logistic, dichotomous and sequential
screening analyses, a nomogram incorporating the risk factors was
created for predicting infection outcomes using the EmpowerStats
statistical software 2.0 (www.empowerstats.com; X&Y Solutions Inc.), which
uses the R packages ‘Survival’ and ‘Rms’ (R version 3.4.3) to
calculate construct the nomograms (Fig. 3, Fig.
4 and Fig. 5).
In all patients with infection, a total score was
calculated based on age, sex, nasogastric feeding, diseases of the
cardiovascular, respiratory, nervous and endocrine systems, aCCI
and intervention measures. However, the application of this model
is relatively complex, therefore it has not been widely applied
clinically. Further simplification is required.
Discussion
The novelty of the present study lies in the
particularity of the study population. The study site was selected
in the geriatric department with a slow turnover, where a large
number of elderly inpatients were included into the study
population. Elderly inpatients are particularly susceptible to
nosocomial infections, especially in the lungs (55,56).
Physiological function in the elderly deteriorates over time, with
characteristics including decreases in age-related lung function,
weakness in the respiratory muscles and loss of the clearance
ability in the respiratory mucosa (57). These factors all contribute to
increasing the incidence of pneumonia and other chronic diseases,
such as chronic obstructive emphysema, chronic pulmonary heart
disease and chronic heart failure, among this demographic (57). In addition, since the majority of
elderly patients have comorbidities, ≥ one complications may occur
based on the disease type. Following cerebrovascular accidents,
including cerebral haemorrhage and cerebral infarction, elderly
patients are also predisposed to succumbing to aspiration
pneumonia, which is a common form of lung infection in elderly
patients (58). Furthermore,
patients with hemiplegia or those who are chronically bedridden
have been reported to be more predisposed to Streptococcus
anginosus-induced pneumonia (59). Patients with malignant tumours may
also develop radiation pneumonia as a result of long-term
radiotherapy (60). Long-term
chemotherapy and chronic management using immunosuppressants may
impair systemic immune system function, increasing the
susceptibility to infection (57).
Additionally, patients with malnutrition have diminished pathogen
resistance, which increases the risk of infection-related diseases
(61). Infection increases the
bodily demand for nutrition, further aggravating malnutrition and
to establish a vicious cycle (62). In addition, elderly patients
frequently display a decreased abilty to perceive their own health
status, leading to treatment delays or even mortality (63). In elderly inpatients with
pneumonia, the lack of clear clinical symptoms, ambiguous
examination and test results, misdiagnosis, delayed treatment and
coexistence of malnutrition, coupled with other diseases, all
contribute to increasing the risk of lung infection (61). This in turn aggravates illness
severity, prolongs the disease course and adds to the difficulty of
accurate diagnosis and treatment, leading to irreversible
consequences. The diagnosis and treatment of pulmonary infection in
elderly inpatients remains a major challenge. Therefore, during the
COVID-19 outbreak, appropriate measures were taken to prevent
nosocomial infections in elderly patients.
During the pandemic, the Geriatric Department of The
First Affiliated Hospital with Nanjing Medical University actively
adopted preventive measures. The present retrospective analysis of
the selected cases showed that implementing preventive and control
measures could effectively control the risk of nosocomial
infections. The key finding was the need to effectively screen
patients with infection before hospitalisation to prevent
cross-infection. During hospitalisation, it remains important to
reduce the flow of people in the hospital, wear masks for
protection, effectively cut off the infection source and route of
transmission (64). In the present
study, preventive measures employed to reduce nosocomial infections
in hospitalised patients in the geriatric wards during the COVID-19
pandemic were summarised. Results of the present retrospective
analysis revealed that these measures significantly reduced the
incidence of nosocomial infection among these patients. Further
implementation of these measures may help to effectively control
nosocomial infections in the future. Empirical recommendations are
required in response to important paroxysmal public health
incidents that occur in the future.
In the subgroup study, the nosocomial infection risk
of 93 inpatients in the Geriatrics Department of The First
Affiliated Hospital with Nanjing Medical University, excluding
patients who recently underwent surgery and anti-tumour therapy,
was analysed. These 93 patients with long-term stable conditions
were selected for longitudinal analysis. The results showed that
preventative measures could also effectively control the rate of
nosocomial infections in this group. In addition, it was found that
the use of indwelling gastric tube was an independent risk factor
for nosocomial infections, especially pulmonary infection, among
elderly inpatients. Clinically, the majority of elderly inpatients
with indwelling gastric tubes also suffer from malnutrition,
dysphagia or eating difficulty and severe coughing, all of which
have clear indications for gastric tube placement (65). Poor nutritional status is common
among elderly patients, especially in those who are hospitalised
long-term. This is due to the impaired ability to chew or swallow,
which also increases the risk of infection. However, regulating
nutrition has been found to contribute to recovery (66-68).
Gastric catheterisation is a common treatment method that can be
used to effectively improve the nutritional status in the elderly
(66). For cerebrovascular
accidents, such as cerebral haemorrhage and cerebral infarction,
swallowing dysfunction or cognitive impairment may lead to
aspiration difficulties, coughing and aspiration pneumonia
(69). Aspiration pneumonia is a
common subtype of lung infection in elderly patients (58), such that gastric tube placement can
effectively prevent aspiration pneumonia. For example, inserting a
catheter into the stomach through the nose can reduce the risk of
aspiration by eating through the mouth, thereby reducing the risk
of aspiration pneumonia. However, the results of the present study
revealed that the presence of an indwelling gastric tube was an
independent risk factor for pneumonia in elderly inpatients.
However, from previous clinical studies (70,71),
it is likely that patients with an indwelling gastric tube are more
predisposed to pneumonia because of their disease state, rather
than the process of gastric tube insertion. Therefore, the focus
should be on the placement, disinfection and replacement of the
gastric tube, combined with the regular removal of oropharyngeal
secretions and good positioning to avoid reflux and aspiration
(69). In the present study,
participants were patients receiving enteral nutrition who had
undergone gastric tube implantation. The results showed that the
indwelling gastric tube increased the risk of nosocomial
infections. Nevertheless, nosocomial infections caused by an
indwelling gastric tube should either be prevented altogether or at
least reduced. Clinically, novel comprehensive nutritional
indicators are required to evaluate the efficacy of specific
interventions, whereby further research is required to explore the
optimal time of intervention and treatment.
In the present study, the preventive and control
measures adopted by the Geriatric Department of The First
Affiliated Hospital with Nanjing Medical University during the
epidemic prevention and control period were discussed. The previous
medical environment was retrospectively summarised. In conclusion,
epidemic preventative and control measures were able to effectively
reduce the occurrence of nosocomial infections in elderly
inpatients. However, owing to the limited research data from
retrospective studies, the study participants in the present study
also had regional limitations. Therefore, the results obtained may
not apply to all geographic regions. It is necessary to further
expand the sample collection area and conduct prospective
multi-centre studies to improve upon existing measures. The
ultimate aim should be to gradually establish a mature medical
management system that is not limited to the prevention and control
of the epidemic. This will certainly contribute to the prevention
of nosocomial infections or reduction of their incidence in the
future.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by The National Key
R&D Program of China (grant nos. 2018YFC2002100 and
2018YFC2002102).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SSW, WL and MJZ conceived and designed the current
study. MJZ, WL, SSW, KW, QS and YJH acquired, analyzed the data and
interpreted the data. MJZ, WL, JL and SSW performed statistical
analysis. ML, SRS and BC participated in the acquisition of data
(including screening of enrolled patients and collection of test
indicators required for the study). JQW designed the study and
interpreted the data. WL and KW confirm the authenticity of all the
raw data. All authors critically revised the manuscript for
important intellectual content, and read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the First Affiliated Hospital with Nanjing Medical
University (approval no. 2020-SR-072; Nanjing). The institutional
review board of the The First Affiliated Hospital with Nanjing
Medical University approved this study and granted a waiver of
informed consent from study participants because of the
retrospective nature of the present study. In the present
retrospective study, patient data were obtained through the
electronic medical record system.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Safiabadi Tali SH, LeBlanc JJ, Sadiq Z,
Oyewunmi OD, Camargo C, Nikpour B, Armanfard N, Sagan SM and
Jahanshahi-Anbuhi S: Tools and techniques for severe acute
respiratory syndrome coronavirus 2 (SARS-CoV-2)/COVID-19 detection.
Clin Microbiol Rev. 34:00228–20. 2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Ahamd F, Almuayqil SN, Humayun M, Naseem
S, Khan WA and Junaid K: Prediction of COVID-19 cases using machine
learning for effective public health management. Comput Mater
Contin. 66:2265–2282. 2021.
|
|
3
|
Ntambara J and Chu M: The risk to child
nutrition during and after COVID-19 pandemic: What to expect and
how to respond. Public Health Nutr. 24:3530–3536. 2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Malekifar P, Pakzad R, Shahbahrami R,
Zandi M, Jafarpour A, Rezayat SA, Akbarpour S, Shabestari AN,
Pakzad I, Hesari E, et al: Viral coinfection among COVID-19 patient
groups: An update systematic review and meta-analysis. Biomed Res
Int. 2021(5313832)2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zandi M, Farahani A, Zakeri A, Akhavan
Rezayat S, Mohammadi R, Das U, Dimmock JR, Afzali S, Nakhaei MA,
Doroudi A, et al: Clinical symptoms and types of samples are
critical factors for the molecular diagnosis of symptomatic
COVID-19 patients: A systematic literature review. Int J Microbiol.
2021(5528786)2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Soltani S, Zakeri A, Zandi M, Kesheh MM,
Tabibzadeh A, Dastranj M, Faramarzi S, Didehdar M, Hafezi H,
Hosseini P and Farahani A: The role of bacterial and fungal human
respiratory microbiota in covid-19 patients. Biomed Res Int.
2021(6670798)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hosseini P, Afzali S, Karimi MR, Zandi M,
Zebardast A, Latifi T, Tabibzadeh A, Ramezani A, Zakeri A, Zakeri
A, et al: The coronavirus disease 2019 and effect on liver
function: A hidden and vital interaction beyond the respiratory
system. Rev Med Microbiol. 33:e161–e179. 2022.
|
|
8
|
Tsatsakis A, Petrakis D, Nikolouzakis TK,
Docea AO, Calina D, Vinceti M, Goumenou M, Kostoff RN, Mamoulakis
C, Aschner M and Hernández AF: COVID-19, an opportunity to
reevaluate the correlation between long-term effects of
anthropogenic pollutants on viral epidemic/pandemic events and
prevalence. Food Chem Toxicol. 141(111418)2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Iaccarino G, Grassi G, Borghi C, Ferri C,
Salvetti M and Volpe M: SARS-RAS Investigators. Age and
multimorbidity predict death among COVID-19 patients: Results of
the SARS-RAS study of the italian society of hypertension.
Hypertension. 76:366–372. 2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Pedersen OB, Nissen J, Dinh KM, Schwinn M,
Kaspersen KA, Boldsen JK, Didriksen M, Dowsett J, Sørensen E,
Thørner LW, et al: Severe acute respiratory syndrome coronavirus 2
(SARS-CoV-2) infection fatality rate among elderly danes: A
cross-sectional study on retired blood donors. Clin Infect Dis.
73:e2962–e2969. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kadambari S, Klenerman P and Pollard AJ:
Why the elderly appear to be more severely affected by COVID-19:
The potential role of immunosenescence and CMV. Rev Med Virol.
30(e2144)2020.PubMed/NCBI View
Article : Google Scholar
|
|
12
|
Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J,
Wang B, Xiang H, Cheng Z, Xiong Y, et al: Clinical Characteristics
of 138 hospitalized patients with 2019 novel coronavirus-infected
pneumonia in Wuhan, China. JAMA. 323:1061–1069. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Smith PW: Nosocomial infections in the
elderly. Infect Dis Clin North Am. 3:763–777. 1989.PubMed/NCBI
|
|
14
|
Plonquet A, Bastuji-Garin S, Tahmasebi F,
Brisacier C, Ledudal K, Farcet J and Paillaud E: Immune risk
phenotype is associated with nosocomial lung infections in elderly
in-patients. Immun Ageing. 8(8)2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu
Y, Zhang L, Fan G, Xu J, Gu X, et al: Clinical features of patients
infected with 2019 novel coronavirus in Wuhan, China. Lancet.
395:497–506. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kollef MH, Torres A, Shorr AF,
Martin-Loeches I and Micek ST: Nosocomial infection. Crit Care Med.
49:169–187. 2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Garner JS, Jarvis WR, Emori TG, Horan TC
and Hughes JM: CDC definitions for nosocomial infections, 1988. Am
J Infect Control. 16:128–140. 1988.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Vickery K, Deva A, Jacombs A, Allan J,
Valente P and Gosbell IB: Presence of biofilm containing viable
multiresistant organisms despite terminal cleaning on clinical
surfaces in an intensive care unit. J Hosp Infect. 80:52–55.
2012.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wenzel RP: Perspective: Attributable
mortality-the promise of better antimicrobial therapy. J Infect
Dis. 178:917–919. 1998.PubMed/NCBI View
Article : Google Scholar
|
|
20
|
Rangel-Frausto MS, Pittet D, Costigan M,
Hwang T, Davis CS and Wenzel RP: The natural history of the
systemic inflammatory response syndrome (SIRS). A prospective
study. JAMA. 273:117–123. 1995.PubMed/NCBI
|
|
21
|
Magill SS, Edwards JR, Bamberg W, Beldavs
ZG, Dumyati G, Kainer MA, Lynfield R, Maloney M, McAllister-Hollod
L, Nadle J, et al: Multistate point-prevalence survey of health
care-associated infections. N Engl J Med. 370:1198–1208.
2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kollef MH, Shorr A, Tabak YP, Gupta V, Liu
LZ and Johannes RS: Epidemiology and outcomes of
health-care-associated pneumonia: Results from a large US database
of culture-positive pneumonia. Chest. 128:3854–3862.
2005.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Labelle A and Kollef MH:
Healthcare-associated pneumonia: Approach to management. Clin Chest
Med. 32:507–515. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kollef MH, Hamilton CW and Ernst FR:
Economic impact of ventilator-associated pneumonia in a large
matched cohort. Infect Control Hosp Epidemiol. 33:250–256.
2012.PubMed/NCBI View
Article : Google Scholar
|
|
25
|
Du Q, Zhang D, Hu W, Li X, Xia Q, Wen T
and Jia H: Nosocomial infection of COVID-19: A new challenge for
healthcare professionals (Review). Int J Mol Med.
47(31)2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Schwierzeck V, König JC, Kühn J, Mellmann
A, Correa-Martínez CL, Omran H, Konrad M, Kaiser T and Kampmeier S:
First reported nosocomial outbreak of severe acute respiratory
syndrome coronavirus 2 in a pediatric dialysis unit. Clin Infect
Dis. 72:265–270. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Carter B, Collins JT, Barlow-Pay F,
Rickard F, Bruce E, Verduri A, Quinn TJ, Mitchell E, Price A,
Vilches-Moraga A, et al: Nosocomial COVID-19 infection: Examining
the risk of mortality. The COPE-nosocomial study (COVID in Older
PEople). J Hosp Infect. 106:376–384. 2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kahl A, du Bois A, Harter P, Prader S,
Schneider S, Heitz F, Traut A, Alesina PF, Meier B, Walz M, et al:
Prognostic value of the age-adjusted charlson comorbidity index
(ACCI) on short- and long-term outcome in patients with advanced
primary epithelial ovarian cancer. Ann Surg Oncol. 24:3692–3699.
2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Brusselaers N and Lagergren J: The
charlson comorbidity index in registry-based research. Methods Inf
Med. 56:401–406. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Frenkel WJ, Jongerius EJ, Mandjes-van
Uitert MJ, van Munster BC and de Rooij SE: Validation of the
charlson comorbidity index in acutely hospitalized elderly adults:
A prospective cohort study. J Am Geriatr Soc. 62:342–346.
2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Arabi YM, Fowler R and Hayden FG: Critical
care management of adults with community-acquired severe
respiratory viral infection. Intensive Care Med. 46:315–328.
2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Sun Q, Qiu H, Huang M and Yang Y: Lower
mortality of COVID-19 by early recognition and intervention:
Experience from Jiangsu Province. Ann Intensive Care.
10(33)2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Pan L, Wang L and Huang X: How to face the
novel coronavirus infection during the 2019-2020 epidemic: The
experience of Sichuan provincial people's hospital. Intensive Care
Med. 46:573–575. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Neagu M, Calina D, Docea AO, Constantin C,
Filippini T, Vinceti M, Drakoulis N, Poulas K, Nikolouzakis TK,
Spandidos DA and Tsatsakis A: Back to basics in COVID-19: Antigens
and antibodies-completing the puzzle. J Cell Mol Med. 25:4523–4533.
2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
The First Affiliated Hospital of Nanjing
Medical University: Patient Admission Procedures During COVID-19
(Edition 7). https://book.yunzhan365.com/ymkhc/oquc/mobile/index.html.
Accessed August 27, 2020.
|
|
36
|
Chung M, Bernheim A, Mei X, Zhang N, Huang
M, Zeng X, Cui J, Xu W, Yang Y, Fayad ZA, et al: CT imaging
features of 2019 novel coronavirus (2019-nCoV). Radiology.
295:202–207. 2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
National Health Commission of the People's
Republic of China: COVID-19 prevention and control protocols.
http://www.gov.cn/zhengce/zhengceku/2020-02/22/5482010/files/310fd7316a89431d977cc8f2dbd2b3e0.pdf.
February 21, 2020.
|
|
38
|
Kok RM and Reynolds CF III: Management of
depression in older adults: A review. JAMA. 317:2114–2122.
2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Farver-Vestergaard I, Jacobsen D and
Zachariae R: Efficacy of psychosocial interventions on
psychological and physical health outcomes in chronic obstructive
pulmonary disease: A systematic review and meta-analysis.
Psychother Psychosom. 84:37–50. 2015.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Sarris J, Moylan S, Camfield DA, Pase MP,
Mischoulon D, Berk M, Jacka FN and Schweitzer I: Complementary
medicine, exercise, meditation, diet, and lifestyle modification
for complementary medicine, exercise, meditation, diet, and
lifestyle modification for anxiety disorders: A review of current
evidence. Evid Based Complement Alternat Med.
2012(809653)2012.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Messika J, Kalfon P and Ricard JD:
Adjuvant therapies in critical care: Music therapy. Intensive Care
Med. 44:1929–1931. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Lim KK, Lee VSY, Tan CS, Kwan YH, Lim ZHX,
Wee HL, Østbye T and Low LL: Examining the heterogeneity inexcess
risks of coronary heart disease, stroke, dialysis, and lower
extremity amputation associated with type 2 diabetes mellitus
across demographic subgroups in an Asian population: A
population-based matched cohort study. Diabetes Res Clin Pract.
171(108551)2021.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Austin PC: Variance estimation when using
inverse probability of treatment weighting (IPTW) with survival
analysis. Stat Med. 35:5642–5655. 2016.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Austin PC and Stuart EA: Moving towards
best practice when using inverse probability of treatment weighting
(IPTW) using the propensity score to estimate causal treatment
effects in observational studies. Stat Med. 34:3661–3679.
2015.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Shoffner M, Owen P, Mostafa J, Lamm B,
Wang X, Schmitt CP and Ahalt SC: The secure medical research
workspace: An IT infrastructure to enable secure research on
clinical data. Clin Transl Sci. 6:222–225. 2013.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Ou HT, Lee TY, Du YF and Li CY:
Comparative risks of diabetes-related complications of basal
insulins: A longitudinal population-based cohort of type 1 diabetes
1999-2013 in Taiwan. Br J Clin Pharmacol. 84:379–391.
2018.PubMed/NCBI View Article : Google Scholar
|
|
47
|
VanderWalde NA, Meyer AM, Deal AM, Layton
JB, Liu H, Carpenter WR, Weissler MC, Hayes DN, Fleming ME and
Chera BS: Effectiveness of chemoradiation for head and neck cancer
in an older patient population. Int J Radiat Oncol Biol Phys.
89:30–37. 2014.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Liang KY and Zeger SL: Longitudinal data
analysis using generalised linear models. Biometrika. 73:13–22.
1986.
|
|
49
|
Allison PD: Longitudinal data analysis
using stata. Statistical Horison, Stockholm, 2018.
|
|
50
|
Koo TK and Li MY: A guideline of selecting
and reporting intraclass correlation coefficients for reliability
research. J Chiropr Med. 15:155–163. 2016.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Wu X, Wu L, Han J, Wu Y, Cao T, Gao Y,
Wang S, Wang S, Liu Q, Li H, et al: Evaluation of the sexual
quality of life and sexual function of cervical cancer survivors
after cancer treatment: A retrospective trial. Arch Gynecol Obstet.
304:999–1006. 2021.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Liu TT, Li R, Huo C, Li JP, Yao J, Ji XL
and Qu YQ: Identification of CDK2-related immune forecast model and
ceRNA in lung adenocarcinoma, a pan-cancer analysis. Front Cell Dev
Biol. 9(682002)2021.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Sun D, Tian L, Zhu Y, Wo Y, Liu Q, Liu S,
Li H and Hou H: Subunits of ARID1 serve as novel biomarkers for the
sensitivity to immune checkpoint inhibitors and prognosis of
advanced non-small cell lung cancer. Mol Med. 26(78)2020.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Hardin JW and Hilbe JM: Generalized
estimating equations. 2nd edition. Chapman & Hall/CRC Press,
London, 2012.
|
|
55
|
Li C, Duan J, Liu S, Meng X, Fu C, Zeng C
and Wu A: Assessing the risk and disease burden of clostridium
difficile infection among patients with hospital-acquired pneumonia
at a university hospital in Central China. Infection. 45:621–628.
2017.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Li C, Wen X, Ren N, Zhou P, Huang X, Gong
R, Feng L, Wu H, Liu Z, Fu C, et al: Point-prevalence of
healthcare-associated infection in China in 2010: A large
multicenter epidemiological survey. Infect Control Hosp Epidemiol.
35:1436–1437. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
57
|
Cho SJ and Stout-Delgado HW: Aging and
lung disease. Annu Rev Physiol. 82:433–459. 2020.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Zdravkovic M, Berger-Estilita J, Sorbello
M and Hagberg CA: An international survey about rapid sequence
intubation of 10,003 anaesthetists and 16 airway experts.
Anaesthesia. 75:313–322. 2020.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Hirai J, Sakanashi D, Haranaga S, Kinjo T,
Hagihara M, Kato H, Suematsu H, Yamagishi Y, Fujita J and Mikamo H:
Case-control study of pneumonia patients with Streptococcus
anginosus group bacteria in their sputum. J Infect Chemother.
22:794–799. 2016.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Sangro B, Martínez-Urbistondo D, Bester L,
Bilbao JI, Coldwell DM, Flamen P, Kennedy A, Ricke J and Sharma RA:
Prevention and treatment of complications of selective internal
radiation therapy: Expert guidance and systematic review.
Hepatology. 66:969–982. 2017.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Eraslan Doganay G and Cirik MO:
Determinants of prognosis in geriatric patients followed in
respiratory ICU; either infection or malnutrition. Medicine
(Baltimore). 100(e27159)2021.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Song P, Man Q, Li Y, Jia S, Yu D, Liu Z
and Zhang J: Trends of underweight malnutrition among chinese
residents aged 60 years and above-China, 1992-2015. China CDC Wkly.
3:232–236. 2021.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Shah FA, Pike F, Alvarez K, Angus D,
Newman AB, Lopez O, Tate J, Kapur V, Wilsdon A, Krishnan JA, et al:
Bidirectional relationship between cognitive function and
pneumonia. Am J Respir Crit Care Med. 188:586–592. 2013.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Yu P, Xia Z, Fei J and Jha SK: An
application review of artificial intelligence in prevention and
cure of COVID-19 pandemic. Comput Mater Contin. 65:743–760.
2020.
|
|
65
|
Bischoff SC, Austin P, Boeykens K,
Chourdakis M, Cuerda C, Jonkers-Schuitema C, Lichota M, Nyulasi I,
Schneider SM, Stanga Z and Pironi L: Espen guideline on home
enteral nutrition. Clin Nutr. 39:5–22. 2019.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Cena H and Chieppa M: Coronavirus disease
(COVID-19-SARS-CoV-2) and nutrition: Is infection in Italy
suggesting a connection? Front Immunol. 11(944)2020.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Willig A, Wright L and Galvin TA: Practice
paper of the academy of nutrition and dietetics: Nutrition
intervention and human immunodeficiency virus infection. J Acad
Nutr Diet. 118:486–498. 2018.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Kris-Etherton PM, Petersen KS, Hibbeln JR,
Hurley D, Kolick V, Peoples S, Rodriguez N and Woodward-Lopez G:
Nutrition and behavioral health disorders: Depression and anxiety.
Nutr Rev. 79:247–260. 2021.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Mandell LA and Niederman MS: Aspiration
pneumonia. N Engl J Med. 380:651–663. 2019.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Liu Y, Wang Y, Zhang B, Wang J, Sun L and
Xiao Q: Gastric-tube versus post-pyloric feeding in critical
patients: A systematic review and meta-analysis of pulmonary
aspiration- and nutrition-related outcomes. Eur J Clin Nutr.
75:1337–1348. 2021.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Alkhawaja S, Martin C, Butler RJ and
Gwadry-Sridhar F: Post-pyloric versus gastric tube feeding for
preventing pneumonia and improving nutritional outcomes in
critically ill adults. Cochrane Database Syst Rev.
2015(CD008875)2015.PubMed/NCBI View Article : Google Scholar
|