Introduction
Diffuse pulmonary lymphangiomatosis (DPL) is a rare
disease which is insidious, and its pathogenesis is unknown. It
usually occurs after birth or in childhood, and can also occur in
adults. There is no significant gender difference (1). Diffuse pulmonary lymphangiomatosis
(DPL) is a relatively rare congenital lymphoproliferative pulmonary
disease characterized by abnormal lymphatic hyperplasia, dilation,
and thickening of the soft tissues of the lungs, pleura, and
mediastinum (2). The younger the
age of onset, the worse the prognosis (3). The main clinical manifestations of
DPL patients are cough, sputum, chest tightness, shortness of
breath, and dyspnea, and some patients also show repeated
intractable chylothorax (4). Lung
CT scans may reveal diffuse interstitial changes, ground glass
manifestations, multiple nodules and diffuse septal thickening, and
diffuse mediastinal and paratracheal soft tissue infiltration
(3), with mediastinal lymph node
enlargement, perihilar soft tissue shadows, and pleural
calcification (5). Bronchoscopy
revealed diffuse translucent vesicular changes in the bronchial
wall mucosa (3). Respiratory
failure and chylous fluid accumulation secondary to infection are
the major causes of death in DPL patients (6,7). We
retrospectively analyzed the clinical data of a case of DPL in a
child admitted to the First Affiliated Hospital of Guangzhou
Medical University in order to improve clinicians' understanding of
this disease and explore new therapeutic directions.
Case report
An 8-year-old boy with the chief complaint of cough
for two weeks, anhelation for one week, and fever for two days was
admitted to our hospital on March 24, 2020. The onset of the
disease was mainly dry cough, and then cough phlegm appeared
gradually, accompanied by blood phlegm once (15 ml). The chest
computed tomography (CT) revealed ‘Interstitial pneumonia is
present in both lungs; Bilateral pleural effusion with local
consolidation.’ The patient received intravenous anti-infection
therapy of cefmetazole, cefoperazone sodium and sulbactam sodium
for two days and closed thoracic drainage in a local hospital, and
the therapeutic effect was not satisfactory, then the patient went
to our hospital for further treatment. The child initially
developed a chylous pericardial effusion at age 1. He underwent
partial pericardiectomy, pericardial drainage and thoracic catheter
ligation at another hospital for chylous pericardial effusion at
the age of 2, during which he also received anti-infection,
nutritional support and somatostatin therapy (The exact course of
treatment was not known). The patient was regularly followed up for
three years after discharge, and reexamination of chest radiographs
showed no abnormality.
Upon physical examination, the child looked
dyspnoeic and could not keep horizontal. The three concave signs
was positive. Respiratory sounds in both lungs were weakened,
obvious in the left lung, and wet rales and wheezing were heard.
Several lymph nodes with a diameter of 0.5-0.8 cm were found in the
neck, armpit, and groin of both sides, which were mobile, without
tenderness and adhesion.
Laboratory examinations revealed severe microcytic
hypochromic anemia, with hemoglobin value of 80 g/l. T-SPOT showed
no abnormality. Blood and pleural effusion cultures were negative.
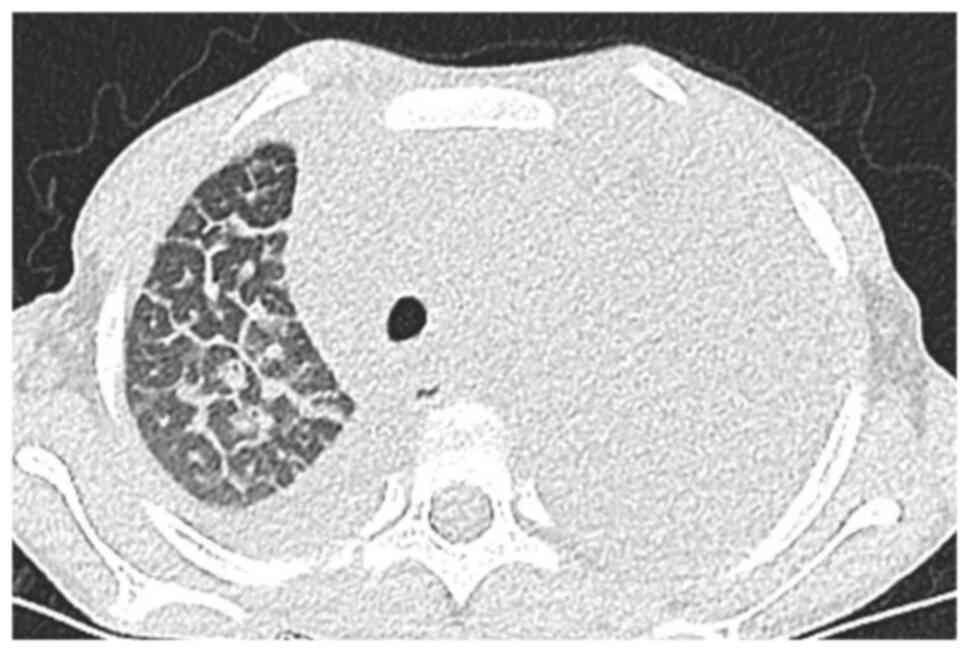
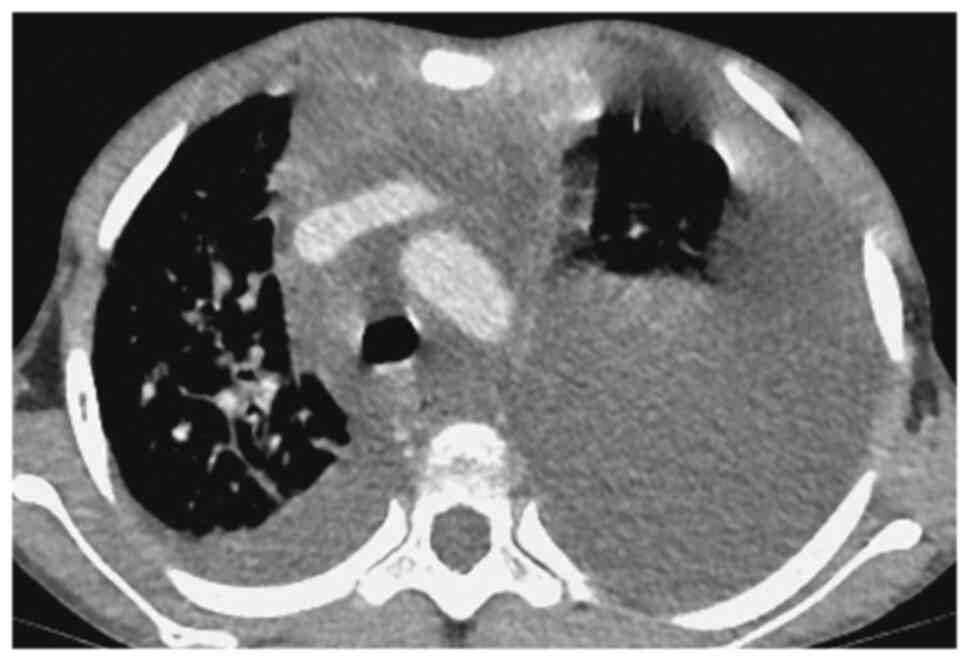
The pleural fluid test suggested chylothorax. Contrast-enhanced
chest CT (Figs. 1 and 2) showed multiple lymph nodes in
bilateral cervical roots and mediastinum, which were fused into
clusters, and the trachea and mediastinal great vessels were
surrounded. Furthermore, there was multiple thickening of
interlobular septa in the right lung, multiple inflammations in
both lungs and atelectasis in the left lung, and massive pleural
effusion on the left side. Positron emission tomography (PET)-CT
showed soft tissue thickening around the trachea, blood vessels,
and thyroid gland in the lower neck and the mediastinum, especially
in the anterior and superior mediastinum. The pulmonary
interlobular septum was significantly thickened, and the bronchial
vascular bundle was thickened, accompanied by multiple patchy
shadows of increased density and consolidation. The pleural
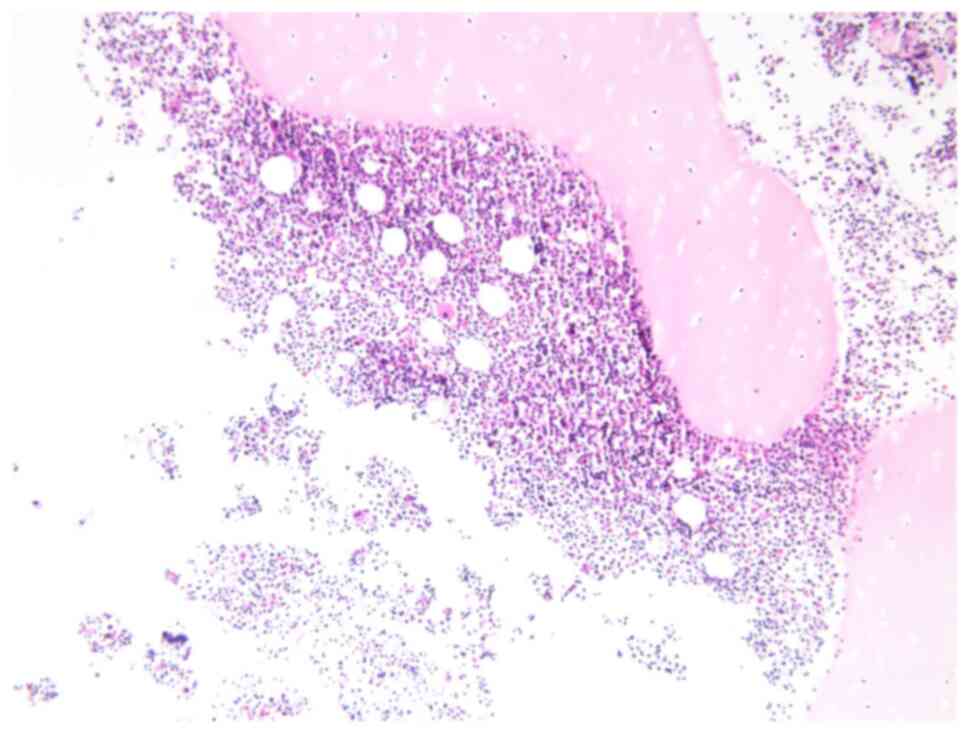
effusion of the child was chylous (Fig. 3). Biopsy of the mediastinal mass
revealed patchy, small lymphocytes with few thymus corpuscles. The
tumor tissue was fissured and had a sparse reticular structure
(Fig. 4). Immunohistochemistry
results revealed the following: D2-40 (+), CD31 (+), Ki67 (1%+),
SALL4 (-), SOX-10 (-), SMA (-), TDT (-), CD5 (-), CD117 (-), PLAP
(-), AFP (-), HMB45 (-) and tissue changes consistent with the
vascular origin of the tumor, inclined to lymphangioma.
After the treatment with prednisone, propranolol,
sirolimus and somatostatin, the clinical symptoms of the child were
improved. At the same time, the patient underwent a thoracentesis
and the drainage tube was removed after laboratory examination of
the pleural effusion. In the treatment of pleural effusion, we
chose conservative treatment instead of repeated drainage.
Afterwards, the pleural fluid in B-mode chest ultrasound was not
significantly increased, blood routine hemoglobin concentration was
stable, and fibrinogen level was not progressively decreased. Thus,
we can consider the treatment was effective. The child was
discharged on June 4, 2020. After discharge, he did not have a
fever or pale face. Regular blood routine examinations were
performed, and hemoglobin level was maintained at 90-110 g/l. He
did not have anhelation and could be supine. The child was admitted
to the hospital for a second time on July 27, 2020, and planned to
receive lymphangiography and occlusion. During hospitalization, the
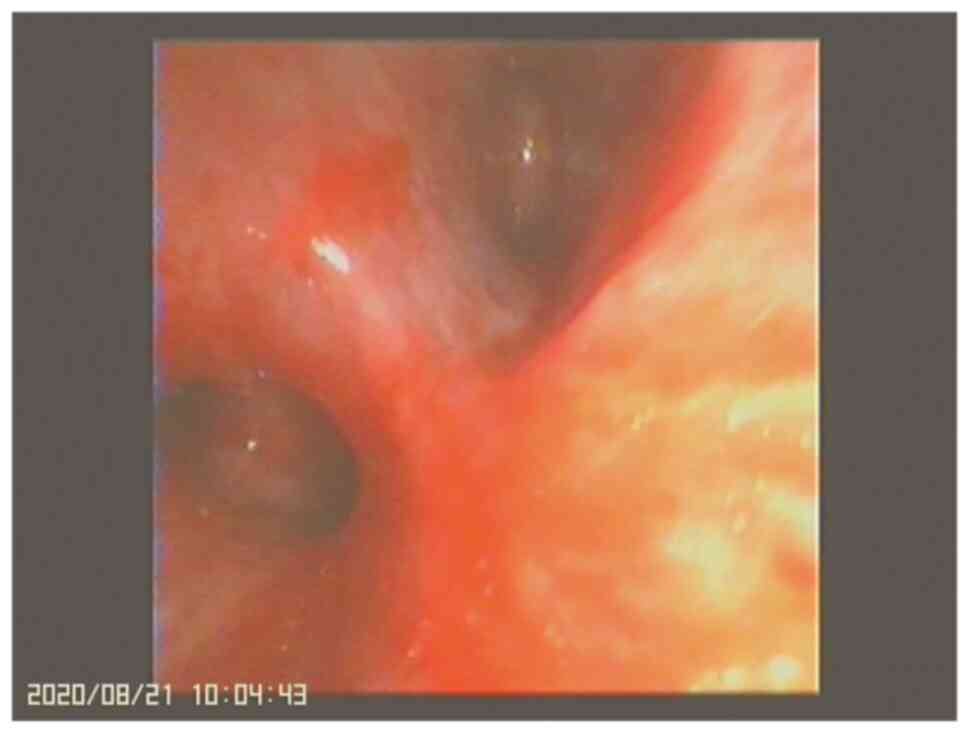
patient's condition worsened due to infection, and hemoptysis
occurred. Bronchoscopy showed obvious congestion, erosion, and
bleeding of the left bronchial mucosa (Fig. 5), which was considered as the cause
of hemoptysis. During the second admission, the patient was treated
with oral prednisone for anti-inflammation, propranolol for
stabilizing endothelial cells, sirolimus for suppressing the immune
response, piperacillin sodium and tazobactam sodium for
anti-infection activity, along with somatostatin, and calcium and
iron supplements, after which the patient's condition was stable.
Lymphangiogram and occlusion were performed on August 20, 2020. The
child's condition was continuously followed up and observed, and
regular outpatient follow-ups were performed until April 2021. The
child's condition is stable, and he has a normal diet, no cough,
hemoptysis, anhelation, chest tightness, and other discomforts.
Routine blood reexamination revealed a hemoglobin concentration of
110 g/l.
Discussion
Lymphangioma is when the original lymphatic sac is
isolated from the lymphatic system during embryonic development,
the remaining lymphoid tissue is hyperplastic, and the lymph fluid
gradually accumulates causing the lymphatic vessels to expand like
a capsule (3). About 10% of DPL
occurs in the mediastinum, and only in the lungs is rare (8).
DPL needs to be differentiated from diffuse
pulmonary lymphangiectasis, pulmonary lymphangiomatosis, pulmonary
capillary angiomatosis, sarcoidosis and other diseases. Diffuse
lymphangiectasia of lung includes primary and secondary 2 kinds.
Primary lymphangiectasia occurs mostly in infants and young
children. It is congenital abnormal development of interstitial
connective tissue of the lung, manifested as lymphangiectasia of
the pulmonary capillary. Secondary lymphangiectasia is mainly due
to surgical operations, radiation, infection, tumors, trauma and
other factors caused by lymphatic circulation disorders (9). Pulmonary capillary angiomatosis is a
pulmonary capillary abnormal hyperplasia disease, its clinical
symptoms are similar to pulmonary hypertension, common pleural
effusion and hemoptysis, can be diagnosed by pulmonary angiography
(10).
The case reported in this paper began to show
clinical symptoms at the age of 1, and was diagnosed as DPL at the
age of 8 according to pathological findings in our hospital. Among
the DPL patients reported in China, this case has the youngest
onset age and the longest diagnosis time span. The disease was not
diagnosed at the beginning, and clinical symptoms appeared again
six years after surgical treatment, indicating that the disease
progressed relatively slowly. Finally, the diagnosis was confirmed
by pathological examination. Because of the lack of clinical
understanding of DPL, which is prone to misdiagnosis, the patient
was not considered for the disease despite the surgical treatment
of chylothorax and pericardial effusion. To define the diagnosis
based on clinical and radiographic features alone is not enough;
most patients need confirmation by bronchoscopy, lung biopsy, or
open lung biopsy, but the risk is huge. Shen (11). reported a case of DPL in children
through bronchoscopy biopsy of the lung hemorrhage and death after
TBLB chylothorax. In our case, the mediastinal puncture was a safe
and effective method under B-ultrasound localization, which can be
confirmed by pathological manifestations and immunohistochemistry.
D2-40 staining was positive, and it can specifically identify
lymphatic endothelium.
There is no universally accepted specific drug for
the treatment of DPL, and treatment is mostly supportive and aimed
at alleviating clinical symptoms. Drug therapy included propranolol
(12), glucocorticoids, and
bevacizumab (13,14). Surgical treatment included thoracic
duct ligation and heart and lung transplantation (15). There have been reports of no
recurrence of the disease after 4 months to 3 years of follow-up
following complete surgical resection of the pulmonary lymphangioma
(16,17,18).
Kandi et al. reported that radiotherapy was used in the
treatment of lymphangioma, and no recurrence was observed during a
follow-up period of 20 months to 8 years (19). In our case, the child was treated
with oral propranolol, as reported by Ozeki et al (12), but there was no considerable
improvement. A review of relevant literature (20). shows that sirolimus is currently
considered to have a considerable effect in hemangioma,
lymphangioma, and other diseases that respond poorly to
propranolol. Gurskyte et al (21). studied the case of a 27-year-old
male patient with DPL and found that sirolimus could effectively
improve the condition and prevent disease progression. Combined
treatment with propranolol and sirolimus showed clinical efficacy.
Sirolimus combined with propranolol may be effective in improving
the condition of children with DPL, which opens up new treatment
options for such children. At present, the treatment of DPL pleural
effusion is still controversial. In the case reported in this
paper, conservative treatment has also achieved good results, which
also provides a new idea for the treatment of pleural effusion.
In summary, children who have difficulty breathing,
unexplained interlobular septal thickening, interstitial pneumonia
combined with hemorrhagic chylothorax, or pericardial effusion,
especially with the diagnosis of diffuse mediastinal soft tissue
infiltration, should be considered for DPL. The diagnosis of DPL
has certain difficulties, mainly based on the pathological
examination, and diagnosis by ultrasound-guided biopsy is effective
and safe. Sirolimus may be an alternative treatment option for
pediatric DPL. Conservative treatment of pleural effusion can
result in better curative effect.
Acknowledgements
Not applicable.
Funding
Funding: This work was supported by the General Project of
National Natural Science Foundation of China (grant no. 81770063)
and Guangzhou Science and Technology Plan Project in 2021, City
School (College) Joint Funding Project (grant no.
202201020419).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XPS and CYL wrote the manuscript and contributed to
the data analysis and interpretation. HYZ collected data. JXX, ZHH,
SY and DHC contributed to the data interpretation and manuscript
revision. DHC designed research and approved the final version of
manuscript. All authors read and approved the final manuscript. XPS
and CYL confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Consent for publication was obtained from the
patient's parents.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Liu JR, Shen WB, Wen Z, An R, Zhou CJ and
Zhao SY: Clinical analysis of two cases with diffuse pulmonary
lymphatic disease. Zhonghua Er Ke Za Zhi. 54:360–364.
2016.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
2
|
Minato H, Kaji S, Kinoshita E, Kurose N,
Nojima T, Kohno M, Konuma K and Ikawa H: Solitary intrapulmonary
cystic lymphangioma in an infant: A case report with literature
review. Pathol Res Pract. 206:851–856. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Faul JL, Berry GJ, Colby TV, Ruoss SJ,
Walter MB, Rosen GD and Raffin TA: Thoracic lymphangiomas,
lymphangiectasis, lymphangiomatosis, and lymphatic dysplasia
syndrome. Am J Respir Crit Care Med. 161 (3 Pt 1):1037–1046.
2000.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Alvarez OA, Kjellin I and Zuppan CW:
Thoracic lymphangiomatosis in a child. J Pediatr Hematol Oncol.
26:136–141. 2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Swensen SJ, Hartman TE, Mayo JR, Colby TV,
Tazelaar HD and Müller NL: Diffuse pulmonary lymphangiomatosis: CT
findings. J Comput Assist Tomogr. 19:348–352. 1995.PubMed/NCBI View Article : Google Scholar
|
|
6
|
de Lima AS, Martynychen MG, Florêncio RT,
Rabello LM, de Barros JA and Escuissato DL: Pulmonary
lymphangiomatosis: A report of two cases. J Bras Pneumol.
33:229–233. 2007.PubMed/NCBI View Article : Google Scholar : (In English,
Portuguese).
|
|
7
|
Sun X, Shen W, Xia S, Wen T and Wang R:
Diffuse pulmonary lymphangiomatosis: MDCT findings after direct
lymphangiography. AJR Am J Roentgenol. 208:300–305. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tazelaar HD, Kerr D, Yousem SA, Saldana
MJ, Langston C and Colby TV: Diffuse pulmonary lymphangiomatosis.
Hum Pathol. 24:1313–1322. 1993.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Run LJ, Yi XH and Zhu XY: Diffuse
pulmonary lymphangiomatosis: Clinical and pathologic analysis and
review of literature. Journal of Diagnostics Concepts &
Practice. 170–174. 2013.
|
|
10
|
Aviv R and McHugh K: Mechanisms of chylous
effusion in lymphangiomatosis. AJR Am J Roentgenol.
175(1191)2000.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Shen QY, Nong GM and Gu YY: diffuse
pulmonary lymphangiomatosis: Two cases report. Zhonghua Er Ke Za
Zhi. 54:781–782. 2016.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
12
|
Ozeki M, Fukao T and Kondo N: Propranolol
for intractable diffuse lymphangiomatosis. N Engl J Med.
364:1380–1382. 2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Onyeforo E, Barnett A, Zagami D, Deller D
and Feather I: Diffuse pulmonary lymphangiomatosis treated with
bevacizumab. Respirol Case Rep. 7(e00384)2018.PubMed/NCBI View
Article : Google Scholar
|
|
14
|
Aman J, Thunnissen E, Paul MA, van Nieuw
Amerongen GP and Vonk-Noordegraaf A: Successful treatment of
diffuse pulmonary lymphangiomatosis with bevacizumab. Ann Intern
Med. 156:839–840. 2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Bermejo Casero EJ, Mongil Poce R, Arrabal
Sánchez R, Fernández de Rota Avecilla A, Benítez Doménech A and
Fernández Bermúdez JL: Diffuse thoracic lymphangiomatosis:
Diagnosis and treatment. Arch Bronconeumol. 40:599–601.
2004.PubMed/NCBI(In Spanish).
|
|
16
|
Nakajima J, Goto A, Takamoto S, Murakawa
T, Fukami T and Kusakabe M: Invasive lymphangioma of the lung
manifesting as a large pulmonary mass with hemoptysis: Report of a
case. Surg Today. 37:418–422. 2007.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Lee CH, Kim YD, Kim KI, Lim YT, Lee KM,
Choi KU, Lee JS and Sol MY: Intrapulmonary cystic lymphangioma in a
2-month-old infant. J Korean Med Sci. 19:458–461. 2004.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Nagayasu T, Hayashi T, Ashizawa K, Muraoka
M, Tagawa T, Akamine S and Oka T: A case of solitary pulmonary
lymphangioma. J Clin Pathol. 56:396–398. 2003.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kandil A, Rostom AY, Mourad WA, Khafaga Y,
Gershuny AR and el-Hosseiny G: Successful control of extensive
thoracic lymphangiomatosis by irradiation. Clin Oncol (R Coll
Radiol). 9:407–411. 1997.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Buhao Sun: Research progress of sirolimus
in the treatment of vascular malformation. Journal of Tissue
Engineering and Reconstructive Surgery. 15:263–265. 2019.
|
|
21
|
Gurskytė V, Zeleckienė I, Maskoliūnaitė V,
Mickys U and Šileikienė V: Successful treatment of diffuse
pulmonary lymphangiomatosis with sirolimus. Respir Med Case Rep.
29(101014)2020.PubMed/NCBI View Article : Google Scholar
|