Introduction
The morbidity and mortality associated with cardiac
arrest (CA) are high worldwide, and the return of spontaneous
circulation (ROSC) causes severe cerebral ischemia-reperfusion
injury (CIRI), which is the main cause of coma in patients with CA
(1,2). Owing to the complex mechanism of
CIRI, the available therapies are not ideal to promote nervous
system recovery (3). Therefore,
the development of new strategies for the clinical management of
CIRI following CA and cardiopulmonary resuscitation (CPR) has been
a challenge.
During CIRI, the fluidity of the mitochondrial
membrane decreases, and the synthesis of adenosine triphosphate is
reduced (4,5). Additionally, the ability of the
plasma membrane and endoplasmic reticulum (ER) calcium pumps to
control calcium levels is compromised, which leads to intracellular
calcium overloads (6,7). Persistent calcium overloads lead to
excessive activation of calpain, which activates or hydrolyzes
other enzymes, resulting in further damage to neuronal function
(8). Therefore, the development of
methods to alleviate CIRI by blocking cellular dysfunction caused
by calpain activation represents an active area of research
(9).
Calpain, a neutral cysteine protease, is widely
expressed in several organisms (humans, fish and amphibians). The
calpain protein family includes >10 subtypes (CAPN1-3, 5-15 and
17), but calpain-1 and calpain-2 are of particular interest because
they are highly expressed in the brain tissue and show
calcium-dependent activities (10,11).
Animal experiments have shown that calpain plays a key role in the
pathophysiology of cerebral ischemic injury (12,13).
Calpastatin (encoded by CAST) is an endogenous specific inhibitor
of calpain and acts on both calpain-1 and calpain-2(14). After a cerebral ischemic injury,
the calpastatin level is not adequate to counterbalance excessively
activated calpain (15).
Therefore, exogenous calpain inhibitors have been developed to
alleviate ischemic brain injury, brain trauma and other
neurological diseases (16).
MDL28170 is a non-selective calpain inhibitor (17,18)
that exerts a neuroprotective effect on cerebral ischemia and
hypoxic injury in adult and neonatal rats by inhibiting
calpain-1(19).
In 1998, Li et al (20) revealed that post-ischemic treatment
with the calpain inhibitor MDL28170 (in a study on bilateral common
carotid artery occlusion-induced global cerebral
ischemia-reperfusion injury) improved brain damage in the gerbil
model of systemic ischemia; however, apart from the inhibition of
calpain-induced proteolysis, other mechanisms are unclear.
Furthermore, MDL28170 can reduce traumatic brain injury in rats by
blocking calpain-2(18). It is
well known that the inflammatory response plays an important role
in CIRI (21). When cerebral
ischemia occurs, the immune system is triggered, immune cells in
the brain tissue are activated and inflammatory cytokines [such as
interleukin (IL)-1β and tumor necrosis factor (TNF)-α] are
released. A large number of inflammatory factors invade the brain
parenchymal tissue and disrupt the blood-brain barrier. It also
stimulates the synthesis of other immune cells and adhesion
molecules in the blood, amplifying the inflammatory cascade and
ultimately exacerbating nerve damage (22). In a study on a traumatic brain
injury mouse model, MDL28170 played a neuroprotective role by
reducing the expression of NF-κB and decreasing the release of
IL-1β and TNF-α (23). Our
previous research on brain injury after cardiac arrest in the same
period demonstrated that calpain-2 is closely associated with
neuroinflammation induced by brain astrocytes and microglia
(24), but the role of different
calpain isoforms in brain injury is unclear. Therefore, it would be
beneficial to explore the changes of inflammatory response in CIRI
by targeting calpain.
Autophagy is an important intracellular
self-degradation process; however, excessive autophagy can lead to
cell component depletion and cell death. Increasing evidence
suggests that autophagy is involved in CIRI (25,26).
Our previous study demonstrated that the inhibition of
mitochondrial autophagy can attenuate the brain damage in rats
after CA and CPR (27). Moreover,
a recent study showed that calpain-2 is involved in TNF-α-mediated
hippocampal neuron autophagy, resulting in neuronal death (28). To the best of our knowledge, the
present study is the first to evaluate calpain-related proteins and
the effect of MDL28170 on inflammation and autophagy in CIRI by
constructing a CA rat model through retroesophageal electrical
stimulation.
Materials and methods
Animals
A total of 81 male Sprague-Dawley rats
(8-10-week-old; body weight, 220-250 g; Experimental Animal Center
of Guangxi Medical University, Nanning, China) were used in the
present study. The rats were housed in facilities with a 12 h/12 h
light/dark cycle at 25˚C and had access to food and water ad
libitum. All animal procedures were conducted in compliance
with the Guide for the Care and Use of Experimental Animals. The
study was approved by the Animal Ethics Committee of Guangxi
Medical University (Nanning, China; approval no. 20190915). Except
for 21 rats that died during CA-CPR modeling, all other rats were
euthanized with 2% pentobarbital sodium (90 mg/kg) as well as via
cervical dislocation at 24 h after CA/CPR; the duration of the
experiment was 24 h. There were two main causes of mortality in the
animals: i) The rats failed to be resuscitated after cardiac arrest
induced during the experiment; or ii) at the end of the experiment,
the rats were euthanized. The vital signs (body temperature, heart
rate and blood pressure) of the rats were continuously monitored
during the entire processes of model-making and euthanasia. Rat
mortality was confirmed when the following four signs were found:
Lack of a heartbeat; lack of respiration; lack of corneal reflex;
and presence of rigor mortis.
Experimental CA/CPR model
All rats were subjected to fasting for 12 h before
surgery. Anesthesia was induced with an intraperitoneal injection
of 2% pentobarbital sodium (30 mg/kg). CA was induced by electrical
stimulation with transesophageal cardiac pacing, as described
previously (29). Before CA
induction, femoral artery blood pressure monitoring, femoral
venipuncture catheterization and endotracheal intubation were
performed. After 7 min of untreated CA, CPR was performed by manual
chest compression (180 compressions/min) to a depth of 25-30% of
the anteroposterior diameter of the thorax and with equal
compression-relaxation duration by the same investigator. ROSC was
defined as an unassisted pulse with a mean arterial pressure of ≥50
mmHg for ≥1 min. When the blood pressure and autonomous respiration
were stable (breaths ≥40/min), mechanical ventilation was withdrawn
after ROSC.
Drug treatment and experimental
groups
MDL28170, a calpain inhibitor (cat. no. M6690;
Sigma-Aldrich; Merck KGaA), was dissolved in 8% dimethyl sulfoxide
(DMSO) to final concentrations of 1.5 and 3.0 mg/kg (30). The vehicle group received an equal
volume of solvent via the same method of administration.
Successfully established model rats were randomly
divided into four groups: i) CA control group, normal saline (NS)
was administered (NS, n=18); ii) control vehicle group, 8% DMSO was
administered (DMSO, n=18); iii) low-dose MDL28170 group (MDL-l, 1.5
mg/kg, n=18); and iv) high-dose MDL28170 group (MDL-h, 3.0 mg/kg,
n=18). Another nine rats were selected for the sham operation
group, which were subjected to the same procedures but without CA
and CPR. All drugs were injected into the left femoral vein within
30 min of ROSC.
Evaluation of survival rate,
neurological deficit score (NDS) and CPR duration
CPR duration was measured as the time from the start
of cardiac compression to the ROSC. The survival rate and NDS of
the experimental rats were evaluated 24 h after the ROSC by an
investigator blinded to the experimental groups. NDS was graded on
a scale of 0-80 based on the arousal, reflex, motor, sensory and
balance responses (most severe deficit, 0; normal performance, 80)
(31). All experimental rats were
used for the subsequent tests to evaluate neuronal function.
Tissue sampling and preparation
Three experimental animals from each group were
deeply anesthetized using 2% pentobarbital sodium (90 mg/kg) at 24
h after CA/CPR. Saline and 4% paraformaldehyde were infused through
the aorta for 2 h at 4˚C. The excised brain tissue was immediately
fixed in 10% paraformaldehyde at room temperature for 48 h, and
paraffin-embedded sections were prepared. Thus, 3-µm-thick
paraffin-embedded sections were used for hematoxylin-eosin (HE) or
double immunofluorescence staining, and 5-µm-thick
paraffin-embedded sections for Nissl staining as detailed
below.
HE and Nissl staining
The prepared brain sections were dehydrated in the
xylene solution to remove the paraffin, then rehydrated in a
descending alcohol series (100, 95, 75 and 50% ethanol) at 25˚C for
5 min/step. Tissue samples were stained with hematoxylin at 25˚C
for 5 min. The tissue samples were stained with HE and 1%
hydrochloric acid in 70% ethanol at 25˚C for 5 min. The specimens
were then dried, covered with cover slides and observed under an
optical microscope (Olympus Corporation).
For Nissl staining, the prepared brain sections also
were dehydrated in the xylene solution to remove the paraffin and
then rehydrated in a descending alcohol series (100, 95, 75 and 50%
ethanol) at 25˚C for 5 min/step. Tissue samples were stained with
toluidine blue solution (cat. no. G3668; Beijing Solarbio Science
& Technology Co., Ltd.) in a dark, airtight container at 70%
humidity and 60˚C for 30 min. Samples were then re-dyed in 0.5%
eosin at 25˚C for 3 sec. Under a light microscope, the Nissl bodies
in the tissue sections were counted in five randomly selected
regions of every sample using ImageJ version 1.46 (National
Institutes of Health).
Transmission electron microscopy (TEM)
analysis
Three rats from each group were used for TEM
analysis. After cerebral perfusion with 4% paraformaldehyde, 40 mg
of the cortex specimen was placed in 2.5% glutaraldehyde and stored
at 4˚C for 24 h. The samples were fixed with 1% osmium tetroxide at
room temperature for 3 h, dehydrated in ethanol and then embedded
in epoxy resin at 37˚C for 1 h. According to the standard operating
principles of three-dimensional localization, resin blocks were cut
into ultrathin slices (100-nm), fixed on metal mesh grids and
double-stained using lead citrate and uranyl acetate at 25˚C for 30
min. Finally, the morphology of the neurons was observed using TEM.
The images were recorded and viewed with an H-7650 TEM unit
(Hitachi, Ltd.).
Western blotting
Rats (n=3 per group) were euthanized after
deep anesthetization, and the brain tissue was immediately excised
and washed with cold saline. The isolated cerebral cortex was
quick-frozen in liquid nitrogen (-196˚C) for 3 h and stored at
-80˚C for subsequent western blotting. The cortex sample (50 mg
from each group) was lysed at 4˚C, and the tissue homogenate was
centrifuged at 13,000 x g for 15 min at 4˚C to extract the
proteins. A bicinchoninic acid protein assay kit (cat. no. P0012;
Beyotime Institute of Biotechnology) was used to test the total
protein concentration. Protein samples (40 µg/lane) were separated
using sodium dodecyl sulfate-polyacrylamide gel (12, 10 or 8%
separation gel) electrophoresis and transferred onto a
polyvinylidene fluoride membrane (pore size, 0.2 µm;
MilliporeSigma), which was blocked for 1 h with 5% fat-free milk
solution at 25˚C and incubated with the corresponding primary
antibodies overnight at 4˚C. The following rabbit anti-rat primary
antibodies were used: Calpain-1 (1:1,000; cat. no. ab28258; Abcam),
calpain-2 (1:1,000; cat. no. 11472-1-AP; Wuhan Sanying
Biotechnology, Inc.), calpastatin (1:5,000; cat. no. ab226249;
Abcam), P62 (1:1,000; cat. no. #5114; Cell Signaling Technology,
Inc.), Beclin-1 (1:1,000; cat. no. #3495; Cell Signaling
Technology, Inc.), LC3 (1:2,000; cat. no. ab192890; Abcam), IL-1β
(1:100; cat. no. sc-32294; Santa Cruz Biotechnology, Inc.), TNF-α
(1:1,000; cat. no. ab6671; Abcam) and glyceraldehyde 3-phosphate
dehydrogenase (GAPDH; 1:1,000; cat. no. #5174; Cell Signaling
Technology, Inc.). After washing with Tris-buffered saline with
0.2% Tween 20, the samples were incubated with a goat anti-rabbit
IgG fluorescence-labeled secondary antibody (1:20,000; cat. no.
#5151T; Cell Signaling Technology, Inc.) at 25˚C for 1 h. Finally,
the strip density was detected using the Tanon™ High-sig ECL
Western Blotting Substrate (Guangzhou Yuwei Biotechnology
Instrument Co., Ltd.). ImageJ version 1.46 (National Institutes of
Health) was used to quantify each protein band, with measured
levels normalized to that of GAPDH.
Double immunofluorescence
staining
The brain tissue sections were dewaxed, hydrated in
a descending alcohol series (100, 95, 75 and 50% ethanol), and
microwaved at 60˚C for 5 min in an ethylenediaminetetraacetic acid
antigen-repair buffer (pH 8.0; Beyotime Institute of
Biotechnology), followed by incubation at 25˚C for 20 min in
phosphate-buffered saline (pH 7.4) containing 0.3% Triton X-100
(phosphate-buffered saline with Tween 20). The tissue sections were
blocked in normal goat serum at 25˚C for 30 min (OriGene
Technologies, Inc.) with bovine serum albumin (Thermo Fisher
Scientific, Inc.). The specimens were initially incubated at 4˚C
with rabbit polyclonal anti-calpain-2 (1:2,000; cat. no.
11472-1-AP; ProteinTech Group, Inc.). The samples were stored at
4˚C and incubated overnight. The following day, the sections were
incubated with horseradish peroxidase-conjugated goat anti-rabbit
secondary antibody (1:500; cat. no. GB23301; Wuhan Servicebio
Technology Co., Ltd.) in the dark at 25˚C for 60 min. Next, the
fluorescein isothiocyanate reagent was added and the sections were
incubated for 10 min in dark at 25˚C. Subsequently, the sections
were placed in a repair box filled with citric acid (pH 6.0)
antigen-repair solution and heated in a microwave oven (heated at
70˚C for 8 min, no heat for 10 min, heated at 60˚C for 7 min and
then allowed to gradually cool down). The sections were incubated
with rabbit monoclonal anti-LC3 (1:1,000; cat. no. ab192890; Abcam)
overnight at 4˚C. Next, the brain tissue was incubated with
Cy3-coupled secondary antibody solution (1:300; cat. no. GB21303;
Wuhan Servicebio Technology Co., Ltd.) at 25˚C for 60 min, followed
by incubation with DAPI at 25˚C for 10 min. Finally, an
auto-fluorescence quenching agent was added to avoid background
fluorescence and the fluorescent markers were observed. The slides
were scanned using the Pannoramic DESK system (3DHISTECH Kft.) for
panoramic scanning. Full-field digital slice images were generated,
and the CaseViewer Windows application (3DHISTECH Kft.) was used to
compile them. Finally, ImageJ version 1.46 (National Institutes of
Health) was used to analyze the rates of cells positive for
calpain-2 and LC3 and the ratio of calpain-2 to LC3 in the
cells.
Statistical analysis
GraphPad Prism 7 (GraphPad Software, Inc.) was used
for all statistical analyses and graph creation. The assumptions of
normality were evaluated using Shapiro-Wilk test. Statistical
differences between groups were analyzed using a one-way analysis
of variance and Tukey's post hoc test. The non-normally distributed
data and the intergroup comparisons were evaluated using the
Kruskal-Wallis test and Dunn's test. P<0.05 was considered to
indicate a statistically significant difference. The survival rates
were analyzed using a χ2 cross-table. The NDS is
expressed as median and interquartile range, and other data are
presented as mean ± standard deviation.
Results
Survival rate, CPR duration and
NDS
There were no significant differences in the CPR
duration among the groups (P>0.05; Table I). The survival rate of rats in the
sham group was 100%. The survival rate of rats in the 0.9% saline
(NS) (61.1%), DMSO (66.7%), MDL-l (72.2%) and MDL-h (83.3%) groups
was lower compared with that of rats in the sham group, but no
significant difference was found in the survival rate among the
groups (P>0.05; Table I).
Additionally, the NDS of the NS (70), DMSO (70), MDL-l (73) and
MDL-h (74) groups was significantly lower compared with that of the
sham group (80) (all P<0.05). However, the NDS of the MDL-h
group was significantly higher compared with that of the NS and
DMSO groups (P<0.05; Table
I).
 | Table ISurvival rate, CPR duration and NDS
in each group. |
Table I
Survival rate, CPR duration and NDS
in each group.
| Group | Survival rate, n
(%) | n | CPR duration,
sec | NDS, median (25th
percentile, 75th percentile) |
|---|
| Sham | 9/9 (100.0) | 9 | - | 80 (80, 80) |
| NS | 11/18 (61.1) | 11 | 98.36±3.331 | 70 (69,
71)a |
| DMSO | 12/18 (66.7) | 12 | 92.25±1.733 | 70 (68,
71.75)a |
| MDL-l | 13/18 (72.2) | 13 | 94.85±1.996 | 73 (70,
74)a |
| MDL-h | 15/18 (83.3) | 15 | 93.53±2.24 | 74 (72,
76)a,b,c |
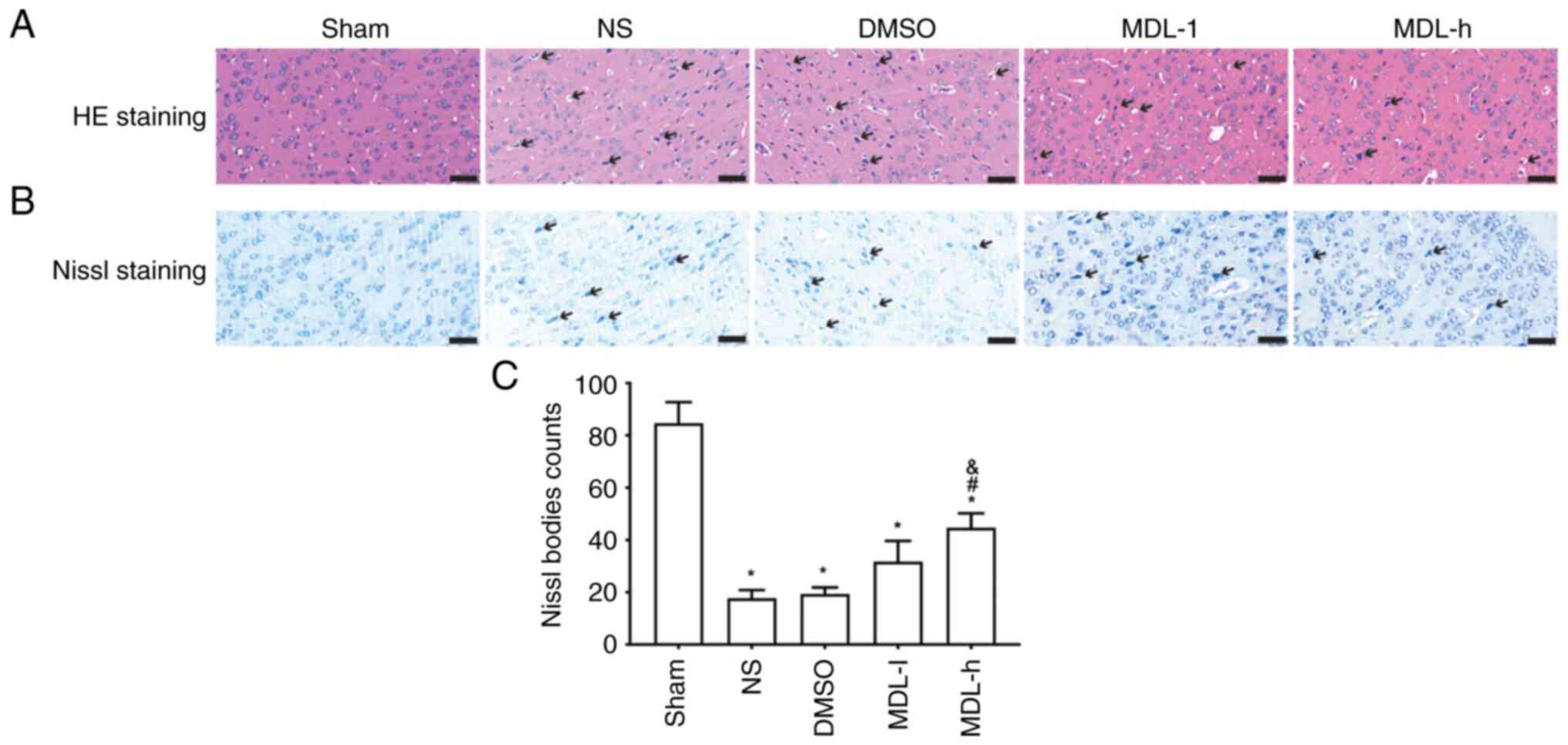
Morphological changes in neurons in
the cortex after CA and CPR
To evaluate the effect of different doses of the
calpain inhibitor MDL28170 on CIRI after CPR, morphological changes
in neurons were observed using HE and Nissl staining. As shown in
the HE-stained images, the nuclei of cells in the sham group were
round and intact, the nucleoli were visible, and the fiber
structure of the brain tissue was normal. In the NS and DMSO
groups, necrotic cells, nuclear deformation and pyknosis, cell
vacuoles and tissue exudation were observed. The morphology of the
cortical neurons in the MDL-l and MDL-h groups was improved
compared with that in the NS and DMSO groups, and the morphology of
the cortical neurons in the MDL-h group was improved compared with
that in any other group (Fig. 1A).
Nissl staining revealed significantly fewer Nissl bodies in the
model groups compared with in the sham group (P<0.05; Fig. 1B and C). The numbers of Nissl bodies were
significantly increased in the MDL-h group compared with in the NS
and DMSO groups (P<0.05; Fig.
1B and C).
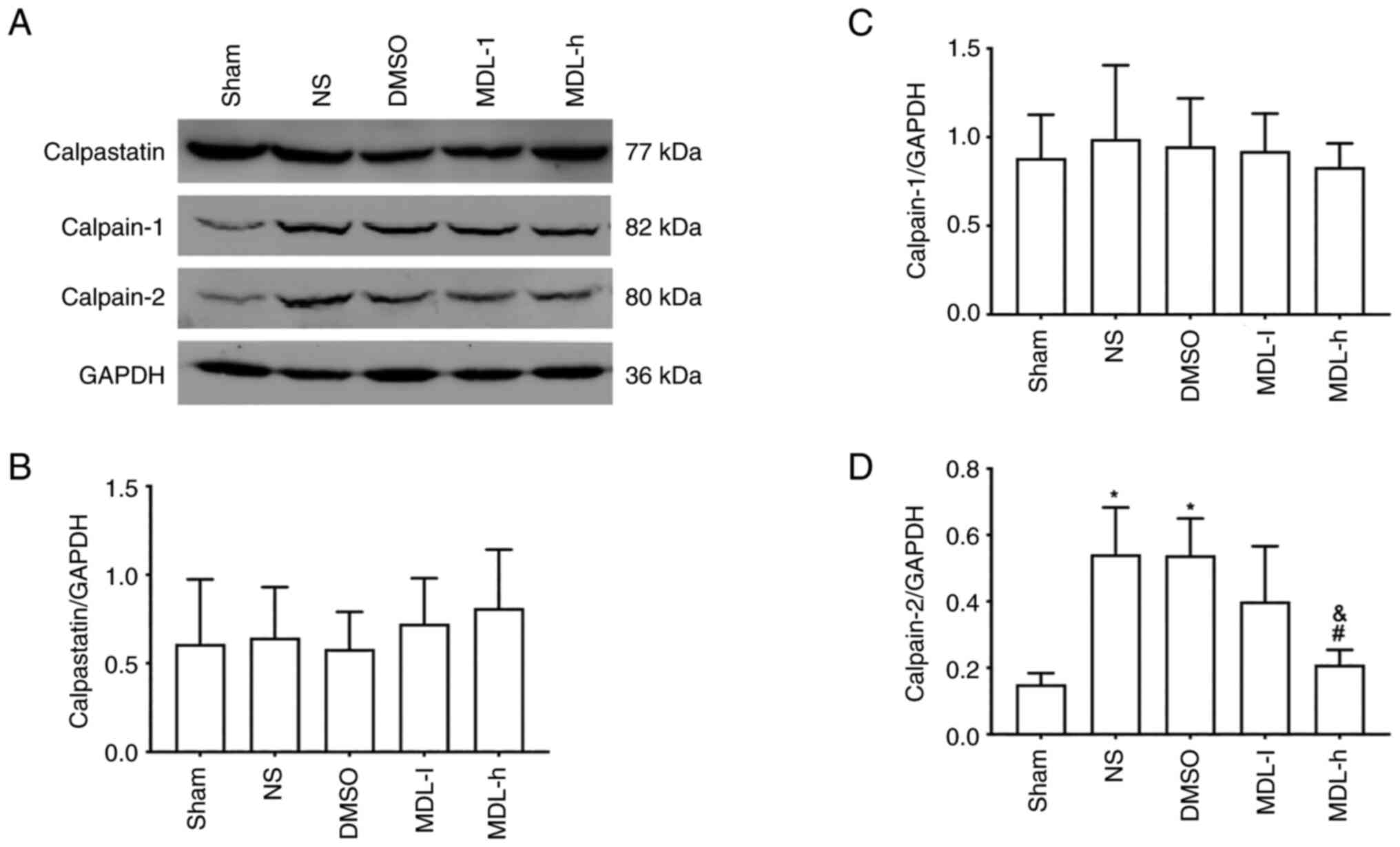
Expression of calpastatin, calpain-1
and calpain-2 in the cortex after CA and CPR
Western blotting of calpastatin, calpain-1 and
calpain-2 revealed no significant differences in the level of
calpastatin or calpain-1 among the groups (Fig. 2A-C). However, the expression of
calpain-2 in the NS and DMSO groups was significantly higher
compared with that in the sham group (P<0.05; Fig. 2A and D). The MDL-h group, which was treated
with 3.0 mg/kg of MDL28170, showed a significantly lower level of
calpain-2 compared with the NS and DMSO groups (P<0.05; Fig. 2A and D).
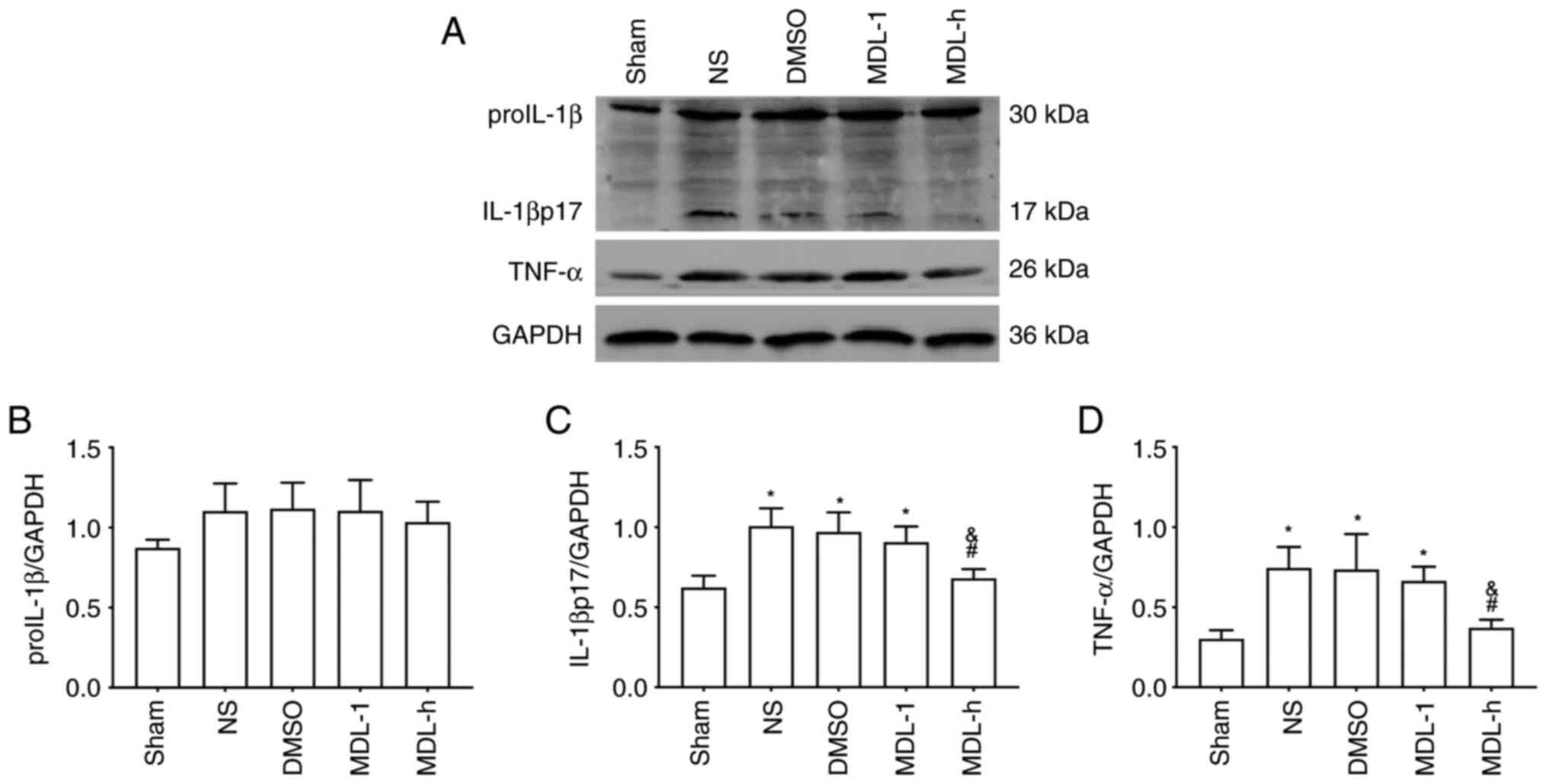
Expression of IL-1β and TNF-α in the
cortex after CA and CPR
Western blotting of ProIL-1β (precursor IL-1β)
showed no significant differences among the groups (P>0.05)
(Fig. 3A and B). The expression of IL-1βp17 (mature
IL-1β) and TNF-α in the NS, DMSO and MDL-L groups was significantly
higher compared with that in the sham group (P<0.05; Fig. 3A, C and D).
However, the expression of IL-1βp17 (mature IL-1β) and TNF-α in the
MDL-h group was significantly lower compared with that in the NS
and DMSO groups (P<0.05; Fig.
3A, C and D).
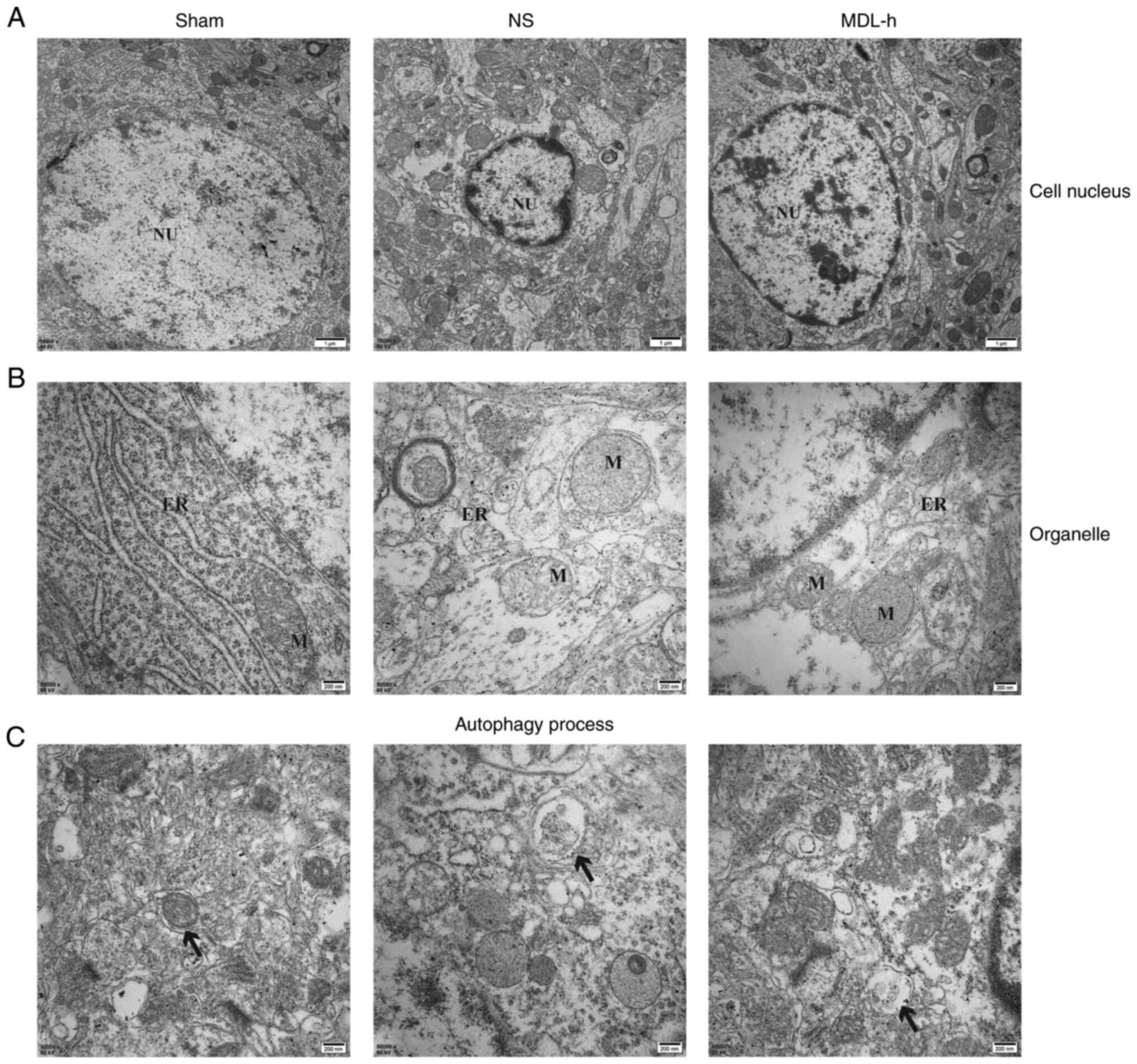
Characterization of ultrastructural
changes and autophagy by TEM
A comparison of the ultrastructural changes in the
cerebral cortex in the sham, NS and MDL-h groups, as determined by
TEM, showed the effect of MDL28170 on autophagy after the
inhibition of calpain-2. In the sham group, the nucleus was round,
and the nuclear membrane was continuous and intact. In the NS and
MDL-h groups, the size of the nucleus changed, the ER and
mitochondria were dilated, and vacuolar degeneration was observed
(Fig. 4A and B). The damage to neurons in the NS group
was more severe compared with that to neurons in the MDL-h group,
and autophagy was detected at every stage in the NS group (Fig. 4C).
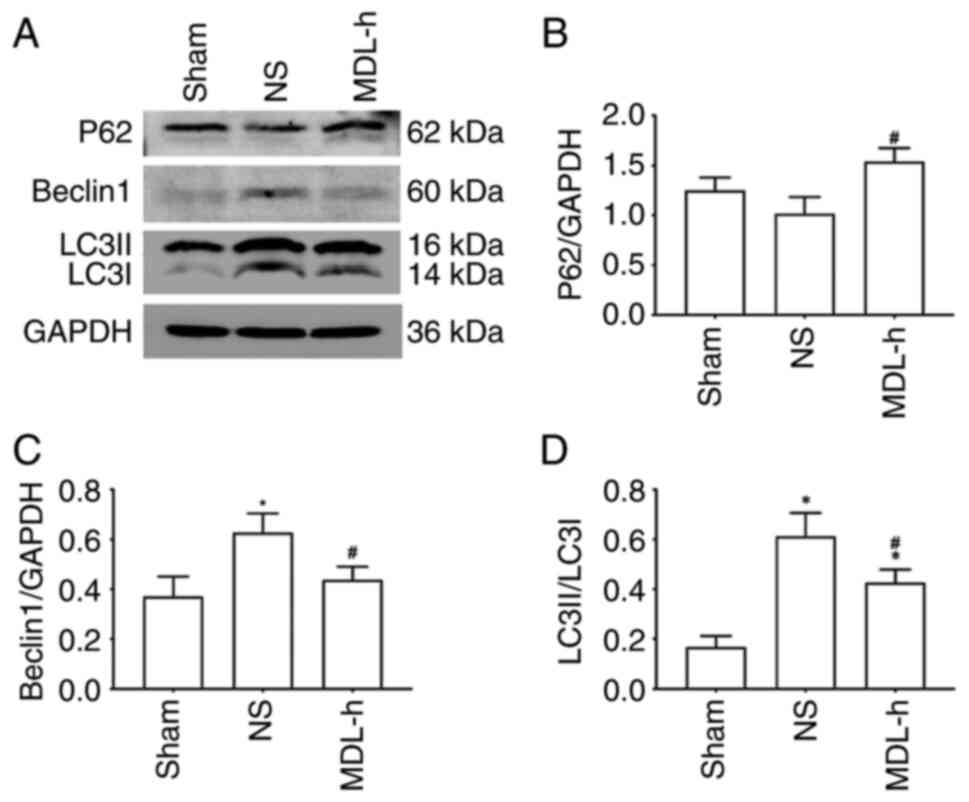
Expression of P62, beclin-1 and LC3 in
the cortex after CA and CPR
To determine the effect of MDL28170 on
autophagy-related proteins after the inhibition of calpain-2, P62,
beclin-1 and LC3 expression levels were detected in the cortex of
rats in the sham, NS and MDL-h groups by western blotting at 24 h
following CA and CPR. As shown in Fig.
5, the levels of beclin-1 and LC3I/LC3II were significantly
upregulated in the NS group compared with that in the sham group.
Additionally, the LC3I/LC3II level in rats in the MDL-h group was
significantly higher compared with that in rats in the sham group,
suggesting that autophagy increased after CIRI. When MDL28170 was
administered to rats in the MDL-h group, the levels of beclin-1 and
LC3I/LC3II decreased, whereas that of P62 increased compared with
the levels in rats in the NS group (P<0.05; Fig. 5).
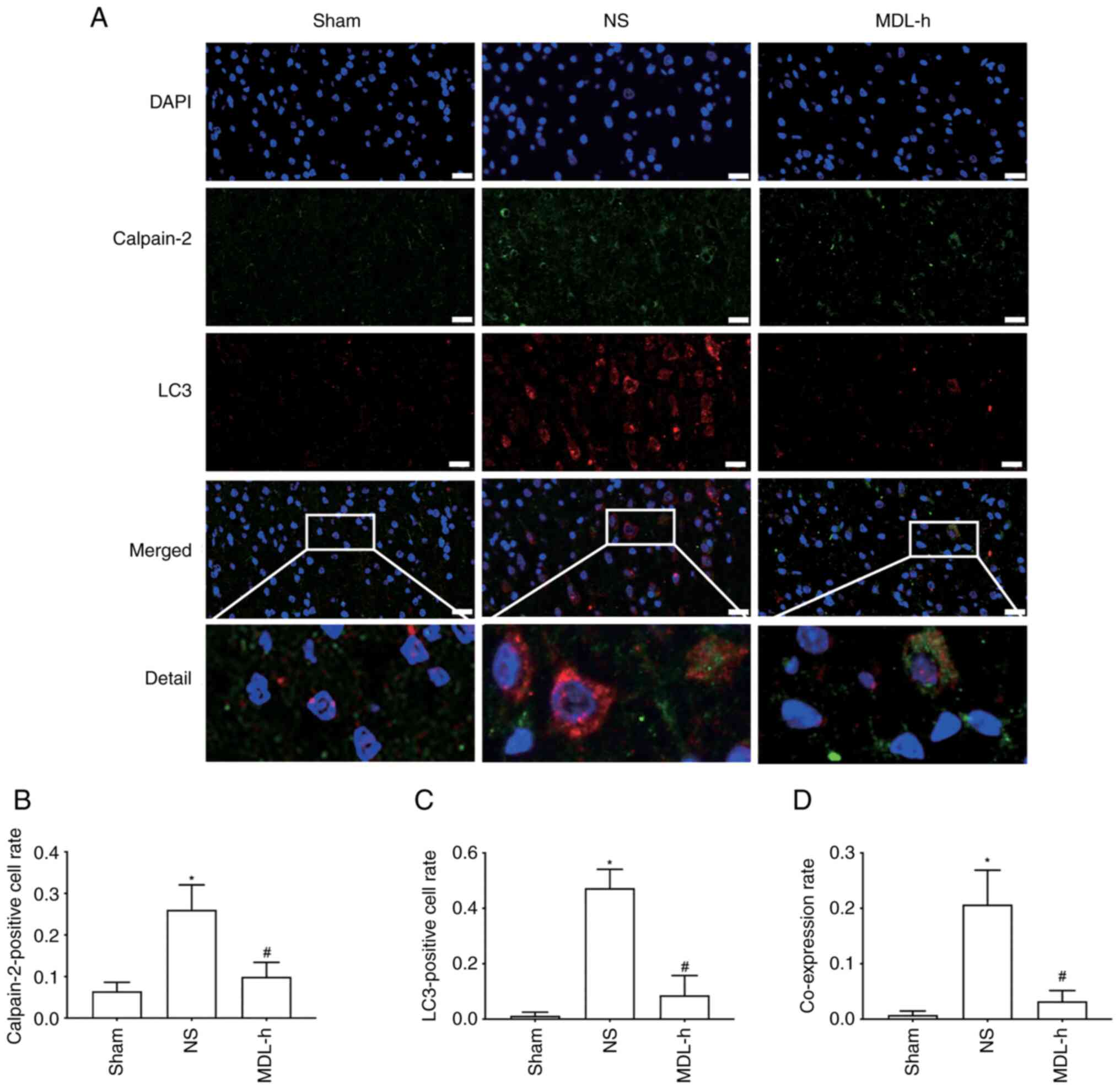
Double immunofluorescence staining of
calpain-2 and LC3 in the cortex following CA and CPR
To clarify the association between calpain-2 and
autophagy, the co-localization of calpain-2 and LC3 in the sham, NS
and MDL-h groups was examined by double immunofluorescence
staining. As shown in Fig. 6A-C,
the rate of cells positive for calpain-2 and LC3 in the NS group
was significantly higher compared with that in the sham group,
whereas the positivity rate of cells in the MDL-h group was
significantly lower compared with that in the NS group (P<0.05).
Moreover, calpain-2 and LC3 were mainly localized in the cytoplasm,
and the co-expression rate of calpain-2 and LC3 in the NS group was
higher compared with that in the sham group (P<0.05). However,
the co-expression rate of calpain-2 and LC3 in the MDL-h group was
significantly lower compared with that in the NS group (P<0.05;
Fig. 6A-D).
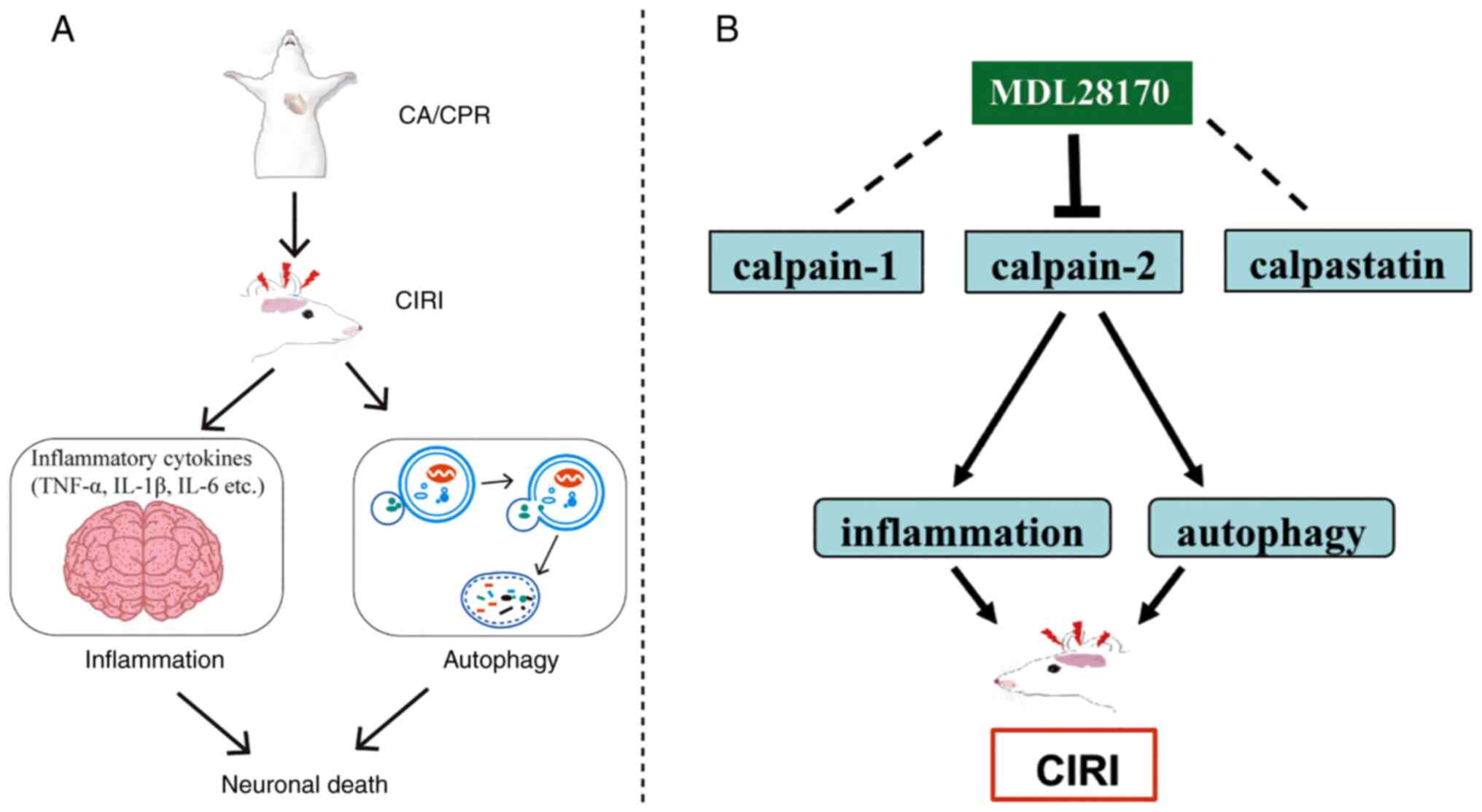
Discussion
In the present study, esophageal electrical
stimulation was used to establish a CA and CPR rat model. The
results showed that the administration of the calpain inhibitor
MDL28170 reduced pathological damage and improved the neural
function by inhibiting calpain-2 (Fig.
7); it further inhibited inflammation and reduced autophagy
level, reflecting a potential relationship between calpain-2 and
autophagy in CIRI.
MDL28170 had no significant effect on the protein
expression levels of calpain-1 and calpastatin, but had a
significant inhibitory effect on the protein expression level of
calpain-2 and a neuroprotective effect. Calpain-1 and calpain-2 are
the two most common subtypes of calpain in the brain tissue, but
there are some differences between them. Although calpain-1 and
calpain-2 share a small subunit (CAPNS1) in their structures, they
have completely different large subunits (32). According to the demand for calcium,
calpain-1 and calpain-2 are activated by intracellular calcium at
the micromole and millimole levels, respectively (33). A number of studies have shown
differences in the effects of calpain-1 and calpain-2 in brain
injury models. For example, calpain-1 induces heat shock protein 70
dysfunction and lysosomal membrane destruction, resulting in
delayed neuronal death after transient global cerebral ischemia
(34). Furthermore, another CIRI
study indicated that calpain-2, rather than calpain-1, participates
in the hydrolysis of the cytoskeleton protein and destroys tissue
structure, consequently aggravating nerve injury (35). The present study found that
MDL28170 inhibited calpain-2, rather than calpain-1, to prevent
neuronal death. In line with these results, a previous study showed
that MDL28170 attenuates neuronal abnormalities in rats with motor
nerve damage by inhibiting calpain-2(36), and a central nervous system
development study indicated a more intimate relationship between
calpain-1 and calpastatin, but calpain-2 resulted in different
effects (37). These results
suggest that calpain-2 induces neuronal injury, and inhibition of
calpain-2 without affecting calpastatin and calpain-1 has a
neuroprotective function.
TNF-α and IL-1β are common pro-inflammatory
cytokines involved in neuroinflammatory responses. TNF-α is
synthesized by macrophages, and its overexpression can lead to the
activation of neutrophils and lymphocytes as well as the
recruitment of adhesion factors, prompting the occurrence of
inflammatory reactions, and then aggravating ischemic brain tissue
damage (38). The inactive
precursor proIL-1β has a molecular weight 31 kDa, and matures to
IL-1β with a molecular weight 17 kDa due to caspase 1 during acute
injury (39). The endogenous
secretion occurs rapidly, produces an inflammatory response and
aggravates cell damage; thus, it is the mature IL-1β instead of the
proIL-1β molecule that participates in the inflammatory response
(39). The present study found
that the expression of TNF-α and mature IL-1β in the cerebral
cortex significantly increased after cardiopulmonary resuscitation,
suggesting that the inflammatory response was involved in CIRI.
MDL28170 inhibited calpain-2 in the cerebral cortex and
downregulated the expression of TNF-α and mature IL-1β, suggesting
that calpain-2 is potentially related to inflammation. Chen et
al (40) showed that there is
a close relationship between calpain and immune cells (especially
macrophages, neutrophils and lymphocytes), and that calpain can
promote the immune response when immune cells are induced.
Therefore, upon the use of MDL28170 after cerebral ischemia,
calpain-2 may be blocked at the same time to suppress the immune
response of related immune cells, thereby reducing the release of
pro-inflammatory cytokines.
The accumulation of autophagosomes and damaged
organelles in the cortex was observed using TEM, indicating the
involvement of autophagy in CIRI after CA/CPR. In the present
study, MDL28170 was found to upregulate P62 and downregulate
beclin-1 and LC3I/LC3II. Moreover, double immunofluorescence
staining indicated that the fluorescence signals produced by LC3
expression were present in the cytoplasm and partially co-expressed
with calpain-2, which revealed a potential relationship between
calpain-2 and autophagy. During CIRI, the ER and mitochondrial
membrane are damaged, resulting in a dynamic imbalance in calcium
concentration and calcium overload (41). Furthermore, the accumulation of
certain proteins, such as calpain, induces ER stress (42), which can lead to the induction of
autophagy degrade abnormally folded proteins (43-47).
Autophagy is regulated by autophagy-specific genes (ATGs),
including beclin-1, which is involved in the initial stages during
the formation of double-membrane structures and is required for
autophagosome formation (48).
Additionally, during autophagy, LC3 is converted to LC3I by ATG4,
followed by conjugation with phosphatidylethanolamine to form
LC3II. Subsequently, P62 in the cytoplasm binds to LC3II on the
membrane of the autophagosome to form a complex that is degraded by
lysosomes (49,50). Thus, both p62 and LC3 are routinely
used as biomarkers to monitor the level of autophagy (51). Autophagy acts as a cellular defense
mechanism during the early stage of injury. However, excessive
autophagy can promote cell death (52). Our previous research indicated that
the inhibition of autophagy can ameliorate nerve functioning and
play a neuroprotective role (53).
In the present study, TEM showed improvements in the ER and
mitochondria structures after MDL28170 administration, indicating
the benefit of inhibiting autophagy by blocking calpain-2 activity,
which might be related to the inhibition of calpain-2-mediated ER
stress (54,55). Similar to the present results, Li
et al (28) found that the
autophagy of hippocampal neurons induced by TNF-α are mediated by
calpain-2 activation, and selective calpain-2 inhibitors (calpain
inhibitor IV and NA101) and calpain-2 knockout can attenuate
autophagy induced by TNF-α, whereas calpain-1 knockout and
inhibition resulted in no such effect. By contrast, another study
showed that calpain-2 inhibition or calpain-2 knockout can enhance
autophagy and reduce cell death after hepatic ischemia-reperfusion
injury (56). Furthermore, Liu
et al (57) showed that
calpain-1-mediated impairment of autophagic flux contributes to
cerebral ischemia-induced neuronal damage, and that MDL28170 can
inhibit calpain-1 to reduce the expression of beclin-1 and ATG-5,
inhibit autophagosome formation and ultimately attenuating brain
damage. Therefore, the present study speculated that the
relationship between calpains and autophagy depends on the degree
of brain damage and activation, and the differences between models
and organs.
Currently, to the best of our knowledge, there are
few reports on calpains and cerebral ischemic injury induced by
cardiac arrest. The most critical innovation of the present study
is that it observed and compared the effects of different subtypes
of calpains on brain injury induced by cardiopulmonary
resuscitation in cases of cardiac arrest from different levels
(pathological changes and expression of molecular proteins).
Furthermore, the present study has a few limitations. First, in
terms of time points, although MDL28170 showed a neuroprotective
effect at 24 h after CIRI, an understanding of its long-term effect
on nerve injury would be beneficial, and this requires further
exploration at 48 or 72 h. Second, the side-effects of MDL28170 on
the liver or kidney were not observed. Third, upstream and
downstream factors of inflammation were presented via western
blotting and double immunofluorescence staining. In the future,
RT-qPCR is planned to be performed to verify the changes in gene
expression and explore the effects of different drug
concentrations. Moreover, in terms of pathophysiological
mechanisms, as calcium overload can activate calpain, the
evaluation of the effects of calcium-channel blockers would provide
additional insight into the role of calcium in the
pathophysiological mechanism.
In summary, calpain-2 activation, inflammation, and
autophagy were found to be involved in CIRI after CA and CPR.
Blocking calpain-2 activation with MDL28170 can significantly
reduce inflammation and autophagy, which plays a neuroprotective
role. These observations may help develop new strategies for
treating nerve injury in patients with CA.
Acknowledgements
Not applicable.
Funding
Funding: This work was supported by the National Natural Science
Foundation of China (grant nos. 81660312 and 81860333).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
WW and JS were responsible for the
conceptualization, methodology, analyzing and interpreting the
data, and writing of the original draft. JS was responsible for
writing, reviewing and editing and funding acquisition. XZ and CD
were responsible for acquiring, analyzing and interpreting the
data. MC was responsible for the methodology, software and funding
acquisition. LX was responsible for the conceptualization,
methodology, reviewing and funding acquisition. WW and LX confirm
the authenticity of all the raw data. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
All animal procedures were conducted in compliance
with the Guide for the Care and Use of Experimental Animals. The
study was approved by the Animal Ethics Committee of Guangxi
Medical University (Nanning, China; approval no. 20190915).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Andersen LW, Holmberg MJ, Berg KM, Donnino
MW and Granfeldt A: In-hospital cardiac arrest: A review. JAMA.
321:1200–1210. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sandroni C, D'Arrigo S and Nolan JP:
Prognostication after cardiac arrest. Crit Care.
22(150)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Moore JC, Bartos JA, Matsuura TR and
Yannopoulos D: The future is now: Neuroprotection during
cardiopulmonary resuscitation. Curr Opin Crit Care. 23:215–222.
2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Goodfellow MJ, Borcar A, Proctor JL, Greco
T, Rosenthal RE and Fiskum G: Transcriptional activation of
antioxidant gene expression by Nrf2 protects against mitochondrial
dysfunction and neuronal death associated with acute and chronic
neurodegeneration. Exp Neurol. 328(113247)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Chen H, Yoshioka H, Kim GS, Jung JE, Okami
N, Sakata H, Maier CM, Narasimhan P, Goeders CE and Chan PH:
Oxidative stress in ischemic brain damage: Mechanisms of cell death
and potential molecular targets for neuroprotection. Antioxid Redox
Signal. 14:1505–1517. 2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Liu J, Liu MC and Wang KKW: Calpain in the
CNS: from synaptic function to neurotoxicity. Sci Signal.
1(re1)2008.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Curcio M, Salazar IL, Mele M, Canzoniero
LMT and Duarte CB: Calpains and neuronal damage in the ischemic
brain: The Swiss knife in synaptic injury. Prog Neurobiol.
143:1–35. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Martinez JA, Zhang Z, Svetlov SI, Hayes
RL, Wang KK and Larner SF: Calpain and caspase processing of
caspase-12 contribute to the ER stress-induced cell death pathway
in differentiated PC12 cells. Apoptosis. 15:1480–1493.
2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sanganalmath SK, Gopal P, Parker JR, Downs
RK, Parker JC Jr and Dawn B: Global cerebral ischemia due to
circulatory arrest: Insights into cellular pathophysiology and
diagnostic modalities. Mol Cell Biochem. 426:111–127.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Dókus LE, Yousef M and Bánóczi Z:
Modulators of calpain activity: Inhibitors and activators as
potential drugs. Expert Opin Drug Discov. 15:471–486.
2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Baudry M and Bi X: Calpain-1 and
calpain-2: The yin and yang of synaptic plasticity and
neurodegeneration. Trends Neurosci. 39:235–245. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Cheng SY, Wang SC, Lei M, Wang Z and Xiong
K: Regulatory role of calpain in neuronal death. Neural Regen Res.
13:556–562. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang Y, Bi X and Baudry M: Calpain-2 as a
therapeutic target for acute neuronal injury. Expert Opin Ther
Targets. 22:19–29. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Jiao W, McDonald DQ, Coxon JM and Parker
EJ: Molecular modeling studies of peptide inhibitors highlight the
importance of conformational prearrangement for inhibition of
calpain. Biochemistry. 49:5533–5539. 2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Betts R, Weinsheimer S, Blouse GE and
Anagli J: Structural determinants of the calpain inhibitory
activity of calpastatin peptide B27-WT. J Biol Chem. 278:7800–7809.
2003.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Potz BA, Abid MR and Sellke FW: Role of
calpain in pathogenesis of human disease processes. J Nat Sci.
2(e218)2016.PubMed/NCBI
|
|
17
|
Hu J, Chen L, Huang X, Wu K, Ding S, Wang
W, Wang B, Smith C, Ren C, Ni H, et al: Calpain inhibitor MDL28170
improves the transplantation-mediated therapeutic effect of bone
marrow-derived mesenchymal stem cells following traumatic brain
injury. Stem Cell Res Ther. 10(96)2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Thompson SN, Carrico KM, Mustafa AG, Bains
M and Hall ED: A pharmacological analysis of the neuroprotective
efficacy of the brain- and cell-permeable calpain inhibitor
MDL-28170 in the mouse controlled cortical impact traumatic brain
injury model. J Neurotrauma. 27:2233–2243. 2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Chen LN, Yan B, Chen DP and Yao YJ:
Protective effect of calpain inhibitor-3 on hypoxic-ischemic brain
damage of neonatal rats. Zhonghua Er Ke Za Zhi. 46:13–17.
2008.PubMed/NCBI(In Chinese).
|
|
20
|
Li PA, Howlett W, He QP, Miyashita H,
Siddiqui M and Shuaib A: Postischemic treatment with calpain
inhibitor MDL 28170 ameliorates brain damage in a gerbil model of
global ischemia. Neurosci Lett. 247:17–20. 1998.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Ravindran S and Kurian GA: Eventual
analysis of global cerebral ischemia-reperfusion injury in rat
brain: A paradigm of a shift in stress and its influence on
cognitive functions. Cell Stress Chaperones. 24:581–594.
2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Tuttolomondo A, Di Raimondo D, Pecoraro R,
Arnao V, Pinto A and Licata G: Inflammation in ischemic stroke
subtypes. Curr Pharm Des. 18:4289–4310. 2012.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Tao XG, Shi JH, Hao SY, Chen XT and Liu
BY: Protective effects of calpain inhibition on neurovascular unit
injury through downregulating nuclear factor-κB-related
inflammation during traumatic brain injury in mice. Chin Med J
(Engl). 130:187–198. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Wang WY, Xie L, Zou XS, Li N, Yang YG, Wu
ZJ, Tian XY, Zhao GY and Chen MH: Inhibition of extracellular
signal-regulated kinase/calpain-2 pathway reduces neuroinflammation
and necroptosis after cerebral ischemia-reperfusion injury in a rat
model of cardiac arrest. Int Immunopharmacol.
93(107377)2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Edinger AL and Thompson CB: Death by
design: Apoptosis, necrosis and autophagy. Curr Opin Cell Biol.
16:663–669. 2004.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Ashrafi G and Schwarz TL: The pathways of
mitophagy for quality control and clearance of mitochondria. Cell
Death Differ. 20:31–42. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zheng JH, Xie L, Li N, Fu ZY, Tan XF, Tao
R, Qin T and Chen MH: PD98059 protects the brain against
mitochondrial-mediated apoptosis and autophagy in a cardiac arrest
rat model. Life Sci. 232(116618)2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Li Y, He Z, Lv H, Chen W and Chen J:
Calpain-2 plays a pivotal role in the inhibitory effects of
propofol against TNF-α-induced autophagy in mouse hippocampal
neurons. J Cell Mol Med. 24:9287–9299. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Chen MH, Liu TW, Xie L, Song FQ, He T,
Zeng ZY and Mo SR: Ventricular fibrillation induced by
transoesophageal cardiac pacing: a new model of cardiac arrest in
rats. Resuscitation. 74:546–551. 2007.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Du PR, Lu HT, Lin XX, Wang LF, Wang YX, Gu
XM, Bai XZ, Tao K and Zhou JJ: Calpain inhibition ameliorates scald
burn-induced acute lung injury in rats. Burns Trauma.
6(28)2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Jia X, Koenig MA, Shin HC, Zhen G, Pardo
CA, Hanley DF, Thakor NV and Geocadin RG: Improving neurological
outcomes post-cardiac arrest in a rat model: Immediate hypothermia
and quantitative EEG monitoring. Resuscitation. 76:431–442.
2008.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Cataldo F, Peche LY, Klaric E, Brancolini
C, Myers MP, Demarchi F and Schneider C: CAPNS1 regulates USP1
stability and maintenance of genome integrity. Mol Cell Biol.
33:2485–2496. 2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zheng P, Chen X, Xie J, Chen X, Lin S, Ye
L, Chen L, Lin J, Yu X and Zheng M: Capn4 is induced by and
required for Epstein-Barr virus latent membrane protein 1 promotion
of nasopharyngeal carcinoma metastasis through ERK/AP-1 signaling.
Cancer Sci. 111:72–83. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Zhu H, Yoshimoto T, Imajo-Ohmi S,
Dazortsava M, Mathivanan A and Yamashima T: Why are hippocampal CA1
neurons vulnerable but motor cortex neurons resistant to transient
ischemia? J Neurochem. 120:574–585. 2012.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Fukuda S, Harada K, Kunimatsu M, Sakabe T
and Yoshida K: Postischemic reperfusion induces alpha-fodrin
proteolysis by m-calpain in the synaptosome and nucleus in rat
brain. J Neurochem. 70:2526–2532. 1998.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zang Y, Chen SX, Liao GJ, Zhu HQ, Wei XH,
Cui Y, Na XD, Pang RP, Xin WJ, Zhou LJ and Liu XG: Calpain-2
contributes to neuropathic pain following motor nerve injury via
up-regulating interleukin-6 in DRG neurons. Brain Behav Immun.
44:37–47. 2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Li Y, Bondada V, Joshi A and Geddes JW:
Calpain 1 and calpastatin expression is developmentally regulated
in rat brain. Exp Neurol. 220:316–319. 2009.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xiong XY, Liu L and Yang QW: Functions and
mechanisms of microglia/macrophages in neuroinflammation and
neurogenesis after stroke. Prog Neurobiol. 142:23–44.
2016.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Lopez-Castejon G and Brough D:
Understanding the mechanism of IL-1β secretion. Cytokine Growth
Factor Rev. 22:189–195. 2011.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Chen Y, Su Z and Liu F: Effects of
functionally diverse calpain system on immune cells. Immunol Res.
69:8–17. 2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Guo MM, Qu SB, Lu HL, Wang WB, He ML, Su
JL, Chen J and Wang Y: Biochanin A alleviates cerebral
ischemia/reperfusion injury by suppressing endoplasmic reticulum
stress-induced apoptosis and p38MAPK signaling pathway in vivo and
in vitro. Front Endocrinol (Lausanne). 12(646720)2021.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Thompson J, Maceyka M and Chen Q:
Targeting ER stress and calpain activation to reverse age-dependent
mitochondrial damage in the heart. Mech Ageing Dev.
192(111380)2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Li W, Zhu J, Dou J, She H, Tao K, Xu H,
Yang Q and Mao Z: Phosphorylation of LAMP2A by p38 MAPK couples ER
stress to chaperone-mediated autophagy. Nat Commun.
8(1763)2017.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Lépine S, Allegood JC, Edmonds Y, Milstien
S and Spiegel S: Autophagy induced by deficiency of
sphingosine-1-phosphate phosphohydrolase 1 is switched to apoptosis
by calpain-mediated autophagy-related gene 5 (Atg5) cleavage. J
Biol Chem. 286:44380–44390. 2011.PubMed/NCBI View Article : Google Scholar
|
|
45
|
So KY, Lee BH and Oh SH: The critical role
of autophagy in cadmium-induced immunosuppression regulated by
endoplasmic reticulum stress-mediated calpain activation in
RAW264.7 mouse monocytes. Toxicology. 393:15–25. 2018.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Demarchi F, Bertoli C, Copetti T, Tanida
I, Brancolini C, Eskelinen EL and Schneider C: Calpain is required
for macroautophagy in mammalian cells. J Cell Biol. 175:595–605.
2006.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Li XY, Meng L, Wang F, Hu XJ and Yu YC:
Sodium fluoride induces apoptosis and autophagy via the endoplasmic
reticulum stress pathway in MC3T3-E1 osteoblastic cells. Mol Cell
Biochem. 454:77–85. 2019.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Qu X, Yu J, Bhagat G, Furuya N, Hibshoosh
H, Troxel A, Rosen J, Eskelinen EL, Mizushima N, Ohsumi Y, et al:
Promotion of tumorigenesis by heterozygous disruption of the beclin
1 autophagy gene. J Clin Invest. 112:1809–1820. 2003.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Glick D, Barth S and Macleod KF:
Autophagy: Cellular and molecular mechanisms. J Pathol. 221:3–12.
2010.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Kaur J and Debnath J: Autophagy at the
crossroads of catabolism and anabolism. Nat Rev Mol Cell Biol.
16:461–472. 2015.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Klionsky DJ, Abdalla FC, Abeliovich H,
Abraham RT, Acevedo-Arozena A, Adeli K, Agholme L, Agnello M,
Agostinis P, Aguirre-Ghiso JA, et al: Guidelines for the use and
interpretation of assays for monitoring autophagy. Autophagy.
8:445–544. 2012.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Gustafsson AB and Gottlieb RA: Autophagy
in ischemic heart disease. Circ Res. 104:150–158. 2009.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Nguyen Thi PAN, Chen MH, Li N, Zhuo XJ and
Xie L: PD98059 protects brain against cells death resulting from
ROS/ERK activation in a cardiac arrest rat model. Oxid Med Cell
Longev. 2016(3723762)2016.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Xie RJ, Hu XX, Zheng L, Cai S, Chen YS,
Yang Y, Yang T, Han B and Yang Q: Calpain-2 activity promotes
aberrant endoplasmic reticulum stress-related apoptosis in
hepatocytes. World J Gastroenterol. 26:1450–1462. 2020.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Ly LD, Xu S, Choi SK, Ha CM, Thoudam T,
Cha SK, Wiederkehr A, Wollheim CB, Lee IK and Park KS: Oxidative
stress and calcium dysregulation by palmitate in type 2 diabetes.
Exp Mol Med. 49(e291)2017.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Zhao Q, Guo Z, Deng W, Fu S, Zhang C, Chen
M, Ju W, Wang D and He X: Calpain 2-mediated autophagy defect
increases susceptibility of fatty livers to ischemia-reperfusion
injury. Cell Death Dis. 7(e2186)2016.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Liu Y, Che X, Zhang H, Fu X, Yao Y, Luo J,
Yang Y, Cai R, Yu X, Yang J and Zhou MS: . CAPN1
(Calpain1)-mediated impairment of autophagic flux contributes to
cerebral ischemia-induced neuronal damage. Stroke. 52:1809–1821.
2021.PubMed/NCBI View Article : Google Scholar
|