Introduction
Acute kidney injury (AKI) is a common clinical
syndrome characterized by rapid-onset kidney function damage, which
leads to chronic and end-stage kidney disease (1). Renal ischemia/reperfusion injury
(RIRI) is the primary pathogenic mechanism underlying AKI in which
the renal blood supply is constrained and then restored (2). Reactive oxygen species (ROS)
overproduction, neutrophil-endothelial cell adhesion and
alterations to the microenvironment are the primary pathological
mechanisms of RIRI (3,4). Oxidative stress and ferroptosis play
crucial roles in RIRI (5-8).
Ferroptosis is a type of cell death that occurs due
to iron-dependent lipid peroxidation (9). Ferroptosis was first proposed by
Dixon et al (10) to
describe a form of programmed cell death dependent on cellular iron
ions that occurs after treatment with the small molecule compound
erastin. Erastin inhibits cystine uptake that leads to depletion of
reduced glutathione (GSH), hindering the antioxidant capacity of
glutathione peroxidase 4 (GPX4), causing an imbalance of
intracellular redox levels and inability to eliminate lipid
peroxidation products (10). Tuo
et al (5) showed that
ferroptosis inhibitors ferrostatin-1 and liproxstatin-1 notably
decrease the infarction volume caused by cerebral IRI in rats by
limiting lipid peroxidation and inhibiting ferroptosis. In
addition, a previous study confirmed the role of ferroptosis in the
pathogenesis of IRI-mediated kidney injury (11). Tao et al (12) confirmed that decreased lipid
peroxidation and ferroptosis inhibition can reduce RIRI.
Various fruits, vegetables and plant-based foods
such as quercetin, citrus, soy and tea contain flavonoids,
compounds that exhibit renal-protective effects by chelating
redox-active metals, which inhibit lipid peroxidation (13). Xanthohumol (XN), the primary active
ingredient in hops, is a flavonoid with various biological
properties, including antioxidation, antitumor and antibacterial
properties (14,15). Previous studies have shown that XN
promotes neuronal differentiation and neurite growth and serves a
neuroprotective role against ischemic stroke in rats (16,17).
To the best of our knowledge, however, the renal protective effect
of XN has not been reported. The present study aimed to investigate
whether XN alleviates IR-induced ferroptosis and its underlying
mechanisms.
Materials and methods
Chemicals
XN was purchased from Shanghai Macklin Biochemical
Co., Ltd. (cat. no. 6754-58-1). Erastin was purchased from
MedChemExpress (cat. no. HY-15763).
Animals
A total of 30 male Sprague-Dawley rats (age, 6 weeks
old; weight, 200-220 g) were obtained from the Animal Center of
Wuhan University, Wuhan, China. The rats were maintained with
standard feeding conditions including food and water ad
libitum, a 12 h light/dark cycle, 20-25˚C and 50-65%
humidity.
RIRI model
Rats were fasted for 8 h before surgery. All rats
were randomly divided into five groups (six rats per group): Sham,
IR, IR + XN, IR + erastin or IR +XN + erastin groups. To explore
the effect of XN on RIRI, XN group rats were intraperitoneally
injected with 0.4 mg/kg XN (16)
10 min before IR. The erastin group rats were intraperitoneally
injected with 10 mg/kg erastin (18) 10 min before IR. A total of 50 mg/kg
pentobarbital sodium was injected intraperitoneally to induce
anesthesia before surgery. The limbs of the rats were fixed and the
surgical skin area was disinfected. The renal pedicle was exposed
via abdominal incision, and bilateral renal arteries were clamped
for 45 min. The hemostatic forceps were loosened during reperfusion
and the color change of the kidney was observed. After blood flow
returned to normal, the incision was sutured. The sham group only
underwent abdominal incision without clamping the renal pedicle.
After 24 h, rats were humanely euthanized under anesthesia by
intraperitoneal injection with 1% pentobarbital sodium (150 mg/kg).
Rat kidneys were harvested and serum samples were collected.
Cell culture
HK-2 cells, a human renal tubular epithelial cell
line, were obtained from the National Collection of Authenticated
Cell Cultures (Beijing, China). HK-2 cells were cultured in DMEM
(cat. no. 12430112) with 1% penicillin-streptomycin solution (cat.
no. 15140122) and 10% fetal bovine serum (cat. no. 16140089; all
Gibco; Thermo Fisher Scientific, Inc.). Cells were cultured in a
37˚C incubator with 5% CO2. All cells were randomly
divided into five groups: Control, IR, IR + XN, IR + erastin or IR
+ XN + erastin.
Hypoxia-reoxygenation (HR) model
Following starvation in serum-free DMEM for 12 h at
37˚C, HK-2 cells were transferred to a 37˚C tri-gas incubator
containing 95% N2 to induce hypoxia for 8 h.
Treatment protocols in vitro
To investigate the effect of ferroptosis and XN,
HK-2 cells were incubated with 10 µM XN (19) or erastin (20) at the beginning of reoxygenation.
HK-2 cells were transfected with either control small interfering
RNA (si-con; GenePharma Co., Ltd.) or Nrf2 siRNA (si-Nrf2;
GenePharma Co., Ltd.) for 48 h before IR to evaluate the effect of
Nrf2. The HK-2 cells were transfected with si-Nrf2
(5'-GCACCUUAUAUCUCGAAGUTT-3') or non-targeting scrambled (used as
si-con) oligonucleotides (5'-UUCUCCGAACGUGUCACGUTT-3') using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the instructions provided by the
manufacturer. The cells were subjected to treatment 24 h after
transfection.
Cell viability assay
Cell viability assay was performed using Cell
Counting Kit-8 (CCK-8; cat. no. C0038; Beyotime Institute of
Biotechnology). HK-2 cells were transferred into 96-well plates at
a density of 1x104 cells/well and cultured for 12 h in a
37˚C thermostatic incubator. Following treatment with either XN or
erastin for 4 h, 10 µl CCK-8 solution was added to the 96-well
plates and cells were incubated for 1 h. A microplate reader
(PerkinElmer, Inc.) was used to measure absorbance at 450 nm.
Renal function and histopathological
analysis
Renal function was measured using serum creatinine
(Scr) and blood urea nitrogen (BUN). Serum was separated by
centrifugation at 3,000 x g and at 4˚C, and Scr and BUN levels were
determined by staff at the Clinical Laboratory of Renmin Hospital
of Wuhan University, who were blinded to the treatments given.
Blood samples were stored at -80˚C for analysis. Rat kidney samples
were collected and fixed in 4% formaldehyde for 24 h at room
temperature, dehydrated with 95% ethanol and embedded in 55˚C
paraffin, and then cut into 4-µm sections. The sections were
stained with hematoxylin and eosin (H&E) to assess the
histopathological changes. The slides were stained with
hematoxylin, and incubated at room temperature for 4 min. The
slides were incubated in water at 25˚C for 30 min. The slides were
dehydrated in 100% ethanol for 10 min three times, and were stained
with eosin at room temperature for 4 min. The slides were
dehydrated in 100% ethanol for 10 min three times, and mounted
under cover slips. The slides were observed under x200
magnification using a light microscope (Olympus Corporation). A
total of five different fields of view were observed. The severity
of RIRI was assessed according to Paller's score (21). The histopathological changes of
kidney tissue were analyzed and graded as follows: 0, no damage; 1,
mild damage, including rounded epithelial cells and dilated tubular
lumen; 2, moderate damage, including substantially dilated lumen,
flattened epithelial cells and nuclear staining loss; and 3, severe
damage, including destroyed tubules with no nuclear staining of
epithelial cells. The evaluation of histological data was performed
by two independent observers, blinded to the experimental
group.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted using TRIzol®
reagent (Vazyme Biotech Co., Ltd.) in accordance with the
manufacturer's instructions. After quantification with a Nanodrop
spectrophotometer (Thermo Fisher Scientific, Inc.), 1 µg RNA sample
was reverse transcribed into cDNA (42˚C, 2 min; 37˚C, 15 min; 85˚C,
5 sec) using a First Strand cDNA Synthesis kit (Thermo Fisher
Scientific, Inc.). Subsequently, the RT-qPCR assay was performed
based on the descriptions of a SYBR Premix Ex Taq II kit (Vazyme
Biotech Co., Ltd.) in an ABI PRISM 7700 Sequence Detection System
(Applied Biosystems; Thermo Fisher Scientific, Inc.). GAPDH was
used as the internal reference to normalize mRNA expression levels.
The calculation of mRNA levels was executed using the
2-∆∆Cq method (22). The qPCR thermocycling conditions
were as follows: Initial denaturation at 95˚C for 30 sec, followed
by annealing and elongation for 39 cycles of 95˚C for 15 sec, 60˚C
for 30 sec and 72˚C for 30 sec, and a final extension at 72˚C for 2
min. The primer sequences used in the present study are shown in
Table I.
 | Table ISequences of primer pairs for
quantitative PCR. |
Table I
Sequences of primer pairs for
quantitative PCR.
| Primer | Forward
(5'-3') | Reverse
(5'-3') |
|---|
| Nrf2 |
CCCAGCACATCCAGACAGAC |
TATCCAGGGCAAGCGACTC |
| Heme
oxygenase-1 |
CCCTTCCTGTGTCTTCCTTTG |
ACAGCCGCCTCTACCGACCACA |
| GAPDH |
GACATGCCGCCTGGAGAAAC |
AGCCCAGGATGCCCTTTAGT |
Western blotting
The cells or tissues were lysed in ice-cold modified
RIPA lysis buffer (50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 50 mM NaF,
0.5% deoxycholic acid, 1% NP-40, 1 mM sodium orthovanadate and 0.1%
SDS). The insoluble material was then removed by centrifugation at
12,000 x g for 15 min at 4˚C. The protein concentration of each
sample was measured using a BCA kit (cat. no. A045-4-2; Nanjing
Jiancheng Bioengineering Institution). Total protein (10 µg/lane)
was separated using SDS-PAGE (10% gels) and transferred to a PVDF
membrane. Membranes were incubated with primary antibodies against
GPX4 (1:1,000; cat. no. 67763-1-Ig; Proteintech Group, Inc.),
long-chain acyl-CoA synthetase (ACSL4; 1:1,000; cat. no.
22401-1-AP; Proteintech Group, Inc.), Nrf2 (1:1,000; cat. no.
80593-1-RR; Proteintech Group, Inc.), heme oxygenase (HO)-1
(1:1,000; cat. no. 10701-1-AP; Proteintech Group, Inc.) or GAPDH
(1:1,000; cat. no. 60004-1-Ig; Proteintech Group, Inc.) overnight
at 4˚C following blocking with 5% BSA for 2 h at room temperature
(Beijing Solarbio Science & Technology Co., Ltd.). Then,
membranes were washed in TBS containing 0.1% Tween-20 and incubated
for 2 h at room temperature with corresponding secondary
antibodies. The secondary antibodies used were HRP-conjugated
anti-rabbit (1:1,000; cat. no. sc-2370) and anti-mouse (1:1,000;
cat. no. sc-516102) (Santa Cruz Biotechnology, Inc.) were used. The
membranes were visualized using an ECL kit (cat. no. W028-2-1;
Nanjing Jiancheng Bioengineering Institution) and band intensities
were quantified using ImageJ (version 1.8.0; National Institutes of
Health).
Detection of oxidative stress
indicators
The kidney tissues or HK-2 cells were homogenized
and centrifuged at 4˚C for 10 min at 10,000 x g. Then, the
activities of superoxide dismutase (SOD), malondialdehyde (MDA) and
GSH were determined using commercial kits (cat. nos. S0131S, S0101S
and S0053, respectively; Beyotime Institute of Biotechnology) based
on the manufacturer's protocols and the OD values were determined
at 560 (SOD), 412 (GSH), 532 and 600 (MDA) nm, respectively, using
a microplate reader.
ROS levels in kidney tissues
Dihydroethidium (DHE) fluorescence probe (cat. no.
D7008; Sigma-Aldrich; Merck KGaA) was used to evaluate the level of
ROS in the kidney tissues. Kidney tissues were placed in optimal
cutting temperature embedding medium (Servicebio, Inc.), frozen for
3 min at -20˚C and cut into 30 µm-thick sections by a freezing
microtome. The DHE probe was dissolved in dimethyl sulfoxide and
then diluted with phosphate-buffered saline to a working solution
of 10 µmol/l. DHE probe working solution (100 µl) was added onto
the surface of the section, which was then promptly placed in a
light-tight wet box for 30 min at 37˚C, in order to oxidize DHE and
generate ethidium bromide (EB). The EB binds to DNA in the cell
nucleus to produce red fluorescence under UV light. Subsequently,
unreacted probes were washed away and the sample was observed under
a fluorescence microscope. Under excitation at a wavelength of 490
nm and an emission wavelength of 520 nm, the signal intensity of
the red fluorescence was observed. Microscopic imaging analysis was
adopted for image acquisition. Image-Pro Plus 6.0 (Media
Cybernetics, Inc.) was used for the quantitative analysis of the
fluorescence intensity of the image.
ROS levels of HK-2 cells
Intracellular ROS production was assessed using a
fluorescent probe, DCFH-DA, using flow cytometric analysis, as
previously described (23).
Detection of ROS was based on the fact that intracellular ROS are
able to oxidize DCFH, yielding the fluorescent product,
2',7'-dichlorofluorescein (DCF). Following an incubation of the
cells with 0, 15, or 30 mM formate at 37˚C for 24 h, the
supernatant was removed (300 x g at room temperature for 10 min),
and the cells were washed with PBS three times. The cells were
subsequently harvested and suspended in PBS. DCFH-DA (10 µM final
concentration) probe (cat. no. D6883; Sigma-Aldrich; Merck KGaA)
was added and the mixture was incubated at 37˚C for 15 min.
Finally, ROS generation was measured, according to the fluorescence
intensity (FL-1; 530 nm) of 104 cells using a flow
cytometer (FACSCalibur; BD Biosciences).
Statistical analysis
GraphPad (version 8.0.1; Dotmatics) was used to
analyze experimental data. Data are presented as mean ± the
standard deviation from three independent experiments. The
comparisons between multiple groups were conducted by one-way ANOVA
followed by Tukey's post hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
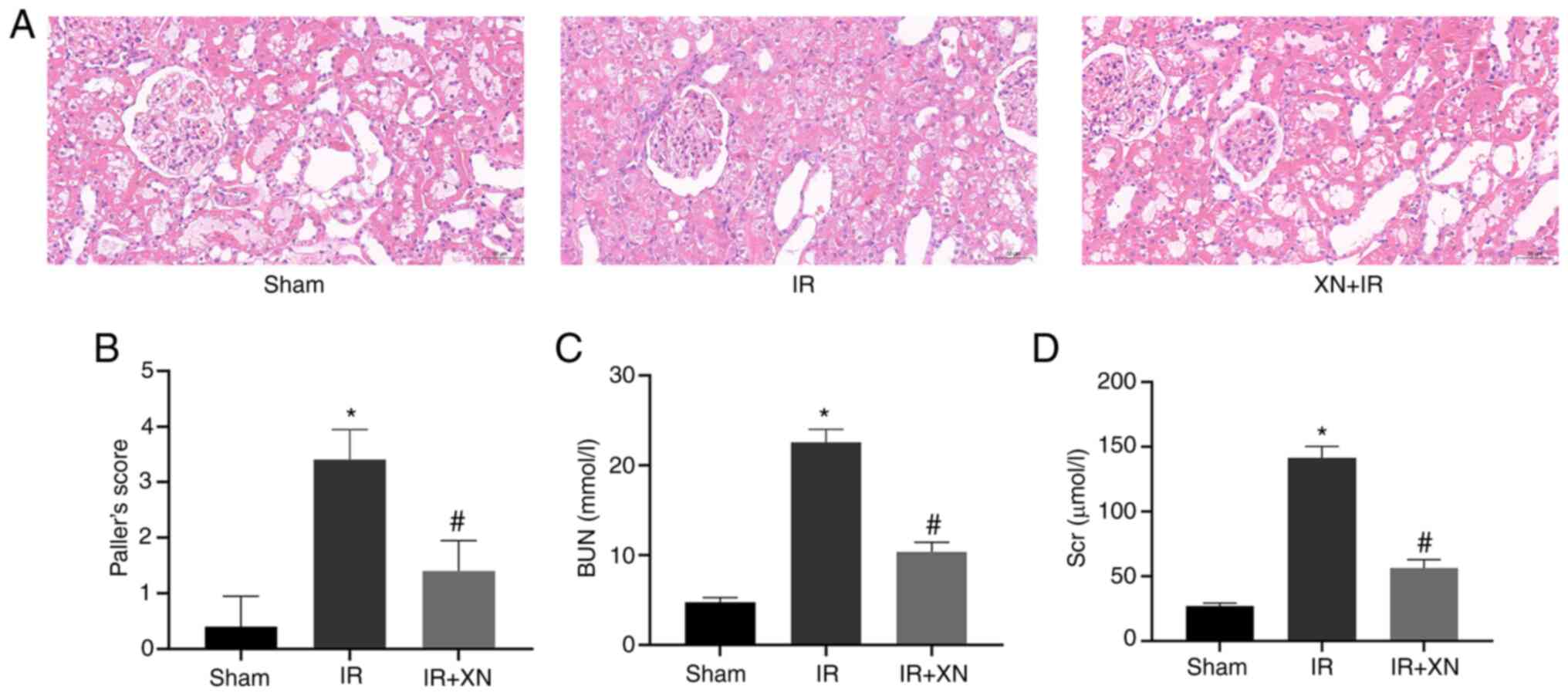
XN significantly decreases RIRI
In the sham group, the renal tubular lumen was not
dilated, glomerular structure was intact, and the renal tubular
epithelial cells were healthy. However, in the IR group, there was
a noticeable glomerular lesion, renal tubular epithelial cells were
flat, the renal tubular lumen was occluded and there were instances
of bleeding and cell abscission. These changes indicated diffuse
damage. By contrast, the XN + IR group showed decreased glomerular
damage, although the renal tubular lumen was still partially
occluded and the morphology of renal tubular epithelial cells was
flattened with loss of the brush border (Fig. 1A and B).
The concentrations of BUN and SCr in the IR group
were significantly higher than those in the sham group, while
levels of BUN and SCr in the XN + IR group were lower than those in
the IR group (Fig. 1C and D).
XN attenuates ferroptosis during
RIRI
To determine whether the protection against RIRI by
XN is related to ferroptosis, the production of ROS and the levels
of SOD, MDA and GSH were measured. IR significantly increased ROS
and MDA levels and decreased the activity of SOD and GSH, while XN
inhibited IR-induced oxidative damage (Fig. 2A-E). The expression of the
ferroptosis-related proteins GPX4 and ACSL4 was determined. The
expression of the pro-ferroptosis protein ACSL4 increased in the IR
group, while expression of the anti-ferroptosis protein GPX4
decreased in the IR group. However, XN partially reversed these
changes and erastin blocked the inhibitory effect of XN on
ferroptosis (Fig. 2F-H). In
general, XN attenuated ferroptosis during RIRI.
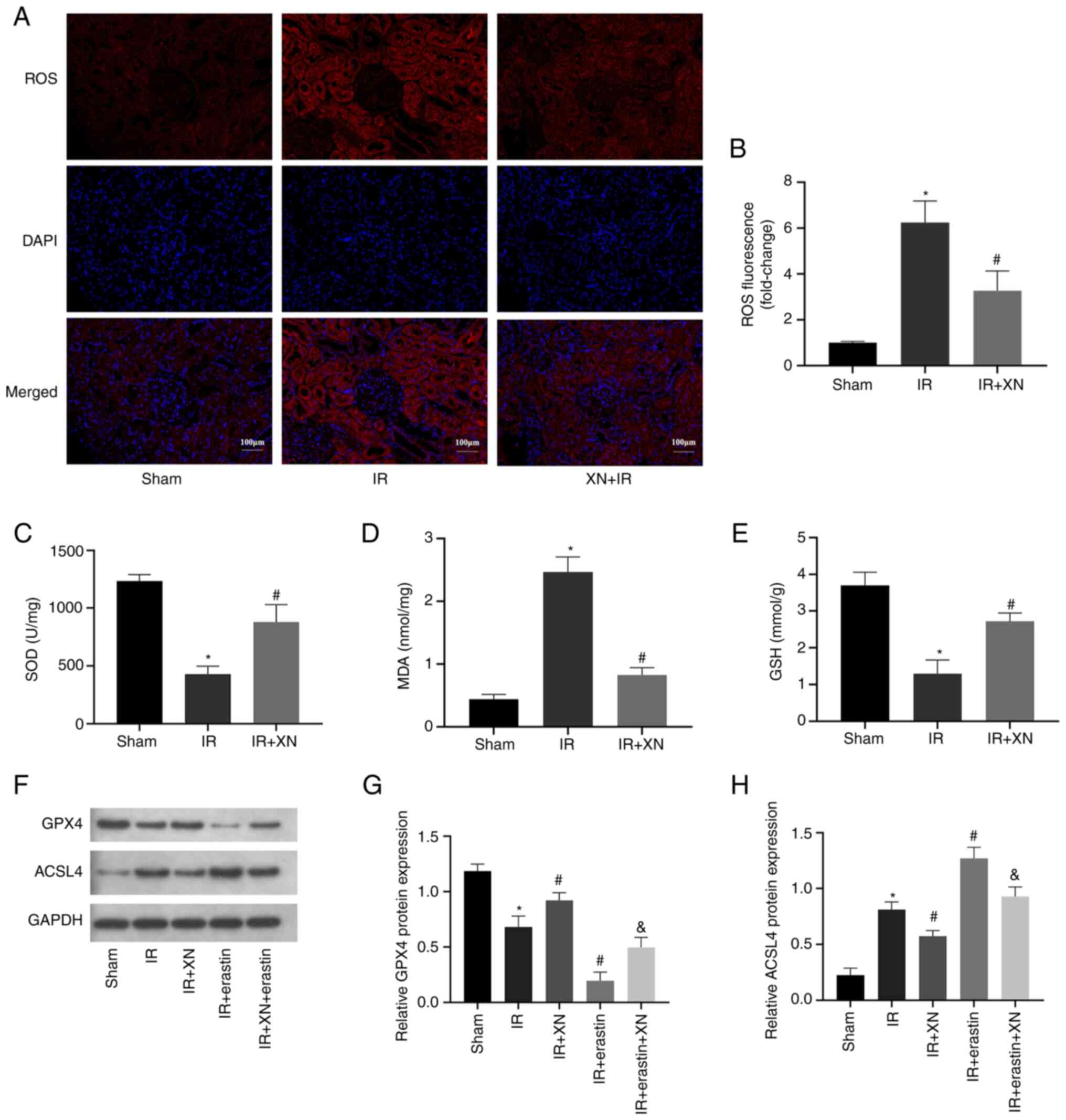 | Figure 2XN prevents ferroptosis during RIRI.
(A) ROS production in kidney tissue was measured by the
dihydroethidium fluorescence probe; magnification, x200. (B)
Quantitative analysis of ROS levels. (C) SOD, (D) MDA and (E) GSH
levels in kidney tissues of rats. (F) Western blotting of (G) GPX4
and (H) ACSL4 in kidney tissues. *P<0.05 vs. sham,
#P<0.05 vs. IR and &P<0.05 vs. XN +
IR. XN, xanthohumol; RIRI, renal ischemia/reperfusion injury; ROS,
reactive oxygen species; SOD, superoxide dismutase; MDA,
malondialdehyde; GSH, glutathione; ACSL4, long-chain acyl-CoA
synthetase. |
XN inhibits HR-induced damage and
ferroptosis in HK-2 cells
An HR model of the renal tubular epithelial cell
line HK-2 was established to determine whether XN prevents
HR-induced damage and ferroptosis. The CCK-8 assay was performed to
evaluate cell viability. HR decreased cell viability and XN
prevented cell death caused by HR. However, erastin reversed the
protective effect of XN on HK-2 cells (Fig. 3A). In addition, HR increased and
decreased the levels of MDA and SOD, respectively, in HK-2 cells.
XN increased SOD and GSH and decreased MDA in HK-2 cells. Erastin
reversed the inhibitory effect of XN on ROS levels in HK-2 cells
(Fig. 3B-D). Similarly, XN
inhibited the increase of ACSL4 expression induced by HR, enhanced
activity of GSH and upregulated the expression of GPX4, while
erastin blocked the inhibitory effect of XN on ferroptosis
(Fig. 3E-G).
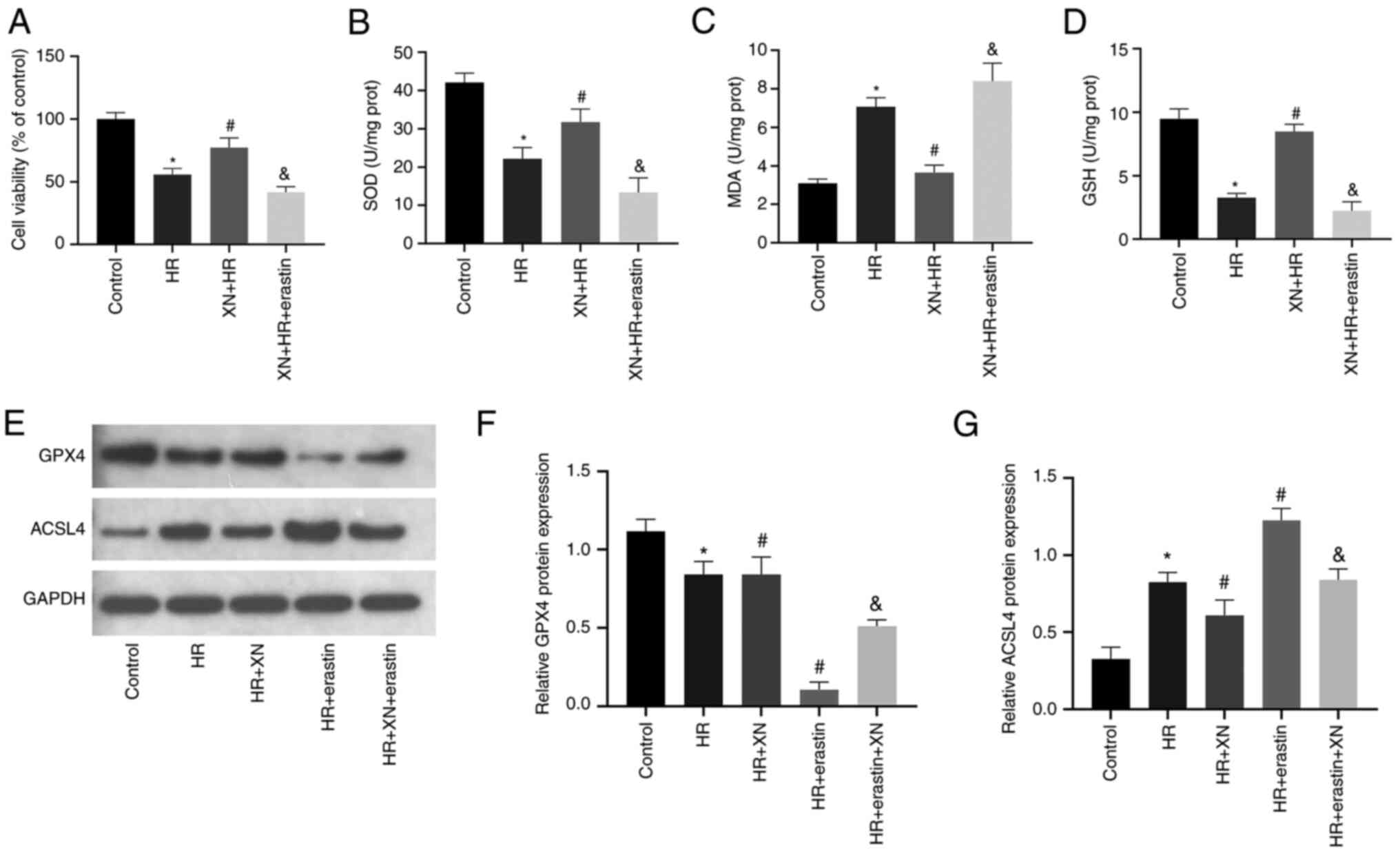 | Figure 3XN inhibits HR-induced ferroptosis in
HK-2 cells. (A) Cell Counting Kit-8 was used to evaluate cell
viability. (B) SOD, (C) MDA and (D) GSH levels in HK-2 cells. (E)
Western blotting of (F) GPX4 and (G) ACSL4 in HK-2 cells.
*P<0.05 vs. control, #P<0.05 vs. HR and
&P<0.05 vs. XN + HR. XN, xanthohumol; HR,
hypoxia-reoxygenation; SOD, superoxide dismutase; MDA,
malondialdehyde; GSH, glutathione; prot, protein; GPX4, glutathione
peroxidase 4; ACSL4, long-chain acyl-CoA synthetase. |
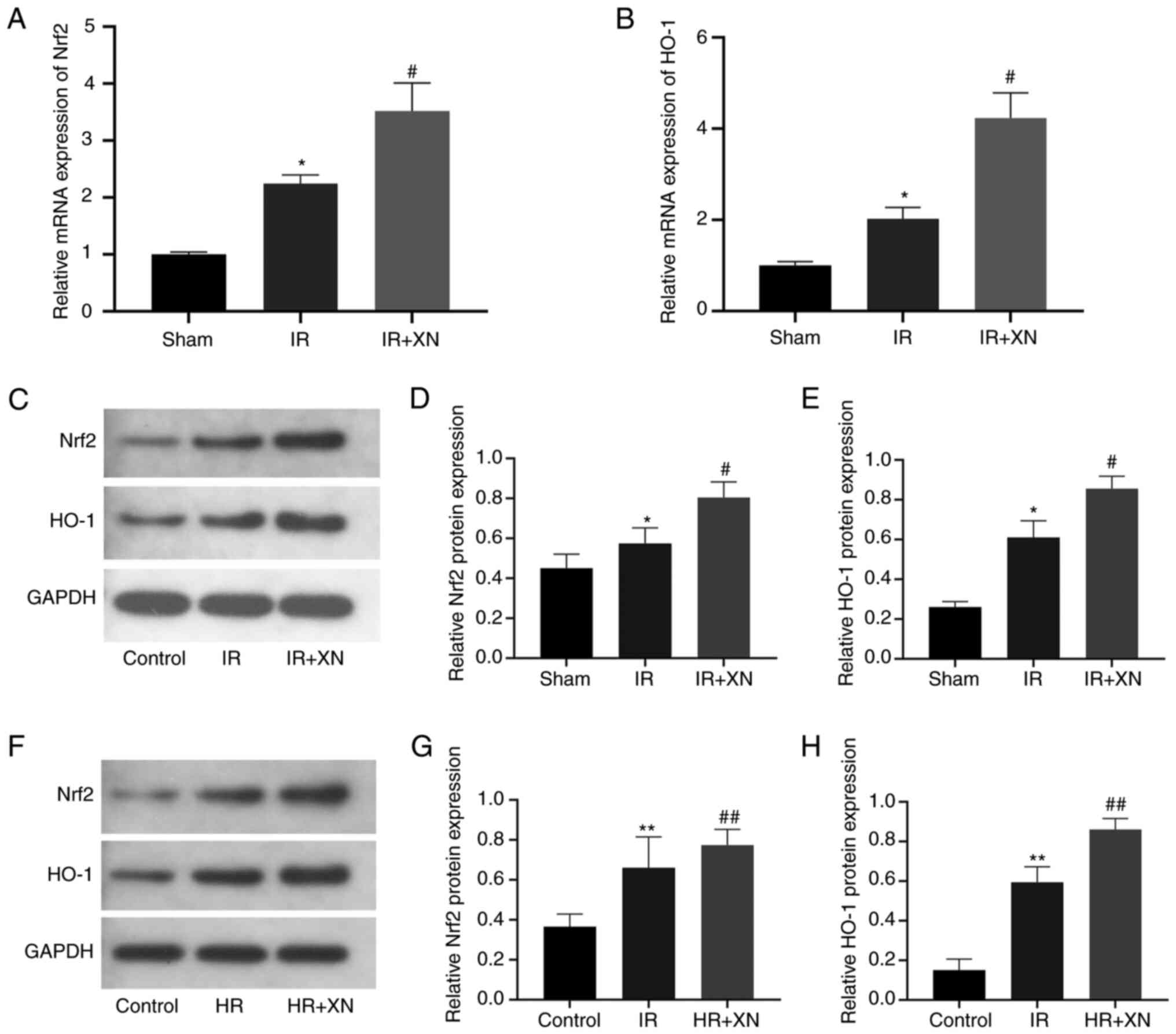
XN upregulates the Nrf2/HO-1 signaling
pathway
RT-qPCR showed that XN significantly upregulated
mRNA levels of Nrf2 and HO-1 in IR+XN group (Fig. 4A and B). Western blotting showed that IR
upregulated the expression of nuclear Nrf2 and cytoplasmic HO-1 in
renal tissue, while XN further upregulated expression of Nrf2 and
HO-1 (Fig. 4C-E). Similar results
were obtained in the HR model of cells (Fig. 4F-H).
XN inhibits IR-induced ferroptosis via
the Nrf2/HO-1 signaling pathway
To explore the role of the Nrf2/HO-1 axis in
XN-mediated protection against IRI, si-Nrf2 was used to knock down
Nrf2. Nrf2 expression was significantly decreased following si-Nrf2
transfection (Fig. 5A and B). XN prevented oxidative damage (XN
upregulated the levels of SOD and GSH levels, and downregulated the
MDA level) and decreased cell viability caused by HR, while si-Nrf2
transfection eliminated this inhibitory effect (Fig. 5C-F). Consistently, si-Nrf2
transfection inhibited the effect of XN on HR-induced upregulation
and downregulation of GPX4 and ACSL4 expression, respectively
(Fig. 5G-K). DCFH-DA fluorescent
probe assessed intracellular ROS levels in HK-2 cells following HR.
The results showed that XN decreased ROS levels, while si-Nrf2
transfection increased ROS levels (Fig. 5L and M).
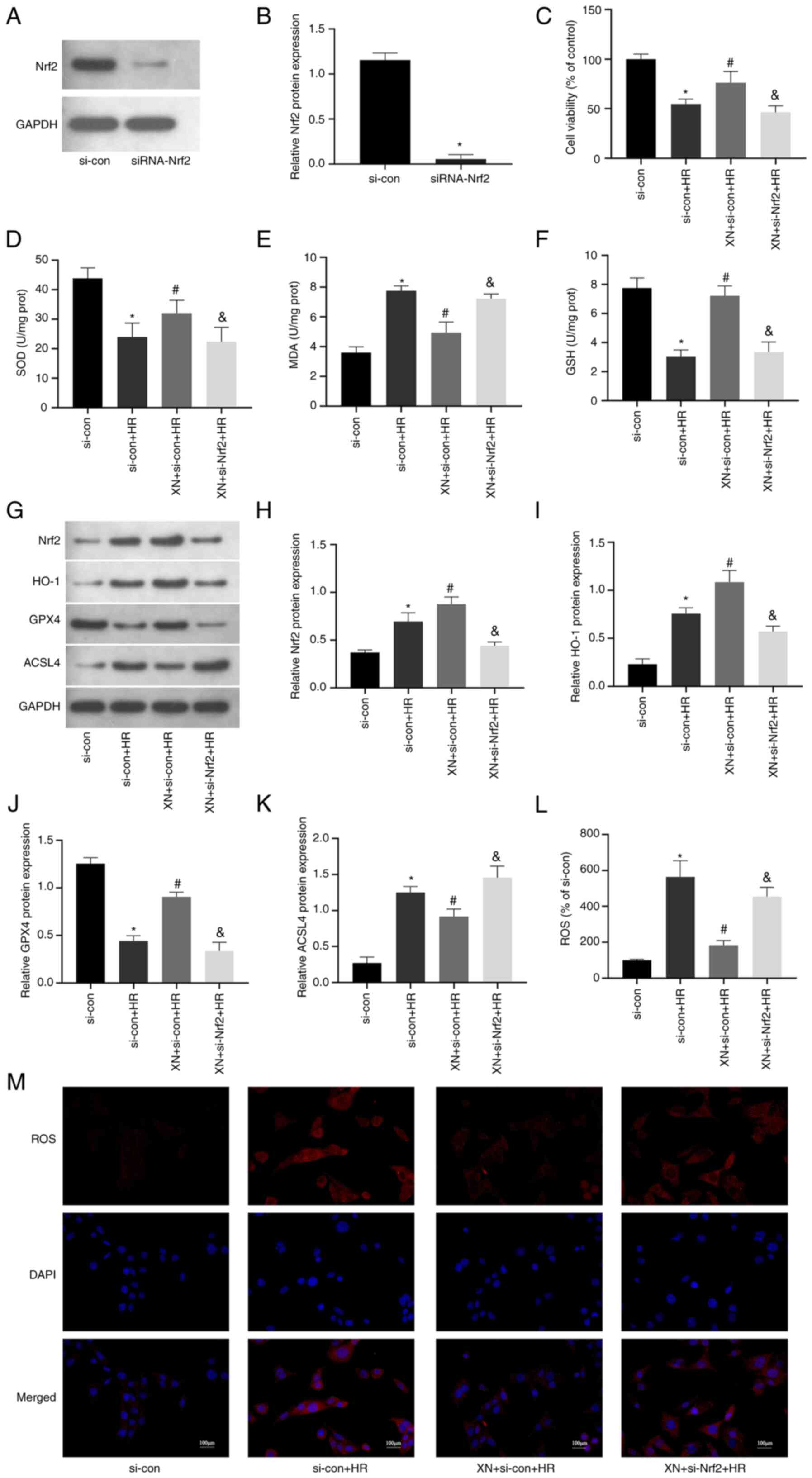 | Figure 5XN inhibits HR-induced ferroptosis
via the Nrf2/HO-1 pathway. (A) HK-2 cells were transfected with
si-Nrf2 or si-con before HR and XN; western blotting was used to
examine Nrf2 in cells. (B) Quantitative analysis of Nrf2
expression. (C) Cell Counting Kit-8 was used to evaluate cell
viability. (D) SOD, (E) MDA and (F) GSH levels in HK-2 cells. (G)
Western blotting of (H) Nrf2, (I) HO-1, (J) GPX4 and (K) ACSL4 in
HK-2 cells. (L and M) ROS production in HK-2 cells was measured by
the 2,7-dichlorofluorescein diacetate fluorescent probe;
magnification, x400. *P<0.05 vs. si-con,
#P<0.05 vs. si-con + HR, &P<0.05
vs. XN + si-con + HR. si-con, negative control small interfering
RNA; XN, xanthohumol; ROS, reactive oxygen species; HR,
hypoxia-reoxygenation; SOD, superoxide dismutase; prot, protein;
MDA, malondialdehyde; GSH, glutathione; GPX4, glutathione
peroxidase 4; ACSL4, long-chain acyl-CoA synthetase. |
Discussion
AKI is a prevalent clinical syndrome characterized
by rapid loss of renal function (24). AKI primarily manifests as a
reduction in glomerular filtration rate and renal excretion
dysfunction, coupled with a rapid increase in BUN and SCr or
hypourocrinia (24,25). Although the pathogenesis of AKI is
complex, RIRI is a common cause of AKI and it is prevalent in
critically ill patients and those undergoing major surgery
(26,27). Additionally, renal insufficiency,
such as that occurring during cardiac surgery using a
cardiopulmonary bypass machine, is a leading cause of severe AKI
(27).
RIRI is a complex pathophysiological process that
results in a decline in kidney function, leading to damage and
life-threatening consequences. Currently, there is a lack of
effective RIRI-specific drugs, and ongoing research aims to develop
potential treatments (28).
Natural products, such as traditional Chinese medicine, have gained
attention due to their low toxicity and high biological activity
(29,30).
XN is a primary active ingredient in hops and
belongs to the isoprene flavonoid compound class (31). XN has various biological functions,
such as anti-tumor and anti-inflammatory effects and regulation of
lipid metabolism (32-37).
XN induces the apoptosis of activated hepatic stellate cells in
vitro in a dose-dependent manner and decreases expression of
some pro-inflammatory response factors, such as monocyte
chemoattractant protein-1 in hepatic stellate cells (38). In addition, XN has a notable
anticancer effect and inhibits the growth of tumor blood vessels by
blocking the endothelial nuclear factor NF-κB and Akt signaling
pathways (39). XN also inhibits
production of tumor-associated vascular growth factors, such as
vascular endothelial growth factor and interleukin-8(40). While previous studies have
indicated the protective effects of XN on organs, including the
liver (41), heart (19) and cranial nerves (42), its renal protective effects have
not been fully explored. The present study found that XN protects
renal function after IRI and decreases the injury of renal tubular
epithelial cells; to the best of our knowledge, the present study
is the first to demonstrate the renal protective effect of XN in
rats.
Ferroptosis was previously identified as a form of
cell death (10). Studies have
reported ferroptosis in certain animal models of IRI in vital
organs and the level of cell death indicates the degree of IRI
(43-45).
Numerous studies have shown that ferroptosis is associated with the
pathophysiological mechanism of RIRI (46-48),
which is essential for exploring disease pathogenesis and
discovering novel therapeutic targets. The present study showed
that XN inhibited oxidative stress levels in rats with RIRI and
expression of ACSL4, but increased the expression of GPX4 during
IR. These results suggested that XN was involved in the ferroptosis
process of renal cells and decreased RIRI by inhibiting ferroptosis
in renal cells. Additionally, the Nrf2/HO-1 signaling pathway
serves a notable role in anti-oxidative stress activation in cells
(49-51).
When IRI occurs, oxidative stress increases and ferroptosis occurs
in renal tubular epithelial cells. At the same time, the
antioxidant system is activated, thus activating the Nrf2/HO-1
signaling pathway to exert its organ protective effect (52,53),
inhibiting oxidative stress and ferroptosis. The present study
showed that XN activated the Nrf2/HO-1 signaling pathway in the
presence of RIRI and blocking of the Nrf2/HO-1 signaling pathway
significantly inhibited the resistance to ferroptosis induced by
XN. Therefore, XN exerted anti-oxidative stress and
anti-ferroptosis effects by activating the Nrf2/HO-1 pathway.
In conclusion, XN decreased RIRI and improved renal
function in rats, and its mechanism may involve activation of the
Nrf2/HO-1 signaling pathway to inhibit ferroptosis in renal cells.
However, the current study has limitations. The effect of blocking
the Nrf2/HO-1 signaling pathway on inhibition of ferroptosis by XN
was only explored in vitro and animal experiments should be
conducted in future studies.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by Science and
Technology Major Project of Hubei Province (grant no.
2019AEA170).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
ZT designed and carried out the experiments and
wrote the manuscript. YF carried out part of the experiment and
completed data collection and analysis. WN carried out part of the
experiment and analyzed data. CL assisted in designing the
experiment and writing the manuscript. All authors have confirmed
the authenticity of all the raw data and have read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the Helsinki Declaration II and approved by the Institutional
Review Boards of Renmin Hospital of Wuhan University (approval no.
K2021-06-023).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Tang J and Zhuang S: Histone acetylation
and DNA methylation in ischemia/reperfusion injury. Clin Sci.
133:597–609. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wu M, Yiang G, Liao W, Tsai AP, Cheng YL,
Cheng PW, Li CY and Li CJ: Current mechanistic concepts in ischemia
and reperfusion injury. Cell Physiol Biochem. 46:1650–1667.
2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Cadenas S: ROS and redox signaling in
myocardial ischemia-reperfusion injury and cardioprotection. Free
Radical Biol Med. 117:76–89. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ampofo E, Berg JJ, Menger MD and Laschke
MW: Maslinic acid alleviates ischemia/reperfusion-induced
inflammation by downregulation of NFkappaB-mediated adhesion
molecule expression. Sci Rep. 9(6119)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Tuo QZ, Lei P, Jackman KA, Li XL, Xiong H,
Li XL, Liuyang ZY, Roisman L, Zhang ST, Ayton S, et al:
Tau-mediated iron export prevents ferroptotic damage after ischemic
stroke. Mol Psychiatry. 22:1520–1530. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Jiang GP, Liao YJ, Huang LL, Zeng XJ and
Liao XH: Effects and molecular mechanism of pachymic acid on
ferroptosis in renal ischemia reperfusion injury. Mol Med Rep.
23(63)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Liu XB and Liu WJ: The role of regulated
cell death in renal ischemia-reperfusion injury. Sheng Li Xue Bao.
74:320–332. 2022.PubMed/NCBI(In Chinese).
|
|
8
|
Cui X, Lin L, Sun X, Wang L and Shen R:
Curcumin protects against renal ischemia/reperfusion injury by
regulating oxidative stress and inflammatory response. Evid Based
Complement Alternat Med. 2021(8490772)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Latunde-Dada GO: Ferroptosis: Role of
lipid peroxidation, iron and ferritinophagy. Biochim Biophys Acta
Gen Subj. 1861:1893–1900. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta
R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS,
et al: Ferroptosis: An iron-dependent form of nonapoptotic cell
death. Cell. 149:1060–1072. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Linkermann A, Skouta R, Himmerkus N, Mulay
SR, Dewitz C, De Zen F, Prokai A, Zuchtriegel G, Krombach F, Welz
PS, et al: Synchronized renal tubular cell death involves
ferroptosis. Proc Natl Acad Sci USA. 111:16836–16841.
2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tao W, Liu F, Zhang J, Fu S, Zhan H and
Qian K: miR-3587 inhibitor attenuates ferroptosis following renal
ischemia-reperfusion through HO-1. Front Mol Biosci.
8(789927)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang J, Chen JJ, Huang JH, Lv BD, Huang
XJ, Hu Q, Fu J, Huang WJ and Tao TT: Protective effects of total
flavonoids from Lysimachia christinae on calcium
oxalate-induced oxidative stress in a renal cell line and renal
tissue. Evid Based Complement Alternat Med.
2021(6667902)2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Peluso MR, Miranda CL, Hobbs DJ, Proteau
RR and Stevens JF: Xanthohumol and related prenylated flavonoids
inhibit inflammatory cytokine production in LPS-activated THP-1
monocytes: structure-activity relationships and in silico binding
to myeloid differentiation protein-2 (MD-2). Planta Med.
76:1536–1543. 2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Liu M, Hansen PE, Wang G, Qiu L, Dong J,
Yin H, Qian Z, Yang M and Miao J: Pharmacological profile of
xanthohumol, a prenylated flavonoid from hops (Humulus
lupulus). Molecules. 20:754–779. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yen TL, Hsu CK, Lu WJ, Hsieh CY, Hsiao G,
Chou DS, Wu GJ and Sheu JR: Neuroprotective effects of xanthohumol,
a prenylated flavonoid from hops (Humulus lupulus), in
ischemic stroke of rats. J Agric Food Chem. 60:1937–1944.
2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Oberbauer E, Urmann C, Steffenhagen C,
Bieler L, Brunner D, Furtner T, Humpel C, Bäumer B, Bandtlow C,
Couillard-Despres S, et al: Chroman-like cyclic prenylflavonoids
promote neuronal differentiation and neurite outgrowth and are
neuroprotective. J Nutr Biochem. 24:1953–1962. 2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Huo H, Zhou Z, Qin J, Liu W, Wang B and Gu
Y: Erastin disrupts mitochondrial permeability transition pore
(mPTP) and induces apoptotic death of colorectal cancer cells. PLoS
One. 11(e0154605)2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lin JH, Yang KT, Lee WS, Ting PC, Luo YP,
Lin DJ, Wang YS and Chang JC: Xanthohumol protects the rat
myocardium against ischemia/reperfusion injury-induced ferroptosis.
Oxid Med Cell Longev. 2022(9523491)2022.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ma H, Wang X, Zhang W, Li H, Zhao W, Sun J
and Yang M: Melatonin suppresses ferroptosis induced by high
glucose via activation of the Nrf2/HO-1 signaling pathway in type 2
diabetic osteoporosis. Oxid Med Cell Longev.
2020(9067610)2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Paller MS: Free radical-mediated
postischemic injury in renal transplantation. Ren Fail. 14:257–260.
1992.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Sundaresan M, Yu ZX, Ferrans VJ, Irani K
and Finkel T: Requirement for generation of H2O2 for
platelet-derived growth factor signal transduction. Science.
270:296–299. 1995.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kellum JA, Romagnani P, Ashuntantang G,
Ronco C, Zarbock A and Anders HJ: Acute kidney injury. Nat Rev Dis
Primers. 7(52)2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ronco C, Bellomo R and Kellum JA: Acute
kidney injury. Lancet. 394:1949–1964. 2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Martin LC: Naringin, trimetazidine and
baroreflex in renal ischemia-reperfusion injury. Arq Bras Cardiol.
117:298–299. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kim K, Yang H, Yi J, Son HE, Ryu JY, Kim
YC, Jeong JC, Chin HJ, Na KY, Chae DW, et al: Real-time clinical
decision support based on recurrent neural networks for in-hospital
acute kidney injury: External validation and model interpretation.
J Med Internet Res. 23(e24120)2021.PubMed/NCBI View
Article : Google Scholar
|
|
28
|
Paragas N, Qiu A, Zhang Q, Samstein B,
Deng SX, Schmidt-Ott KM, Viltard M, Yu W, Forster CS, Gong G, et
al: The Ngal reporter mouse detects the response of the kidney to
injury in real time. Nat Med. 17:216–222. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
29
|
Su X, Zhou M, Li Y, Zhang J, An N, Yang F,
Zhang G, Yuan C, Chen H, Wu H and Xing Y: Protective effects of
natural products against myocardial ischemia/reperfusion:
Mitochondria-targeted therapeutics. Biomed Pharmacother.
149(112893)2022.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Li K, Xiao K, Zhu S, Wang Y and Wang W:
Chinese herbal medicine for primary liver cancer therapy:
Perspectives and challenges. Front Pharmacol.
13(889799)2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chen W, Becker T, Qian F and Ring J: Beer
and beer compounds: Physiological effects on skin health. J Eur
Acad Dermatol Venereol. 28:142–150. 2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Krajnovic T, Kaluderovic GN, Wessjohann
LA, Mijatovic S and Maksimovic-Ivanic D: Versatile antitumor
potential of isoxanthohumol: Enhancement of paclitaxel activity in
vivo. Pharmacol Res. 105:62–73. 2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Negrao R, Costa R, Duarte D, Taveira GT,
Mendanha M, Moura L, Vasques L, Azevedo I and Soares R:
Angiogenesis and inflammation signaling are targets of beer
polyphenols on vascular cells. J Cell Biochem. 111:1270–1279.
2010.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Negrao R, Duarte D, Costa R and Soares R:
Isoxanthohumol modulates angiogenesis and inflammation via vascular
endothelial growth factor receptor, tumor necrosis factor alpha and
nuclear factor kappa B pathways. Biofactors. 39:608–622.
2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Cho YC, You SK, Kim HJ, Cho CW, Lee IS and
Kang BY: Xanthohumol inhibits IL-12 production and reduces chronic
allergic contact dermatitis. Int Immunopharmacol. 10:556–561.
2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kiyofuji A, Yui K, Takahashi K and Osada
K: Effects of xanthohumol-rich hop extract on the differentiation
of preadipocytes. J Oleo Sci. 63:593–597. 2014.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Izzo G, Soder O and Svechnikov K: The
prenylflavonoid phytoestrogens 8-prenylnaringenin and
isoxanthohumol diferentially suppress steroidogenesis in rat Leydig
cells in ontogenesis. J Appl Toxicol. 31:589–594. 2011.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Dorn C, Kraus B, Motyl M, Weiss TS, Gehrig
M, Scholmerich J, Heilmann J and Hellerbrand C: Xanthohumol, a
chalcon derived from hops, inhibits hepatic inflammation and
fibrosis. Mol Nutr Food Res. 54 (Suppl 2):S205–S213.
2010.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Albini A, Dell'Eva R, Vene R, Ferrari N,
Buhler DR, Noonan DM and Fassina G: Mechanisms of the
antiangiogenic activity by the hop flavonoid xanthohumol: NF-kappaB
and Akt as targets. FASEB J. 20:527–529. 2006.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Saito K, Matsuo Y, Imafuji H, Okubo T,
Maeda Y, Sato T, Shamoto T, Tsuboi K, Morimoto M, Takahashi H, et
al: Xanthohumol inhibits angiogenesis by suppressing nuclear
factor-κB activation in pancreatic cancer. Cancer Sci. 109:132–140.
2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Dorn C, Massinger S, Wuzik A, Heilmann J
and Hellerbrand C: Xanthohumol suppresses inflammatory response to
warm ischemia-reperfusion induced liver injury. Exp Mol Pathol.
94:10–16. 2013.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Jiao Y, Cao Y, Lu X, Wang J, Saitgareeva
A, Kong X, Song C, Li J, Tian K, Zhang S, et al: Xanthohumol
protects neuron from cerebral ischemia injury in experimental
stroke. Mol Biol Rep. 47:2417–2425. 2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Friedmann AJP, Schneider M, Proneth B,
Tyurina YY, Tyurin VA, Hammond VJ, Herbach N, Aichler M, Walch A,
Eggenhofer E, et al: Inactivation of the ferroptosis regulator Gpx4
triggers acute renal failure in mice. Nat Cell Biol. 16:1180–1191.
2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Gao M, Monian P, Quadri N, Ramasamy R and
Jiang X: Glutaminolysis and transferrin regulate ferroptosis. Mol
Cell. 59:298–308. 2015.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Gascon S, Murenu E, Masserdotti G, Ortega
F, Russo GL, Petrik D, Deshpande A, Heinrich C, Karow M, Robertson
SP, et al: Identification and successful negotiation of a metabolic
checkpoint in direct neuronal reprogramming. Cell Stem Cell.
18:396–409. 2016.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Pan J, Zhao J, Feng L, Xu X, He Z and
Liang W: Inhibition of USP14 suppresses ROS-dependent ferroptosis
and alleviates renal ischemia/reperfusion injury. Cell Biochem
Biophys. 81:87–96. 2023.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Qi Y, Hu M, Qiu Y, Zhang L, Yan Y, Feng Y,
Feng C, Hou X, Wang Z, Zhang D and Zhao J: Mitoglitazone
ameliorates renal ischemia/reperfusion injury by inhibiting
ferroptosis via targeting mitoNEET. Toxicol Appl Pharmacol.
465(116440)2023.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Dong B, Ding C, Xiang H, Zheng J, Li X,
Xue W and Li Y: USP7 accelerates FMR1-mediated ferroptosis by
facilitating TBK1 ubiquitination and DNMT1 deubiquitination after
renal ischemia-reperfusion injury. Inflamm Res. 71:1519–1533.
2022.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Shen B, Zhao C, Wang Y, Peng Y, Cheng J,
Li Z, Wu L, Jin M and Feng H: Aucubin inhibited lipid accumulation
and oxidative stress via Nrf2/HO-1 and AMPK signalling pathways. J
Cell Mol Med. 23:4063–4075. 2019.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Di Tu Q, Jin J, Hu X, Ren Y, Zhao L and He
Q: Curcumin improves the renal autophagy in rat experimental
membranous nephropathy via regulating the PI3K/AKT/mTOR and
Nrf2/HO-1 signaling pathways. Biomed Res Int.
2020(7069052)2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Loboda A, Damulewicz M, Pyza E, Jozkowicz
A and Dulak J: Role of Nrf2/HO-1 system in development, oxidative
stress response and diseases: An evolutionarily conserved
mechanism. Cell Mol Life Sci. 73:3221–3247. 2016.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Qiao J, Ma H, Chen M and Bai J: Vitamin D
alleviates neuronal injury in cerebral ischemia-reperfusion via
enhancing the Nrf2/HO-1 antioxidant pathway to counteract
NLRP3-mediated pyroptosis. J Neuropathol Exp Neurol. 82:722–733.
2023.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Li X, Yi L, Liu X, Chen X, Chen S and Cai
S: Isoquercitrin played a neuroprotective role in rats after
cerebral ischemia/reperfusion through Up-regulating neuroglobin and
anti-oxidative stress. Transplant Proc. 55:1751–1761.
2023.PubMed/NCBI View Article : Google Scholar
|