Introduction
The periodontal ligament (PDL) is located between
the tooth root and alveolar bone (1). Most PDL cells are fibroblasts with
relatively high alkaline phosphatase (ALP) activity (2,3).
Fibroblasts derived from the PDL have the ability to form bone-like
tissues in vitro, similar to osteoblasts (2,3),
and thus, PDL cells function similarly to osteoblasts in hard
tissue formation. Recently, several studies have demonstrated that
PDL cells also differentiate into cementoblastic and adipogenic
cells in vitro (4).
Therefore, the PDL probably contains pluripotent progenitor cells
or putative stem cells. However, the mechanism of PDL cell
migration is poorly understood.
Stromal cell-derived factor 1α (SDF-1α, also known
as CXCL12) is an α-chemokine that strongly chemoattracts
mesenchymal stem cells (MSCs) and endothelial progenitor cells
(EPCs) via interaction with its unique receptor CXCR4 (5). In adults, tissue repair and
regeneration after injury are thought to involve the selective
recruitment of circulating or resident stem cell populations. The
importance of SDF-1α in stem and progenitor cell recruitment has
been established by showing that its expression in injured tissue
correlates with the recruitment of adult stem cells and tissue
regeneration (5–8). Therefore, SDF-1α, as a type of stem
cell-development factor and chemokine, plays an important role in
coordinating tissue injury and repair.
For regenerative therapy, biologically active
soluble factors such as cytokines and growth factors are being
evaluated for clinical use in the regeneration of periodontal
tissue damaged or lost as a result of periodontitis. Of these
factors, fibroblast growth factor 2 (FGF-2) is a multifunctional
growth factor that has a variety of effects, including the
induction of proliferation and differentiation in a wide range of
mesodermal and neuro-ectodermal cells (9). Therefore, we investigated whether
FGF-2 could regulate the expression of SDF-1α in cultured PDL cells
in vitro.
Materials and methods
Reagents
FGF-2 was obtained from R&D Systems
(Minneapolis, MN, USA). Anti-SDF-1α polyclonal antibody for the
western blot analysis was obtained from Abcam (ab9797, Cambridge,
UK). SU5402 (10 μM), SP600125 (10 μM), U0126 (10 μM), SB203580 (10
μM) and LY294002 (10 μM) were purchased from EMD Chemicals, Inc.
(Calbiochem; Gibbstown, NJ, USA).
Cell culture
PDL tissues were obtained from the middle third of
the root surfaces of healthy human permanent teeth (3 donors, aged
7–8 years), as previously described (10,11). Informed consent was obtained from
the donors' parents before tooth extraction, which was carried out
in our hospital during the course of orthodontic treatment. This
study protocol was approved by the Ethics Committee of the Iwate
Medical University, School of Dentistry (no. 01101).
The PDL tissues were cut into pieces using a
surgical blade and were digested with collagenase (2 mg/ml) at 37˚C
for 30 min. The tissues were then washed with Dulbecco's
phosphate-buffered saline (PBS), placed on culture dishes, and
maintained in α-modified minimum essential medium (α-MEM; Life
Technologies Corp., Carlsbad, CA, USA) supplemented with 10% fetal
bovine serum (FBS; Life Technologies Corp.). Fibroblastic cells
that grew out from the PDL tissues were used as PDL cells. When the
cells reached confluence, they were detached with 0.2% trypsin and
0.02% EDTA·4Na in PBS and subcultured at a 1:4 split ratio. All of
the experiments were performed using fourth passage cells cultured
in α-MEM supplemented with 10% FBS in the absence or presence of 10
ng/ml FGF-2 for between 24 and 48 h. All of the cultures were
maintained at 37˚C in a humidified atmosphere of 5% CO2
in air.
Isolation of total-RNA
Total-RNA was extracted from the cultured PDL cells
using ISOGEN (Nippon Gene, Tokyo, Japan) as described previously
(10,11). The pellet of total-RNA was washed
briefly with 75% ethanol, resuspended in 30 μl of
diethylpyrocarbonate (DEPC)-treated water, and stored at −80˚C. The
concentration of total-RNA was determined spectrophotometrically by
measuring the optical density at 260 nm.
Quantitative real-time reverse
transcription-polymerase chain reaction
One microgram of the RNA sample was
reverse-transcribed to first-strand cDNA using a PrimeScript RT
reagent kit (Takara Bio, Inc., Shiga, Japan) according to the
manufacturer's protocol. A Thermal Cycler Dice real-time system
(Takara Bio, Inc.) was used for the two-step reverse
transcription-polymerase chain reaction. The cDNA was amplified
with SYBR Premix ExTaq (Takara Bio, Inc.) and specific
oligonucleotide primers for target sequences encoding parts of
SDF-1α. The primers (Table I)
were designed based on the cDNA sequences of human mRNA for SDF-1α
and glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Amplification
conditions consisted of 10 sec at 95˚C, followed by 40 cycles at
95˚C for 5 sec and 60˚C for 30 sec, with a final 15 sec at 95˚C and
30 sec at 60˚C in the Thermal Cycler Dice real-time system
(12,13).
 | Table IPrimers used in the quantitative
real-time PCR reverse transcription-polymerase chain reaction
(real-time PCR). |
Table I
Primers used in the quantitative
real-time PCR reverse transcription-polymerase chain reaction
(real-time PCR).
| Gene name | Origonucleotide
sequence (5′-3′) |
|---|
| SDF-1 | F:
GAGCCAACGTCAAGCATCTCAA
R: TTTAGCTTCGGGTCAATGCACA |
| GAPDH | F:
GCACCGTCAAGGCTGAGAAC
R: TGGTGAAGACGCCAGTGGA |
Western blot analysis of SDF-1α
expression from PDL cells
After treatment with FGF-2 for 7 days, the
conditioned media from PDL cell culture were collected for SDF-1α
analysis. Subsequently, 20 μl of conditioned media were dissolved
in SDS buffer without dithiothreitol, incubated at 95˚C for 5 min,
resolved electrophoretically on 10% SDS-polyacrylamide gels and
transferred to a polyvinylidene difluoride membrane (Millipore
Corp., Bedford, MA, USA). After being blocked with 5% skim milk in
Tris-buffered saline containing 0.1% Tween-20 (TBST), the membrane
was incubated with mouse anti-human SDF-1α antibodies and
subsequently with anti-mouse secondary antibodies (Life
Technologies Corp.). Specific protein bands on the membrane were
detected using an enhanced AP conjugate substrate kit (Bio-Rad
Laboratories, Inc., Hercules, CA, USA) as previously described
(10–13).
Statistical analysis
The results are expressed as means ± SEM.
Statistical significance was determined using one-way analysis of
variance with Bonferroni post hoc comparisons between pairs of
groups. The threshold for statistical significance was set a
priori at P<0.01.
Results
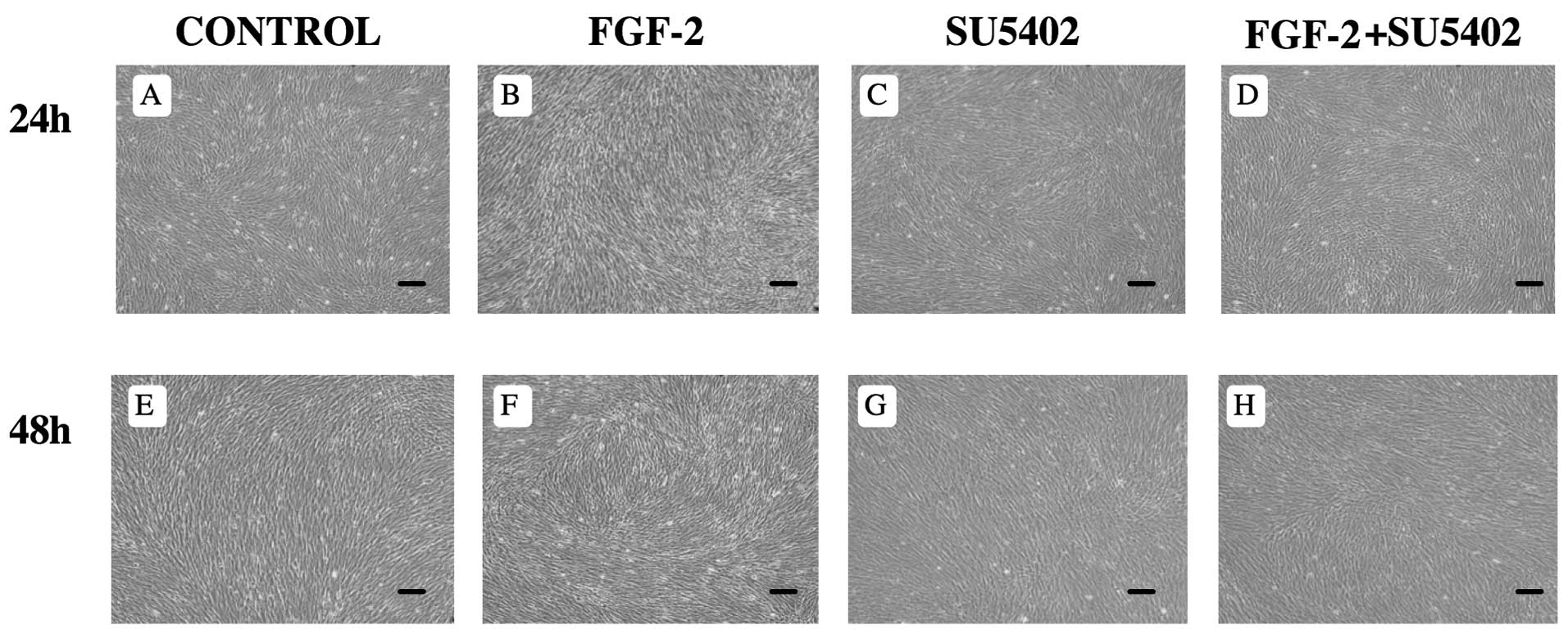
FGF-2 induces morphological changes
Morphological changes in PDL cells were induced by
treatment with FGF-2 for 24–48 h. After culturing for 24–48 h, PDL
cells reached confluence in control media (Fig. 1A and E) and in the presence of
FGF-2 (Fig. 1B and F). When
treated with FGF-2, PDL cells altered their morphology into long,
thin, spindle-shaped fibroblasts (Fig. 1B and F). There were no differences
in the appearance of PDL cells between control and SU5402 treatment
(Fig. 1A, C, E and G), or between
control and FGF-2 + SU5402 (Fig. 1A,
C, D and H).
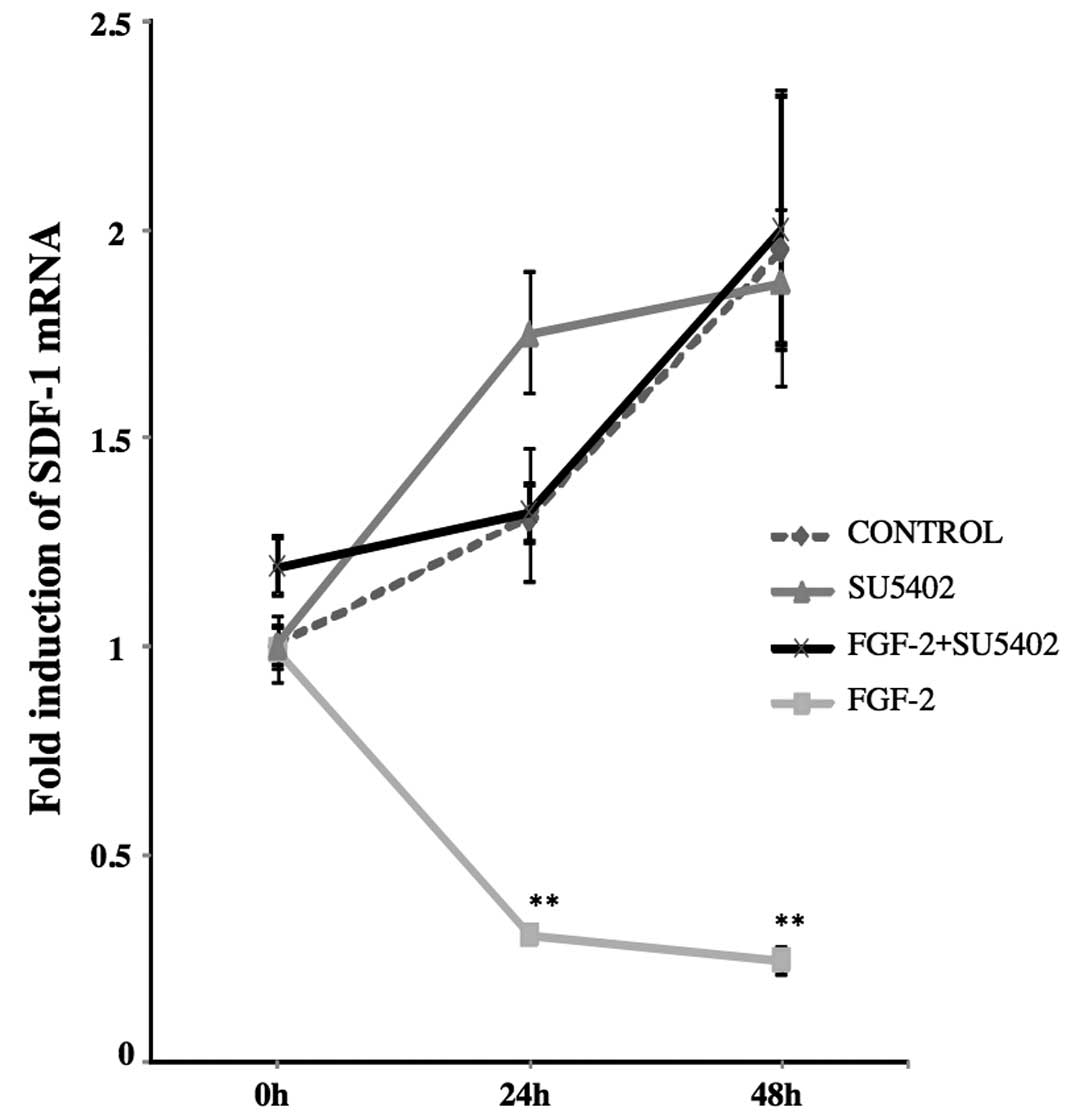
FGF-2 suppresses SDF-1α mRNA
expression
Expression of SDF-1α mRNA was suppressed in PDL
cells cultured in the presence of FGF-2 for 24 and 48 h. When PDL
cells were cultured in the presence of FGF-2 for 24 and 48 h,
SDF-1α mRNA expression was significantly decreased compared to the
0 h level (0 h, 1; 24 h, 0.3; 48 h, 0.2; P<0.01). However, after
treating with SU5402 alone and FGF-2 + SU5402, SDF-1α expression
was slightly increased (Fig.
2).
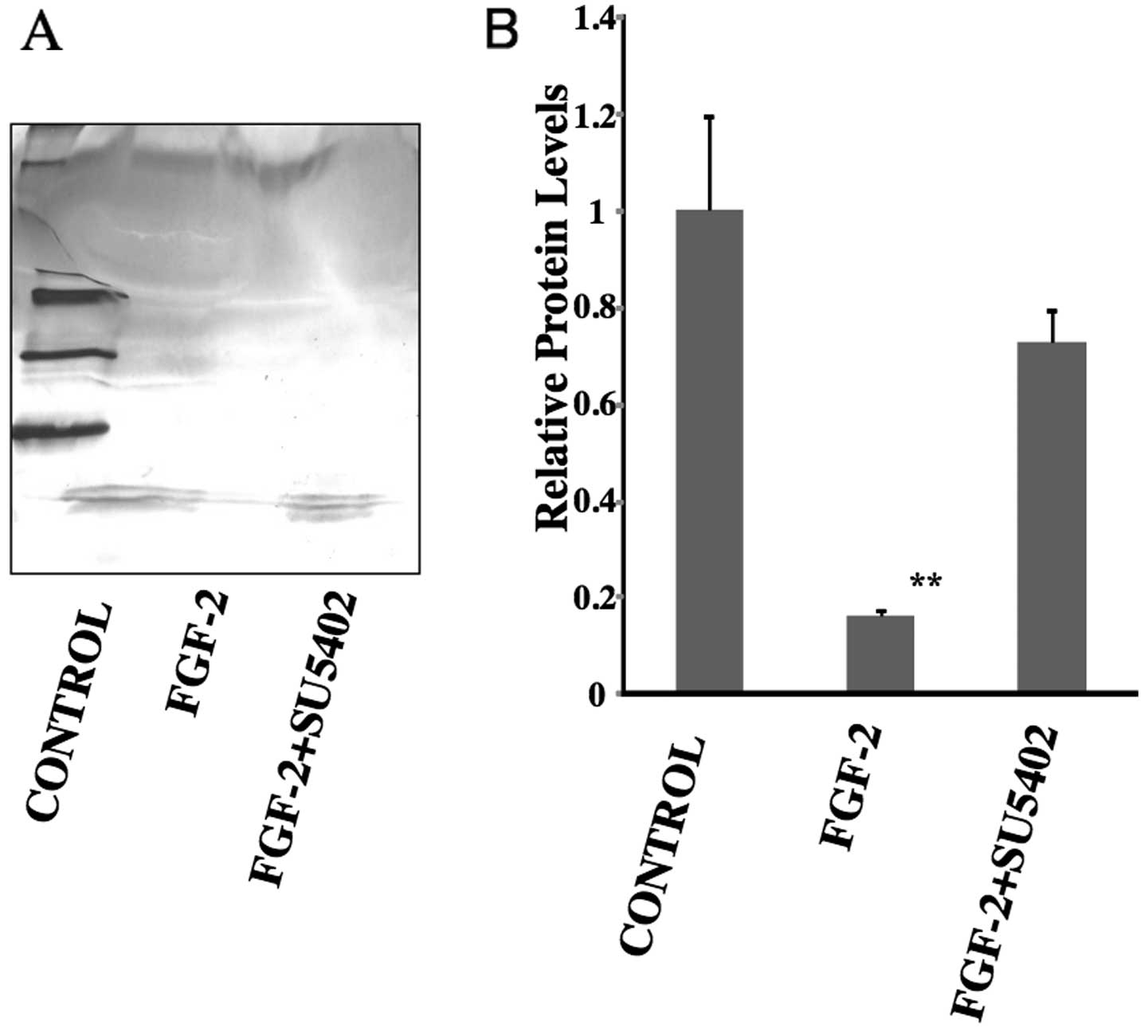
FGF-2 decreased SDF-1α expression
SDF-1α expression decreased in PDL cells cultured in
the presence of FGF-2. After treatment with FGF-2 for 7 days, the
production of SDF-1α was noticeably decreased in PDL cells compared
to the control (control, 1; FGF-2, 0.16; P<0.01) (Fig. 3). However, in the presence of
FGF-2 + SU5402, SDF-1α expression was slightly decreased compared
with control, although this was not statistically significant
(control, 1; FGF-2 + SU5402, 0.72).
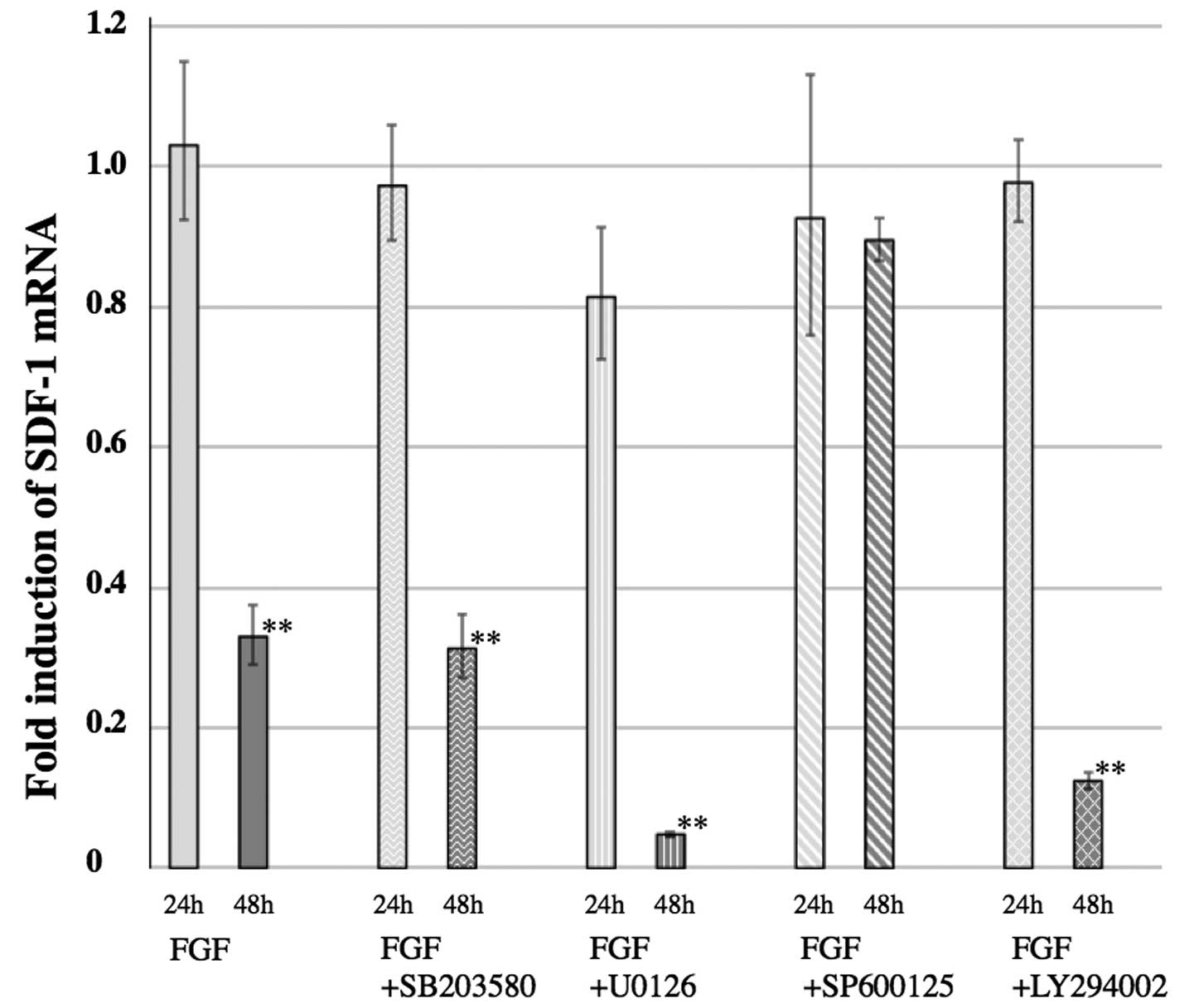
SP600125 inhibites the FGF-2-mediated
decrease in SDF-1α expression
The decreased expression of SDF-1α in PDL cells,
mediated by FGF-2, was inhibited by SP600125, an inhibitor of JNK.
Only the treatment of FGF-2 + SP600125 inhibited the decreased
expression of SDF-1α observed when PDL cells were cultured with
FGF-2 alone (Fig. 4). Other MAP
kinase inhibitors, U0126 (an MEK1/2 inhibitor) and SB203580 (a p38
kinase inhibitor), had no effect on the decreased expression of
SDF-1α. LY294002, a phosphatidylinositol-3 kinase (PI3K) inhibitor,
also had no effect on the decreased expression of SDF-1α (Fig. 4).
Discussion
This study demonstrated that SDF-1α mRNA was
inhibited in cultured PDL cells by treatment with FGF-2. Decreased
expression of SDF-1α was also demonstrated in conditioned media
from PDL cells cultured with FGF-2. This is the first report of
altered SDF-1α expression regulated by FGF-2 treatment in PDL cells
derived from human permanent teeth.
PDL tissue regeneration and homeostasis in response
to pathological and environmental changes, such as injury and
orthodontic treatment, are thought to depend on recruitment of
circulating or resident stem cells. SDF-1α plays an important role
in tissue healing by recruiting endothelial progenitor cells and
MSCs from the bone marrow through its receptor, CXCR4 (5–8).
However, it remains unclear whether the recruitment of stem and
progenitor cells can be regulated in PDL tissue.
In this study, PDL cells derived from human
permanent teeth were used to investigate the effects of FGF-2 on
the expression of SDF-1α, using real-time PCR. Previous studies
have shown that PDL cells not stimulated with FGF-2 basally express
SDF-1α (14,15). The results of the current study
support those findings; PDL cells expressed SDF-1α in the absence
of FGF-2 (Fig. 2). Surprisingly,
PDL cells significantly decreased the expression of SDF-1α when
cultured in the presence of FGF-2 (Fig. 2). In addition, this study
demonstrated that SDF-1α expression was regulated by FGF-2 via the
fibroblast growth factor receptor (FGFR). Results showed levels of
SDF-1α expression similar to those of the control when cultured in
media with combined FGF-2 and an FGF receptor antagonist, SU5402.
No statistical differences in SDF-1α expression were found between
untreated cells and cells treated with SU5402 alone.
Western blot analysis showed that SDF-1α protein
production in conditioned media was significantly decreased by
treatment with FGF-2 compared to the control group. In the presence
of FGF-2 + SU5402, SDF-1α expression recovered almost to control
levels (Fig. 3). Similarly to the
real-time PCR results, this analysis suggests that FGF-2 inhibits
SDF-1α protein expression in PDL cells via the FGFR.
A phosphatidylinositol-3 kinase (PI3K) inhibitor,
LY2940002, blocks PI3K and results in the inhibition of Akt pathway
activity. It has previously been shown that FGF-2-induced Akt
phosphorylation depends upon PI3K in MSCs (16). In the presence of FGF-2 +
LY2940002, FGF-2-reduced expression of SDF-1α was not inhibited in
PDL cell culture. Therefore, the current results indicate that the
PI3K/Akt pathway is not related to FGF-2-reduced expression of
SDF-1α in PDL cells.
Furthermore, the specific MEK inhibitor U0126 or the
p38 MAP kinase inhibitor SB202190 in combination with FGF-2 had no
effect on FGF-2-induced SDF-1α inhibition (Fig. 4). However, the JNK inhibitor
SP600125 in combination with FGF-2 inhibited FGF-2-reduced
expression of SDF-1α (Fig. 4).
These results indicate that the JNK pathway plays an important role
in FGF-2-mediated effects in PDL cells.
FGF-2 has been described as a multipotent cytokine
that regulates cell proliferation as well as differentiation,
matrix composition, and migration in a number of cell types
(9). It has also been shown that
FGF-2 itself as well as SDF-1α could control MSC migration
(5–8,17).
SDF-1α expression levels in clinically inflamed dental pulp tissues
were higher than those in healthy dental pulp (18). Moreover, human gingival
fibroblasts constitutively expressed SDF-1α (19). This expression was enhanced by
stimulation with tumor necrosis factor-α (TNF-α) and transforming
growth factor-β (TGF-β) (19).
Together with these results, it can be concluded that PDL cells in
an environment where FGF-2 is abundant may decrease SDF-1α
expression because FGF-2 is capable of inducing the migration of
MSCs.
This is the first study to demonstrate that the
treatment of PDL cells derived from human permanent teeth with
FGF-2 in vitro regulates SDF-1α expression. These findings
can aid in understanding of mechanisms of PDL tissue regeneration
by suggesting that with effective regulation of SDF-1α expression,
MSC migration can be controlled.
Acknowledgements
This study was supported in part by Grants-in-Aid
for Scientific Research (nos. 18592026, 23592896 to A.I., no.
19791370 to N.C., no. 21390548 to M.M., and nos. 18592239 and
22592296 to T.H.) and Grant-in-Aid for Challenging Exploratory
Research (no. 23659965 to M.M.) from the Ministry of Education,
Culture, Sports, Science and Technology of Japan; the Open Research
Project and High-Tech Research Project from the Ministry of
Education, Culture, Sports, Science and Technology of Japan; and
the Medical Innovation by Advanced Science and Technology (MIAST) a
project; Grant-in-Aid for Strategic Medical Science Research Center
from the Ministry of Education, Culture, Sports, Science and
Technology of Japan, 2010–2014; the Akiyama Foundation (to T.H.,
2005); and a grant from the Keiryokai Research Foundation (no. 100
to N.C., 2008; no. 103 to A.I. and no. 106 to T.H., 2009).
References
|
1
|
E FreemanPeriodontiumOral Histology:
Development, Structure and FunctionAR Ten CateMosbySt
Louis2763121994
|
|
2
|
MC GroeneveldV EvertsW BeertsenAlkaline
phosphatase activity in the periodontal ligament and gingiva of the
rat molar: its relation to cementum formationJ Dent
Res7413741381199510.1177/002203459507400709017560388
|
|
3
|
W BeertsenT van den BosAlkaline
phosphatase induces the mineralization of sheets of collagen
implanted subcutaneously in the ratJ Clin
Invest8919741980199210.1172/JCI1158051602003
|
|
4
|
BM SeoM MiuraS GronthosPM BartoldS
BatouliJ BrahimM YoungPG RobeyCY WangS ShiInvestigation of
multipotent postnatal stem cells from human periodontal
ligamentLancet364149155200410.1016/S0140-6736(04)16627-015246727
|
|
5
|
J WangR LobergRS TaichmanThe pivotal role
of CXCL12 (SDF-1)/CXCR4 axis in bone metastasisCancer Metastasis
Rev25573587200610.1007/s10555-006-9019-x17165132
|
|
6
|
A ZerneckeA SchoberI BotP von
HundelshausenEA LiehnB MöppsM MericskayP GierschikEA BiessenC
WeberSDF-1alpha/CXCR4 axis is instrumental in neointimal
hyperplasia and recruitment of smooth muscle progenitor cellsCirc
Res96784791200510.1161/01.RES.0000162100.52009.3815761195
|
|
7
|
M KuciaJ RatajczakR RecaA
Janowska-WieczorekMZ RatajczakTissue-specific muscle, neural and
liver stem/progenitor cells reside in the bone marrow, respond to
an SDF-1 gradient and are mobilized into peripheral blood during
stress and tissue injuryBlood Cells Mol
Dis325257200410.1016/j.bcmd.2003.09.025
|
|
8
|
MZ RatajczakM KuciaR RecaM MajkaA
Janowska-WieczorekJ RatajczakStem cell plasticity revisited:
CXCR4-positive cells expressing mRNA for early muscle, liver and
neural cells 'hide out' in the bone
marrowLeukemia182940200410.1038/sj.leu.240318414586476
|
|
9
|
S MurakamiPeriodontal tissue regeneration
by signaling molecule(s): what role does basic fibroblast growth
factor (FGF-2) have in periodontal
therapy?Periodontology56188208201110.1111/j.1600-0757.2010.00365.x21501244
|
|
10
|
T HasegawaY YoshimuraT KikuiriY YawakaS
TakeyamaA MatsumotoH OguchiT ShirakawaExpression of receptor
activator of NF-kappa B ligand and osteoprotegerin in culture of
human periodontal ligament cellsJ Periodont
Res37405411200210.1034/j.1600-0765.2002.01603.x12472833
|
|
11
|
T HasegawaT KikuiriS TakeyamaY YoshimuraM
MitomeH OguchiT ShirakawaHuman periodontal ligament cells derived
from deciduous teeth induce osteoclastogenesis in vitroTissue
Cell344451200210.1054/tice.2002.022311989970
|
|
12
|
T HasegawaN ChosaT AsakawaY YoshimuraA
AsakawaA IshisakiM TanakaEffect of fibroblast growth factor-2 on
periodontal ligament cells derived from human deciduous teeth in
vitroExp Ther Med1337341201022993547
|
|
13
|
T HasegawaN ChosaT AsakawaY YoshimuraA
AsakawaA IshisakiM TanakaEffect of fibroblast growth factor-2 on
dental pulp cells derived from human deciduous teeth in vitroExp
Ther Med14774802010
|
|
14
|
O TrubianiA IsgroN ZiniI AntonucciF AiutiR
Di PrimioA NanciS CaputiR PaganelliFunctional
interleukin-7/interleukin-7Ralpha, and SDF-1alpha/CXCR4 are
expressed by human periodontal ligament derived mesenchymal stem
cellsJ Cell Physiol214706713200810.1002/jcp.2126617894415
|
|
15
|
K NagatomoM KomakiI SekiyaY SakaguchiK
NoguchiS OdaT MunetaI IshikawaStem cell properties of human
periodontal ligament cellsJ Periodontal
Res41303310200610.1111/j.1600-0765.2006.00870.x16827724
|
|
16
|
SC ChoiSJ KimJH ChoiCY ParkWJ ShimDS
LimFibroblast growth factor-2 and -4 promote the proliferation of
bone marrow mesenchymal stem cells by the activation of the
PI3K-Akt and ERK1/2 signaling pathwaysStem Cells
Dev17725736200810.1089/scd.2007.023018788932
|
|
17
|
A SchmidtD LadageT SchinkötheU KlausmannC
UlrichsFJ KlinzK BrixiusS ArnholdB DesaiU MehlhornBasic fibroblast
growth factor controls migration in human mesenchymal stem
cellsStem
Cells2417501758200610.1634/stemcells.2005-019116822883
|
|
18
|
L JiangYQ ZhuR DuYX GuL XiaF QinHH
RitchieThe expression and role of stromal cell-derived
factor-1alpha-CXCR4 axis in human dental pulpJ
Endod34939944200810.1016/j.joen.2008.05.01518634924
|
|
19
|
Y HosokawaI HosokawaK OzakiH NakaeK
MurakamiY MiyakeT MatsuoCXCL12 and CXCR4 expression by human
gingival fibroblasts in periodontal diseaseClin Exp
Immunol141467474200510.1111/j.1365-2249.2005.02852.x16045736
|