Introduction
Polycystic ovary syndrome (PCOS) is one of the most
common endocrine disorders, affecting 6–10% of women of
reproductive age, and is diagnosed by combination of polycystic
ovaries, hyperandrogenism or oligo-anovulation (1–3).
Serum luteinizing hormone (LH) hypersecretion, compensatory
hyperinsulinemia from insulin resistance and reduced fecundity are
common features of PCOS and at least 50% of PCOS women are insulin
resistant when compared with age- and weight-matched controls
(4). Genetic studies on this
disorder have certain difficulties due to heterogeneity in the
phenotype of PCOS patients. Therefore, association studies have
been used as a powerful genetic approach to analyze the complex
traits in PCOS. Most molecular and genetic analyses have focused on
genes involved in folliculogenesis and insulin signaling (5–12).
Follicular fluid, secreted by granulose cells and by
diffusion from the theca capillaries, plays an essential role in
the physiology of follicular growth, oocyte maturation and
ovulation (13). A number of
studies have revealed that follicular fluid inhibits the binding of
spermatozoa to the zona pellucida (14), and promotes hyperactivation and
acrosome reaction of sperm (15,16), and oocyte maturation and embryo
development (17,18). Moreover, follicular fluid contains
high levels of gonadotropin and growth factors that are important
to oocyte maturation and embryo development (19). Therefore, the chemical composition
of fluid from dominant follicles can be used as an indicator of the
secretory activities and metabolism of follicular cells, which
regulate the follicular quality. Recent proteomic studies
demonstrated that most proteins in human follicular fluid are also
present in the plasma, suggesting that these proteins can be used
as biomarkers for reproductive diseases using both follicular fluid
and plasma (20–23). In this study, we performed genetic
and proteomic analyses to identify novel PCOS-associated factors in
follicular fluid from PCOS patients.
Materials and methods
Subjects and biochemical
determinations
Women with PCOS were recruited at the Fertility
Center at CHA General Hospital in Seoul, Korea. The criteria for
diagnosis of PCOS rely on the combination of clinical symptoms,
ultrasonographic examination, and biochemical data based on the
revised diagnostic criteria announced in the 2003 ASRM/ESHRE
Rotterdam consensus (24).
Healthy female volunteers were enrolled and used as the control
group. Their healthy state was determined based on medical history,
physical and pelvic examination and complete blood chemistry. Their
normal ovulatory state was confirmed by transvaginal
ultrasonography and a plasma hormone assay was conducted during the
luteal phase of the cycle.
The clinical characteristics of normal and PCOS
patients are listed in Table I.
Informed written consent was obtained from all participants.
Follicular fluid was prepared as previously described (20). To carry out western blot analysis,
serum samples were prepared as previously described (20). Venous blood samples were obtained
prior to follicular aspiration, and were collected in sterile
plastic tubes containing ethylenediaminetetraacetic acid (EDTA) as
an anticoagulant and immediately centrifuged. All samples were
stored at −80°C until use.
 | Table IClinical characteristics of normal
and PCOS patients for follicular fluid and blood serum. |
Table I
Clinical characteristics of normal
and PCOS patients for follicular fluid and blood serum.
| Normal | PCOS |
|---|
| No. | 5 (3) | 5 (7) |
| Body mass index
(kg/m2) | 20.94 (19.78–22.38)
(20.02–21.36) | 23.07 (19.97–26.35)
[22.37 (18.02–25.24)] |
| FSH (IU/l) | 6.1 (3.7–8.2) [6.4
(5.0–7.5)] | 5.74 (2.9–8.2) [5.8
(3.1–8.5)] |
| LH (IU/l) | 4.98 (2.3–6.7)
[2.49 (1.5–4.0)] | 10.46 (8.0–13.0)
[7.28 (0.4–24.1)] |
| E2
(pmol/l) | 27.86 (9.3–49.0)
[17 (11.76–25.13)] | 32.6 (19.0–61.0)
[27 (7.81–52.66)] |
For genetic analysis, 233 patients with PCOS and 120
healthy women of reproductive age were recruited from the Fertility
Center at CHA General Hospital. Blood samples were obtained in
tubes containing EDTA, which acts as an anticoagulant. The
follicular fluid and blood serum used for proteomic analysis were
also used in the genomic study. Genomic DNA was extracted using a
DNA purification system kit (Gentra Systems, Minneapolis, MN, USA).
Biochemical analysis of various hormones from blood including
insulin, DHEA-S, testosterone, DHEA-S, FSH and LH was conducted.
Furthermore, the participants underwent a series of physical
examinations including weight, height and waist/hip ratio
measurements, and the history of the patient and family were
recorded (Table I). The human
follicular fluid and blood samples obtained from controls and PCOS
patients were approved by the CHA General Hospital Institutional
Review Board.
Two-dimensional electrophoresis (2-DE)
and image analysis
Immobiline DryStrips (IPG) pH 3–10 nonlinear 18 cm,
and Pharmalytes pH 3–10 were purchased from Amersham Pharmacia
Biotech (Uppsala, Sweden); Griess reagent, CHAPS, urea, DTT, Tris
base, thiourea, glycine, ammonium persulfate and SDS were obtained
from Sigma (St. Louis, MO, USA); Coomassie brilliant blue (CBB)
G-250 and TEMED were from Bio-Rad (Hercules, CA, USA). All other
chemicals were obtained from various commercial sources and were of
the highest grade.
Depletion of the six most abundant proteins (i.e.,
albumin, transferrin, IgG, IgA, haptoglobin and antitrypsin) in
follicular fluid was carried out using a Multiple Affinity Removal
Column (MARC) (Agilent Technologies, Inc., Wilmington, DE, USA).
Then, 2-DE and image analyses were performed, as previously
described (20).
In-gel digestion and LC-MS/MS
Excised spots were destained, reduced, alkylated,
and then digested with trypsin (Promega, Madison, WI, USA) as
previously described (25). For
matrix-assisted laser desorption/ionization-time-of-flight mass
spectrometry (MALDI-TOF-MS) analysis, the tryptic peptides were
concentrated using a poros R2 and oligo R3 column (Applied
Biosystems, Foster City, CA, USA) and eluted in
α-cyano-4-hydroxycinnamic acid. Spectra were obtained using a 4700
TOF/TOF spectrophotometer (Applied Biosystems). Proteins were
identified from the peptide mass maps using MASCOT (http://www.matrixscience.com/search_form_select.html),
MS-Fit (http://prospector.ucsf.edu), and
ProFound (http://prowl.rockfeller.edu/prowl-cgi/profound.exe) to
search for the proteins in the protein database, Swiss-Prot and
GenBank. For nano LC-ESI-MS/MS analysis, the peptides digested with
trypsin were concentrated using a poros R2 and oligo R3 column
(Applied Biosystems).
Bioinformatics
Each MS/MS spectrum obtained was searched against
the non-redundant protein sequence database using the Spectrum Mill
software tool. Sequences of uninterpreted CID spectra were
identified by correlation with the peptide sequences present in the
protein sequence database (NCBInr 2012.1) using the Spectrum Mill
MS Proteomics Workbench (Rev A.03.00.015; Agilent Technologies,
Inc.).
Western blot analysis
Human follicular fluid proteins diluted to 1/10 with
PBS buffer and human sera were subjected to SDS-PAGE. Albumin was
removed from the follicular fluid to detect kininogen 1,
cytokeratin 9, antithrombin, fibrinogen γ-chain, apolipoprotein
A-IV (apoA-IV) precursor, and α-1-B-glycoprotein (A1BG) using the
Aurum serum protein mini kit (Bio-Rad). The proteins were loaded in
each lane of a 7.5% SDS-PAGE and blotted onto nitrocellulose
membranes. The membranes were incubated with a blocking solution,
containing a 1:200 dilution of anti-kininogen 1 (Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA), or anti-cytokeratin 9
antibodies (Abcam, Cambridge, UK). In addition, an antithrombin
antibody (Abcam) diluted to 1:1,000 with the blocking solution was
used. Also, the membranes were incubated with a blocking solution,
containing a 1:500 dilution of anti-fibrinogen λ, anti-apoA-IV
(Santa Cruz Biotechnology, Inc.), or anti-A1BG (Aviva Systems
Biology, San Diego, CA, USA) antibodies. The membranes were then
incubated with a blocking solution containing a 1:5,000 dilution of
horseradish peroxidase-conjugated secondary antibodies (KPL,
Gaithersburg, MD, USA). An ECL system (Elpis Biotech, Daejeon,
Korea) was used to detect signals. Bands from the western blot
analysis were scanned and digitized using Fluor-S™ MultiImager
(Bio-Rad). Values are expressed as the means ± standard error of
the mean (SEM).
Genetic analysis
Restriction fragment length polymorphism (RFLP)
analysis was performed for apoA-IV using isolated genomic
DNA from the blood samples obtained from all subjects. PCR primers
were designed and used to amplify the DNA for the genotyping
analysis. To determine the Gln360His variant genotype, the
following primers were designed: forward, 5′-CTT CCA GAT GAA GAA
GAA CG-3′ and reverse, 5′-AGT TTC TGC CTG AGC TGT T-3′. The PCR
conditions were as follows: denaturation at 94°C for 30 sec,
annealing at 55°C for 10 sec and polymerization at 72°C for 15 sec.
Amplified PCR products were purified using a PCR purification kit
(AccuPrep®; Bioneer, Daejeon, Korea) and screened by
restriction digestion with Fnu4HI (New England Biolabs,
Beverly, MA, USA) at 37°C for 6 h. In addition, amplified PCR
products for each genotype were sequenced in order to confirm the
genotyping results from the RFLP analysis.
Statistical analysis
For the statistical analysis, the HapAnalyzer
(National Genome Research Institute, Seoul, Korea) and SAS ver. 9.1
(2006) were used to analyze the distributions of the apoA-IV
genotype. P-value <0.05 was considered to indicate a
statistically significant difference.
Results
2-DE analysis on follicular fluid of
PCOS
A typical 2-DE separation was performed in the pH
range of 3–10 on proteins extracted from the follicular fluid of
PCOS patients and healthy women as a control to identify proteins
that were aberrantly expressed in PCOS patients. In order to
deplete the samples of the most abundant proteins found in
follicular fluid, an immunoaffinity column (MARC) was applied to
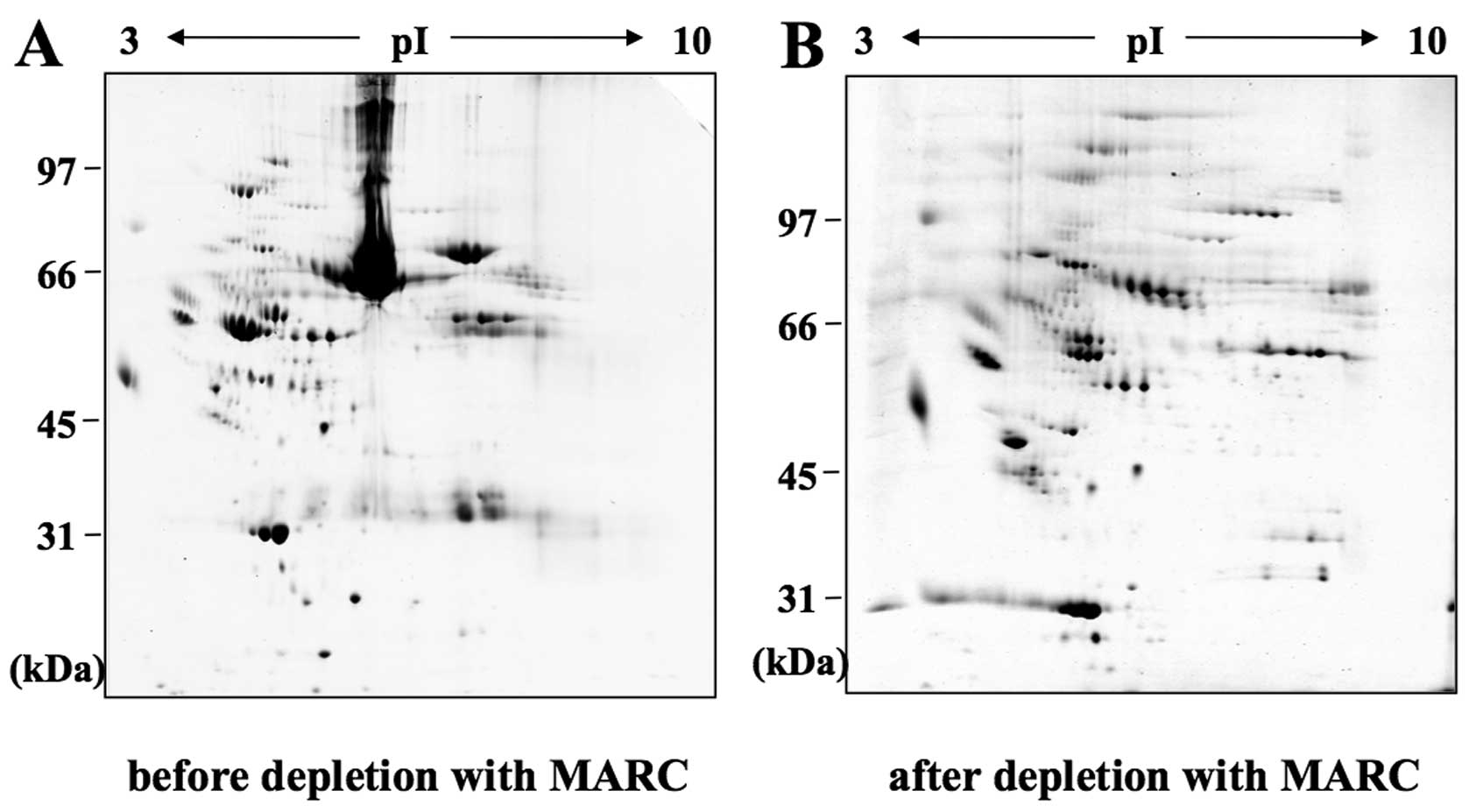
collect flow-through fractions (Fig.
1), as previously described (20).
Follicular fluids were collected from in
vitro fertilization (IVF) patients suffering from PCOS, and
from normal controls who underwent an IVF cycle due to male
infertility factors with the treatment of the GnRH agonist for
pituitary downregulation. Clinical and biochemical profiles of PCOS
patient and normal control groups are shown in Table I. To ascertain differences in
protein expression between normal and PCOS patients, we applied the
isoelectric focusing technique using differential-display proteomic
analysis methods described previously (20). Follicular fluid depleted of the
most abundant proteins with an immunoaffinity column revealed
better distinguishable spots on 2-DE gels (Fig. 1). The same separation method was
then performed on proteins extracted from the follicular fluid of
PCOS patients and healthy women as a control to identify proteins
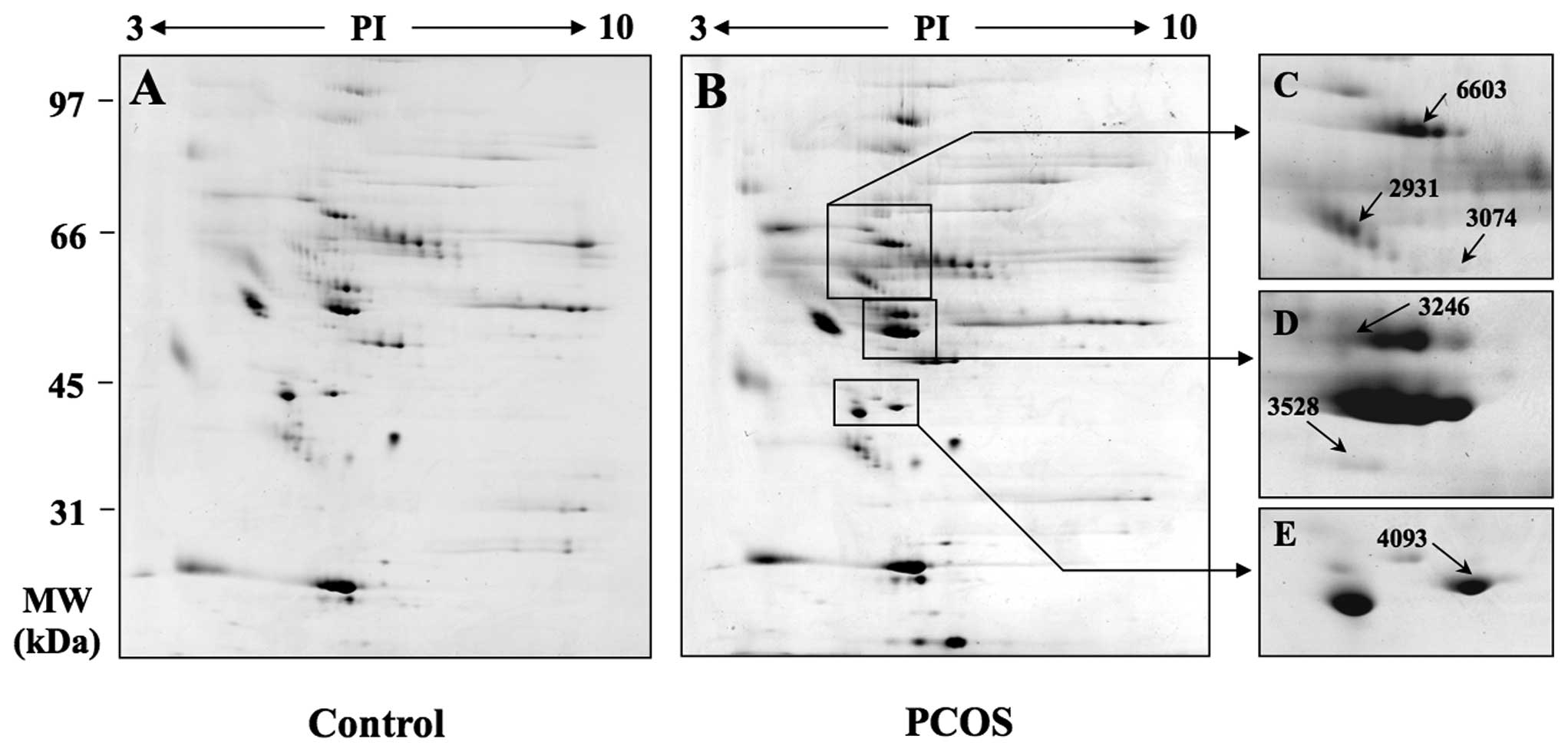
that were aberrantly expressed in PCOS patients (Fig. 2A and B).
Identification of differentially
expressed proteins in PCOS patients
When protein expression levels in the follicular
fluid from five PCOS patients with polycystic ovaries, oligo- or
amenorrhea and hyperandrogenism, were compared with five normal
women as a control, six spots were determined to be overexpressed
in the 2-DE analysis based on relative fold increase in volume
intensity (Fig. 2C–E). The
expression level was determined by examining the ratio of the
relative spot volume of a protein in the gel (Fig. 2F). The identification of these
different proteins was performed by MALDI-TOF-MS or nano LC-MS/MS.
The identified proteins included kininogen 1, cytokeratin 9,
antithrombin chain B, fibrinogen γ-chain, apoA-IV precursor and
A1BG (Table II).
 | Table IIProtein identities determined by mass
analysis. |
Table II
Protein identities determined by mass
analysis.
| Spot ID | Accession no. | Protein name | Scorea | Matched peptides
no. | Sequence
coverage | Matching
sequence | Theoretical
MW/pI |
|---|
| 2931 | gi|4504893 | Kininogen 1 | 89 | 11/44 (25%) | 21% | MKLITILFLCSR,
TVGSDTFYSFKYEIK, QVVAGLNFR, VQVVAGKKY FIDFVAR, | 47853/6.29 |
|
RPPGFSPFRSSRIGEIKEETTSHLR,
GRPPKAGAEPASER |
| 3074 | gi|435476 | Cytokeratin 9 | 83 | 17/184 (9%) | 41% |
LSRSGGGGGGGLGSGGSIRSSYSRFSSSGGR, | 62092/5.29 |
|
FSSSSGYGGGSSRVCGRGGGGSFGYSYGGGSGGGFSASSLGGGFGGGSR, |
|
VQALEEANNDLENK, |
|
NYSPYYNTIDDLKDQIVDLTVGNNKTLLDIDNTRMTLDDFRIKFEMEQNLR, |
|
SDLEMQYETLQEELMALKK, TLNDMRQEY
EQLIAK, |
| HGVQELEIELQSQLSKK,
GGSGGSYGGGGSGGGYGGGSGSR, |
|
GGSGGSHGGGSGFGGESGGSYGGGEEASGSGGGYGGGSGK |
| 3246 | gi|999514 | Chain B,
antithrombin | 68 | 17/184 (9%) | 53% |
HGSPVDICTAKPRDIPMNPMCIAR,
RVWELSKANSRFATTFYQHLADSK, | 48916/5.95 |
| TSDQIHFFFAK,
LQPLDFKENAEQSR, SKFS PENTR, |
|
VAEGTQVLELPFKGDDITMVLILPKPEK, |
|
VEKELTPEVLQEWLDELEEMMLVVHMPRFRIEDGFSLKEQLQDMGLVDLFSPEK, |
|
LPGIVAEGRDDLYVSDAFHKAFLEVNEEGSEAAASTAVVIAGR, |
|
ANRPFLVFIREVPLNTIIFMGR |
| 3528 | gi|182489 | Fibrinogen
γ-chain | 68 | 16/165 (10%) | 45% |
NLILYFYALLFLSSTCVAYVATRDNCCILDER, | 49450/5.61 |
|
TSEVKQLIKAIQLTYNPDESSKPNMIDAATLK, |
|
YEASILTHDSSIRYLQEIYNSNNQK,
EKVAQLEAQCQEPCKDTVQIHDITGK, |
| NWIQYKEGFGHLSPTGTT
EFWLGNEK, VELEDWNGRTSTADYAMFKVGPEADKYR, |
| TRWYSMK, TTM
KIIPFNRLTIGEGQQHHLGG AK |
| 4093 | gi|71773110 | Apolipoprotein A-IV
precursor | 277 | 25/50 (50%) | 50% |
SELTQQLNALFQDKLGEVNTYAGDLQKKLVPFATELHER, | 45344/5.28 |
| LLPHANEVSQK,
LEPYADQLRTQVSTQAEQLRRQLTPYAQR, |
|
ENADSLQASLRPHADELK, |
|
IDQNVEELKGRLTPYADEFKVKIDQTVEELRRSLAPYAQDTQEKLNHQLEGLTFQMK, |
| LAPLAEDVRGNLR,
SLAELGGHLDQQVEEFRRRVEPYGENFNKALVQQMEQLR, |
| LGPHAGDVEGHLSFLEK,
DKVNSFFSTFK |
| 6603 | gi|69990 |
α-1-B-glycoprotein | 82 | 16/135 (12%) | 45% |
RLETPDFQLFKNGVAQEPVHLDSPAIKHQFLLTGDTQGR, | 51908/5.65 |
|
SGLSTGWTQLSKLLELTGPK, GVTFLLRR, |
|
GEKELLVPRSSTSPDRIFFHLNAVALGDGGHYTCR, |
|
LHDNQNGWSGDSAPVELILSDETLPAPEFSPEPESGR,
FALVREDR, |
|
LELHVDGPPPRPQLRATWSGAVLAGR,
TPGAAANLELIFVGPQHAGNYR, |
| SWVPHTFE
SELSDPVELLVAES |
Western blot analysis using follicular
fluid and plasma
The follicular fluids were analyzed by western blot
analysis using the appropriate antibodies to validate their
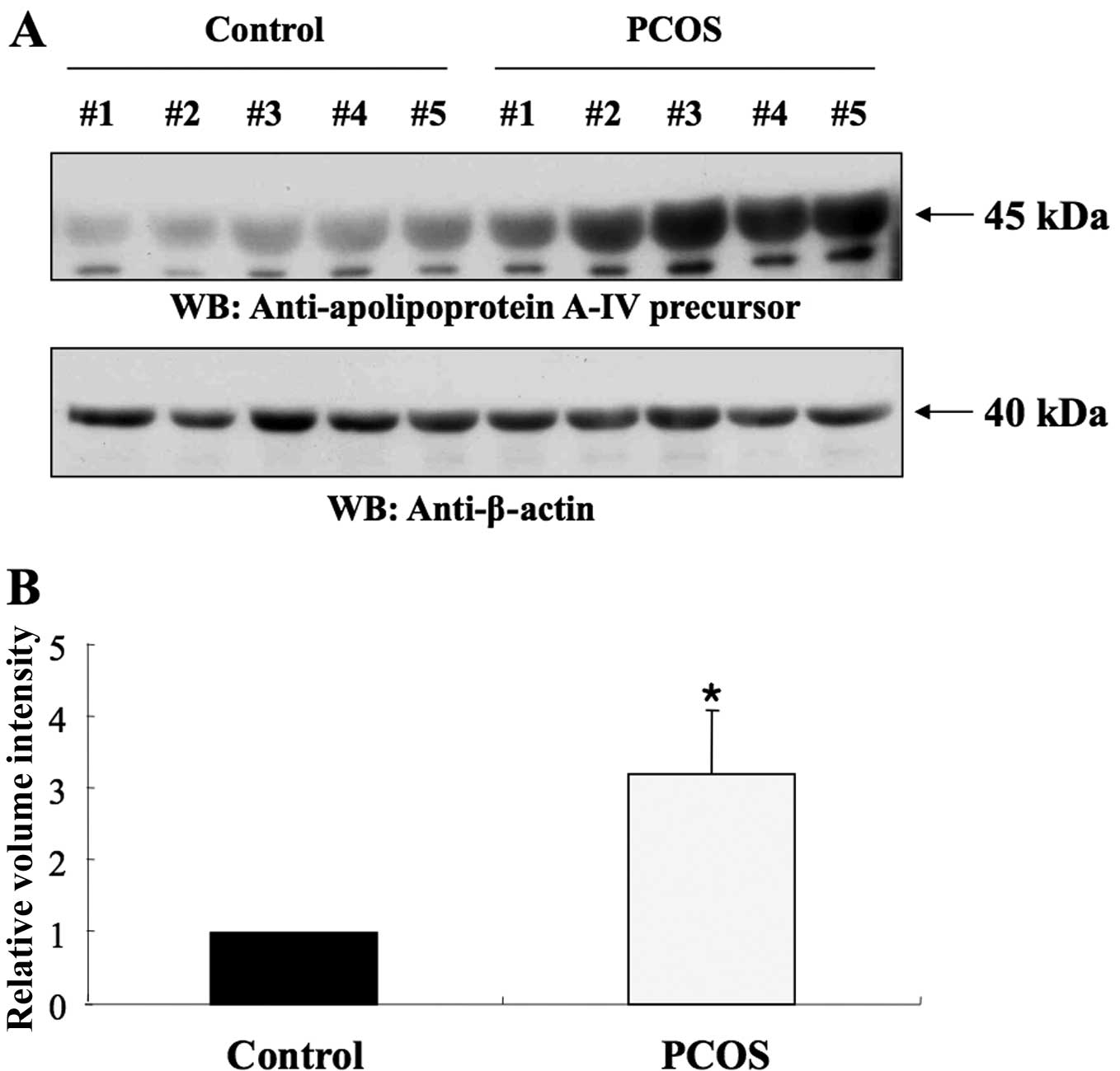
identities. As shown in Fig. 3A,
apoA-IV was detected in the follicular fluid of both the PCOS and
the control group. ApoA-IV (45 kDa) expression in PCOS patients was
significantly higher than that in controls (1.00 vs. 3.21±0.89,
P<0.005) (Fig. 3B). Fig. 3C shows the western blot analysis
of the A1BG (52 kDa), which was detected in the follicular fluid of
controls and PCOS patients. A1BG expression in PCOS patients was
also increased by ∼3-fold compared with the control (1.00 vs.
2.70±0.31, P<0.005) (Fig. 3D).
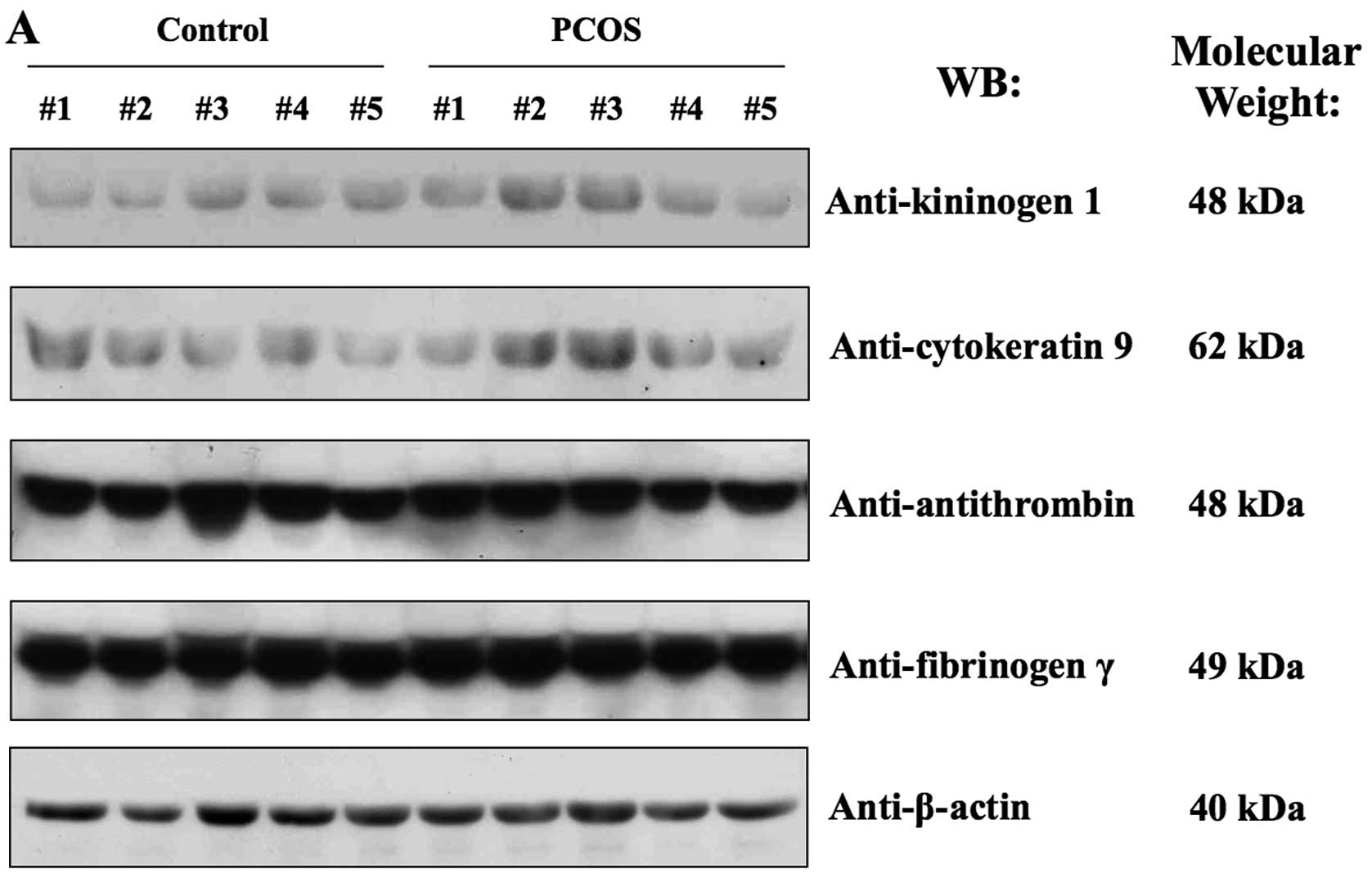
On the other hand, western blot analysis revealed individual
variations in the expression of kininogen 1, cytokeratin 9,
fibrinogen γ and antithrombin between the PCOS and the control
group (Fig. 4A). Fig. 4A shows that the expression level
of these proteins varied among normal or PCOS patients.
Since the plasma protein enrichment process occurs
during the production of follicular fluid, western blot analysis
was conducted using human sera derived from the control and PCOS
patients (Fig. 4B). Clinical and
biochemical profiles of PCOS patient and normal control groups are
shown in parentheses in Table I.
In human sera from PCOS patients, apoA-IV, A1BG, antithrombin and
fibrinogen γ were not significantly altered in their expression
levels (Fig. 4B). However,
cytokeratin 9, kininogen 1 and actin were not detected at the
protein level (data not shown).
Association study between apoA-IV and
PCOS using RFLP analysis
Several researchers have performed association
studies between various genes and PCOS for genetic analysis to
identify the etiology of PCOS. Therefore, in this report, an
association study for apoA-IV, which was highly expressed at
the protein level in follicular fluid and sera of PCOS patients,
was also conducted. RFLP analysis was performed on the samples
collected from 233 PCOS and 120 control patients. All PCOS patients
had clinical and biochemical characteristics in accordance with the
definition by ASRM/ESHRE (24);
149 patients (63.95%) showed polycystic ovaries and oligo- or
amenorrhea, 31 patients (13.3%) had oligo- or amenorrhea and
hyperandrogenism, 27 patients (11.59%) had polycystic ovaries and
hyperandrogenism, and 26 patients (11.16%) exhibited polycystic
ovaries, oligo- or amenorrhea, and hyper-androgenism. The level of
LH, DHEA-S and testosterone were significantly high in the PCOS
group (Table III).
 | Table IIIClinical and biochemical
characteristics of normal controls (n=120) and patients with PCOS
(n=233). |
Table III
Clinical and biochemical
characteristics of normal controls (n=120) and patients with PCOS
(n=233).
|
Characteristics | Normal controls
(n=120) | PCOS patients
(n=233) |
|---|
| Age (year) | 33.13±3.36
(29–36) | 32.41±3.04
(29–35) |
| BMI
(kg/m2) | 20.88±2.62 | 22.78±3.76 |
| Waist/hip ratio
(WHR) | 0.80±0.05 | 0.82±0.06 |
| Obesity | n=6 (5.45%) | n=44 (18.88%) |
| Polycystic ovaries
and oligo- or amenorrhea | n=0 (0.00%) | n=149 (63.95%) |
| Polycystic ovaries
and hyperandrogenism | n=0 (0.00%) | n=27 (11.59%) |
| Oligo- or
amenorrhea and hyperandrogenism | n=0 (0.00%) | n=31 (13.30%) |
| Polycystic ovaries,
oligo- or amenorrhea, and hyperandrogenism | n=0 (0.00%) | n=26 (11.16%) |
| FSH levels
(mlU/ml) | 6.13±2.23
(3.20–8.49) | 6.61±7.17
(3.00–11.50) |
| LH levels
(mlU/ml)a | 3.32±3.06
(1.00–17.02) | 6.63±4.78
(1.00–7.10) |
| E2
levels (pg/ml) | 71.49±298.60
(9.10–81.40) | 52.92±128.92
(5.00–219.90) |
| Prolactin levels
(ng/ml) | 13.79±10.47
(2.30–20.90) | 14.59±13.44
(4.10–46.40) |
| TSH levels
(μlU/ml) | 2.98±6.57
(0.46–5.47) | 2.59±5.08
(0.03–4.06) |
| DHEA-S levels
(μg/dl)a | 168.10±70.35
(45.30–377.20) | 194.86±77.93
(67.20–257.40) |
| Testosterone
(ng/ml)a | 0.23±0.14
(0.06–0.86) | 0.38±0.22
(0.01–0.54) |
In the RFLP analysis, the association of
apoA-IV and PCOS was confirmed by the G→T substitution,
which led to a change from glutamine to histidine, by restriction
enzyme Fnu4HI (Fig. 5).
While restriction enzyme Fnu4HI cleaved normal DNA fragments
into 101 and 26 bp (Fig. 5A),
variant DNA fragments by the G→T substitution were not cleaved
(Fig. 5B).
The homozygote of the variant allele (TT) was not
detected in the healthy control group and heterozygote genotype
(GT) was only observed in three control patients. Of note, 50 PCOS
patients (21.5%) had the TT variant homozygote genotype, and 45
PCOS patients (19.3%) had the GT variant heterozygote genotype
(Table IV). Statistical analysis
using HapAnalyzer and SAS programs identified that the Gln360His
variant was significantly associated with PCOS in dominant
(P<0.05) and co-dominant (P<0.05) (Table IV). However, the genotype of PCOS
patients used for proteomic analysis only displayed the GG
homozygote.
 | Table IVAllele frequencies of the Gln360His
ApoA-IV gene. |
Table IV
Allele frequencies of the Gln360His
ApoA-IV gene.
| PCOS patient
group | Control group |
|---|
| Genotype | | |
| GG | 138 (59.2%) | 117 (97.5%) |
| GT | 45 (19.3%) | 3 (2.5%) |
| TT | 50 (21.5%) | 0 (0.0%) |
| Co-dominant | OR (95% CI) = 15.68
(5.14–47.89), P-value <0.001 | |
| Dominant | OR (95% CI) = 28.71
(8.83–93.33), P-value <0.001 | |
| Recessive | OR (95% CI) = N/A,
P-value =0.997 | |
| Total | OR (95% CI) = 37.33
(11.74–118.70), P-value <0.001 | |
Discussion
PCOS is a common endocrinopathy affecting 6–10% of
women of reproductive age (1).
There is clear evidence of the underlying genetic cause for PCOS
based on familiar clustering of cases. Most studies have been
consistent with an autosomal dominant pattern of inheritance.
However, it has been difficult to draw solid conclusions from
several of these studies due to the small sample sizes, errors in
statistical analysis, and differences in diagnostic criteria, an
inevitable consequence of PCOS being a heterogeneous disorder. It
has been suggested that PCOS develops as a consequence of a primary
genetic abnormality in ovarian androgen production, interacting
with environmental factors or other factors causing
hyperinsulinemia.
Body fluids such as follicular fluid, amniotic fluid
and cervical-vaginal fluid have been used to investigate the
pathogenesis for pregnancy and birth including recurrent pregnancy
loss (RPL), preeclampsia, intra-amniotic infection (20–23,25,26). In order to investigate novel
etiology factors of PCOS, the proteins expressed in the follicular
fluid of PCOS patients were investigated using 2-DE analysis, and
six proteins were found to be aberrantly expressed. These proteins
were kininogen 1, cytokeratin 9, antithrombin chain B, fibrinogen
γ, apoA-IV precursor and A1BG (Fig.
3). However, it is unclear if these proteins were induced due
to the ovarian hyperstimulation caused by IVF therapy even though
control samples did not show upregulation of these proteins.
ApoA-IV is a 45 kDa glycoprotein triglyceride-rich
glycoprotein (27). It is
expressed in the small intestine and its synthesis and secretion
increases in response to fatty meals (28). ApoA-IV has several proposed roles,
including lipid transport, lipoprotein metabolism (29), control of food intake (30) and gastric function (31). apoA-IV has been shown to be
antiatherogenic (32), and there
is also evidence supporting apoA-IV antioxidant activity (32–34). Two mechanisms have been suggested
for apoA-IV’s antiatherogenic action: enhancement of cellular lipid
efflux (i.e., ‘reverse cholesterol transport’) (35) and antioxidant activity (33,34).
We showed that the expression of apoA-IV was
significantly increased in the follicular fluid from PCOS patients.
One of the possibilities is that this may result in aberration of
lipoprotein metabolism and transport, causing the generation of
polycystic ovaries. Identifying the etiology of PCOS is extremely
difficult as it is caused by a combination of hormonal, genetic and
environmental factors. For these reasons, a combination of
proteomic and genetic analyses has been used to identify new
factors associated with these diseases (35–37). Several researchers have
investigated a diverse range of genes that may be associated with
PCOS to identify the etiology of this disease, and apoA-IV has also
been studied in numerous diseases, including obesity, using genetic
analysis (38). Gln360His and
Thr347Ser are well-studied functional polymorphisms of apoA-IV. It
has been demonstrated that these affect obesity and
lipid/lipoprotein metabolism (39,40). In particular, Gln360His
polymorphism is related with type 1 diabetes mellitus (T1DM) and
obesity (41,42).
In women with PCOS, insulin resistance or deficiency
is one of the main characteristics of patients with obesity
(43,44). Therefore, we performed a single
nucleotide polymorphism (SNP) study for Gln360His polymorphism of
apoA-IV to investigate its association with PCOS. In these
experiments, this polymorphism was shown to be highly related to
PCOS. The homozygote of the variant allele was not detected in the
control group. This suggests that the polymorphism affects the
development of PCOS by regulating apoA-IV protein expression or by
impacting the function of the protein. However, the relationship
between the aberrant expression of apoA-IV in follicular fluid and
Gln360His polymorphism and their role in the generation of PCOS
remains unclear. The study had certain challenges due to the fact
that PCOS is an endocrine disorder influenced by diverse
factors.
We also determined the expression level of
α-1-B-glycoprotein (A1BG) by western blot analysis, and found that
the expression of this protein is increased in PCOS patients
relative to healthy women. A1BG is a known plasma protein with
unknown function and a member of the immunoglobulin superfamily.
Little is known about the biological functions of A1BG with regard
to infertility diseases. Therefore, further studies are needed to
explain why the A1BG protein is increased in PCOS patients.
Although we showed that the expression of kininogen
1 and cytokeratin 9 was increased in the follicular fluids from
PCOS patients by 2-DE analysis, western blot analysis showed
individual variation between PCOS and normal patients. However, the
expression level also varied among normal or PCOS patients,
suggesting that these proteins may be involved in the etiology of
PCOS in certain cases. Therefore, continuous investigation with
2-DE analysis is required for the identification of proteins
associated with the pathogenesis of PCOS.
In addition, we showed that the expression of
antithrombin and fibrinogen γ was increased in the follicular
fluids from PCOS patients by 2-DE analysis. Western blot analysis,
however, showed individual variation between PCOS and normal
patients. In the case of antithrombin, Tsanadis et al
(45) reported that the
concentration difference was not statistically significant between
women with PCOS and the control women, which is in agreement with
our data. There are several studies in the literature regarding the
concentration of fibrinogen in females with PCOS. Atiomo et
al (46) reported a
statistically higher concentration of fibrinogen in women with PCOS
than in normal control women. Dahlgren et al (47) found that the concentration of
fibrinogen was lower in females with PCOS. Kelly et al
(48) did not find statistically
significant differences between these two groups. Considering the
discrepancies in the literature with regard to the expression level
of fibrinogen in PCOS patients, further studies are required in
order to more accurately understand the function of fibrinogen in
PCOS patients.
Acknowledgements
This study was supported by a grant of
the Korea Health 21 R&D Project, Ministry of Health and
Welfare, Republic of Korea (01-PJ10-PG6-01GN13-0002) and the
Biomedical Proteome Research Center (A030003).
References
|
1
|
Goodarzi MO, Dumesic DA, Chazenbalk G and
Azziz R: Polycystic ovary syndrome: etiology, pathogenesis and
diagnosis. Nat Rew Endocrinol. 7:219–231. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hart R, Hickey M and Franks S:
Definitions, prevalence and symptoms of polycystic ovaries and
polycystic ovary syndrome. Best Pract Res Clin Obstet Gynaecol.
18:671–683. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Pasquali R, Stener-Victorin E, Yildiz BO,
Duleba AJ, Hoeger K, Mason H, Homburg R, Hickey T, Franks S,
Tapanainen JS, et al: PCOS Forum: research in polycystic ovary
syndrome today and tomorrow. Clin Endocrinol (Oxf). 74:424–433.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dumesic DA, Abbott DH and Padmanabhan V:
Polycystic ovary syndrome and its developmental origins. Rev Endocr
Metab Disord. 8:127–141. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hourvitz A, Kuwahara A, Hennebold JD,
Tavares AB, Negishi H, Lee TH, Erickson GF and Adashi EY: The
regulated expression of the pregnancy-associated plasma protein-A
in the rodent ovary: a proposed role in the development of dominant
follicles and of corpora lutea. Endocrinology. 143:1833–1844. 2002.
View Article : Google Scholar
|
|
6
|
Teixeira Filho FL, Baracat EC, Lee TH, Suh
CS, Matsui M, Chang RJ, Shimasaki S and Erickson GF: Aberrant
expression of growth differentiation factor-9 in oocytes of women
with polycystic ovary syndrome. J Clin Endocrinol Metab.
87:1337–1344. 2002.PubMed/NCBI
|
|
7
|
Munir I, Yen HW, Baruth T, Tarkowki R,
Azziz R, Magoffin DA and Jakimiuk AJ: Resistin stimulation of
17alpha-hydroxylase activity in ovarian theca cells in vitro:
relevance to polycystic ovary syndrome. J Clin Endocrinol Metab.
90:4852–4857. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wickenheisser JK, Nelson-Degrave VL and
McAllister JM: Dysregulation of cytochrome P450 17alpha-hydroxylase
messenger ribonucleic acid stability in theca cells isolated from
women with polycystic ovary syndrome. J Clin Endocrinol Metab.
90:1720–1727. 2005. View Article : Google Scholar
|
|
9
|
Goodarzi MO, Jones MR, Chen YD and Azziz
R: First evidence of genetic association between AKT2 and
polycystic ovary syndrome. Diabetes Care. 31:2284–2287. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim JJ, Choi YM, Hong MA, Hwang SS, Yoon
SH, Chae SJ, Jee BC, Ku SY, Kim JG and Moon SY:
Phosphatidylinositol 3-kinase p85alpha regulatory subunit gene
Met326Ile polymorphism in women with polycystic ovary syndrome. Hum
Reprod. 24:1184–1190. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gu BH and Baek KH: Pro12Ala and His447His
polymorphisms of PPAR-gamma are associated with polycystic ovary
syndrome. Reprod Biomed Online. 18:644–650. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Choi SW, Gu BH, Ramakrishna S, Park JM and
Baek KH: Association between a single nucleotide polymorphism in
MTHFR gene and polycystic ovary syndrome. Eur J Obstet Gynecol
Reprod Biol. 145:85–88. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fortune JE: Ovarian follicular growth and
development in mammals. Biol Reprod. 50:225–232. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chiu PC, Koistinen R, Koistinen H, Seppala
M, Lee KF and Yeung WS: Zona-binding inhibitory factor-1 from human
follicular fluid is an isoform of glycodelin. Biol Reprod.
69:365–372. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Coy P, Gadea J, Romar R, Matas C and
Garcia E: Effect of in vitro fertilization medium on the acrosome
reaction, cortical reaction, zona pellucida hardening and in vitro
development in pigs. Reproduction. 124:279–288. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yao Y, Ho P and Yeung WS: Effects of human
follicular fluid on the capacitation and motility of human
spermatozoa. Fertil Steril. 73:680–686. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang TH, Chang CL, Wu HM, Chiu YM, Chen CK
and Wang HS: Insulin-like growth factor-II (IGF-II), IGF-binding
protein-3 (IGFBP-3), and IGFBP-4 in follicular fluid are associated
with oocyte maturation and embryo development. Fertil Steril.
86:1392–1401. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu YT, Tang L, Cai J, Lu XE, Xu J, Zhu XM,
Luo Q and Huang HF: High bone morphogenetic protein-15 level in
follicular fluid is associated with high quality oocyte and
subsequent embryonic development. Hum Reprod. 22:1526–1531. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rosen MP, Zamah AM, Shen S, Dobson AT,
McCulloch CE, Rinaudo PF, Lamb JD and Cedars MI: The effect of
follicular fluid hormones on oocyte recovery after ovarian
stimulation: FSH level predicts oocyte recovery. Reprod Biol
Endocrinol. 7:352009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim YS, Kim MS, Lee SH, Choi BC, Lim JM,
Cha KY and Baek KH: Proteomic analysis of recurrent pregnancy loss:
Identification of an inadequately expressed set of proteins in
human follicular fluid. Proteomics. 6:3445–3454. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kim MS, Gu BH, Song S, Choi BC, Cha DH and
Baek KH: ITI-H4, as a biomarker in the serum of recurrent pregnancy
loss (RPL) patients. Mol Biosyst. 7:1430–1440. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Atiomo W, Khalid S, Parameshweran S, Houda
M and Layfield R: Proteomic biomarkers for the diagnosis and risk
stratification of polycystic ovary syndrome: a systematic review.
BJOG. 116:137–143. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jarkovska K, Martinkova J, Liskova L,
Halada P, Moos J, Rezabek K, Gadher SJ and Kovarova H: Proteome
mining of human follicular fluid reveals a crucial role of
complement cascade and key biological pathways in women undergoing
in vitro fertilization. J Proteome Res. 9:1289–1301. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Revised 2003 consensus on diagnostic
criteria and long health risks related to polycystic ovary syndrome
(PCOS). Hum Reprod. 19:41–47. 2004. View Article : Google Scholar
|
|
25
|
Gravett MG, Thomas A, Schneider KA, Reddy
AP, Dasari S, Jacob T, Lu X, Rodland M, Pereira L, Sadowsky DW, et
al: Proteomic analysis of cervical vaginal fluid: identification of
novel biomarkers for detection of intra-amniotic infection. J
Proteome Res. 6:89–96. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Park JS, Oh KJ, Norwitz ER, Han JS, Choi
HJ, Seong HS, Kang YD, Park CW, Kim BJ, Jun JK, et al:
Identification of proteomic biomarkers of preeclampsia in amniotic
fluid using SELDI-TOF mass spectrometry. Reprod Sci. 15:457–468.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sadeghi M, Roohafza H, Afshar H, Rajabi F,
Ramzani M, Shemirani H and Sarafzadeghan N: Relationship between
depression and apolipoproteins A and B: a case-control study.
Clinics (Sao Paulo). 66:113–117. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ferrer F, Nazih H, Zair Y, Krempf M and
Bard JM: Postprandial changes in the distribution of apolipoprotein
AIV between apolipoprotein B- and non apolipoprotein B-containing
lipoproteins in obese women. Metabolism. 52:1537–1541. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Araki S, Okazaki M and Goto S: Impaired
lipid metabolism in aged mice as revealed by fasting-induced
expression of apolipoprotein mRNAs in the liver and changes in
serum lipids. Gerontology. 50:206–215. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qin X and Tso P: The role of
apolipoprotein AIV on the control of food intake. Curr Drug
Targets. 6:145–151. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Whited KL, Lu D, Tso P, Kent Lloyd KC and
Raybould HE: Apolipoprotein A-IV is involved in detection of lipid
in the rat intestine. J Physiol. 569:949–958. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Culnan DM, Cooney RN, Stanley B and Lynch
CJ: Apolipoprotein A-IV, a putative satiety/antiatherogenic factor,
rises after gastric bypass. Obesity. 17:46–52. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ostos MA, Conconi M, Vergnes L, Baroukh N,
Ribalta J, Girona J, Caillaud JM, Ochoa A and Zakin MM:
Antioxidative and antiatherosclerotic effects of human
apolipoprotein A-IV in apolipoprotein E-deficient mice.
Arterioscler Thromb Vasc Biol. 21:1023–1028. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Broedl UC, Schachinger V, Lingenhel A,
Lehrke M, Stark R, Seibold F, Göke B, Kronenberg F, Parhofer KG and
Konrad-Zerna A: Apolipoprotein A-IV is an independent predictor of
disease activity in patients with inflammatory bowel disease.
Inflamm Bowel Dis. 13:391–397. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Remaley AT, Stonik JA, Demosky SJ, Neufeld
EB, Bocharov AV, Vishnyakova TG, Eggerman TL, Patterson AP,
Duverger NJ, Santamarina-Fojo S, et al: Apolipoprotein specificity
for lipid efflux by the human ABCAI transporter. Biochem Biophys
Res Commun. 280:818–823. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Stylianou IM, Affourtit JP, Shockley KR,
Wilpan RY, Abdi FA, Bhardwaj S, Rollins J, Churchill GA and Paigen
B: Applying gene expression, proteomics and single-nucleotide
polymorphism analysis for complex trait gene identification.
Genetics. 178:1795–1805. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jin EH, Shim SC, Kim HG, Chae SC and Chung
HT: Polymorphisms of COTL1 gene identified by proteomic approach
and their association with autoimmune disorders. Exp Mol Med.
41:354–361. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Weinberg RB: Apolipoprotein A-IV
polymorphisms and diet-gene interactions. Curr Opin Lipidol.
13:125–134. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Vincent S, Planells R, Defoort C, Bernard
MC, Gerber M, Prudhomme J, Vague P and Lairon D: Genetic
polymorphisms and lipoprotein responses to diets. Proc Nutr Soc.
61:427–434. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Bai H, Liu R, Liu Y, Saku K and Liu BW:
Distribution and effect of apo A-IV genotype on plasma lipid and
apolipoprotein levels in a Chinese population. Acta Cardiol.
63:315–322. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Heilbronn LK, Noakes M, Morris AM, Kind KL
and Clifton PM: 360His polymorphism of the apolipoproteinA-IV gene
and plasma lipid response to envergy restricted diets in overweight
subjects. Atherosclerosis. 150:187–192. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kretowski A, Hokanson JE, McFann K, Kinney
GL, Snell-Bergeon JK, Maahs DM, Wadwa RP, Eckel RH, Ogden LG and
Garg SK: The apolipoprotein A-IV Gln360His polymorphism predicts
progression of coronary artery calcification in patients with type
1 diabetes. Diabetologia. 49:1946–1954. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Antoine HJ, Pall M, Trader BC, Chen YD,
Azziz R and Goodarzi MO: Genetic variants in peroxisome
proliferator-activated receptor gamma influence insulin resistance
and testosterone levels in normal women, but not those with
polycystic ovary syndrome. Fertil Steril. 87:862–869. 2007.
View Article : Google Scholar
|
|
44
|
Codner E and Cassorla F: Puberty and
ovarian function in girls with type 1 diabetes mellitus. Horm Res.
71:12–21. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Tsanadis G, Vartholomatos G, Korkontzelos
I, Avgoustatos F, Kakosimos G, Sotiriadis A, Tatsioni A,
Eleftheriou A and Lolis D: Polycystic ovarian syndrome and
thrombophilia. Hum Reprod. 17:314–319. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Atiomo WU, Bates SA, Condon JE, Shaw S,
West JH and Prentice AG: The plasminogen activator system in women
with polycystic ovary syndrome. Fertil Steril. 69:236–241. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Dahlgren E, Janson PO, Johansson S,
Lapidus L, Lindstedt G and Tengborn L: Hemostatic and metabolic
variables in women with polycystic ovary syndrome. Fertil Steril.
61:455–460. 1994.PubMed/NCBI
|
|
48
|
Kelly CJ, Lyall H, Petrie JR, Gould GW,
Connell JM, Rumley A, Lowe GD and Sattar N: A specific elevation in
tissue plasminogen activator antigen in women with polycystic
ovarian syndrome. J Clin Endocrinol Metab. 87:3287–3290. 2002.
View Article : Google Scholar : PubMed/NCBI
|