Introduction
Respiratory syncytial virus (RSV) is a
non-segmented, negative-stranded RNA virus and a member of the
Paramyxoviridae family. RSV is the leading cause of serious
respiratory infections in children as well as in elderly and
immune-suppressed individuals (1,2).
The role of reactive oxygen species (ROS) as mediators of the
virus-induced epithelial damage in RSV infected mice has been
previously reported (3,4). Oxidative stress is one of the
components of the pathophysiology of chronic obstructive pulmonary
disease (5). RSV infection led to
a ROS induction, which increases the expression of pro-inflammatory
molecules such as interleukin-8 (IL-8), IL-6, CCL5 or CXCL10
(6). This production of
pro-inflammatory cytokines induced by RSV infection resulted in
type 1 and 2 cytokine imbalance. It was strongly suggested that
excess type 2 and/or deficient type 1 immune responses were
involved in the pathogenesis of RSV bronchiolitis (7).
Herbal medicines have been previously used in humans
to treat medical illness or to improve physical performance.
Panax ginseng is one of the most well-known herbal medicines
that have been consumed for thousands of years. Experimental
evidence suggests that ginseng modulates the host immune system and
improves outcomes of inflammatory human diseases (8,9).
Ginseng or its component ginsenoside protopanaxatriol was also
reported to protect endothelial cells by scavenging hydroxyl
radicals and modulating the antioxidant defense systems such as
superoxide dismutase and glutathione peroxidase enzymes (10–12). Ginsenosides of ginseng were shown
to protect human endothelial cells against influenza H9N2-induced
inflammation and apoptosis (13).
However, the potential antiviral effects of ginseng on RSV
infection remain unknown.
In this study, we investigated the effect of Korean
red ginseng extract (KRGE) on RSV replication, on RSV-induced
cytokine expression, and RSV-induced cellular oxidative stress in a
human epithelial cell line. In addition, we evaluated the possible
in vivo antiviral effects of KRGE on clearing lung viral
loads and host immune responses following RSV infection in a mouse
model.
Materials and methods
Cells, virus and reagents
RSV A2 strain (a biosafety level 2 human pathogen)
and HEp2 cells were used as previously described (14,15). The human alveolar type II-like
epithelial cell line (A549 cell), was kindly provided by Dr
Jae-Hyang Lim (Center for Inflammation, Immunity and Infection,
Institute for Biomedical Sciences, Georgia State University). KRGE,
a concentrated form of the commercial ginseng product was kindly
provided by Korea Ginseng Corporation (Daejeon, Korea). Briefly,
fresh roots of the Panax ginseng were washed, steamed at
100°C, and dried. The dried red ginseng roots were boiled in water
for 3 h and the supernatants were concentrated. This preparation
was designated as ‘KRGE’ and contained ~36% water content. Fetal
bovine serum (FBS), penicillin-streptomycin, and Dulbecco’s
modified Eagle’s medium (DMEM) were purchased from Gibco (Grand
Island, NY, USA). 2′,7′-Dichlorodihydrofluorescein diacetate
(H2DCFDA) was purchased from Molecular Probes (Carlsbad,
CA, USA).
Cell viability, RSV immunoplaque, and
cytopathogenic effect (CPE) assays
Cell viability was determined using the
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
assay, which is based on the reduction of a tetrazolium salt by
mitochondrial dehydrogenase in viable cells (16). Cell viability was expressed as a
percentage of the control cells in the absence of RSV infection.
RSV titers were determined by an immunoplaque assay (14,15). Virus stock or lung homogenates
from infected mice were serially diluted and added to HEp2 cells
that were grown to confluence. After 3 days of incubation, the
infected cells were fixed with ice-cold acetone-methanol, and
air-dried. Anti-F monoclonal antibody (Millipore, Billerica, MA,
USA) and HRP-conjugated anti-mouse IgG antibodies (Southern
Biotech, Birmingham, AL, USA) were used. Plaques were developed
using a DAB substrate (Invitrogen, Grand Island, NY, USA). The CPE
assay was performed as previously described (17). Treatment with KRGE was initiated 1
day prior to RSV infection. Confluent cell monolayer A549 cells
grown in 96-well plates were infected with RSV, incubated for
another two days in the presence or absence of KRGE, and
virus-induced CPE was recorded.
Reverse transcriptase-polymerase chain
reaction (RT-PCR)
Total RNA was isolated using an RNeasy mini kit
(Qiagen), according to the manufacturer’s instructions. Relative
quantities of mRNA for each gene of interest were determined by
semi-quantitative RT-PCR, as previously described (18). In brief, total RNA (1 μg) from
each sample was used for the preparation of cDNA by using oligo(dT)
primers and SuperScript III RT (Invitrogen). mRNA level of IL-6,
IL-8 and 18S rRNA was determined by using the oligonucleotides as
primers: IL-6 sense, 5′-GACAGCCACTCACCTCTTCA-3′ and antisense,
5′-CATCTTTGGAAGGTTCAGGTTGT-3′; IL-8 sense,
5′-CAGCCTTCCTGATTTCTGC-3′ and antisense,
5′-ACTTCTCCACAACCCTCTGC-3′; 18S rRNA sense,
5′-ATCCTGCCAGTAGCATATGC-3′ and antisense,
5′-ACCCGGGTTGGTTTTGATCTG-3′. RT-PCR products were visualized on 2%
agarose gels containing ethidium bromide. 18S rRNA was the internal
control. The relative quantity of PCR products was expressed as
fold increase relative to phosphate-buffered saline (PBS)
controls.
Intracellular ROS measurement and image
analysis
The change in fluorescence resulting from the
oxidation of the fluorescent probe H2DCFDA was used to
evaluate the level of intracellular ROS (19). After RSV and KRGE treatments, the
cells were incubated with 50 mM of the fluorescent probe
H2DCFDA. The degree of fluorescence was detected at an
excitation and emission of 485 and 535 nm, respectively, using a
microplate spectrofluorometer (Molecular Devices Corp., Sunnyvale,
CA, USA). Coverslip-loaded 6-well plates were used for image
collection during cell culture. After various treatments,
H2DCFDA solution was added to each well of the plate,
which was incubated for 2 h at 37°C. Images of the stained cells
were collected using a fluorescence microscope (Nikon, Melville,
NY, USA).
Treatment of mice with KRGE and RSV
virus
KRGE was dissolved in sterile PBS and filtered
through 0.4 μm Millipore membrane. For animal experiments, 8- to
10-week-old female BALB/c mice (Harlan Laboratories, Indianapolis,
IN, USA) were lightly anesthetized by isoflurane and then orally
administered KRGE in water feeding at a dose of 25 mg/kg/day for 60
days. In addition, 8- to 10-week-old female mice serving as
controls were treated with PBS at the beginning of this experiment.
This dose was based on previous studies with regard to oral intake
of ginseng extracts in humans and mouse/kg of body weight (20,21). To determine the effects of KRGE
treatment on RSV infection, the mice (n=5/group) were anesthetized
by isoflurane inhalation and intranasally infected with RSV A2
(2×106 PFU) in a volume of 50 μl. All the animal
experiments presented in this study were approved by the Georgia
State University (GSU) Institutional Animal Care and Use Committee
review board and carried out in the animal biosafety level 2
facility.
Lung virus titer and cytokine assays
Mice were anesthetized with isoflurane and
exsanguinated after severing of the right caudal artery. The
individual lungs were removed aseptically at day 5 post challenge,
and lung extracts were prepared as homogenates after challenge
using frosted glass slides (15).
Lung homogenates for viral titers from individual mice were
prepared using a mechanical tissue grinder with 1.5 ml of PBS. The
homogenates were centrifuged at 1,100 × g for 10 min to collect
supernatants. Virus titers in the supernatants were determined by
immunoplaque assay. Ready-Set-Go IL-4, IL-5, IL-13, tumor necrosis
factor-α (TNF-α), and interferon-γ (IFN-γ) kits (eBioscience) were
used to detect cytokine levels in bronchoalveolar fluids (BALF)
following the manufacturer’s recommended procedures (22).
Preparation of bronchoalveolar lavage
(BAL) and flow cytometric analysis
Five days after RSV infection, the mice were
sacrificed using carbon dioxide inhalation to collect BAL fluids
and lung samples. BAL fluid samples were obtained by infusing 1 ml
of PBS into the lungs via the trachea using a 25-gauge catheter
(Exelint International Co., Los Angeles, CA, USA) as previously
described (23,24). The cells from BAL fluids were
pooled (n=5) and then stimulated with phorbol myristate acetate (50
ng/ml) and ionomycin (500 ng/ml) for 4 h. After staining with
surface antibodies (anti-CD3, CD4, CD8α, CD11b and CD11c antibodies
from eBiosciences), intracellular IFN-γ cytokine staining was
followed according to the manufacturer’s instructions (BD
Cytofix/Cytoperm™ Fixation/Permeabilization Solution kit). The
percentage of gated cells was calculated by FlowJo software (Tree
Star Inc., Ashland, OR, USA).
Statistical analysis
Data are expressed as the means ± standard error
(SE), and the results were taken from at least three independent
experiments performed in triplicate. The data were analyzed by the
Student’s t-test to evaluate significant differences. P<0.05 was
considered statistically significant.
Results
Influence of KRGE on RSV replication in
human epithelial cells
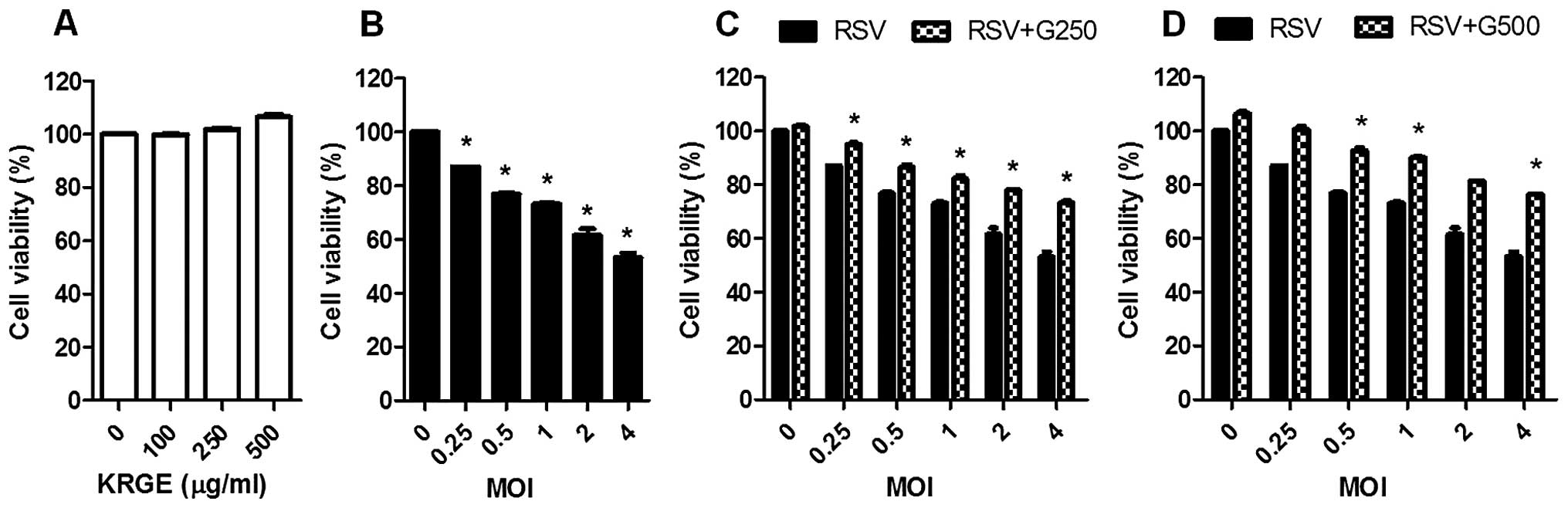
To investigate the effects of KRGE on RSV
replication in human epithelial cells, confluent A549 cells were
infected with RSV at different multiplicity of infection (MOI) in
the presence or absence of KRGE treatment. KRGE did not affect A549
cell viability (Fig. 1A). After
RSV infection, A549 cells exhibited marked morphological changes,
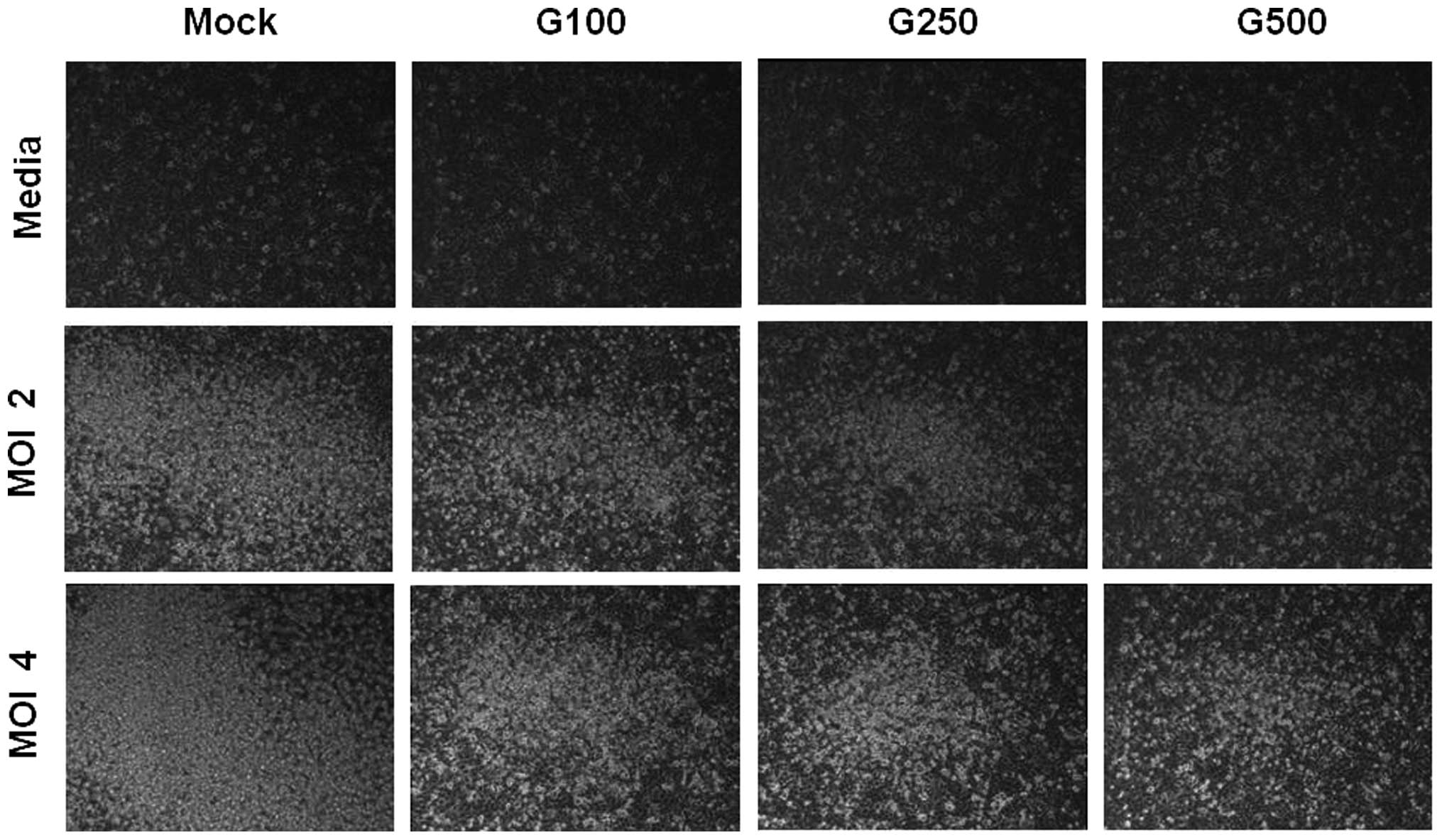
suggesting cell death in a MOI-dependent manner (Fig. 1B), such as cell rounding and
detachment (Fig. 2). When A549
cells were treated with KRGE, RSV-induced cell death was partially
reduced and cell survival was increased at the concentrations of
250 and 500 μg/ml (Fig. 1C and
D). CPE was also examined under the microscope after KRGE
treatment and RSV infection. KRGE treatments resulted in lower CPE
levels as evidenced by reduction of the number of floating cells
due to RSV infection (Fig. 2).
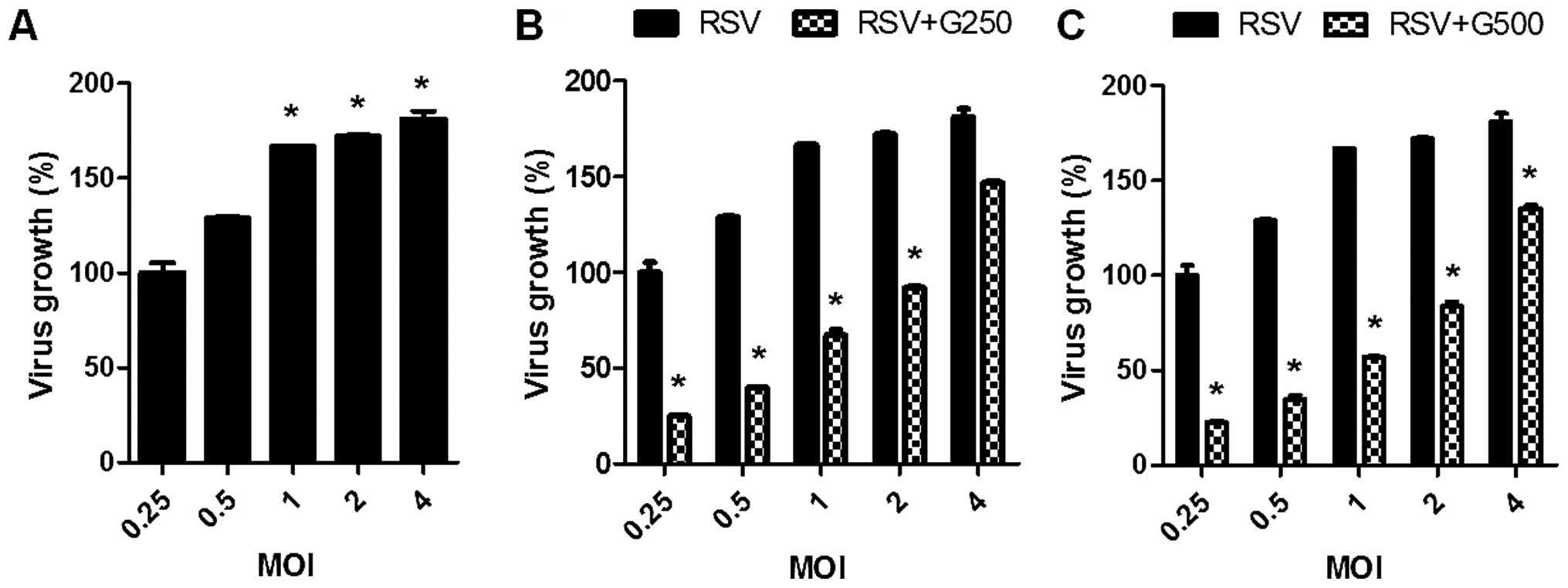
Higher viral growth was observed by increasing the MOI (Fig. 3A). In the presence of KRGE, much
lower levels of RSV growth were observed. The magnitude of growth
inhibition was more pronounced at lower MOIs of RSV (Fig. 3B and C). Therefore, these results
showed that KRGE treatment reduced cell death by RSV infection.
More importantly, KRGE treatment significantly inhibited the growth
of RSV in vitro A549 cell cultures.
Influence of KRGE on inflammatory
cytokine production in human epithelial cells
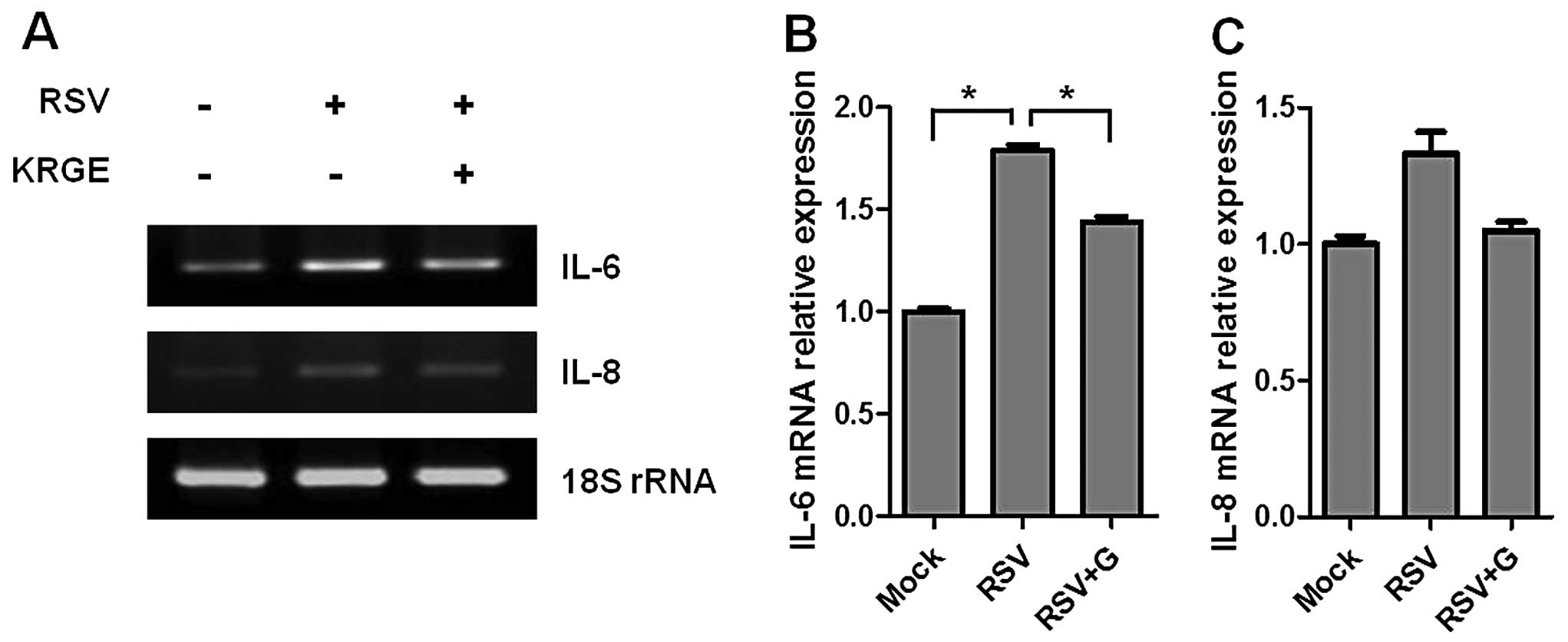
To investigate RSV-induced cytokine expression,
confluent cell layers were infected with RSV at a MOI of 1 in the
presence or absence of KRGE treatment. Total RNA was extracted from
the cells, and the gene expression was evaluated by RT-PCR.
RSV-infected A549 cells significantly increased the expression of
IL-6 and IL-8 cytokines compared to the mock-treated cells. IL-6
was expressed more prominently than IL-8 cytokine. KRGE treatment
resulted in lower levels of RSV-induced expression of IL-6 cytokine
gene in RSV-infected human epithelial cells (Fig. 4). KRGE treatment also showed a
trend of lowering IL-8 cytokine. Therefore, these results suggested
that KRGE inhibits the production of inflammatory cytokines induced
by RSV infection.
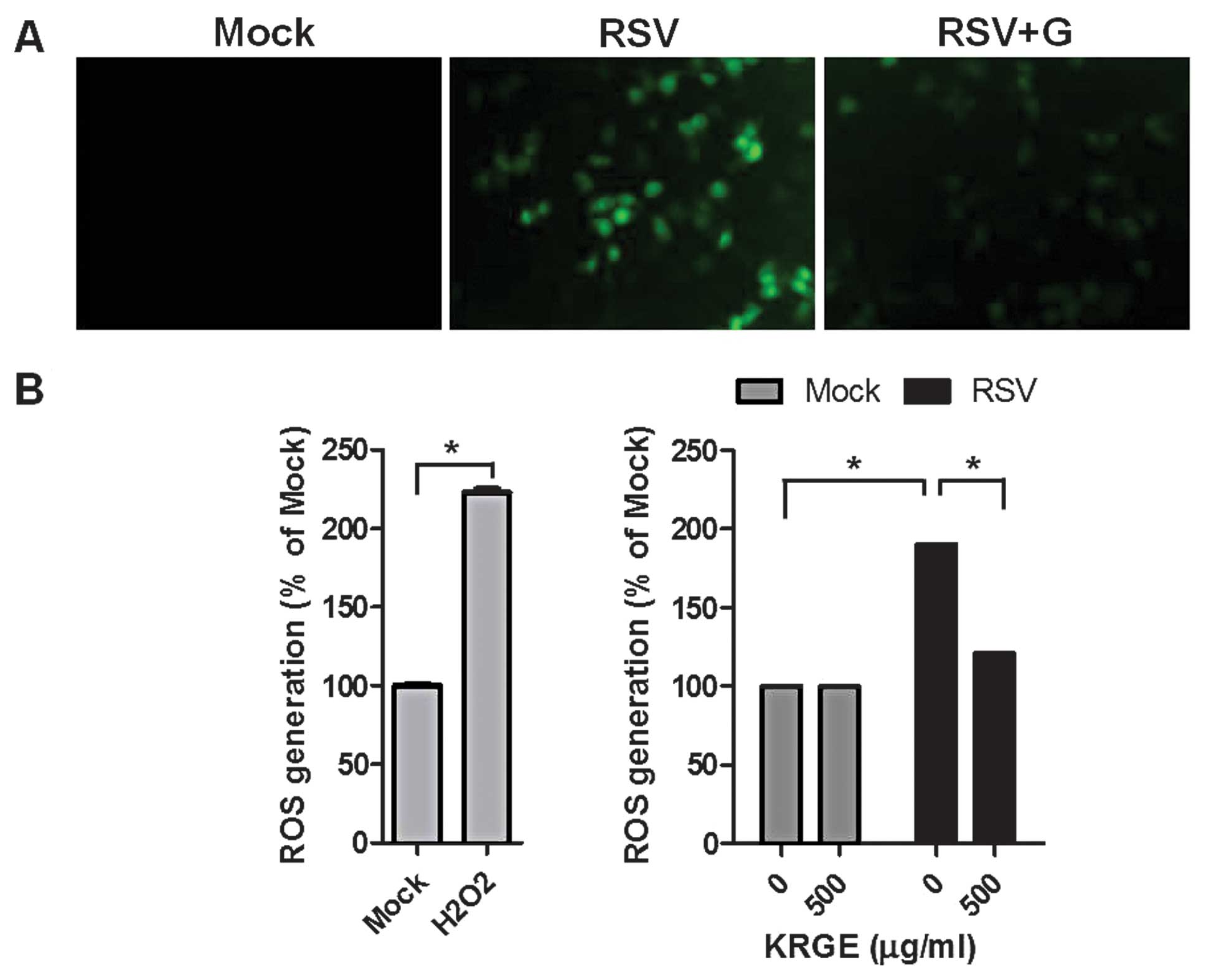
Influence of KRGE on ROS formation in
human epithelial cells
Oxidative stress-mediated events are known to be
involved in virus infection mechanisms and correlated with the
release of inflammatory mediators from epithelial cells (25). Intracellular ROS levels in
RSV-infected A549 cells were determined using the ROS-sensitive
fluorescent probe H2DCFDA. KRGE treatment did not affect
ROS formation of the non-infected cells. The fluorescence intensity
of the dichlorofluorescein diacetate stain was significantly
enhanced in RSV-infected human epithelial cells. Of nore, KRGE
treatment significantly inhibited RSV-induced ROS formation as
indicated by lowering green fluorescence intensity (Fig. 5). These results suggested that
ginseng treatment inhibits RSV-induced ROS generation.
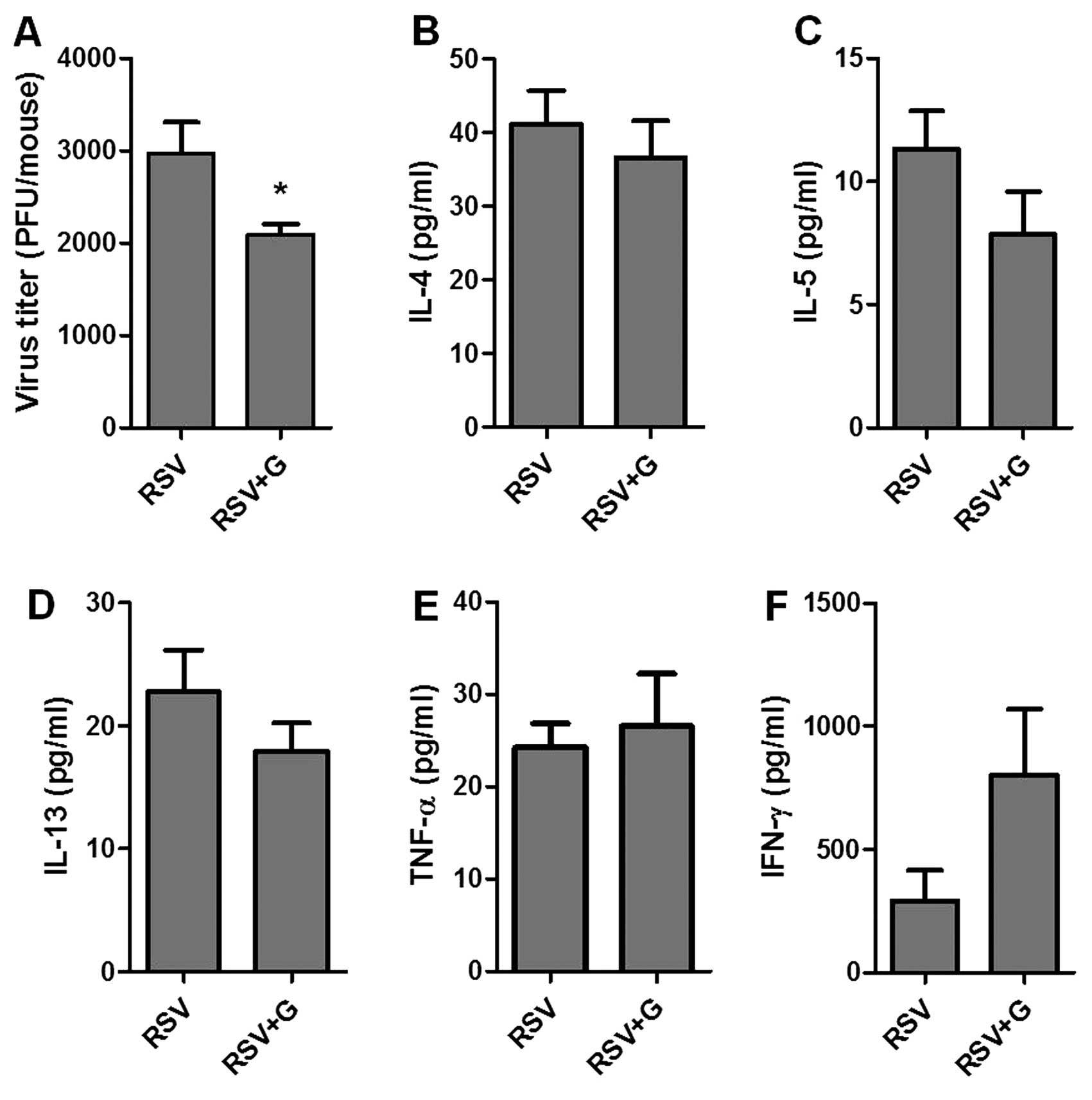
KRGE protects mice from RSV
infection
To better understand the potential role of KRGE in
conferring protection against RSV infection, we tested the in
vivo effects of KRGE on viral infection in a mouse model.
BALB/c mice were orally administered 25 mg/kg/day of drinking water
for 60 days. KRGE and mock-treated mice were infected by intranasal
inoculation of RSV (2×106 PFU/mouse). Lungs and BALF
were collected at day 5 post-infection for analysis of lung viral
titers and cytokine levels. The group of mice that were orally
treated with ginseng for 60 days showed a lower level of lung viral
titers compared to that in the RSV-infected naïve control (Fig. 6A). In addition, the levels of
pro-inflammatory cytokines IL-4, IL-5 and IL-13 were found to be
lower in the KRGE-treated group following RSV infection compared to
the untreated control, with no statistical significance being
observed (Fig. 6). Levels of
TNF-α in BALF were similar in the two groups. The levels of
antiviral cytokine IFN-γ showed a higher level in the
KRGE-administered mice compared to the infected naïve control mice
(Fig. 7), with no statistical
significance being observed.
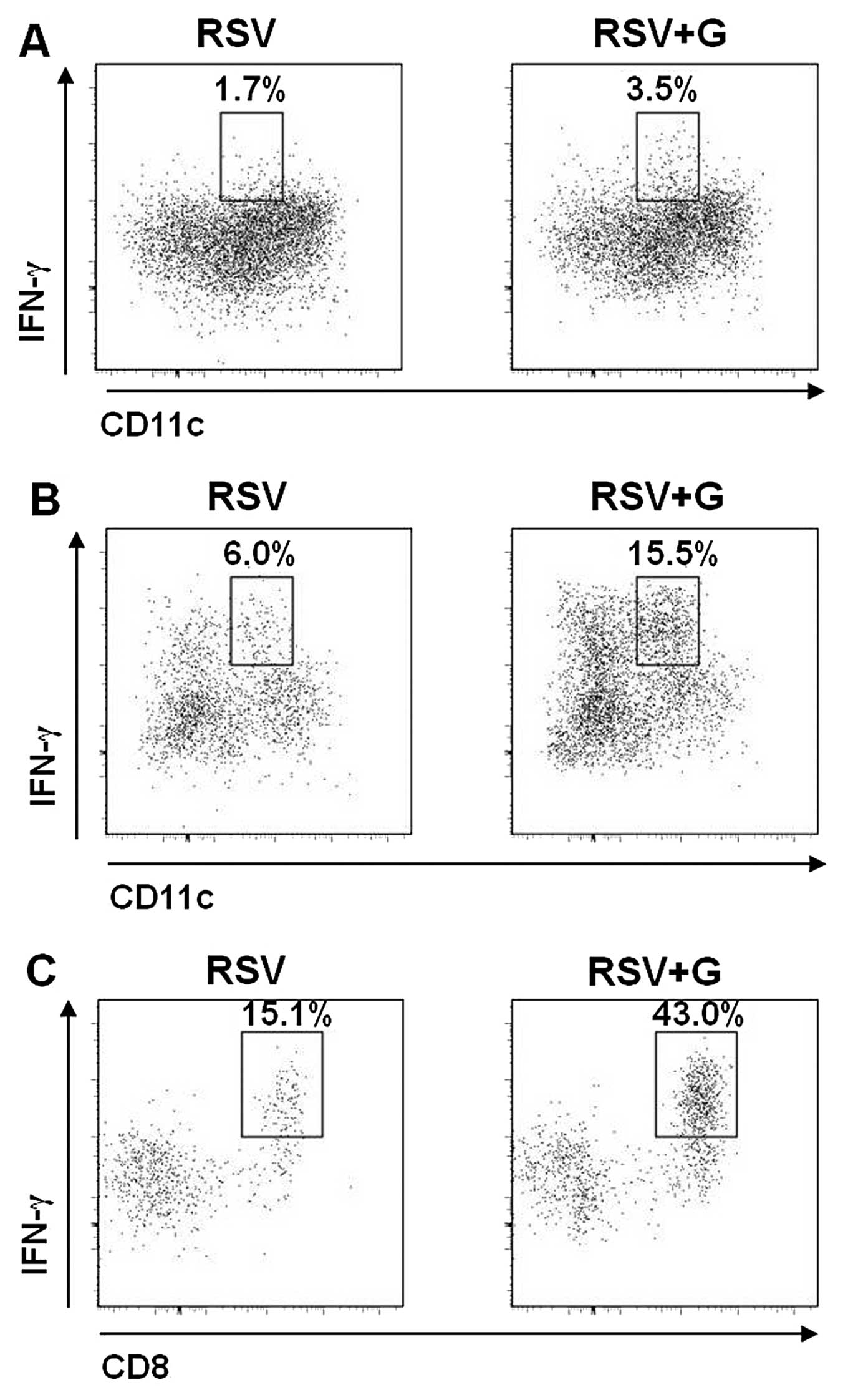
 | Figure 7In vivo effects of Korean red
ginseng extract (KRGE) on phenotypes of bronchoalveolar cells
producing interferon-γ (IFN-γ). KRGE was administered orally at a
dose of 25 mg/kg/day for 60 days and then mice were challenged with
respiratory syncytial virus (RSV) (2×106 PFU).
Bronchoalveolar lavage (BAL) was collected 5 days after RSV
challenge, and BAL cells were pooled (n=5 BALB/c mice/group), and
stained with CD3, CD4, CD8, CD11b, CD11c and IFN-γ antibodies, and
analyzed by flow cytometry. The cells were first gated by their
cell size and IFN-γ-producing (A) CD3+CD4+ T
cells, (B) CD11b+ cells and (C) CD11c+ cells
were observed. The numbers in dot plots indicate the cell
percentages of the double-positive population. Representative data
are shown of two independent sets of experiments (two sets of
experiments n=10). RSV, naïve mice challenged with RSV; RSV + G,
KRGE was administered orally at a dose of 25 mg/kg/day for 60 days
and then mice were challenged with RSV. |
KRGE increases antiviral IFN-γ-producing
cells in mice following RSV infection
To better understand the potential role of KRGE in
conferring protection against RSV infection, the levels of
IFN-γ-producing cells in BALF were examined by intracellular IFN-γ
cytokine staining at day 5 post infection with RSV. From the flow
cytometric analysis of pooled BAL cells of 5 mice in each group, we
observed that oral administration of KRGE to mice showed higher
levels of various phenotypic cells producing IFN-γ antiviral
cytokine following RSV infection (Figs. 7 and 8). A higher level of IFN-γ-producing
CD4+ T cells was detected in BALF from mice that were
orally treated with KRGE compared to that from the control group
without KRGE following RSV infection (11.7 vs. 17.2%) (Fig. 7A). Additionally, CD11b+
granulocytes producing IFN-γ were higher in the KRGE-treated mouse
group than those in the untreated mice with RSV infection (1.8 vs.
4.5%) (Fig. 7B). In particular,
CD11c+ dendritic cells producing IFN-γ were found to be
3-fold higher in BALF samples of KRGE-treated mice compared to that
from the control group following RSV infection (2.7 vs. 8.5%)
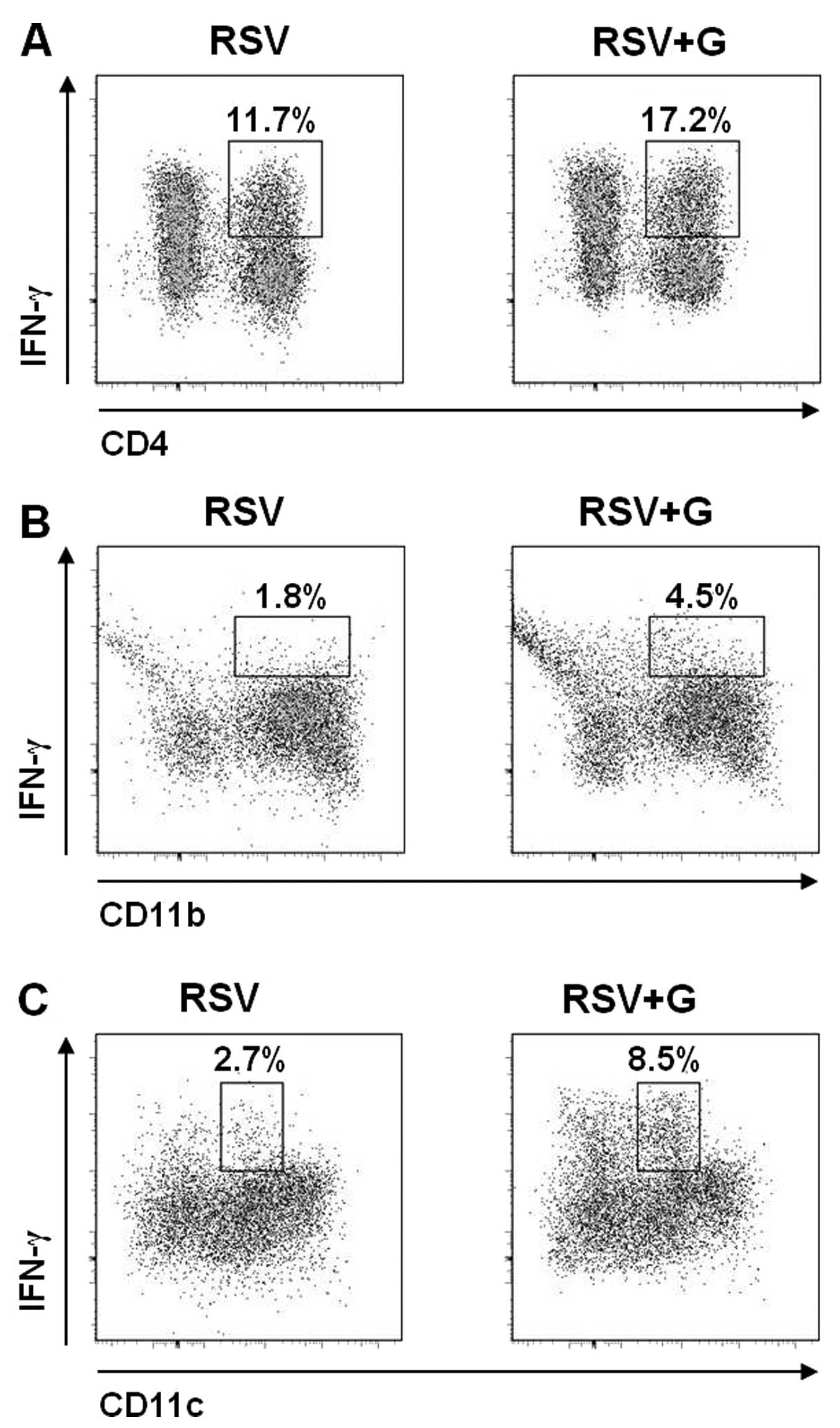
(Fig. 7C). Sub-populations
producing IFN-γ were further dissected with additional phenotypic
markers CD11b and CD8α (Fig. 8).
CD11c+CD11b+IFN-γ+ granulocytes
were 2-fold higher in the KRGE-treated group compared to the
non-treated controls, although these granulocytes are not a major
population producing IFN-γ (Fig.
8A). The majority of granulocytes expressing IFN-γ in the
KRGE-treated group were found to be
CD11b−CD11c+IFN-γ+ dendritic cells
(Fig. 8B), expressing
CD8α+ (Fig. 8C). The
lymphocytic marker CD8α+ expressing CD11c-positive
dendritic cells is known to contribute to inducing T-helper type 1
(Th1) immune responses (26).
Thus, KRGE treatment may stimulate innate and adaptive Th1-type
immune responses by stimulating
CD11b−CD8α+CD11c+ dendritic
cells.
Discussion
Ginseng has been known to have various
immunomodulatory functions. However, the potential anti-viral
activity, oxidative stress, inflammatory cytokine production, and
immunomodulatory effects of Panax ginseng on RSV infection
remain unknown. In the present study, we showed that KRGE inhibited
the replication of RSV, RSV-induced cell death, expression of
pro-inflammatory cytokines, and suppressed RSV-induced ROS
formation in human epithelial cells. Oral intake of ginseng
products is the most common means of consumption as a nutrient
supplement in healthy individuals. Therefore, we assessed the
potential antiviral effects and immunomodulatory functions of KRGE
following RSV viral infection in a mouse model. Oral administration
of KRGE to mice conferred moderate but significant resistance to
RSV infection. Therefore, the present study provides evidence that
KRGE has antiviral activity against RSV in vitro and in
vivo. Anti-RSV activity of KRGE has a high significance
considering the fact that RSV is the most frequent viral cause of
respiratory diseases, asthma, and chronic obstructive pulmonary
disease exacerbations early and later in life (27).
The results showed that RSV caused significant cell
death following RSV infection in a dose-dependent manner,
presumably due to RSV-induced oxidative damage to the cells
(3). Cellular oxidative damage
due to RSV infection was reported as a result of inducing an
imbalance between ROS production and antioxidant cell defenses
(3). Various cytokines and
chemokines are known to be expressed during RSV infection, playing
a central role in pathogenesis (28). These effects have been associated
with activation of the transcription factor nuclear factor-κB and
mitogen-activated protein kinase through oxidant-dependent
mechanisms (25). KRGE treatment
partially protected the cell death from RSV infection. The
underlying mechanisms by which KRGE treatment improves cell
survival during RSV infection remain to be defined. KRGE treatment
significantly inhibited the in vitro growth of RSV by
3–5-fold as well as the production of RSV-induced pro-inflammatory
cytokines and ROS formation. A possible mechanism is that the
KRGE-mediated inhibition of RSV replication in alveolar epithelial
cells may have resulted in a reduction of ROS and pro-inflammatory
cytokine production, which subsequently resulted in improving cell
survival. Alternatively, an anti-oxidant effect by KRGE may have
independently contributed to improving cell survival protection.
Thus, it is hypothesized that KRGE may exhibit antiviral activity
against RSV in various ways during RSV infection.
Findings of a previous study demonstrated that oral
administration of KRGE prior to infection significantly increased
survival rates of mice following infection with 2009 pandemic H1N1
virus (20). Results of the
present study have shown that oral administration of KRGE to mice
prior to infection resulted in lowering lung viral loads subsequent
to RSV infection. This reduction is highly significant as oral
intake of KRGE protects the local infection of RSV in lungs. The
anti-inflammatory effects of KRGE on inflammatory cytokines in BALF
samples were minimal, likely because RSV is not highly pathogenic
to mice although RSV replicates to a certain level (29).
The ginsenosides, the constituents of KRGE were
shown to reduce IL-4 production but increase IFN-γ production,
resulting in Th1-type immune responses in an ovalbumin-induced
murine model of asthma (30).
Dendritic cells are crucial in determining the fate of naïve T
cells, whether Th1 or Th2 cells (26). However, the effects of ginseng on
dendritic cells and resulting T-cell responses remain to be
determined. A significant finding in this study on RSV is that KRGE
oral treatment increased IFN-γ production in BALF following RSV
infection. A detailed analysis of BALF cells provided evidence that
KRGE may stimulate the production of IFN-γ-producing CD4 T
lymphocytes as well as granulocytes such as CD11b+ and
CD11c+ cells. Particularly, the increase in
CD11b−/lowCD11c+CD8α+ cell
populations was prominent as a result of KRGE treatment, which may
contribute to the production of IFN-γ and the increased induction
of CD4 T cells producing IFN-γ. The cellular phenotypes of IFN-γ
production and RSV pathogenesis as a result of ginseng treatment
may be different from those by RSV-infected epithelial cells that
were associated with severe pneumonia due to RSV infection
(31). Taken together, this study
suggests a mechanism that ginseng may have antiviral activity
against respiratory virus by modulating host cellular phenotypes
producing cytokines.
In conclusion, the modulation of oxidative stress is
a potential novel pharmacologic approach that may be used to
ameliorate RSV-induced acute lung inflammation. Ginseng inhibited
RSV-induced cellular oxidative damage and blocked the induction of
RSV-induced pro-inflammatory gene expression in the human alveolar
epithelial cell line. Furthermore, ginseng suppressed lung viral
titer and contributed to protective immunity by enhancing IFN-γ
production following RSV viral infection in an experimental RSV
infection murine model. Based on the results, although the exact
underlying anti-virus mechanism of ginseng remains to be
determined, consumption of ginseng in healthy individuals would
have beneficial effects in the prevention of unexpected RSV
infections and/or reduction of the severity of RSV disease.
Acknowledgements
This study was supported in part by NIH/NIAID grants
AI105170 (S.M.K.) and AI093772 (S.M.K.), and the Korean Ginseng
Corp. (S.M.K). The findings and conclusions in this study are those
of the authors and do not necessarily represent the views of the
funding agents.
References
|
1
|
Murphy BR, Prince GA, Collins PL, Van Wyke
Coelingh K, Olmsted RA, et al: Current approaches to the
development of vaccines effective against parainfluenza and
respiratory syncytial viruses. Virus Res. 11:1–15. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kim HW, Canchola JG, Brandt CD, Pyles G,
Chanock RM, et al: Respiratory syncytial virus disease in infants
despite prior administration of antigenic inactivated vaccine. Am J
Epidemiol. 89:422–434. 1969.PubMed/NCBI
|
|
3
|
Collins PL and Graham BS: Viral and host
factors in human respiratory syncytial virus pathogenesis. J Virol.
82:2040–2055. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chin J, Magoffin RL, Shearer LA, Schieble
JH and Lennette EH: Field evaluation of a respiratory syncytial
virus vaccine and a trivalent parainfluenza virus vaccine in a
pediatric population. Am J Epidemiol. 89:449–463. 1969.PubMed/NCBI
|
|
5
|
Fulginiti VA, Eller JJ, Sieber OF, Joyner
JW, Minamitani M and Meiklejohn G: Respiratory virus immunization.
I. A field trial of two inactivated respiratory virus vaccines; an
aqueous trivalent parainfluenza virus vaccine and an
alum-precipitated respiratory syncytial virus vaccine. Am J
Epidemiol. 89:435–448. 1969.
|
|
6
|
Garofalo RP, Kolli D and Casola A:
Respiratory syncytial virus infection: mechanisms of redox control
and novel therapeutic opportunities. Antioxid Redox Signal.
18:186–217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Becker S, Soukup J and Yankaskas JR:
Respiratory syncytial virus infection of human primary nasal and
bronchial epithelial cell cultures and bronchoalveolar macrophages.
Am J Respir Cell Mol Biol. 6:369–374. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hong CE and Lyu SY: Anti-inflammatory and
anti-oxidative effects of Korean red ginseng extract in human
keratinocytes. Immune Netw. 11:42–49. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Culley FJ, Pennycook AM, Tregoning JS,
Hussell T and Openshaw PJ: Differential chemokine expression
following respiratory virus infection reflects Th1- or Th2-biased
immunopathology. J Virol. 80:4521–4527. 2006. View Article : Google Scholar
|
|
10
|
Jiang Z, Kunimoto M and Patel JA:
Autocrine regulation and experimental modulation of interleukin-6
expression by human pulmonary epithelial cells infected with
respiratory syncytial virus. J Virol. 72:2496–2499. 1998.PubMed/NCBI
|
|
11
|
Hammad H and Lambrecht BN: Dendritic cells
and epithelial cells: linking innate and adaptive immunity in
asthma. Nat Rev Immunol. 8:193–204. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kwok HH, Ng WY, Yang MS, Mak NK, Wong RN
and Yue PY: The ginsenoside protopanaxatriol protects endothelial
cells from hydrogen peroxide-induced cell injury and cell death by
modulating intracellular redox status. Free Radic Biol Med.
48:437–445. 2010. View Article : Google Scholar
|
|
13
|
Chan LY, Kwok HH, Chan RW, Peiris MJ, Mak
NK, et al: Dual functions of ginsenosides in protecting human
endothelial cells against influenza H9N2-induced inflammation and
apoptosis. J Ethnopharmacol. 137:1542–1546. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Moore ML, Chi MH, Luongo C, Lukacs NW,
Polosukhin VV, et al: A chimeric A2 strain of respiratory syncytial
virus (RSV) with the fusion protein of RSV strain line 19 exhibits
enhanced viral load, mucus, and airway dysfunction. J Virol.
83:4185–4194. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Quan FS, Kim Y, Lee S, Yi H, Kang SM, et
al: Viruslike particle vaccine induces protection against
respiratory syncytial virus infection in mice. J Infect Dis.
204:987–995. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Panuska JR, Merolla R, Rebert NA, Hoffmann
SP, Tsivitse P, et al: Respiratory syncytial virus induces
interleukin-10 by human alveolar macrophages. Suppression of early
cytokine production and implications for incomplete immunity. J
Clin Invest. 96:2445–2453. 1995. View Article : Google Scholar
|
|
17
|
Arnold R, König B, Galatti H, Werchau H
and König W: Cytokine (IL-8, IL-6, TNF-alpha) and soluble TNF
receptor-I release from human peripheral blood mononuclear cells
after respiratory syncytial virus infection. Immunology.
85:364–372. 1995.
|
|
18
|
Zhang Y, Luxon BA, Casola A, Garofalo RP,
Jamaluddin M and Brasier AR: Expression of respiratory syncytial
virus-induced chemokine gene networks in lower airway epithelial
cells revealed by cDNA microarrays. J Virol. 75:9044–9058. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lee DC and Lau AS: Effects of Panax
ginseng on tumor necrosis factor-α-mediated inflammation: a
mini-review. Molecules. 16:2802–2816. 2011.
|
|
20
|
Legg JP, Hussain IR, Warner JA, Johnston
SL and Warner JO: Type 1 and type 2 cytokine imbalance in acute
respiratory syncytial virus bronchiolitis. Am J Respir Crit Care
Med. 168:633–639. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Matsuse H, Hirose H, Tsuchida T, Fukahori
S, Fukushima C, et al: Effects of respiratory syncytial virus
infection on dendritic cells and cysteinyl leukotrienes in lung
tissues of a murine model of asthma. Allergol Int. 56:165–169.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Song JM, Hossain J, Yoo DG, Lipatov AS,
Davis CT, et al: Protective immunity against H5N1 influenza virus
by a single dose vaccination with virus-like particles. Virology.
405:165–175. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Song JM, Van Rooijen N, Bozja J, Compans
RW and Kang SM: Vaccination inducing broad and improved cross
protection against multiple subtypes of influenza A virus. Proc
Natl Acad Sci USA. 108:757–761. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Attele AS, Wu JA and Yuan CS: Ginseng
pharmacology: multiple constituents and multiple actions. Biochem
Pharmacol. 58:1685–1693. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jie YH, Cammisuli S and Baggiolini M:
Immunomodulatory effects of Panax Ginseng C.A. Meyer in the
mouse. Agents Actions. 15:386–391. 1984.
|
|
26
|
Pulendran B: Modulating vaccine responses
with dendritic cells and Toll-like receptors. Immunol Rev.
199:227–250. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Panuska JR, Midulla F, Cirino NM, Villani
A, Gilbert IA, et al: Virus-induced alterations in macrophage
production of tumor necrosis factor and prostaglandin E2. Am J
Physiol. 259:L396–L402. 1990.PubMed/NCBI
|
|
28
|
Zeng R, Li C, Li N, Wei L and Cui Y: The
role of cytokines and chemokines in severe respiratory syncytial
virus infection and subsequent asthma. Cytokine. 53:1–7. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cormier SA, You D and Honnegowda S: The
use of a neonatal mouse model to study respiratory syncytial virus
infections. Expert Rev Anti Infect Ther. 8:1371–1380. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mata M, Morcillo E, Gimeno C and Cortijo
J: N-acetyl-L-cysteine (NAC) inhibit mucin synthesis and
pro-inflammatory mediators in alveolar type II epithelial cells
infected with influenza virus A and B and with respiratory
syncytial virus (RSV). Biochem Pharmacol. 82:548–555. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Watanabe W, Shimizu T, Sawamura R, Hino A,
Konno K, et al: Effects of tetrabromobisphenol A, a brominated
flame retardant, on the immune response to respiratory syncytial
virus infection in mice. Int Immunopharmacol. 10:393–397. 2010.
View Article : Google Scholar : PubMed/NCBI
|