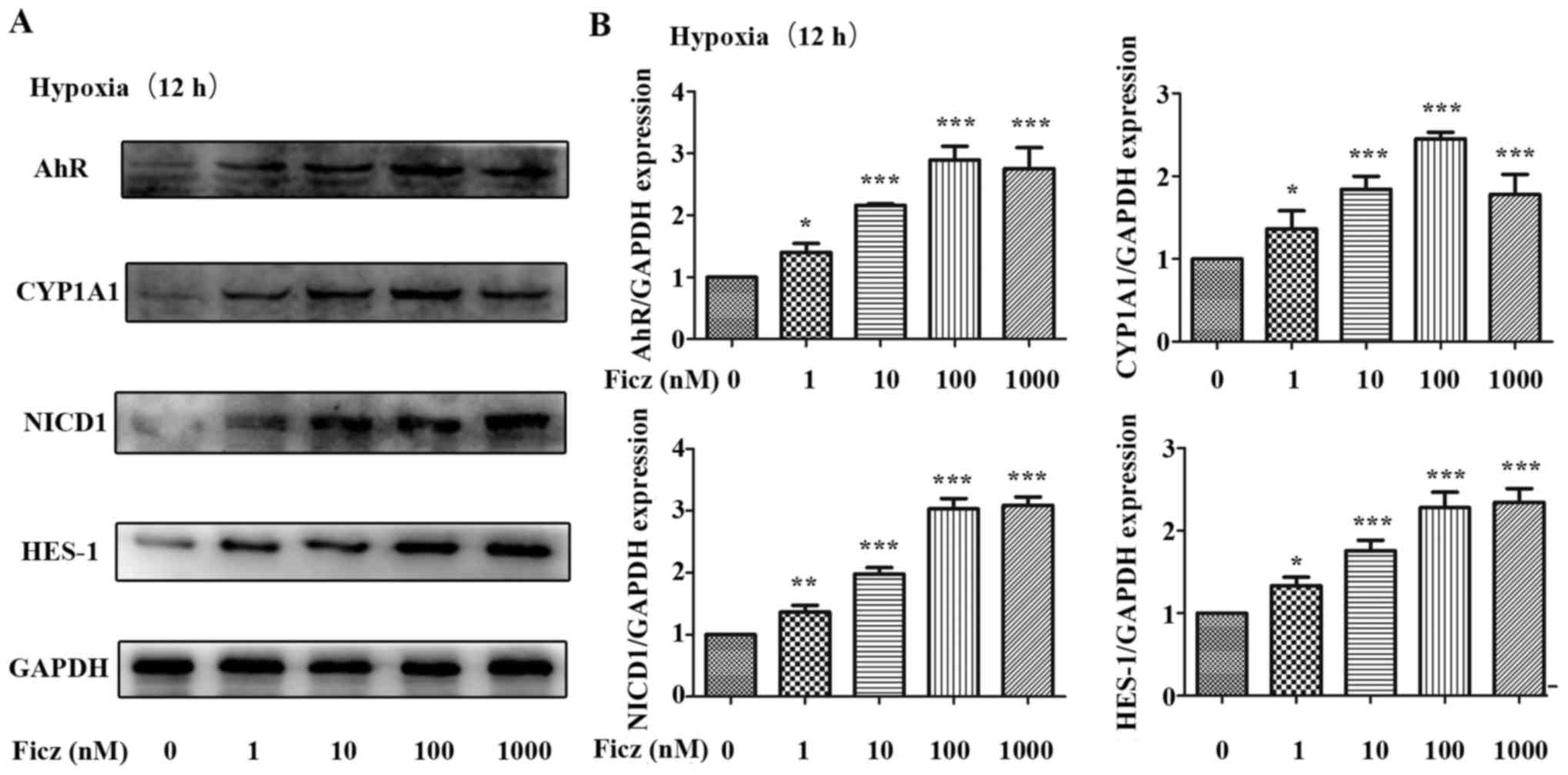
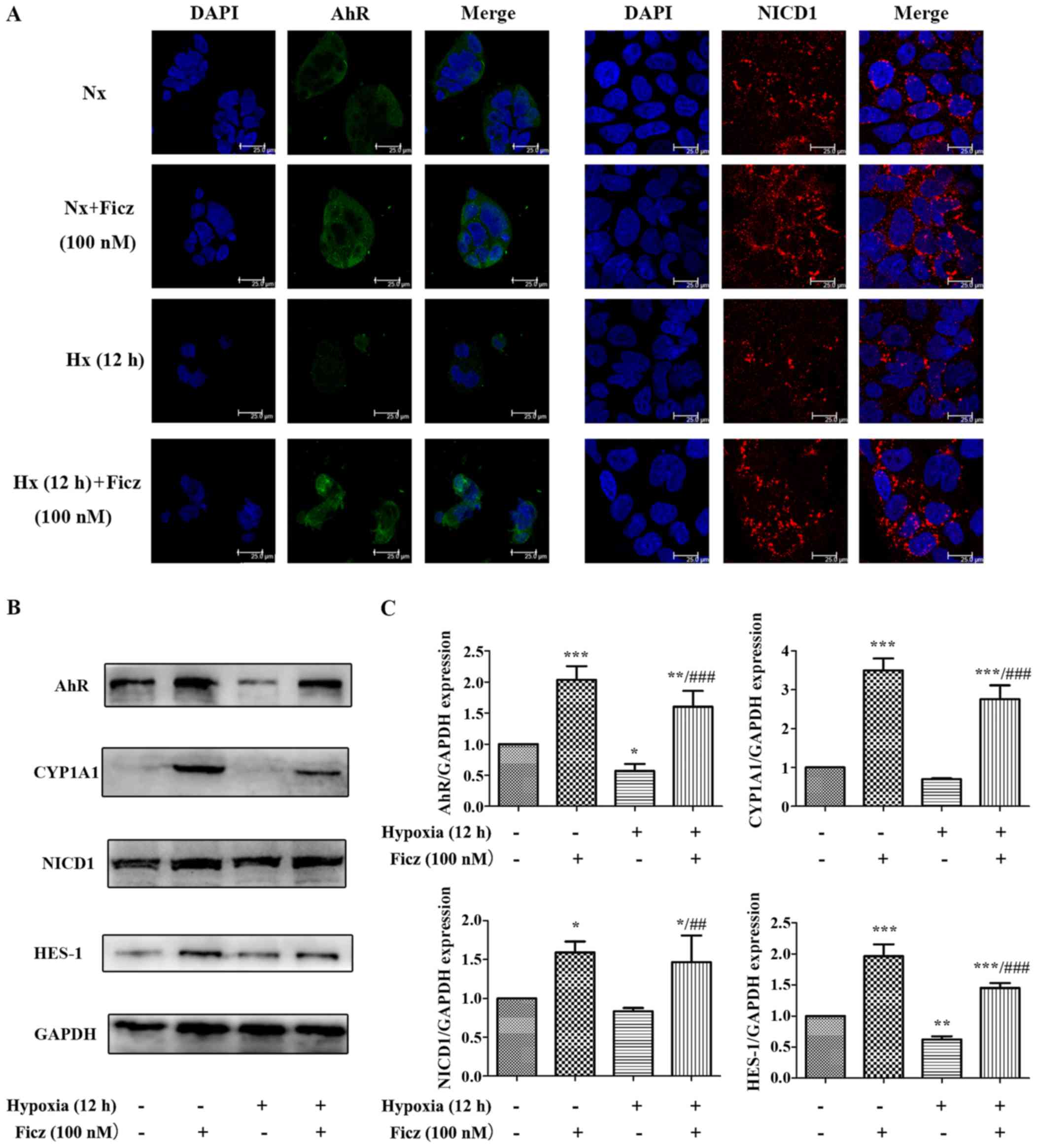
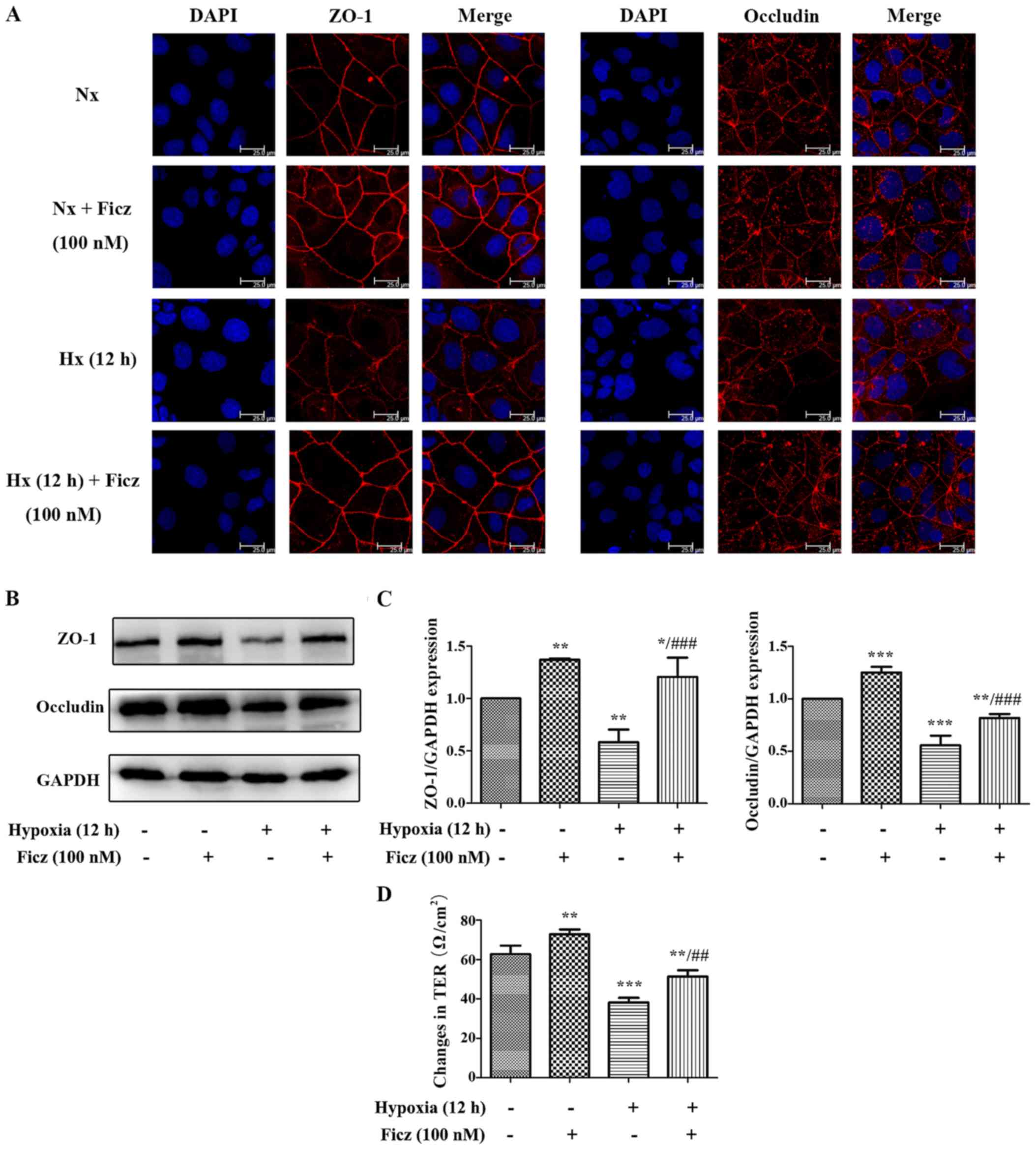
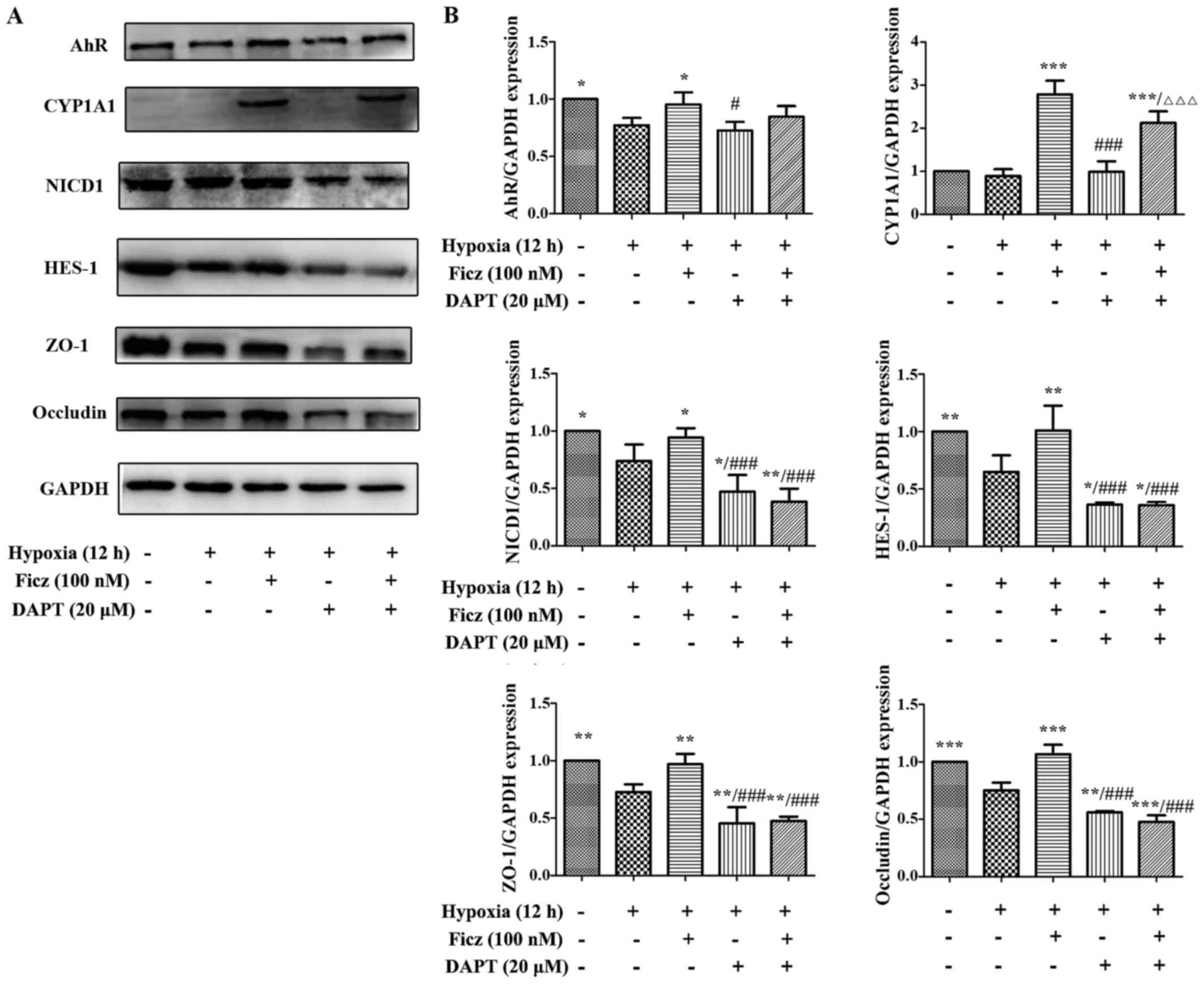
|
1
|
Arda-Pirincci P and Bolkent S: The role of
epidermal growth factor in prevention of oxidative injury and
apoptosis induced by intestinal ischemia/reperfusion in rats. Acta
Histochem. 116:167–175. 2014. View Article : Google Scholar
|
|
2
|
Granger DN and Korthuis RJ: Physiologic
mechanisms of post-ischemic tissue injury. Annu Rev Physiol.
57:311–332. 1995. View Article : Google Scholar
|
|
3
|
Matthijsen RA, Derikx JP, Kuipers D, van
Dam RM, Dejong CH and Buurman WA: Enterocyte shedding and
epithelial lining repair following ischemia of the human small
intestine attenuate inflammation. PLoS One. 4:e70452009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee SH: Intestinal permeability regulation
by tight junction: implication on inflammatory bowel diseases.
Intest Res. 13:11–18. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yang Y, Qiu Y, Wang W, Xiao W, Liang H,
Zhang C and Yang H, Teitelbaum DH, Sun LH and Yang H: Adenosine A2B
receptor modulates intestinal barrier function under hypoxic and
ischemia/reperfusion conditions. Int J Clin Exp Pathol.
7:2006–2018. 2014.PubMed/NCBI
|
|
6
|
Cai Y, Wang W, Liang H, Sun L, Teitelbaum
DH and Yang H: Keratinocyte growth factor improves epithelial
structure and function in a mouse model of intestinal
ischemia/reperfusion. PLoS One. 7:e447722012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xie G, Peng Z and Raufman JP: Src-mediated
aryl hydrocarbon and epidermal growth factor receptor cross talk
stimulates colon cancer cell proliferation. Am J Physiol
Gastrointest Liver Physiol. 302:G1006–G1015. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Marlowe JL, Fan Y, Chang X, Peng L,
Knudsen ES, Xia Y and Puga A: The aryl hydrocarbon receptor binds
to E2F1 and inhibits E2F1-induced apoptosis. Mol Biol Cell.
19:3263–3271. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mandavia C: TCDD-induced activation of
aryl hydrocarbon receptor regulates the skin stem cell population.
Med Hypotheses. 84:204–208. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Qiu J, Heller JJ, Guo X, Chen ZM, Fish K,
Fu YX and Zhou L: The aryl hydrocarbon receptor regulates gut
immunity through modulation of innate lymphoid cells. Immunity.
36:92–104. 2012. View Article : Google Scholar :
|
|
11
|
Monteleone I, Rizzo A, Sarra M, Sica G,
Sileri P, Biancone L, MacDonald TT, Pallone F and Monteleone G:
Aryl hydrocarbon receptor-induced signals up-regulate IL-22
production and inhibit inflammation in the gastrointestinal tract.
Gastroenterology. 141:237–248. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ji T, Xu C, Sun L, Yu M, Peng K, Qiu Y,
Xiao W and Yang H: Aryl hydrocarbon receptor activation
down-regulates IL-7 and reduces inflammation in a mouse model of
DSS-induced colitis. Dig Dis Sci. 60:1958–1966. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Qiu J, Guo X, Chen ZM, He L, Sonnenberg
GF, Artis D, Fu YX and Zhou L: Group 3 innate lymphoid cells
inhibit T-cell-mediated intestinal inflammation through aryl
hydrocarbon receptor signaling and regulation of microflora.
Immunity. 39:386–399. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Arsenescu R, Arsenescu V, Zhong J, Nasser
M, Melinte R, Dingle RW, Swanson H and de Villiers WJ: Role of the
xenobiotic receptor in inflammatory bowel disease. Inflamm Bowel
Dis. 17:1149–1162. 2011. View Article : Google Scholar :
|
|
15
|
Li Y, Innocentin S, Withers DR, Roberts
NA, Gallagher AR, Grigorieva EF, Wilhelm C and Veldhoen M:
Exogenous stimuli maintain intraepithelial lymphocytes via aryl
hydrocarbon receptor activation. Cell. 147:629–640. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jing H, Shen G, Wang G, Zhang F, Li Y, Luo
F, Yao J and Tian XF: MG132 alleviates liver injury induced by
intestinal ischemia/reperfusion in rats: involvement of the AhR and
NFκB pathways. J Surg Res. 176:63–73. 2012. View Article : Google Scholar
|
|
17
|
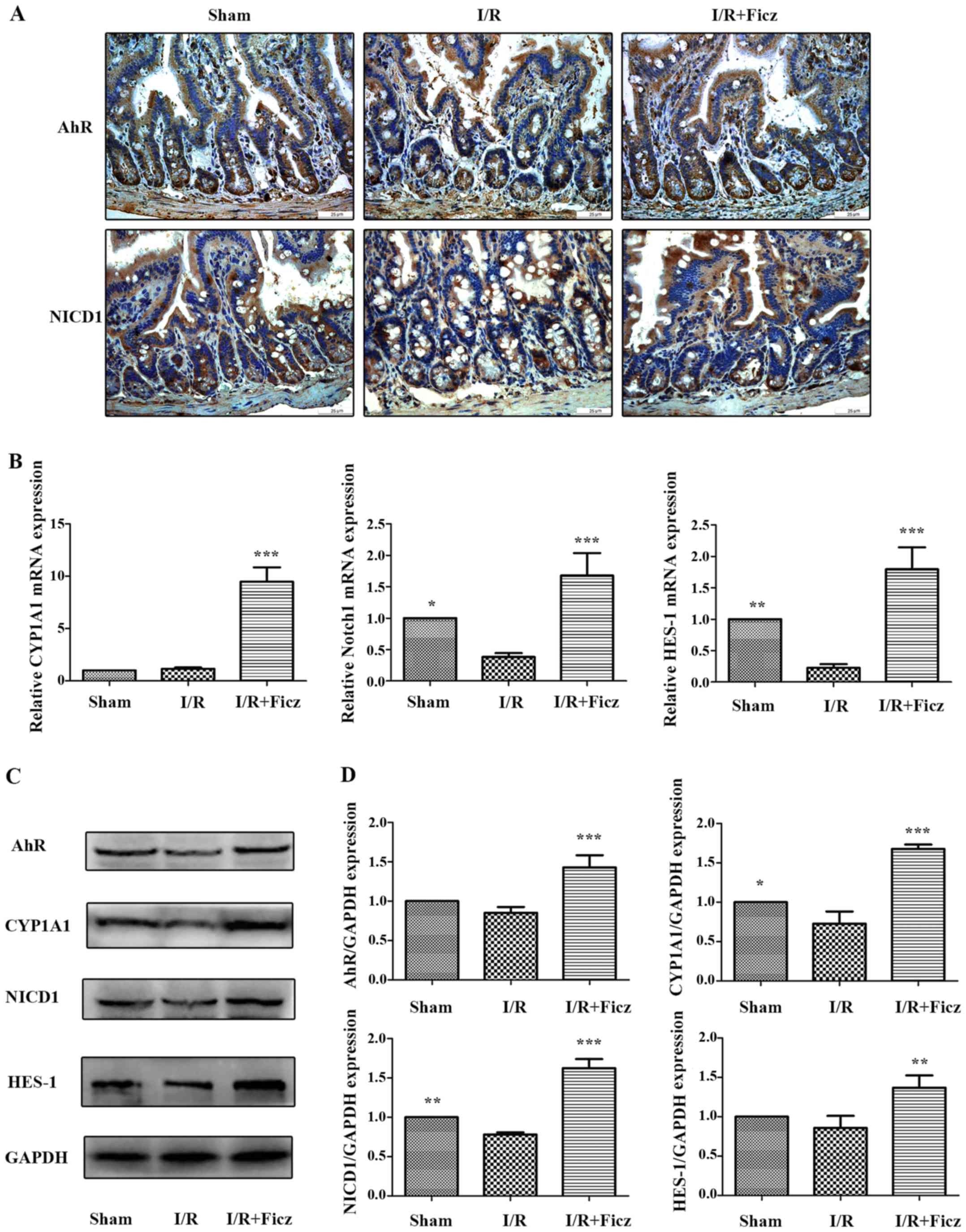
Dahan S, Rabinowitz KM, Martin AP, Berin
MC, Unkeless JC and Mayer L: Notch-1 signaling regulates intestinal
epithelial barrier function, through interaction with
CD4+ T cells, in mice and humans. Gastroenterology.
140:550–559. 2011. View Article : Google Scholar
|
|
18
|
Mathern DR, Laitman LE, Hovhannisyan Z,
Dunkin D, Farsio S, Malik TJ, Roda G, Chitre A, Iuga AC, Yeretssian
G, et al: Mouse and human Notch-1 regulate mucosal immune
responses. Mucosal Immunol. 7:995–1005. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen G, Sun L, Yu M, Meng D, Wang W, Yang
Y and Yang H: The Jagged-1/Notch-1/Hes-1 pathway is involved in
intestinal adaptation in a massive small bowel resection rat model.
Dig Dis Sci. 58:2478–2486. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen G, Qiu Y, Sun L, Yu M, Wang W, Xiao
W, Yang Y, Liu Y, Yang S, Teitelbaum DH, et al: The
jagged-2/notch-1/hes-1 pathway is involved in intestinal epithelium
regeneration after intestinal ischemia-reperfusion injury. PLoS
One. 8:e762742013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lee JS, Cella M, McDonald KG, Garlanda C,
Kennedy GD, Nukaya M, Mantovani A, Kopan R, Bradfield CA, Newberry
RD, et al: AHR drives the development of gut ILC22 cells and
postnatal lymphoid tissues via pathways dependent on and
independent of Notch. Nat Immunol. 13:144–151. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Stevens EA, Mezrich JD and Bradfield CA:
The aryl hydrocarbon receptor: a perspective on potential roles in
the immune system. Immunology. 127:299–311. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xia M, Viera-Hutchins L, Garcia-Lloret M,
Noval Rivas M, Wise P, McGhee SA, Chatila ZK, Daher N, Sioutas C
and Chatila TA: Vehicular exhaust particles promote allergic airway
inflammation through an aryl hydrocarbon receptor-Notch signaling
cascade. J Allergy Clin Immunol. 136:441–453. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Alam MS, Maekawa Y, Kitamura A, Tanigaki
K, Yoshimoto T, Kishihara K and Yasutomo K: Notch signaling drives
IL-22 secretion in CD4+ T cells by stimulating the aryl
hydrocarbon receptor. Proc Natl Acad Sci USA. 107:5943–5948. 2010.
View Article : Google Scholar
|
|
25
|
Qiu Y, Yu M, Yang Y, Sheng H, Wang W, Sun
L, Chen G, Liu Y, Xiao W and Yang H: Disturbance of intraepithelial
lymphocytes in a murine model of acute intestinal
ischemia/reperfusion. J Mol Histol. 45:217–227. 2014. View Article : Google Scholar
|
|
26
|
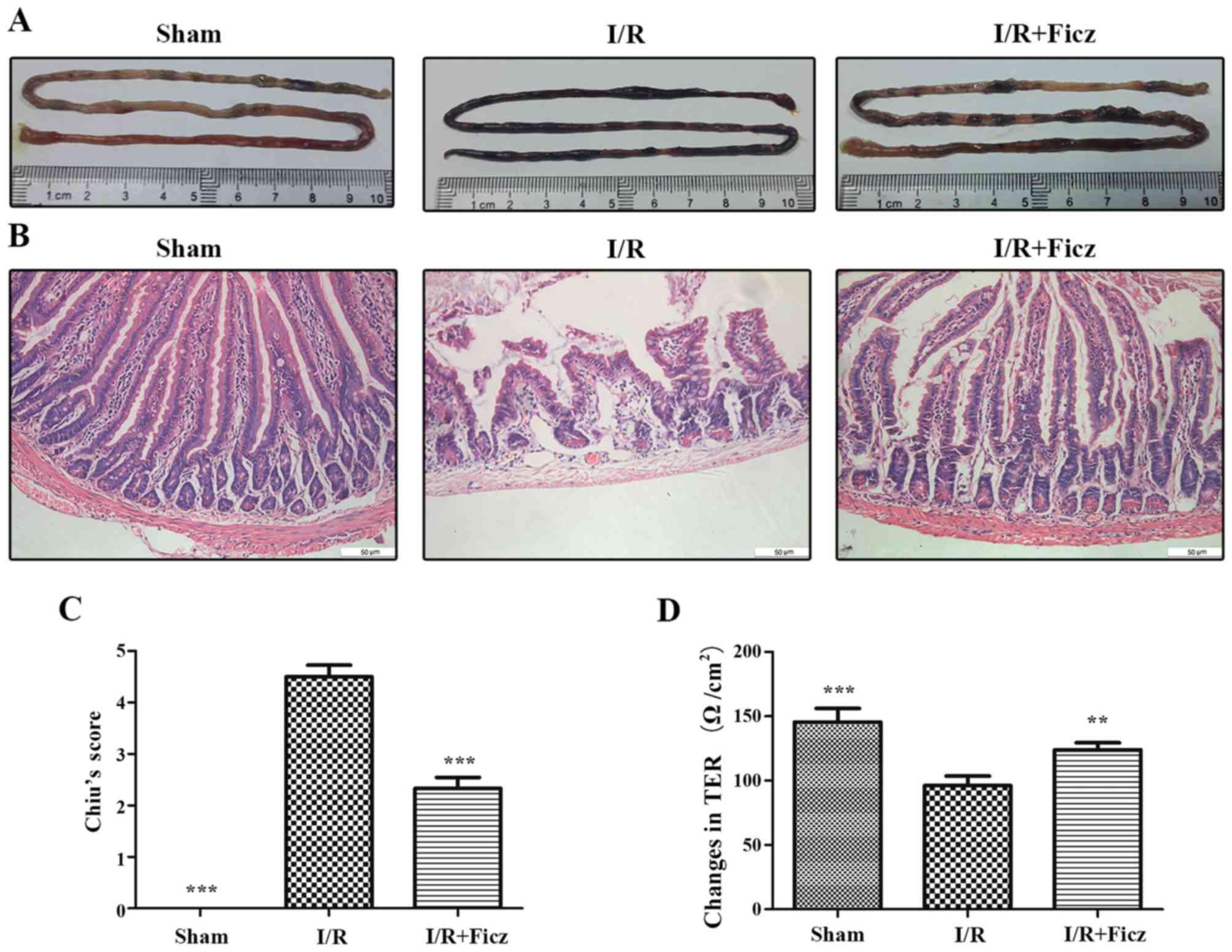
Chiu CJ, McArdle AH, Brown R, Scott HJ and
Gurd FN: Intestinal mucosal lesion in low-flow states. I. A
morphological, hemodynamic, and metabolic reappraisal. Arch Surg.
101:478–483. 1970. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Polentarutti BI, Peterson AL, Sjöberg AK,
Anderberg EK, Utter LM and Ungell AL: Evaluation of viability of
excised rat intestinal segments in the Ussing chamber:
investigation of morphology, electrical parameters, and
permeability characteristics. Pharm Res. 16:446–454. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
29
|
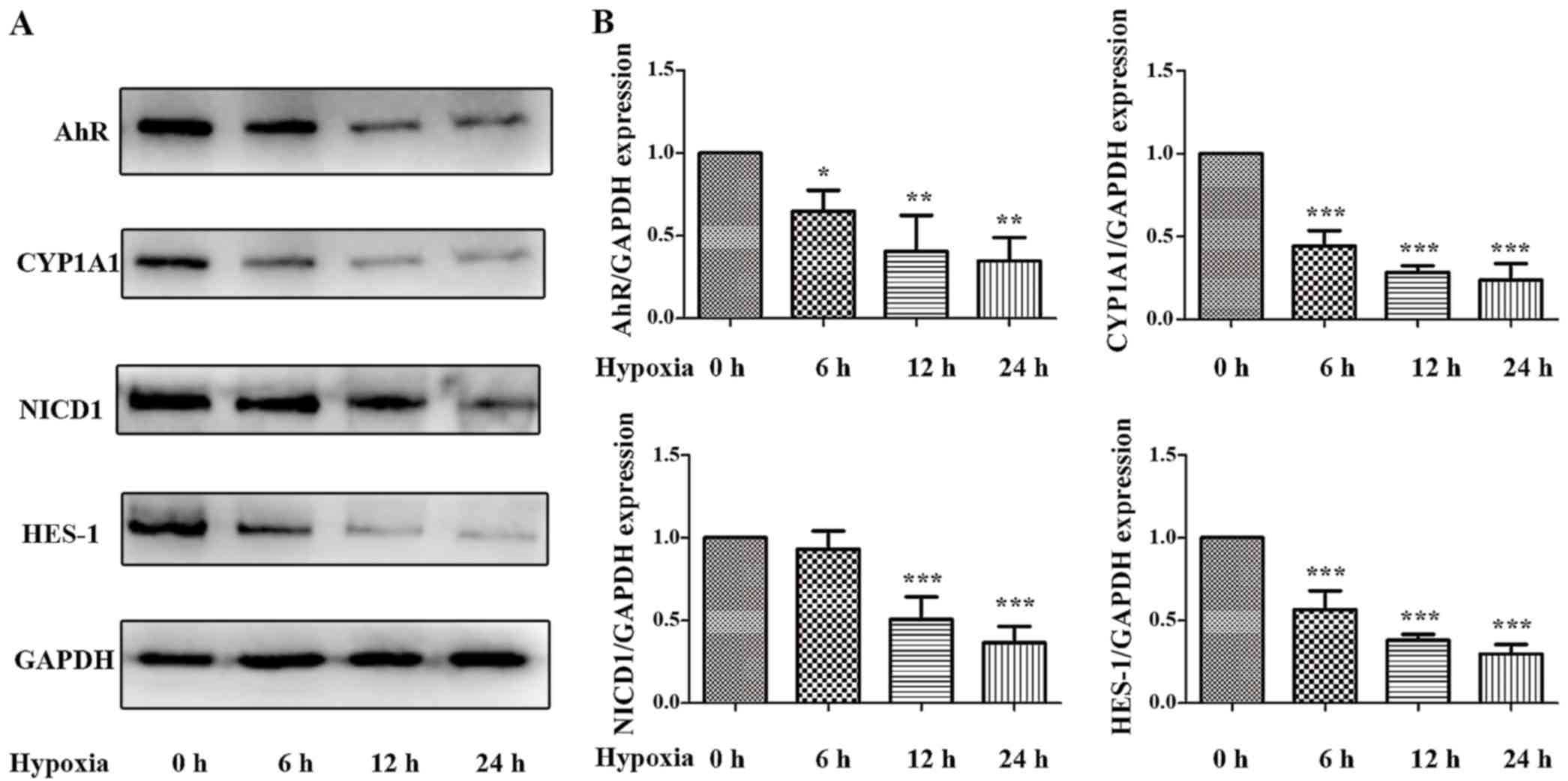
Nebert DW and Roe AL: Role of the aromatic
hydrocarbon receptor and [Ah] gene battery in the oxidative stress
response, cell cycle control, and apoptosis. Biochem Pharmacol.
59:65–85. 2000. View Article : Google Scholar
|
|
30
|
Delescluse C, Lemaire G, de Sousa G and
Rahmani R: Is CYP1A1 induction always related to AHR signaling
pathway. Toxicology. 153:73–82. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen G, Zhang Z, Cheng Y, Xiao W, Qiu Y,
Yu M, Sun L, Wang W, Du G, Gu Y, et al: The canonical Notch
signaling was involved in the regulation of intestinal epithelial
cells apoptosis after intestinal ischemia/reperfusion injury. Int J
Mol Sci. 15:7883–7896. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Huang CY, Hsiao JK, Lu YZ, Lee TC and Yu
LC: Anti-apoptotic PI3K/Akt signaling by sodium/glucose transporter
1 reduces epithelial barrier damage and bacterial translocation in
intestinal ischemia. Lab Invest. 91:294–309. 2011. View Article : Google Scholar
|
|
33
|
Jönsson ME, Franks DG, Woodin BR, Jenny
MJ, Garrick RA, Behrendt L, Hahn ME and Stegeman JJ: The tryptophan
photo-product 6-formylindolo[3,2-b]carbazole (FICZ) binds multiple
AHRs and induces multiple CYP1 genes via AHR2 in zebrafish. Chem
Biol Interact. 181:447–454. 2009. View Article : Google Scholar
|
|
34
|
Daniel RA, Cardoso VK, Góis E Jr, Parra
RS, Garcia SB, Rocha JJ and Féres O: Effect of hyperbaric oxygen
therapy on the intestinal ischemia reperfusion injury. Acta Cir
Bras. 26:463–469. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ogando J, Tardáguila M, Díaz-Alderete A,
Usategui A, Miranda-Ramos V, Martínez-Herrera DJ, de la Fuente L,
García-León MJ, Moreno MC, Escudero S, et al: Notch-regulated
miR-223 targets the aryl hydrocarbon receptor pathway and increases
cytokine production in macrophages from rheumatoid arthritis
patients. Sci Rep. 6:202232016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
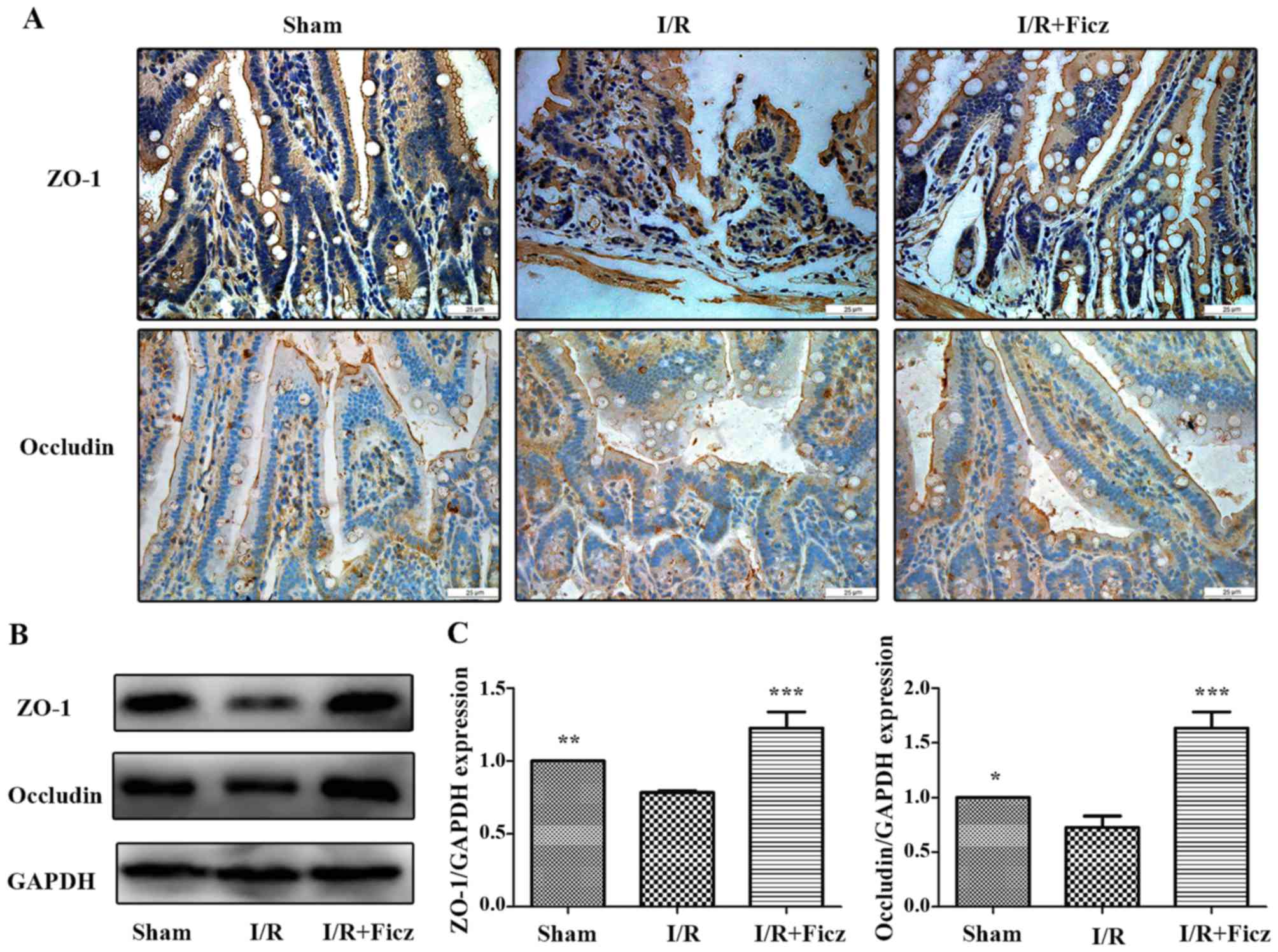
González-Mariscal L, Tapia R and Chamorro
D: Crosstalk of tight junction components with signaling pathways.
Biochim Biophys Acta. 1778:729–756. 2008. View Article : Google Scholar
|
|
37
|
Pope JL, Bhat AA, Sharma A, Ahmad R,
Krishnan M, Washington MK, Beauchamp RD, Singh AB and Dhawan P:
Claudin-1 regulates intestinal epithelial homeostasis through the
modulation of Notch-signalling. Gut. 63:622–634. 2014. View Article : Google Scholar
|
|
38
|
Movahedan A, Afsharkhamseh N, Sagha HM,
Shah JR, Milani BY, Milani FY, Logothetis HD, Chan CC and Djalilian
AR: Loss of Notch1 disrupts the barrier repair in the corneal
epithelium. PLoS One. 8:e691132013. View Article : Google Scholar : PubMed/NCBI
|