Introduction
Bladder cancer is one of the most common malignant
tumors of the urological system, and is also one of the main causes
of cancer-related mortality (1).
The prognosis of bladder cancer is dependent on its potential to
invade and the development of chemoresistance (2–4).
There are several risk factors for bladder cancer, including
smoking, environmental and occupational exposure and gender
(5). Although combined therapies,
including surgery and chemotherapy, have been developed over the
past decades, the prognosis for bladder cancer in the advanced
stage remains poor (6). The
identification of novel molecular targets and therapeutic
strategies are required in order to achieve long-term survival of
patients. Resistance to chemotherapy drives the search for
identification of more effective targets to overcome
chemoresistance.
MicroRNA (miRNA) are small non-coding RNA that
downregulate gene expression by binding to the 3′-untranslated
region (3′-UTR) of target mRNA (7,8).
miRNA have been reported to be dysregulated in various types of
human cancer, including lung, colorectal and bladder cancer, and
they serve important roles in carcinogenesis, invasion, metastasis
and development of chemoresistance (9–11).
miR-214 is regarded as a tumor suppressor that is
downregulated in various cancer types, including breast, lung and
colorectal carcinoma (12–14).
The expression status of miR-214 was also reported in bladder
cancer (15). miR-214 was
downregulated in bladder cancer tissues and significantly
associated with tumor stage, lymph node status and tumor grade
(15). miR-214 may serve as an
independent factor of recurrence-free survival and overall survival
for patients with muscle-invasive bladder cancer. Urinary levels of
cell-free miR-214 could be an independent prognostic parameter for
non-muscle-invasive bladder cancer recurrence (16). miR-214 exerts tumor-suppressive
effects in bladder cancer by downregulating oncogenic P53 and DNA
damage regulated 1 (PDRG1) expression (15). Currently, the role of miR-214 in
cancer cell apoptosis and drug resistance remains unclear. To
clarify this issue, the present study transfected miR-214 mimics
into human bladder cancer cell lines and examined its role in
cisplatin resistance. The underlying molecular mechanism was also
explored.
Materials and methods
Clinical specimens
Fresh bladder cancer samples and corresponding
normal adjacent tissues were obtained from 30 patients (22 males
and 8 females; age, 56.1±11.6 years) at The First Affiliated
Hospital of China Medical University (Shenyang, China) between
September 2013 and August 2015. The present study was conducted
with the approval of the Ethics Committee of China Medical
University. Written informed consent was obtained from all patients
and bladder cancer was confirmed by pathological diagnosis.
Cell culture and transfection
SV-HUC-1, RT-4, J82 and T24 cell lines were obtained
from the American Type Culture Collection (Manassas, VA, USA).
Cells were cultured in Dulbecco's modified Eagle's medium (DMEM;
Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
supplemented with 10% fetal bovine serum (FBS; Invitrogen; Thermo
Fisher Scientific, Inc.) at 37°C in 5% CO2. miR-214
mimic (5′-ACAGCAGGCACAGACAGGCAGU-3′) and corresponding control were
obtained from Guangzhou RiboBio Co., Ltd., (Guangzhou, China).
Cells were transfected with 50 nM concentration. Netrin-1 plasmid
(pCMV6-NTN1) was obtained from OriGene Technologies, Inc.
(Rockville, MD, USA). Plasmid was transfected at 1 μg/ml
concentration. miR-214 mimic was transfected using DharmaFECT 1
Reagent (GE Healthcare, Chicago, IL, USA). Lipofectamine 3000
reagent was used for plasmid transfection (Invitrogen; Thermo
Fisher Scientific, Inc.). Cells were harvested for subsequent
experiments 4 h after transfection. For cell viability and
apoptosis assays, cells were treated with cisplatin (10
μg/ml; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) for
24, 48 and 72 h at 37°C in 5% CO2.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
RNA was extracted from tissues and cells using
RNAiso plus (Takara Biotechnology Co., Ltd., Dalian, China). For
quantification of miR-214, primers (Guangzhou RiboBio Co., Ltd.)
for miR-214 (Bulge-LoopTM miRNA qRT-PCR Primer Set for hsa-mir-214)
and U6 (U6 small nuclear RNA qRT-PCR Primer) were used. Reverse
transcription (37°C for 15 min and 85°C for 5 sec) was performed
using a PrimeScript RT reagent kit from Takara Biotechnology Co.,
Ltd. qPCR was performed using ABI SYBR-Green Master Mix (Applied
Biosystems; Thermo Fisher Scientific, Inc.) on an Analytik Jena
QTOWER PCR system (Analytik Jena AG, Jena, Germany), according to
the manufacturer's protocol. The thermocyclying conditions were as
follows: 95°C for 30 sec, followed by 40 cycles of 95°C for 5 sec
and 60°C for 30 sec. The relative levels of gene expression were
represented as ΔCt=Ct miR-214-Ct U6. The change of gene expression
was calculated by the 2−ΔΔCq method (15).
For quantification of netrin-1 mRNA, total RNA was
reverse transcribed to cDNA using PrimeScript RT Master Mix (Takara
Biotechnology Co., Ltd.). qPCR was performed using an ABI SYBR
Green MasterMix on an Analytik Jena QTOWER PCR system, according to
the manufacturer's protocol. The thermocyclying conditions were as
follows: 95°C for 30 sec, followed by 40 cycles of 95°C for 5 sec
and 60°C for 30 sec. The relative levels of gene expression were
represented as ΔCt=Ct gene-Ct reference, and the fold change of
gene expression was calculated using the 2−ΔΔCq method
(15). The primer sequences used
were as follows: Netrin-1, forward 5′-GTCAATGCGGCCTTCGG-3′ and
reverse 5′-CTGCTCGTTCTGCTTGGTGAT-3′; and β-actin, forward
5′-ATAGCACAGCCTGGATAGCAACGTAC-3′ and reverse
5′-CACCTTCTACAATGAGCTGCGTGTG-3′.
Western blot analysis
Proteins were extracted using
radioimmunoprecipitation assay lysis buffer (Thermo Fisher
Scientific, Inc.) and quantified using the Bradford method. Protein
samples (~40 μg) were separated by 10% SDS-PAGE and then
transferred to a polyvinylidene difluoride membrane. Blocking was
performed using 5% bovine serum albumin (Beijing Solarbio Science
& Technology Co., Ltd., Beijing, China) at room temperature for
2 h. The membrane was incubated at 4°C overnight with primary
antibodies, including netrin-1 (1:500; ab126729; Abcam, Cambridge,
MA, USA), caspase-3 (9665), cleaved caspase-3 (9664), poly
(ADP-ribose) polymerase (PARP) (9532), cleaved PARP (5625),
phosphorylated (p)-AKT (4060), AKT (4691) B-cell lymphoma (Bcl)-2
(15071) (all 1:1,000; Cell Signaling Technology, Inc., Danvers, MA,
USA) and GAPDH (sc-32233) (1:2,000; Santa Cruz Biotechnology, Inc.,
Dallas, TX, USA). Following incubation with horseradish
peroxidase-conjugated secondary antibody (7071/7072; 1:2,000; Cell
Signaling Technology, Inc.) at 37°C for 2 h, visualization was
performed using an enhanced chemiluminescent kit (Pierce; Thermo
Fisher Scientific, Inc.) and a DNR Bio-Imaging System (DNR
Bio-Imaging Systems, Ltd., Neve Yamin, Israel).
Cell Counting Kit-8 (CCK-8) assay
CCK-8 assay was conducted to detect cell viability
using a CCK-8 kit from Dojindo Molecular Technologies, Inc.
(Rockville, MD, USA), according to the manufacturer's protocol.
Briefly, cultured cells (2,000 cells/well) were incubated with 10
μl/well CCK-8 solution for 4 h at 37°C in an incubator.
Subsequently, the optical density of each well was measured at a
wavelength of 490 nm.
Colony formation assay
Cells were seeded into three 6-cm cell culture
dishes with DMEM supplemented with 10% FBS (1,000 cells/dish) and
incubated at 37°C in 5% CO2 for at least 2 weeks.
Subsequently, the plates were washed with PBS and fixed using cold
100% methanol at 4°C for 10 min. Following this, the plates were
stained with Giemsa at room temperature for 30 min. Colony number
was counted manually using an Olympus CKX41 light microscope
(Olympus Corporation, Tokyo, Japan) with a magnification of
×100.
Matrigel invasion assay
A Matrigel invasion assay was conducted using a
24-well Transwell chamber from Costar (Corning Incorporated,
Corning, NY, USA) coated with 20 μl Matrigel (BD
Biosciences, Franklin Lakes, NJ, USA), with a dilution rate of 1:8.
A total of 100,000 cells were transferred into the upper Transwell
chamber in DMEM without serum and incubated for 18 h to allow the
cells to invade. Remaining cells were wiped out using a cotton tip
and invading cells were fixed with 4% paraformaldehyde at room
temperature for 10 min and stained with hematoxylin at room
temperature for 10 min. Colony number was counted using an Olympus
BX53 light microscope (Olympus Corporation) under a magnification
of ×200.
Flow cytometry for apoptosis
The apoptosis rate was detected using an Annexin
V/propidium iodide (PI) staining kit (BD Biosciences), according to
the manufacturer's protocol. The cells were analyzed using a
FACSCalibur flow cytometer (BD Biosciences) and data was analyzed
using FlowJo v. 10 software (FlowJo LLC, Ashland, OR, USA).
Validation of target gene and miRNA
interaction
A p-MIR-reporter vector (Thermo Fisher Scientific,
Inc.) was used for 3′-UTR luciferase reporter assays to determine
the interaction of miR-214 with netrin-1. The wild-type (WT)
miR-214 target site in netrin-1 3′-UTR was CCU GCU G. The mutant
(MUT) miR-214 target site was CCU UUU G. TargetScan (targetscan.org) was used to identify miR-214
targets.
Cells with 80% confluence were co-transfected with
the firefly luciferase reporter plasmid (0.1 μg) along with
the reference Renilla luciferase reporter plasmid (0.01
μg) using Lipofectamine 3000, according to the protocols
provided by the manufacturers. After 48 h of transfection, the
luciferase activity was measured using a Promega Dual-Luciferase
Reporter Assay System (Promega Corporation, Madison, WI, USA). The
relative activity of the reporter gene was calculated by dividing
the signals from firefly luciferase reporter by the signals
obtained from Renilla luciferase reporter.
Statistical analysis
Data were presented as the mean ± standard
deviation, and experiments were repeated in triplicates. SPSS v. 16
for Windows (SPSS, Inc., Chicago, IL, USA) was used for all
statistical analyses. A Student's t-test was used to compare data
between control and treatment groups. One-way analysis of variance
with Bonferroni's post hoc analysis was used to compare the means
of more than two groups. All P-values were based on a two-sided
statistical analysis. P<0.05 was considered to indicate a
statistically significant difference.
Results
miR-214 expression is downregulated in
bladder cancer
The expression of miR-214 was examined in 30 samples
of fresh bladder carcinoma tissues with corresponding normal
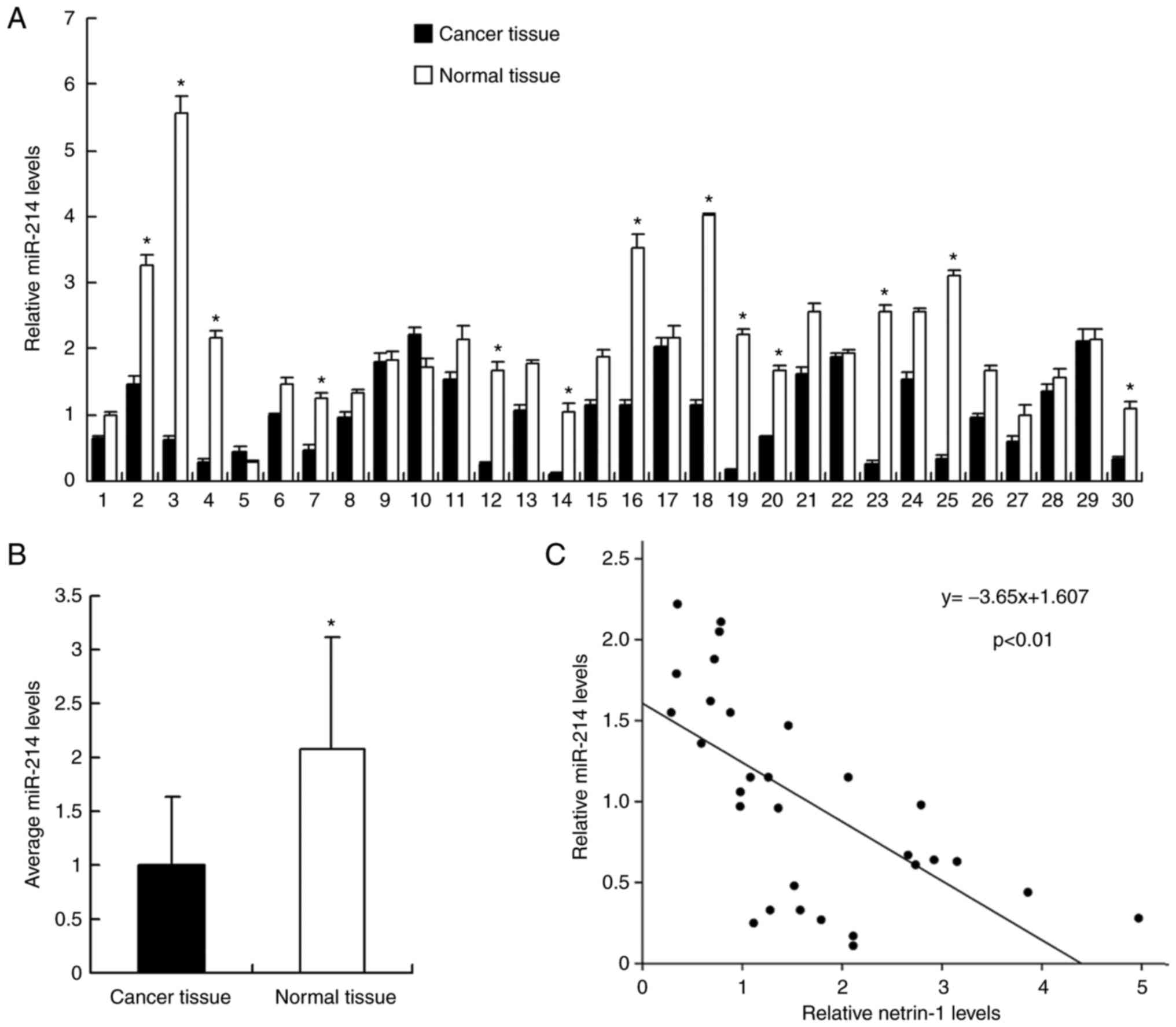
tissues by RT-qPCR. As demonstrated in Fig. 1, miR-214 expression levels were
significantly downregulated in cancer tissues compared with normal
tissues. miR-214 cancer/normal ratio <0.5 was regarded as
significant downregulation. It was revealed that miR-214 expression
was significantly downregulated compared with the level in normal
tissue in 13 out of 30 samples (significant was defined as ratio of
cancer tissue/normal tissue <1/2) (Fig. 1A). The mean value of miR-214
expression between cancer and normal tissues was examined and it
was demonstrated that the mean miR-214 expression level in normal
tissue was significantly higher than that in cancer tissue
(P<0.05; Fig. 1B). In
addition, the association between miR-214 expression and T stage
was evaluated. As demonstrated in Table I, the rate of miR-214
downregulation was 58.3% in T3 + T4 bladder cancer and 33.3% in T1
+ T2 cancer, suggesting that miR-214 may be downregulated in cancer
with a higher T stage. However, the difference did not reach a
statistical significance.
 | Table IDistribution of miR-214 expression in
bladder cancer according to T status. |
Table I
Distribution of miR-214 expression in
bladder cancer according to T status.
| Tumor local invasion
status | Number of
patients | miR-214 expression
level
| P-value |
|---|
| Low | High |
|---|
| T1 + T2 | 18 | 6 | 12 | 0.264 |
| T3 + T4 | 12 | 7 | 5 | |
miR-214 is downregulated in bladder
cancer cell lines and inhibits cell proliferation and invasion
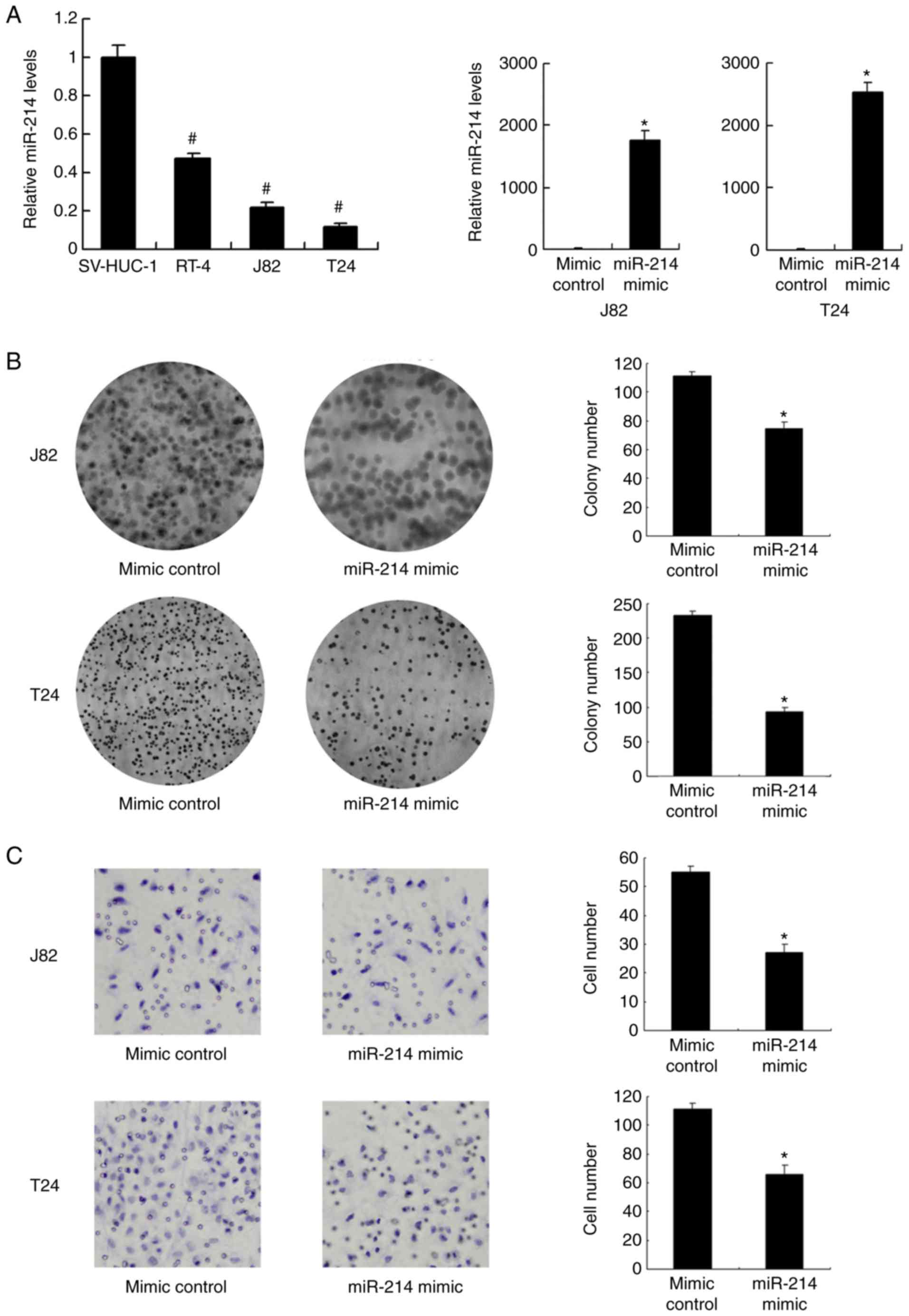
The expression level of miR-214 in normal epithelial
cell line SV-HUC-1 and three bladder cancer cell lines (J82, J82
and T24) was investigated. Expression of miR-214 was significantly
lower in all bladder cancer cell lines compared with that in
SV-HUC-1 cells (P<0.001; Fig.
2A). The T24 and J82 cell lines, with low endogenous
expression, were selected for transfection with miR-214 mimic. The
transfection efficiency was confirmed by RT-qPCR. Transfection with
miR-214 mimic significantly increased the expression level of
miR-214 in J82 and T24 cells compared with the level in the mimic
control group (P<0.05; Fig.
2A). Using a colony formation assay, it was demonstrated that
miR-214 mimic significantly reduced the colony formation ability of
the J82 and T24 cell lines compared with that observed in the mimic
control group (P<0.05; Fig.
2B). Matrigel invasion assays indicated that miR-214 mimic
significantly inhibited the invading ability of the J82 and T24
cell lines compared with that observed in the mimic control group
(P<0.05; Fig. 2C).
miR-214 reduces cisplatin resistance and
inhibits AKT phosphorylation
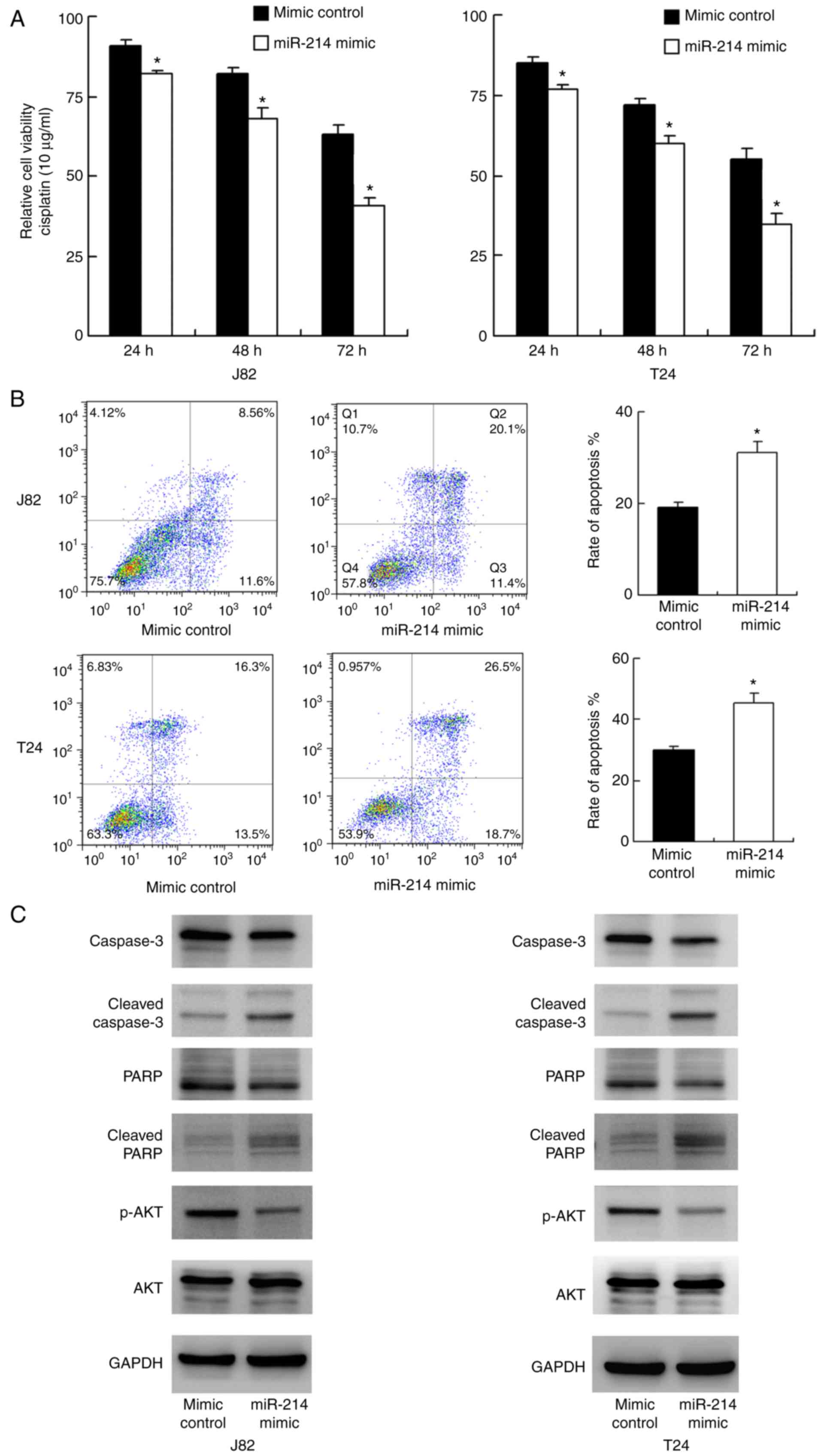
The role of miR-214 on chemoresistance of bladder
cancer cells was explored. A CCK-8 assay was conducted in T24 and
J82 cells treated with cisplatin (10 μg/ml). As demonstrated
in Fig. 3A, miR-214 mimic
significantly reduced cell viability in both cell lines at 24, 48
and 72 h following treatment with cisplatin compared with that
observed in the mimic control group (P<0.05). These results
suggest that miR-214 reduced chemoresistance of bladder cancer
cells. Annexin V/PI staining was conducted to measure the rate of
apoptosis. As indicated in Fig.
3B, transfection with miR-214 mimic significantly upregulated
the apoptosis rate after 24 h of cisplatin treatment in the J82 and
T24 cell lines. Western blotting was performed to examine the
change of related proteins. As indicated in Fig. 3C, transfection with miR-214 mimic
downregulated total caspase-3 and total PARP expression, and
upregulated cleaved caspase-3 and cleaved PARP. Several signaling
pathways involved in cancer cell survival were also analyzed, and
it was revealed that miR-214 inhibited AKT phosphorylation.
miR-214 targets and downregulates
netrin-1
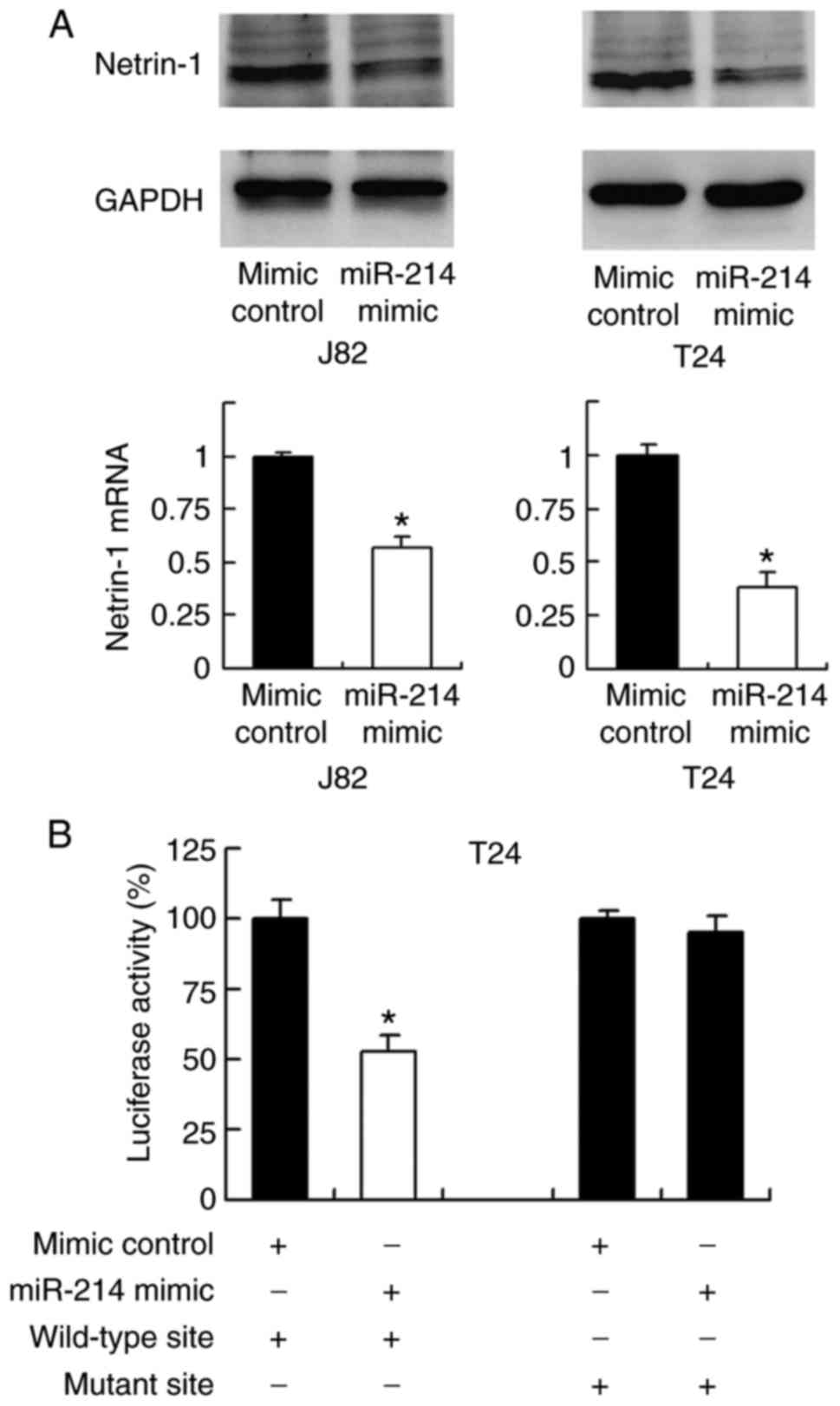
TargetScan software was used to predict potential
targets of miR-214, and this revealed that netrin-1 is one of the
target genes (17). To validate
the relationship between netrin-1 and miR-214, the change in
expression level of netrin-1 mRNA and protein was examined
following transfection with miR-214 mimic. As demonstrated in
Fig. 4A, transfection with
miR-214 mimic markedly and significantly downregulated netrin-1
expression at the protein and mRNA levels, respectively, compared
with the level in the mimic control group (P<0.05; Fig. 4A).
To validate if netrin-1 is the direct target of
miR-214, a luciferase reporter assay was conducted. Reporters with
wild-type (CCUGCUG) and mutant (CCUUUUG) 3′-UTR binding sites of
netrin-1 were introduced into J82 cells together with miR-214
mimic. The luciferase reporter assay demonstrated that miR-214
mimic significantly suppressed the luciferase intensity of the
wild-type reporter compared with that in the control group
(P<0.05); however, no significant change was observed in the
luciferase intensity of the mutant reporter (Fig. 4B). The above data indicates that
miR-214 binds to the 3′-UTR of netrin-1 to reduce its mRNA and
protein expression. In addition, the correlation between miR-214
and netrin-1 mRNA expression in bladder cancer tissues was
examined. Linear regression analysis demonstrated that the miR-214
expression level in cancer tissues was negatively correlated with
netrin-1 mRNA expression (P<0.01; Fig. 1C).
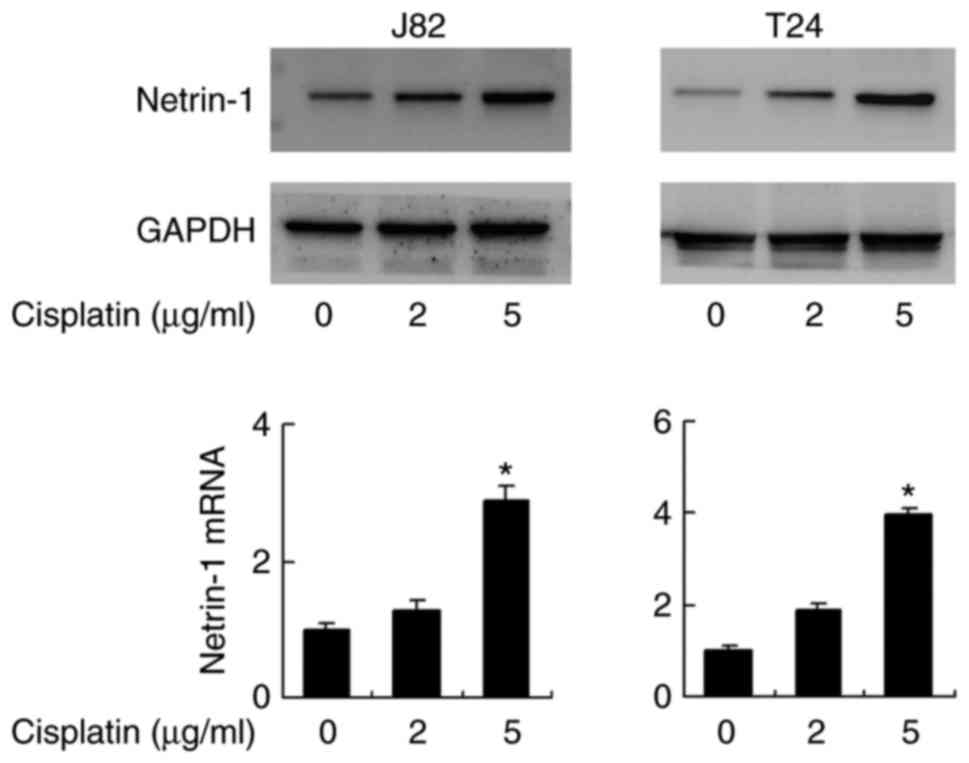
Cisplatin treatment induces netrin-1
expression in bladder cancer cells
Chemotherapeutic agents, including cisplatin and
doxorubicin, have been reported to induce netrin-1 expression,
which appears to be a survival mechanism, as the depletion of
netrin-1 results in cancer cell death (18). To confirm this, the alteration of
the netrin-1 expression level in bladder cancer cells following
cisplatin treatment was investigated. As demonstrated in Fig. 5, cisplatin treatment (2 and 5
μg/ml for 2 days) induced an increase of protein expression
in the J82 and T24 cell lines compared with the level in cells that
did not receive cisplatin treatment. At the mRNA expression level,
this increase was significant in cells treated with 5 μg/ml
cisplatin (P<0.05). mRNA expression levels of netrin-1 increased
significantly following treatment with 5 μg/ml cisplatin,
which was confirmed by one-way analysis of variance with post hoc
comparisons in both cell lines (J82: 5 μg/ml vs. control,
P=0.004 and 5 vs. 2 μg/ml, P=0.006; T24: 5 μg/ml vs.
control, P=0.001 and 5 vs. 2 μg/ml, P=0.002; Fig. 5).
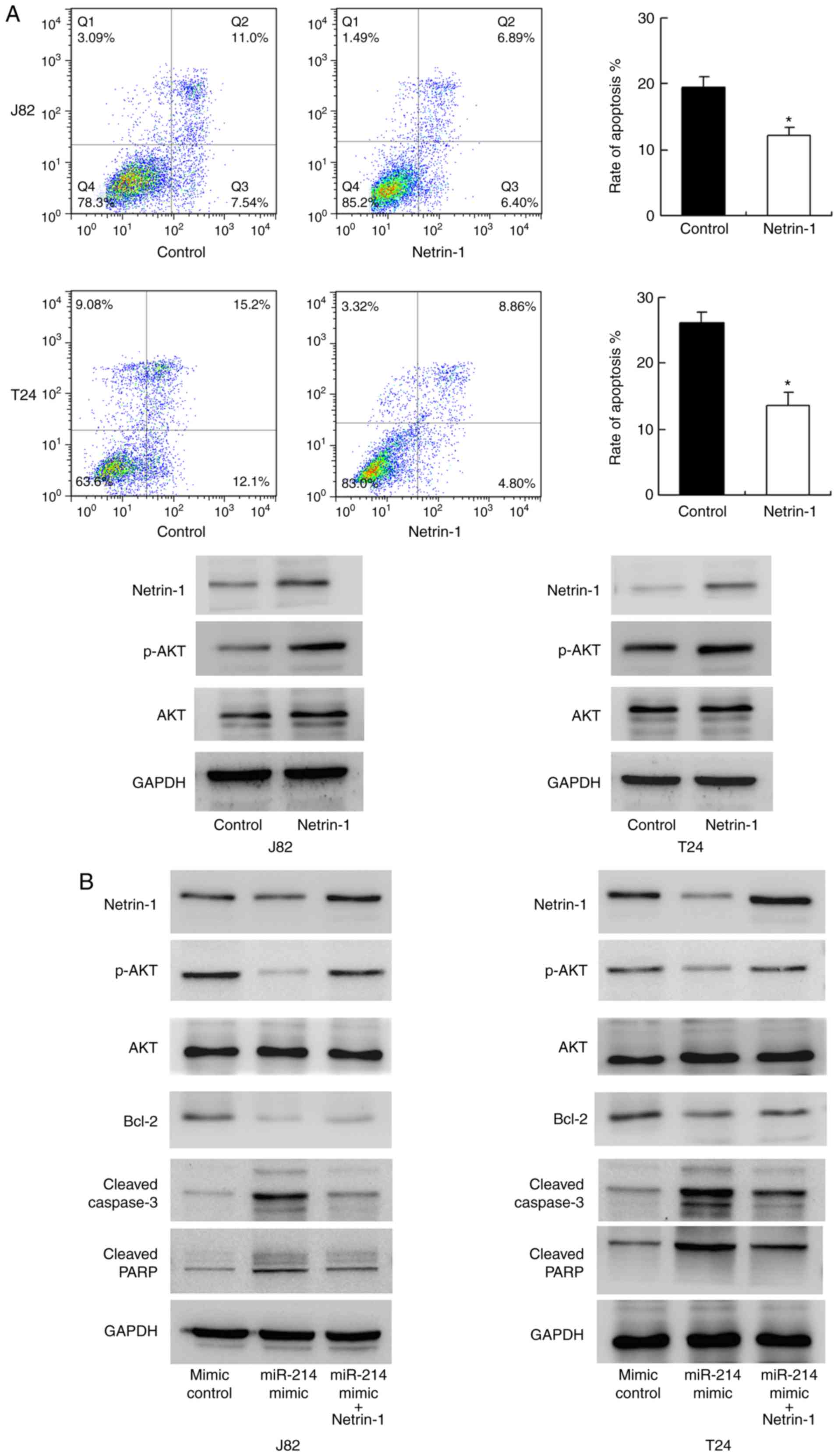
miR-214 regulates cisplatin resistance
through netrin-1
To validate the biological function of netrin-1,
netrin-1 was overexpressed in T24 and J82 cell lines. As
demonstrated in Fig. 6A, Annexin
V/PI analysis indicated that netrin-1 overexpression significantly
inhibited cisplatin-induced apoptosis compared with the level in
the control group (P<0.05). Western blotting indicated that
netrin-1 transfection upregulated AKT phosphorylation. These data
suggest that netrin-1 is associated with chemoresistance in bladder
cancer cells. To confirm the involvement of netrin-1 in the
biological effects of miR-214, netrin-1 plasmid transfection was
conducted in bladder cancer cells with miR-214 mimic. As indicated
in Fig. 6B, plasmid transfection
restored netrin-1 and p-AKT status in cells transfected with
miR-214 mimic, with downregulation of caspase/PARP cleavage. In
addition, the expression of the AKT target protein, Bcl-2, was
studied. miR-214 mimic downregulated Bcl-2 expression and netrin-1
transfection restored Bcl-2 status. Together, these data
demonstrate that the function of miR-214 in chemoresistance is, at
least partly, through netrin-1 regulation.
Discussion
Accumulating evidence has demonstrated the role of
miRNA during cancer development and progression. miR-214 is
regarded as a tumor suppressor that is downregulated in various
cancer types, including breast, lung and colorectal carcinoma
(15,19–21). The expression status of miR-214
has been reported to be downregulated in bladder cancer tissues and
significantly associated with tumor stage, lymph node status and
cancer grade (15). Urinary
levels of cell-free miR-214 could be an independent prognostic
parameter for non-muscle-invasive bladder cancer recurrence
(16). miR-214 exerts
tumor-suppressive effects in bladder cancer by downregulating
oncogenic PDRG1 expression (15).
In the present study, it was demonstrated that miR-214 expression
levels are downregulated in bladder cancer tissues and cell lines.
The suppressive effects of miR-214 on bladder cancer proliferation
and invasion were also validated in the present study. However, its
involvement in the development of chemoresistance of bladder cancer
cells had yet to be explored.
Thus, the present study investigated the influence
of miR-214 on resistance to chemotherapy by using cisplatin
treatment in J82 and T24 cells. The results demonstrated that
miR-214 mimic significantly reduced drug resistance by upregulating
the apoptosis rate. A series of apoptosis-related proteins were
examined. The present results revealed that total caspase-3 and
total PARP expression levels were decreased following transfection
with miR-214 mimic, while cleaved caspase-3 and cleaved PARP were
markedly upregulated. These results indicate that miR-214 promotes
apoptosis. In addition, the present study indicated that miR-214
was able to suppress AKT phosphorylation. AKT signaling is involved
in the regulation of cell apoptosis by upregulating pro-survival
Bcl-2 family proteins, including Bcl-2 and Bcl-extra large
(22). Thus, the effects of
miR-214 on chemoresistance may be dependent on its regulation of
AKT/Bcl-2 signaling.
The present study further explored the mechanism by
which miR-214 regulates apoptosis and chemoresistance. Potential
target genes of miR-214 were predicted using TargetScan software,
and netrin-1 was identified as one of the targets. Netrin-1 has
been identified as an oncoprotein in several cancer types, such as
bladder cancer (23). Previously
we reported that netrin-1 overexpression was associated with poor
prognosis in patients with bladder cancer (23). A previous report suggested that
netrin-1 may enhance chemoresistance of cancer cells through
regulation of its receptor activity (19). In the present study, the effects
of netrin-1 on bladder cancer apoptosis and AKT signaling were
investigated. The results demonstrated that netrin-1 plasmid
transfection significantly downregulated apoptosis and notably
upregulated AKT phosphorylation in bladder cancer cells. In
addition, miR-214 mimic significantly and markedly decreased
netrin-1 mRNA and protein expression levels, respectively. Notably,
a negative relationship was identified in bladder cancer tissues
between miR-214 and netrin-1 mRNA expression levels in the present
study. Furthermore, luciferase reporter assays revealed that
miR-214 could directly bind to the 3′-UTR of netrin-1,
demonstrating that netrin-1 is a direct target of miR-214. These
results together suggest that miR-214 downregulates netrin-1, which
in turn inhibits p-AKT and reduces chemoresistance. To further
confirm this, netrin-1 plasmid was used to restore its function in
the present study. Netrin-1 plasmid transfection restored cisplatin
resistance downregulated by miR-214. Taken together, these data
suggest that miR-214 inhibits chemoresistance in bladder cancer by
targeting netrin-1.
In conclusion, to the best of our knowledge, the
present study is the first to demonstrate the relationship between
miR-214 and chemoresistance of bladder cancer cells. miR-214 may
reduce cisplatin resistance and AKT signaling by targeting
netrin-1. The present findings may contribute to an improved
understanding of the mechanism involved in bladder cancer
chemoresistance.
Acknowledgments
The present study was supported by the National
Natural Science Foundation of China (grant no. 81402088).
Notes
[1] Competing
interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Reddy OL, Cates JM, Gellert LL, Crist HS,
Yang Z, Yamashita H, Taylor JA III, Smith JA Jr, Chang SS, Cookson
MS, et al: Loss of FOXA1 drives sexually dimorphic changes in
urothelial differentiation and is an independent predictor of poor
prognosis in bladder cancer. Am J Pathol. 185:1385–1395. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Szarvas T, László V, Vom Dorp F, Reis H,
Szendröi A, Romics I, Tilki D, Rübben H and Ergün S: Serum
endostatin levels correlate with enhanced extracellular matrix
degradation and poor patients' prognosis in bladder cancer. Int J
Cancer. 130:2922–2929. 2012. View Article : Google Scholar
|
|
4
|
Yang GL, Zhang LH, Bo JJ, Huo XJ, Chen HG,
Cao M, Liu DM and Huang YR: Increased expression of HMGB1 is
associated with poor prognosis in human bladder cancer. J Surg
Oncol. 106:57–61. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dobruch J, Daneshmand S, Fisch M, Lotan Y,
Noon AP, Resnick MJ, Shariat SF, Zlotta AR and Boorjian SA: Gender
and bladder cancer: A collaborative review of etiology, biology,
and outcomes. Eur Urol. 69:300–310. 2016. View Article : Google Scholar
|
|
6
|
Mitra AP: Molecular substratification of
bladder cancer: Moving towards individualized patient management.
Ther Adv Urol. 8:215–233. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lu J, Getz G, Miska EA, Alvarez-Saavedra
E, Lamb J, Peck D, Sweet-Cordero A, Ebert BL, Mak RH, Ferrando AA,
et al: MicroRNA expression profiles classify human cancers. Nature.
435:834–838. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Takamizawa J, Konishi H, Yanagisawa K,
Tomida S, Osada H, Endoh H, Harano T, Yatabe Y, Nagino M, Nimura Y,
et al: Reduced expression of the let-7 microRNAs in human lung
cancers in association with shortened postoperative survival.
Cancer Res. 64:3753–3756. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang DQ, Zhou CK, Jiang XW, Chen J and
Shi BK: Increased expression of miR-222 is associated with poor
prognosis in bladder cancer. World J Surg Oncol. 12:2412014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kohler CU, Bryk O, Meier S, Lang K,
Rozynek P, Brüning T and Käfferlein HU: Analyses in human
urothelial cells identify methylation of miR-152, miR-200b and
miR-10a genes as candidate bladder cancer biomarkers. Biochem
Biophys Res Commun. 438:48–53. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Puerta-Gil P, Garcia-Baquero R, Jia AY,
Ocaña S, Alvarez-Múgica M, Alvarez-Ossorio JL, Cordon-Cardo C, Cava
F and Sánchez-Carbayo M: miR-143, miR-222, and miR-452 are useful
as tumor stratification and noninvasive diagnostic biomarkers for
bladder cancer. Am J Pathol. 180:1808–1815. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang J, Su B, Gong C, Xi Q and Chao T:
miR-214 promotes apoptosis and sensitizes breast cancer cells to
doxorubicin by targeting the RFWD2-p53 cascade. Biochem Biophys Res
Commun. 478:337–342. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yu X, Luo A, Liu Y, Wang S, Li Y, Shi W,
Liu Z and Qu X: MiR-214 increases the sensitivity of breast cancer
cells to tamoxifen and fulvestrant through inhibition of autophagy.
Mol Cancer. 14:2082015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li QQ, Xie YK, Wu Y, Li LL, Liu Y, Miao
XB, Liu QZ, Yao KT and Xiao GH: Sulforaphane inhibits cancer
stem-like cell properties and cisplatin resistance through
miR-214-mediated downregulation of c-MYC in non-small cell lung
cancer. Oncotarget. 8:12067–12080. 2017.PubMed/NCBI
|
|
15
|
Wang J, Zhang X, Wang L, Yang Y, Dong Z,
Wang H, Du L and Wang C: MicroRNA-214 suppresses oncogenesis and
exerts impact on prognosis by targeting PDRG1 in bladder cancer.
PLoS One. 10:e01180862015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang J, Zhang X, Wang L, Dong Z, Du L,
Yang Y, Guo Y and Wang C: Downregulation of urinary cell-free
microRNA-214 as a diagnostic and prognostic biomarker in bladder
cancer. J Surg Oncol. 111:992–999. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pan X, Peng G, Liu S, Sun Z, Zou Z and Wu
G: MicroRNA-4649-3p inhibits cell proliferation by targeting
protein tyrosine phosphatase SHP-1 in nasopharyngeal carcinoma
cells. Int J Mol Med. 36:559–564. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Paradisi A, Creveaux M, Gibert B, Devailly
G, Redoulez E, Neves D, Cleyssac E, Treilleux I, Klein C,
Niederfellner G, et al: Combining chemotherapeutic agents and
netrin-1 interference potentiates cancer cell death. EMBO Mol Med.
5:1821–1834. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
20
|
Long LM, He BF, Huang GQ, Guo YH, Liu YS
and Huo JR: microRNA-214 functions as a tumor suppressor in human
colon cancer via the suppression of ADP-ribosylation factor-like
protein 2. Oncol Lett. 9:645–650. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen DL, Wang ZQ, Zeng ZL, Wu WJ, Zhang
DS, Luo HY, Wang F, Qiu MZ, Wang DS, Ren C, et al: Identification
of microRNA-214 as a negative regulator of colorectal cancer liver
metastasis by way of regulation of fibroblast growth factor
receptor 1 expression. Hepatology. 60:598–609. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang YB, Qin J, Zheng XY, Bai Y, Yang K
and Xie LP: Diallyl trisulfide induces Bcl-2 and
caspase-3-dependent apoptosis via downregulation of Akt
phosphorylation in human T24 bladder cancer cells. Phytomedicine.
17:363–368. 2010. View Article : Google Scholar
|
|
23
|
Liu J, Kong CZ, Gong DX, Zhang Z and Zhu
YY: PKC α regulates netrin-1/UNC5B-mediated survival pathway in
bladder cancer. BMC Cancer. 14:932014. View Article : Google Scholar
|