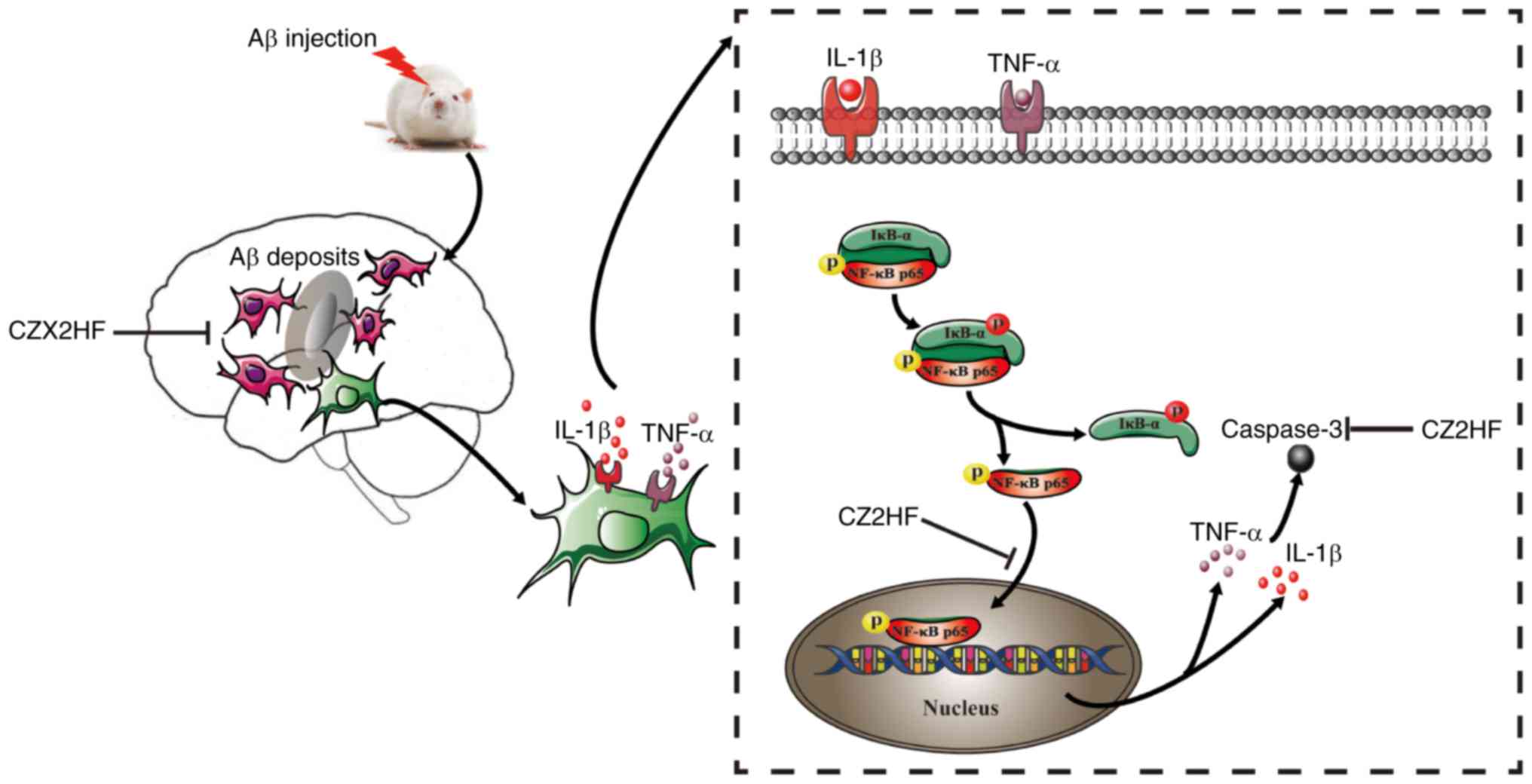
Introduction
Alzheimer’s disease (AD) is a major type of dementia
in the elderly, which is characterized by progressive learning and
memory impairment. The present consensus is that the typical
pathological features of AD are extracellular β-amyloid (Aβ)
plaques and intracellular neurofibrillary tangles in the brain,
accompanied by neuronal damage or loss (1,2).
Due to an increase in the aging population in recent years, there
are currently ~46 million dementia patients worldwide and the
number of patients enrolled in 2,050 is expected to increase to 135
million, which will put a great economic burden on society and
patients’ families (3). The
pathogenesis of AD is complex, and remains to be fully elucidated.
It has been previously established that the Aβ cascade theory has
an important role in the development of AD (4). Aβ is derived from β-secretase and
γ-secretase and the deposition of Aβ, particularly
Aβ1-42 leads to neurotoxicity and neurodegeneration
during the progress of AD (5).
Physiologically, the formation and removal of Aβ in the brain is
maintained in a dynamic equilibrium state; however, when this
balance is dysregulated, it may lead to abnormal deposition of Aβ
and disturb the physiological activity of the neuronal cells
(6). A previous study showed that
Aβ deposition produced a sequence of cascade reactions, such as
exacerbation of the inflammatory response, including increased
interleukin-1β (IL-1β), tumor necrosis factor (TNF-α),
cyclooxygenase-2 (COX-2) expression and levels of nuclear factor-κB
(NF-κB) (7). Additionally, these
inflammatory factors may increase β-amyloid precursor protein (APP)
expression in the brain and upregulate the activity of γ-secretase;
therefore, the levels of Aβ were increased in turn (8). In addition, a previous study
indicated that Aβ deposition in the brain may induce neuronal
apoptosis, which may lead to further learning and memory
impairment. B-cell lymphoma-2 (Bcl-2) gene family members, such as
Bcl-2 and Bcl-2-associated X, apoptosis regulator (Bax), have
important roles in the process of apoptosis (9). It is of note that it has been
previously demonstrated that the learning and memory impairment of
the Aβ1-40-induced AD rats may be ameliorated through
regulation of the apoptosis-associated genes, such as Bax and
Bcl-2. Therefore, inhibition of neuroinflammation and neuronal
apoptosis may be used as a promising strategy for the clinical
treatment of AD.
Evidence from clinical and experimental trials
revealed that cholinesterase inhibitors such as donepezil, tacrine
and galantamine, and a N-methyl-D-aspartate receptor antagonists,
such as memantine may be used as AD-treatment agents (10-12). However, these drugs had limited
use in a clinical setting due to their single target, their price
and multiple side effects (13).
Therefore, Traditional Chinese Medicine (TCM) may be a promising
treatment method for AD as it has advantages of multi-targets and
reduced side effects.
The cu-zhi-2-hao-fang (CZ2HF) decoction, an
empirical formula of TCM, which consisted of Herba Epimedium,
Rhizoma curculiginis, Morinda officinalis, Acorus gramineus, Lycium
barbarum, Scrophularia ningpoensis, Cinnamomum cassia Presl,
Rhizoma zingiberis (Table I).
According to TCM theory, Herba Epimedium is the primary component
in this decotion, which is one of the traditional Chinese herbs for
treating various diseases, such as AD and its main active
ingredients, icariin and icariside II downregulate
Aβ1-40 and Aβ1-42 expression levels in the
brain of Tg2576 transgenic mice to mitigate learning and memory
impairment and reduce TNF-α, COX-2, IL-1β expression levels and
neuronal apoptosis to improve Aβ25-35-induced learning
and memory impairments in rats (14-16). CZ2HF was used to clinically
prevent and treat amnesia. However, whether CZ2HF may alleviate
Aβ25-35-induced learning and memory impairment and its
underlying mechanism remains to be elucidated. Therefore, the
present study was designed to investigate the effect of CZ2HF on
Aβ25-35-induced learning and memory impairment and
further elucidate its possible action mechanism.
 | Table IComposition of cu-zhi-2-hao-fang. |
Table I
Composition of cu-zhi-2-hao-fang.
| Common name | Latin name | Quantity (g) |
|---|
| Herba
Epimedium | Epimedium
brevicornu Maxim. | 9 |
| Rhizoma
curculiginis | Curculigo
orchioides Gaertn. | 9 |
| Morinda
officinalis | Morinda
officinalis How. | 9 |
| Acorus
gramineus | Acorus
tatarinowii. | 9 |
| Lycium
barbarum | Lycium
barbarum L. | 9 |
| Scrophularia
ningpoensis | Scrophularia
ningpoensis Hemsl. | 9 |
| Cinnamomum
cassia Presl | Cassia
Twig | 5 |
| Rhizoma
zingiberis | Zingiber
officinale Roscoe | 5 |
Materials and methods
Agents
Donepezil hydrochloride (1511010) was obtained from
Affiliated Hospital of Zunyi Medical University (Zunyi, China),
Aβ25-35 (the amino acid sequence is
Gly-Ser-Asn-Lys-Gly-A la-Ile-Ile-Gly-Leu-Met) was purchased from
Sigma-Aldrich (cat. no. A4559; Merck Millipore, Darmstadt,
Germany), primary antibodies of COX-2 (cat. no. ab15191),
active-caspase-3 (cat. no. ab13847), Bax (cat. no. ab7977), Bcl-2
(cat. no. ab7973), IL-1β (cat. no. ab9787), TNF-α (cat. no.
ab66579), Aβ1-42 (cat. no. ab10148) were acquired from
Abcam (Cambridge, UK), pro-caspase-3 antibody (cat. no. sc-7148)
was obtained from Santa Cruz Biotechnology, Inc. (Dallas, TX, USA),
primary antibodies of inhibitor of κB (IκB-α; cat. no. 9242),
phosphorylated (p)-nuclear factor (NF)-κB p65 (cat. no. 3033),
NF-κB p65 (cat. no. 8242) were acquired from Cell Signaling
Technology, Inc. (Danvers, MA, USA).
Preparation of CZ2HF decoction
CZ2HF consisted of 8 ingredients, as presented in
Table I. CZ2HF was provided by
Affiliated Hospital of Zunyi Medical University and identified by
Professor Jianwen Yang (School of Pharmacy, Zunyi Medical
University, Zunyi, China). Briefly, a mixture of 9 g Herba
Epimedium, 9 g Rhizoma curculiginis, 9 g Morinda
officinalis, 9 g Acorus calamus, 9 g Lycium barbarum, 9 g
Scrophularia ningpoensis, 5 g Cinnamomum cassia
Presl, 5 g Zingiberis rhizome was soaked for 60 min with 1,000 ml
distilled water and boiled for 1.5 h. Subsequently, the filtrate
was gathered and the residue was boiled again for an additional 1 h
with distilled water. The extraction was further condensed,
combined and lyophilized according to the protocol as previously
described (17). The yield was
21.6% relative to the original crude quantity.
Preparation of animal model and drug
treatment
A total of 98 healthy male Sprague-Dawley (SD) rats
(250-300 g) were purchased from the Laboratory Animal Center of the
Third Military Medical University (Chongqing, China; certificate
no. SCXK2012-0011). The rats were housed in specific pathogen-free
conditions, under a 12-h light/dark cycle, temperature was 22±1°C,
humidity was 60±2% and were given free access to food and water.
All experiments were approved by the Ethics Committee and performed
according to the current guide for the care and use of laboratory
animal standard, which was set up by Zunyi Medical University
Animal Studies Committee (argument number [2015] 2-043). The rats
were randomly divided into 7 groups as follows: i) Sham; ii)
sham+CZ2HF (400 mg/kg); iii) model (Aβ25-35); iv)
Aβ25-35+CZ2HF (100 mg/kg); v) Aβ25-35+CZ2HF
(200 mg/kg); vi) Aβ25-35+CZ2HF (400 mg/kg); and vii)
Aβ25-35+donepezil (1.0 mg/kg) as the positive drug group
(n=14 rats per group. Briefly, Aβ25-35 (1 mg) was
dissolved in 500 μl saline, configured as 2.0
μg/μl solution, placed in 37°C incubator for 4 days,
in order to induce a clustered state to enhance its toxicity
(18). Subsequently, SD rats were
anesthetized with an intraperitoneal injection of 2% pentobarbital
sodium, the rat’s brain was fixed a stereotaxic device, and the
following hippocampus needle coordinates were used: 3.5 mm
posterior to the bregma, 2.5 mm lateral to the sagittal suture, 3.5
mm beneath the surface of brain. The needle was retained in the
bilateral hippocampi for 5 min and the 5 μl
Aβ25-35 was injected. Various doses of CZ2HF were
administered orally daily for a continuous period for 15 days. The
rats in CZ2HF group were treated with CZ2HF alone, and the sham
group were given double-distilled water at an equal volume to the
CZ2HF solution.
Morris water maze test
The Morris water maze test (MWM) was performed in
order to determine the learning and memory function of the rats
from the 7 groups as described in our previous study (19). The rats were trained and exposed
to 4 successive memory acquisition trials in the MWM to analyze
their capacity to escape and find the platform, which was performed
daily between days 11 and 16 after the Aβ25-35
injection. On day 16, the spatial probe experiment was performed to
detect the ability of spatial memory. All SD rats were subjected to
anesthesia by 0.3 ml of 2% pentobarbital sodium injection after
intraperitoneal examination of the MWM.
Hematoxylin and eosin (H&E)
staining
Following fixation in 4% for 48 h (pH 7.4), the
brains were removed, fixed with 4% paraformaldehyde at 4°C,
dehydrated and embedded in paraffin. Subsequently, 3-μm
thick frozen sections were prepared and H&E staining at room
temperature for 12 min was used to detect pathological changes in
the CA1 region of hippocampal tissue by an independent pathologist.
Images of the histopathological examination were observed using a
light microscope. Three rats per group were used for H&E
staining.
Nissl staining
Brains were fixed with 4% paraformaldehyde at 4°C
for 48 h and subsequently embedded in paraffin. Sections
(3-μm thick) of rat brain tissue were stained with toluidine
blue at 60°C for 10 min. The Nissl bodies were stained blue-purple
in the CA1 region of the hippocampus to estimate the morphological
changes of neurons in the CA1 area and assessed by a light
microscope.
Terminal
deoxynucleotidyltransferase-mediated dUTP nick-end labeling (TUNEL)
staining
Apoptosis was evaluated using TUNEL staining with
the In Situ Cell Death Detection kit (cat. no. 11684817910; Roche
Diagnostics GmbH, Mannheim, Germany), according to the
manufacturer’s protocol. The brain slices of rats from each group
were washed twice with double distilled water for 5 min, then the
sections were soaked in 3% H2O2 solution for
15 min. Subsequently, the slices were washed with PBS 3 times for 5
min, placed in a dark chamber and proteinase K was added to the
working solution. Next, the brain slices were incubated for 60 min
at 37 °C with TUNEL reaction mixture and the sections were washed
with PBS again and incubated for 30 min at 37°C with converter-POD
work liquid. The sections were subsequently treated with DAB
substrate solution and washed again with PBS. A total of 3 images
were captured randomly for each section and counted using a
fluorescent microscope as described in our previous study (19).
Western blotting
Expression levels of TNF-α, IL-1β, COX-2, IκB-α,
NF-κB p65, p-NF-κB p65, Bax, Bcl-2, caspase-3, Aβ1-42
were dretermined using western blotting. Briefly, three rats were
randomly selected from each group and sacrificed, the hippocampal
tissues were dissected and immediately frozen at −80°C. Then, the
subsequent procedures were performed as described in our previous
study (20). The corresponding
proteins in this study were analyzed using primary antibodies
against TNF-α (1:2,000), IL-1β (1:1,000), COX (1:1,000), IκB-α
(1:2,000), NF-κB p65 (1:1,000), p-NF-κB p65 (1:1,000); Bax (1:500),
Bcl-2 (1: 500), pro-caspase-3 (1:1,000), active-caspase-3 (1:1,000)
and Aβ1-42 (1:2,000). The membranes were incubated
overnight with the primary antibodies at 4°C. Susbequently, the
membranes were washed twice with TBST and incubated with secondary
antibodies goat anti-rabbit IgG H&L (cat. no. ab6702; 1:1,000;
Abcam) for 2 h at room temperature. The blots were visualized using
Davinch-Chemi™ imaging system and the relative band optical
intensity was quantified using Quantity One 1-D analysis software
version 4.52 (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Statistical analysis
Data were expressed as the mean ± standard error of
the mean and analyzed using SPSS version 17.0 (SPSS, Inc., Chicago,
IL, USA). Data were analyzed by one-way analysis of variance and
differences among means were analyzed using Dunnett’s test or
Tukey-Kramer’s multiple comparison test. P<0.05 was considered
to indicate a statistically significant difference.
Results
CZ2HF mitigates
Aβ25-35-induced learning and memory impairment in
rats
In order to investigate whether CZ2HF may alleviate
the learning and memory impairment induced by Aβ25-35 in
rats, the spatial learning and memory function of rats was
determined using the MWM test, which was performed from day 11 to
day 16 after the Aβ25-35 injection (Fig. 1A). The findings revealed that the
model group rats had a notably elevated escape latency compared
with the sham group, indicating that injection of
Aβ25-35 impaired their ability of spatial learning.
However, CZ2HF attenuated escape latency compared with model group
(F6,85=2.217; P< 0.05; Fig. 1B). Following the hidden platform
training on day 16, a spatial probe test was performed to determine
spatial memory abilities by counting the time spent in the target
quadrant of the rats of various groups (14). The findings revealed that the rats
in the model group spent shorter time in the target quadrant
compared with the sham group rats. However, CZ2HF increased the
retention time in the target quadrant compared with the model group
(Fig. 1C). No significant
difference was identified between the swimming speed of the
different treatment rat groups (Fig.
1D), indicating that CZ2HF and donepezil did not affect the
motor function of rats.
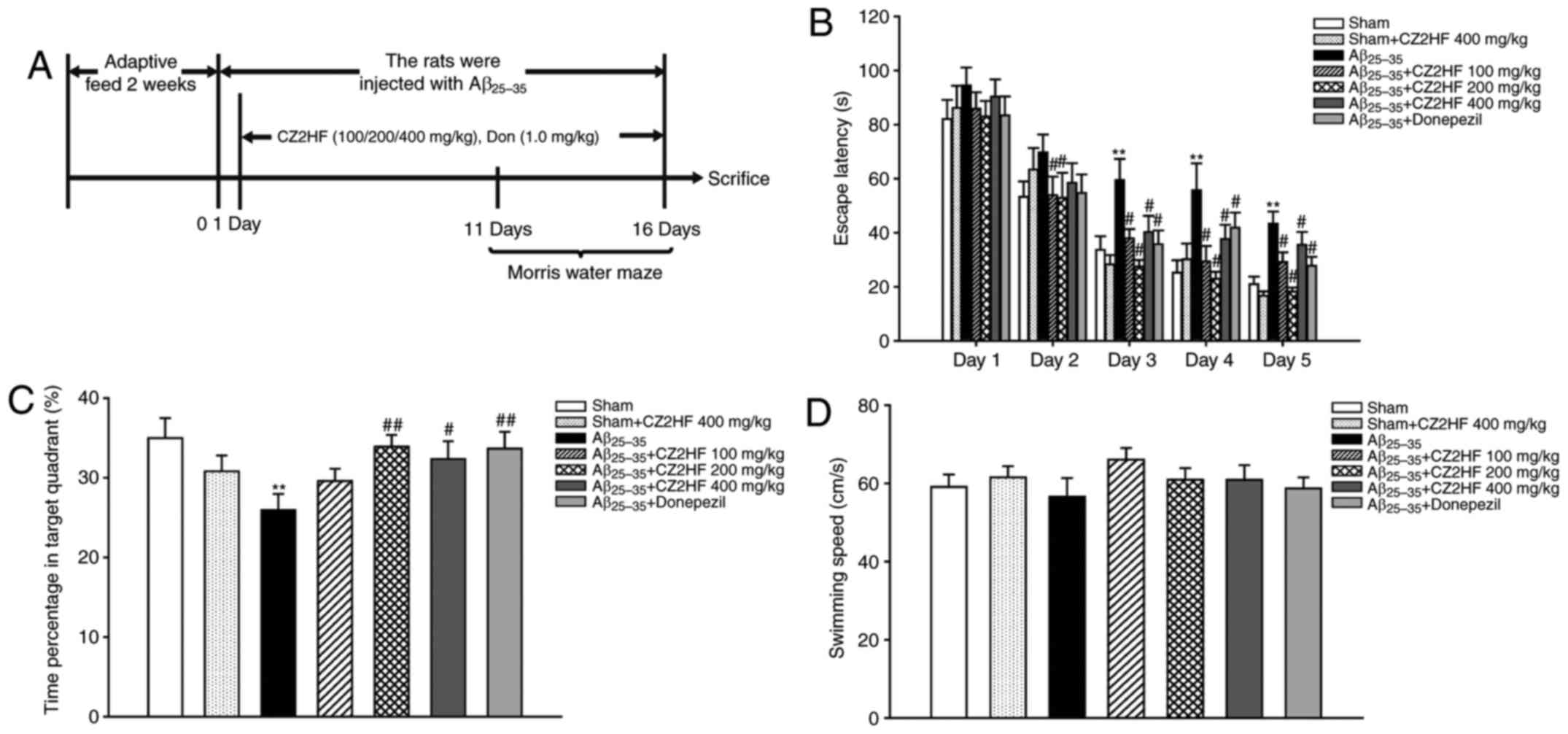 | Figure 1Effect of CZ2HF on
Aβ25-35-induced learning and memory impairments in rats.
(A) Experimental design. (B) Escape latency. (C) Time spent in
target quadrant. (D) Swimming speed. **P<0.01 vs.
sham; #P<0.05, ##P<0.01 vs. the
Aβ25-35. Sham (n=14), Aβ25-35 (n=11),
Sham+CZ2HF (400 mg/kg) (n=13), Aβ25-35+CZ2HF (100 mg/kg)
(n=12), Aβ25-35+CZ2HF (200 mg/kg) (n=12),
Aβ25-35+CZ2HF (400 mg/kg) (n=13), Aβ25-35+Don
(1.0 mg/kg) (n=13). CZ2HF, cu-zhi-2-hao-fang; Aβ25-35,
β-amyloid 25-35; Don, donepezil. |
CZ2HF reduces Aβ25-35-induced
neuronal injury of hippo- campus
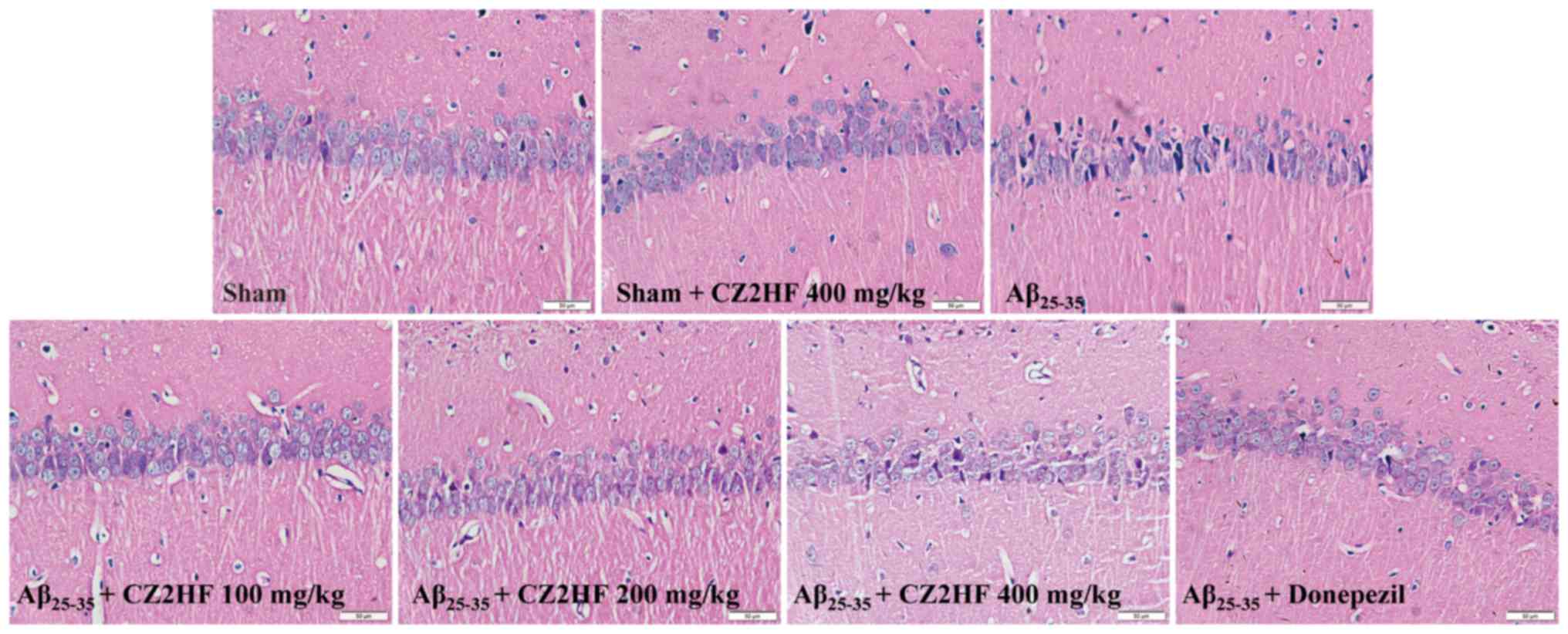
H&E and Nissl staining were used to evaluate the
effects of CZ2HF on the morphology of hippocampal neurons and
neuronal injury. The current findings revealed that the neurons in
the CA1 region of the hippocampus of rats in the sham group had
high density, the nuclei and cytoplasm were homogeneous and the
edges were clear. However, in the model group, the neurons were
disordered, their density was low and a large number of cells were
nucleated, indicating that Aβ25-35 damaged the neuronal
cells in the rat hippocampus. However, CZ2HF (100, 200, 400 mg/kg)
and donepezil treatment notably ameliorated the neuronal structure
and density (Fig. 2).
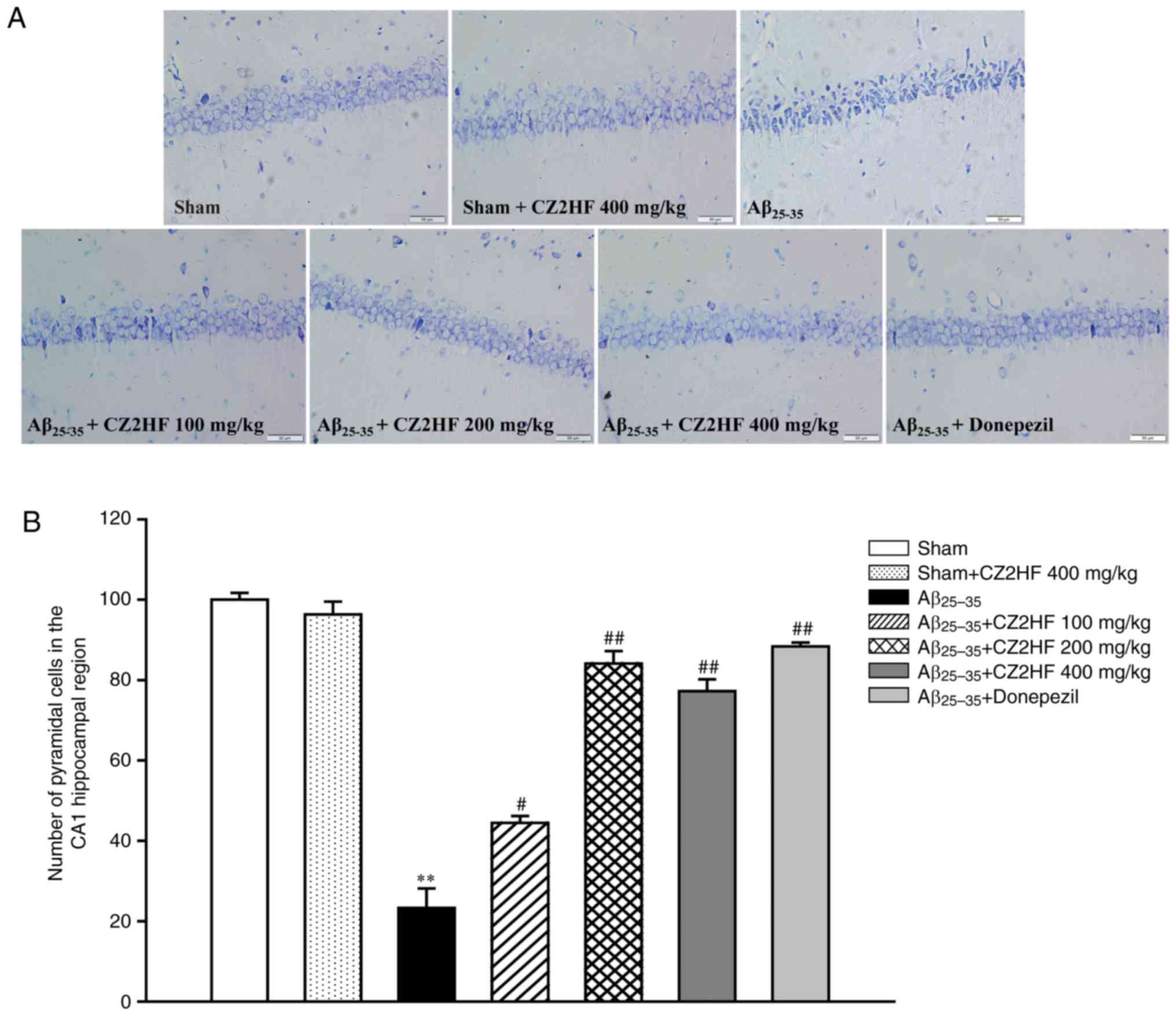
Furthermore, the results of Nissl staining revealed that the
neurons in CA1 region of sham were arranged neatly and densely.
Additionally, the nuclei and cytoplasm were stained uniformly and
the structure of neuron was clear and complete (Fig. 3A). However, the cellular structure
became unclear, the cell density was lower and the neurons were
disordered in the model group, indicating that Aβ25-35
damaged the hippocampal neurons. CZ2HF treatment ameliorated the
Aβ25-35-induced the injury of neuronal structure
(Fig. 3A). Meanwhile, these
effects were also confirmed by the number of pyramidal cells
counted in the CA1 hippocampal region of the rats (Fig. 3B).
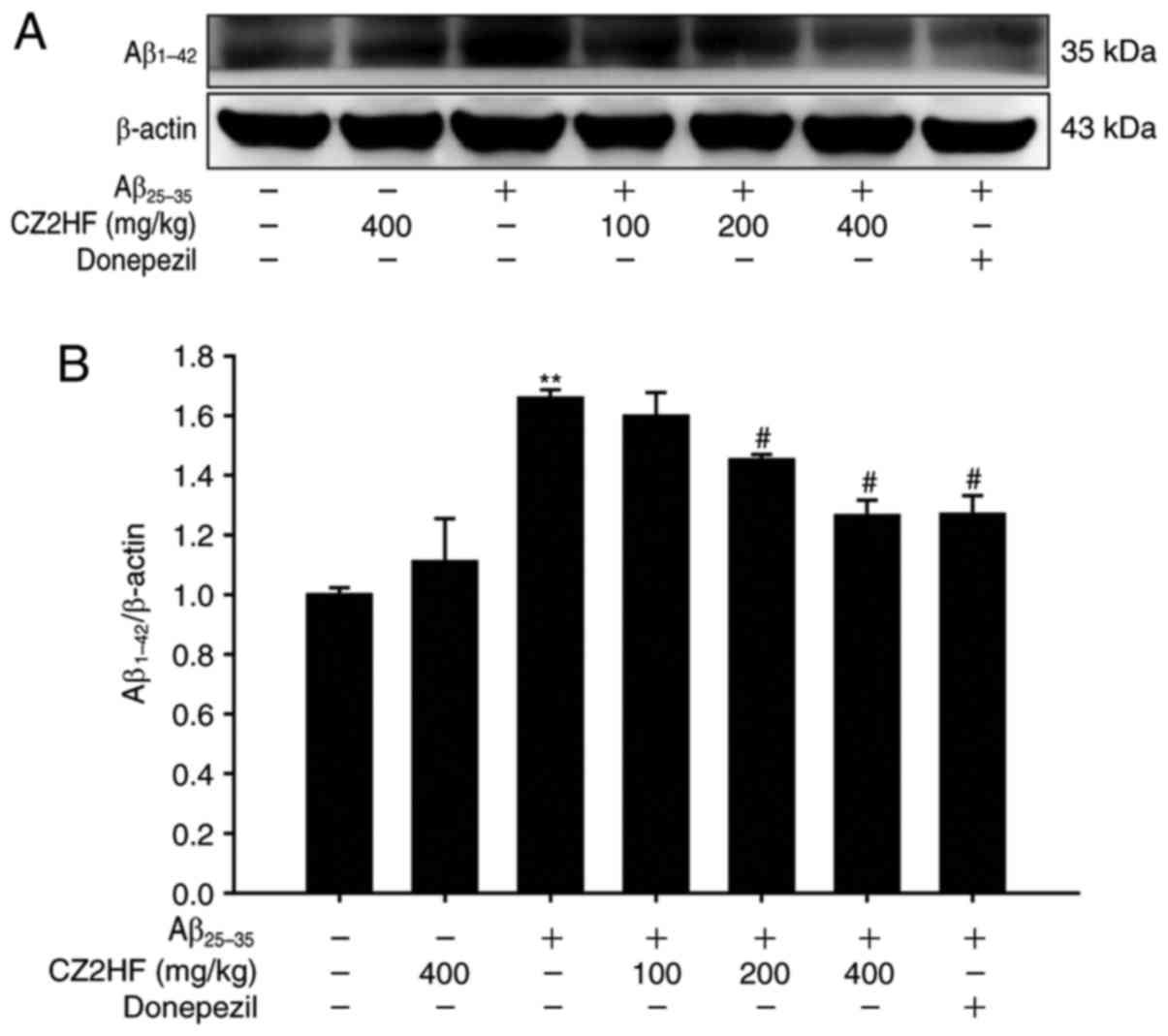
CZ2HF attenuates the level of hippocampal
Aβ1-42 induced by Aβ25-35
Western blotting was used to detect the level of
Aβ1-42 in the rat hippocampus induced by
Aβ25-35 exposure (Fig.
4A). The present study determined that the level of
Aβ1-42 was significantly increased in model group;
however, CZ2HF (100, 200, 400 mg/kg) and donepezil notably reduced
the level of Aβ1-42 in the rat hippocampus injected with
Aβ25-35, suggesting that CZ2HF and donepezil may block
Aβ25-35-induced Aβ1-42 increase
(F6,14=6.283; P<0.01; Fig. 4B).
CZ2HF represses Aβ25-35
-induced neuroinflammatory factors in rats
In order to determine the role of neuroinflammation
during the process of Aβ25-35-induced cognitive
impairment in rats, the inflammatory factors in hippocampi were
determined using western blotting (Fig. 5A). It was shown that TNF-α, IL-1β,
and COX2 expression levels were increased in the model group,
suggesting that Aβ25-35 triggered the inflammatory
response. However, CZ2HF (200, 400 mg/kg) and donepezil treatment
significantly reduced the increase in these inflammatory factors
including COX2 (F6,14=2.96; P<0.05; Fig. 5B), IL-1β (F6,14=7.802;
P<0.01; Fig. 5C) and TNF-α
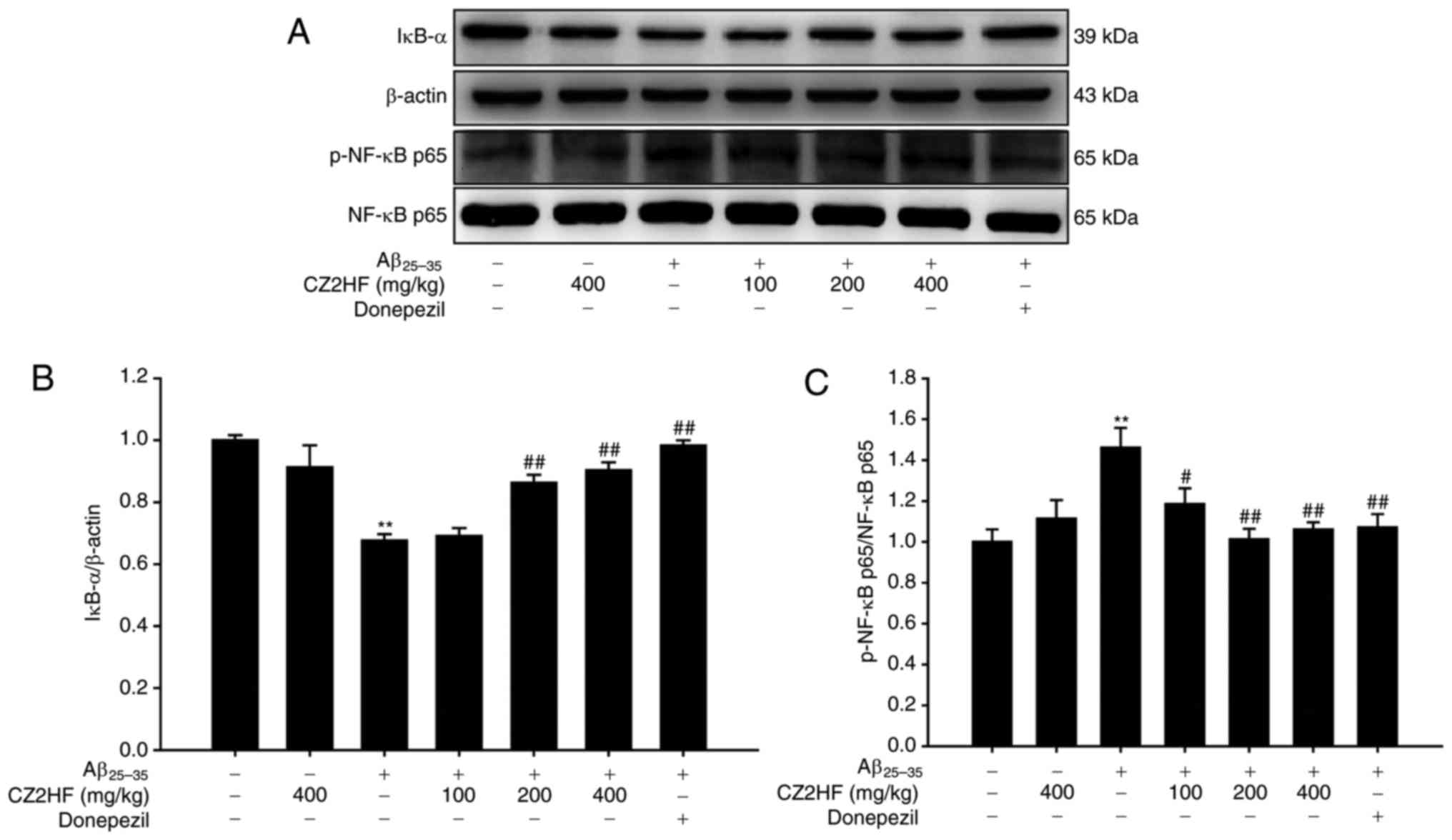
(F6,14=6.082; P<0.01; Fig. 5D). Furthermore, as NF-κB has a key
role in the development of AD (Fig.
6A) and it is upstream of the aforementioned inflammatory
factors, these findings were confirmed that CZ2HF significantly
increased the level of IκB-α (F6,14=13.850; P<0.05;
Fig. 6B) and reduced the p-NF-κB
p65 level (F6,14=3.829; P<0.05; Fig. 6C). Therefore, CZ2HF may block the
phosphorylation of NF-κB p65 induced by Aβ25-35.
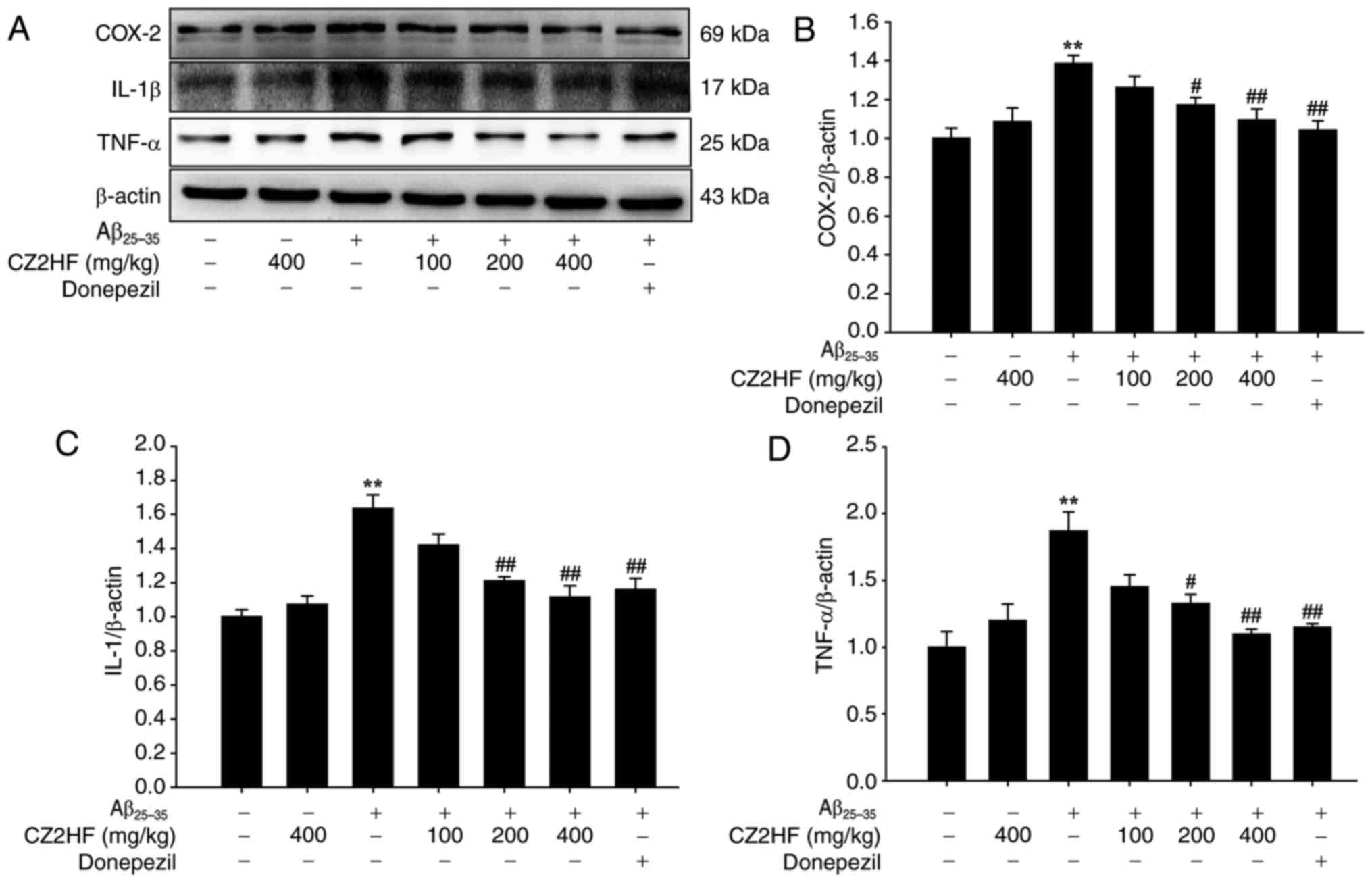 | Figure 5Effect of CZ2HF on COX-2, TNF-α and
IL-1β expression levels in hippocampus induced by
Aβ25-35 in rats. (A) Representative images of western
blotting for COX-2, TNF-α, IL-1β protein expression. Quantification
of (B) COX-2, (C) IL-1β and (D) TNF-α protein expression levels.
**P<0.01 vs. sham; #P<0.05,
##P<0.01 vs. Aβ25-35 (n=3). CZ2HF,
cu-zhi-2-hao-fang; Aβ25-35, β-amyloid 25-35; COX-2,
cyclooxygenase-2; IL-1β, interleukin-1β; TNF-α, tumor necrosis
factor-α. |
CZ2HF inhibits Aβ25-35-induced
hippocampal neuronal apoptosis in rats
The effect of CZ2HF on the apoptosis of hippocampal
neurons exposed to Aβ25-35 was evaluated using TUNEL
staining (Fig. 7A). The findings
demonstrated that the number of apoptotic cells in the model group
was significantly increased compared with the sham group. However,
the different doses of CZ2HF markedly attenuated the number of
apoptotic cells (Fig. 7B).
CZ2HF suppresses the increase in Bax and
caspase-3 and the decrease in Bcl-2 expression levels
To further investigate the possible effects of CZ2HF
on apoptosis-associated proteins in Aβ25-35-induced
hippocampal neuronal apoptosis, the Bax and Bcl-2 protein levels,
and active-caspase-3 level were determined by western blotting
(Fig. 8). The present study
determined that CZ2HF (100, 200 and 400 mg/kg) downregulated Bax
expression and upregulated Bcl-2 expression; therefore, the ratio
of Bax/Bcl-2 was reduced, which reversed the
Aβ25-35-induced increase in the Bax/Bcl-2 ratio
(F6,14=2.955, P<0.05; F6,14=3.063,
P<0.05). Additionally, CZ2HF (200, 400 mg/kg) also reduced the
level of active-caspase-3 and limited the decrease of pro-caspase-3
compared with the model group (F6,14=7.867; P<0.01).
These findings suggested that CZ2HF may reduce
Aβ25-35-induced hippocampal neuronal apoptosis by
regulating apoptosis-associated proteins (Fig. 8).
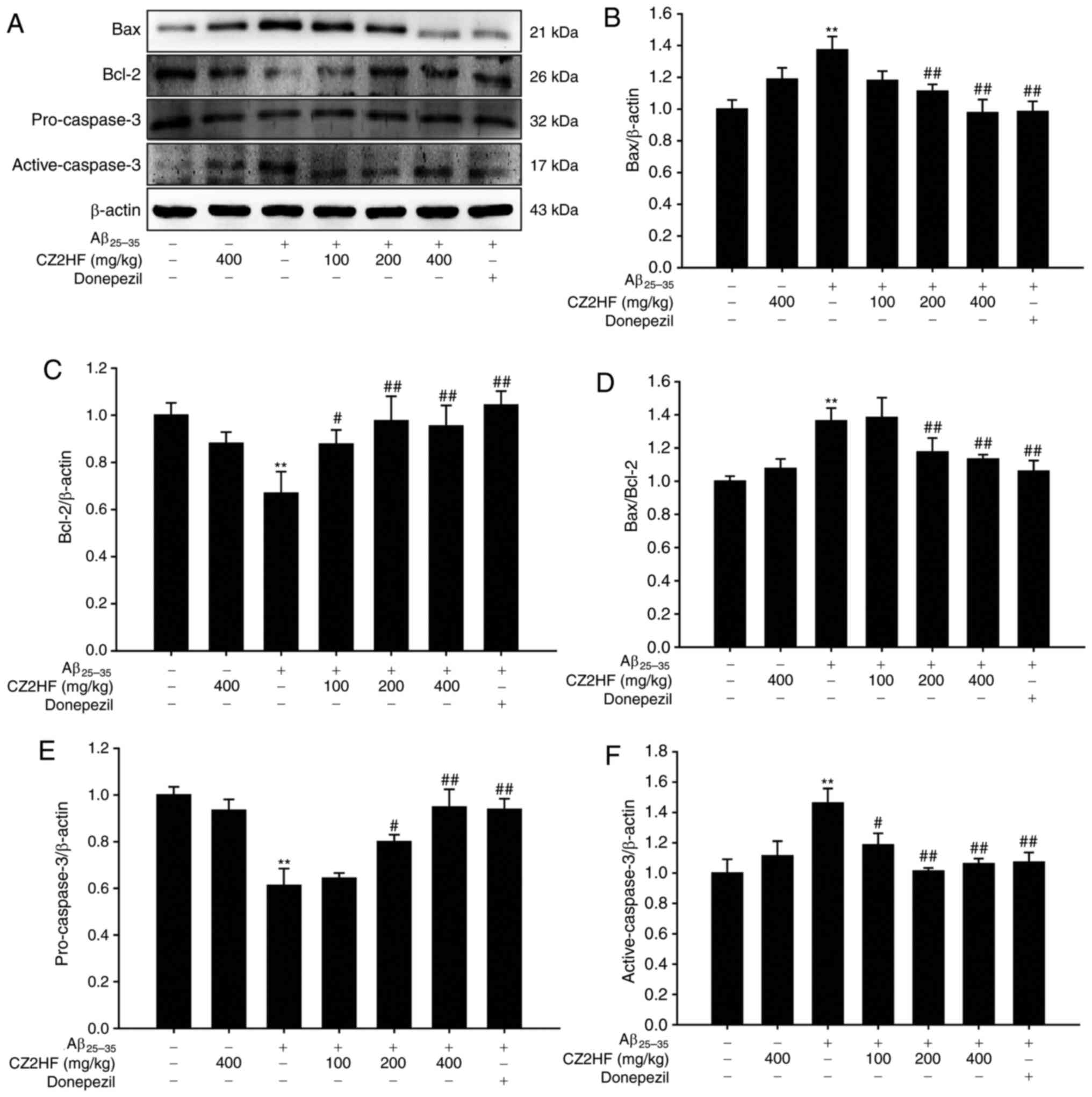 | Figure 8Effect of CZ2HF on the protein
expression of Bax, Bcl-2 and caspase-3 induced by
Aβ25-35 in rats. (A) Representative western blotting
images of Bax, Bcl-2, pro-caspase-3 and active-caspase-3.
Quantification of (B) Bax, (C) Bcl-2, (D) Bax/Bcl-2 ratio, (E)
pro-caspase-3 and (F) active-caspase-3. **P<0.01 vs.
sham; #P<0.05, ##P<0.01 vs.
Aβ25-35 (n=3). CZ2HF, cu-zhi-2-hao-fang;
Aβ25-35, β-amyloid 25-35; Bax, Bcl-2-associated X,
apoptosis regulator; Bcl-2, B cell leukemia/lymphoma 2. |
Discussion
The findings in the current study suggested that
CZ2HF may be a promising agent for the treatment of AD. CZ2HF
significantly attenuated Aβ25-35-induced cognitive
impairments and inhibited neuronal damage and deletions in rats.
Additionally, CZ2HF reduced the protein expression of TNF-α, IL-1β,
COX-2, the Bax/Bcl-2 ratio, and reduced the levels of
Aβ1-42 and active-caspase-3. Furthermore, IκB-α
degradation and p-NF-κB p65 activation was repressed by CZ2HF.
Accumulating evidence demonstrated that
Aβ25-35 is the key fragment of full-length
Aβ1-42 and the acute injection of Aβ25-35
into rat cerebral ventricle may lead to neurotoxic effects similar
to those produced by the Aβ1-40; however, the presence
of the Aβ25-35 fragment in the AD brains remains to be
determined (21,22). Previous studies indicated that
bilateral hippocampal injection of Aβ25-35 may induce an
AD learning and memory impairment model in rats, which has been
widely used in AD research (23,24). Therefore,
Aβ25-35-induced AD rat model was used to investigate the
effects and mechanism of CZ2HF on learning and memory impairment in
the current study using methodology described in our previous study
(14). Additionally, a MWM test
was performed to identify the primary effect of CZ2HF on
Aβ25-35-induced learning and memory function injury in
the rats. As Aβ25-35-induced learning and memory
function exhibits self-limitation, Contextual and Cued Fear
Conditioning Test, which may confirm the association between
hippocampal dependence and learning, and memory function will be
performed in future studies. In addition, as the present study was
preliminary in order to determine the effect of CZ2HF on
Aβ25-35-induced AD; therefore, 3 or 4 rats were used
from each group in the present study. Therefore, the number of rats
used for each molecular or histological experiment was 3 or 4. The
current findings revealed that CZ2HF significantly ameliorated
learning and memory dysfunction in AD rats using the MWM test.
Furthermore, CZ2HF inhibited the
Aβ25-35-induced reduction of the number of neurons,
which was observed by the H&E and Nissl staining. This
indicated that CZ2HF had beneficial effects on learning and memory
impairment, attenuation of hippocampal neuronal damage or loss.
Nevertheless, the underlying mechanism of CZ2HF must be further
elucidated.
It has been previously established that Aβ is
derived from the ordered hydrolysis of APP and Aβ fragments of
39-43 amino acids were formed in this process, including
Aβ1-40 and Aβ1-42 (25). The present study showed that
Aβ1-42 with highest toxicity were significantly
increased in Aβ25-35-induced learning and memory
impairment in the rat brains, which was consistent with the report
that Aβ25-35-induced learning and memory impairment was
accompanied with a greater Aβ1-42 protein level
(26). Previous studies indicated
that the inflammatory factors, including COX-2, IL-1 and TNF-α may
be agglutinated by upregulating Aβ1-42 in the central
nervous system and subsequently lead to a learning and memory
disorder (27). The findings in
the present study demonstrated that Aβ1-42 and COX-2,
IL-1β and TNF-α levels were increased in Aβ25-35-induced
AD rats, which was consistent with the fact that the inflammatory
cytokines increase the activity of β-secretase and the content of
APP, leading to increased Aβ1-42 levels, which creates a
positive feedback effect aggravates cognitive dysfunction (28). However, CZ2HF and donepezil
significantly downregulated TNF-α, IL-1β and COX-2 protein
expression, indicating that CZ2HF reduced the decrease in
Aβ25-35-induced learning and memory impairment both
through reducing Aβ1-42 level and the inflammatory
factors, such as COX-2, IL-1β and TNF-α. Additionally, NF-κB, a
vital nuclear transcription factor, is located in the cytoplasm and
binds to IκB (29). When the
cells are stimulated, IκB is phosphorylated and degraded, which
activates NF-κB p65, and upregulates inflammatory factors during
the inflammatory process. Additionally, NF-κB is also identified to
be upstream of the inflammatory factors (30). The present findings revealed that
degradation of IκB-α and the subsequent activation of NF-κB p65
were increased by the Aβ25-35 injection treatment.
However, CZ2HF significantly reduced the degradation of IκB-α and
inhibited NF-κB p65 phosphorylation in Aβ25-35-induced
AD rats. Therefore, it is possible that CZ2HF ameliorated the
learning and memory impairment, at least partly, through regulation
of the NF-κB signaling pathway.
Previous studies have indicated that the NF-κB
pathway may induce an inflammatory response to release inflammatory
factors and lead to neuronal apoptosis, which also contributes to
the development and progression of AD (31,32). Bcl-2 family proteins such as the
anti-apoptotic protein Bcl-2 and the pro-apoptotic protein Bax have
a key role in the process of neuronal apoptosis. Additionally,
caspase-3 is the key terminal cleavage enzyme during apoptosis and
also executes apoptosis, thereby leading to neuronal cell death
(33). The present findings
revealed that Bcl-2 was reduced, whereas Bax and caspase-3 were
increased in Aβ25-35-induced AD rats, which was
consistent with a previous report which stated that
Aβ25-35 may increase the ratio of Bax/Bcl-2 and activate
caspase-3, inducing neuronal cell apoptosis (34). However, CZ2HF reversed the
aforementioned effects, which confirmed that the beneficial effects
of CZ2HF on learning and memory impairment may be associated with
inhibition of neuronal cell apoptosis. Additionally, the present
findings also indicated that inflammatory response and neuronal
apoptosis have an imperative role in the progression of AD and
there is a connection between Aβ deposition, neuroinflammation and
apoptosis. However, since aqueous extract of TCM contained a large
number of polysaccharides, CZ2HF did not exhibit a dose-dependent
effect for the treatment of Aβ25-35-induced symptoms in
AD-like rats. It is of note that on the protein level, CZ2HF
exerted beneficial effects in a dose-dependent manner, which may be
associated with a potential indirect effect; therefore, compared
with other doses, CZ2HF at a dose of 400 mg/kg promoted the
apoptosis. The in-depth mechanism of CZ2HF on learning and memory
impairment requires further investigation. Additionally,
considering that the components of CZ2HF were complex, the
mechanism which allows CZ2HF or its exact components to pass though
the blood-brain barrier should be investigated in future
studies.
In conclusion, the present study demonstrated that
CZ2HF ameliorates Aβ25-35-induced learning and memory
impairment in rats and inhibits the damage of hippocampal neurons.
To the best of our knowledge the present study was the first to
determine the underlying mechanisms, which may be attributed, at
least in part, to repressing the inflammatory response and
apoptosis (Fig. 9). The present
study provided a scientific foundation and information for the use
of CZ2HF for the treatment of AD.
Acknowledgments
Not applicable.
Abbreviations:
|
AD
|
Alzheimer’s disease
|
|
CZ2HF
|
cu-zhi-2-hao-fang
|
|
Aβ25-35
|
β-amyloid 25-35
|
|
MWM
|
Morris water maze
|
|
H&E
|
hematoxylin and eosin
|
|
TUNEL
|
terminal
deoxynuc-leotidyltransferase-mediated dUTP nick-end labeling
|
|
COX-2
|
cyclooxygenase-2
|
|
TNF-α
|
tumor necrosis factor-α
|
|
IL-1β
|
interleukin-1β
|
|
NF-κB p65
|
nuclear factor-κB p65
|
Funding
The current study was supported by the Natural
Science Foundation of China (grant no. 81560585), Program for
Excellent Young Talents of Zunyi Medical Uiverstity (grant no.
15zy-002), Science and Technology Innovation Talent Team of Guizhou
Province (grant no. 20154023), The ‘Hundred’ Level of High-Level
Innovative Talents in Guizhou Province (grant no. QKHRCPT 20165684)
and Program for Changjiang Scholars and Innovative Research Team in
University, China (grant no. IRT17R113).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors’ contributions
QG and JS designed the experimental approaches. LZ
performed all the other studies described herein, except the
western blotting conducted by YD and JG. LZ and JG wrote the
manuscript with the help from QG. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Zunyi Medical University (Zunyi, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ding C, Li F, Long Y and Zheng J:
Chloroquine attenuates lipopolysaccharide-induced inflammatory
responses through upregulation of USP25. Can J Physiol Pharmacol.
95:481–491. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Huang L, Luo Y, Pu Z, Kong X, Fu X, Xing
H, Wei S, Chen W and Tang H: Oxoisoaporphine alkaloid derivative
8-1 reduces Aβ1-42 secretion and toxicity in human cell
and Caenorhabditis elegans models of Alzheimer’s disease. Neurochem
Int. 108:157–168. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kreutzer AG, Yoo S, Spencer RK and Nowick
JS: Stabilization, assembly, and toxicity of trimers derived from
Aβ. J Am Chem Soc. 139:966–975. 2017. View Article : Google Scholar
|
|
4
|
Liu C, Cao L, Yang S, Xu L, Liu P, Wang F
and Xu D: Subretinal injection of amyloid-β peptide accelerates RPE
cell senescence and retinal degeneration. Int J Mol Med.
35:169–176. 2015. View Article : Google Scholar
|
|
5
|
Yin K, Jin J, Zhu X, Yu L, Wang S, Qian L,
Han L and Xu Y: CART modulates beta-amyloid metabolism-associated
enzymes and attenuates memory deficits in APP/PS1 mice. Neurol Res.
39:885–894. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Puzzo D, Gulisano W, Arancio O and Palmeri
A: The keystone of Alzheimer pathogenesis might be sought in Aβ
physiology. Neuroscience. 307:26–36. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Park SY, Kim MJ, Kim YJ, Lee YH, Bae D,
Kim S, Na Y and Yoon HG: Selective PCAF inhibitor ameliorates
cognitive and behavioral deficits by suppressing NF-κB-mediated
neuroin-flammation induced by Aβ in a model of Alzheimer’s disease.
Int J Mol Med. 35:1109–1118. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ling IF, Golde TE, Galasko DR and Koo EH:
Modulation of Aβ42 in vivo by γ-secretase modulator in primates and
humans. Alzheimers Res Ther. 7:552015. View Article : Google Scholar
|
|
9
|
Kang EB, Kwon IS, Koo JH, Kim EJ, Kim CH,
Lee J, Yang CH, Lee YI, Cho IH and Cho JY: Treadmill exercise
represses neuronal cell death and inflammation during Aβ-induced ER
stress by regulating unfolded protein response in aged presenilin 2
mutant mice. Apoptosis. 18:1332–1347. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Um YH, Kim TW, Jeong JH, Seo HJ, Han JH,
Hong SC, Lee CU and Lim HK: Prediction of treatment response to
donepezil using automated hippocampal subfields volumes
segmentation in patients with mild Alzheimer’s disease. Psychiatry
Investig. 14:698–702. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Darreh-Shori T, Hosseini SM and Nordberg
A: Pharmacodynamics of cholinesterase inhibitors suggests add-on
therapy with a low-dose carbamylating inhibitor in patients on
long-term treatment with rapidly reversible inhibitors. J
Alzheimers Dis. 39:423–440. 2014. View Article : Google Scholar
|
|
12
|
Deardorff WJ and Grossberg GT:
Pharmacotherapeutic strategies in the treatment of severe
Alzheimer’s disease. Expert Opin Pharmacother. 17:1789–1800. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pérez DI, Martínez A, Gil C and Campillo
NE: From bitopic inhibitors to multitarget drugs for the future
treatment of Alzheimer’s disease. Curr Med Chem. 22:3789–3806.
2015. View Article : Google Scholar
|
|
14
|
Deng Y, Long L, Wang K, Zhou J, Zeng L, He
L and Gong Q: Icariside II, a broad-spectrum anti-cancer agent,
reverses beta-amyloid-induced cognitive impairment through reducing
inflammation and apoptosis in rats. Front Pharmacol. 8:392017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gao J, Deng Y, Yin C, Liu Y, Zhang W, Shi
J and Gong Q: Icariside II, a novel phosphodiesterase 5 inhibitor,
protects against H2O2-induced PC12 cells
death by inhibiting mitochondria-mediated autophagy. J Cell Mol
Med. 21:375–386. 2017. View Article : Google Scholar
|
|
16
|
Li F, Dong HX, Gong QH, Wu Q, Jin F and
Shi JS: Icariin decreases both APP and Aβ levels and increases
neurogenesis in the brain of Tg2576 mice. Neuroscience. 304:29–35.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jia KK, Zheng YJ, Zhang YX, Liu JH, Jiao
RQ, Pan Y and Kong LD: Banxiahoupu decoction restores glucose
intolerance in CUMS rats through improvement of insulin signaling
and suppression of NLRP3 inflammasome activation in liver and
brain. J Ethnopharmacol. 209:219–229. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gong QH, Pan LL, Liu XH, Wang Q, Huang H
and Zhu YZ: S-propargyl-cysteine (ZYZ-802), a sulphur-containing
amino acid, attenuates beta-amyloid-induced cognitive deficits and
pro-inflammatory response: Involvement of ERK1/2 and NF-κB pathway
in rats. Amino Acids. 40:601–610. 2011. View Article : Google Scholar
|
|
19
|
Yan L, Deng Y, Gao J, Liu Y, Li F, Shi J
and Gong Q: Icariside II effectively reduces spatial learning and
memory impairments in Alzheimer’s disease model mice targeting
beta-amyloid production. Front Pharmacol. 8:1062017. View Article : Google Scholar
|
|
20
|
Liu Y, Deng Y, Liu H, Yin C, Li X and Gong
Q: Hydrogen sulfide ameliorates learning memory impairment in
APP/PS1 transgenic mice: A novel mechanism mediated by the
activation of Nrf2. Pharmacol Biochem Behav. 150-151:207–216. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li XH, Deng YY, Li F, Shi JS and Gong QH:
Neuroprotective effects of sodium hydrosulfide against
β-amyloid-induced neurotoxicity. Int J Mol Med. 38:1152–1160. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tikhonova LA, Kaminsky YG, Reddy VP, Li Y,
Solomadin IN, Kosenko EA and Aliev G: Impact of amyloid
β25-35 on membrane stability, energy metabolism, and
antioxidant enzymes in erythrocytes. Am J Alzheimers Dis Other
Demen. 29:685–695. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wu J, Yang H, Zhao Q, Zhang X and Lou Y:
Ginsenoside Rg1 exerts a protective effect against
Aβ25-35-induced toxicity in primary cultured rat
cortical neurons through the NF-κB/NO pathway. Int J Mol Med.
37:781–788. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schimidt HL, Garcia A, Martins A,
Mello-Carpes PB and Carpes FP: Green tea supplementation produces
better neuroprotective effects than red and black tea in
Alzheimer-like rat model. Food Res Int. 100:442–448. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Colvin MT, Silvers R, Ni QZ, Can TV,
Sergeyev I, Rosay M, Donovan KJ, Michael B, Wall J, Linse S and
Griffin RG: Atomic resolution structure of monomorphic Aβ42 amyloid
fibrils. J Am Chem Soc. 138:9663–9674. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Patricio-Martínez A, Mendieta L, Martínez
I, Aguilera J and Limón ID: The recombinant C-terminal fragment of
tetanus toxin protects against cholinotoxicity by intraseptal
injection of beta-amyloid peptide (25-35) in rats. Neuroscience.
315:18–30. 2016. View Article : Google Scholar
|
|
27
|
Yu X, Wang LN, Du QM, Ma L, Chen L, You R,
Liu L, Ling JJ, Yang ZL and Ji H: Akebia Saponin D attenuates
amyloid β-induced cognitive deficits and inflammatory response in
rats: Involvement of Akt/NF-κB pathway. Behav Brain Res.
235:200–209. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Carrero I, Gonzalo MR, Martin B,
Sanz-Anquela JM, Arévalo-Serrano J and Gonzalo-Ruiz A: Oligomers of
β-amyloid protein (Aβ1-42) induce the activation of
cyclooxygenase-2 in astrocytes via an interaction with
interleukin-1β, tumour necrosis factor-α, and a nuclear factor κ-B
mechanism in the rat brain. Exp Neurol. 236:215–227. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu W, Yang JJ, Yang HM, Huang MM, Fang QJ,
Shi G, Mao ZM, Han WB, Shen SM and Wan YG: Multi-glycoside of
Tripterygium wilfordii Hook. f. attenuates glomerulosclerosis in a
rat model of diabetic nephropathy by exerting
anti-microinflammatory effects without affecting hyperglycemia. Int
J Mol Med. 40:721–730. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Choi JH, Chung KS, Jin BR, Cheon SY,
Nugroho A, Roh SS and An HJ: Anti-inflammatory effects of an
ethanol extract of Aster glehni via inhibition of NF-κB activation
in mice with DSS-induced colitis. Food Funct. 8:2611–2620. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Cha HY, Ahn SH, Cheon JH, Park SY and Kim
K: Hataedock treatment has preventive therapeutic effects for
atopic dermatitis through skin barrier protection in
Dermatophagoides farinae-in duced NC/Nga mice. J Ethnopharmacol.
206:327–336. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Orban Z, Mitsiades N, Burke TR Jr, Tsokos
M and Chrousos GP: Caffeic acid phenethyl ester induces leukocyte
apoptosis, modulates nuclear factor-kappa B and suppresses acute
inflammation. Neuroimmunomodulation. 7:99–105. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang H, Xu YS, Wang ML, Cheng C, Bian R,
Yuan H, Wang Y, Guo T, Zhu LL and Zhou H: Protective effect of
naringin against the LPS-induced apoptosis of PC12 cells:
Implications for the treatment of neurodegenerative disorders. Int
J Mol Med. 39:819–830. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang Q, Li J, Liu C, Song C, Li P, Yin F,
Xiao Y, Li J, Jiang W, Zong A, et al: Protective effects of low
molecular weight chon-droitin sulfate on amyloid beta (Aβ)-induced
damage in vitro and in vivo. Neuroscience. 305:169–182. 2015.
View Article : Google Scholar : PubMed/NCBI
|