Introduction
Vascular dementia (VD) is the second most common
cognitive disorder, following Alzheimer's disease (AD). VD is
caused by a series of cardiac-cerebral vascular conditions that are
characterized by loss of cognitive functions (1-3).
Previous studies have demonstrated that the incidence of chronic
cerebral hypoperfusion increases with advancing age and dementia
(4,5). As VD is a progressive
neurodegenerative disease, treatment strategies should focus on the
early stages of the pathological process. It was previously
demonstrated that ischemia-induced neuronal apoptosis and synaptic
reduction are major causes of early VD (6,7).
Accumulating evidence has shown that synaptic changes are closely
associated with a number of dementia-related diseases, and are not
specific to AD (8,9). Permanent bilateral occlusion of the
common carotid artery (2-VO) is an effective VD model in rats. A
previous study reported that rats exhibited significant cognitive
deficits after 4 weeks of chronic ischemia, suggesting that the
early synaptic pathology of VD is associated with cognitive
impairment (10). The aim of the
present study was to investigate the effects of microRNA
(miRNA/miR)-134-5p on cognitive deficits and elucidate the
potential underlying mechanism in a chronic global cerebral
ischemia rat model induced by 2-VO.
Synapses are critical for early cognition and are
strongly associated with cognitive deficits, including amyloid and
tau pathology (11,12). Synapse-associated proteins act as
synaptic morphogens, initiating functional changes; synapsin I
(Syn1) and synaptosome-associated protein 25 (Snap25) are important
markers involved in synaptic transmission through different
mechanisms (13-15). Syn1 is implicated in the
modulation of neurotransmitter release and synaptogenesis, and is
involved in the transmission and regulation of synaptic
information. Syn1 has a potential role in several neurological
diseases, such as epilepsy and autism spectrum disorders (16,17). Previous studies demonstrated that
the loss of synaptic proteins is associated with VD-associated
cognitive impairment (18,19).
The transcription factor forkhead box P2 (Foxp2) is
the only gene known to be involved in the Mendelian form of human
speech and language impairment to date, and is primarily involved
in influencing complex vocal cord movement and vocal learning
behavior (20). Mutations in the
human Foxp2 gene cause severe speech and language disorders
(21,22). In different vertebrate species,
Foxp2 is highly expressed in the central nervous system during
development and adulthood (23).
As a transcription factor, Foxp2 regulates a number of downstream
target genes and plays important roles in the development of the
central nervous system, including synaptic plasticity,
neurotransmission and neurite outgrowth (24). Foxp2 plays important roles in the
gene networks linked to nervous system development and function.
Moreover, reducing the functional dose of mouse Foxp2 led to
impaired synaptic plasticity in the cortical striatum and impaired
motor learning (25).
Furthermore, previous studies have indicated that various genetic
and environmental factors may contribute to speech and language
development, and related neural developmental disorders, through
the miRNA/Foxp2 regulatory network (26). However, the role of epigenetic
regulation of Foxp2 in VD remains elusive.
Several miRNAs have been identified as important
therapeutic targets involved in synaptic plasticity and
neuro-development (27). The
targeting of Ca2+/calmodulin kinase II (CaMKII) by
miR-219 negatively regulates arsenic-induced hippocampus structural
damage and the causes impairment of memory and learning (28). However, the overexpression of
miR-132 can increase the miniature excitatory postsynaptic current
amplitude and dendritic protrusion width (29,30). miR-12 has been reported to
regulate specific synaptic plasticity during long-term olfactory
habituation in Drosophila (31). miR-134 was previously reported to
be widely expressed in primary cultured neurons and dendrites.
miR-134 has also been reported to regulate the development of the
dendritic spine by targeting Lim domain kinase 1 (LimK1), which is
inhibited by brain-derived neurotrophic factor (BDNF) (32). In addition, miR-134 has been shown
to exert an adverse regulatory effect on the volume of neuronal
dendritic spines (33). A
previous study has demonstrated that miR-134 is closely associated
with the expression of synaptic proteins in the brain (34).
In the present study, 4 weeks of cerebral ischemia
was induced using 2-VO in rats in order to simulate the early stage
of the pathological process in VD. This model allows investigation
of the role of miRNAs in cognitive impairment.
Materials and methods
Animals and VD model
In total, 18 male Sprague-Dawley rats (mean weight,
180-220 g; age, 6-7 weeks) were supplied by the Laboratory Animal
Center of Guangzhou University of Chinese Medicine. The rats were
housed in an environment with a constant room humidity (50-70%), a
temperature of 25±1°C and a 12-h light/dark cycle (lights on at
7:00 am). Water and food were available ad libitum. The
experiments were approved by the Care and Use of Experimental
Animals Committee of Guangzhou University of Chinese Medicine and
performed according to the National Institute of Health Guide for
the Care and Use of Laboratory Animals. Each cage housed 5 rats to
prevent any effects of social isolation.
VD surgery
The VD model was induced in rats by global cerebral
ischemia using 2-VO. The rats used in the experiments were
anesthetized by intraperitoneal injection (0.15 ml/100 g body
weight) of 100 mg/kg ketamine and 10 mg/kg xylazine. The surgery
separated the bilateral common carotid arteries, which were
permanently ligated with small-diameter nylon sutures. Rats in the
control group underwent the same procedure, but without ligation of
the common carotid arteries. The body temperature of the rats was
monitored throughout the procedure and maintained at 37°C. After
surgery, the rats were placed back in their cages and normal
feeding was resumed.
Intracerebroventricular (ICV) injection
of the antagomir
To determine the effect of miR-134-5p on cognitive
function and synaptic changes in VD rats, the ICV injection method
was used to administer the miR-134-5p antagomir
(5′-CCCCUCUGGUCAACCAGU CAC A-3′; Guangzhou RiboBio Co., Ltd.). The
animals were randomly divided into three groups (n=6/group): The
control group, the VD model group and the miR-134-5p antagomir
group (2-VO + miR-134-5p antagomir). Each group of rats were
continuously injected via ICV for 3 days. The miR-134-5p antagomir
was dissolved in sterile double-distilled water (ddH2O)
before use and a volume of 5 μl was injected (200 pmol/rat)
into both sides of the lateral ventricle. Rats in the control and
VD model groups were injected with the same volume of sterile
ddH2O. For the injection, the rats were anesthetized
using an intraperitoneal injection (0.15 ml/100 g body weight) of
100 mg/kg ketamine and 10 mg/kg xylazine and placed on a
stereotactic device with their backs on the panel. The antagomir
was immediately injected into the lateral ventricle. According to
the rat brain atlas, two small holes were drilled carefully in the
skull bilaterally using a surgical drill. The stereotaxic
coordinates of the ICV injection were as follows: -0.8 mm anterior,
1.5 mm lateral and -4.5 mm depth.
Morris water maze (MWM) test
To evaluate spatial memory and learning, MWM tests
were conducted 24 h after the last ICV injection. The apparatus
consisted of a black circular pool (diameter, 180 cm) filled with
water to a depth of 40 cm. The maze was divided into four
quadrants. The temperature of the water was 23±2°C. A submerged
circular escape platform (diameter, 20 cm), 1.5 cm below the water
surface, was placed in a random quadrant 25-30 cm from the edge of
the pool. The platform remained in the same position throughout the
experiment. On days 1-5, the rats swam freely for 90 sec to find
the platform. The rats were given 4 trials/day to train their
spatial learning ability (n=6/group). If a rat failed to reach the
platform, it was gently guided to the platform and placed on it for
20 sec. Each rat required a 15-min rest before the next trial. A
spatial probe test was conducted on the 6th day, during which the
hidden platform was removed. Rats were allowed to swim freely for
90 sec to determine their memory retention. After each trial, the
rats were dried. The cleanliness of the water was maintained
throughout the trails.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) analysis
Rats from each group were randomly selected and
anesthetized using an intraperitoneal injection (0.15 ml/100 g body
weight) of 100 mg/kg ketamine and 10 mg/kg xylazine. The rats were
then sacrificed using CO2 asphyxiation with a fill rate
of 20% of the chamber volume/min with CO2. Death was
confirmed using a combination of criteria, including lack of pulse,
breathing, corneal reflex, response to a firm toe pinch and graying
of the mucous membranes. The brains were quickly removed and stored
at -80°C. Total RNA was extracted using TRIzol® (Thermo
Fisher Scientific, Inc.) from cultured PC12 cells or tissue on ice
for 20 min, and then isolated using chloroform and isopropyl
alcohol. The concentration and purity of the extracted RNA was
determined using a UV spectrophotometer. RT was carried out using
the Prime Script™ RT reagent kit (Takara Biotechnology, Co., Ltd.),
according to the manufacturer's protocol. The thermocycling
conditions were 37°C for 15 min and 85°C for 4 sec. A specific RT
primer was used for miR-134-5p
(5′-GTCGTATCCAGTGCAGGGTCCGAGTATTCGCACTGGATACGA-3′) and random
primers were used for RT of U6. The qPCR was performed using a SYBR
Premix EX TaqⅡ kit (Takara Biotechnology, Co., Ltd.). Relative
quantification of gene expression was performed using CFX96™
Real-Time PCR Detection System (Bio-Rad Laboratories Inc.). The
thermocy-cling conditions were as follows: 95°C for 30 sec,
followed by 40 cycles at 95°C for 5 sec, 55°C for 30 sec and 72°C
for 1 min, with a final extension at 55°C for 5 sec. The following
primers were synthesized by Sangon Biotech Co., Ltd. and used for
qPCR of rno-miR-134-5p: Forward, 5′-CGCGTGTGACTGGTTGACCA-3′ and
reverse, 5′-AGTGCAGGGTCCGAGGTATT-3′. The U6 qPCR primers were
purchased from Sangon Biotech Co., Ltd. (cat. no. MQP-0101).
Differences in gene expression were analyzed using the
2−ΔΔCq method (35).
U6 was used as an internal control for the detection of miRNA. Each
experiment was repeated three times.
Bioinformatics prediction of the target
genes of miR-134-5p
The target genes of Rattus norvegicus
(rno)-miR-134-5p were predicted using bioinformatics tools,
including TargetScan (version 7.2; www.targetscan.org/mmu_71), miRBase (version 22.1;
www.mirbase.org) and miRWalk (version Nov/2018
Release; http://mirwalk.umm.uni-heidelberg.de). The predicted
gene targets were screened using Venn diagrams and the DAVID
Bioinformatics Resources 6.8 (david.ncifcrf.gov) for gene function analysis. The
target genes of rno-miR-134-5p were verified using western blotting
and dual-luciferase reporter gene analysis.
Histopathological observation using
hematoxylin and eosin (H&E) staining
After 4 weeks of 2-VO and following the MWM test,
rats in each group were randomly selected and anesthetized. Rats
were immediately perfused transcardially with cold normal saline
followed by ~100 ml of cold 4% para-formaldehyde to prefix the
brain tissue. The brains were washed with cold saline and fixed in
4% paraformaldehyde at 4̊C overnight. Paraffin sections of 4
μm were used for H&E, TUNEL and immunohistochemical
staining. Sections from the cortex and hippocampus were stained
using an H&E staining kit (Beijing Solarbio Science &
Technology). The sections were visualized using a light microscope
(Leica dMI400; Leica Microsystems GmbH) and photographed. Each
experiment was repeated three times.
TUNEL staining
TUNEL staining was used to identify apop-totic cells
in the cortex, following the manufacturer's protocol (one-step
TUNEL apoptosis assay kit; Beyotime Institute of Biotechnology).
Nuclei were stained with DAPI (1:100; Beijing Solarbio Science
& Technology) for 10 min at 37°C in a humidified atmosphere in
the dark. Images of apoptotic cells were captured using a confocal
laser scanning microscope (LSM 800; Carl Zeiss AG) at x200
magnification. Each experiment was repeated three times.
Immunohistochemical staining
Immunohistochemical staining was performed using a
HistostainTM-Plus kit (Beijing Biosynthesis Biotech Co.,
Ltd.) according to the manufacturer's instructions. The dehydrated
and transparent tissue block was placed in dissolved paraffin, and
after the tissue block was completely immersed in paraffin, it was
embedded and sliced. Paraffin sections were incubated with
anti-Syn1 primary antibody (1:100; cat. no. ab8; Abcam) at 4°C
overnight, and incubated with a biotinylated secondary antibody for
20 min at 37°C. The staining was examined and images were captured
using a light microscope (Leica dMI400; Leica Microsystems GmbH) at
x400 magnification. In total, five fields of view were randomly
selected on each section to measure the expression level of Syn1.
Each experiment was repeated three times.
Western blot analysis
Proteins were extracted using RIPA lysis buffer
(Thermo Fisher Scientific, Inc.) from cultured PC12 cells or tissue
(pulverized frozen fresh cortex) on ice for 20 min. The
supernatants were obtained by centrifugation at 12,000 x g at 4°C
for 20 min. Protein concentrations were quantified using a
bicinchoninic acid protein assay (Fdbio Science). Equal amounts of
protein (20-50 μg/lane) were separated using 10% SDS-PAGE
gels and transferred onto nitrocellulose membranes after
electrophoresis. After blocking with 5% skimmed milk in TBS
containing 0.01% Tween-20 (TBST) at room temperature for 2 h, the
blots were incubated with anti-Foxp2 (1:1,000; cat. no. ab16046;
Abcam), anti-Snap25 (1:1,000; cat no. A0986; ABclonal Biotech Co.,
Ltd.), anti-Syn1 (1:1,000; cat. no. ab8; Abcam) and anti-β-actin
(1:5,000; cat. no. ab8227; Abcam) primary antibodies at 4°C
overnight. Following washing with TBST, the blots were incubated
with a secondary horseradish peroxidase-conjugated goat anti-rabbit
antibody (1:1,000; cat. no HAF008, R&D Systems, Inc.) for 1 h
at room temperature and washed with TBST. Protein bands were
visualized using an enhanced chemiluminescence kit (Beyotime
Institute of Biotechnology), according to the manufacturer's
protocol. Protein bands were analyzed using ImageJ software
(version 1.4; National Institutes of Health). The expression of the
target protein was calculated by comparing the gray value of each
group with the corresponding internal reference gene. Each
experiment was repeated three times.
PC12 cell culture
PC12 rat pheochromocytoma cells are frequently used
as a model in neuronal research (36). PC12 cells were purchased from
Procell Life Science & Technology Co., Ltd. and were cultured
at 37°C in a 5% CO2 incubator in RPMI 1640 medium
(Gibco; Thermo Fisher Scientific, Inc.), 10% FBS (Gibco; Thermo
Fisher Scientific, Inc.) and 1% solution penicillin/streptomycin
(Thermo Fisher Scientific, Inc.). The medium was replaced every 3
days. PC12 cells were plated into 6- or 24-well plates at a density
of 9×105 or 1.5×105 cells/well,
respectively.
Luciferase reporter assay
PC12 cells were seeded in 24-well plates at a
density of 1x105 cells/well and were subsequently
transfected with miR-134-5p mimic, miR-134-5p inhibitor, miR-134-5p
mimic-NC or miR-134-5p inhibitor-NC (Guangzhou RiboBio Co., Ltd.),
and co-transfected with pLUC-Foxp2-wild-type (WT) 3′-untranslated
region (UTR) or pLUC-Foxp2-mutant (MUT) 3′-UTR plasmids (Shenzhen
Huaan Ping Kang Bio Technology Co., Inc.) using
Lipofectamine® 2000 (Thermo Fisher Scientific, Inc.),
according to the manufacturer's protocol. In the pLUC-Foxp2 cloning
vector, miRNA target sites, predicted using TargetScan software
(version 7.2; http://www.targetscan.Org/vert_71/), were inserted
after the Renilla luciferase region. The final concentration
of miR-134-5p mimic and miRNA mimic negative control transfected
into PC12 cells was 50 nM, and the final concentration of
miR-134-5p inhibitor and miRNA inhibitor negative control was 100
nM; 200 ng plasmid was used for transfection. PC12 cells were lysed
and the luciferase activity was quantified using the
Dual-Luciferase® Reporter assay kit (Promega
Corporation), according to the manufacturer's protocol, 48 h after
transfection. Each experiment was repeated three times.
Transfection of PC12 cells with
miR-134-5p mimic, miR-134-5p inhibitor and the respective miRNA
negative controls (miRNA mimic-NC and miRNA inhibitor-NC) or siRNA
(siR)-Foxp2
PC12 cells were seeded into 6-well plates at a
density of 9x105 cells/well 1 day before transfection in
order to reach a confluence of 60-80%. Transfection of miR-134-5p
mimic (5′-UGUGACUGGUUGACCAGAGGGG-3′ and
5′-ACACUGACCAACUGGUCUCCCC-3′), miR-134-5p mimic-NC (cat. No
miR1N0000001-1-5; sequence unavailable; Guangzhou RiboBio Co.,
Ltd.), miR-134-5p inhibitor (5′-CCCCUCUGGUCAACCAGUCACA-3′),
miR-134-5p inhibitor-NC (cat. no miR2N0000001-1-5; sequence
unavailable; Guangzhou RiboBio Co., Ltd.) or siRNA-Foxp2
(siR-Foxp2-001, AGCAGCAACAACTACAAG A; siR-Foxp2-002,
CAAAGCTTCACCGCCAATA; siR-Foxp2-003, CGACATTCAGACAAATACA) was
performed using Lipofectamine® 2000 (Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol. The
final concentration of mimic, mimic-NC and siRNAs was 50 nM, and
the final concentration of inhibitor and inhibitor-NC was 100 nM.
Cells were lysed with RIPA buffer 48 h after transfection for
western blot analysis. Each experiment was repeated three
times.
Chromatin immunoprecipitation
(ChIP)-qPCR
Using ChIP assays, the molecular interaction between
Foxp2 and the Syn1 promoter binding site was investigated. Nerve
growth factor (NGF; 50 ng/ml) was used to induce PC12 cells for 1
week. According to the manufacturer's instructions (Magna
ChIPTM A; EMD Millipore), the culture medium was removed
and 1% formaldehyde was added for crosslinking for 10 min at 37°C;
2 ml of 0.125 M glycine was added to quench unreacted formaldehyde.
The cells were washed using ice-cold PBS twice and then collected
and lysed in SDS Lysis Buffer (EMD Millipore). Chromatin was
extracted from the nuclei. The chromatin was then sheared to
200-1,000 bp fragments by ultrasonication at 4°C for 40 min, and
the effect of ultrasonic crushing was examined by agarose gel
electrophoresis. The Foxp2 antibody (Abcam) was applied to
immunoprecipitated DNA by using magnetic protein G beads. Following
reverse crosslinking, ChIP-DNA complexes were extracted, washed and
eluted using 100 μl ChIP Elution Buffer (EMD Millipore) with
1 μl protease K (EMD Millipore). DNA was purified by Spin
Filter. The ChIP-DNA fragments were directly used in qPCR assays
with specific primers for the promoter region of the Syn1. GAPDH
was used as an internal control for the detection of mRNA. The
primers used were as follows: Syn1-1: Forward,
5′-GGACCCCTAAGTTCCTTCCTCCA-3′ and reverse,
5′-AGACACAAACATTGGCAAAGGTGG-3′; Syn1-2: Forward,
5′-CTCCCAAATCCGCATGGGGT-3′ and reverse,
5′-GTCTCCTCTTGGCTTTGGGGATAGT-3′; Syn1-3: Forward,
5′-CTGAGGCAGTATCAGGGCACAG-3′ and reverse,
5′-CTGCCTTCTCAGCGCAGCC-3′; and GAPDH: Forward,
5′-GTCCATGCCATCACTGCCACT C-3′ and reverse,
5′-CGCCTGCTTCACCACCTTCTTG-3′. ChIP was analyzed by qPCR using the
CFX96™ Real-Time PCR Detection System (Bio-Rad Laboratories, Inc.).
Each experiment was repeated three times.
Statistical analysis
All data are presented as the mean ± standard
deviation. Data were analyzed using Excel 2017 (Microsoft
Corporation) and GraphPad Prism7 (version 7.0; GraphPad Software,
Inc.). Comparisons between two groups were performed using a
t-test. Comparison among multiple groups were analyzed using
one-way ANOVA followed by Tukey's multiple comparisons test for
post hoc analysis. P<0.05 was considered to indicate a
statistically significant difference.
Results
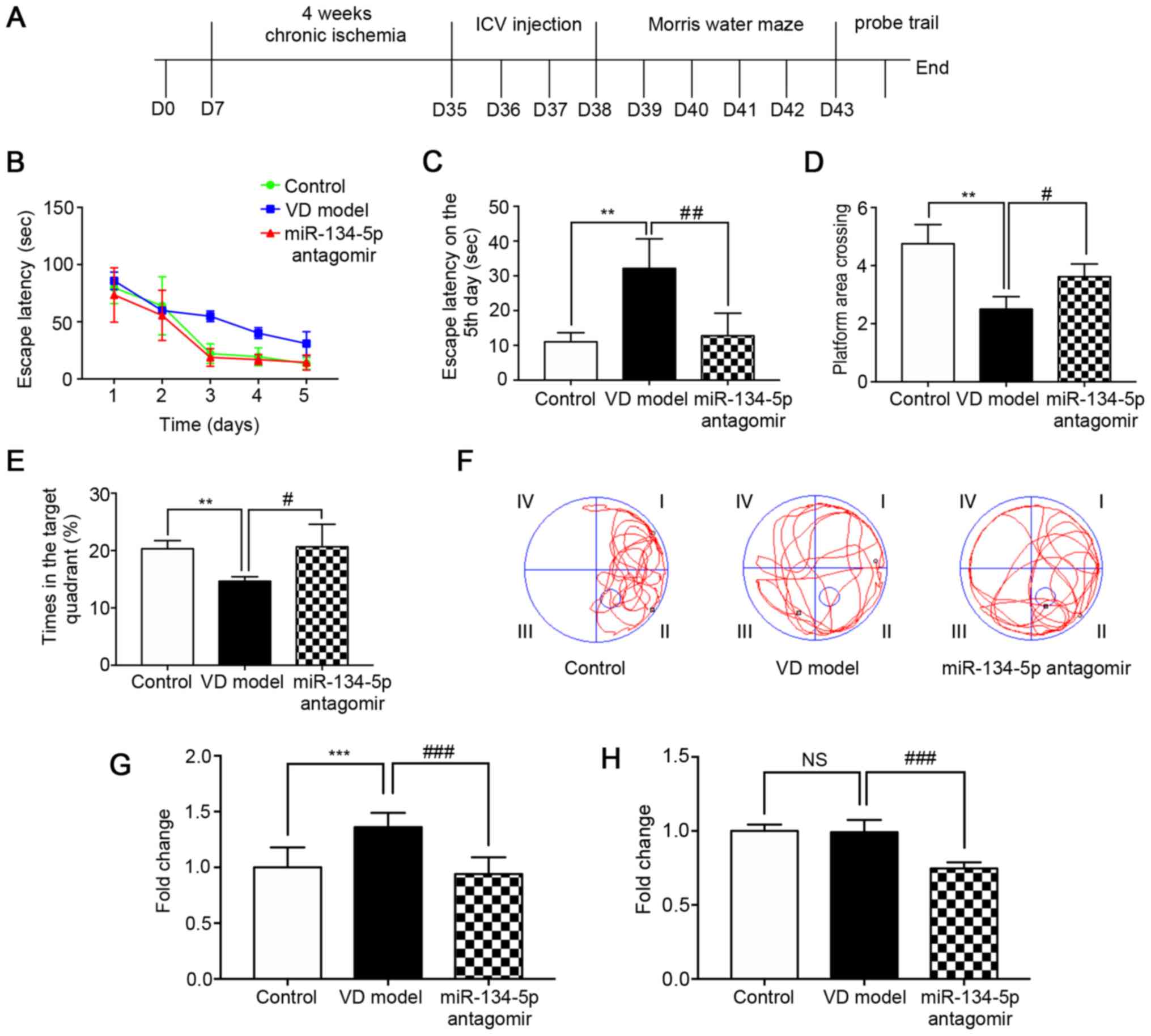
miR-134-5p inhibitor relieves cognitive
dysfunction in VD rats following 4 weeks of chronic ischemia
A rat model of dementia was produced using 2-VO for
4 weeks. In order to verify whether miR-134-5p could affect the
spatial learning and memory ability of VD rats, MWM experiments
were conducted after 3 days of lateral ventricular injection of the
miR-134-5p antagomir (n=6/group). During MWM training, the escape
latency of rats decreased significantly, indicating that the
platform could be found with training. In consecutive trials, the
VD rats were impaired while training to find the submerged escape
platform, as indicated by longer escape latencies compared with the
control group. The escape latency of the miR-134-5p antagomir group
was shorter compared with that of the VD model group; this was also
the case on the 5th day (Fig.
1A-C). In the MWM test, the number of rats crossing over the
platform position, and the percentage of time in the target
quadrant relative to the total escape latency time in the pool, was
significantly decreased in VD model rats and significantly
increased in the miR-134-5p antagomir group compared with the
control group (Fig. 1D and E).
Representative movement traces of the MWM test on the 6th day
revealed that the number of VD model rats crossing the platform was
significantly lower compared with that of rats in the control
group. In addition, the number of miR-134-5p antagomir rats
crossing the platform was significantly higher compared with in the
VD model group (Fig. 1F). Using
the MWM experiment, it was demonstrated that rats exhibited spatial
learning impairment following 4 weeks of chronic ischemia.
Behavioral tests also indicated that inhibiting miR-134-5p could
relieve cognitive dysfunction, as well as enhance learning and
memory ability, in VD rats.
miR-134-5p expression is increased in the
cortex of VD rats
To determine whether miR-134-5p is upregulated in
the brains of VD rats, RT-qPCR analysis was performed (n=3/group).
The results demonstrated that the expression of miR-134-5p in the
cortical tissue of VD model rats was increased significantly
compared with the control group (Fig.
1G). No significant differences were found in the hippocampus
of VD model rats compared with the control group (Fig. 1H). These results revealed that
miR-134-5p is implicated in chronic ischemia-induced VD. These
results also indicated that miR-134-5p is differentially expressed
in the cortex, but not in the hippocampus, of VD rats.
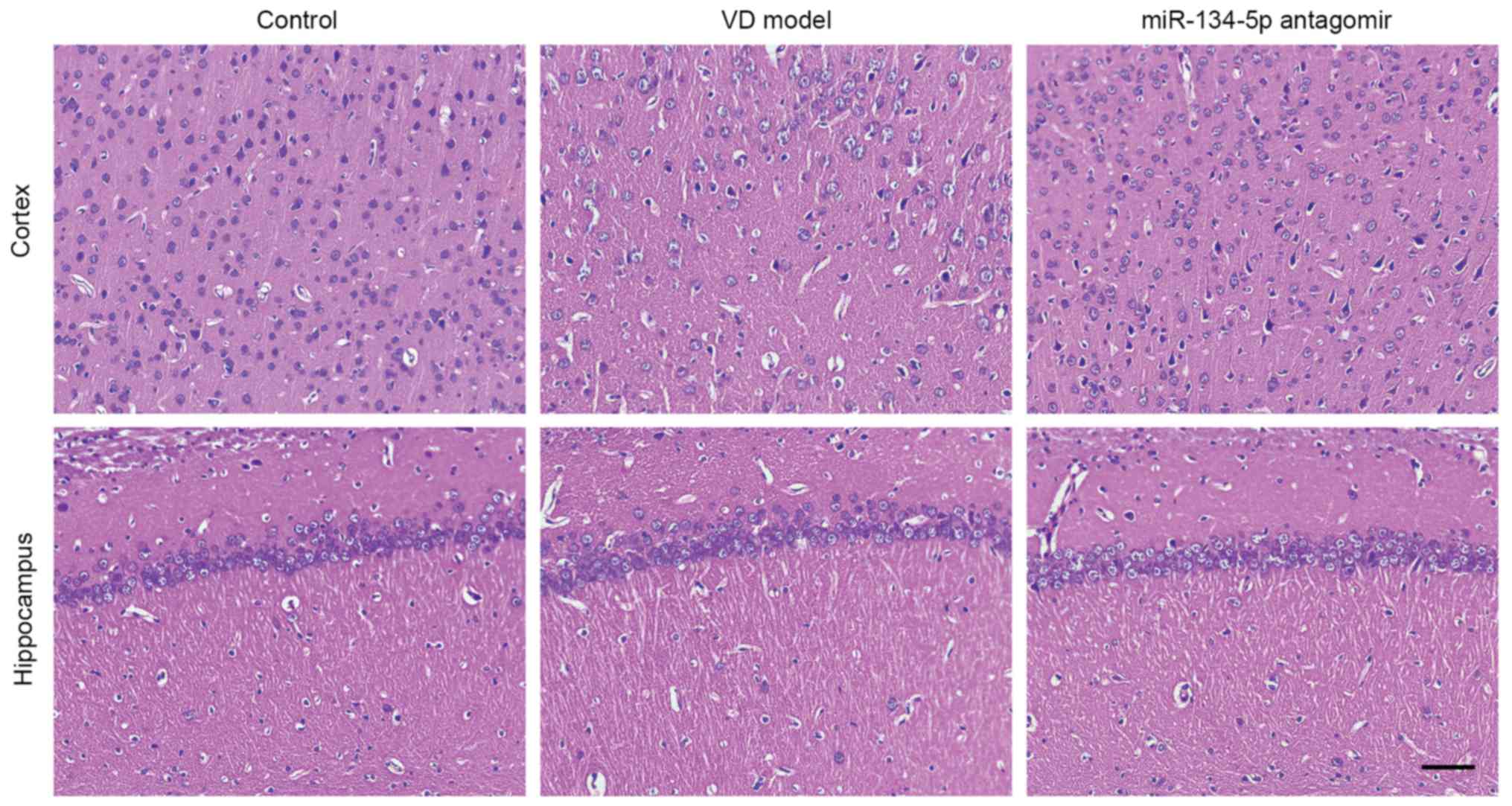
Histopathological changes and apoptosis
levels are increased in the cortex of VD rats
Histopathological changes in the cortex and the
cornu ammonis 1 (CA1) region of the hippocampus were observed
following staining with H&E at 4 weeks after 2-VO, and are
shown in Fig. 2 (n=3/group). In
the cortex of VD rats, neuronal cell loss, shrinkage, dark staining
of neurons and marked vacuolar changes were observed in the cortex.
Additionally, the space between neurons was enlarged. The nucleus
was pyknotic and the structure was not clear. Inhibition of
miR-134-5p attenuated chronic hypoperfusion-induced neuronal cell
injury. The structure of the nucleus in the hippocampal CA1 region
of VD rats was not sufficiently clear compared with the control
group; however, no significant damage in hippocampal CA1 neurons
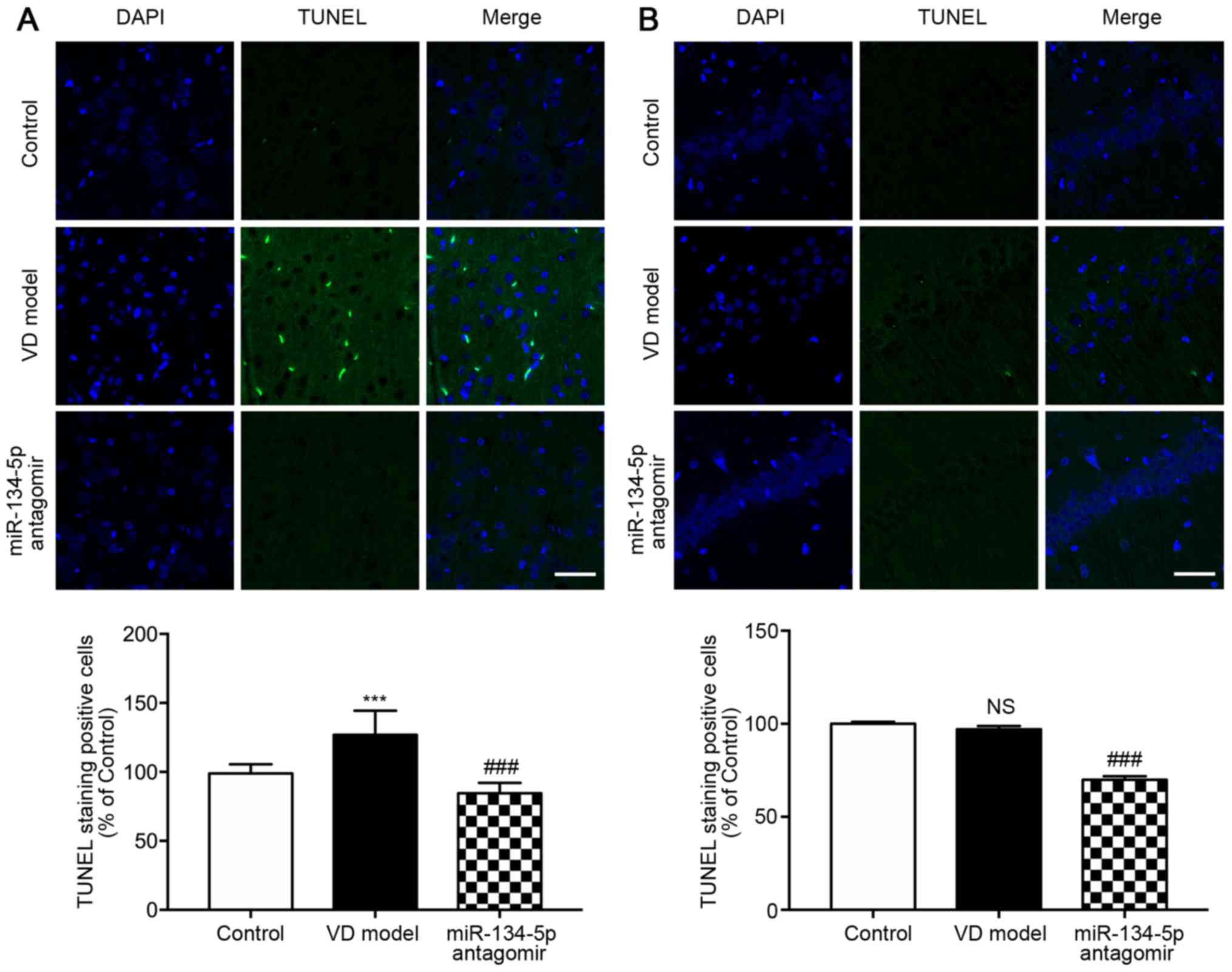
was observed in VD rats. Neurons undergoing apoptosis were detected
using TUNEL staining (n=3/group). As shown in Fig. 3A and B, there were more
TUNEL-positive neurons in the cortex of VD rats compared with the
hippocampal CA1 region. By contrast to VD rats, the miR-134-5p
antagomir group had markedly fewer TUNEL-positive neurons. These
results suggested that cortical injury was more prominent compared
with that of the hippocampus in early VD rats following chronic
ischemia for 4 weeks.
Foxp2 is a potential target of
miR-134-5p
Bioinformatics analysis predicted that the
miR-134-5p sequence GUCAGUG was associated with the Foxp2 gene
sequence CAGUCAC (Fig. 4A). To
assess the direct effect of miR-134-5p on Foxp2 gene expression,
dual-luciferase reporters carrying the miR-134-5p target site in
pLUC-Foxp2 were constructed (Fig.
4B). This analysis demonstrated that an miR-134-5p mimic
significantly decreased the luciferase activity of Foxp2. By
contrast, the miR-134-5p mimic did not reduce luciferase activity
when the miR-134-5p seed sequence at the 3'UTR of Foxp2 was mutated
(Fig. 4C). To determine whether
the miRNA mimic and inhibitor transfections were successful,
RT-qPCR analysis was performed (n=3/group). The results revealed
that the expression of miR-134-5p was significantly increased in
the mimic group compared with that in the mimic-NC group, and
significantly reduced in the inhibitor group compared with that in
the inhibitor-NC group (Fig. 4D).
Western blot analysis identified Foxp2 as a potential target of
miR-134-5p. The expression of Foxp2 was reduced in the mimic group
compared with the mimic-NC group and increased in the inhibitor
group compared with the inhibitor-NC group (Fig. 4E). These results indicated that
miR-134-5p regulates Foxp2 directly.
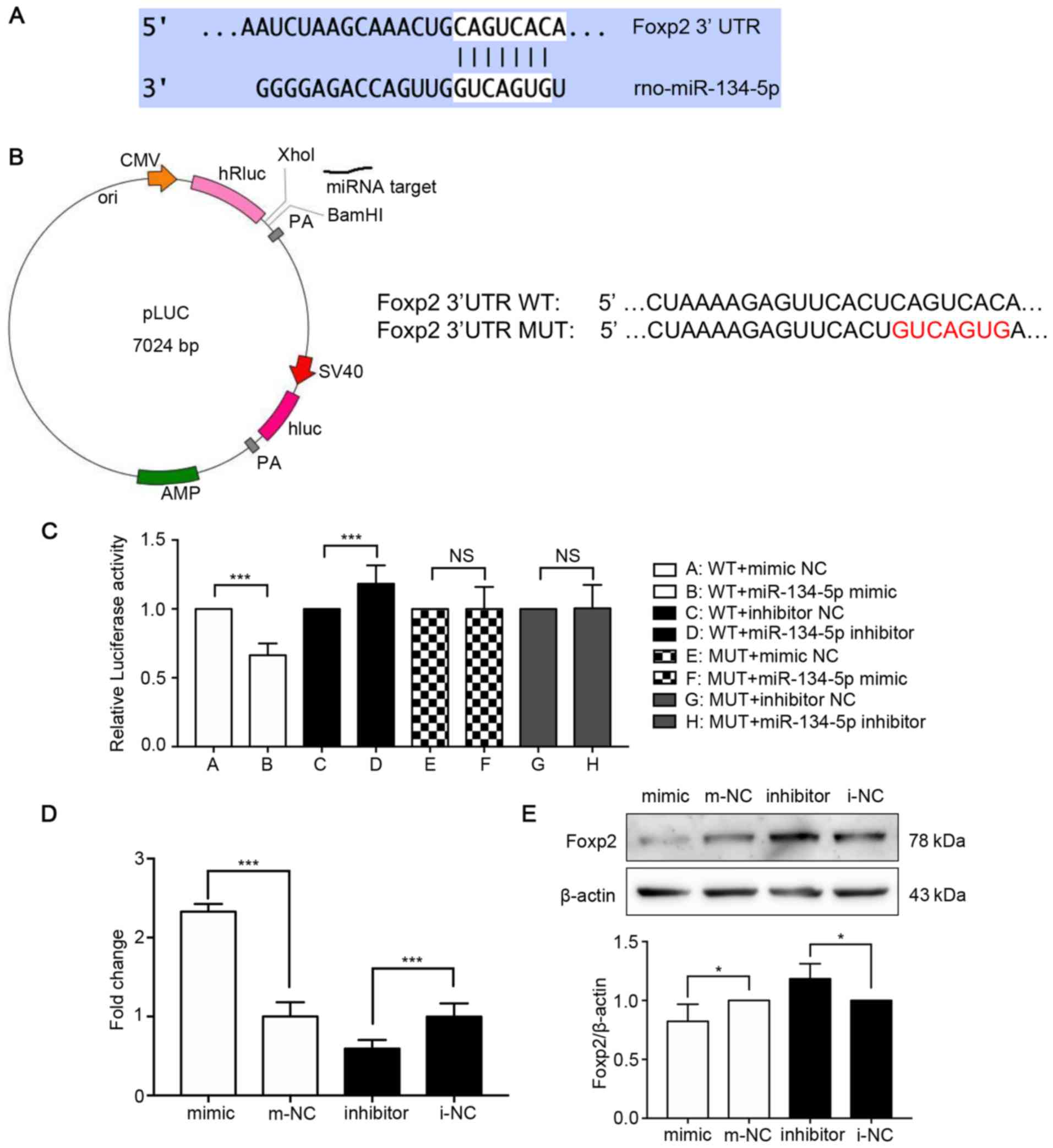 | Figure 4Foxp2 is a potential target of
miR-134-5p. (A) miR-134 binding sites in the 3′-UTR of Foxp2. The
target sites were analyzed by Targetscan, miRBase and miRWalk. (B)
Construction of pLUC-Foxp2-WT and pLUC-Foxp2-MUT containing the
miR-134-5p target sites in the Foxp2 3′-UTR. (C) pLUC-Foxp2-WT and
the corresponding mutant construct pLUC-Foxp2-MUT were
co-transfected into PC12 cells together with miR-134-5p mimic,
inhibitor or the corresponding miRNA negative controls. The
relative luciferase activity of PC12 cells indicated that there was
an association between miR-134-5p and Foxp2. (D) RT-qPCR analysis
demonstrated the miRNA mimic and inhibitor transfections were
effective and successful (n=3/group). (E) Western blot analysis
revealed a decrease in the expression of Foxp2 in PC12 cells
transfected with miR-134-5p mimic and an increase in the expression
of Foxp2 in cells transfected with the miR-134-5p inhibitor. Data
were analyzed using one-way ANOVA and are presented as the mean ±
standard deviation (n=3/group). *P<0.05,
***P<0.001 vs. the respective miRNA NC groups. Foxp2,
forkhead box P2; miR, microRNA; m-NC, miRNA mimic negative control;
i-NC, miRNA inhibitor negative control; WT, wild-type; MUT, mutant;
UTR, untranslated region. |
miR-134-5p inhibitor relieves synaptic
reduction in vivo and in vitro
Western blotting verified that the expression levels
of the representative synaptic-associated proteins, Syn1 and
Snap25, were significantly reduced in the cortex of VD model rats
compared with the control group, and were markedly increased in the
cortex of miR-134-5p antagomir rats compared with VD model rats
(Fig. 5A). According to the
results of immunohistochemical staining (n=3/group), the expression
of Syn1 was significantly reduced in the cortex of VD rats compared
with the control group. The miR-134-5p antagomir significantly
enhanced Syn1 expression compared with the VD model group (Fig. 5B). To determine whether Foxp2
expression in the cortex was associated with synaptic reduction in
the development of early VD and the targeting of miR-134-5p to
Foxp2 in vivo, western blot analysis was performed to detect
Foxp2 expression (n=3/group). The results indicated that the
expression of Foxp2 in the cortex of VD rats was significantly
decreased compared with that in the control group, and was
significantly higher in the cortex of miR-134-5p antagomir rats
compared with VD model rats (Fig.
5C). In order to further elucidate the effect of miR-134-5p on
loss of synaptic proteins, miR-134-5p was transfected into PC12
cells. The results of the RT-qPCR analysis demonstrated that the
miRNA mimic and inhibitor transfections were successful
(n=3/group). The expression of miR-134-5p was significantly
increased in the mimic group compared with the mimic-NC group, and
significantly reduced in the inhibitor group compared with the
inhibitor-NC group (Fig. 5D).
Western blotting revealed that the expression levels of Snap25 and
Syn1 were decreased in the miR-134-5p mimic group and increased in
the miR-134-5p inhibitor group, compared with the respective miRNA
NC groups (Fig. 5E). These
results indicated concomitant synaptic protein loss in VD rats, and
the downregulation of miR-134-5p relieved synaptic protein loss in
the cortex of VD rats. In addition, these results indicated that
miR-134-5p regulated the loss of synaptic proteins in vivo
and in vitro, and the targeting of miR-134-5p to Foxp2 was
further verified in vivo.
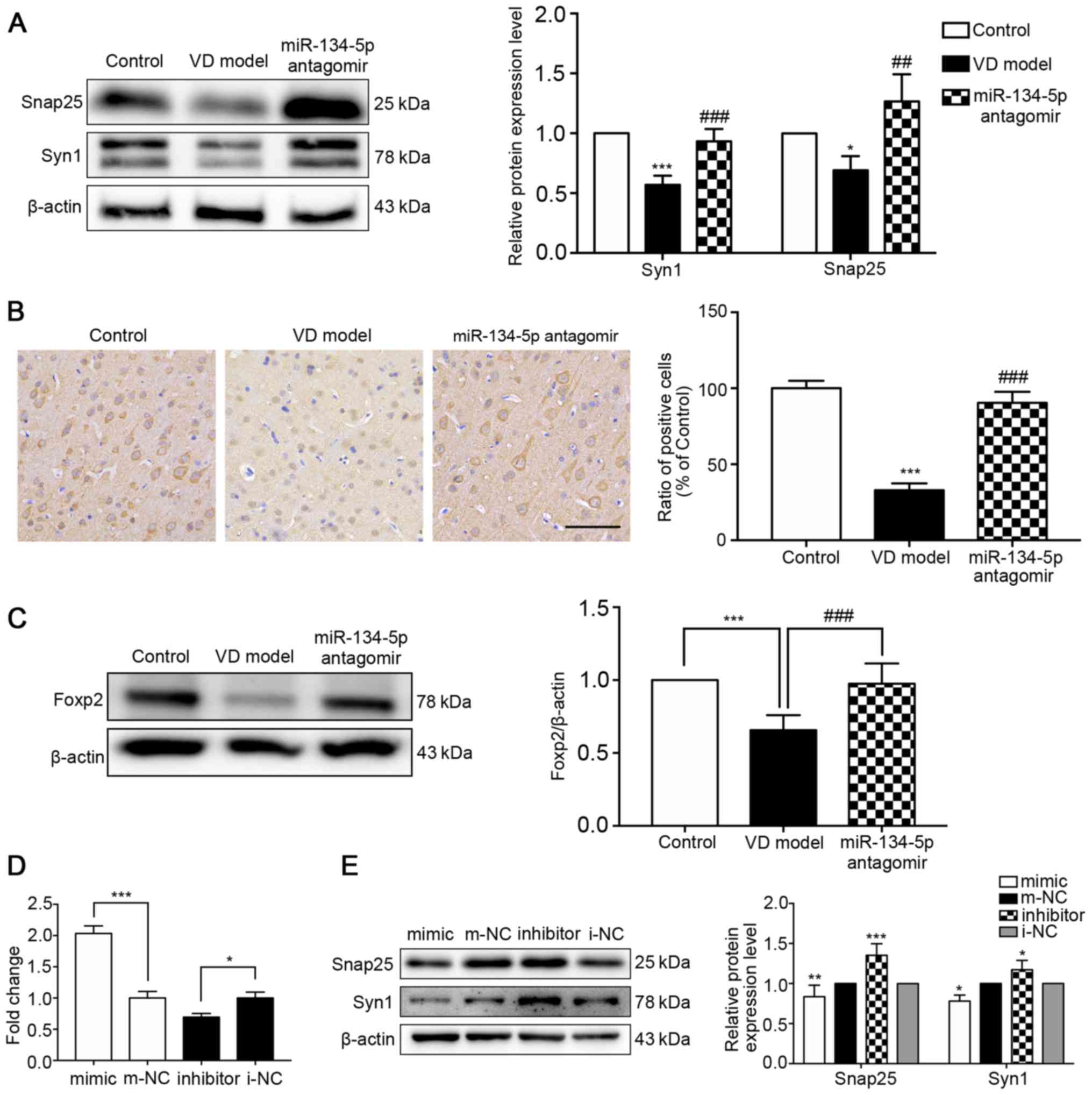 | Figure 5Inhibiting the expression of
miR-134-5p reduces the loss of synaptic proteins in vivo and
in vitro. Expression of (A) Syn1 and Snap25. Two bands for
Syn1 are shown, as the Syn1 gene is subject to alternative
splicing, producing two isoforms (synapsin Ia and Ib) which are
differentially expressed in animal tissues and cells.
*P<0.05, ***P<0.001 vs. the control
group; ##P<0.01, ###P<0.001 vs. the VD
model group. (B) miR-134-5p antagomir attenuated the loss of the
Syn1 protein in the cortex of VD rats. Immunohistochemical staining
of paraffin sections was used to observe the expression of Syn1
(n=3/group). ***P<0.001 vs. the control group;
###P<0.001 vs. the VD model group. (C) Foxp2 was
determined using western blotting (n=3/group).
***P<0.001 vs. the control group;
###P<0.001 vs. the VD model group. (D) RT-qPCR
analysis confirmed that the miRNA mimic and inhibitor transfections
were effective and successful (n=3/group). *P<0.05,
***P<0.001 vs. the respective miRNA NC groups. (E)
Western blot analysis revealed decreased expression of Syn1 and
Snap25 in PC12 cells transfected with miR-134-5p mimic and an
increased expression of Syn1 and Snap25 in cells transfected with
miR-134-5p inhibitor (n=3/group). *P<0.05,
**P<0.01, ***P<0.001 vs. the respective
miRNA NC groups. Data were analyzed by one-way ANOVA and are
presented as the mean ± standard deviation. Scale bar, 50
μm. VD, vascular dementia; miR, microRNA; m-NC, miRNA mimic
negative control; i-NC, miRNA inhibitor negative control; Syn1,
synapsin I; Snap25, synaptosome-associated protein 25; Foxp2,
forkhead box P2. |
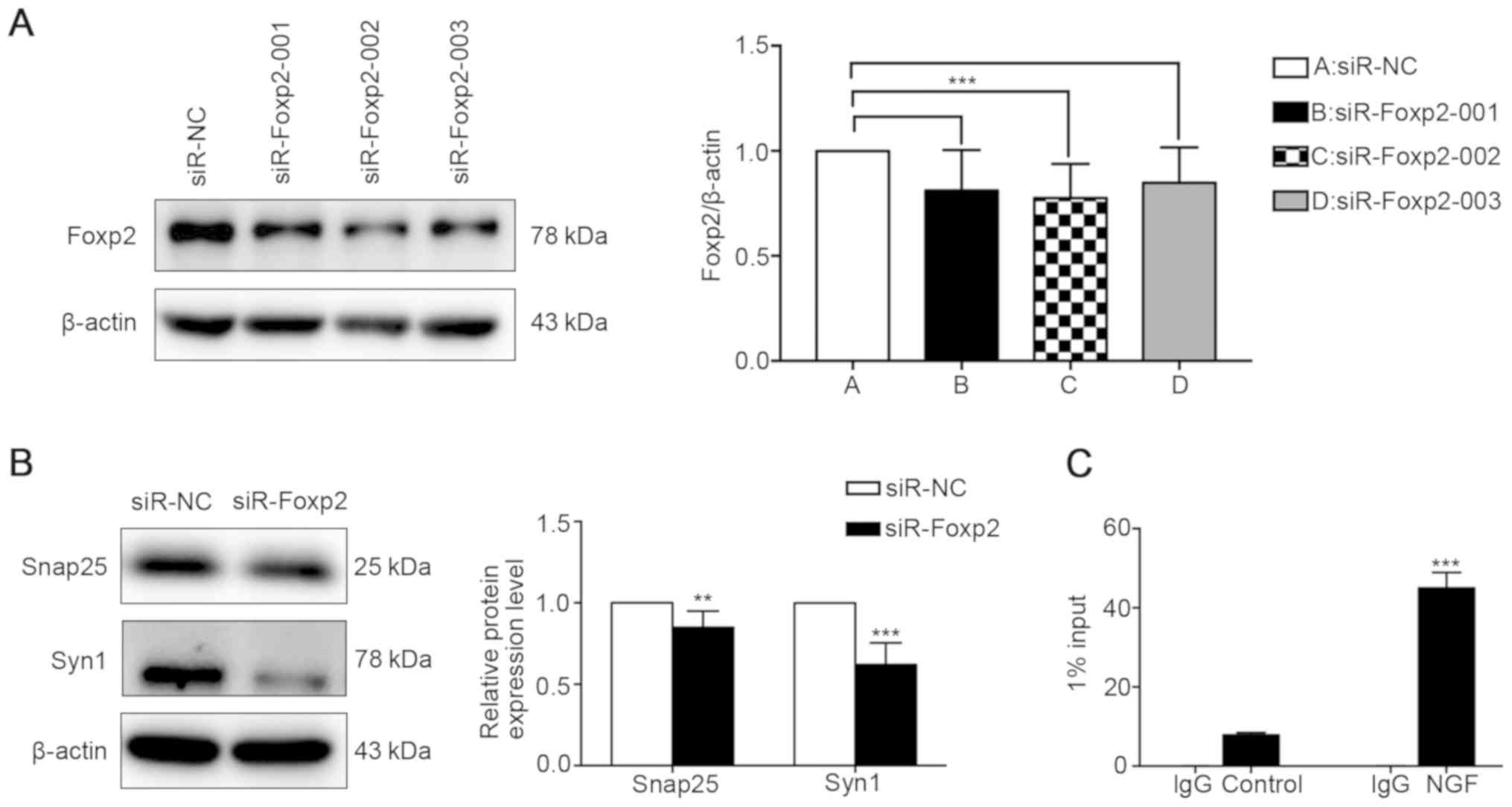
Silencing Foxp2 decreases the expression
of synaptic-associated proteins
To investigate whether miR-134-5p regulates the
expression of synaptic-associated proteins by targeting Foxp2,
siR-Foxp2-001, siR-Foxp2-002 or siR-Foxp2-003 were transfected into
PC12 cells to determine the effect of Foxp2 on the expression of
Syn1 and Snap25. Western blot analysis revealed that siR-Foxp2-002
significantly suppressed the expression of Foxp2 at the protein
level compared with siR-NC (Fig.
6A). The expression of synaptic-associated proteins, such as
Syn1 and Snap25, was decreased in the siR-Foxp2-002 group compared
with the siR-NC group (Fig. 6B).
These data confirmed that the downregulation of Foxp2 significantly
inhibited the expression of the synapse-associated proteins Syn1
and Snap25, thereby affecting the morphology and function of
synapses. Due to the direct targeting effect of miR-134-5p on
Foxp2, these results also indicated that miR-134-5p affects the
expression of synaptic-associated proteins by regulating Foxp2,
rather than by directly regulating synaptic-associated
proteins.
Syn1 is a potential downstream target of
Foxp2
To investigate whether Syn1 is a downstream target
of Foxp2, ChIP assays were performed. The model group was PC12
cells induced with NGF (50 ng/ml) for 1 week. The control group
received no treatment. Verified using an RT-qPCR assay, the level
of DNA fragment in the model group was significantly increased
compared with the control group (Fig.
6C). ChIP analysis revealed the binding of Foxp2 to the Syn1
promoter at −400/−600 bp upstream of the transcription start site
in PC12 cells induced by NGF. These results indicated that Foxp2
regulates synaptic changes through its interaction with the Syn1
promoter.
Discussion
VD is often accompanied by changes in cognitive
function, which is closely associated with early synaptic changes
(37). Ischemic VD leads to
cognitive dysfunction (38,39). However, it is not known how
miR-134-5p regulates cognitive impairment in VD caused by chronic
ischemia. In the present study, 2-VO was used to create a VD model
and was found to mimic several prominent pathological
characteristics of early VD. Whether miR-134-5p/Foxp2/Syn1
participates in cognitive dysfunction in the early stage of VD was
also investigated. The principal findings of the present study were
as follows: i) miR-134-5p was increased in the cortex of VD rats;
ii) downregulation of miR-134-5p significantly relieved the
cognitive deficit of VD rats; iii) Foxp2 was identified as a direct
target of miR-134-5p; and iv) Foxp2 acts as a transcription factor
and Syn1 may be a downstream target of Foxp2. miR-134-5p was found
to indirectly regulate the loss of synaptic proteins through the
direct targeting of Foxp2. These results suggested that the
miR-134-5p/Foxp2/Syn1 signaling pathway may play an important role
in cognitive dysfunction in early VD.
2-VO is a well-characterized model for investigating
the cognitive and histopathological consequences of chronic
cerebral hypoperfusion (4). This
model more accurately simulates VD caused by vascular factors,
which is similar to the pathological process in the brain tissue in
chronic cerebral hypoperfusion (40). In the present study, rats were
found to exhibit spatial learning impairment and synaptic protein
loss following 4 weeks of chronic ischemia. Several miRNAs may be
used as diagnostic markers for VD. It has been reported that plasma
miR-409-3p, miR-502-3p and miR-451a may be used to differentiate
small-vessel VD patients from healthy controls (41). In addition, a number of miRNAs
have been shown to be involved in synapses and may participate in
the regulation of synaptic development, as well as functional
regulation (42). A previous
study reported that miR-134-5p is involved in the development and
morphological changes of synapses (43). miR-134 is found in neurons and
dendrites of primary cultured neurons, where it regulates the
development of the dendritic spine by targeting LimK1 mRNA, which
can be inhibited by BDNF (18).
Furthermore, the overexpression of miR-134 leads to a markedly
decreased the size of neuronal dendritic spines (33). miR-146, miR-125 and miR-485 were
previously found to be involved in the regulation of synaptic
plasticity, including control of dendritic spine number and synapse
formation (29,44-46). However, there is currently no
definitive and effective method for preventing the progression of
cognitive dysfunction.
The key finding of the present study was that
miR-134-5p is involved in cognitive dysfunction in VD rats. RT-qPCR
analysis revealed that miR-134-5p expression was increased in the
cortex of VD rats; however, no significant change in hippocampal
tissue was observed. Therefore, it was inferred that the cortex is
one of the initial lesion sites in the early stage of VD induced by
4 weeks of ischemia. H&E and TUNEL staining in the present
study indicated that pathological damage is more prominent in the
cortex of early VD rats compared with that in the hippocampus after
chronic cerebral hypoperfusion for 4 weeks. Inhibition of
miR-134-5p expression ameliorated the cerebral hypoperfusioninduced
neuronal damage. This observation was consistent with the results
of the MWM test. Specific deficits were found in animals with
damage to the hippocampus, striatum, basal forebrain, cerebellum
and several cortical areas, which may affect MWM performance in
various ways (47).
Another important finding of the present study was
that miR-134-5p contributes to the loss of synaptic proteins in VD
rats. Western blotting and immunohistochemical staining suggested
that the inhibition of miR-134-5p increased the loss of synaptic
proteins. miR-134-5p was also demonstrated to regulate the
expression of synaptic proteins in vivo and in vitro.
A number of previous studies have reported that synaptic protein
loss is associated with cognitive impairment in VD rats (8,48).
The present study revealed that miR-134-5p promotes cognitive
impairment through the loss of synaptic proteins. According to the
results of the histopathological analysis, the miR-134-5p antagomir
significantly improved pathological tissue injury in the brains of
VD rats.
An important mechanism of action of miR-134-5p was
revealed in the present study. miR-134-5p was found to regulate the
loss of synaptic proteins by targeting the mRNA of Foxp2. Foxp2 was
identified as a target of miR-134-5p using bioinformatics target
prediction tools. As a transcription factor, Foxp2 regulates the
expression of hundreds of downstream target genes, many of which
are involved in regulating neural development and function in
different ways (49). A number of
these target genes play important roles in synaptic plasticity,
including axon growth, axon guidance and neurotransmission
(23,50). Foxp2 is enriched in the cerebral
cortex, striatum and cerebellum of the rodent brain, which are
areas participating in motor control (51,52). In Foxp2 mutant mice, in addition
to vocal cord defects, changes in synaptic plasticity and impaired
motor learning ability have been observed (53).
To verify the effect of Foxp2 on synaptic proteins,
siRNA experiments were conducted to silence Foxp2 in the present
study. These experiments indicated that Foxp2 inhibited the
expression of Syn1 and Snap25. The data confirmed that miR-134-5p
promoted cognitive impairment in VD rats by inhibiting Foxp2
directly. Syn1 also acts as a downstream target gene of Foxp2 and
regulates the development and alterations of synapses (54). ChIP-qPCR analysis suggested that
Syn1 is a downstream target gene of Foxp2. Syn1 plays multiple
roles in synaptic transmission and plasticity by differentially
affecting important steps of synaptic vesicle trafficking in
excitatory and inhibitory synapses (55). The results of the present study
indicated that Foxp2 directly binds to the Syn1 promoter sequence
to regulate synaptic damage. The Foxp2/Syn1 pathway may also
represent a novel target for the functional study of the effect of
Foxp2 on synaptic plasticity.
In conclusion, the results of the present study have
several important clinical implications for VD. First, miR-134-5p
may be a potential biomarker for the early diagnosis of VD. It was
demonstrated that miR-134-5p aggravates cognitive impairment in VD
rats by damaging cortical neurons and reducing synaptic proteins.
Second, the data suggested that miR-134-5p promotes cognitive
impairment in early VD rats by inhibiting Foxp2. Therefore, the
miR-134-5p/Foxp2/Syn1 pathway may represent a novel target for
early VD synaptic damage and molecular-based therapeutic
strategies.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 81673770) and the
Natural Science Foundation of Guangdong Province (grant no.
2017A030312009).
Availability of data and materials
The datasets generated and analyzed during the
present study are available from the corresponding author on
reasonable request.
Authors' contributions
XL performed the experiments and wrote the
manuscript; RZ and ZW performed experiments, including the
animal-associated experiments and molecular biology; WS and ZR
provided experimental technical guidance; SZ and JZ provided
technical assistance; DC conceived and designed the experiments,
and secured the project funds. All authors have read and approved
the final version of this manuscript for publication.
Ethics approval and consent to
participate
All animal experiments were approved by the Care and
Use of Experimental Animals Committee of Guangzhou University of
Chinese Medicine and were performed according to the National
Institute of Health Guide for the Care and Use of Laboratory
Animals.
Patient consent for publication
Not applicable.
Competing interests
All authors declare that they have no competing
interests.
References
|
1
|
Guo LL, Wang DS, Xu YY and Cui KG: Effects
of IL-1β on hippocampus cell apoptosis and learning ability of
vascular dementia rats. Eur Rev Med Pharmacol Sci. 22:6042–6048.
2018.PubMed/NCBI
|
|
2
|
Yang JW, Wang XR, Zhang M, Xiao LY, Zhu W,
Ji CS and Liu CZ: Acupuncture as a multifunctional neuroprotective
therapy ameliorates cognitive impairment in a rat model of vascular
dementia: A quantitative iTRAQ proteomics study. CNS Neurosci Ther.
24:1264–1274. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yang HY, Liu Y, Xie JC, Liu NN and Tian X:
Effects of repetitive transcranial magnetic stimulation on synaptic
plasticity and apoptosis in vascular dementia rats. Behav Brain
Res. 281:149–155. 2015. View Article : Google Scholar
|
|
4
|
Farkas E and Luiten PG: Cerebral
microvascular pathology in aging and Alzheimer's disease. Prog
Neurobiol. 64:575–611. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Damodaran T, Müller CP and Hassan Z:
Chronic cerebral hypoperfusion-induced memory impairment and
hippocampal long-term potentiation deficits are improved by
cholinergic stimulation in rats. Pharmacol Rep. 71:443–448. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Waldmeier PC: Prospects for antiapoptotic
drug therapy of neurodegenerative diseases. Prog
Neuropsychopharmacol Biol Psychiatry. 27:303–321. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jinglong T, Weijuan G, Jun L, Tao Q,
Hongbo Z and Shasha L: The molecular and electrophysiological
mechanism of buyanghuanwu decoction in learning and memory ability
of vascular dementia rats. Brain Res Bull. 99:13–18. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Scheff SW, Neltner JH and Nelson PT: Is
synaptic loss a unique hallmark of Alzheimer's disease? Biochem
Pharmacol. 88:517–528. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kalaria RN: The pathology and
pathophysiology of vascular dementia. Neuropharmacology.
134:226–239. 2018. View Article : Google Scholar
|
|
10
|
Ren Z, Yu J, Wu Z, Si W, Li X, Liu Y, Zhou
J, Deng R and Chen D: MicroRNA-210-5p contributes to cognitive
impairment in early vascular dementia rat model through targeting
Snap25. Front Mol Neurosci. 11:3882018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Terry RD, Masliah E, Salmon DP, Butters N,
DeTeresa R, Hill R, Hansen LA and Katzman R: Physical basis of
cognitive alterations in Alzheimer's disease: Synapse loss is the
major correlate of cognitive impairment. Ann Neurol. 30:572–580.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Blennow K, Bogdanovic N, Alafuzoff I,
Ekman R and Davidsson P: Synaptic pathology in Alzheimer's disease:
Relation to severity of dementia, but not to senile plaques,
neurofibrillary tangles, or the ApoE4 allele. J Neural Transm
(Vienna). 103:603–618. 1996. View Article : Google Scholar
|
|
13
|
Guo Y, Zhao Y, Nan Y, Wang X, Chen Y and
Wang S: (-)-Epigallocatechin-3-gallate ameliorates memory
impairment and rescues the abnormal synaptic protein levels in the
frontal cortex and hippocampus in a mouse model of Alzheimer's
disease. Neuroreport. 28:590–597. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guarnieri FC, Pozzi D, Raimondi A, Fesce
R, Valente MM, Delvecchio VS, Van Esch H, Matteoli M, Benfenati F,
D'Adamo P and Valtorta F: A novel SYN1 missense mutation in
non-syndromic X-linked intellectual disability affects synaptic
vesicle life cycle, clustering and mobility. Hum Mol Genet.
26:4699–4714. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Batista AFR, Martinez JC and Hengst U:
Intra-axonal synthesis of SNAP25 is required for the formation of
presynaptic terminals. Cell Rep. 20:3085–3098. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fassio A, Patry L, Congia S, Onofri F,
Piton A, Gauthier J, Pozzi D, Messa M, Defranchi E, Fadda M, et al:
SYN1 loss-of-function mutations in autism and partial epilepsy
cause impaired synaptic function. Hum Mol Genet. 20:2297–2307.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shupliakov O, Haucke V and Pechstein A:
How synapsin I may cluster synaptic vesicles. Semin Cell Dev Biol.
22:393–399. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sinclair LI, Tayler HM and Love S:
Synaptic protein levels altered in vascular dementia. Neuropathol
Appl Neurobiol. 41:533–543. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gallart-Palau X, Serra A, Qian J, Chen CP,
Kalaria RN and Sze SK: Temporal lobe proteins implicated in
synaptic failure exhibit differential expression and deamidation in
vascular dementia. Neurochem Int. 80:87–98. 2015. View Article : Google Scholar
|
|
20
|
Rodenas-Cuadrado PM, Mengede J, Baas L,
Devanna P, Schmid TA, Yartsev M, Firzlaff U and Vernes SC: Mapping
the distribution of language related genes FoxP1, FoxP2, and
CntnaP2 in the brains of vocal learning bat species. J Comp Neurol.
526:1235–1266. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chiu YC, Li MY, Liu YH, Ding JY, Yu JY and
Wang TW: Foxp2 regulates neuronal differentiation and neuronal
subtype specification. Dev Neurobiol. 74:723–738. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vernes SC, Spiteri E, Nicod J, Groszer M,
Taylor JM, Davies KE, Geschwind DH and Fisher SE: High-throughput
analysis of promoter occupancy reveals direct neural targets of
FOXP2, a gene mutated in speech and language disorders. Am J Hum
Genet. 81:1232–1250. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Konopka G, Bomar JM, Winden K, Coppola G,
Jonsson ZO, Gao F, Peng S, Preuss TM, Wohlschlegel JA and Geschwind
DH: Human-specific transcriptional regulation of CNS development
genes by FOXP2. Nature. 462:213–217. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Spiteri E, Konopka G, Coppola G, Bomar J,
Oldham M, Ou J, Vernes SC, Fisher SE, Ren B and Geschwind DH:
Identification of the transcriptional targets of FOXP2, a gene
linked to speech and language, in developing human brain. Am J Hum
Genet. 81:1144–1157. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vernes SC, Oliver PL, Spiteri E, Lockstone
HE, Puliyadi R, Taylor JM, Ho J, Mombereau C, Brewer A, Lowy E, et
al: Foxp2 regulates gene networks implicated in neurite outgrowth
in the developing brain. PLoS Genet. 7:pp. e10021452011, View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fu L, Shi Z, Luo G, Tu W, Wang X, Fang Z
and Li X: Multiple microRNAs regulate human FOXP2 gene expression
by targeting sequences in its 3' untranslated region. Mol Brain.
7:712014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Higuchi Y, Soga T and Parhar IS: Potential
roles of microRNAs in the regulation of monoamine oxidase a in the
brain. Front Mol Neurosci. 11:3392018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang D, Wang X, Liu X, Jiang L, Yang G,
Shi X, Zhang C and Piao F: Inhibition of miR-219 alleviates
arsenic-induced learning and memory impairments and synaptic damage
through up-regulating CaMKII in the hippocampus. Neurochem Res.
43:948–958. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Edbauer D, Neilson JR, Foster KA, Wang CF,
Seeburg DP, Batterton MN, Tada T, Dolan BM, Sharp PA and Sheng M:
Regulation of synaptic structure and function by FMRP-associated
microRNAs miR-125b and miR-132. Neuron. 65:373–384. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wayman GA, Davare M, Ando H, Fortin D,
Varlamova O, Cheng HY, Marks D, Obrietan K, Soderling TR, Goodman
RH and Impey S: An activity-regulated microRNA controls dendritic
plasticity by down-regulating p250GAP. Proc Natl Acad Sci USA.
105:9093–9098. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
McCann C, Holohan EE, Das S, Dervan A,
Larkin A, Lee JA, Rodrigues V, Parker R and Ramaswami M: The
Ataxin-2 protein is required for microRNA function and
synapse-specific long-term olfactory habituation. Proc Natl Acad
Sci USA. 108:E655–E662. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schratt GM, Tuebing F, Nigh EA, Kane CG,
Sabatini ME, Kiebler M and Greenberg ME: A brain-specific microRNA
regulates dendritic spine development. Nature. 439:283–289. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bicker S, Lackinger M, Weiß K and Schratt
G: MicroRNA-132, -134, and -138: A microRNA troika rules in
neuronal dendrites. Cell Mol Life Sci. 71:3987–4005. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yu H, Fan C, Yang L, Yu S, Song Q, Wang P
and Mao X: Ginsenoside Rg1 prevents chronic stress-induced
depression-like behaviors and neuronal structural plasticity in
rats. Cell Physiol Biochem. 48:2470–2482. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
36
|
Liu C, Guo Y, Zhao F, Qin H, Lu H, Fang L,
Wang J and Min W: Potential mechanisms mediating the protective
effects of a peptide from walnut (Juglans mandshurica Maxim.)
against hydrogen peroxide induced neurotoxicity in PC12 cells. Food
Funct. 10:3491–3501. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Niu XL, Jiang X, Xu GD, Zheng GM, Tang ZP,
Yin N, Li XQ, Yang YY and Lv PY: DL-3-n-butylphthalide alleviates
vascular cognitive impairment by regulating endoplasmic reticulum
stress and the Shh/Ptch1 signaling-pathway in rats. J Cell Physiol.
234:12604–12614. 2019. View Article : Google Scholar
|
|
38
|
Komatani A, Yamaguchi K, Sugai Y,
Takanashi T, Kera M, Shinohara M and Kawakatsu S: Assessment of
demented patients by dynamic SPECT of inhaled xenon-133. J Nucl
Med. 29:1621–1626. 1988.PubMed/NCBI
|
|
39
|
Ohnishi T, Hoshi H, Nagamachi S, Jinnouchi
S, Flores LG II, Futami S and Watanabe K: High-resolution SPECT to
assess hippocampal perfusion in neuropsychiatric diseases. J Nucl
Med. 36:1163–1169. 1995.PubMed/NCBI
|
|
40
|
Tsuchiya M, Sako K, Yura S and Yonemasu Y:
Local cerebral glucose utilisation following acute and chronic
bilateral carotid artery ligation in Wistar rats: Relation to
changes in local cerebral blood flow. Exp Brain Res. 95:1–7. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Prabhakar P, Chandra SR and Christopher R:
Circulating microRNAs as potential biomarkers for the
identification of vascular dementia due to cerebral small vessel
disease. Age Ageing. 46:861–864. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Baek D, Villén J, Shin C, Camargo FD, Gygi
SP and Bartel DP: The impact of microRNAs on protein output.
Nature. 455:64–71. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
McGowan H, Mirabella VR, Hamod A,
Karakhanyan A, Mlynaryk N, Moore JC, Tischfield JA, Hart RP and
Pang ZP: Hsa-let-7c miRNA regulates synaptic and neuronal function
in human neurons. Front Synaptic Neurosci. 10:192018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Prada I, Gabrielli M, Turola E, Iorio A,
D'Arrigo G, Parolisi R, De Luca M, Pacifici M, Bastoni M, Lombardi
M, et al: Glia-to-neuron transfer of miRNAs via extracellular
vesicles: A new mechanism underlying inflammation-induced synaptic
alterations. Acta Neuropathol. 135:529–550. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wang X, Liu D, Huang HZ, Wang ZH, Hou TY,
Yang X, Pang P, Wei N, Zhou YF, Dupras MJ, et al: A novel
microRNA-124/PTPN1 signal pathway mediates synaptic and memory
deficits in Alzheimer's disease. Biol Psychiatry. 83:395–405. 2018.
View Article : Google Scholar
|
|
46
|
Cohen JE, Lee PR, Chen S, Li W and Fields
RD: MicroRNA regulation of homeostatic synaptic plasticity. Proc
Natl Acad Sci USA. 108:11650–11655. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
D'Hooge R and De Deyn PP: Applications of
the Morris water maze in the study of learning and memory. Brain
Res Brain Res Rev. 36:60–90. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zhu Y, Zhang Q, Zhang W, Li N, Dai Y, Tu
J, Yang F, Brann DW and Wang R: Protective effect of 17β-estradiol
upon hippo-campal spine density and cognitive function in an animal
model of vascular dementia. Sci Rep. 7:426602017. View Article : Google Scholar
|
|
49
|
Estruch SB, Graham SA, Quevedo M, Vino A,
Dekkers DHW, Deriziotis P, Sollis E, Demmers J, Poot RA and Fisher
SE: Proteomic analysis of FOXP proteins reveals interactions
between cortical transcription factors associated with
neurodevelopmental disorders. Hum Mol Genet. 27:1212–1227. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Hachigian LJ, Carmona V, Fenster RJ,
Kulicke R, Heilbut A, Sittler A, Pereira de Almeida L, Mesirov JP,
Gao F, Kolaczyk ED and Heiman M: Control of huntington's
disease-associated phenotypes by the striatum-enriched
transcription factor Foxp2. Cell Rep. 21:2688–2695. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ferland RJ, Cherry TJ, Preware PO,
Morrisey EE and Walsh CA: Characterization of Foxp2 and Foxp1 mRNA
and protein in the developing and mature brain. J Comp Neurol.
460:266–279. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hisaoka T, Nakamura Y, Senba E and
Morikawa Y: The fork-head transcription factors, Foxp1 and Foxp2,
identify different subpopulations of projection neurons in the
mouse cerebral cortex. Neuroscience. 166:551–563. 2010. View Article : Google Scholar
|
|
53
|
Groszer M, Keays DA, Deacon RM, de Bono
JP, Prasad- Mulcare S, Gaub S, Baum MG, French CA, Nicod J,
Coventry JA, et al: Impaired synaptic plasticity and motor learning
in mice with a point mutation implicated in human speech deficits.
Curr Biol. 18:354–362. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Kang HJ, Voleti B, Hajszan T, Rajkowska G,
Stockmeier CA, Licznerski P, Lepack A, Majik MS, Jeong LS, Banasr
M, et al: Decreased expression of synapse-related genes and loss of
synapses in major depressive disorder. Nat Med. 18:1413–1417. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Park HJ, Kim SK, Kang WS, Chung JH and Kim
JW: Increased activation of synapsin 1 and mitogen-activated
protein kinases/extracellular signal-regulated kinase in the
amygdala of maternal separation rats. CNS Neurosci Ther.
20:172–181. 2014. View Article : Google Scholar
|