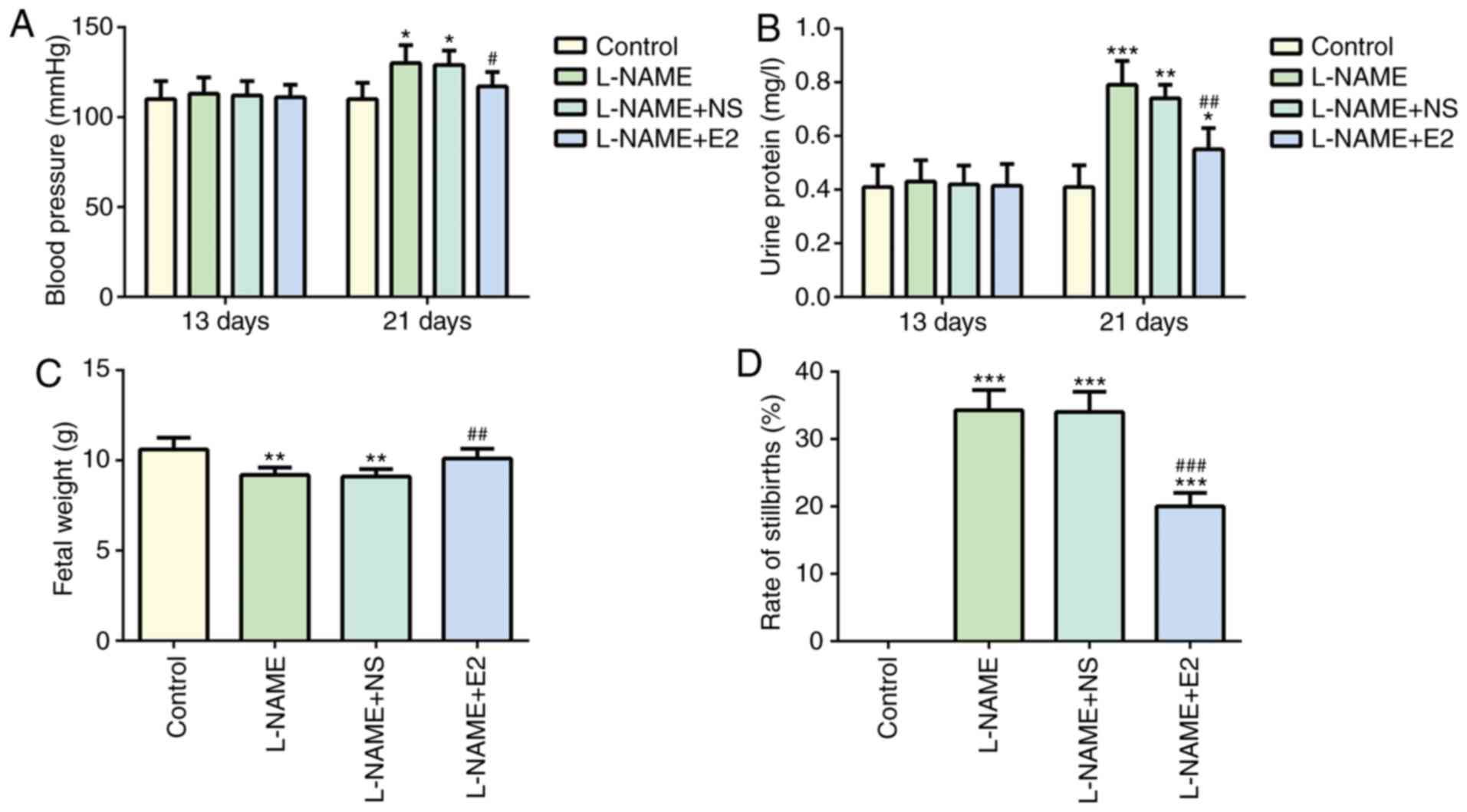
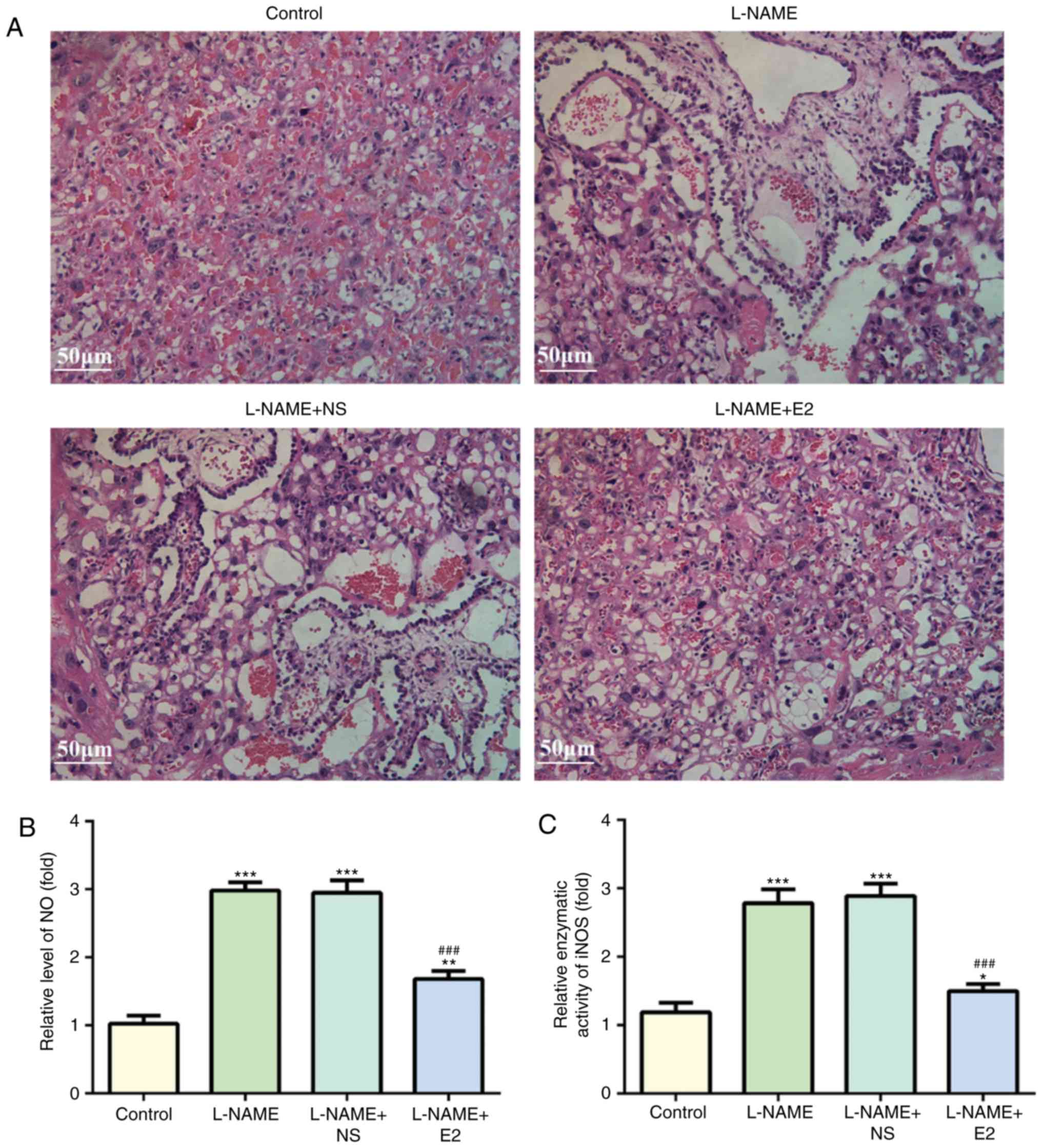
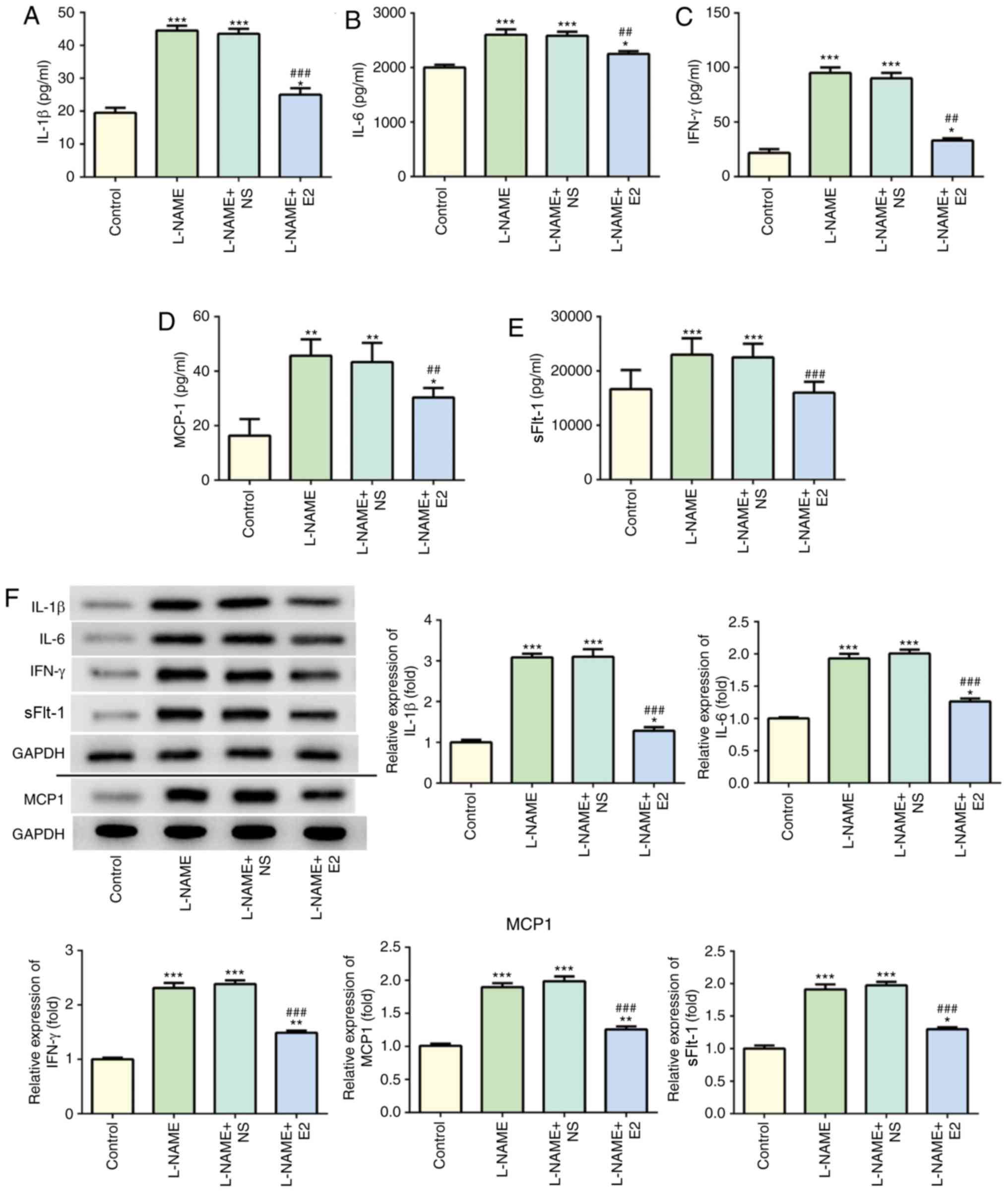
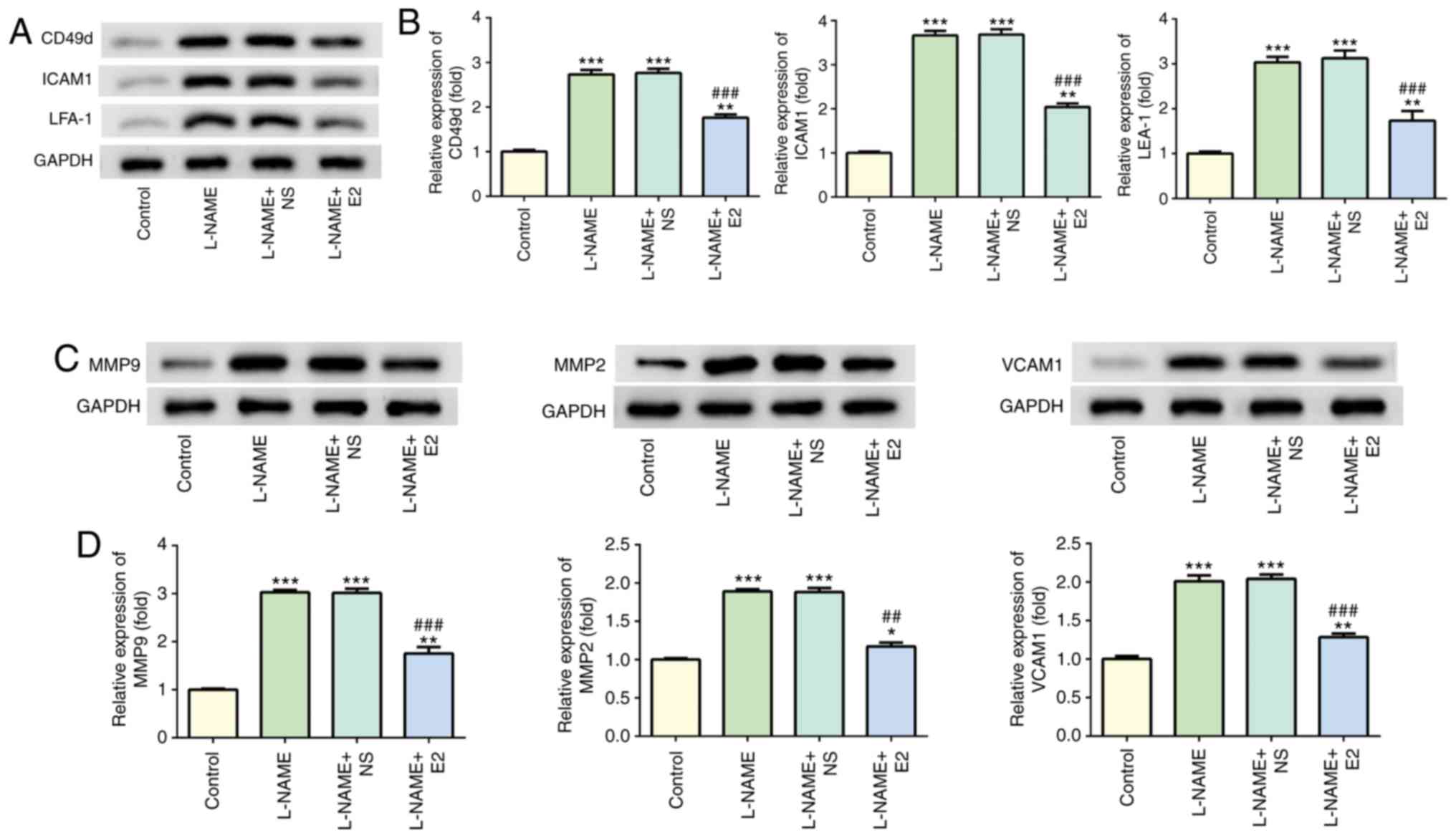
|
1
|
Walker CK, Krakowiak P, Baker A, Hansen
RL, Ozonoff S and Hertz-Picciotto I: Preeclampsia, placental
insufficiency, and autism spectrum disorder or developmental delay.
JAMA Pediatr. 169:154–162. 2015. View Article : Google Scholar :
|
|
2
|
Hod T, Cerdeira AS and Karumanchi SA:
Molecular mechanisms of preeclampsia. Cold Spring Harb Perspect
Med. 5:2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ramos JGL, Sass N and Costa SHM:
Preeclampsia. Rev Bras Ginecol Obstet. 39:496–512. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Borzychowski AM, Croy BA, Chan WL, Redman
CW and Sargent IL: Changes in systemic type 1 and type 2 immunity
in normal pregnancy and pre-eclampsia may be mediated by natural
killer cells. Eur J Immunol. 35:3054–3063. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Biondi C, Pavan B, Lunghi L, Fiorini S and
Vesce F: The role and modulation of the oxidative balance in
pregnancy. Curr Pharm Des. 11:2075–2089. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kiprono LV, Wallace K, Moseley J, Martin J
Jr and Lamarca B: Progesterone blunts vascular endothelial cell
secretion of endothelin-1 in response to placental ischemia. Am J
Obstet Gynecol. 209:44.e1–e6. 2013. View Article : Google Scholar
|
|
7
|
Wei J, Satomi M, Negishi Y, Matsumura Y,
Miura A, Nishi Y, Asakura H and Takeshita T: Effect of sera on the
adhesion of natural killer cells to the endothelium in severe
pre-eclampsia. J Obstet Gynaecol Res. 32:443–448. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mihu D, Razvan C, Malutan A and Mihaela C:
Evaluation of maternal systemic inflammatory response in
preeclampsia. Taiwan J Obstet Gynecol. 54:160–166. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
de Oliveira LG, Karumanchi A and Sass N:
Preeclampsia: Oxidative stress, inflammation and endothelial
dysfunction. Rev Bras Ginecol Obstet. 32:609–616. 2010.In
Portuguese.
|
|
10
|
Tranquilli AL, Dekker G, Magee L, Roberts
J, Sibai BM, Steyn W, Zeeman GG and Brown MA: The classification,
diagnosis and management of the hypertensive disorders of
pregnancy: A revised statement from the ISSHP. Pregnancy Hypertens.
4:97–104. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wei J, Chen Q, James JL, Stone PR and
Chamley LW: IL-1 beta but not the NALP3 inflammasome is an
important determinant of endothelial cell responses to
necrotic/dangerous trophoblastic debris. Placenta. 36:1385–1392.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Southcombe JH, Redman CW, Sargent IL and
Granne I: Interleukin-1 family cytokines and their regulatory
proteins in normal pregnancy and pre-eclampsia. Clin Exp Immunol.
181:480–490. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Khalil RA: Sex hormones as potential
modulators of vascular function in hypertension. Hypertension.
46:249–254. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Duckles SP and Miller VM: Hormonal
modulation of endothelial NO production. Pflugers Arch.
459:841–851. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Salas SP, Marshall G, Gutiérrez BL and
Rosso P: Time course of maternal plasma volume and hormonal changes
in women with preeclampsia or fetal growth restriction.
Hypertension. 47:203–208. 2006. View Article : Google Scholar
|
|
16
|
Zheng JJ, Wang HO, Huang M and Zheng FY:
Assessment of ADMA, estradiol, and progesterone in severe
preeclampsia. Clin Exp Hypertens. 38:347–351. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cikos S, Bukovská A and Koppel J: Relative
quantification of mRNA: Comparison of methods currently used for
real-time PCR data analysis. BMC Mol Biol. 8:1132007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Feng J, Wang X, Li H, Wang L and Tang Z:
Silencing of Annexin A1 suppressed the apoptosis and inflammatory
response of preeclampsia rat trophoblasts. Int J Mol Med.
42:3125–3134. 2018.PubMed/NCBI
|
|
19
|
Kanashiro CA, Cockrell KL, Alexander BT,
Granger JP and Khalil RA: Pregnancy-associated reduction in
vascular protein kinase C activity rebounds during inhibition of NO
synthesis. Am J Physiol Regul Integr Comp Physiol. 278:R295–R303.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Souza CO, Peracoli MT, Weel IC, Bannwart
CF, Romão M, Nakaira-Takahagi E, Medeiros LT, Silva MG and Peraçoli
JC: Hepatoprotective and anti-inflammatory effects of silibinin on
experimental preeclampsia induced by L-NAME in rats. Life Sci.
91:159–165. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Doering TP, Haller NA, Montgomery MA,
Freeman EJ and Hopkins MP: The role of AT1 angiotensin receptor
activation in the pathogenesis of preeclampsia. Am J Obstet
Gynecol. 178:1307–1312. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Beckers KF and Sones JL: Maternal
microbiome and the hypertensive disorder of pregnancy,
preeclampsia. Am J Physiol Heart Circ Physiol. 2019.PubMed/NCBI
|
|
23
|
de Moura RS, Resende AC, Moura AS and
Maradei MF: Protective action of a hydroalcoholic extract of a
vinifera grape skin on experimental preeclampsia in rats. Hypertens
Pregnancy. 26:89–100. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Conrad KP, Joffe GM, Kruszyna H, Kruszyna
R, Rochelle LG, Smith RP, Chavez JE and Mosher MD: Identification
of increased nitric oxide biosynthesis during pregnancy in rats.
FASEB J. 7:566–571. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Akram SK, Sahlin L, Ostlund E, Hagenäs L,
Fried G and Söder O: Placental IGF-I, estrogen receptor, and
progesterone receptor expression, and maternal anthropometry in
growth-restricted pregnancies in the Swedish population. Horm Res
Paediatr. 75:131–137. 2011. View Article : Google Scholar
|
|
26
|
Fodor P, White B and Khan R:
Inflammation-The role of ATP in pre-eclampsia. Microcirculation.
2019.PubMed/NCBI
|
|
27
|
Kauma S, Takacs P, Scordalakes C, Walsh S,
Green K and Peng T: Increased endothelial monocyte chemoattractant
protein-1 and interleukin-8 in preeclampsia. Obstet Gynecol.
100:706–714. 2002.PubMed/NCBI
|
|
28
|
Blois S, Tometten M, Kandil J, Hagen E,
Klapp BF, Margni RA and Arck PC: Intercellular adhesion
molecule-1/LFA-1 cross talk is a proximate mediator capable of
disrupting immune integration and tolerance mechanism at the
feto-maternal interface in murine pregnancies. J Immunol.
174:1820–1829. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Springer TA: Traffic signals for
lymphocyte recirculation and leukocyte emigration: The multistep
paradigm. Cell. 76:301–314. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chaiworapongsa T, Romero R, Yoshimatsu J,
Espinoza J, Kim YM, Park K, Kalache K, Edwin S, Bujold E and Gomez
R: Soluble adhesion molecule profile in normal pregnancy and
pre-eclampsia. J Matern Fetal Neonatal Med. 12:19–27. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Oggé G, Romero R, Chaiworapongsa T,
Gervasi MT, Pacora P, Erez O, Kusanovic JP, Vaisbuch E, Mazaki-Tovi
S, Gotsch F, et al: Leukocytes of pregnant women with
small-for-gestational age neonates have a different phenotypic and
metabolic activity from those of women with preeclampsia. J Matern
Fetal Neonatal Med. 23:476–487. 2010. View Article : Google Scholar
|
|
32
|
Xu Z, Liu J, Jianxin C, Yongliang Z and
Pan X: 17β-Estradiol inhibits testosterone-induced cell
proliferation in HepG2 by modulating the relative ratios of 3
estrogen receptor isoforms to the androgen receptor. J Cell
Biochem. 119:8659–8671. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zou Y, Ding L, Coleman M and Wang Z:
Estrogen receptor-alpha (ER-alpha) suppresses expression of its
variant ER-alpha 36. FEBS Lett. 583:1368–1374. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cocciadiferro L, Miceli V, Granata OM and
Carruba G: Abstract 1852: Merlin/NF2 is associated with elevated
aromatase expression and estrogen formation in human liver tissues
and liver cancer cells: An unifying model for hepatocellular
carcinoma development and progression. Cancer Res. 1852:2015.
|
|
35
|
Liu J, Xu Z, Ma X, Huang B and Pan X: Role
of ER-α36 in breast cancer by typical xenoestrogens. Tumour Biol.
36:7355–7364. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Xu H, Wei Y, Zhang Y, Xu Y, Li F, Liu J,
Zhang W, Han X, Tan R and Shen P: Oestrogen attenuates tumour
progression in hepato-cellular carcinoma. J Pathol. 228:216–229.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang Z, Zhang X, Shen P, Loggie BW, Chang
Y and Deuel TF: Identification, cloning, and expression of human
estrogen receptor-alpha36, a novel variant of human estrogen
receptor-alpha66. Biochem Biophys Res Commun. 336:1023–1027. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rao Y and Su J: Insights into the
antiviral immunity against grass carp (Ctenopharyngodon idella)
reovirus (GCRV) in grass carp. J Immunol Res. 2015:6704372015.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Meng M, Wang H, Li Z, Guo M and Hou L:
Protective effects of polysaccharides from Cordyceps gunnii mycelia
against cyclophosphamide-induced immunosuppression to
TLR4/TRAF6/NF-κB signalling in BALB/c mice. Food Funct.
10:3262–3271. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang X, Han C, Qin J, Wei Y, Qian X, Bao Y
and Shi W: Pretreatment with salvia miltiorrhiza polysaccharides
protects from lipopolysaccharides/d-galactosamine-induced liver
injury in mice through inhibiting TLR4/MyD88 signaling pathway. J
Interferon Cytokine Res. 39:495–505. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Verstak B, Nagpal K, Bottomley SP,
Golenbock DT, Hertzog PJ and Mansell A: MyD88 adapter-like
(Mal)/TIRAP interaction with TRAF6 is critical for TLR2- and
TLR4-mediated NF-kappaB proinflammatory responses. J Biol Chem.
284:24192–24203. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shi S, Liang D, Chen Y, Xie Y, Wang Y,
Wang L, Wang Z and Qiao Z: Gx-50 reduces β-amyloid-induced TNF-α,
IL-1β, NO, and PGE2 expression and inhibits NF-κB signaling in a
mouse model of Alzheimer's disease. Eur J Immunol. 46:665–676.
2016. View Article : Google Scholar
|
|
43
|
Song J, Han X, Yao YL, Li YM, Zhang J,
Shao DY, Hou LS, Fan Y, Song SZ, Lian LH, et al: Acanthoic acid
suppresses lipin1/2 via TLR4 and IRAK4 signalling pathways in EtOH-
and lipopolysaccharide-induced hepatic lipogenesis. J Pharm
Pharmacol. 70:393–403. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Leon-Martinez D, Mulla MJ, Han CS, Chamley
LW and Abrahams VM: Modulation of trophoblast function by
concurrent hyperglycemia and antiphospholipid antibodies is in part
TLR4-dependent. Am J Reprod Immunol. 80:e130452018. View Article : Google Scholar : PubMed/NCBI
|