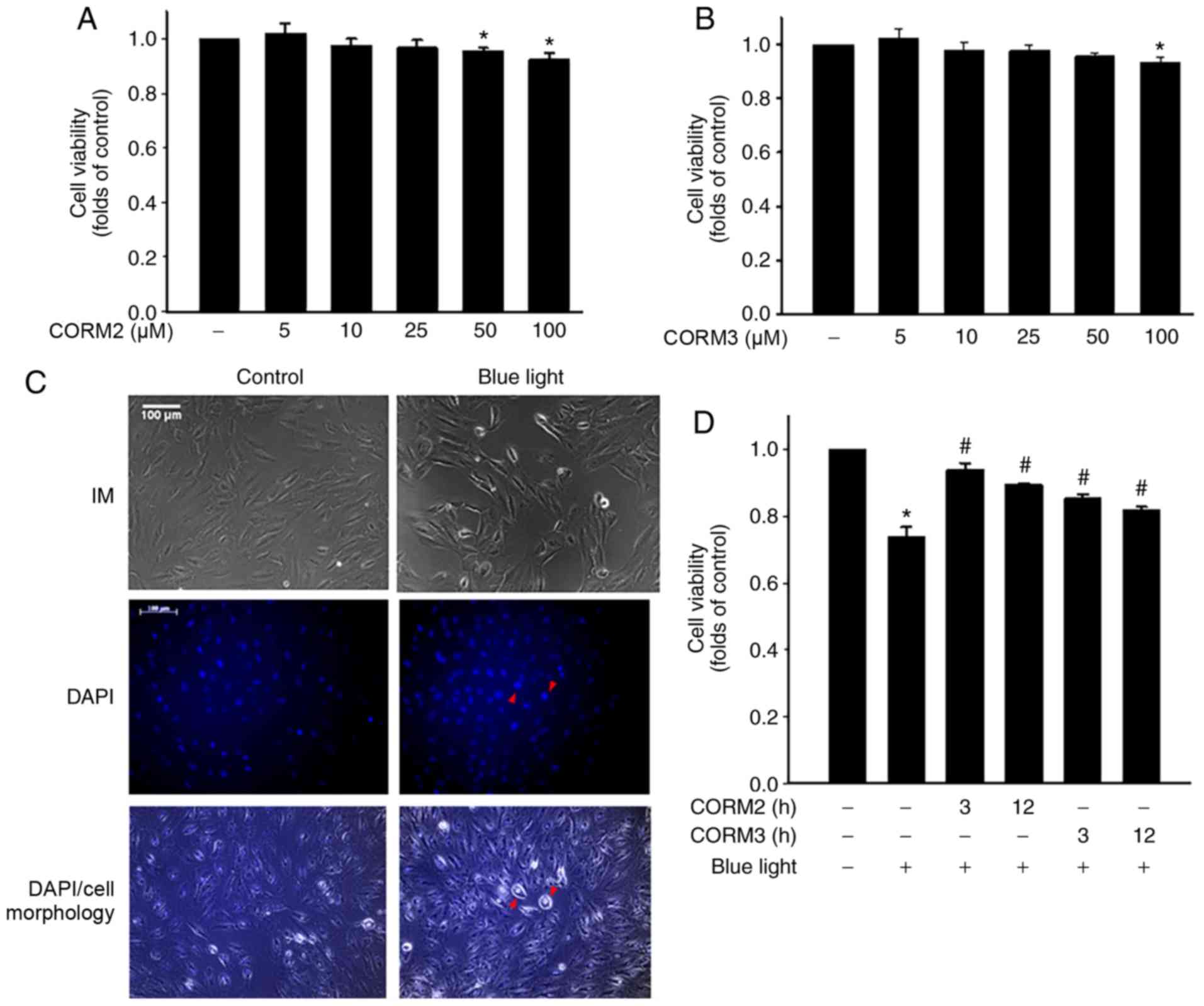
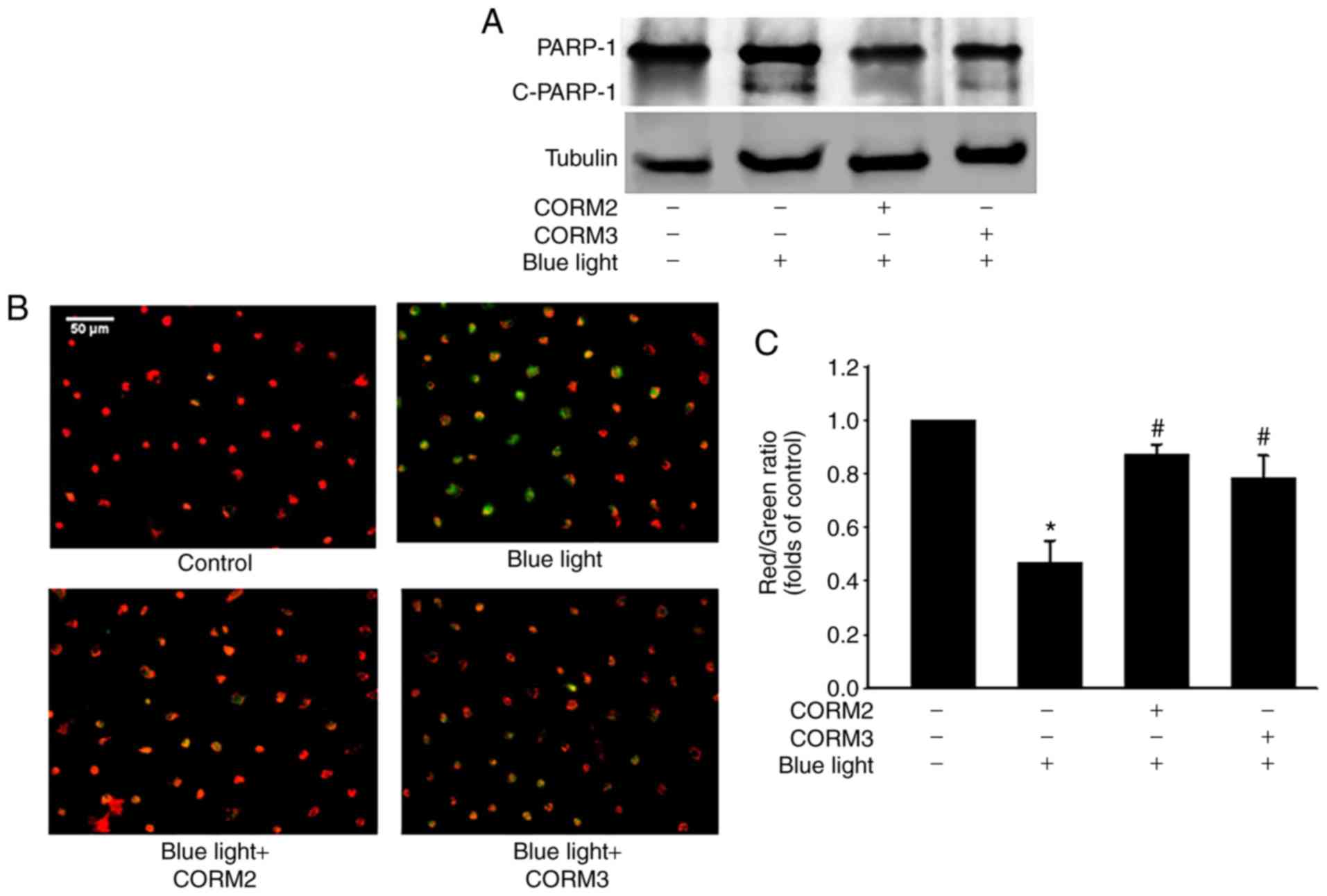
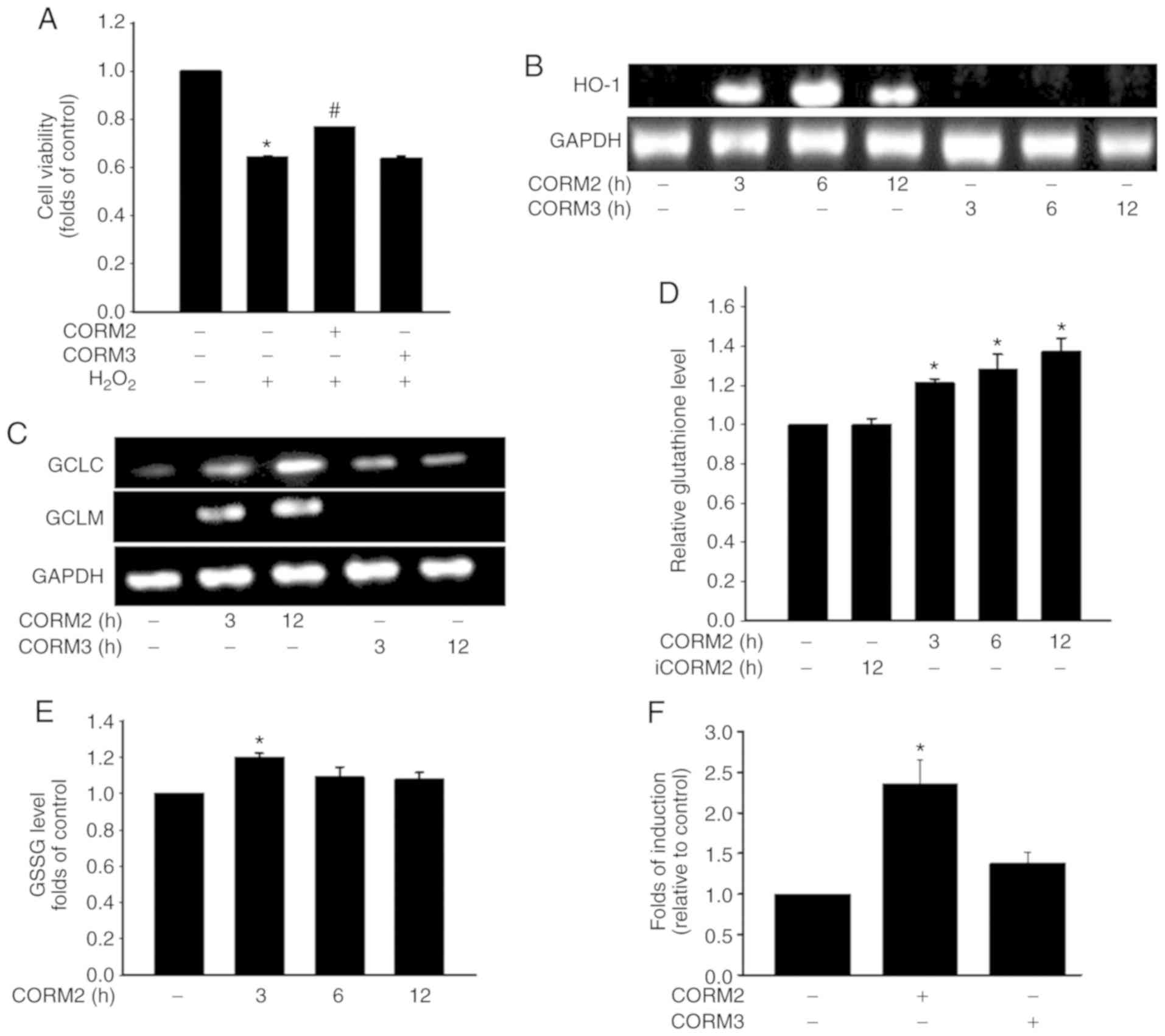
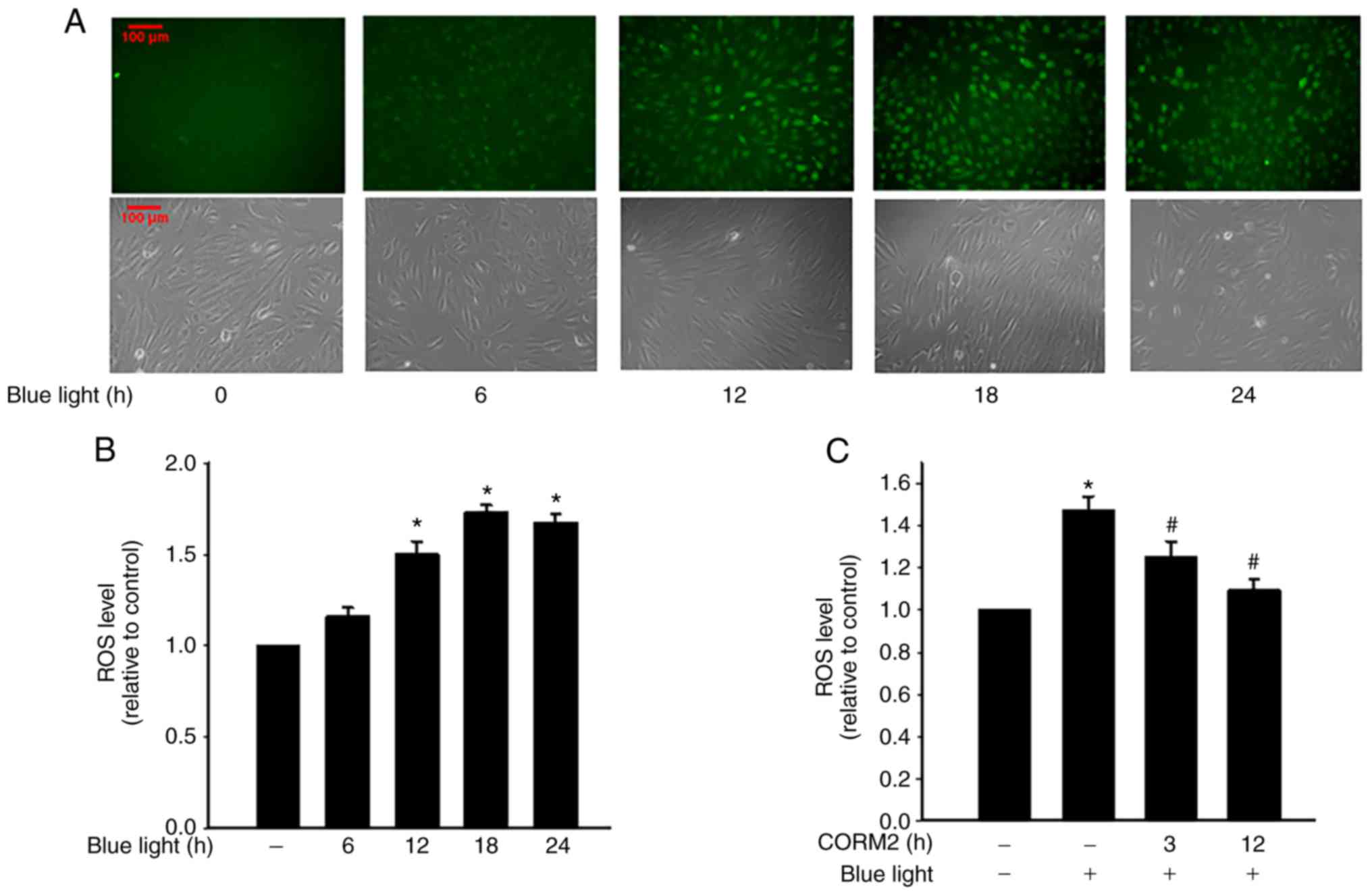
|
1
|
Kokotas H, Grigoriadou M and Petersen MB:
Age-related macular degeneration: Genetic and clinical findings.
Clin Chem Lab Med. 49:601–616. 2011. View Article : Google Scholar
|
|
2
|
Kent D and Sheridan C: Choroidal
neovascularization: A wound healing perspective. Mol Vis.
9:747–755. 2003.
|
|
3
|
Vicente-Tejedor J, Marchena M, Ramírez L,
García-Ayuso D, Gómez-Vicente V, Sánchez-Ramos C, de la Villa P and
Germain F: Removal of the blue component of light significantly
decreases retinal damage after high intensity exposure. PLoS One.
13:e01942182018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Strauss O: The retinal pigment epithelium
in visual function. Physiol Rev. 85:845–81. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cousins SW, Espinosa-Heidmann DG and Csaky
KG: Monocyte activation in patients with age-related macular
degeneration: A biomarker of risk for choroidal neovascularization?
Arch Ophthalmol. 122:1013–1018. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang L, Froio RM, Sciuto TE, Dvorak AM,
Alon R and Luscinskas FW: ICAM-1 regulates neutrophil adhesion and
transcellular migration of TNF-alpha-activated vascular endothelium
under flow. Blood. 106:584–592. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ledebur HC and Parks TP: Transcriptional
regulation of the inter-cellular adhesion molecule-1 gene by
inflammatory cytokines in human endothelial cells. Essential roles
of a variant NF-kappaB site and p65 homodimers. J Biol Chem.
270:933–943. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tak PP and Firestein GS: NF-kappaB: A key
role in inflammatory diseases. J Clin Invest. 107:7–11. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nakanishi-Ueda T, Majima HJ, Watanabe K,
Ueda T, Indo HP, Suenaga S, Hisamitsu T, Ozawa T, Yasuhara H and
Koide R: Blue LED light exposure develops intracellular reactive
oxygen species, lipid peroxidation, and subsequent cellular
injuries in cultured bovine retinal pigment epithelial cells. Free
Radic Res. 47:774–780. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Itoh K, Chiba T, Takahashi S, Ishii T,
Igarashi K, Katoh Y, Oyake T, Hayashi N, Satoh K, Hatayama I, et
al: An Nrf2/small Maf heterodimer mediates the induction of phase
II detoxifying enzyme genes through antioxidant response elements.
Biochem Biophys Res Commun. 236:313–322. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Alam J, Stewart D, Touchard C, Boinapally
S, Choi AM and Cook JL: Nrf2, a Cap'n'Collar transcription factor,
regulates induction of the heme oxygenase-1 gene. J Biol Chem.
274:26071–26078. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McMahon M, Itoh K, Yamamoto M, Chanas SA,
Henderson CJ, McLellan LI, Wolf CR, Cavin C and Hayes JD: The
Cap'n'Collar basic leucine zipper transcription factor Nrf2 (NF-E2
p45-related factor 2) controls both constitutive and inducible
expression of intestinal detoxification and glutathione
biosynthetic enzymes. Cancer Res. 61:3299–3307. 2001.PubMed/NCBI
|
|
13
|
Townsend DM, Tew KD and Tapiero H: The
importance of glutathione in human disease. Biomed Pharmacother.
57:145–155. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim KM, Pae HO, Zheng M, Park R, Kim YM
and Chung HT: Carbon monoxide induces heme oxygenase-1 via
activation of protein kinase R-like endoplasmic reticulum kinase
and inhibits endothelial cell apoptosis triggered by endoplasmic
reticulum stress. Circ Res. 101:919–927. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Schwer CI, Mutschler M, Stoll P, Goebel U,
Humar M, Hoetzel A and Schmidt R: Carbon monoxide releasing
molecule-2 inhibits pancreatic stellate cell proliferation by
activating p38 mitogen-activated protein kinase/heme oxygenase-1
signaling. Mol Pharmacol. 77:660–669. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shin DY, Chung J, Joe Y, Pae HO, Chang KC,
Cho GJ, Wolf CR, Cavin C and Hayes JD: Pretreatment with
CO-releasing molecules suppresses hepcidin expression during
inflammation and endoplasmic reticulum stress through inhibition of
the STAT3 and CREBH pathways. Blood. 119:2523–2532. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ryter SW, Alam J and Choi AM: Heme
oxygenase-1/carbon monoxide: From basic science to therapeutic
applications. Physiol Rev. 86:583–650. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bannenberg GL and Vieira HL: Therapeutic
applications of the gaseous mediators carbon monoxide and hydrogen
sulfide. Expert Opin Ther Pat. 19:663–682. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Queiroga CS, Vercelli A and Vieira HL:
Carbon monoxide and the CNS: Challenges and achievements. Br J
Pharmacol. 172:1533–1545. 2015. View Article : Google Scholar :
|
|
20
|
Lian KC, Chuang JJ, Hsieh CW, Wung BS,
Huang GD, Jian TY and Sun YW: Dual mechanisms of NF-kappaB
inhibition in carnosol-treated endothelial cells. Toxicol Appl
Pharmacol. 245:21–35. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kamencic H, Lyon A, Paterson P and
Juurlink BH: Monochlorobimane fluorometric method to measure tissue
glutathione. Anal Biochem. 286:35–37. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Braut-Boucher F, Pichon J, Rat P, Adolphe
M, Aubery M and Font J: A non-isotopic, highly sensitive,
fluorimetric, cell-cell adhesion microplate assay using calcein
AM-labeled lymphocytes. J Immunol Methods. 178:41–51. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yang D, Elner SG, Bian ZM, Till GO, Petty
HR and Elner VM: Pro-inflammatory cytokines increase reactive
oxygen species through mitochondria and NADPH oxidase in cultured
RPE cells. Exp Eye Res. 85:462–472. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sparrow JR, Hicks D and Hamel CP: The
retinal pigment epithelium in health and disease. Curr Mol Med.
10:802–823. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Pavan B and Dalpiaz A: Retinal pigment
epithelial cells as a therapeutic tool and target against
retinopathies. Drug Discov Today. 23:1672–1679. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
King A, Gottlieb E, Brooks DG, Murphy MP
and Dunaief JL: Mitochondria-derived reactive oxygen species
mediate blue light-induced death of retinal pigment epithelial
cells. Photochem Photobiol. 79:470–475. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Choi YK, Kim CK, Lee H, Jeoung D, Ha KS,
Kwon YG, Kim KW and Kim YM: Carbon monoxide promotes VEGF
expression by increasing HIF-1alpha protein level via two distinct
mechanisms, translational activation and stabilization of
HIF-1alpha protein. J Biol Chem. 285:32116–32125. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Samiec PS, Drews-Botsch C, Flagg EW, Kurtz
JC, Sternberg P Jr, Reed RL and Jones DP: Glutathione in human
plasma: Decline in association with aging, age-related macular
degeneration, and diabetes. Free Radic Biol Med. 24:699–704. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang PM, Wu ZZ, Zhang YQ and Wung BS:
Lycopene inhibits ICAM-1 expression and NF-κB activation by
Nrf2-regulated cell redox state in human retinal pigment epithelial
cells. Life Sci. 155:94–101. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mandal MN, Patlolla JM, Zheng L, Agbaga
MP, Tran JT, Wicker L, Kasus-Jacobi A, Elliott MH, Rao CV and
Anderson RE: Curcumin protects retinal cells from light-and oxidant
stress-induced cell death. Free Radic Biol Med. 46:672–679. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liao BC, Hsieh CW, Liu YC, Tzeng TT, Sun
YW and Wung BS: Cinnamaldehyde inhibits the tumor necrosis
factor-alpha-induced expression of cell adhesion molecules in
endothelial cells by suppressing NF-kappaB activation. Effects upon
IkappaB and Nrf2. Toxicol Appl Pharmacol. 229:161–171. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yeh PY, Li CY, Hsieh CW, Yang YC, Yang PM
and Wung BS: CO-releasing molecules and increased heme oxygenase-1
induce protein S-glutathionylation to modulate NF-kappaB activity
in endothelial cells. Free Radic Biol Med. 70:1–13. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hernández-Zimbrón LF, Zamora-Alvarado R,
Ochoa-De la Paz L, Velez-Montoya R, Zenteno E, Gulias-Cañizo R,
Quiroz-Mercado H and Gonzalez-Salinas R: Age-related macular
degeneration: New paradigms for treatment and management of AMD.
Oxid Med Cell Longev. 2018:83746472018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Martin DF, Maguire MG, Ying GS, Grunwald
JE, Fine SL and Jaffe GJ: Ranibizumab and bevacizumab for
neovascular age-related macular degeneration. N Engl J Med.
364:1897–1908. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ahmad S, Hewett PW, Fujisawa T, Sissaoui
S, Cai M, Gueron G, Al-Ani B, Cudmore M, Ahmed SF, Wong MK, et al:
Carbon monoxide inhibits sprouting angiogenesis and vascular
endo-thelial growth factor receptor-2 phosphorylation. Thromb
Haemost. 113:329–337. 2015. View Article : Google Scholar
|
|
36
|
Yang PM, Huang YT, Zhang YQ, Hsieh CW and
Wung BS: Carbon monoxide releasing molecule induces endothelial
nitric oxide synthase activation through a calcium and
phosphati-dylinositol 3-kinase/Akt mechanism. Vascul Pharmacol.
87:209–218. 2016. View Article : Google Scholar : PubMed/NCBI
|