The innate immune response, often known as
non-specific immunity, is the body's first line of defense
(1) and recognizes
pathogen-associated molecular patterns (PAMPs) and host-derived
danger-associated molecular patterns (DAMPs) via pattern
recognition receptors (PRRs) (2).
NOD-like receptors (NLRs), which belong to the evolutionarily well
conserved PRR family are located in the cytoplasm. By recruiting
downstream adaptor proteins, they can form inflammasome complexes
that promote the maturation and secretion of inflammatory
mediators, including interleukin (IL)-1β and IL-18, resulting in
inflammatory reactions. A total of five major inflammasomes
currently exist, i.e., NLRP1, NLRP3, NLRC4, NLRP6 and absent in
melanoma 2 (AIM2), which are activated to promote
inflammasome-dependent immune responses when they recognize PAMPs
and DAMPs from pathogenic microorganisms (3). The activation of the majority of
inflammasomes is dependent on a few highly specific agonists;
however, the NLRP3 inflammasome can be activated by various
unrelated stimuli, including K+, Cl−,
Ca2+, lysosomal destruction, mitochondrial dysfunction
and metabolic alterations (4).
Additionally, inflammasomes are activated when PAMP receptors,
including Toll-like receptors, recognize their ligands. Concomitant
with the cleavage of IL-1β and IL-18, gasdermin D (GSDMD) is
cleaved by activated caspase-1, resulting in a lytic-regulated cell
death mode, termed pyroptosis. Upon cleavage, the N-terminus of
GSDMD binds to membrane lipids and forms micropores, causing cell
rupture and the occurrence of an inflammatory cascade (5). Accordingly, apart from being crucial
to the resistance to pathogen invasion, the NLRP3 inflammasome
modulates inflammation (6). There
is increasing evidence to suggest that inflammatory diseases can be
treated more effectively by targeting the NLRP3 inflammasome
(7), including atherosclerosis
(8), ischemic stroke (9), Alzheimer's disease (AD) (10), diabetes mellitus (DM) (11) and inflammatory bowel disease (IBD)
(12). A therapeutic strategy for
inflammatory disorders focuses on recombinant cytokine receptor
antagonists and neutralizing antibodies targeting the IL-1 family
(7). Nonetheless, there is an
increased risk of infection associated with cytokine therapy.
Inhibitors targeting the NLRP3 inflammasome pathway rather than
effector molecules currently exhibit desired prevention or
therapeutic effects in animal models of inflammatory diseases, as
discussed below. In the present review, systematic searches in the
title, key words and abstract of articles were performed using the
PubMed and Web of Science databases with 'NLRP3 inhibitor(s)' and
'inflammatory disease(s)' as key words at initial retrieval. By
browsing the literature from 2012 to 2022, 100 articles were
retrieved, of which 35 were excluded as they were reviews,
editorials, retracted, or unavailable online, and 65 articles were
included. NLRP3 inhibitors were shown to relieve inflammatory
diseases. Therefore, when discussing specific inflammatory
diseases, the present review included further literature by
combining 'Atherosclerosis', 'ischemic stroke', 'Alzheimer's
diseaseʼ, 'Diabetes mellitus' and 'Inflammatory bowel disease',
respectively with 'NLRP3 inhibitor(s)' as key words. According to
the retrieved literature, a brief review of studies on LRP3
inflammasome inhibitors is presented herein, in an aim to aid the
development of NLRP3 inflammasome-related disease drugs.
Due to inflammatory stimuli, the NLRP3 inflammasome
is predominantly found in immune and inflammatory cells (13,14), and is equipped with NLRP3, the
adaptor protein apoptosis-associated speck-like protein (ASC) and
pro-caspase-1 (15). The NLRP3
protein comprises three main components, i.e., a leucine-rich
repeat (LRR), a central nucleotide-binding oligomerization domain
(NOD), also known as NACHT, in the carboxy terminus, and a pyrin
domain (PYD) in the amino-terminal. Similar to Toll-like receptor
(TLR), LRR recognizes and binds PAMP or DAMP stimuli; PYD is the
functional region connecting downstream bridging proteins to
effector molecules; NOD is the core part of NLRs that undergoes
oligomerization when the LRR recognizes and binds PAMPs or DAMPs
and exerts adenosine triphosphate (ATPase) activity for the
self-association and function of NLRP3 (4).
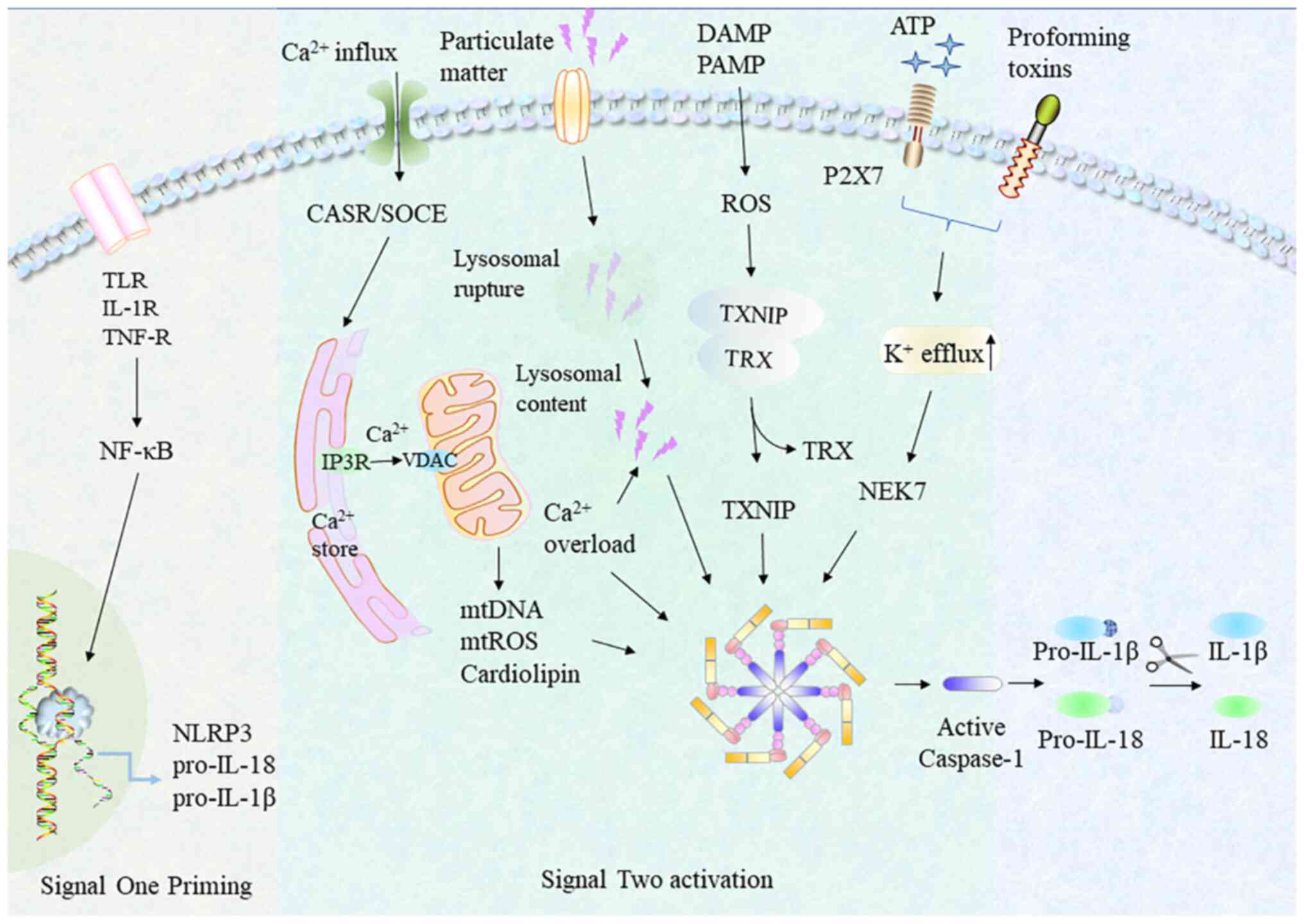
NLRP3 inflammasome activation is tightly regulated,
due to a two-step process known as priming and assembly (16) (Fig.
1). The priming step, indicated as 'the first signal', is
initiated by TLR and nuclear factor-κB (NF-κB) to increase the
intracellular transcript levels of pro-IL-1β, pro-IL-18 and NLRP3
(17,18). Once primed, subsequent NLRP3
inflammasome activation by NLRP3 oligomerization and the later
NLRP3 inflammasome assembly is termed 'the second signal' (19).
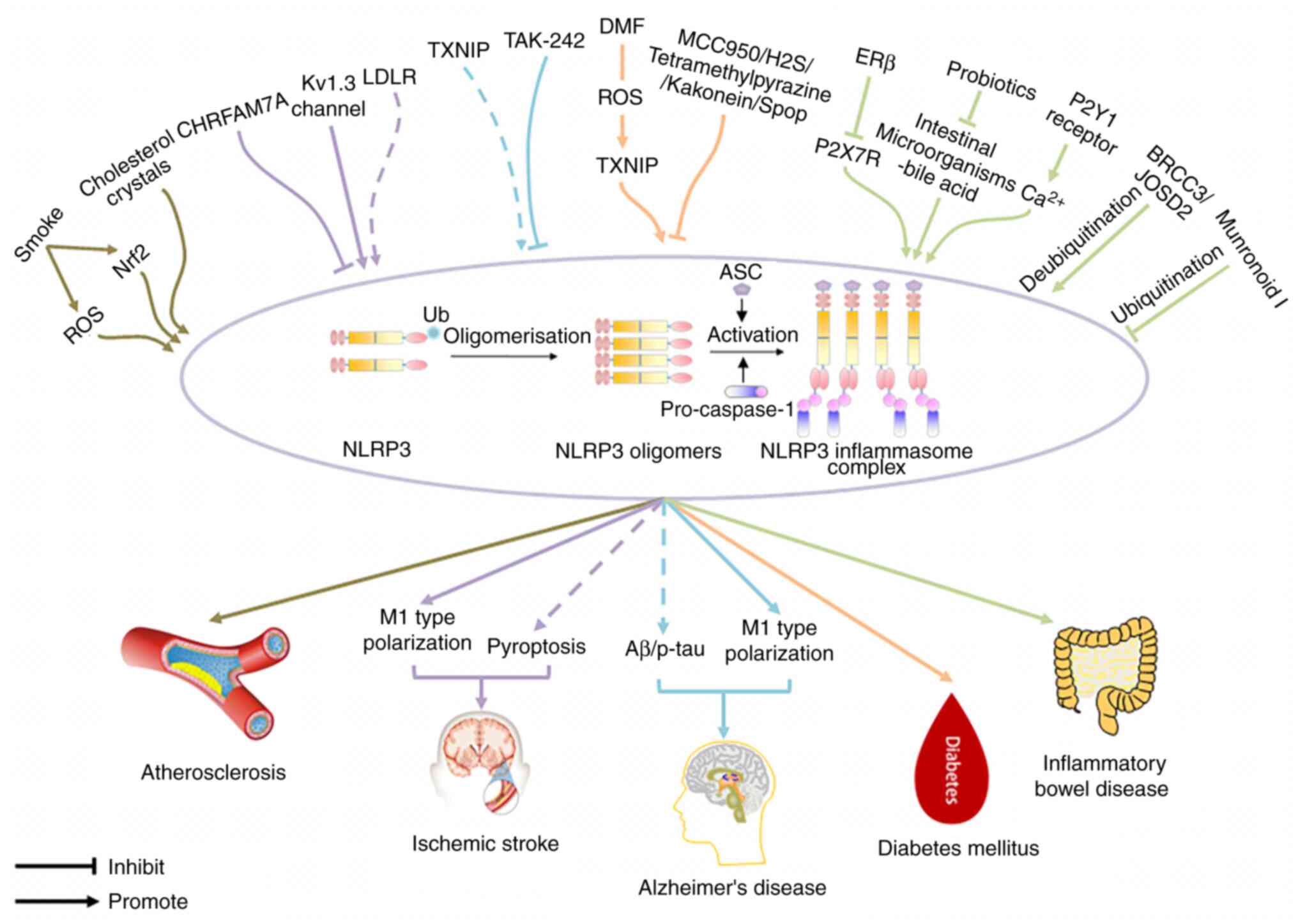
The NLRP3 inflammasome is crucial for innate
immunity; however, its aberrant activation promotes various
inflammatory disorders, including atherosclerosis, ischemic stroke,
AD, DM and IBD (Fig. 2).
Cardiovascular disease (CVD) remains the leading
cause of morbidity and mortality across the globe (35). Ischemic CVD is largely caused by
atherosclerosis, a chronic inflammatory disease of the arterial
wall caused by lipids (36). In
patients with atherosclerosis, the NLRP3 inflammasome is highly
expressed in the aorta (37).
NLRP3 activation significantly increases macrophage lipid
deposition susceptibility and migration capacity, hence promoting
atherosclerosis. In advanced atherosclerosis, the NLRP3
inflammasome is crucial for necrotic core formation, and its
silencing increases plaque stability (38). However, other researchers have
observed opposite effects. NLRP3 also promotes the proliferation
and migration of vascular smooth muscle cells (VSMCs) in vessels
(39), which may contribute to
vascular remodeling and plaque stability. Smoking affects the
stages of atherosclerosis, and is hence one of the major
independent risk factors. Cigarette smoke extracts impair the
cardiovascular system in vitro by activating the nuclear
factor erythroid2-related factor 2 pathway and inducing ROS
generation, hence activating the NLRP3 inflammasome (40,41). Furthermore, cigarette smoke
condensate induces THP-1 monocyte differentiation into macrophages
(42), which, in combination with
a high-fat diet (HFD), exert a synergistic promoting effect on
atherosclerosis (43). During
atherogenesis, the formation of cholesterol crystals in the vessel
wall initiates plaque inflammation by activating the NLRP3
inflammasome during atherogenesis (44). Similarly, the NLRP3 inflammasome
is involved in hyperglycemia-induced endothelial inflammation and
diabetes-related atherosclerosis (45).
Ischemic stroke is caused by cerebral ischemia,
which eventually causes lifelong disability or mortality (46). It is characterized by an
inflammatory response responsible for its pathophysiology (47). Neuroinflammation due to ischemic
stroke is controlled by microglia, which are categorized into
M1-like (pro-inflammatory) and M2-like (anti-inflammatory
pro-regenerative) phenotypes (48). An increased number of M1 microglia
is caused by dysregulated microglia polarization dynamics,
resulting in post-stroke injury expansion (49,50). The NLRP3 inflammasome promotes the
development of ischemic stroke, primarily by promoting microglial
polarization. For instance, ischemic stroke increases NLRP3
inflammasome expression and activation (51). Liu et al (52) and Zhao et al (53) found that the NLRP3 inflammasome
was activated in the microglia and astrocytes affected by cerebral
ischemia/reperfusion injury (CIRI). The Kv1.3 channel, a
transmembrane protein, is involved in the production of
inflammatory cytokines and ROS (49), and even promotes neuronal death
(54). Cerebral ischemic injury
is alleviated by inhibiting Kv1.3 channels, which may be related to
the remodeling of the microglial phenotypic response from M1 to M2
as well as inhibiting NLRP3 inflammasome activation and IL-1β
release (49). In addition, the
injection of salvianolic acids has been found to generate similar
effects, altering the microglial phenotype from M1 to M2 by
suppressing the pyroptosis mediated by the NLRP3 inflammasome
(55). CHRFAM7A, a
dominant-negative inhibitor of α7 nicotinic acetylcholine receptor
(α7nAChR, coded by CHRNA7), causes brain disorders (56). CHRFAM7A overexpression attenuates
CIRI by inhibiting microglial pyroptosis via the NLRP3/caspase-1
pathway and promoting M2 microglial polarization (57). In addition to microglia, NLRP3
expression is upregulated in endothelial cells and neurons
following stroke (58,59). Low-density lipoprotein (LDL)
receptors (LDLRs) regulate cholesterol uptake and exhibit
anti-inflammatory properties (47). Research indicates that LDLRs play
a modulatory role in LRP3-mediated neuronal pyroptosis and
inflammation following ischemic stroke (47). Furthermore, LDLR knockout
increases caspase-1/GSDMD expression, resulting in severe neuronal
pyroptosis (47). By contrast,
opposite findings have also been reported, demonstrating that
ischemic brain injury is reduced in ASC−/−,
AIM2−/− and NLRC4−/− mice and not in mice
deficient for the canonical sensor of sterile injury, NLRP3
(60).
AD is the most prevalent type of dementia among the
elderly, characterized by hyper-phosphorylated tau protein and Aβ
accumulation (61). Moreover,
numerous inflammatory markers are present in the AD-affected brain,
including inflammatory cytokines and chemokines (62). Senile plaques activate microglia,
contributing to cerebral neuroinflammation, which is termed the
third core pathological characteristic of AD (63). TLR4 functions as a 'priming'
signal for the NLRP3 inflammasome activation (64), unlike TLR4, whose inhibitor
(TAK-242) provides neuroprotection and promotes microglial M2-like
phenotype in AD (65). Trained
microglia respond to subsequent unspecific stimuli in an enhanced
manner and microglial training is a major pro-AD factor, augmenting
the subsequent inflammatory response (66). In a previous study, in mice with
sporadic AD injected with streptozotocin, microglial training
worsened Aβ accumulation, neuronal loss and cognitive impairment,
effects which were attenuated by the microglial NLRP3 inhibitor
(66). Moreover, increased ER
stress has been observed in AD (67). TXNIP, an endogenous inhibitor of
thioredoxin, is a key antioxidant reductive protein and
anti-apoptotic protein (66),
which may also represent a connection between ER stress and
neuroinflammation (67).
According to a previous study, using double immunofluorescence
staining, TXNIP and IL-1β were shown to be co-localized near Aβ
plaques and p-tau (68). TXNIP
also directly interacts with the NLRP3 inflammasome in AD-affected
brains, modulating inflammatory cascades. Therefore, inhibiting the
NLRP3 inflammasome activation may help to control AD. However, Tang
and Harte (69) indicated that
the levels of NLRP3 activation markers were not significantly
altered in in the temporal cortex of patients with AD, and in age-
and sex-matched controls.
DM is a prevalent metabolic disorder characterized
by hyperglycemia, marked by a chronic state of low-grade
inflammation (70); it is a
highly prevalent disease with high morbidity and mortality rates
(71). Several common clinical
complications of DM have been reported, including CVD, stroke,
diabetic nephropathy and diabetic retinopathy (72,73), which are all closely associated
with NLRP3 inflammasome activation (74). Additionally, there is evidence to
indicate that hyperglycemic conditions cause endothelial cell
dysfunction and NLRP3 inflammasome activation (75). MCC950, an NLRP3 inhibitor
(11), tetramethylpyrazine
(76), hydrogen sulfide (77) and Kakonein (78) have been shown to improve
endothelial dysfunction by suppressing NLRP3 inflammasome
activation or the production of its effectors, caspase-1 and IL-1β.
Moreover, MCC950 targets NLRP3-mediated inflammation, and reduces
plaque development, promotes plaque stability and improves
diabetes-associated vascular disease (79). Similarly, MCC950 is a promising
treatment that prevents neurovascular remodeling and cognitive
impairment in diabetic patients following stroke (80). Dimethyl fumarate exerts
vasculoprotective effects on diabetic aortas by suppressing the
activation of the ROS/TXNIP/NLRP3 inflammasome pathway (81). Furthermore, NLRP3 inflammasome
activation has been shown to exacerbate cardiac dysfunction
following ischemic stroke in diabetic mice (82). By contrast, sodium-glucose
cotransporter 2 inhibitor exerts cardioprotective effects by
suppressing the NLRP3 inflammasome (83). Differing from NLRP3, IL-1β has a
more complex effect on systemic glucose metabolism. It has been
shown that IL-1β contributes to the postprandial stimulation of
insulin secretion (84).
Moreover, the deletion of IL-1R impairs glucose tolerance and
reduces the insulin-positive area in pancreatic tissue of db/db and
C57BL/6 mice (85).
Diabetic nephropathy is a prevalent complication of
DM and a major cause of end-stage renal disease. The inflammatory
response induced by NLRP3 inflammasome activation modulates the
pathological process of diabetic nephropathy (86). Curcumin, a principal and most
active curcuminoid (87),
attenuates the progression of diabetic nephropathy by limiting the
activation of the NLRP3 inflammasome (88). Similarly, the E3 ubiquitin ligase
speckle BTB-POZ protein, a suppressor of the NLRP3 inflammasome,
promotes NLRP3 degradation by improving the K48-linked
polyubiquitin of NLRP3, thereby suppressing renal dysfunction and
pathological changes to ameliorate diabetic nephropathy (89). Moreover, the NLRP3 inflammasome
may promote pathological neovascularization in the advanced stages
of diabetic retinopathy (90). Li
et al (91) discovered
that quercetin, a bioactive flavonoid pigment in several fruits,
had therapeutic potential in diabetic retinopathy-associated
retinal neovascularization by suppressing the NLRP3 inflammasome.
Furthermore, isoflurane pre-treatment has been shown to inhibit
NLRP3 inflammasome activation in the retina and provide substantial
retinal protection against retinal injury induced by stroke
associated with DM (92). Taken
together, these findings demonstrate that NLRP3 inflammasome
participates in the development and progression of DM and its
related complications.
IBD is an idiopathic disease of the gut
characterized by chronic, recurrent inflammation (93). Its pathogenesis is directly
associated with changes in the immune environment (94,95). It has been demonstrated that the
NLRP3 inflammasome in childhood IBD may be involved in the
regulation of immune mechanisms by upregulating caspase-1 and IL-1β
expression (96). There is
evidence to suggest that the NLRP3 inflammasome is persistently
activated and plays a key role in IBD (97). Consequently, it is a potential
therapeutic target for the treatment of IBD. Adenosine diphosphate,
which is abundant in injured colonic tissue, activates the NLRP3
inflammasome by regulating P2Y1 receptor-mediated Ca2+
signaling, resulting in the maturation and secretion of IL-1β,
further aggravating the progression of colitis (98). On the other hand, dextran sulfate
sodium salt (DSS)-induced colitis and endotoxic shock have been
shown to be significantly ameliorated by genetic ablation or the
pharmacological blockade of the P2Y1 receptor (98). Additionally, the
BRCA1/BRCA2-containing complex 3 and Josephin domain containing 2
mediate NLRP3-R779C deubiquitination (99) and the interaction between NEK7 and
NLRP3 (100), both of which
promote NLRP3 inflammasome activation and an increased risk of IBD.
Munronoid I is a novel diterpenoid isolated and purified from the
Meliaceae family. In mice with DSS-induced IBD, NLRP3 has been
found to be ubiquitinated and degraded to regulate canonical
pyroptosis (101). Moreover,
estrogen receptor β is a crucial anti-inflammatory agent in rats
with IBD, related to P2X7R downregulation, the inhibition of NLRP3
inflammasome activation, as well as the release of IL-1β from
macrophages via the JAK2/STAT3 signaling pathway (102). A disrupted intestinal microbiota
is also a feature of IBD (103).
Notably, probiotics alleviate IBD by modulating the intestinal
microorganisms-bile acid-NLRP3 inflammasome pathway (104).
Currently available clinical treatment agents for
NLRP3-related diseases include drugs targeting IL-1β, including
anakinra, canakinumab and rilonacept (105). However, there are some concerns
that these treatments may increase the risks of infection (106). Therefore, inhibitors targeting
the NLRP3 inflammasome may be more effective than those targeting
IL-1β in the treatment of NLRP3-driven diseases (12). In recent years, researchers have
suppressed the NLRP3 inflammasome through various targets by
exploiting their complex signaling pathways, including the priming
step of NLRP3 inflammasome activation, the content of
K+, Ca2+, Cl− and ROS in the
microenvironment, the assembly of NLRP3 the inflammasome and GSDMD
cleavage. As such, the present review summarizes recent inhibitors
of the NLRP3 inflammasome pathway and their roles in inflammatory
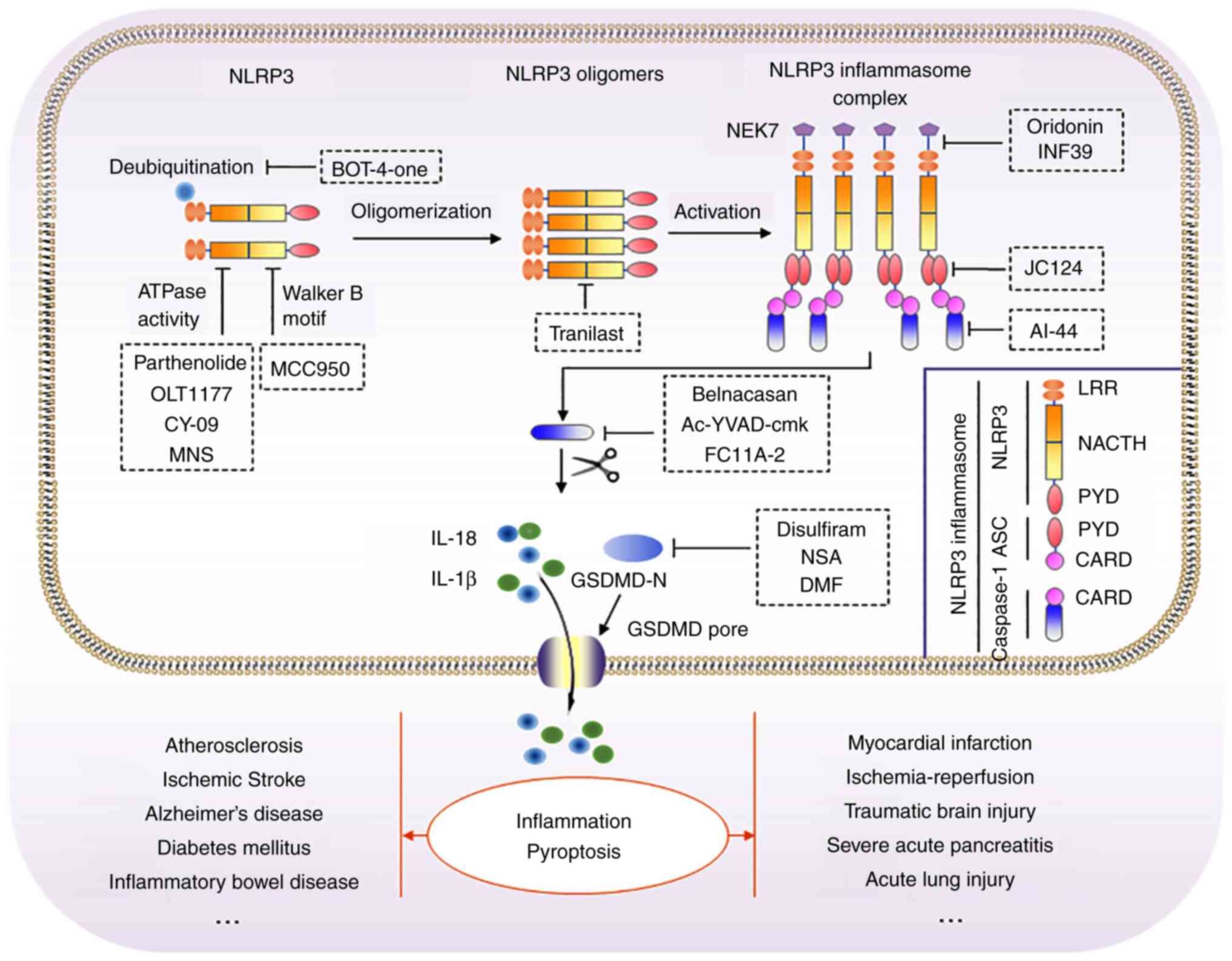
diseases (Table I and Fig. 3).
LPS, oxidized LDL (ox-LDL) and cholesterol are
recognized by TLR4 and IL-1R to mediate NF-κB entry and upregulated
the expression of pro-caspase-1, NLRP3, pro-IL-1β and pro-IL-18,
which is defined as the priming step.
TLRs are a type of transmembrane protein, which can
be combined with a corresponding ligand to trigger intracellular
signal transduction cascade responses, hence stimulating chemokines
and proinflammatory cytokines (107). As the PRR, TLR4 regulates
neuroinflammation. In the priming step of NLRP3 inflammasome
activation, TLR4 signals are activated by LPS via myeloid
differentiation primary response 88 (MyD88), which ultimately
activates NF-κB, thereby upregulating pro-IL-1β, pro-IL-18 and
NLRP3 expression (108,109). Therefore, it is possible to
develop chronic/sustained inflammation caused by a vicious circle
of NLRP3 inflammasome activation via TLR4 signaling (110). Consequently, the development of
small molecule pharmacological antagonists for TLR4 is a novel
molecular therapeutic approach. TAK-242, or resatorvid, is a TLR4
inhibitor that binds to the TIR domain of TLR4 and competes with
TLR4 interacting molecules, thereby suppressing the TLR4-mediated
release of several cytokines (111). TAK-242 penetrates the
blood-brain barrier (BBB) and is an effective inhibitor of
congenital inflammation (112),
as well as neuroinflammation (112-114). TAK-242 inhibits the TLR4/NLRP3
inflammasome signaling pathway induced by Aβ in microglia (115). A similar mechanism is adopted by
TAK-242 to provide neuroprotection and promote M2 microglial
polarization by suppressing the TLR4/MyD88/NF-κB/NLRP3 signaling
pathway (65). Moreover, a HFD
exacerbates the extent and severity of acute pancreatitis via the
inflammatory response. The inhibition of TLR4 signaling by TAK-242
decreases inflammatory reaction, exerting a protective effect
during acute pancreatitis in HFD rats (116). Moreover, TAK-242 improves
symptoms of myocardial infarction (MI) (117), periodontitis (118), renal/retinal lesions (107,119), ischemia-reperfusion and acute
lung injury by inhibiting TLR4 and its downstream inflammatory
markers (120).
BAY 11-7082 is an NF-κB inhibitor that targets the
phosphorylation of IκBα (inhibitor of NF-κB) (121). BAY 11-7082 suppresses the
phosphorylation of IκBα and NF-κB translocation to the nucleus
induced by TNF-α, thereby suppressing NLRP3 inflammasome activation
(122). Following oxygen-glucose
deprivation and re-oxygenation, BAY 11-7082 decreases the levels of
the NLRP3 inflammasome and cleaved caspase-1 protein in BV2
microglial cells, presenting a pharmacological effect in stroke
(123). Moreover, chronic cold
stress activates the microglia, causing neuroinflammation that can
be significantly inhibited by BAY 11-7082 by targeting the
GABA-induced NLRP3 inflammasome (124). Sulfasalazine is a drug used in
the treatment of rheumatoid arthritis and ulcerative colitis. It
can also inhibit NF-κB activity (125). Sulfasalazine significantly
inhibits NF-κB expression to dose-dependently ameliorate acetic
acid-induced inflammasome activation by reducing NLRP3 and
caspase-1 expression, thereby reducing ulcerative colitis in rats
(126). Moreover, the
therapeutic administration of sulfasalazine effectively
downregulates NF-κB activation, as well as IL-1β and IL-8 mRNA
expression, whereas IκBα levels have been shown to be stable in
biopsy specimens from patients with ulcerative colitis (125). Analogous data were also obtained
when sulfasalazine was used to attenuate oxazolone-induced
ulcerative colitis in mice (127).
NLRP3 inflammasome complex formation is dependent on
NLRP3 oligomerization and the recruitment of ASC to NLRP3 oligomers
(136) (Fig. 3). NLRP3 is oligomerized by the
ATPase activity of NLRP3 NACHT domain to recruit and oligomerize
ASC, hence activating caspase-1 (137,138). Consequently, NLRP3
inflammasome-specific inhibitors targeting NLRP3 are considered
attractive targets (Table I).
MCC950 (CP-456,773, CRID3), an inhibitor of the
NLRP3 inflammasome, has demonstrated excellent in vivo
efficacy in several species and disease models. There is ample
evidence to suggest that MCC950 inhibits ATP hydrolysis, ASC
oligomerization and chloride efflux by directly interacting with
the Walker B motif of the NLRP3 NACHT domain, thereby suppressing
NLRP3 inflammasome activation (139). However, its inhibitory effects
are independent of NLRP3 inflammasome priming, calcium signaling,
potassium efflux, mitochondrial respiration, ROS production,
NLRP3-NLRP3, NLRP3-ASC and NEK7-NLRP3 interaction (140-142). It has been demonstrated that
MCC950 is responsible for the treatment of inflammatory-based
diseases and their complications. For instance, MCC950 attenuates
macrophage inflammation and pyroptosis to prevent atherosclerosis
(8). Similarly, MCC950
significantly reduces plaque sizes in hyperlipidemic murine models,
suggesting that NLRP3 inhibitors may be candidates for the
treatment of atherosclerosis (143). The oral administration of MCC950
is a recently identified approach for reducing the severity of
spontaneous chronic colitis in Winnie mice (144) and for suppressing human retinal
endothelial cell dysfunction for the treatment of diabetic
retinopathy induced by high glucose conditions (145). Furthermore, NLRP3 inflammasome
activation in neurons mediates neuroinflammation in acute ischemic
stroke, whereas MCC950 reduces CIRI by mitigating inflammation and
preserving BBB integrity (146).
Similarly, MCC950 treatment significantly improves insulin
sensitivity to alleviate diabetic encephalopathy in db/db mice
(147). Additionally, MCC950
ameliorates diabetic kidney injury in db/db mice by decreasing the
fibrosis markers in high glucose-induced mesangial cells to prevent
diabetic nephropathy progression (148). Due to its effects on
inflammation, MCC950 may be effective in treating such
disorders.
INF39 is a non-toxic, irreversible, specific
inhibitor of the NLRP3 inflammasome, which specifically inhibits
NLRP3 activation, but not the NLRC4 or AIM2 inflammasomes (162). However, INF39 affects neither
the upstream events of NLRP3 inflammasome activation, including
K+ efflux, ROS generation or mitochondrial membrane
potential, nor the downstream signal, GSDMD (162). The inhibition of NEK7-NLRP3
interaction is a major mechanism of the anti-inflammatory effects
of INF39, followed by the inhibition of NLRP3 oligomerization,
NLRP3-ASC, ASC oligomerization and speckle formation (162). According to bioluminescence
resonance energy transfer analyses, INF39 suppresses the release of
IL-1β from macrophages by directly interfering with NLRP3
activation (163). In rats with
type 2 DM (T2DM), INF39 has been shown to effectively suppress the
expression of ICAM-1, NLRP3, as well as other inflammatory factors,
and to reduce intimal-media thickness, as well as platelet
activation (76). INF39 also
promotes the effect of Arctigenin on DSS-induced acute colitis by
suppressing the NLRP3 inflammasome (164). Pellegrini et al (165) also demonstrated that directly
inhibiting NLRP3, reduced systemic and bowel inflammation more
effectively than inhibiting caspase-1 or IL-1 receptors.
Tranilast is a tryptophan metabolite that
suppresses NLRP3 inflammasome activation; it is also used in the
treatment of allergies and asthma, without affecting AIM2 or NLRC4
inflammasome activation (166).
Tranilast suppresses NLRP3 oligomerization by binding to the NACHT
domain and subsequent NLRP3 inflammasome assembly, caspase-1
activation and IL-1β production with no effects on its ATPase
activity, K+ efflux, mitochondrial damage, or
CL− efflux (166,167). In recent research, tranilast was
shown to inhibit NLRP3 oligomerization in an ATPase-independent
manner and exert profound treatment and preventive effects in mouse
models of gout, cryopyrin-associated periodic syndrome (CAPS), and
T2DM (28). According to Cao and
Peng (168), tranilast
ameliorated the symptoms of gestational DM, including
hyperglycemia, insulin deficiency, glucose intolerance and insulin
resistance by suppressing NLRP3 inflammasome activation, as well as
inflammatory responses. Furthermore, tranilast has been shown to
inhibit NLRP3 inflammasome activation by improving NLRP3
ubiquitination to reduce vascular inflammation, and ameliorate
atherosclerosis in both LDLR- and apolipoprotein E-deficient mice
(169).
JC124 is an active and selective NLRP3 inflammasome
inhibitor that targets ASC oligomerization in macrophages, and
constitutively expresses active NLRP3 inflammasome (171). A previous photoaffinity labeling
probe experiment indicated that JC124 directly targets NLRP3
inflammasome complex without changing its ATPase activity (172). Furthermore, JC124 targets the
NLRP3 inflammasome and exerts beneficial effects in APP/PS1 mice,
significantly decreasing Aβ accumulation and improving cognitive
function (173,174). Moreover, JC124 significantly
decreases the number of degenerating neurons, the inflammatory
response, and cortical lesion volume post-injury (172). In TBI, JC124 substantially
downregulates NLRP3, ASC, IL-1β, TNF-α, inducible nitric oxide
synthase and caspase-1 expression (172). SAR adds more information to the
JC124 structure, leading to the discovery of two novel lead
compounds, i.e.,14 and 17, with improved inhibitory potency
(175).
MNS, parthenolide and BOT-4-one impair NLRP3 ATPase
activity, thereby suppressing NLRP3 inflammasome activation. MNS
does not inhibit K+ efflux or influence NLRC4 or AIM2
inflammasome activation, suggesting that it specifically inhibits
NLRP3 inflammasome (176). Apart
from targeting NLRP3, parthenolide is a direct inhibitor of
caspase-1 protease activity (177). Moreover, BOT-4-one increases
NLRP3 ubiquitination and suppresses NLRP3 inflammasome activation
(178). Summarily, for NLRP3
inflammasome inhibitors targeting NLRP3 protein and NLRP3
inflammasome assembly, tranilast, CY-09 and BOT-4-one can only
suppress NLRP3 oligomerization by binding to the NACHT domain or
affect its ATPase activity; hence, they are NLRP3
inflammasome-specific inhibitors. Additionally, INF39, OLT1177,
oridonin and X-11-5-27 inhibit NLRP3 inflammasome assembly. Among
all the inhibitors, only parathenolide can inhibit NLRP3, NLRC4 and
AIM2, whereas others exhibit NLRP3 specificity.
As the protease that matures IL-1β, IL-18 and
GSDMD, caspase-1 is a key initial event at the onset of NLRP3
inflammasome activation and canonical caspase-1-dependent
pyroptosis (179). Therefore,
the pharmaceutical industry has focused on developing
clinical-grade molecules that suppress caspase protease
activity.
Belnacasan (VX-765), an efficient and selective
caspase-1 inhibitor, can hinder the development and progression of
atherosclerosis at least by targeting ox-LDL-induced VSMC
pyroptosis (180). Caspase-1
inhibition with VX-765 has been shown to significantly reduce
neuropathological damage and pyroptosis following prolonged
ketamine exposure (181). It has
also been shown that VX-765 significantly attenuates cerebral
ischemic injury and cerebral edema, as well as reduces
ischemia-associated BBB permeability in rats sujected to middle
cerebral artery occlusion by suppressing pyroptosis and the
RAGE/MAPK pathway (182).
Similarly, it has been shown that VX-765 not only attenuates brain
injury, but also suppresses microglial pyroptosis and
neuroinflammation by downregulating GSDMD, TNF-α and MPO in an
in vivo model of intracerebral hemorrhage (ICH) (183). In addition, overactivated N9
microglia treated with VX-765 are responsible for the reduction in
the NLRP3 inflammasome and pyroptosis-associated proteins
expression in vitro (184). Other research has shown that
VX-765 inhibits silica nanoparticle-induced cardiomyocytic
pyroptosis and cardiac hypertrophy (185).
Ac-YVAD-cmk is a peptide whose sequence is
homologous to a known caspase substrate sequence, confirming its
capacity in suppressing caspase-1 activation (186). As a selective caspase-1
inhibitor, Ac-YVAD-cmk effectively inhibits pyroptosis, and IL-1β
and IL-18 expression in numerous diseases (187). As previously demonstrated, in
rat H9C2 cardiomyocytes, LPS pre-treatment can efficiently mediate
pyroptosis by activating the NLRP3 inflammasome, exacerbating high
glucose and hypoxia/reoxygenation injury. Ac-YVAD-cmk responds by
counteracting these effects (188). Ac-YVAD-cmk reduces the
expression of mature IL-1β/IL-18, improves behavioral performance,
and alleviates microglia in the perihematoma region in rats with
ICH (189). The SARS-CoV-2 N
protein promotes NLRP3 inflammasome activation and generates
excessive inflammatory responses, which are blocked by Ac-YVAD-cmk
(190).
AI-44, a curcumin analogue, binds to peroxiredoxin
1 (PRDX1) and promotes the interaction of PRDX1 with pro-caspase-1,
thereby demonstrating an association between pro-caspase-1 and ASC
(191). However, the inhibitory
effect of AI-44 on NLRP3 inflammasome is markedly diminished after
PRDX1 is knocked out (192).
FC11A-2, another caspase-1 inhibitor, has been shown to
significantly attenuate experimental colitis in mice induced by
DSS, primarily by targeting caspase-1 activation prior to
IL-1β/IL-18 production in macrophages (193). VX-765 and Ac-YVAD-cmk are the
most broadly used inhibitors. Both can reduce the related
inflammatory diseases by suppressing caspase-1 expression to
varying degrees, such as atherosclerosis, CVD (cerebral ischemia
injury, cerebral edema and cerebral hemorrhage), myocardial
pyroptosis and myocardial hypertrophy.
As a candidate for pyroptotic pore formation, GSDMD
is a downstream effector of caspase-1 that not only regulates
pyroptosis, but also releases IL-1β and IL-18 to the extracellular
space (194). As described
above, caspase-1 cleaves GSDMD following NLRP3 inflammasome
activation, allowing GSDMD-N-mediated pore formation in the plasma
membrane to promote pyroptosis (195). In total, 12 Cys residues are
present in the sequence of GSDMD, and the reactivity of Cys191/192
(human/mouse) is crucial for pore formation, since it is well
exposed and is highly reactive in the protein structure.
Suppressing GSDMD can alleviate inflammasome-induced pyroptosis
(196), thus suggesting that
GSDMD may be an attractive novel target for regulating
inflammation.
NSA was initially identified as an inhibitor in
mixed lineage kinase domain-like protein-mediated necroptotic cell
death (197). However, Rathkey
et al (198) found that
the NSA also bound to GSDMD via Cys191, thereby inhibiting the
oligomerization of p30-GSDMD and preventing pyroptosis through both
primary and immortalized macrophages. Additionally, pre-treatment
with NSA was shown to suppress Aβ-142-induced mouse cortical neuron
(MCN) pyroptosis, primarily by targeting the permeability of cell
membrane and inflammatory factor release (199). Notably, the inhibition of
p30-GSDMD oligomerization blocks the opening of membrane pores,
confirming its importance in MCN pyroptosis and its potential as an
NSA target (199). In A549 and
H1299 cells, Teng et al (200) similarly found that NSA inhibited
the polyphyllin VI-induced activation of the NLRP3 inflammasome
(200).
Disulfiram inhibits membrane pore formation in
GSDMD, but not in other GSDMs families, hence covalently modifying
GSDMD in Cys191/Cys192 of human/mice to inhibit pore formation.
Disulfiram and its metabolism exhibit anti-inflammatory activities,
which can alleviate inflammation in vitro and in vivo
(201). Disulfiram relieves
severe acute pancreatitis induced by caerulein and related lung
injury, and inhibits IL-1β and IL-18 production by targeting GSDMD
cleavage (202). In both human
and mouse monocyte/macrophage cells, disulfiram has been shown to
inhibit the release of IL-1β and pyroptosis (203). Similarly, disulfiram has been
shown to block pyroptosis and cytokine release in phorbol
12-myristate 13-acetate-differentiated, as well as LPS-primed human
THP-1 cells and LPS-induced sepsis-associated mortality in mice
(204). It has also been
demonstrated that disulfiram significantly promotes macrophage M2
phenotype polarization based on a small-molecule compound library
(205). Mechanistically,
disulfiram targets GSDMD to attenuate macrophagic pyroptosis, IL-1β
and high mobility group box 1 protein release (205). In mouse experiments, Hu et
al revealed that disulfiram inhibited the function of GSDMD by
covalently modifying its cys192, hence blocking the IL-1β release
without affecting caspase-1 and pro-IL-1β expression (204). Thus, disulfiram does not affect
IL-1β production and maturation, but rather blocks pores formation
in the cell membrane to prevent IL-1β release and pyroptosis
(204,206).
As described above, the treatment efficacy of NLRP3
inhibitors in inflammatory diseases has been largely documented in
animal and cellular experiments. However, their practical
application in treating these diseases is limited because of
insufficient clinical research. Tranilast, OLT1177, NAC, DMF and
disulfiram have been tested thus far in clinical trials (Table II). For instance, tranilast at a
dose of 300 mg/day for 1 year, is safe for patients with both
early-stage and advanced diabetic nephropathy. Increased mesangial
cell proliferation, and the accumulation of extracellular matrix
components, such as collagen in the glomeruli, is one of the
pathologic features during in the early stages of diabetic
nephropathy (211). Tranilast
treatment may suppress collagen accumulation in renal tissue and
may be therapeutically beneficial in reducing the progression of
advanced diabetic nephropathy. Moreover, tranilast may be
therapeutically beneficial for early-stage diabetic nephropathy
(212,213). In another study, tranilast was
administered to patients with coronary artery disease following a
successful directional coronary atherectomy (DCA) at a dose of 600
mg daily for 3 months. Consequently, the oral administration of
tranilast significantly prevented restenosis following DCA
(214). In a phase I study,
OLT1177 was analyzed for safety in patients with heart failure and
reduced ejection fraction; as a result, it was found to be safe and
well-tolerated after 14 days of treatment (215). Furthermore, the trial
demonstrated that NAC was safe when preoperatively administered;
however, its efficacy as an antioxidant and anti-inflammatory agent
was not statistically significant and thus, additional
investigations using a larger sample are warranted (trial no.
NCT03589495). In patients ICH with oxidative stress, was shown to
NAC substantially reduce perihematomal edema volume and shorten
intensive care unit stay (216).
Furthermore, NAC has been shown to significantly improve remission
maintenance in ulcerative colitis patients receiving 800 mg NAC for
16 weeks unlike the placebo (217). Alcohol-dependent patients are
subjected to disulfiram-treatment to discourage
alcohol-consumption. Besides, disulfiram can increase A disintegrin
and metalloprotease 10 expression (218), which inhibits the production of
Aβ, the hallmarks of AD pathology (219). Therefore, NLRP3 inflammasome
pathway inhibitors have been demonstrated in vitro or in
vivo and in clinical trials. Among these candidate molecules
are tranilast and OLT1177, which are safe and effective in both
clinical and basic studies. Furthermore, tranilast is the most
extensively studied, with apparent treatment effects on early or
late diabetes nephropathy. Additionally, tranilast can also prevent
stenosis following DCA in patients with coronary artery
disease.
The NLRP3 inflammasome is present at low levels
under normal circumstances, which is important for innate immunity
regulation. However, NLRP3 inflammasome activation ultimately
results in inflammation and pyroptosis. Therefore, the NLRP3
inflammasome may provide novel targets for the treatment of various
inflammatory diseases. Notably, both priming and activation steps
are crucial for NLRP3 inflammasome activation. Therefore, beginning
from the priming step, the present review summarized the related
TLR4 and NF-κB inhibitors, among which TAK-242, BAY11-7082 and
sulfasalazine inhibit inflammatory diseases caused by the NLRP3
inflammasome, without any notable adverse toxic side-effects.
Subsequently, the present review also summarized the associated ion
inhibitors to preserve the associated ion homeostasis during NLRP3
inflammasome activation. Of note, NLRP3, ASC and caspase-1
inhibitors for the NLRP3 inflammasome itself were also described.
NLRP3-induced pyroptosis is an important mechanism causing
inflammatory disease. Therefore, the present review described the
related inhibitors of pyroptosis executor, GSDMD, which may serve
as an effective target for inflammatory diseases. In conclusion,
the present review comprehensively described the inhibitors that
can trigger NLRP3 inflammasome activation from the priming step to
the activation step, illustrating their promising roles in the
treatment of NLRP3 inflammasome-induced inflammatory diseases.
However, future research is necessary to elucidate certain issues.
First, TLR4 and NF-κB, as common membrane receptors and
transcription factors activate the NLRP3 inflammasome. Secondly,
K+ and CL− efflux are two independent, yet
indispensable events that activate the NLRP3 inflammasome. The
small-molecule inhibitors of the NLRP3 inflammasome documented thus
far have not been confirmed in clinical trials or approved by the
FDA or other institutions. Therefore, their pharmacokinetic
characteristics and comprehensive mechanisms warrant further
investigation, given their promising prospects as NLRP3
inflammasome inhibitors.
Not applicable.
XZ drafted the manuscript. XJ designed and
supervised the study. JG verified the contents and revised the
manuscript. ZW, YZ, QY, MZ and LB critically revised the
manuscript. LY and MG edited the manuscript. All authors reviewed,
and have read and approved the final manuscript. Data
authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present study was supported by the National Natural Science
Foundation of China (grant nos. 82074211, 81873130 and 82174470),
the Tianjin Natural Science Foundation (grant no. 21JCQNJC01170)
and the 2019 Annual Graduate Students Innovation Fund, School of
Integrative Medicine, Tianjin University of Traditional Chinese
Medicine, Tianjin, China (grant no. ZXYCXLX201902).
|
1
|
Fu C, Ye S, Liu Y and Li S: Role of CARD
region of MDA5 gene in canine influenza virus infection. Viruses.
12:3072020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang Y and Okamoto CT: Nucleotide binding
domain and leucine-rich repeat pyrin domain-containing protein 12:
Characterization of its binding to hematopoietic cell kinase. Int J
Biol Sci. 16:1507–1525. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhao C and Zhao W: NLRP3 inflammasome-A
key player in antiviral responses. Front Immunol. 11:2112020.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Swanson KV, Deng M and Ting JP: The NLRP3
inflammasome: Molecular activation and regulation to therapeutics.
Nat Rev Immunol. 19:477–489. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zuo Y, Chen L, Gu H, He X, Ye Z, Wang Z,
Shao Q and Xue C: GSDMD-mediated pyroptosis: A critical mechanism
of diabetic nephropathy. Expert Rev Mol Med. 23:e232021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Arioz BI, Tarakcioglu E, Olcum M and Genc
S: The role of melatonin on NLRP3 inflammasome activation in
diseases. Antioxidants (Basel). 10:10202021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vong CT, Tseng H, Yao P, Yu H, Wang S,
Zhong Z and Wang Y: Specific NLRP3 inflammasome inhibitors:
Promising therapeutic agents for inflammatory diseases. Drug Discov
Today. 26:1394–1408. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zeng W, Wu D, Sun Y, Suo Y, Yu Q, Zeng M,
Gao Q, Yu B, Jiang X and Wang Y: The selective NLRP3 inhibitor
MCC950 hinders atherosclerosis development by attenuating
inflammation and pyroptosis in macrophages. Sci Rep. 11:193052021.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Feng YS, Tan ZX, Wang MM, Xing Y, Dong F
and Zhang F: Inhibition of NLRP3 inflammasome: A prospective target
for the treatment of ischemic stroke. Front Cell Neurosci.
14:1552020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Holbrook JA, Jarosz-Griffiths HH, Caseley
E, Lara-Reyna S, Poulter JA, Williams-Gray CH, Peckham D and
McDermott MF: Neurodegenerative disease and the NLRP3 inflammasome.
Front Pharmacol. 12:6432542021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ferreira NS, Bruder-Nascimento T, Pereira
CA, Zanotto CZ, Prado DS, Silva JF, Rassi DM, Foss-Freitas MC,
Alves-Filho JC, Carlos D, et al: NLRP3 inflammasome and
mineralocorticoid receptors are associated with vascular
dysfunction in type 2 diabetes mellitus. Cells. 8:15952019.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jiang H, He H, Chen Y, Huang W, Cheng J,
Ye J, Wang A, Tao J, Wang C, Liu Q, et al: Identification of a
selective and direct NLRP3 inhibitor to treat inflammatory
disorders. J Exp Med. 214:3219–3238. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Guarda G, Zenger M, Yazdi AS, Schroder K,
Ferrero I, Menu P, Tardivel A, Mattmann C and Tschopp J:
Differential expression of NLRP3 among hematopoietic cells. J
Immunol. 186:2529–2534. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhong Y, Kinio A and Saleh M: Functions of
NOD-like receptors in human diseases. Front Immunol. 4:3332013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Park WJ and Han JS: Gryllus bimaculatus
extract protects against lipopolysaccharide and palmitate-induced
production of proinflammatory cytokines and inflammasome formation.
Mol Med Rep. 23:2062021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Flores-Costa R, Duran-Guell M, Casulleras
M, Lopez-Vicario C, Alcaraz-Quiles J, Diaz A, Lozano JJ, Titos E,
Hall K, Sarno R, et al: Stimulation of soluble guanylate cyclase
exerts antiinflammatory actions in the liver through a
VASP/NF-κB/NLRP3 inflammasome circuit. Proc Natl Acad Sci USA.
117:28263–28274. 2020. View Article : Google Scholar
|
|
17
|
Dowling JK and O'Neill LA: Biochemical
regulation of the inflammasome. Crit Rev Biochem Mol. 47:424–443.
2012. View Article : Google Scholar
|
|
18
|
Ulland TK, Ferguson PJ and Sutterwala FS:
Evasion of inflammasome activation by microbial pathogens. J Clin
Invest. 125:469–477. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Trojan E, Tylek K, Leskiewicz M, Lason W,
Brandenburg LO, Leopoldo M, Lacivita E and Basta-Kaim A: The
N-Formyl peptide receptor 2 (FPR2) agonist MR-39 exhibits
anti-inflammatory activity in LPS-stimulated organotypic
hippocampal cultures. Cells. 10:15242021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ming SL, Zeng L, Guo YK, Zhang S, Li GL,
Ma YX, Zhai YY, Chang WR, Yang L, Wang J, et al: The human-specific
STING agonist G10 activates type I interferon and the NLRP3
inflammasome in porcine cells. Front Immunol. 11:5758182020.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Munoz-Planillo R, Kuffa P, Martinez-Colon
G, Smith BL, Rajendiran TM and Nunez G: K(+) efflux is the common
trigger of NLRP3 inflammasome activation by bacterial toxins and
particulate matter. Immunity. 38:1142–1153. 2013. View Article : Google Scholar
|
|
22
|
Tang T, Lang X, Xu C, Wang X, Gong T, Yang
Y, Cui J, Bai L, Wang J, Jiang W and Zhou R: CLICs-dependent
chloride efflux is an essential and proximal upstream event for
NLRP3 inflammasome activation. Nat Commun. 8:2022017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
He Y, Zeng MY, Yang D, Motro B and Nunez
G: NEK7 is an essential mediator of NLRP3 activation downstream of
potassium efflux. Nature. 530:354–357. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Maejima I, Takahashi A, Omori H, Kimura T,
Takabatake Y, Saitoh T, Yamamoto A, Hamasaki M, Noda T, Isaka Y, et
al: Autophagy sequesters damaged lysosomes to control lysosomal
biogenesis and kidney injury. EMBO J. 32:2336–2347. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Otsuki T, Holian A and Di Gioacchino M:
Immunological effects of environmental factors: Focus on the
fibrous and particulated materials. J Immunol Res. 2014:6974382014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang Y, Qin X and Paudel HK: Amyloid beta
peptide promotes lysosomal degradation of clusterin via sortilin in
hippocampal primary neurons. Neurobiol Dis. 103:78–88. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ismael S, Ahmed HA, Adris T, Parveen K,
Thakor P and Ishrat T: The NLRP3 inflammasome: A potential
therapeutic target for traumatic brain injury. Neural Regen Res.
16:49–57. 2021. View Article : Google Scholar :
|
|
28
|
Paik S, Kim JK, Silwal P, Sasakawa C and
Jo EK: An update on the regulatory mechanisms of NLRP3 inflammasome
activation. Cell Mol Immunol. 18:1141–1160. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bauernfeind F, Bartok E, Rieger A, Franchi
L, Nunez G and Hornung V: Cutting edge: Reactive oxygen species
inhibitors block priming, but not activation, of the NLRP3
inflammasome. J Immunol. 187:613–617. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang C, Fu KK, Dai J, Lacey SD, Yao Y,
Pastel G, Xu L, Zhang J and Hu L: Inverted battery design as ion
generator for interfacing with biosystems. Nat Commun. 8:156092017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ma C, Liu S, Zhang S, Xu T, Yu X, Gao Y,
Zhai C, Li C, Lei C, Fan S, et al: Evidence and perspective for the
role of the NLRP3 inflammasome signaling pathway in ischemic stroke
and its therapeutic potential (Review). Int J Mol Med.
42:2979–2990. 2018.PubMed/NCBI
|
|
32
|
Lee GS, Subramanian N, Kim AI,
Aksentijevich I, Goldbach-Mansky R, Sacks DB, Germain RN, Kastner
DL and Chae JJ: The calcium-sensing receptor regulates the NLRP3
inflammasome through Ca2+ and cAMP. Nature. 492:123–127. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhong Z, Zhai Y, Liang S, Mori Y, Han R,
Sutterwala FS and Qiao L: TRPM2 links oxidative stress to NLRP3
inflammasome activation. Nat Commun. 4:16112013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jo EK, Kim JK, Shin DM and Sasakawa C:
Molecular mechanisms regulating NLRP3 inflammasome activation. Cell
Mol Immunol. 13:148–159. 2016. View Article : Google Scholar :
|
|
35
|
Hosen MR, Goody PR, Zietzer A, Nickenig G
and Jansen F: MicroRNAs as master regulators of atherosclerosis:
From pathogenesis to novel therapeutic options. Antioxid Redox
Sign. 33:621–644. 2020. View Article : Google Scholar
|
|
36
|
Saigusa R, Winkels H and Ley K: T cell
subsets and functions in atherosclerosis. Nat Rev Cardiol.
17:387–401. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Martinet W, Coornaert I, Puylaert P and De
Meyer G: Macrophage death as a pharmacological target in
atherosclerosis. Front Pharmacol. 10:3062019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xu H, Jiang J, Chen W, Li W and Chen Z:
Vascular macrophages in atherosclerosis. J Immunol Res.
2019:43547862019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ren XS, Tong Y, Ling L, Chen D, Sun HJ,
Zhou H, Qi XH, Chen Q, Li YH, Kang YM and Zhu GQ: NLRP3 Gene
deletion attenuates Angiotensin II-Induced phenotypic
transformation of vascular smooth muscle cells and vascular
remodeling. Cell Physiol Biochem. 44:2269–2280. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wu X, Zhang H, Qi W, Zhang Y, Li J, Li Z,
Lin Y, Bai X, Liu X, Chen X, et al: Nicotine promotes
atherosclerosis via ROS-NLRP3-mediated endothelial cell pyroptosis.
Cell Death Dis. 9:1712018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhao Z, Wang X, Zhang R, Ma B, Niu S, Di
X, Ni L and Liu C: Melatonin attenuates smoking-induced
atherosclerosis by activating the Nrf2 pathway via NLRP3
inflammasomes in endothelial cells. Aging (Albany NY).
13:11363–11380. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Mehta S and Dhawan V: Exposure of
cigarette smoke condensate activates NLRP3 inflammasome in THP-1
cells in a stage-specific manner: An underlying role of innate
immunity in atherosclerosis. Cell Signal. 72:1096452020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mehta S, Srivastava N, Bhatia A and Dhawan
V: Exposure of cigarette smoke condensate activates NLRP3
inflammasome in vitro and in vivo: A connotation of innate immunity
and atherosclerosis. Int Immunopharmacol. 84:1065612020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Keping Y, Yunfeng S, Pengzhuo X, Liang L,
Chenhong X and Jinghua M: Sestrin1 inhibits oxidized low-density
lipoprotein-induced activation of NLRP3 inflammasome in macrophages
in a murine atherosclerosis model. Eur J Immunol. 50:1154–1166.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ma Q, Yang Q, Chen J, Yu C, Zhang L, Zhou
W and Chen M: Salvianolic acid A ameliorates early-stage
atherosclerosis development by inhibiting NLRP3 inflammasome
activation in zucker diabetic fatty rats. Molecules. 25:10892020.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Li W, Liu D, Xu J, Zha J, Wang C, An J,
Xie Z and Qiao S: Astrocyte-Derived TNF-alpha-Activated platelets
promote cerebral Ischemia/Reperfusion injury by regulating the
RIP1/RIP3/AKT signaling pathway. Mol Neurobiol. 59:5734–5749. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sun R, Peng M, Xu P, Huang F, Xie Y, Li J,
Hong Y, Guo H, Liu Q and Zhu W: Low-density lipoprotein receptor
(LDLR) regulates NLRP3-mediated neuronal pyroptosis following
cerebral ischemia/reperfusion injury. J Neuroinflamm. 17:3302020.
View Article : Google Scholar
|
|
48
|
Shimizu T, Smits R and Ikenaka K:
Microglia-induced activation of non-canonical Wnt signaling
aggravates neurodegeneration in demyelinating disorders. Mol Cell
Biol. 36:2728–2741. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ma DC, Zhang NN, Zhang YN and Chen HS:
Kv1.3 channel blockade alleviates cerebral ischemia/reperfusion
injury by reshaping M1/M2 phenotypes and compromising the
activation of NLRP3 inflammasome in microglia. Exp Neurol.
332:1133992020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Pozzo ED, Tremolanti C, Costa B,
Giacomelli C, Milenkovic VM, Bader S, Wetzel CH, Rupprecht R,
Taliani S, Settimo FD and Martini C: Microglial Pro-Inflammatory
and Anti-Inflammatory phenotypes are modulated by translocator
protein activation. Int J Mol Sci. 20:44672019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Chen C, Ai Q, Chu S, Zhang Z, Zhou X, Luo
P, Liu Y and Chen N: IMM-H004 protects against oxygen-glucose
deprivation/reperfusion injury to BV2 microglia partly by
modulating CKLF1 involved in microglia polarization. Int
Immunopharmacol. 70:69–79. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liu H, Wu X, Luo J, Zhao L, Li X, Guo H,
Bai H, Cui W, Guo W, Feng D and Qu Y: Adiponectin peptide
alleviates oxidative stress and NLRP3 inflammasome activation after
cerebral ischemia-reperfusion injury by regulating AMPK/GSK-3β. Exp
Neurol. 329:1133022020. View Article : Google Scholar
|
|
53
|
Zhao J, Piao X, Wu Y, Liang S, Han F,
Liang Q, Shao S and Zhao D: Cepharanthine attenuates cerebral
ischemia/reperfusion injury by reducing NLRP3 inflammasome-induced
inflammation and oxidative stress via inhibiting 12/15-LOX
signaling. Biomed Pharmacother. 127:1101512020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Kaushal V, Koeberle PD, Wang Y and
Schlichter LC: The Ca2+-activated K+ channel KCNN4/KCa3.1
contributes to microglia activation and nitric oxide-dependent
neurodegeneration. J Neurosci. 27:234–244. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Ma DC, Zhang NN, Zhang YN and Chen HS:
Salvianolic Acids for injection alleviates cerebral
ischemia/reperfusion injury by switching M1/M2 phenotypes and
inhibiting NLRP3 inflammasome/pyroptosis axis in microglia in vivo
and in vitro. J Ethnopharmacol. 270:1137762021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Li ZG, Shui SF, Han XW and Yan L: NLRP10
ablation protects against ischemia/reperfusion-associated brain
injury by suppression of neuroinflammation. Exp Cell Res.
389:1119122020. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Cao X, Wang Y and Gao L: CHRFAM7A
overexpression attenuates cerebral ischemia-reperfusion injury via
inhibiting microglia pyroptosis mediated by the NLRP3/Caspase-1
pathway. Inflammation. 44:1023–1034. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Bellut M, Papp L, Bieber M, Kraft P, Stoll
G and Schuhmann MK: NLPR3 inflammasome inhibition alleviates
hypoxic endothelial cell death in vitro and protects blood-brain
barrier integrity in murine stroke. Cell Death Dis. 13:202021.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Fu C, Zhang X, Zeng Z, Tian Y, Jin X, Wang
F, Xu Z, Chen B, Zheng H and Liu X: Neuroprotective effects of
qingnao dripping pills against cerebral ischemia via Inhibiting
NLRP3 inflammasome signaling pathway: In vivo and in vitro. Front
Pharmacol. 11:652020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Denes A, Coutts G, Lenart N, Cruickshank
SM, Pelegrin P, Skinner J, Rothwell N, Allan SM and Brough D: AIM2
and NLRC4 inflammasomes contribute with ASC to acute brain injury
independently of NLRP3. Proc Natl Acad Sci USA. 112:4050–4055.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Severini C, Barbato C, Di Certo MG,
Gabanella F, Petrella C, Di Stadio A, de Vincentiis M, Polimeni A,
Ralli M and Greco A: Alzheimer's disease: New concepts on the role
of autoimmunity and NLRP3 inflammasome in the pathogenesis of the
disease. Curr Neuropharmacol. 19:498–512. 2021.
|
|
62
|
Lee YJ, Han SB, Nam SY, Oh KW and Hong JT:
Inflammation and Alzheimer's disease. Arch Pharm Res. 33:1539–1556.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Li G, Dong Y, Liu D, Zou Z, Hao G, Gao X,
Pan P and Liang G: NEK7 coordinates rapid neuroinflammation after
subarachnoid hemorrhage in mice. Front Neurol. 11:5512020.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Liang S, Zhong Z, Kim SY, Uchiyama R, Roh
YS, Matsushita H, Gottlieb RA and Seki E: Murine macrophage
autophagy protects against alcohol-induced liver injury by
degrading interferon regulatory factor 1 (IRF1) and removing
damaged mitochondria. J Biol Chem. 294:12359–12369. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Cui W, Sun C, Ma Y, Wang S, Wang X and
Zhang Y: Inhibition of TLR4 induces M2 microglial polarization and
provides neuroprotection via the NLRP3 inflammasome in Alzheimer's
disease. Front Neurosci. 14:4442020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
He XF, Xu JH, Li G, Li MY, Li LL, Pei Z,
Zhang LY and Hu XQ: NLRP3-dependent microglial training impaired
the clearance of amyloid-beta and aggravated the cognitive decline
in Alzheimer's disease. Cell Death Dis. 11:8492020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ismael S, Wajidunnisa, Sakata K, McDonald
MP, Liao FF and Ishrat T: ER stress associated TXNIP-NLRP3
inflammasome activation in hippocampus of human Alzheimer's
disease. Neurochem Int. 148:1051042021. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Li L, Ismael S, Nasoohi S, Sakata K, Liao
FF, McDonald MP and Ishrat T: Thioredoxin-interacting protein
(TXNIP) associated NLRP3 inflammasome activation in human
Alzheimer's disease brain. J Alzheimers Dis. 68:255–265. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Tang H and Harte M: Investigating markers
of the NLRP3 inflammasome pathway in Alzheimer's disease: A human
post-mortem study. Genes (Basel). 12:17532021. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Heneka MT, Kummer MP, Stutz A, Delekate A,
Schwartz S, Vieira-Saecker A, Griep A, Axt D, Remus A, Tzeng TC, et
al: NLRP3 is activated in Alzheimer's disease and contributes to
pathology in APP/PS1 mice. Nature. 493:674–678. 2013. View Article : Google Scholar
|
|
71
|
Garcia-Serrano AM and Duarte J: Brain
metabolism alterations in type 2 diabetes: What did we learn from
diet-induced diabetes models? Front Neurosci. 14:2292020.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
An X, Jin D, Duan L, Zhao S, Zhou R, Lian
F and Tong X: Direct and indirect therapeutic effect of traditional
Chinese medicine as an add-on for non-proliferative diabetic
retinopathy: A systematic review and meta-analysis. Chin Med.
15:992020. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Omar SM, Musa IR, ElSouli A and Adam I:
Prevalence, risk factors, and glycaemic control of type 2 diabetes
mellitus in eastern Sudan: A community-based study. Ther Adv
Endocrinol. 10:19061866492019.
|
|
74
|
Yu ZW, Zhang J, Li X, Wang Y, Fu YH and
Gao XY: A new research hot spot: The role of NLRP3 inflammasome
activation, a key step in pyroptosis, in diabetes and diabetic
complications. Life Sci. 240:1171382020. View Article : Google Scholar
|
|
75
|
Wanrooy BJ, Kumar KP, Wen SW, Qin CX,
Ritchie RH and Wong C: Distinct contributions of hyperglycemia and
high-fat feeding in metabolic syndrome-induced neuroinflammation. J
Neuroinflamm. 15:2932018. View Article : Google Scholar
|
|
76
|
Zhang H, Chen H, Wu X, Sun T, Fan M, Tong
H, Zhu Y, Yin Z, Sun W, Zhang C, et al: Tetramethylpyrazine
alleviates diabetes-induced high platelet response and endothelial
adhesion via inhibiting NLRP3 inflammasome activation.
Phytomedicine. 96:1538602022. View Article : Google Scholar
|
|
77
|
Zheng Q, Pan L and Ji Y: H2S
protects against diabetes-accelerated atherosclerosis by preventing
the activation of NLRP3 inflammasome. J Biomed Res. 34:94–102.
2019. View Article : Google Scholar
|
|
78
|
Lian D, Liu J, Han R, Jin J, Zhu L, Zhang
Y, Huang Y, Wang X, Xian S and Chen Y: Kakonein restores
diabetes-induced endothelial junction dysfunction via promoting
autophagy-mediated NLRP3 inflammasome degradation. J Cell Mol Med.
25:7169–7180. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Sharma A, Choi J, Stefanovic N, Al-Sharea
A, Simpson DS, Mukhamedova N, Jandeleit-Dahm K, Murphy AJ, Sviridov
D, Vince JE, et al: Specific NLRP3 inhibition protects against
diabetes-associated atherosclerosis. Diabetes. 70:772–787. 2021.
View Article : Google Scholar
|
|
80
|
Ward R, Li W, Abdul Y, Jackson L, Dong G,
Jamil S, Filosa J, Fagan SC and Ergul A: NLRP3 inflammasome
inhibition with MCC950 improves diabetes-mediated cognitive
impairment and vasoneuronal remodeling after ischemia. Pharmacol
Res. 142:237–250. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Amin FM, Abdelaziz RR, Hamed MF, Nader MA
and Shehatou G: Dimethyl fumarate ameliorates diabetes-associated
vascular complications through ROS-TXNIP-NLRP3 inflammasome
pathway. Life Sci. 256:1178872020. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Lin HB, Wei GS, Li FX, Guo WJ, Hong P,
Weng YQ, Zhang QQ, Xu SY, Liang WB, You ZJ, et al: Macrophage-NLRP3
inflammasome activation exacerbates cardiac dysfunction after
ischemic stroke in a mouse model of diabetes. Neurosci Bull.
36:1035–1045. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Kim SR, Lee SG, Kim SH, Kim JH, Choi E,
Cho W, Rim JH, Hwang I, Lee CJ, Lee M, et al: SGLT2 inhibition
modulates NLRP3 inflammasome activity via ketones and insulin in
diabetes with cardiovascular disease. Nat Commun. 11:21272020.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Dror E, Dalmas E, Meier DT, Wueest S,
Thevenet J, Thienel C, Timper K, Nordmann TM, Traub S, Schulze F,
et al: Postprandial macrophage-derived IL-1beta stimulates insulin,
and both synergistically promote glucose disposal and inflammation.
Nat Immunol. 18:283–292. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Burke SJ, Batdorf HM, Burk DH, Martin TM,
Mendoza T, Stadler K, Alami W, Karlstad MD, Robson MJ, Blakely RD,
et al: Pancreatic deletion of the interleukin-1 receptor disrupts
whole body glucose homeostasis and promotes islet β-cell
de-differentiation. Mol Metab. 14:95–107. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Yang M, Wang X, Han Y, Li C, Wei L, Yang
J, Chen W, Zhu X and Sun L: Targeting the NLRP3 inflammasome in
diabetic nephropathy. Curr Med Chem. 28:8810–8824. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Den Hartogh DJ, Gabriel A and Tsiani E:
Antidiabetic properties of Curcumin II: Evidence from in vivo
studies. Nutrients. 12:582019. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Oltean S, Coward R, Collino M and Baelde
H: Diabetic nephropathy: Novel molecular mechanisms and therapeutic
avenues. Biomed Res Int. 2017:31465242017. View Article : Google Scholar
|
|
89
|
Wang B, Dai Z, Gao Q, Liu Y, Gu G and
Zheng H: Spop ameliorates diabetic nephropathy through restraining
NLRP3 inflammasome. Biochem Bioph Res Commun. 594:131–138. 2022.
View Article : Google Scholar
|
|
90
|
Tassetto M, Scialdone A, Solini A and Di
Virgilio F: The P2X7 receptor: A promising pharmacological target
in diabetic retinopathy. Int J Mol Sci. 22:71102021. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li R, Chen L, Yao GM, Yan HL and Wang L:
Effects of quercetin on diabetic retinopathy and its association
with NLRP3 inflammasome and autophagy. Int J Ophthalmol. 14:42–49.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Lin HB, Lin YH, Zhang JY, Guo WJ, Ovcjak
A, You ZJ, Feng ZP, Sun HS, Li FX and Zhang HF: NLRP3 inflammasome:
A potential target in isoflurane pretreatment alleviates
Stroke-induced retinal injury in diabetes. Front Cell Neurosci.
15:6974492021. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Shujun W, Huijie Z, Xia B and Hongjian W:
Cerebral venous sinus thrombosis in patients with inflammatory
bowel disease: A retrospective study. Sci Rep. 11:170042021.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Fu Y, Lee CH and Chi CC: Association of
psoriasis with inflammatory bowel disease: A systematic review and
Meta-analysis. JAMA Dermatol. 154:1417–1423. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Ginwala R, Bhavsar R, Chigbu DI, Jain P
and Khan ZK: Potential role of flavonoids in treating chronic
inflammatory diseases with a special focus on the anti-inflammatory
activity of apigenin. Antioxidants (Basel). 8:352019. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Wang H and Ma YC: Role of NLRP1 and NLRP3
inflammasome signaling pathways in the immune mechanism of
inflammatory bowel disease in children. Zhongguo Dang Dai Er Ke Za
Zhi. 22:854–859. 2020.In Chinese. PubMed/NCBI
|
|
97
|
Zhou W, Liu X, Zhang X, Tang J, Li Z, Wang
Q and Hu R: Oroxylin A inhibits colitis by inactivating NLRP3
inflammasome. Oncotarget. 8:58903–58917. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Zhang C, Qin J, Zhang S, Zhang N, Tan B,
Siwko S, Zhang Y, Wang Q, Chen J, Qian M, et al:
ADP/P2Y1 aggravates inflammatory bowel disease through
ERK5-mediated NLRP3 inflammasome activation. Mucosal Immunol.
13:931–945. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Zhou L, Liu T, Huang B, Luo M, Chen Z,
Zhao Z, Wang J, Leung D, Yang X, Chan KW, et al: Excessive
deubiquitination of NLRP3-R779C variant contributes to
very-early-onset inflammatory bowel disease development. J Allergy
Clin Immun. 147:267–279. 2021. View Article : Google Scholar
|
|
100
|
Chen X, Liu G, Yuan Y, Wu G, Wang S and
Yuan L: NEK7 interacts with NLRP3 to modulate the pyroptosis in
inflammatory bowel disease via NF-κB signaling. Cell Death Dis.
10:9062019. View Article : Google Scholar
|
|
101
|
Ma X, Di Q, Li X, Zhao X, Zhang R, Xiao Y,
Li X, Wu H, Tang H, Quan J, et al: Munronoid I ameliorates
DSS-induced mouse colitis by inhibiting NLRP3 inflammasome
activation and pyroptosis via modulation of NLRP3. Front Immunol.
13:8531942022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Jiang Q, Li W, Zhu X, Yu L, Lu Z, Liu Y,
Ma B and Cheng L: Estrogen receptor β alleviates inflammatory
lesions in a rat model of inflammatory bowel disease via
down-regulating P2X7R expression in macrophages. Int J Biochem Cell
Biol. 139:1060682021. View Article : Google Scholar
|
|
103
|
Sang H, Xie Y, Su X, Zhang M, Zhang Y, Liu
K and Wang J: Mushroom Bulgaria Inquinans modulates host
immunological response and gut microbiota in mice. Front Nutr.
7:1442020. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Liu Z and Wang H: Probiotics alleviate
inflammatory bowel disease in mice by regulating intestinal
microorganisms-bile acid-NLRP3 inflammasome pathway. Acta Biochim
Pol. 68:687–693. 2021.PubMed/NCBI
|
|
105
|
Dinarello CA, Simon A and van der Meer JW:
Treating inflammation by blocking interleukin-1 in a broad spectrum
of diseases. Nat Rev Drug Discov. 11:633–652. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Liu M, Saredy J, Zhang R, Shao Y, Sun Y,
Yang WY, Wang J, Liu L, Drummer CT, Johnson C, et al: Approaching
inflammation paradoxes-proinflammatory cytokine blockages induce
inflammatory regulators. Front Immunol. 11:5543012020. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Liu J, Zhang N, Zhang M, Yin H, Zhang X,
Wang X, Wang X and Zhao Y: N-acetylserotonin alleviated the
expression of interleukin-1beta in retinal ischemia-reperfusion
rats via the TTLR4/NF-κB/NLRP3 pathway. Exp Eye Res.
208:1085952021. View Article : Google Scholar
|
|
108
|
Bauernfeind FG, Horvath G, Stutz A,
Alnemri ES, MacDonald K, Speert D, Fernandes-Alnemri T, Wu J, Monks
BG, Fitzgerald KA, et al: Cutting edge: NF-kappaB activating
pattern recognition and cytokine receptors license NLRP3
inflammasome activation by regulating NLRP3 expression. J Immunol.
183:787–791. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Fernandes-Alnemri T, Kang S, Anderson C,
Sagara J, Fitzgerald KA and Alnemri ES: Cutting edge: TLR signaling
licenses IRAK1 for rapid activation of the NLRP3 inflammasome. J
Immunol. 191:3995–3999. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Yang J, Wise L and Fukuchi KI: TLR4
cross-talk with NLRP3 inflammasome and complement signaling
pathways in Alzheimer's disease. Front Immunol. 11:7242020.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Matsunaga N, Tsuchimori N, Matsumoto T and
Ii M: TAK-242 (resatorvid), a small-molecule inhibitor of Toll-like
receptor (TLR) 4 signaling, binds selectively to TLR4 and
interferes with interactions between TLR4 and its adaptor
molecules. Mol Pharmacol. 79:34–41. 2011. View Article : Google Scholar
|
|
112
|
Plunk MA, Alaniz A, Olademehin OP,
Ellington TL, Shuford KL and Kane RR: Design and catalyzed
activation of Tak-242 prodrugs for localized inhibition of
TLR4-induced inflammation. Acs Med Chem Lett. 11:141–146. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Feng Y, Gao J, Cui Y, Li M, Li R, Cui C
and Cui J: Neuroprotective effects of resatorvid against traumatic
brain injury in rat: Involvement of neuronal autophagy and TLR4
signaling pathway. Cell Mol Neurobiol. 37:155–168. 2017. View Article : Google Scholar
|
|
114
|
Karimy JK, Reeves BC and Kahle KT:
Targeting TLR4-dependent inflammation in post-hemorrhagic brain
injury. Expert Opin Ther Tar. 24:525–533. 2020. View Article : Google Scholar
|
|
115
|
Liu Y, Dai Y, Li Q, Chen C, Chen H, Song
Y, Hua F and Zhang Z: Beta-amyloid activates NLRP3 inflammasome via
TLR4 in mouse microglia. Neurosci Lett. 736:1352792020. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Hong YP, Yu J, Su YR, Mei FC, Li M, Zhao
KL, Zhao L, Deng WH, Chen C and Wang WX: High-fat diet aggravates
acute pancreatitis via TLR4-mediated necroptosis and inflammation
in rats. Oxid Med Cell Longev. 2020:81727142020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Xu M, Ye Z, Zhao X, Guo H, Gong X and
Huang R: Deficiency of tenascin-C attenuated cardiac injury by
inactivating TLR4/NLRP3/caspase-1 pathway after myocardial
infarction. Cell Signal. 86:1100842021. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Huang X, Shen H, Liu Y, Qiu S and Guo Y:
Fisetin attenuates periodontitis through FGFR1/TLR4/NLRP3
inflammasome pathway. Int Immunopharmacol. 95:1075052021.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Zhu Y, Zhu WP, Li W, Zhang HT, Chen BH,
Ding A, Yang H and Zhang H: Implications of EET in renal
ischemia/reperfusion by regulating NLRP3 expression and pyroptosis.
Zhonghua Yi Xue Za Zhi. 100:779–784. 2020.In Chinese. PubMed/NCBI
|
|
120
|
Xu Q, Wang M, Guo H, Liu H, Zhang G, Xu C
and Chen H: Emodin alleviates severe acute pancreatitis-associated
acute lung injury by inhibiting the cold-inducible RNA-binding
protein (CIRP)-mediated activation of the NLRP3/IL-1β/CXCL1
signaling. Front Pharmacol. 12:6553722021. View Article : Google Scholar
|
|
121
|
Pierce JW, Schoenleber R, Jesmok G, Best
J, Moore SA, Collins T and Gerritsen ME: Novel inhibitors of
cytokine-induced IkappaBalpha phosphorylation and endothelial cell
adhesion molecule expression show anti-inflammatory effects in
vivo. J Biol Chem. 272:21096–21103. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Irrera N, Vaccaro M, Bitto A, Pallio G,
Pizzino G, Lentini M, Arcoraci V, Minutoli L, Scuruchi M, Cutroneo
G, et al: BAY 11-7082 inhibits the NF-κB and NLRP3 inflammasome
pathways and protects against IMQ-induced psoriasis. Clin Sci
(Lond). 131:487–498. 2017.
|
|
123
|
Chen X, Wang Y, Yao N and Lin Z:
Immunoproteasome modulates NLRP3 inflammasome-mediated
neuroinflammation under cerebral ischaemia and reperfusion
conditions. J Cell Mol Med. 26:462–474. 2022. View Article : Google Scholar
|
|
124
|
Lang L, Xu B, Yuan J, Li S, Lian S, Chen
Y, Guo J and Yang H: GABA-mediated activated microglia induce
neuroinflammation in the hippocampus of mice following cold
exposure through the NLRP3 inflammasome and NF-κB signaling
pathways. Int Immunopharmacol. 89:1069082020. View Article : Google Scholar
|
|
125
|
Gan HT, Chen YQ and Ouyang Q:
Sulfasalazine inhibits activation of nuclear factor-kappaB in
patients with ulcerative colitis. J Gastroen Hepatol. 20:1016–1024.
2005. View Article : Google Scholar
|
|
126
|
Hafez HM, Ibrahim MA, Yehia AW, Gad AA,
Mohammed NAHS and Abdel-Gaber SA: Protective effect of mirtazapine
against acetic acid-induced ulcerative colitis in rats: Role of
NLRP3 inflammasome pathway. Int Immunopharmacol. 101:1081742021.
View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Ullah H, Saba E, Lee YY, Hong SB, Hyun SH,
Kwak YS, Park CK, Kim SD and Rhee MH: Restorative effects of
Rg3-enriched Korean Red Ginseng and Persicaria tinctoria extract on
oxazolone-induced ulcerative colitis in mice. J Ginseng Res.
46:628–635. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Zhang Y and Shi Y: Intracellular potassium
ion measurements by inductively coupled plasma optical emission
spectrometer (ICP-OES). Methods Mol Biol. 2459:85–92. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Leu WJ, Chu JC, Hsu JL, Du CM, Jiang YH,
Hsu LC, Huang WJ and Guh JH: Chalcones display anti-NLRP3
inflammasome activity in macrophages through inhibition of both
priming and activation steps-structure-activity-relationship and
mechanism studies. Molecules. 25:59602020. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Li Q, Feng H, Wang H, Wang Y, Mou W, Xu G,
Zhang P, Li R, Shi W, Wang Z, et al: Licochalcone B specifically
inhibits the NLRP3 inflammasome by disrupting NEK7-NLRP3
interaction. EMBO Rep. 23:e534992022. View Article : Google Scholar
|
|
131
|
Abais JM, Xia M, Zhang Y, Boini KM and Li
PL: Redox regulation of NLRP3 inflammasomes: ROS as trigger or
effector? Antioxid Redox Sign. 22:1111–1129. 2015. View Article : Google Scholar
|
|
132
|
Lan XF, Zhang XJ, Lin YN, Wang Q, Xu HJ,
Zhou LN, Chen PL and Li QY: Estradiol regulates Txnip and prevents
intermittent hypoxia-induced vascular injury. Sci Rep. 7:103182017.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Zhao Y, Guo Q, Zhu Q, Tan R, Bai D, Bu X,
Lin B, Zhao K, Pan C, Chen H, et al: Flavonoid VI-16 protects
against DSS-induced colitis by inhibiting Txnip-dependent NLRP3
inflammasome activation in macrophages via reducing oxidative
stress. Mucosal Immunol. 12:1150–1163. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Al KH, Brown LJ, Hossain KR, Hudson AL,
Sinclair-Burton AA, Ng JP, Daniel EL, Hare JE, Cornell BA, Curmi
PM, et al: Members of the chloride intracellular ion channel
protein family demonstrate glutaredoxin-like enzymatic activity.
PLoS One. 10:e1156992015. View Article : Google Scholar
|
|
135
|
Ye L, Zeng Q, Ling M, Ma R, Chen H, Lin F,
Li Z and Pan L: Inhibition of IP3R/Ca2+ Dysregulation Protects mice
from ventilator-induced lung injury via endoplasmic reticulum and
mitochondrial pathways. Front Immunol. 12:7290942021. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Davis BK, Wen H and Ting JP: The
inflammasome NLRs in immunity, inflammation, and associated
diseases. Annu Rev Immunol. 29:707–735. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Dick MS, Sborgi L, Ruhl S, Hiller S and
Broz P: ASC filament formation serves as a signal amplification
mechanism for inflammasomes. Nat Commun. 7:119292016. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Duncan JA, Bergstralh DT, Wang Y,
Willingham SB, Ye Z, Zimmermann AG and Ting JP: Cryopyrin/NALP3
binds ATP/dATP, is an ATPase, and requires ATP binding to mediate
inflammatory signaling. Proc Natl Acad Sci USA. 104:8041–8046.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Wu D, Chen Y, Sun Y, Gao Q, Li H, Yang Z,
Wang Y, Jiang X and Yu B: Target of MCC950 in inhibition of NLRP3
inflammasome activation: A literature review. Inflammation.
43:17–23. 2020. View Article : Google Scholar
|
|
140
|
Dempsey C, Rubio AA, Bryson KJ, Finucane
O, Larkin C, Mills EL, Robertson A, Cooper MA, O'Neill L and Lynch
MA: Inhibiting the NLRP3 inflammasome with MCC950 promotes
non-phlogistic clearance of amyloid-beta and cognitive function in
APP/PS1 mice. Brain Behav Immun. 61:306–316. 2017. View Article : Google Scholar
|
|
141
|
Coll RC, Robertson AA, Chae JJ, Higgins
SC, Munoz-Planillo R, Inserra MC, Vetter I, Dungan LS, Monks BG,
Stutz A, et al: A small-molecule inhibitor of the NLRP3
inflammasome for the treatment of inflammatory diseases. Nat Med.
21:248–255. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Gross CJ, Mishra R, Schneider KS, Medard
G, Wettmarshausen J, Dittlein DC, Shi H, Gorka O, Koenig PA, Fromm
S, et al: K+ Efflux-independent NLRP3 inflammasome
activation by small molecules targeting mitochondria. Immunity.
45:761–773. 2016. View Article : Google Scholar
|
|
143
|
van der Heijden T, Kritikou E, Venema W,
van Duijn J, van Santbrink PJ, Slutter B, Foks AC, Bot I and Kuiper
J: NLRP3 inflammasome inhibition by MCC950 reduces atherosclerotic
lesion development in apolipoprotein E-deficient Mice-brief report.
Arterioscl Throm Vas. 37:1457–1461. 2017. View Article : Google Scholar
|
|
144
|
Perera AP, Fernando R, Shinde T,
Gundamaraju R, Southam B, Sohal SS, Robertson A, Schroder K, Kunde
D and Eri R: MCC950, a specific small molecule inhibitor of NLRP3
inflammasome attenuates colonic inflammation in spontaneous colitis
mice. Sci Rep. 8:86182018. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Ye J, Li L, Wang M, Ma Q, Tian Y, Zhang Q,
Liu J, Li B, Zhang B, Liu H, et al: Diabetes mellitus promotes the
development of atherosclerosis: The role of NLRP3. Front Immunol.
13:9002542022. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Franke M, Bieber M, Kraft P, Weber A,
Stoll G and Schuhmann MK: The NLRP3 inflammasome drives
inflammation in ischemia/reperfusion injury after transient middle
cerebral artery occlusion in mice. Brain Behav Immun. 92:223–233.
2021. View Article : Google Scholar
|
|
147
|
Zhai Y, Meng X, Ye T, Xie W, Sun G and Sun
X: Inhibiting the NLRP3 inflammasome activation with MCC950
ameliorates diabetic encephalopathy in db/db Mice. Molecules.
23:5222018. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Zhang C, Zhu X, Li L, Ma T, Shi M, Yang Y
and Fan Q: A small molecule inhibitor MCC950 ameliorates kidney
injury in diabetic nephropathy by inhibiting NLRP3 inflammasome
activation. Diabetes Metab Syndr Obes. 12:1297–1309. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Kuo LM, Kuo CY, Lin CY, Hung MF, Shen JJ
and Hwang TL: Intracellular glutathione depletion by oridonin leads
to apoptosis in hepatic stellate cells. Molecules. 19:3327–3344.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
He H, Jiang H, Chen Y, Ye J, Wang A, Wang
C, Liu Q, Liang G, Deng X, Jiang W, et al: Oridonin is a covalent
NLRP3 inhibitor with strong anti-inflammasome activity. Nat Commun.
9:25502018. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Gao RF, Li X, Xiang HY, Yang H, Lv CY, Sun
XL, Chen HZ, Gao Y, Yang JS, Luo W, et al: The covalent
NLRP3-inflammasome inhibitor Oridonin relieves myocardial
infarction induced myocardial fibrosis and cardiac remodeling in
mice. Int Immunopharmacol. 90:1071332021. View Article : Google Scholar
|
|
152
|
Jia Y, Tong Y, Min L, Li Y and Cheng Y:
Protective effects of oridonin against cerebral
ischemia/reperfusion injury by inhibiting the NLRP3 inflammasome
activation. Tissue Cell. 71:1015142021. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Yan C, Yan H, Mao J, Liu Y, Xu L, Zhao H,
Shen J, Cao Y, Gao Y, Li K, et al: Neuroprotective effect of
oridonin on traumatic brain injury via inhibiting NLRP3
inflammasome in experimental mice. Front Neurosci. 14:5571702020.
View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Liang L, Zheng Y, Xie Y, Xiao L and Wang
G: Oridonin ameliorates insulin resistance partially through
inhibition of inflammatory response in rats subjected to chronic
unpredictable mild stress. Int Immunopharmacol. 91:1072982021.
View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Marchetti C, Swartzwelter B, Koenders MI,
Azam T, Tengesdal IW, Powers N, de Graaf DM, Dinarello CA and
Joosten L: NLRP3 inflammasome inhibitor OLT1177 suppresses joint
inflammation in murine models of acute arthritis. Arthritis Res
Ther. 20:1692018. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Jankowsky JL, Fadale DJ, Anderson J, Xu
GM, Gonzales V, Jenkins NA, Copeland NG, Lee MK, Younkin LH, Wagner
SL, et al: Mutant presenilins specifically elevate the levels of
the 42 residue beta-amyloid peptide in vivo: Evidence for
augmentation of a 42-specific gamma secretase. Hum Mol Genet.
13:159–170. 2004. View Article : Google Scholar
|
|
157
|
Marchetti C, Swartzwelter B, Gamboni F,
Neff CP, Richter K, Azam T, Carta S, Tengesdal I, Nemkov T,
D'Alessandro A, et al: OLT1177, a β-sulfonyl nitrile compound, safe
in humans, inhibits the NLRP3 inflammasome and reverses the
metabolic cost of inflammation. Proc Natl Acad Sci USA.
115:E1530–E1539. 2018. View Article : Google Scholar
|
|
158
|
Lonnemann N, Hosseini S, Marchetti C,
Skouras DB, Stefanoni D, D'Alessandro A, Dinarello CA and Korte M:
The NLRP3 inflammasome inhibitor OLT1177 rescues cognitive
impairment in a mouse model of Alzheimer's disease. Proc Natl Acad
Sci USA. 117:32145–32154. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Sanchez-Fernandez A, Skouras DB, Dinarello
CA and Lopez-Vales R: OLT1177 (Dapansutrile), a selective NLRP3
inflammasome inhibitor, ameliorates experimental autoimmune
encephalomyelitis pathogenesis. Front Immunol. 10:25782019.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Oizumi T, Mayanagi T, Toya Y, Sugai T,
Matsumoto T and Sobue K: NLRP3 Inflammasome inhibitor OLT1177
suppresses onset of inflammation in mice with dextran sulfate
sodium-induced colitis. Digest Dis Sci. 67:2912–2921. 2022.
View Article : Google Scholar
|
|
161
|
Toldo S, Mauro AG, Cutter Z, Van Tassell
BW, Mezzaroma E, Del BM, Prestamburgo A, Potere N and Abbate A: The
NLRP3 inflammasome inhibitor, OLT1177 (Dapansutrile), reduces
infarct size and preserves contractile function after ischemia
reperfusion injury in the mouse. J Cardiovasc Pharm. 73:215–222.
2019. View Article : Google Scholar
|
|
162
|
Shi Y, Lv Q, Zheng M, Sun H and Shi F:
NLRP3 inflammasome inhibitor INF39 attenuated NLRP3 assembly in
macrophages. Int Immunopharmacol. 92:1073582021. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Cocco M, Pellegrini C, Martinez-Banaclocha
H, Giorgis M, Marini E, Costale A, Miglio G, Fornai M, Antonioli L,
Lopez-Castejon G, et al: Development of an acrylate derivative
targeting the NLRP3 inflammasome for the treatment of inflammatory
bowel disease. J Med Chem. 60:3656–3671. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Pu Z, Han C, Zhang W, Xu M, Wu Z, Liu Y,
Wu M, Sun H and Xie H: Systematic understanding of the mechanism
and effects of Arctigenin attenuates inflammation in dextran
sulfate sodium-induced acute colitis through suppression of NLRP3
inflammasome by SIRT1. Am J Transl Res. 11:3992–4009.
2019.PubMed/NCBI
|
|
165
|
Pellegrini C, Fornai M, Colucci R,
Benvenuti L, D'Antongiovanni V, Natale G, Fulceri F, Giorgis M,
Marini E, Gastaldi S, et al: A Comparative study on the efficacy of
NLRP3 inflammasome signaling inhibitors in a Pre-clinical model of
bowel inflammation. Front Pharmacol. 9:14052018. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Huang Y, Jiang H, Chen Y, Wang X, Yang Y,
Tao J, Deng X, Liang G, Zhang H, Jiang W, et al: Tranilast directly
targets NLRP3 to treat inflammasome-driven diseases. EMBO Mol Med.
10:e86892018. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Saeedi-Boroujeni A, Mahmoudian-Sani MR,
Nashibi R, Houshmandfar S, Tahmaseby GS and Khodadadi A: Tranilast:
A potential anti-Inflammatory and NLRP3 inflammasome inhibitor drug
for COVID-19. Immunopharm Immunot. 43:247–258. 2021. View Article : Google Scholar
|
|
168
|
Cao J and Peng Q: NLRP3 inhibitor
Tranilast attenuates gestational diabetes mellitus in a genetic
mouse model. Drugs R D. 22:105–112. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Chen S, Wang Y, Pan Y, Liu Y, Zheng S,
Ding K, Mu K, Yuan Y, Li Z, Song H, et al: Novel role for Tranilast
in regulating NLRP3 ubiquitination, vascular inflammation, and
atherosclerosis. J Am Heart Assoc. 9:e155132020. View Article : Google Scholar
|
|
170
|
Shen K, Jiang W, Zhang C, Cai L, Wang Q,
Yu H, Tang Z, Gu Z and Chen B: Molecular mechanism of a specific
NLRP3 inhibitor to alleviate seizure severity induced by
pentylenetetrazole. Curr Mol Pharmacol. 14:579–586. 2021.
View Article : Google Scholar
|
|
171
|
Marchetti C, Toldo S, Chojnacki J,
Mezzaroma E, Liu K, Salloum FN, Nordio A, Carbone S, Mauro AG, Das
A, et al: Pharmacologic inhibition of the NLRP3 inflammasome
preserves cardiac function after ischemic and nonischemic injury in
the mouse. J Cardiovasc Pharm. 66:1–8. 2015. View Article : Google Scholar
|
|
172
|
Kuwar R, Rolfe A, Di L, Xu H, He L, Jiang
Y, Zhang S and Sun D: A novel small molecular NLRP3 inflammasome
inhibitor alleviates neuroinflammatory response following traumatic
brain injury. J Neuroinflamm. 16:812019. View Article : Google Scholar
|
|
173
|
Kuwar R, Rolfe A, Di L, Blevins H, Xu Y,
Sun X, Bloom GS, Zhang S and Sun D: A novel inhibitor targeting
NLRP3 inflammasome reduces neuropathology and improves cognitive
function in Alzheimer's disease transgenic mice. J Alzheimers Dis.
82:1769–1783. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Yin J, Zhao F, Chojnacki JE, Fulp J, Klein
WL, Zhang S and Zhu X: NLRP3 Inflammasome inhibitor ameliorates
amyloid pathology in a mouse model of Alzheimer's disease. Mol
Neurobiol. 55:1977–1987. 2018. View Article : Google Scholar
|
|
175
|
Fulp J, He L, Toldo S, Jiang Y, Boice A,
Guo C, Li X, Rolfe A, Sun D, Abbate A, et al: Structural insights
of Benzenesulfonamide analogues as NLRP3 inflammasome inhibitors:
Design, synthesis, and biological characterization. J Med Chem.
61:5412–5423. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
He Y, Varadarajan S, Munoz-Planillo R,
Burberry A, Nakamura Y and Nunez G:
3,4-methylenedioxy-β-nitrostyrene inhibits NLRP3 inflammasome
activation by blocking assembly of the inflammasome. J Biol Chem.
289:1142–1150. 2014. View Article : Google Scholar
|
|
177
|
Juliana C, Fernandes-Alnemri T, Wu J,
Datta P, Solorzano L, Yu JW, Meng R, Quong AA, Latz E, Scott CP, et
al: Anti-inflammatory compounds parthenolide and Bay 11-7082 are
direct inhibitors of the inflammasome. J Biol Chem. 285:9792–9802.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Shim DW, Shin WY, Yu SH, Kim BH, Ye SK,
Koppula S, Won HS, Kang TB and Lee KH: BOT-4-one attenuates NLRP3
inflammasome activation: NLRP3 alkylation leading to the regulation
of its ATPase activity and ubiquitination. Sci Rep. 7:150202017.
View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Zheng D, Liwinski T and Elinav E:
Inflammasome activation and regulation: Toward a better
understanding of complex mechanisms. Cell Discov. 6:362020.
View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Li Y, Niu X, Xu H, Li Q, Meng L, He M,
Zhang J and Zhang Z and Zhang Z: VX-765 attenuates atherosclerosis
in ApoE deficient mice by modulating VSMCs pyroptosis. Exp Cell
Res. 389:1118472020. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Zhang Z, Bai H, Ma X, Shen M, Li R, Qiu D,
Li S and Gao L: Blockade of the NLRP3/caspase-1 axis attenuates
ketamine-induced hippocampus pyroptosis and cognitive impairment in
neonatal rats. J Neuroinflamm. 18:2392021. View Article : Google Scholar
|
|
182
|
Liang Y, Song P, Chen W, Xie X, Luo R, Su
J, Zhu Y, Xu J, Liu R, Zhu P, et al: Inhibition of Caspase-1
Ameliorates Ischemia-Associated Blood-Brain barrier dysfunction and
integrity by suppressing pyroptosis activation. Front Cell
Neurosci. 14:5406692020. View Article : Google Scholar
|
|
183
|
Gu L, Sun M, Li R, Zhang X, Tao Y, Yuan Y,
Luo X and Xie Z: Didymin suppresses microglia pyroptosis and
neuroinflammation through the Asc/Caspase-1/GSDMD pathway following
experimental intracerebral hemorrhage. Front Immunol.
13:8105822022. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Tian DD, Wang M, Liu A, Gao MR, Qiu C, Yu
W, Wang WJ, Zhang K, Yang L, Jia YY, et al: Antidepressant effect
of paeoniflorin is through inhibiting pyroptosis CASP-11/GSDMD
pathway. Mol Neurobiol. 58:761–776. 2021. View Article : Google Scholar
|
|
185
|
Wang F, Liang Q, Ma Y, Sun M, Li T, Lin L,
Sun Z and Duan J: Silica nanoparticles induce pyroptosis and
cardiac hypertrophy via ROS/NLRP3/Caspase-1 pathway. Free Radical
Bio Med. 182:171–181. 2022. View Article : Google Scholar
|
|
186
|
Xu S, Li X, Liu Y, Xia Y, Chang R and
Zhang C: Inflammasome inhibitors: Promising therapeutic approaches
against cancer. J Hematol Oncol. 12:642019. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Wang F, Li G, Ning J, Chen L, Xu H, Kong
X, Bu J, Zhao W, Li Z, Wang X, et al: Alcohol accumulation promotes
esophagitis via pyroptosis activation. Int J Biol Sci.
14:1245–1255. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Qiu Z, He Y, Ming H, Lei S, Leng Y and Xia
ZY: Lipopolysaccharide (LPS) aggravates high glucose- and
Hypoxia/Reoxygenation-induced injury through activating
ROS-dependent NLRP3 inflammasome-mediated pyroptosis in H9C2
Cardiomyocytes. J Diabetes Res. 2019:81518362019. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Liang H, Sun Y, Gao A, Zhang N, Jia Y,
Yang S, Na M, Liu H, Cheng X, Fang X, et al: Ac-YVAD-cmk improves
neurological function by inhibiting caspase-1-mediated inflammatory
response in the intracerebral hemorrhage of rats. Int
Immunopharmacol. 75:1057712019. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Pan P, Shen M, Yu Z, Ge W, Chen K, Tian M,
Xiao F, Wang Z, Wang J, Jia Y, et al: SARS-CoV-2 N protein promotes
NLRP3 inflammasome activation to induce hyperinflammation. Nat
Commun. 12:46642021. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Zheng X, Chen W, Gong F, Chen Y and Chen
E: The role and mechanism of pyroptosis and potential therapeutic
targets in sepsis: A review. Front Immunol. 12:7119392021.
View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Liu W, Guo W, Zhu Y, Peng S, Zheng W,
Zhang C, Shao F, Zhu Y, Hang N, Kong L, et al: Targeting
Peroxiredoxin 1 by a Curcumin analogue, AI-44, inhibits NLRP3
inflammasome activation and attenuates lipopolysaccharide-induced
sepsis in mice. J Immunol. 201:2403–2413. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Liu W, Guo W, Wu J, Luo Q, Tao F, Gu Y,
Shen Y, Li J, Tan R, Xu Q and Sun Y: A novel benzo[d]imidazole
derivate prevents the development of dextran sulfate sodium-induced
murine experimental colitis via inhibition of NLRP3 inflammasome.
Biochem Pharmacol. 85:1504–1512. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Sborgi L, Ruhl S, Mulvihill E, Pipercevic
J, Heilig R, Stahlberg H, Farady CJ, Muller DJ, Broz P and Hiller
S: GSDMD membrane pore formation constitutes the mechanism of
pyroptotic cell death. EMBO J. 35:1766–1778. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Shi J, Zhao Y, Wang K, Shi X, Wang Y,
Huang H, Zhuang Y, Cai T, Wang F and Shao F: Cleavage of GSDMD by
inflammatory caspases determines pyroptotic cell death. Nature.
526:660–665. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Liu Z, Gan L, Xu Y, Luo D, Ren Q, Wu S and
Sun C: Melatonin alleviates inflammasome-induced pyroptosis through
inhibiting NF-κB/GSDMD signal in mice adipose tissue. J Pineal Res.
63:2017. View Article : Google Scholar
|
|
197
|
Sun L, Wang H, Wang Z, He S, Chen S, Liao
D, Wang L, Yan J, Liu W, Lei X and Wang X: Mixed lineage kinase
domain-like protein mediates necrosis signaling downstream of RIP3
kinase. Cell. 148:213–227. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Rathkey JK, Zhao J, Liu Z, Chen Y, Yang J,
Kondolf HC, Benson BL, Chirieleison SM, Huang AY, Dubyak GR, et al:
Chemical disruption of the pyroptotic pore-forming protein
gasdermin D inhibits inflammatory cell death and sepsis. Sci
Immunol. 3:eaat27382018. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Han C, Yang Y, Guan Q, Zhang X, Shen H,
Sheng Y, Wang J, Zhou X, Li W, Guo L, et al: New mechanism of nerve
injury in Alzheimer's disease: β-amyloid-induced neuronal
pyroptosis. J Cell Mol Med. 24:8078–8090. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Teng JF, Mei QB, Zhou XG, Tang Y, Xiong R,
Qiu WQ, Pan R, Law BY, Wong VK, Yu CL, et al: Polyphyllin VI
induces Caspase-1-mediated Pyroptosis via the induction of
ROS/NF-κB/NLRP3/GSDMD signal axis in Non-small cell lung cancer.
Cancers (Basel). 12:1932020. View Article : Google Scholar
|
|
201
|
Guo W, Chen S, Li C, Xu J and Wang L:
Application of Disulfiram and its metabolites in treatment of
inflammatory disorders. Front Pharmacol. 12:7950782021. View Article : Google Scholar
|
|
202
|
Wu J, Zhang J, Zhao J, Chen S, Zhou T and
Xu J: Treatment of severe acute pancreatitis and related lung
injury by targeting gasdermin D-mediated pyroptosis. Front Cell Dev
Biol. 9:7801422021. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Pandeya A, Li L, Li Z and Wei Y: Gasdermin
D (GSDMD) as a new target for the treatment of infection.
Medchemcomm. 10:660–667. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Hu JJ, Liu X, Xia S, Zhang Z, Zhang Y,
Zhao J, Ruan J, Luo X, Lou X, Bai Y, et al: FDA-approved disulfiram
inhibits pyroptosis by blocking gasdermin D pore formation. Nat
Immunol. 21:736–745. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Zhou Q, Wang W, Yang F, Wang H, Zhao X,
Zhou Y, Fu P and Xu Y: Disulfiram suppressed Peritendinous fibrosis
through inhibiting macrophage accumulation and its pro-inflammatory
properties in tendon bone healing. Front Bioeng Biotech.
10:8239332022. View Article : Google Scholar
|
|
206
|
Yan H, Yang H, Wang L, Sun X, Han L, Cong
P, Chen X, Lu D and Che C: Disulfiram inhibits IL-1β secretion and
inflammatory cells recruitment in Aspergillus fumigatus keratitis.
Int Immunopharmacol. 102:1084012022. View Article : Google Scholar
|
|
207
|
Cattani-Cavalieri I, Da MVH, Moraes JA,
Brito-Gitirana L, Romana-Souza B, Schmidt M and Valenca SS:
Dimethyl fumarate attenuates lung inflammation and oxidative stress
induced by chronic exposure to diesel exhaust particles in mice.
Int J Mol Sci. 21:96582020. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Humphries F, Shmuel-Galia L,
Ketelut-Carneiro N, Li S, Wang B, Nemmara VV, Wilson R, Jiang Z,
Khalighinejad F, Muneeruddin K, et al: Succination inactivates
gasdermin D and blocks pyroptosis. Science. 369:1633–1637. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Muhammad JS, Jayakumar MN, Elemam NM,
Venkatachalam T, Raju TK, Hamoudi RA and Maghazachi AA: Gasdermin D
Hypermethylation inhibits pyroptosis and LPS-Induced IL-1β release
from NK92 cells. Immunotargets Ther. 8:29–41. 2019. View Article : Google Scholar :
|
|
210
|
Xia L, Liu L, Cai Y, Zhang Y, Tong F, Wang
Q, Ding J and Wang X: Inhibition of Gasdermin D-mediated pyroptosis
attenuates the severity of seizures and astroglial damage in kainic
Acid-induced epileptic mice. Front Pharmacol. 12:7516442021.
View Article : Google Scholar
|
|
211
|
Sohn E, Kim J, Kim CS, Jo K and Kim JS:
Osteomeles schwerinae extract prevents diabetes-induced renal
injury in spontaneously diabetic Torii rats. Evid Based Complement
Alternat Med. 2018:68242152018. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Soma J, Sugawara T, Huang YD, Nakajima J
and Kawamura M: Tranilast slows the progression of advanced
diabetic nephropathy. Nephron. 92:693–698. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Soma J, Sato K, Saito H and Tsuchiya Y:
Effect of Tranilast in early-stage diabetic nephropathy. Nephrol
Dial Transpl. 21:2795–2799. 2006. View Article : Google Scholar
|
|
214
|
Kosuga K, Tamai H, Ueda K, Hsu YS, Ono S,
Tanaka S, Doi T, Myou-U W, Motohara S and Uehata H: Effectiveness
of Tranilast on restenosis after directional coronary atherectomy.
Am Heart J. 134:712–718. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Wohlford GF, Van Tassell BW, Billingsley
HE, Kadariya D, Canada JM, Carbone S, Mihalick VL, Bonaventura A,
Vecchie A, Chiabrando JG, et al: Phase 1B, randomized,
Double-blinded, dose escalation, single-center, repeat dose safety
and pharmacodynamics study of the oral NLRP3 inhibitor dapansutrile
in subjects with NYHA II-III systolic heart failure. J Cardiovasc
Pharm. 77:49–60. 2020. View Article : Google Scholar
|
|
216
|
Kim M, Byun J, Chung Y, Lee SU, Park JE,
Park W, Park JC, Ahn JS and Lee S: Reactive Oxygen species
scavenger in acute intracerebral hemorrhage patients: A
multicenter, randomized controlled trial. Stroke. 52:1172–1181.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Masnadi SK, Sotoudeh S, Masnadi SA,
Moaddab SY, Nourpanah Z and Nikniaz Z: Effect of N-acetylcysteine
on remission maintenance in patients with ulcerative colitis: A
randomized, double-blind controlled clinical trial. Clin Res
Hepatol Gas. 45:1015322021. View Article : Google Scholar
|
|
218
|
Reinhardt S, Stoye N, Luderer M, Kiefer F,
Schmitt U, Lieb K and Endres K: Identification of disulfiram as a
Secretase-modulating compound with beneficial effects on
Alzheimer's disease hallmarks. Sci Rep. 8:13292018. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Yuan XZ, Sun S, Tan CC, Yu JT and Tan L:
The role of ADAM10 in Alzheimer's disease. J Alzheimers Dis.
58:303–322. 2017. View Article : Google Scholar : PubMed/NCBI
|