Introduction
Liver transplantation is the treatment of choice for
end-stage liver disease (1).
However, the rising prevalence of liver disease has led to a severe
shortage of donor organs, which limits the broader clinical
application of this procedure (2). The estimated global
age-standardized liver disease-associated mortality rates increased
from 1990 to 2021 across 112 countries, rising from 103.4 deaths
per 1,000,000 people in 1990 to 173.0 deaths per 1,000,000 people
in 2021 (3). Donation after
circulatory death (DCD) has expanded the donor pool by using
marginal grafts, which refer to organs with higher transplant risks
and borderline quality due to factors such as donor age,
pathological conditions, or prolonged ischemia time (4). However, DCD liver grafts are
vulnerable to more severe ischemia-reperfusion injury (IRI). IRI in
liver transplantation refers to tissue damage that occurs during
the ischemic period and is exacerbated following restoration of
blood flow. This triggers an inflammatory response, impairs hepatic
microcirculation and induces hepatocyte injury, thereby hindering
the functional recovery and long-term survival of the transplanted
liver (5,6).
To mitigate IRI, several dynamic graft preservation
techniques have been developed, with hypothermic machine perfusion
(HMP) serving as one of the most promising strategies (7). Compared with static cold storage
(CS), HMP better protects donor organs, decreases the severity of
IRI and promotes graft repair, thereby helping to alleviate the
organ shortage crisis (8).
However, the protective mechanism of HMP against IRI in DCD liver
grafts has not yet been fully elucidated.
Kruppel-like factor 2 (KLF2) is a transcription
factor that maintains endothelial cell (EC) quiescence. Its
expression in ECs is upregulated in response to physiological
laminar shear stress (LSS) from blood flow, activating a
transcriptional program that confers vascular protection (9). Conversely, under CS conditions,
where hemodynamic stimulation is absent, the endothelial
KLF2-mediated protective pathway is significantly downregulated,
leading to loss of the vascular protective phenotype (10,11). Previous studies have demonstrated
that during organ preservation, HMP maintains a form of
physiological laminar flow, which upregulates KLF2 expression in
ECs and exerts protective effects (12,13). Our preliminary study has also
confirmed that pharmacologically induced overexpression of KLF2 in
donors can alleviate IRI in rat DCD livers (14). Yet, the specific role of KLF2
during HMP in the context of DCD liver IRI requires further
elucidation.
Macrophage efferocytosis refers to the process by
which macrophages efficiently clear apoptotic cells, thereby
preventing a larger inflammatory cascade and maintaining immune
balance and organismal stability (15). This process also plays a key role
in modulating hepatic IRI (16,17). Macrophages can adapt their
phenotype and function in response to signals from the
microenvironment (18,19). Such microenvironmental cues
typically originate from intercellular communication. For example,
ECs shape macrophage phenotypes through the secretion of paracrine
factors (20,21). Exosomes secreted by vascular ECs
under physiologically protective LSS polarizes macrophages toward
an anti-inflammatory phenotype (22). Moreover, exosomes derived from
KLF2-upregulated ECs modulate macrophage behavior regulate
macrophages by enhancing immune-modulatory responses and
attenuating pro-inflammatory responses (23). Additionally, in atherosclerosis,
disturbed blood flow leads to the downregulation of KLF2 expression
(24,25), which is associated with impaired
macrophage efferocytosis (26-28). Simvastatin and resveratrol
enhance macrophage efferocytosis (29,30). It was hypothesized that
upregulation of EC KLF2 expression during HMP may alter the
microenvironment via paracrine signaling, thereby enhancing
macrophage efferocytosis. This mechanism may represent a novel
pathway contributing to the protective effects of HMP on DCD liver
grafts.
Materials and methods
Animals and treatments
A total of 48 adult male Sprague-Dawley (SD) rats
aged 8-10 weeks (body weight, 250-300 g) were purchased from Hubei
Research Center of Laboratory Animals. They were housed under room
temperature of 20-25°C, humidity of 50-70% and a 12/12-h light-dark
cycle. They were fed standard chow and provided with adequate water
freely and housed in a specific pathogen-free facility. All animal
procedures were conducted according to the Guide for the Care and
Use of Laboratory Animals published by the National Research
Council (31) and approved by
the Institutional Animal Care and Use Committee of the First
Affiliated Hospital of Nanchang University (Nanchang, China;
approval nos. CDYFY-IACUC-202211QR219 and
CDYFY-IACUC-202407QR199).
As previously described (5), a DCD rat model was established.
Briefly, rats were anesthetized by intraperitoneal injection of 30
mg/kg sodium pentobarbital. Without portal vein clamping or prior
heparinization, the diaphragm was transected to induce cardiac
arrest. Rats were placed on a thermostatic heating pad to maintain
liver temperature at 29±1.45°C. After 30 min, in situ
perfusion of the liver was performed at 4°C using 50 ml
histidine-tryptophan-ketoglutarate (HTK) solution through the
abdominal aorta to flush out the blood. The portal vein and
suprahepatic inferior vena cava were cannulated, while the
infrahepatic inferior vena cava and right adrenal vein were
ligated. Finally, the liver was excised.
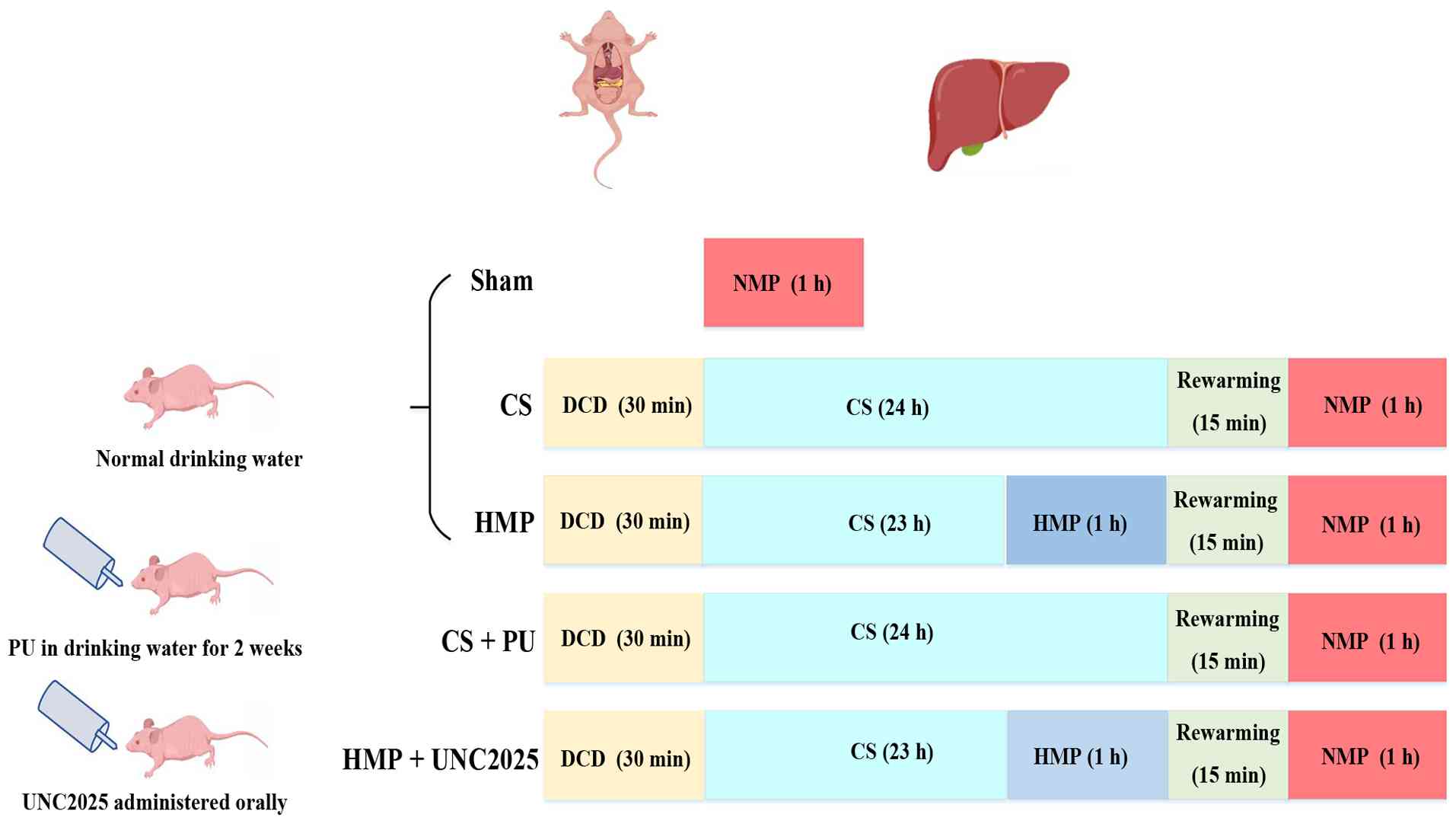
Rats were randomly divided into groups (n=6/group;
Fig. 1): Sham, cardiac arrest
was not induced, but all other procedures were performed as
aforementioned; CS, following DCD modeling, the isolated liver was
preserved in a 4°C University of Wisconsin (UW) solution for 24 h;
HMP, liver was preserved in a 4°C UW solution for 23 h, followed by
1 h HMP and 1 h normothermic perfusion (NMP); CS + putrescine (PU),
rats were provided with drinking water supplemented with 3 mM PU
(32) (cat. no. HY-Y1781;
MedChemExpress); and HMP + UNC2025, SD rats received a single
intraperitoneal injection of the c-mer tyrosine kinase (MerTK)
inhibitor UNC2025 (cat. no. HY-12344; MedChemExpress) at a dose of
10 mg/kg 1 h prior to DCD induction. Subsequently, the livers were
processed, preserved, and perfused as aforementioned. The HMP and
isolated perfused rat liver system have been described previously
(33). Portal vein perfusion
pressure was 4 mmHg, the flow rate was controlled at 0.1 ml/g·min,
and the HMP duration was 1 h. Under NMP, the perfusate temperature
was maintained at 37±1°C, the portal vein perfusion pressure was ≤8
mmHg, the perfusion flow rate was controlled at ~1 ml/g·min, and
the reperfusion duration was 1 h (33,34). HMP was conducted using UW
solution, while reperfusion was performed with HTK solution. At the
end of NMP, perfusate and liver tissue samples were collected for
further analysis. The water for both the control and
PU-supplemented groups was changed every 2-3 days. After 2 weeks,
venous blood was collected via inferior vena cava puncture under
anesthesia (administered via intraperitoneal injection of 30 mg/kg
pentobarbital sodium), with ~1 ml drawn per animal for liver
function analysis, followed by liver retrieval as
aforementioned.
Euthanasia was performed by an overdose of sodium
pentobarbital (>100 mg/kg, administered intraperitoneally) to
ensure a painless procedure and compliance with animal ethics
guidelines. The rats were checked for cardiac arrest, respiratory
cessation, body stiffness and pupillary dilation to confirm
death.
Liver injury assessment
The collected serum and perfusate following
normothermic reperfusion were stored at −80°C. Alanine transferase
(ALT) and aspartate transferase (AST) concentration in rat serum
and perfusate was assessed using the BS-2000 automatic biochemical
analyzer (Shenzhen Mindray Bio-Medical Electronics Co., Ltd.). The
liver tissues were fixed using 4% buffered paraformaldehyde at room
temperature for 24 h. The paraffin-embedded tissues were sectioned
at a thickness of 4 μm. Staining was performed with
hematoxylin for 5 min and eosin for 15 min, both at room
temperature. Finally, morphological observation and image
acquisition were conducted under a light microscope and graded
using the Suzuki scoring system (35) in a blinded manner. Suzuki scoring
system was assessed on a scale from 0 to 4 based on the severity of
cellular vacuolization, sinusoidal congestion and hepatocyte
necrosis.
TUNEL assay and in situ efferocytosis
measurement
Following fixation in 4% paraformaldehyde (24 h,
room temperature), the paraffin-embedded liver sections were
stained for hepatocyte apoptosis using the terminal
deoxynucleotidyl TUNEL assay kit (cat. no. G1504; Wuhan Servicebio
Technology Co., Ltd.). Immunofluorescence (IF) staining was
performed using the TUNEL assay kit and anti-CD68 antibody
(1:1,000; cat. no. ab303565, Abcam). The sections were incubated
with TUNEL staining reagent at room temperature for 60 min,
followed by three washes with PBS. Sections were blocked with 10%
normal goat serum at room temperature for 30 min (Beijing Solarbio
Science & Technology Co., Ltd.) and incubated overnight at 4°C
with the anti-CD68 antibody. Finally, the slides were incubated
with a fluorescently labeled secondary antibody (1:1,000; cat. no.
ab205718; Abcam) at 37°C for 1 h and counterstained with DAPI
before applying antifade mounting medium (cat. no. 0100-01,
SouthernBiotech). Fluorescence microscopy (Olympus Corporation) was
used to observe the slides and TUNEL+ nuclei associated
with CD68+ macrophages were counted to quantify in
situ efferocytosis. Number of macrophages per field of view:
30-50. All researchers responsible were blinded to the group
allocation.
Immunohistochemistry (IHC) and IF
staining
Following fixation in 4% paraformaldehyde (24 h,
room temperature), rat liver tissues were embedded in paraffin and
cut into 4 μm-thick sections. Paraffin-embedded specimens
were sectioned, deparaffinized with xylene and rehydrated in
decreasing concentrations of ethanol. Add proteinase K working
solution, then incubate the slides at 37°C for 20 min for antigen
retrieval. Endogenous enzymes were blocked by incubating with 3%
H2O2 at room temperature for 20 min. The
slides were blocked with 10% normal goat serum at room temperature
for 1 h for IHC staining. The slides were incubated overnight at
4°C with the primary antibody. The HRP-conjugated secondary
antibody Goat Anti-Rabbit IgG H&L (1:1,000; cat. no. ab205718,
Abcam) was applied for 1 h at room temperature. The primary
antibodies were as follows: Anti-caspase 3 (1:200; cat. no. 9661,
Cell Signaling Technology, Inc.), anti-myeloperoxidase (MPO;
1:1,000; cat. no. ab208670, Abcam), anti-KLF2 (1:200; cat. no.
bs-2772R, BIOSS) and anti-T cell immunoglobulin and mucin
domain-containing protein 4 (TIMD4; 1:200; cat. no. a17807,
ABclonal Biotech Co., Ltd.). The DAB working solution was then
applied to the tissue sections for chromogenic development. Cell
nuclei were counterstained with Mayer's hematoxylin (cat. no.
G1080; Beijing Solarbio Science & Technology Co., Ltd.) for 1
min at room temperature, and the slides were observed under a
microscope. The staining results were quantitatively analyzed using
the image analysis software Image J (6.0, National Institutes of
Health).
Transmission electron microscopy
(TEM)
Fresh liver tissue and cell samples (size, 1-2
mm2) were fixed in 2.5% glutaraldehyde (Wuhan Servicebio
Technology Co., Ltd.) at 4°C for 24 h. Cell samples were pelleted
by centrifugation at 2,000 × g (4°C, 5 min) in an EP tube. The
supernatant and glutaraldehyde fixative were removed. A total of ~1
mm3 of melted 1% agarose was added to the pellet and
gently mixed. After cooling and solidification, the resulting
agarose-embedded cell block was processed for subsequent steps. The
samples were dehydrated through a gradient of ethanol and acetone
and embedded (37°C, overnight) in epoxy resin. Samples were
sectioned into thin slices (60-80 nm), which were placed on copper
grids. Finally, the sections were performed with 2% uranyl acetate
(room temperature) for 8 min, followed by 2% lead citrate (both
room temperature) for 8 min. The images were analyzed using TEM
(cat. no. JEM1400, JEOL Ltd.). All researchers were blinded to the
group allocation information.
Cell culture and treatment
Human umbilical vein ECs (PUMC-HUVEC-T1, cat. no.
CL-0675), THP-1 (human monocytic leukemia cell line; cat. no.
CL-0233) and Jurkat (cat. no. CL-0129; all Procell Life Science
& Technology Co., Ltd.) cells were cultured at 37°C in a 5%
CO2 humidified incubator. HUVECs were cultured in DMEM
(cat. no. G4511; Wuhan Servicebio Technology co., Ltd.) with high
glucose, supplemented with 1% penicillin/streptomycin and 10% fetal
bovine serum (cat. no. A5256701; Gibco; Thermo Fisher Scientific,
Inc.). THP-1 and Jurkat cells were cultured in THP-1 cell culture
medium (cat. no. CM-0233) and Jurkat cell culture medium (cat. no.
CM-0129; both Procell Life Science & Technology Co., Ltd.),
respectively. THP-1 cells were cultured (37°C) in medium
supplemented with 100 ng/ml phorbol 12-myristate 13-acetate (PMA;
cat. no. HY-18739, MedChemExpress) for 48 h to induce
differentiation into macrophages. To establish the CS/reperfusion
(CS/Rep) model, the cell culture medium was replaced with UW
solution and the cells were incubated at 4°C for 24 h.
Subsequently, the solution was replaced with high-glucose DMEM)
(cat. no. G4511; Wuhan Servicebio Technology co., Ltd.) containing
FBS (cat. no. A5256701; Gibco; Thermo Fisher Scientific, Inc; 10%).
and the cells were incubated at 37°C with 5% CO2 for 3 h
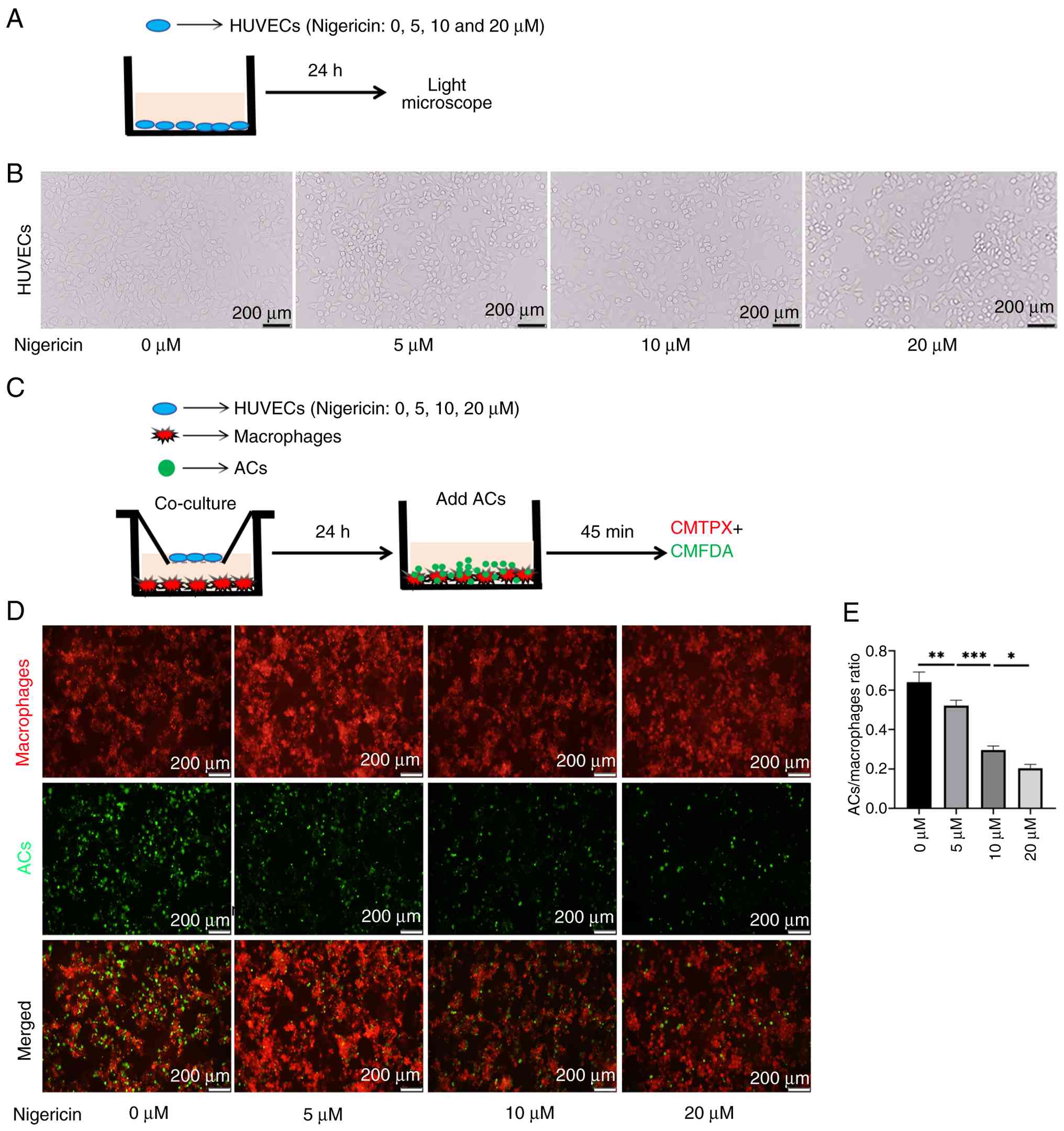
to simulate the reperfusion process. Nigericin was dissolved in
DMSO and diluted in DMEM containing 10% FBS. To verify that the
pyroptosis of ECs affects macrophage phagocytosis, HUVECs were
treated (37°C, 24 h) with nigericin (cat. no. HY-100381,
MedChemExpress; 0, 5, 10 and 20 μM) to induce
pyroptosis.
In vitro shear stress experiment
construction
HUVECs (1×106) were seeded on
collagen-coated glass slides to form a monolayer. As previously
described (36), the slides were
placed into a flow chamber system, and DMEM supplemented with 10%
FBS (Gibco; Thermo Fisher Scientific, Inc.) was injected into the
chamber to cover the ECs. The flow rate was regulated using a
unidirectional flow pump and ECs on the slides were exposed to LSS
at 12 dyne/cm2 for 0, 3, 6 and 12 h before performing
cell viability assay.
Lentivirus construction
The recombinant lentivirus for KLF2 overexpression
(ov-KLF2) was designed and constructed by OBiO Technology
(Shanghai) Corp., Ltd. KLF2 knockdown was achieved using lentiviral
particles expressing short hairpin RNA targeting KLF2 (sh-KLF2)
with vector backbone (cat. no. GV493; both Shanghai GeneChem Co.,
Ltd.), and the concentration of nucleic acid used was 20 μg.
The sequence of sh-KLF2 was 5'-GCCTTCGGTCTCTTCGACGAC; the
sequence-3' of sh-control is 5'-TTCTCCGAACGTGTCACGT-3'. To
transduce HUVECs, the lentivirus was added to wells containing
cells in serum-free and antibiotic-free DMEM along with the
transfection reagent Lipofectamine® (cat. no. 11668019;
Thermo Fisher Scientific). The mixture was incubated at 37°C for 48
h. Stable cell lines were obtained following puromycin (cat. no.
BL528A, Biosharp Life Sciences) selection for subsequent
experiments. The incubation period between transfection and
subsequent treatment was 72 h. To verify whether NLRP3 inhibition
rescues the phenotypes resulting from KLF2 deficiency,
sh-KLF2-transfected cells were treated (37°C) with 10 μM
NLRP3 inhibitor MCC950 (cat. no. HY12815, MedChemExpress) for 24 h
and subjected to CS/Rep as aforementioned. Expression levels of
pyroptosis-associated proteins were assessed by western
blotting.
Cell viability assay
Treated HUVECs were washed three times with PBS,
then incubated (37°C with 5% CO2) with
calcein-AM/propidium iodide (PI; cat. no. L6037, Suzhou Youyilandi
Biotechnology Co., Ltd.) for 30 min, according to the
manufacturer's instructions. Following fixation with 4%
formaldehyde at room temperature for 15 min, the cells were
observed under a fluorescence microscope.
Flow cytometry
1×105 HUVECs were collected in flow tubes
and centrifuged at 1,000 × g at 4°C for 5 min. The supernatant was
discarded, and the cells were resuspended in pre-cooled PBS.
Following an additional centrifugation step as aforementioned, the
cell pellet was resuspended in 1X binding buffer) (Wuhan Servicebio
Technology Co., Ltd.). The Annexin V-FITC/PI Apoptosis Detection
kit (Wuhan Servicebio Technology Co., Ltd.) was used to measure
early and late apoptosis of HUVECs. Briefly, cells were stained
with Annexin V-FITC/PI at 37°C for 30 min. HUVECs were washed twice
with PBS and the stained cells were resuspended in cold PBS.
Analysis was performed using a flow cytometer (CytoFlex S, Beckman
Coulter, Inc.) and data were analyzed using FlowJo software (v10.8;
BD Biosciences).
Indirect co-culture
In a Transwell co-culture system, 0.2 ml HUVECs at a
concentration of 1×104 cells/ml was added to the upper
chamber, while 0.5 ml PMA-induced macrophages (THP-1 derived) at a
concentration of 1×104 cells/ml was seeded in the lower
chamber. High-glucose DMEM supplemented with 10% FBS was used as
the culture medium for both monoculture and co-culture conditions.
All groups were subjected to indirect co-culture. HUVECs in the
upper chamber were removed, while macrophages in the lower chamber
were retained for phagocytosis assay.
Induction of apoptosis in Jurkat cells
and in vitro phagocytosis assay
To induce apoptosis in Jurkat cells, the cells were
exposed to UV light (254 nm) for 15 min, followed by incubation
under normal cell culture conditions for 2-3 h to obtain apoptotic
Jurkat cells (ACs) (37). ACs
were labeled with 5-chloromethylfluorescein diacetate (CMFDA) (cat.
no. 40721ES) and macrophages were labeled with CellTracker™ Red
CMTPX dye (CMTPX) (cat. no. 40717ES; both Shanghai Yeasen
Biotechnology Co., Ltd.), according to the manufacturer's
instructions. Labeled ACs were added to the macrophages at a ratio
of 5:1, followed by co-incubation (37°C, 5% CO2) for 45
min. Cells were washed three times with PBS to remove unbound ACs.
The macrophages were fixed (37°C) with 4% formaldehyde for 20 min,
followed by three washes with PBS. The ability of macrophages to
phagocytose ACs was visualized using IF microscopy. For microscopic
analysis, ≥3 fields of view per sample were examined, and ≥30
macrophages/field were evaluated. The phagocytic index was
calculated using the following formula: (Number of macrophages
containing apoptotic cells/total number of macrophages) ×100%. Data
were normalized to the control group (set to 100%).
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was extracted from liver tissue and HUVECs
using TRIzol (cat. no. 15596026CN; Thermo Fisher Scientific, Inc.).
RT was performed using Hifair®V one-step RT-gDNA
digestion SuperMix kit (cat. no. 11142ES10; Shanghai Yeasen
Biotechnology co., Ltd.). as follows: Primer annealing at 30°C for
5 min, cDNA synthesis 55°C for 30 min and the reaction was
terminated by heating at 85°C for 5 min. SYBR Green (Biosharp Life
Sciences) was used for qPCR. Thermocycling conditions were as
follows: Initial denaturation at 95°C for 5 min, followed by 40
cycles of 95°C for 10 sec (denaturation) and 60°C for 30 sec
(combined annealing/extension), with fluorescence signal
measurement at the end of each extension step. β-actin was used as
the reference gene. Relative expression of target genes was
calculated according to the 2−ΔΔCq method (38). The primers for all target genes
are listed in Table I.
 | Table IPrimer sequences of the target
genes. |
Table I
Primer sequences of the target
genes.
| Gene | Primer name | Primer sequence,
5'➔3' |
|---|
| IL-1β (rat) | Forward |
AATCTCACAGCAGCATCTCGACAAG |
| Reverse |
TCCACGGGCAAGACATAGGTAGC |
| IL-6 (rat) | Forward |
ACTTCCAGCCAGTTGCCTTCTTG |
| Reverse |
TGGTCTGTTGTGGGTGGTATCCTC |
| TNF-α (rat) | Forward |
AAAGGACACCATGAGCACGGAAAG |
| Reverse C |
GCCACGAGCAGGAATGAGAAG |
| KLF2 (human) | Forward |
GGCTGCGGCTGGAAGTTTG |
| Reverse |
GCACAGATGGCACTGGAATGG |
| NLRP3 (human) | Forward |
GCTTGCCGACGATGCCTTCC |
| Reverse |
GCTGTCATTGTCCTGGTGTCTTCC |
| β-actin
(human) | Forward |
CAGATGTGGATCAGCAAGCAGGAG |
| Reverse C |
GCAACTAAGTCATAGTCCGCCTAG |
| β-actin (rat) | Forward |
GCTGTGCTATGTTGCCCTAGACTTC |
| Reverse |
GGAACCGCTCATTGCCGATAGTG |
Western blotting
The western blotting procedure was performed as
previously described (6). The
primary antibodies were as follows: Anti-KLF2 (1:1,000; cat. no.
23384-1-AP, Proteintech Group, Inc.), anti-KLF2 (1:1,000; cat. no.
PAB40163; Bioswamp; Wuhan Bienle Biotechnology Co., Ltd.), anti-BAX
(1:1,000; cat. no. 50599-2-Ig), anti-BCL-2 (1:1,000; cat. no.
60178-1-Ig; both Proteintech Group, Inc.), anti-p53 upregulated
modulator of apoptosis (1:1,000; PUMA; cat. no. A3752), anti-growth
arrest and DNA damage-inducible α (1:500; GADD45A; cat. no. A13487;
both ABclonal Biotech Co., Ltd.), anti-NLRP3 (1:1,000; cat. no.
ab283819, Abcam), anti-gasdermin D (1:1,000; GSDMD; cat. no.
20770-1-AP, Proteintech Group, Inc.), anti-caspase1 (1:1,000; cat.
no. BD-PC0002; Suzhou Botron Immunotherapy Co., Ltd.), anti-IL-18
(1:1,000; cat. no. 10663-1-AP, Proteintech Group, Inc.) and
anti-β-actin (cat. no. AC026, ABclonal Biotech Co., Ltd.).
Co-immunoprecipitation (Co-IP)
Liver tissue was homogenized in ice-cold lysis
buffer (cat. no. P2179S, Beyotime Institute of Biotechnology)
supplemented with protease inhibitors. The lysates were centrifuged
at 12,000 × g for 15 min at 4°C and the supernatants were collected
for protein quantification using the BCA assay. Using the Protein
A+G Magnetic Beads Immunoprecipitation kit (cat. no. P2179S,
Beyotime Institute of Biotechnology), 200 μl lysate was
centrifuged at 10,000-14,000 × g and 4°C for 5 min. The supernatant
was collected, and Protein A+G magnetic beads were magnetically
separated with the supernatant removed. Subsequently, 500 μl
antibody working solution (prepared at a final antibody
concentration of 30 μg/ml) was added to resuspend the beads,
followed by incubation on a rotary mixer at room temperature for 1
h. Protein A+G magnetic beads coupled with either the target
antibody or normal IgG were added to the protein samples at a ratio
of 20 μl bead suspension per 500 μl protein sample,
and the mixture was incubated overnight at 4°C on a rocking
platform or rotary mixer. The mixture was placed on a magnetic
stand for 10 sec to allow bead separation, and the supernatant was
discarded. After incubation, the complexes were placed on a
magnetic stand for 10 sec to allow bead separation. The eluted
proteins were separated by 10% SDS-PAGE and transferred onto PVDF
membranes for western blot analysis, as aforementioned Then the
membranes were probed with anti-NLRP3 (1:1,000; cat. no. ab283819,
Abcam), or anti-KLF2 (1:1,000; cat. no. 23384-1-AP, Proteintech
Group, Inc.) at 4°C overnight to detect interacting proteins. IgG
was used as a negative control to confirm the specificity of the
immunoprecipitation.
RNA sequencing (RNA-seq)
RNA-seq was performed to elucidate the global
transcriptomic alterations in response to simulated IRI across
varying levels of KLF2 expression. Stable ov-KLF2 HUVECs were
generated via lentiviral transduction, with empty vector-transduced
cells serving as the control (ov-control). Following the CS/Rep
treatment, total RNA was extracted from three biological
replicates/group. RNA was extracted from samples using TRIzol (cat.
no. 15596026CN; Thermo Fisher Scientific Inc.), and its integrity
was assessed using the Agilent 2100 Bioanalyzer. mRNA was enriched
via poly(A) selection, fragmented and reverse-transcribed using
SuperScript™ II Reverse Transcriptase (Invitrogen, cat. 1896649,
USA), according to the manufacturer's instructions. A cDNA library
was constructed using the Hieff NGS EvoMax RNA Library Prep kit
(Shanghai Yeasen Biotechnology Co., Ltd.; cat. no. 12340ES97), with
a final library concentration of 5 nM. High-throughput sequencing
was performed on the Illumina platform using the NovaSeq X Series
25B Reagent kit (300 cycles; Illumina, Inc.; cat. no. 20104706) for
paired-end 150 bp sequencing, with an insert size of 300-400 bp.
The sequencing orientation was 3'-end sequencing. The polyA tail of
mRNA was captured using Oligo(dT) magnetic beads, followed by
sequencing of the fragmented sequences near the 3'end. Following
quality control of the raw sequencing data, differentially
expressed genes (DEGs) were identified using DESeq2 (1.22.2;
github.com/mikelove/dESeq2). Then
sequence quality was verified using FastQC (http://www.bioinformatics.babraham.ac.uk/projects/fastqc/,
0.11.9), including the Q20, Q30 and GC-content of the clean data.
DEGs were defined as absolute log2 fold change ≥1 and an adjusted
P-value <0.05. Gene Ontology (GO; geneontology.org/) analysis was performed
(significance threshold: P<0.05).
Statistical analysis
Statistical analyses were performed using GraphPad
Prism 8.0.1 (Dotmatics). All data are expressed as mean ± SD of ≥3
biological replicates. Normality was assessed using the
Shapiro-Wilk test and homogeneity of variances was evaluated with
Levene's test. For comparisons between two groups, unpaired
Student's t-test (or Mann-Whitney U test for non-normal data) was
used. For comparisons among >2 groups, one- or two-way ANOVA
followed by Tukey's post hoc test was applied. P<0.05 was
considered to indicate a statistically significant difference.
Results
HMP improves DCD donor liver injury
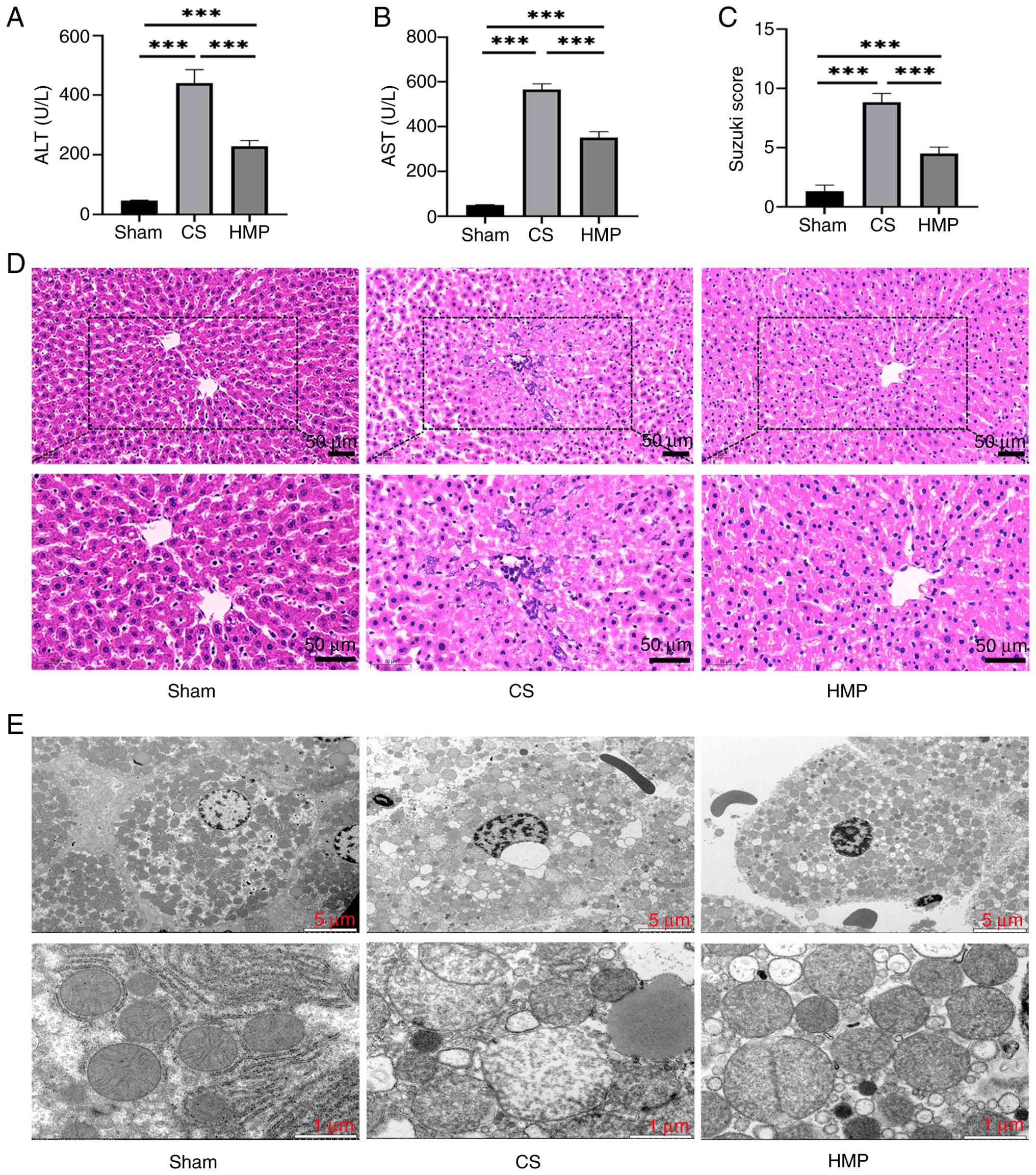
To assess the protective effect of HMP on DCD donor
livers, reperfusion fluid of the CS and HMP groups was analyzed.
Compared with the CS group, the HMP group showed a significant
reduction in AST and ALT levels (Fig. 2A and B). Additionally, staining
showed milder liver sinusoidal congestion, less lobular edema and
reduced hepatocyte vacuolation and necrosis in the HMP group
compared with the CS group (Fig. 2C
and D). TEM also showed that mitochondria displayed visible
cristae and less swelling and vacuolation in the HMP group compared
with CS group (Fig. 2E).
To validate the protective effect of HMP on
inflammation, IF staining was performed to detect neutrophil
infiltration marker MPO. Neutrophil infiltration was significantly
higher in the CS than in the HMP group (Fig. 3A and B). Furthermore, the mRNA
expression levels of inflammatory factors such as IL-6, IL-1β and
tumor necrosis factor (TNF)-α were significantly elevated in the CS
group compared with the HMP group (Fig. 3C-E).
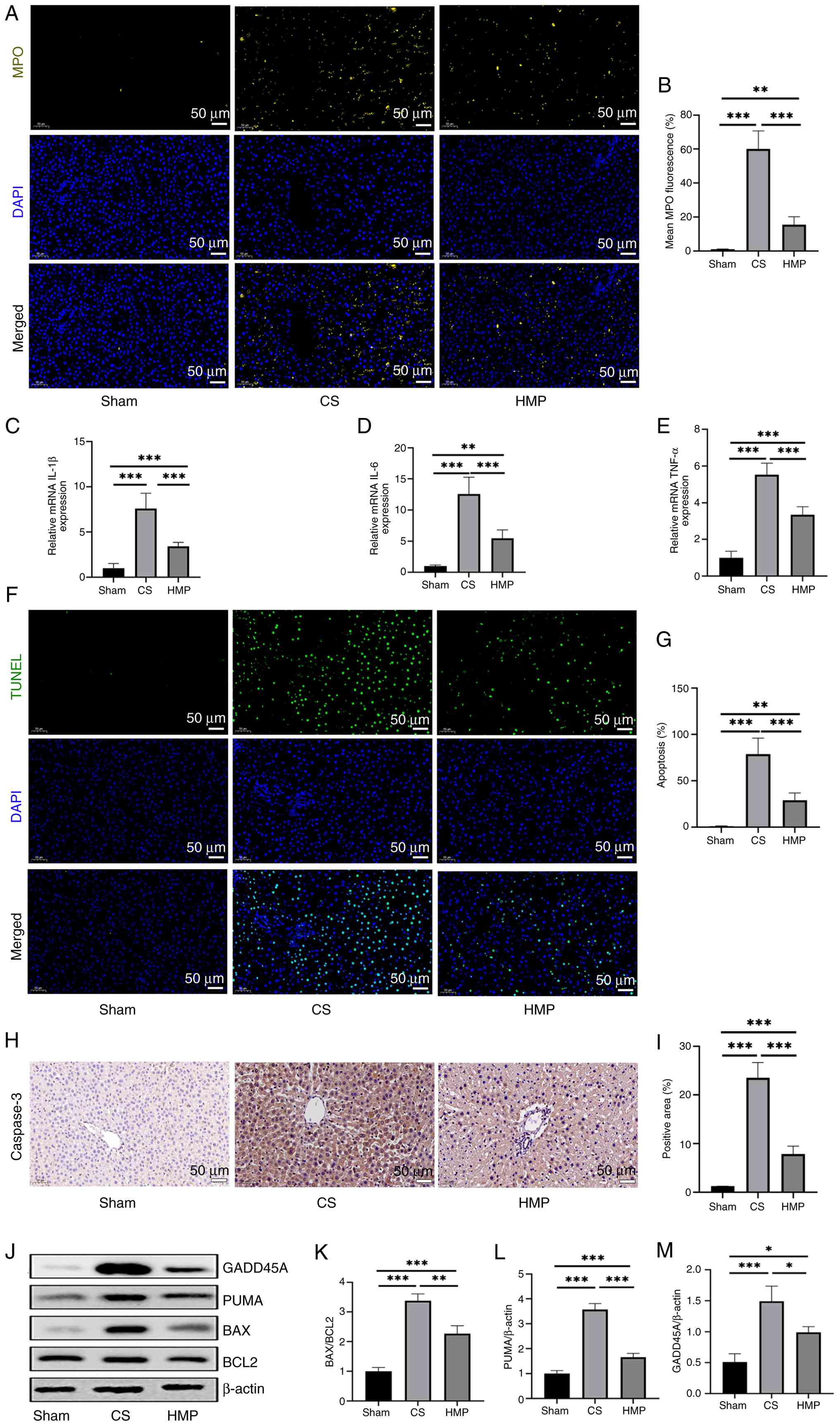 | Figure 3HMP inhibits hepatocyte apoptosis in
donation after circulatory death donor livers. (A) IF staining of
MPO was performed on paraffin-embedded rat liver tissues. (B) IF of
MPO was quantified. Expression levels of mRNA for (C) IL-1β, (D)
IL-6 and (E) TNF-α in liver tissue were quantified using reverse
transcription-quantitative PCR. (F) Hepatocyte apoptosis was
assessed using TUNEL assay. (G) Number of TUNEL-positive cells was
quantified. (H) Immunohistochemical staining of caspase-3 was
performed on paraffin-embedded rat liver tissues. (I)
Caspase-3-positive cells were quantified (n=6). (J) Expression
levels of (K) BAX/BCL2, (L) PUMA and (M) GADD45A in rat liver
tissue were assessed using western blotting (n=3).
*P<0.05, **P<0.01,
***P<0.001. HMP, hypothermic machine perfusion; IF,
immunofluorescence; MPO, myeloperoxidase; PUMA, P53 upregulated
modulator of apoptosis; GADD45A, growth arrest and DNA
damage-inducible alpha; CS, cold storage. |
To assess apoptosis rates in liver tissue, d TUNEL
staining was performed and IHC was performed to detect caspase-3
expression. The number of TUNEL- and caspase-3-positive cells in
the HMP group was significantly lower compared with the CS group
(Fig. 3F-I). Furthermore, the
expression of anti-apoptotic proteins (BAX/BCL2) and pro-apoptotic
proteins (PUMA, GADD45A) in the HMP group, indicating a lower
apoptotic level compared with the CS group (Fig. 3J-M). These findings suggest that
HMP mitigated IRI in DCD donor livers.
HMP protects ECs via shear stress-induced
KLF2 upregulation
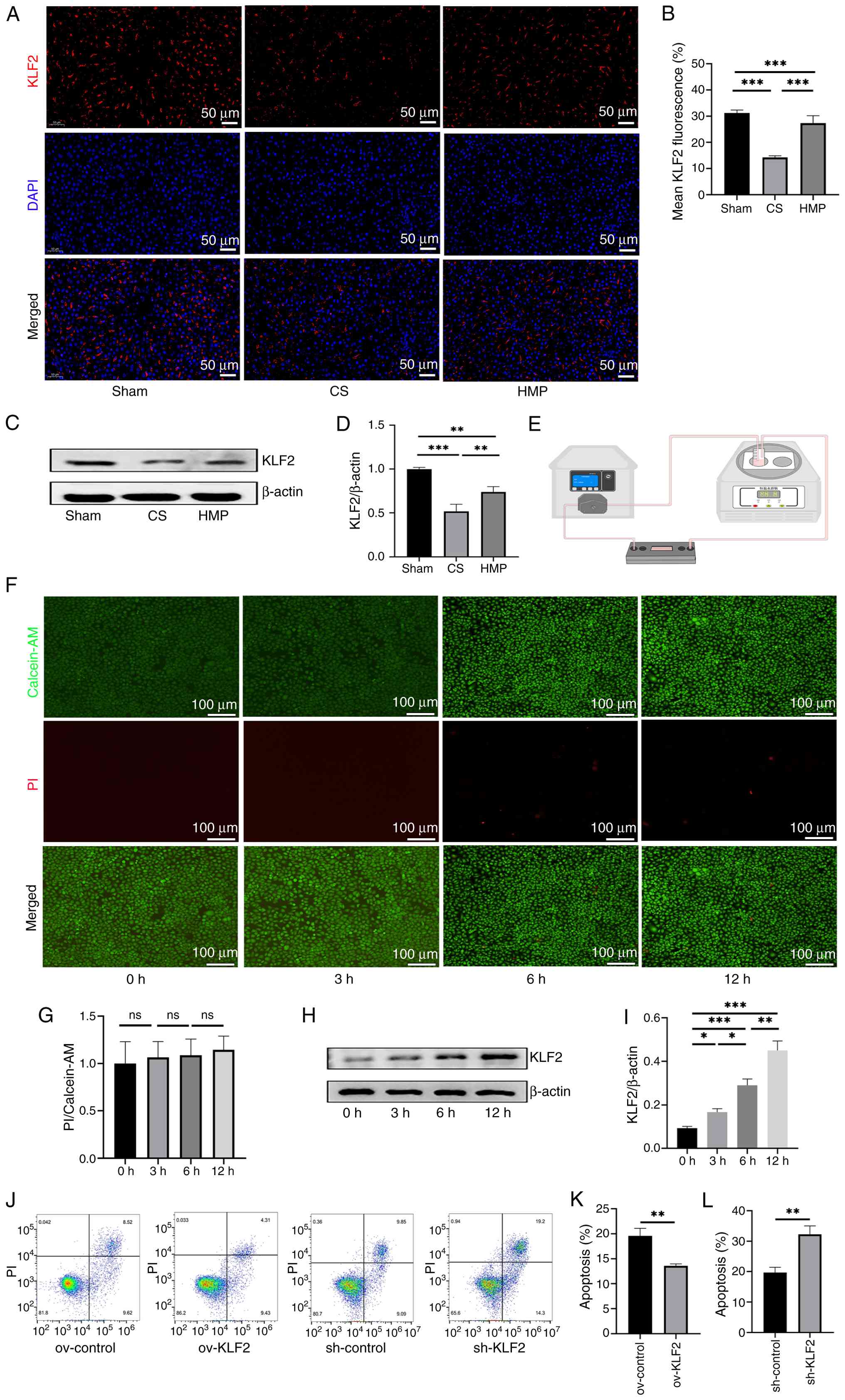
To investigate whether the protective effect of HMP
on DCD liver grafts was due to sustained shear stress leading to
KLF2 upregulation in ECs, IF staining was performed. Liver tissue
was labeled with KLF2 antibodies. The results showed that the
colocalization of KLF2 fluorescence in liver sinusoidal ECs (LSECs)
was significantly higher in the HMP than in the CS group (Fig. 4A and B). Western blot analysis
confirmed that KLF2 protein expression was significantly higher in
the HMP group compared with the CS group (Fig. 4C and D).
A parallel plate flow chamber system was used to
simulate in vivo shear stress conditions in vitro
(Fig. 4E) to validate that KLF2
expression is specifically induced by ECs under laminar flow
conditions. Calcein-AM/PI staining was performed at 0, 3, 6 and 12
h, showing no significant differences in EC damage caused by shear
stress at these time points (Fig. 4F
and G). Western blot analysis of cell samples revealed that
KLF2 expression increased with duration of laminar flow (Fig. 4H and I). These findings suggested
that HMP may protect DCD liver grafts through upregulation of KLF2.
Additionally, the protective phenotype of EC KLF2 upregulation
under CS/Rep conditions was verified. In vitro experiments
using an EC CS/Rep model revealed that overexpression of KLF2
decreased apoptosis, whereas knockdown of KLF2 increased apoptosis
(Fig. 4J-L). These results
suggested that the protective effect of HMP on DCD livers was
closely associated with the upregulation of KLF2.
KLF2 upregulation in ECs promotes
macrophage efferocytosis
To explore how the upregulation of KLF2 in ECs,
induced by HMP, promotes macrophage efferocytosis, TUNEL-positive
apoptotic cells and CD68-positive macrophages were quantified in
DCD liver grafts. Fluorescence colocalization was performed to
assess macrophage efferocytosis in vivo. Compared with the
CS group, the HMP group exhibited increased efferocytosis in
hepatocytes. (Fig. 5A and
B).
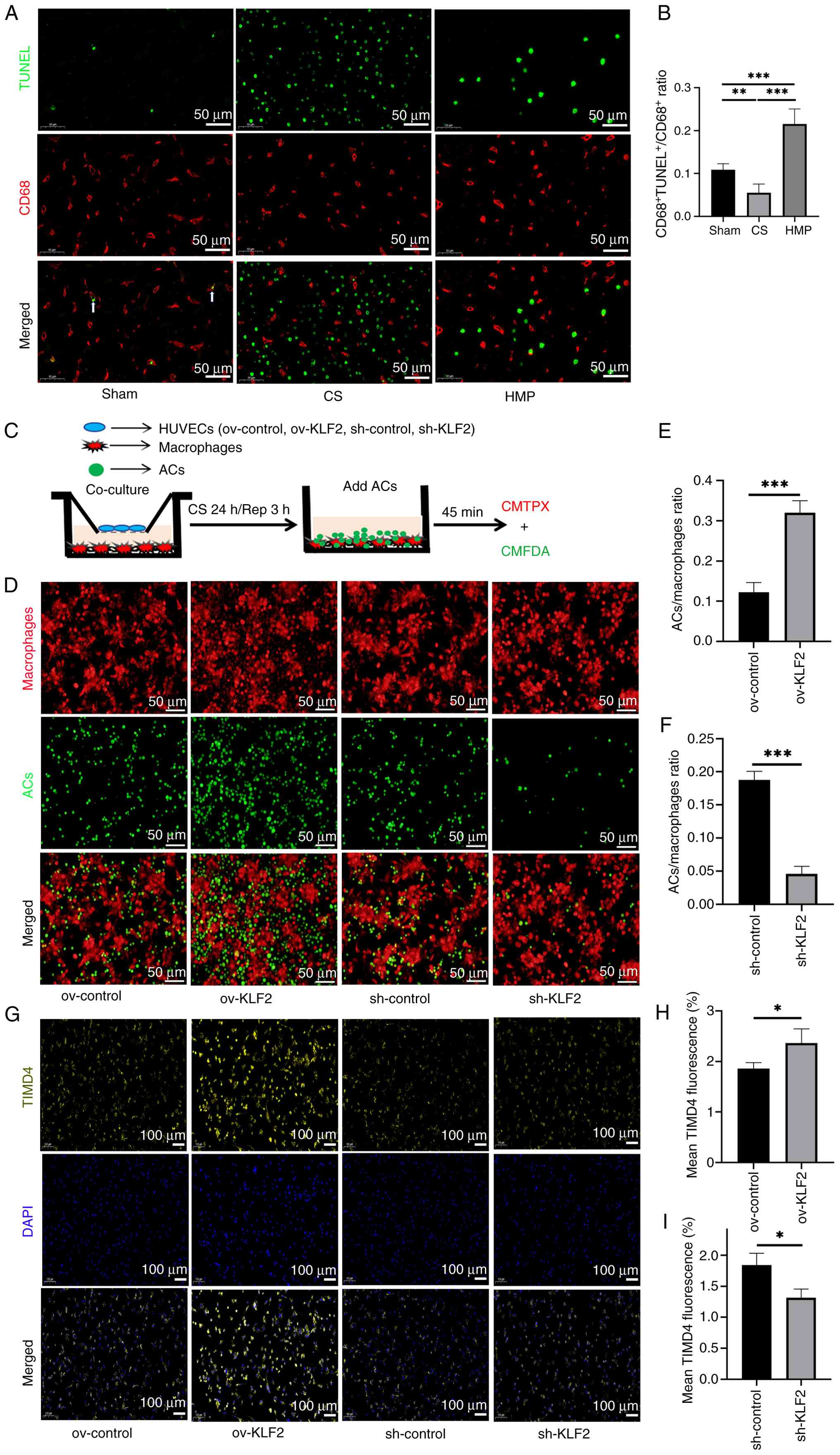 | Figure 5Upregulation of the endothelial cell
protective molecule KLF2 promotes macrophage efferocytosis. (A)
In vivo assessment and (B) quantification of efferocytosis
(proportion of CD68-positive cells engulfing TUNEL-positive
apoptotic cells relative to the total CD68-positive cell
population). Arrows represent macrophage efferocytosis events. (C)
Schematic diagram of the co-culture protocol for HUVECs with
varying KLF2 expression levels and macrophages. (D) Macrophages
(CMTPX+) were co-cultured with apoptotic Jurkat cells
(CMFDA+) for 45 min, and efferocytosis was
quantitatively assessed in vitro by fluorescence
co-localization analysis. (E) Statistical analysis of fluorescence
signals from HUVECS of ov-control and ov-KLF2 groups was performed
using Image-Pro Plus 6.0 software (n=3). (F) Statistical analysis
of fluorescence signals from HUVECS of sh-control and sh-KLF2
groups was performed using Image-Pro Plus 6.0 software (n=3). (G)
Representative staining of TIMD4. (H) Immunofluorescence of TIMD4
from HUVECS of ov-control and ov-KLF2 groups was quantified using
Image-pro Plus 6.0. (I) Immunofluorescence of TIMD4 from HUVECS of
ov-control and ov-KLF2 groups was quantified using Image-pro Plus
6.0 (n=3). *P<0.05, **P<0.01,
***P<0.001. HMP, hypothermic machine perfusion; KLF2,
Kruppel-like Factor 2; AC, apoptotic Jurkat cell; HUVEC, human
umbilical vein endothelial cells; CMTPX, CellTracker™ Red CMTPX
dye; CMFDA, 5-Chloromethylfluorescein Diacetate; CS, cold storage;
Rep, reperfusion; TIMD4, T-cell immunoglobulin and mucin
domain-containing protein 4; ov, overexpression; sh, short
hairpin. |
To confirm that KLF2 upregulation in ECs promoted
macrophage efferocytosis, PMA-induced THP-1-derived macrophages
were co-cultured with HUVECs expressing different levels of KLF2
(Fig. 5C). Under the Transwell
non-contact co-culture condition, both cell types were placed in a
cold IR model to simulate their interaction during cold IR. ACs
were co-incubated with macrophages. The present study assessed
macrophage efferocytosis in vitro by quantifying the
co-positive expression of CMTPX (red tracer for macrophages) and
CMFDA (green tracer for ACs). Macrophages co-incubated with
ov-KLF2-expressing HUVECs showed significantly higher efferocytosis
compared with the ov-control group (Fig. 5D-F). However, compared with the
sh-control group, the sh-KLF2 group decreased efferocytosis. TIMD4,
a marker of macrophage efferocytosis ability, expression was
detected. Comparison of TIMD4 fluorescence intensity among these
groups confirmed these findings (Fig. 5G-I). Specifically, macrophages in
the ov-KLF2 group showed higher TIMD4 intensity than those in the
ov-control group; compared with the sh-control group, the TIMD4
fluorescence intensity of macrophages in the sh-KLF2 group was
significantly lower. These results indicated that the positive
effect of HMP on DCD livers was mediated by upregulation of KLF2
expression to promote efferocytosis.
Increased macrophage efferocytosis
decreases hepatocellular injury in DCD liver grafts
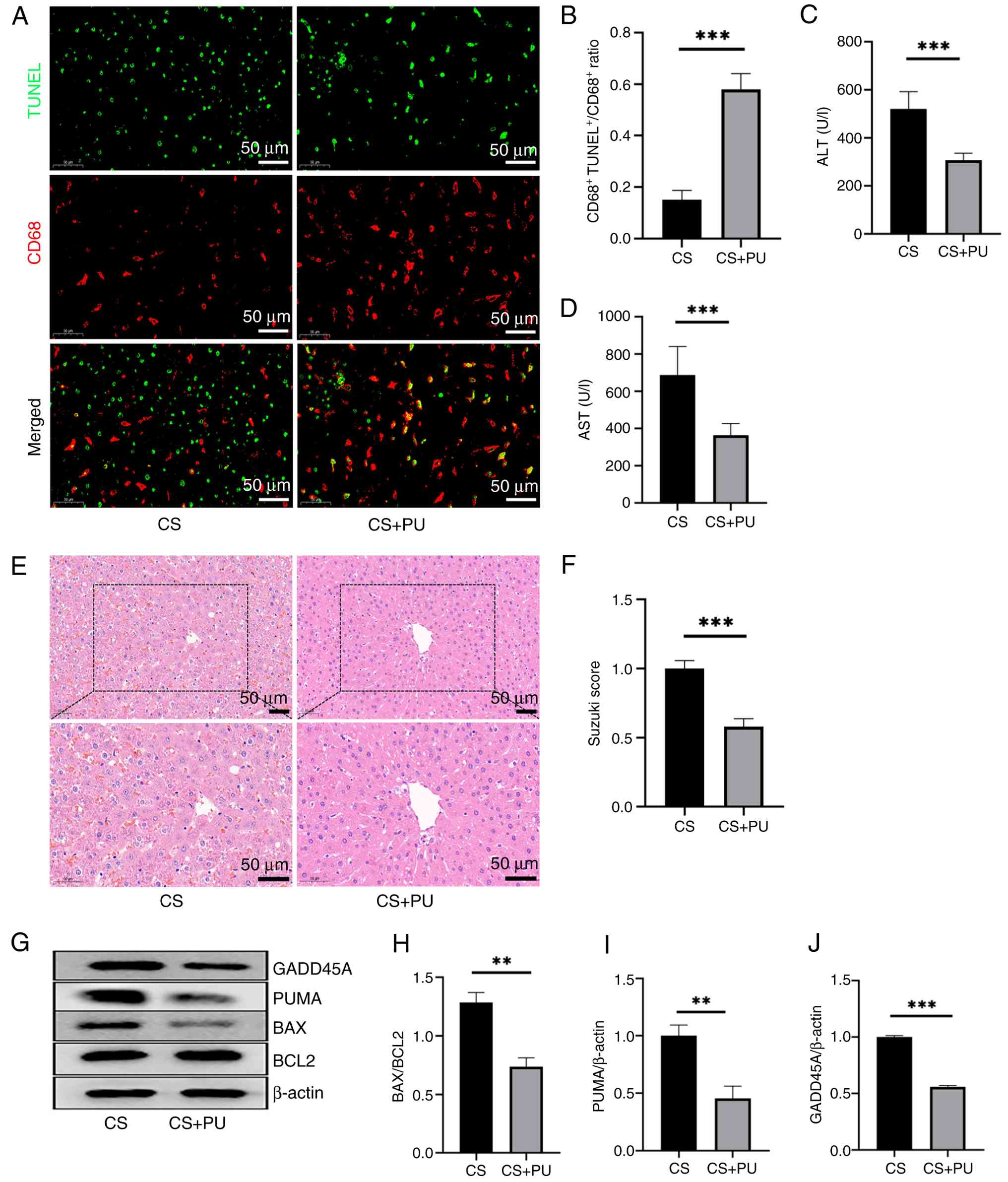
PU is an agonist that promotes macrophage
efferocytosis (32,39). PU was administered to
experimental animals and livers from DCD donors were preserved by
CS. These livers were compared with those from conventional DCD
donors that were not treated with PU to investigate whether
increased efferocytosis events enhanced the protection of DCD
livers. In the CS + PU group, efferocytosis events occurred more
frequently than in the CS group (Fig. 6A and B). Assessment of liver
function indicators and histopathological analysis demonstrated a
significant attenuation of hepatocellular injury in the CS + PU
group, as evidenced by decreased serum transaminase levels and
reduced histopathological scores (Fig. 6C-F). Western blot analysis of
BCL2, BAX, PUMA, and GADD45A indicated that enhanced efferocytosis
reduced hepatocellular apoptosis in DCD livers. This was evidenced
by a downregulation of pro-apoptotic proteins (BAX, PUMA, and
GADD45A) and a concomitant increase in the anti-apoptotic BCL2/BAX
ratio, collectively shifting the protein expression profile toward
an anti-apoptotic phenotype. (Fig.
6G-J). These findings suggested that promoting efferocytosis
events improved the IRI of DCD livers. Moreover, the specific MerTK
inhibitor UNC2025 significantly attenuated the effects of HMP on
graft preservation and inflammatory response (Fig. S1A-D). In summary, the concurrent
reduction in serum transaminase levels, improvement in
histopathological scores, and decrease in apoptosis alongside
enhanced efferocytosis in DCD liver grafts collectively demonstrate
that the therapeutic efficacy of HMP depends on intact efferocytic
function.
KLF2 inhibits the NLRP3-mediated
pyroptosis pathway during CS/Rep in ECs
To investigate how KLF2 affects macrophage
phagocytosis under CS/Rep conditions, RNA-seq was performed on the
ov-control and the ov-KLF2 HUVECs after CS/Rep treatment. RNA-seq
analysis revealed that 1,061 genes were up- and 1,584 genes were
downregulated (Fig. 7A). GO
enrichment analysis showed that these DEGs were primarily enriched
in biological processes such as 'angiogenesis', 'positive
regulation of apoptotic process', 'cell-cell signaling' and
'inflammatory response' (Fig.
S2), suggesting that KLF2 overexpression significantly
modulates inflammation- and immune-associated pathways during cold
IRI in HUVECs.
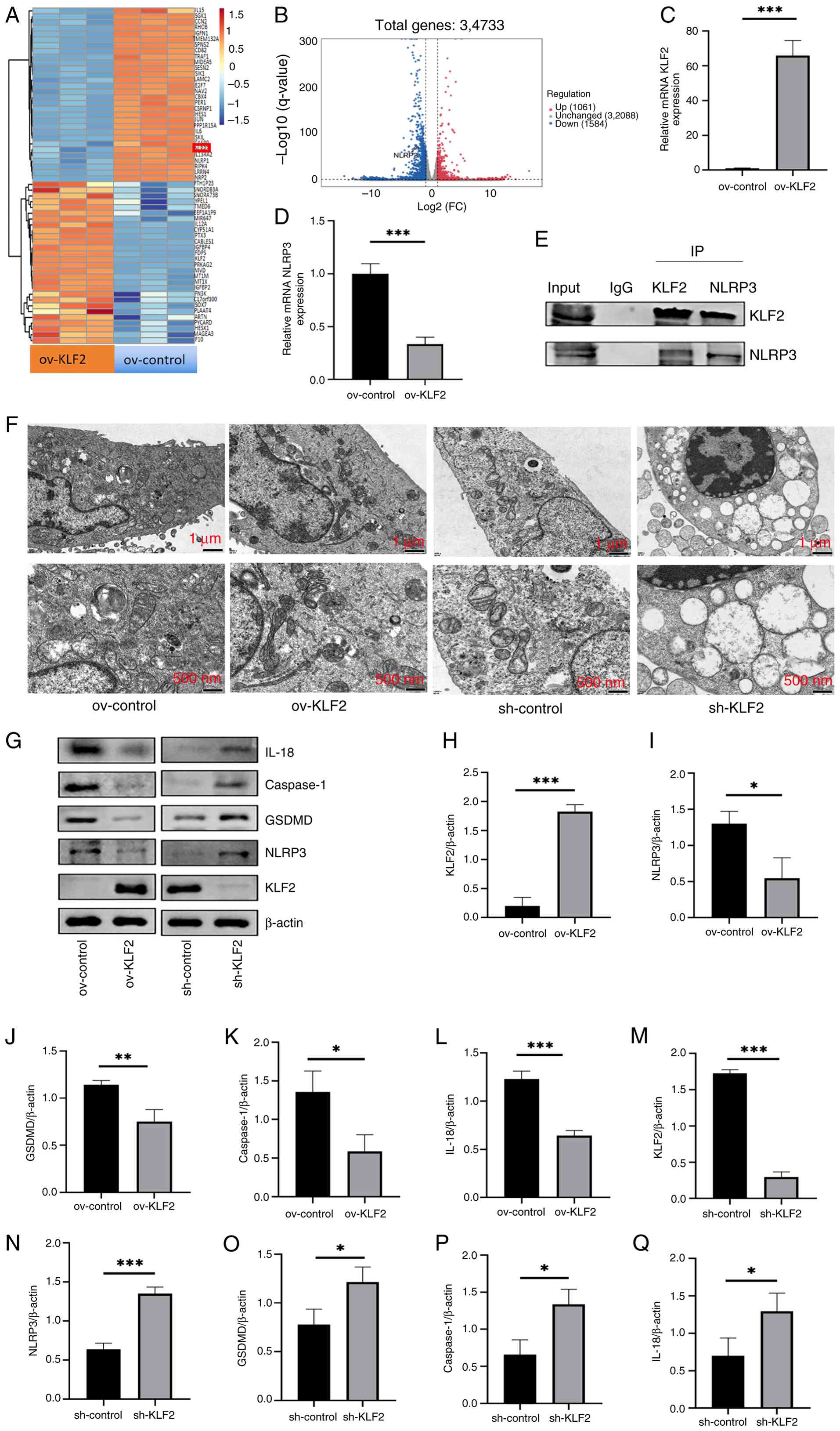 | Figure 7KLF2 inhibits the NLRP3-mediated
pyroptosis pathway during CS/Rep in ECs. (A) Heatmap and (B)
volcano plot of differentially expressed genes between control and
ov-KLF2 HUVECs after CS/Rep treatment. mRNA levels of (C) KLF2 and
(D) NLRP3 in ov-control and ov-KLF2 group of HUVECs following
CS/Rep treatment were quantified using reverse
transcription-quantitative PCR. (E) Co-IP analysis of the
interaction between KLF2 and NLRP3. (F) Microstructure of HUVECS.
(G) Representative Western blot images for KLF2, NLRP3, GSDMD,
Caspase-1 and IL-18 in CS/Rep HUVECs. Quantitative analysis of KLF2
(H), NLRP3 (I), GSDMD (J), Caspase-1 (K) and IL-18 (L) protein
expression based on western blot results from HUVECS of ov-control
and ov-KLF2 groups. Quantitative analysis of KLF2 (M), NLRP3 (N),
GSDMD (O), Caspase-1 (P) and IL-18 (Q) protein expression based on
western blot results from HUVECS of sh-control and sh-KLF2 groups.
(n=3). *P<0.05, **P<0.01,
***P<0.001. KLF2, Kruppel-like Factor 2; CS/Rep, cold
storage/reperfusion; HUVEC, human umbilical vein endothelial cells;
ov, overexpression; IP, immunoprecipitation; GSDMD, gasdermin D;
sh, short hairpin;; FC, fold-change. |
It is known that the inflammatory microenvironment
of macrophages influences their phenotypical characteristics and
phagocytic function (40). The
present differential gene analysis focused on the expression of
NLRP3 (Fig. 7B) as
NLRP3-mediated pyroptosis amplifies the inflammatory response
(41). In addition to NLRP3,
RNA-seq revealed significant changes in the expression of other
genes associated with inflammation. Specifically, levels of
pro-inflammatory factors such as IL-6 and JUN were significantly
downregulated.. NLRP3 expression levels were significantly lower in
the ov-KLF2 compared with the ov-control group (Fig. 7C and D). To investigate the
potential interaction between KLF2 and NLRP3, Co-IP was performed.
There was a specific physical interaction between KLF2 and NLRP3,
as evidenced by the presence of NLRP3 in the KLF2 immunoprecipitate
and KLF2 in the NLRP3 immunoprecipitate (Fig. 7E). TEM was performed to observe
signs of pyroptosis following CS/Rep treatment. Compared with the
ov-control group, the ov-KLF2 group maintained relatively intact
cell membranes. By contrast with the sh-control group, the sh-KLF2
group exhibited notable mitochondrial swelling with loss of most
cristae, characteristic of pyroptosis (Fig. 7F). Western blot analysis of
pyroptosis-associated molecules such as GSDMD, Caspase-1 and IL-18
(42-46) was performed. Overexpression of
KLF2 significantly decreased the expression of NLRP3 and
pyroptosis-related proteins, while the sh-KLF2 group showed the
opposite trend (Fig. 7G-Q).
Compared with the sh-KLF2 group, the sh-KLF2 + MCC950 group showed
a significant inhibition in the expression of key
pyroptosis-related proteins (Fig.
S3A-E). These results collectively indicated that KLF2
upregulation suppressed the NLRP3-mediated pyroptosis pathway in
ECs during CS/Rep. NLRP3 inhibitor under KLF2-deficient conditions
effectively rescued the exacerbated pyroptotic phenotype.
EC pyroptosis-induced inflammation
inhibits macrophage efferocytosis
To explore the impact of EC pyroptosis on macrophage
efferocytosis, pyroptosis of ECs was altered using the pyroptosis
agonist nigericin (Fig. 8A). As
the dose of nigericin increased, HUVECs exhibited distinct
morphological changes. The cells shrank or swelled, the outlines
became blurred, the cytoplasm appeared sparse or expanded and
structure became irregular (Fig.
8B). To assess how EC pyroptosis affect macrophages
efferocytosis, ECs were co-cultured with macrophages for 24 h in a
non-contact setting (Fig. 8C).
Macrophages were co-incubated with ACs for 45 min. The
efferocytosis capacity of macrophages was quantified based on the
co-localization of the tracers of the two cell types. When the
degree of EC pyroptosis increased, macrophages co-cultured with
these cells demonstrated a decreased ability to phagocytose
apoptotic cells (Fig. 8D and E).
Therefore, increased pyroptosis of ECs impaired macrophage
efferocytosis.
Discussion
The present study investigated the protective
crosstalk between ECs and macrophage efferocytosis during HMP for
DCD livers. The present study confirmed the key role of endothelial
KLF2 in DCD livers. Additionally, conventional static CS
significantly downregulated KLF2 expression in LSECs, which was
associated with impaired macrophage efferocytosis. Mechanistically,
KLF2 negatively regulated the pyroptosis signaling pathway by
directly interacting with NLRP3 in ECs, thereby attenuating the
inflammatory microenvironment. This decrease facilitated macrophage
efferocytosis. These findings demonstrate the regulatory role of
KLF2 in the NLRP3 signaling cascade in ECs, underscoring the
potential therapeutic value of targeting KLF2 in HMP to improve
macrophage efferocytosis and counteract the impaired immune
response during DCD liver storage.
Optimizing organ preservation methods serves a key
role in minimizing IRI, especially in DCD livers. ECs, which line
blood vessels, are vital integrators and sensors of physiological
stimuli (47), actively
participating in numerous processes by responding to changes in
hemodynamic forces. Under physiological conditions, endothelial
cells respond to changes in hemodynamic forces to maintain
circulatory homeostasis. Under pathological conditions, however,
this responsive mechanism becomes dysregulated, thereby promoting
the development of vascular diseases such as hypertension,
thrombosis, aneurysm, and atherosclerosis (48). In conventional static CS, the
absence of biomechanical stimulation on the blood vessel walls
results in damage to ECs (10,49-51), negatively impacting graft
recovery. HMP is an advanced technology applied in organ
preservation and transplantation. By mimicking physiological
conditions in a hypothermic environment to match organ metabolic
demands, HMP enhances physiological adaptability and increases the
success rate of transplantation (52,53). HMP effectively alleviates EC
damage caused by the lack of biomechanical stimulation during CS by
providing flow stimulation, thereby improving EC function (13,54). A key mechanism underlying this
positive effect is the upregulation of KLF2, a critical molecule
that induces endothelial shear stress. KLF2 regulates endothelial
gene expression, which is key for processes such as angiogenesis
and the maintenance of vascular endothelial health under
flow-induced shear stress (55).
KLF2 influences the inflammatory response (56), angiogenesis (57) and antioxidant (58) defense in blood vessels by
regulating the gene expression of ECs. Here, conventional static CS
significantly downregulated KLF2 expression in LSECs, whereas HMP
markedly upregulated its expression. Parallel plate flow chamber
experiments in vitro confirmed that KLF2 served as a key
effector molecule produced by vascular ECs in response to LSS
stimulation. Moreover, HUVECs overexpressing KLF2 exhibited
significantly decreased apoptosis following CS/Rep injury, which is
consistent with previous studies (12,59). KLF2 is a key factor in
maintaining the functional properties and homeostasis of ECs in a
shear stress environment, highlighting its vital role in optimizing
organ preservation and improving transplant outcomes. However, the
specific role of HMP by elevating KLF2 expression in vascular ECs
to improve DCD liver injury remains unclear.
In the present EC CS/Rep model, KLF2 exerted a
potent anti-inflammatory effect by inhibiting the activation of the
NLRP3 inflammasome. The NLRP3 protein belongs to the
nucleotide-binding oligomerization domain-like receptor family
(60). The assembly of the NLRP3
inflammasome is the canonical upstream event for caspase-1
activation. Detection of caspase-1 activation provides direct
evidence of functional inflammasome activity and represents a key
hallmark distinguishing the canonical pyroptosis pathway. GSDMD,
following cleavage by caspase-1, forms pores in the plasma
membrane, representing the definitive downstream step in pyroptosis
execution. Mature IL-18 serves as a key substrate of caspase-1. Its
secretion not only validates caspase-1 activity but also directly
explains the inflammatory response driven by pyroptosis. As a
pattern recognition receptor, its key function is to assemble the
NLRP3 inflammasome. NLRP3 has the broadest functional range of all
inflammasomes in both the innate and adaptive immune systems
(61,62), and its dysregulation is the root
cause of numerous inflammatory diseases (63,64). For example, the pathogenesis of
various immune- and inflammation-related diseases, such as
including arthritis, Alzheimer's disease, inflammatory bowel
disease, and other autoimmune or autoinflammatory disorders-is
closely linked to the function of the NLRP3 inflammasome (65). Downregulating the NLRP3
inflammasome can exert a protective effect against liver IRI
(33,66,67). ECs at inflammatory sites act as
both active participants and regulators of the inflammatory process
(68). Studies (68,69) have confirmed that activation of
the NLRP3 inflammasome in ECs exacerbates endothelial dysfunction.
This activation triggers the NLRP3 inflammasome, leading to the
secretion of mature forms of IL-1β and IL-18, thereby intensifying
the inflammatory response in ECs (70). This aligns with the present
findings: Compared with the sh-control group, ECs with sh-KLF2,
exhibited increased NLRP3 expression following CS/Rep, along with
upregulation of NLRP3 inflammasome-associated proteins.
Correspondingly, apoptosis levels in ECs were significantly
elevated. sh-KLF2-transfected cells were treated with the specific
NLRP3 inhibitor MCC950, followed by CS/Rep. The expression levels
of pyroptosis-associated proteins were then assessed by western
blotting. Compared with the sh-KLF2 group, MCC950 attenuated the
increased expression of pyroptosis-related proteins induced by
sh-KLF2, suggesting NLRP3 inhibition rescued the pyroptosis
phenotype resulting from KLF2 deficiency. Mechanistically, KLF2
inhibits the activation of the NLRP3 inflammasome, decreases
pyroptosis and ameliorates endothelial dysfunction. Additionally,
NLRP3 inflammasome activation releases damage-associated molecular
patterns such as high mobility group box 1 protein (HMGB1) into the
extracellular environment (71).
The inflammatory molecules released by NLRP3 inflammasome-mediated
pyroptosis directly shape a persistently pro-inflammatory
microenvironment. This microenvironment is associated with the
regulation of macrophage efferocytosis; multiple inflammatory
mediators, including HMGB1 (19)
and IL-10 (16), modulate
macrophage efferocytosis.
The present study not only observed enhanced
efferocytosis but also demonstrated, through co-culture
experiments, that endothelial KLF2 status regulated macrophage
function. Efferocytosis serves as a key hub linking innate immunity
and tissue repair, playing a key role in physiological and
pathological processes by regulating the clearance of apoptotic
cells (72,73). Macrophages efficiently remove
apoptotic cells, preventing secondary necrosis and the release of
inflammatory factors, thereby maintaining tissue homeostasis.
Moreover, this process promotes macrophage polarization toward an
anti-inflammatory and reparative (M2) phenotype, characterized by
the release of anti-inflammatory cytokines such as IL-10 and TGF-β,
which suppress excessive inflammatory responses (74). In the liver, efferocytosis serves
a central role in maintaining homeostasis and suppressing
inflammatory responses. In non-alcoholic steatohepatitis, impaired
efferocytosis leads to the accumulation of apoptotic hepatocytes,
exacerbating inflammation and fibrosis (75). By contrast, during liver IRI,
efferocytosis exhibits notable anti-inflammatory and protective
effects (16,76). This mechanism explains why the
HMP group demonstrated milder inflammatory responses and enhanced
tissue repair compared with CS group.
Nevertheless, the present study has several
important limitations. First, the precise molecular mechanisms and
signaling pathways by which pyroptosis in LSECs influences
macrophage efferocytosis via paracrine signals remain incompletely
elucidated. Future investigations should employ a multi-dimensional
approach to explore this cascade: At the molecular level,
identifying key mediators released by ECs following pyroptosis is
key; at the cellular level, establishing more sophisticated
co-culture systems is needed to identify intercellular
communication and at the tissue level, techniques such as spatial
transcriptomics could be leveraged to visualize the signaling
networks in situ.
Due to technical and time constraints, the present
study did not conduct in vivo endothelial-specific KLF2
loss-of-function experiments to validate the role of KLF2 in liver
protection. However, such investigations should be performed in
future research. The present study demonstrated through rescue
experiments that NLRP3 served as a key downstream mediator of
KLF2-driven cytoprotection and efferocytosis. However, a
complementary gain-of-function test (activating NLRP3 in a
KLF2-overexpression setting) was not performed, which undermines
the robustness of the conclusion. Future work should address this
to delineate the KLF2-NLRP3 inhibitory axis. Furthermore,
functional validation of TIMD4 in efferocytosis is limited by the
lack of specific, well-characterized inhibitors or blocking
antibodies, precluding direct loss-of-function evidence for
TIMD4-dependent efferocytosis as a key phenotypical mediator. To
address the broader role of efferocytosis in the observed
phenotype, the present study performed loss-of-function experiments
using a specific inhibitor of MerTK, another key efferocytosis
receptor. PU, a central molecule in polyamine metabolism,
participates broadly in physiological processes including cell
proliferation and inflammation regulation (77). The pro-efferocytosis effect may
reflect one facet of its pleiotropic functions rather than a
specific agonist activity.
Second, the rat DCD model used in the present study
presents limitations for clinical translation. The model lacks
long-term functional assessment metrics post-transplantation and
the short observation window precludes insights into long-term
outcomes. Furthermore, the standardized experimental setting fails
to recapitulate the donor-recipient heterogeneity inherent in
clinical practice, such as variations in age, underlying
comorbidities and immune status, which limit the direct
extrapolation of the present findings to the clinical realm.
Notwithstanding these limitations, the proposed
KLF2-NLRP3-efferocytosis axis has a well-defined trajectory for
clinical translation. In the context of organ preservation,
incorporating KLF2 agonists or NLRP3 inhibitors into machine
perfusion solutions represents a promising ex vivo strategy
to improve the quality of marginal donor livers. For transplant
assessment, key molecular components of this axis, including KLF2,
NLRP3 and efferocytosis markers, exhibit potential as novel
biomarkers to predict post-transplant rejection and functional
recovery. Development of integrated therapeutic strategies based on
this mechanism (donor preconditioning, intraoperative organ
protection and postoperative immunomodulation in recipients) may
establish a novel treatment paradigm for DCD liver
transplantation.
The present study highlighted the role of HMP in
regulating the KLF2-derived transcriptional program in LSECs,
contributing to vascular protection and promoting macrophage
efferocytosis. Overexpression of KLF2 in ECs activated a
KLF2/NLRP3-mediated pyroptosis paracrine mechanism that influenced
macrophage efferocytosis, shaping the inflammatory
microenvironment. The present study deepens the understanding of
how HMP protects DCD donor livers, with implications for developing
therapeutic strategies aimed at improving macrophage function and
mitigating liver IRI. Targeting ECs to regulate macrophage
efferocytosis may offer a promising approach for enhancing liver
preservation and recovery.
Supplementary Data
Availability of data and materials
The data generated in the present study may be found
in the Gene Expression Omnibus database under accession number
GSE296118 or at the following URL: ncbi.nlm.nih.gov/gds/?term=GSE296118.
Authors' contributions
QD performed experiments, analyzed data and wrote
and revised the manuscript. ZLi and QY designed the methodology and
analyzed data. JL, XZ and ZF analyzed data. JL, ZLu and PY analyzed
data and supervised the study. JX and QX designed and conceived the
study and revised the manuscript. All authors have read and
approved the final manuscript. QD and ZLi confirm the authenticity
of all the raw data.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee for Laboratory Animal Welfare of the First Affiliated
Hospital of Nanchang University (approval no.
CDYFY-IACUC-202407QR199, Nanchang, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
The present study was supported by National Natural Science
Foundation of China (grant nos. 82460131, 82060122, 82200707 and
82300728) and Natural Science Foundation of JiangXi province (grant
no. 20224ACB206027).
References
|
1
|
Sugawara Y and Hibi T: Recent trends and
new developments in liver transplantation. Biosci Trends.
18:206–211. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Saidi RF and Hejazii Kenari SK: Challenges
of organ shortage for transplantation: Solutions and opportunities.
Int J Organ Transplant Med. 5:87–96. 2014.PubMed/NCBI
|
|
3
|
Hahn JW, Woo S, Park J, Lee H, Kim HJ, Ko
JS, Moon JS, Rahmati M, Smith L, Kang J, et al: Global, regional,
and national trends in liver Disease-related mortality across 112
countries from 1990 to 2021, with projections to 2050:
Comprehensive analysis of the WHO mortality database. J Korean Med
Sci. 39:e2922024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Monbaliu D, Pirenne J and Talbot D: Liver
transplantation using donation after cardiac death donors. J
Hepatol. 56:474–485. 2012. View Article : Google Scholar
|
|
5
|
Yue P, Lv X, You J, Zou Y, Luo J, Lu Z,
Cao H, Liu Z, Fan X and Ye Q: Hypothermic oxygenated perfusion
attenuates DCD liver ischemia-reperfusion injury by activating the
JAK2/STAT3/HAX1 pathway to regulate endoplasmic reticulum stress.
Cell Mol Biol Lett. 28:552023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Czigany Z, Craigie EC, Lurje G, Song S,
Yonezawa K, Yamamoto Y, Minor T and Tolba RH: Adenosine A2a
receptor stimulation attenuates Ischemia-reperfusion injury and
improves survival in A porcine model of DCD liver transplantation.
Int J Mol Sci. 21:67472020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sousa Da Silva RX, Weber A, Dutkowski P
and Clavien PA: Machine perfusion in liver transplantation.
Hepatology. 76:1531–1549. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Feng GY, Feng X, Tao J, Ao YP, Wu XH, Qi
SG, He ZB and Shi ZR: Benefits of hypothermic oxygenated perfusion
versus static cold storage in liver transplant: A comprehensive
systematic review and Meta-analysis. J Clin Exp Hepatol.
14:1013372024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Parmar KM, Larman HB, Dai G, Zhang Y, Wang
ET, Moorthy SN, Kratz JR, Lin Z, Jain MK, Gimbrone MA Jr and
García-Cardeña G: Integration of flow-dependent endothelial
phenotypes by Kruppel-like factor 2. J Clin Invest. 116:49–58.
2006. View Article : Google Scholar :
|
|
10
|
Gracia-Sancho J, Villarreal G Jr, Zhang Y,
Yu JX, Liu Y, Tullius SG and García-Cardeña G: Flow cessation
triggers endothelial dysfunction during organ cold storage
conditions: Strategies for pharmacologic intervention.
Transplantation. 90:142–149. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Russo L, Gracia-Sancho J, García-Calderó
H, Marrone G, García-Pagán JC, García-Cardeña G and Bosch J:
Addition of simvastatin to cold storage solution prevents
endothelial dysfunction in explanted rat livers. Hepatology.
55:921–930. 2012. View Article : Google Scholar
|
|
12
|
Hu X, Wang W, Zeng C, He W, Zhong Z, Liu
Z, Wang Y and Ye Q: Appropriate timing for hypothermic machine
perfusion to preserve livers donated after circulatory death. Mol
Med Rep. 22:2003–2011. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Burlage LC, Karimian N, Westerkamp AC,
Visser N, Matton APM, van Rijn R, Adelmeijer J, Wiersema-Buist J,
Gouw ASH, Lisman T and Porte RJ: Oxygenated hypothermic machine
perfusion after static cold storage improves endothelial function
of extended criteria donor livers. HPB (Oxford). 19:538–546. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu Z, Zhang X, Xiao Q, Ye S, Lai CH, Luo
J, Huang X, Wang W, Zeng C, Zhong Z, et al: Pretreatment donors
after circulatory death with simvastatin alleviates liver ischemia
reperfusion injury through a KLF2-dependent mechanism in rat. Oxid
Med Cell Longev. 2017:38619142017. View Article : Google Scholar
|
|
15
|
Doran AC, Yurdagul A Jr and Tabas I:
Efferocytosis in health and disease. Nat Rev Immunol. 20:254–267.
2020. View Article : Google Scholar
|
|
16
|
Ni M, Zhang J, Sosa R, Zhang H, Wang H,
Jin D, Crowley K, Naini B, Reed FE, Busuttil RW, et al: T-Cell
immunoglobulin and mucin Domain-containing Protein-4 is critical
for kupffer cell homeostatic function in the activation and
resolution of liver ischemia reperfusion injury. Hepatology.
74:2118–2132. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Miao L, Yu C, Guan G, Luan X, Jin X, Pan
M, Yang Y, Yan J, Chen P and Di G: Extracellular vesicles
containing GAS6 protect the liver from ischemia-reperfusion injury
by enhancing macrophage efferocytosis via MerTK-ERK-COX2 signaling.
Cell Death Discov. 10:4012024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mihaila AC, Ciortan L, Tucureanu MM,
Simionescu M and Butoi E: Anti-Inflammatory neutrophils reprogram
macrophages toward a Pro-healing phenotype with increased
efferocytosis capacity. Cells. 13:2082024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pisetsky DS: The role of HMGB1 in
efferocytosis: When the dead go unburied. Focus on 'HMGB1 inhibits
macrophage activity in efferocytosis through binding to the
alphavbeta3-integrin'. Am J Physiol Cell Physiol. 299:C1253–C1255.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang J, Muri J, Fitzgerald G, Gorski T,
Gianni-Barrera R, Masschelein E, D'Hulst G, Gilardoni P, Turiel G,
Fan Z, et al: Endothelial lactate controls muscle regeneration from
ischemia by inducing M2-like macrophage polarization. Cell Metab.
31:1136–1153.e7. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhou B, Magana L, Hong Z, Huang LS,
Chakraborty S, Tsukasaki Y, Huang C, Wang L, Di A, Ganesh B, et al:
The angiocrine Rspondin3 instructs interstitial macrophage
transition via metabolic-epigenetic reprogramming and resolves
inflammatory injury. Nat Immunol. 21:1430–1443. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li C, Fang F, Wang E, Yang H, Yang X, Wang
Q, Si L, Zhang Z and Liu X: Engineering extracellular vesicles
derived from endothelial cells sheared by laminar flow for
anti-atherosclerotic therapy through reprogramming macrophage.
Biomaterials. 314:1228322025. View Article : Google Scholar
|
|
23
|
He S, Wu C, Xiao J, Li D, Sun Z and Li M:
Endothelial extracellular vesicles modulate the macrophage
phenotype: Potential implications in atherosclerosis. Scand J
Immunol. 87:e126482018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ni CW, Qiu H, Rezvan A, Kwon K, Nam D, Son
DJ, Visvader JE and Jo H: Discovery of novel mechanosensitive genes
in vivo using mouse carotid artery endothelium exposed to disturbed
flow. Blood. 116:e66–e73. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nam D, Ni CW, Rezvan A, Suo J, Budzyn K,
Llanos A, Harrison D, Giddens D and Jo H: Partial carotid ligation
is a model of acutely induced disturbed flow, leading to rapid
endothelial dysfunction and atherosclerosis. Am J Physiol Heart
Circ Physiol. 297:H1535–H1543. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lv JJ, Wang H, Zhang C, Zhang TJ, Wei HL,
Liu ZK, Ma YH, Yang Z, He Q, Wang LJ, et al: CD147 sparks
atherosclerosis by driving M1 phenotype and impairing
efferocytosis. Circ Res. 134:165–185. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Adkar SS and Leeper NJ: Efferocytosis in
atherosclerosis. Nat Rev Cardiol. 21:762–779. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kojima Y, Weissman IL and Leeper NJ: The
role of efferocytosis in atherosclerosis. Circulation. 135:476–489.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Parmar KM, Nambudiri V, Dai G, Larman HB,
Gimbrone MA Jr and García-Cardeña G: Statins exert endothelial
atheroprotective effects via the KLF2 transcription factor. J Biol
Chem. 280:26714–26719. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yao X, Liu Y, Mao M, Yang L, Zhan Q and
Xiao J: Calorie restriction mimetic, resveratrol, attenuates
hepatic ischemia and reperfusion injury through enhancing
efferocytosis of macrophages via AMPK/STAT3/S1PR1 pathway. J Nutr
Biochem. 126:1095872024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen F, Xu W, Tang M, Tian Y, Shu Y, He X,
Zhou L, Liu Q, Zhu Q, Lu X, et al: hnRNPA2B1 deacetylation by SIRT6
restrains local transcription and safeguards genome stability. Cell
Death Differ. 32:382–396. 2025. View Article : Google Scholar
|
|
32
|
Yurdagul A Jr, Subramanian M, Wang X,
Crown SB, Ilkayeva OR, Darville L, Kolluru GK, Rymond CC, Gerlach
BD, Zheng Z, et al: Macrophage metabolism of apoptotic Cell-derived
arginine promotes continual efferocytosis and resolution of injury.
Cell Metab. 31:518–533.e10. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
He W, Ye S, Zeng C, Xue S, Hu X, Zhang X,
Gao S, Xiong Y, He X, Vivalda S, et al: Hypothermic oxygenated
perfusion (HOPE) attenuates ischemia/reperfusion injury in the
liver through inhibition of the TXNIP/NLRP3 inflammasome pathway in
a rat model of donation after cardiac death. FASEB J. Jun
5–2018.Epub ahead of print. View Article : Google Scholar
|
|
34
|
Zeng X, Li M, Fan X, Xue S, Liang W, Fang
Z, Zeng C, Fan L, Xiong Y, Wang Y and Ye Q: Hypothermic oxygenated
machine perfusion alleviates donation after circulatory death liver
injury through regulating P-selectin-dependent and -independent
pathways in mice. Transplantation. 103:918–928. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Suzuki S, Toledo-Pereyra LH, Rodriguez FJ
and Cejalvo D: Neutrophil infiltration as an important factor in
liver ischemia and reperfusion injury. Modulating effects of FK506
and cyclosporine. Transplantation. 55:1265–1272. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hu Q, Chen H, Lan J, Chen Y, Liu Z, Xiong
Y, Zhou W, Zhong Z and Ye Q: KLF10 induced by hypothermic machine
perfusion alleviates renal inflammation through BIRC2/Noncanonical
NF-κB pathway. Transplantation. 109:e273–e286. 2025. View Article : Google Scholar
|
|
37
|
Petrusca DN, Gu Y, Adamowicz JJ, Rush NI,
Hubbard WC, Smith PA, Berdyshev EV, Birukov KG, Lee CH, Tuder RM,
et al: Sphingolipid-mediated inhibition of apoptotic cell clearance
by alveolar macrophages. J Biol Chem. 285:40322–40332. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
39
|
Yurdagul A Jr, Kong N, Gerlach BD, Wang X,
Ampomah P, Kuriakose G, Tao W, Shi J and Tabas I: ODC (Ornithine
Decarboxylase)-Dependent putrescine synthesis maintains MerTK (MER
Tyrosine-Protein Kinase) expression to drive resolution.
Arterioscler Thromb Vasc Biol. 41:e144–e159. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang Y, Zhang W, Xu Y, Wu D, Gao Z, Zhou
J, Qian H, He B and Wang G: Extracellular HMGB1 impairs
macrophage-mediated efferocytosis by suppressing the
Rab43-controlled cell surface transport of CD91. Front Immunol.
13:7676302022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Vasudevan SO, Behl B and Rathinam VA:
Pyroptosis-induced inflammation and tissue damage. Semin Immunol.
69:1017812023. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jiang K, Tu Z, Chen K, Xu Y, Chen F, Xu S,
Shi T, Qian J, Shen L, Hwa J, et al: Gasdermin D inhibition confers
antineutrophil-mediated cardioprotection in acute myocardial
infarction. J Clin Invest. 132:e1512682022. View Article : Google Scholar :
|
|
43
|
Li S, Sun Y, Song M, Song Y, Fang Y, Zhang
Q, Li X, Song N, Ding J, Lu M and Hu G:
NLRP3/caspase-1/GSDMD-mediated pyroptosis exerts a crucial role in
astrocyte pathological injury in mouse model of depression. JCI
Insight. 6:e1468522021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liang F, Zhang F, Zhang L and Wei W: The
advances in pyroptosis initiated by inflammasome in inflammatory
and immune diseases. Inflamm Res. 69:159–166. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Liu Y, Pan R, Ouyang Y, Gu W, Xiao T, Yang
H, Tang L, Wang H, Xiang B and Chen P: Pyroptosis in health and
disease: Mechanisms, regulation and clinical perspective. Signal
Transduct Target Ther. 9:2452024. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhang X, Wang Z, Li X, Chen J, Yu Z, Li X,
Sun C, Hu L, Wu M and Liu L: Polydatin protects against
atherosclerosis by activating autophagy and inhibiting pyroptosis
mediated by the NLRP3 inflammasome. J Ethnopharmacol.
309:1163042023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Gimbrone MA Jr, Topper JN, Nagel T,
Anderson KR and Garcia-Cardeña G: Endothelial dysfunction,
hemodynamic forces, and atherogenesis. Ann N Y Acad Sci.
902:230–240. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ando J and Yamamoto K: Hemodynamic forces,
endothelial mechanotransduction, and vascular diseases. Magn Reson
Med Sci. 21:258–266. 2022. View Article : Google Scholar :
|
|
49
|
Caldwell-Kenkel JC, Currin RT, Tanaka Y,
Thurman RG and Lemasters JJ: Reperfusion injury to endothelial
cells following cold ischemic storage of rat livers. Hepatology.
10:292–299. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Peralta C, Jiménez-Castro MB and
Gracia-Sancho J: Hepatic ischemia and reperfusion injury: Effects
on the liver sinusoidal milieu. J Hepatol. 59:1094–1106. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Upadhya GA, Topp SA, Hotchkiss RS, Anagli
J and Strasberg SM: Effect of cold preservation on intracellular
calcium concentration and calpain activity in rat sinusoidal
endothelial cells. Hepatology. 37:313–323. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Knijff LWD, van Kooten C and Ploeg RJ: The
effect of hypothermic machine perfusion to ameliorate
Ischemia-Reperfusion injury in donor organs. Front Immunol.
13:8483522022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Schlegel A, de Rougemont O, Graf R,
Clavien PA and Dutkowski P: Protective mechanisms of end-ischemic
cold machine perfusion in DCD liver grafts. J Hepatol. 58:278–286.
2013. View Article : Google Scholar
|
|
54
|
Chatauret N, Coudroy R, Delpech PO,
Vandebrouck C, Hosni S, Scepi M and Hauet T: Mechanistic analysis
of nonoxygenated hypothermic machine perfusion's protection on warm
ischemic kidney uncovers greater eNOS phosphorylation and
vasodilation. Am J Transplant. 14:2500–2514. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hergenreider E, Heydt S, Tréguer K,
Boettger T, Horrevoets AJ, Zeiher AM, Scheffer MP, Frangakis AS,
Yin X, Mayr M, et al: Atheroprotective communication between
endothelial cells and smooth muscle cells through miRNAs. Nat Cell
Biol. 14:249–256. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Jha P and Das H: KLF2 in regulation of
NF-κB-Mediated immune cell function and inflammation. Int J Mol
Sci. 18:23832017. View Article : Google Scholar
|
|
57
|
Dekker RJ, Boon RA, Rondaij MG, Kragt A,
Volger OL, Elderkamp YW, Meijers JC, Voorberg J, Pannekoek H and
Horrevoets AJ: KLF2 provokes a gene expression pattern that
establishes functional quiescent differentiation of the
endothelium. Blood. 107:4354–4363. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Nayak L, Lin Z and Jain MK: 'Go with the
flow': How Krüppel-like factor 2 regulates the vasoprotective
effects of shear stress. Antioxid Redox Signal. 15:1449–1461. 2011.
View Article : Google Scholar
|
|
59
|
Gallinat A, Efferz P, Paul A and Minor T:
One or 4 h of 'in-house' reconditioning by machine perfusion after
cold storage improve reperfusion parameters in porcine kidneys.
Transpl Int. 27:1214–1219. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Xiao L, Magupalli VG and Wu H: Cryo-EM
structures of the active NLRP3 inflammasome disc. Nature.
613:595–600. 2023. View Article : Google Scholar
|
|
61
|
Lamkanfi M and Dixit VM: Inflammasomes and
their roles in health and disease. Annu Rev Cell Dev Biol.
28:137–161. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zhang Y, Yang W, Li W and Zhao Y: NLRP3
Inflammasome: Checkpoint connecting innate and adaptive immunity in
autoimmune diseases. Front Immunol. 12:7329332021. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Mangan MSJ, Olhava EJ, Roush WR, Seidel
HM, Glick GD and Latz E: Targeting the NLRP3 inflammasome in
inflammatory diseases. Nat Rev Drug Discov. 17:588–606. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Franchi L, Eigenbrod T, Muñoz-Planillo R
and Nuñez G: The inflammasome: A caspase-1-activation platform that
regulates immune responses and disease pathogenesis. Nat Immunol.
10:241–247. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Chen Y, Ye X, Escames G, Lei W, Zhang X,
Li M, Li M, Jing T, Yao Y, Qiu Z, et al: The NLRP3 inflammasome:
Contributions to inflammation-related diseases. Cell Mol Biol Lett.
28:512023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Dai J, Chen Q, Huang W, Shi K, Zhang Y, Li
T, Mou T, Huang Z and Wu Z: Liver kinase B1 attenuates liver
ischemia/reperfusion injury via inhibiting the NLRP3 inflammasome.
Acta Biochim Biophys Sin (Shanghai). 53:601–611. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Pu JL, Huang ZT, Luo YH, Mou T, Li TT, Li
ZT, Wei XF and Wu ZJ: Fisetin mitigates hepatic
ischemia-reperfusion injury by regulating GSK3β/AMPK/NLRP3
inflammasome pathway. Hepatobiliary Pancreat Dis Int. 20:352–360.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Bai B, Yang Y, Wang Q, Li M, Tian C, Liu
Y, Aung LHH, Li PF, Yu T and Chu XM: NLRP3 inflammasome in
endothelial dysfunction. Cell Death Dis. 11:7762020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Xu T, Chen T, Fang H, Shen X, Shen X, Tang
Z and Zhao J: Human umbilical cord mesenchymal stem cells repair
endothelial injury and dysfunction by regulating NLRP3 to inhibit
endothelial cell pyroptosis in Kawasaki disease. Inflammation.
47:483–502. 2024. View Article : Google Scholar
|
|
70
|
Chen Z, Martin M, Li Z and Shyy JY:
Endothelial dysfunction: The role of sterol regulatory
element-binding protein-induced NOD-like receptor family pyrin
domain-containing protein 3 inflammasome in atherosclerosis. Curr
Opin Lipidol. 25:339–349. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Keyel PA: How is inflammation initiated?
Individual influences of IL-1, IL-18 and HMGB1. Cytokine.
69:136–145. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Poon IKH and Ravichandran KS: Targeting
efferocytosis in inflammaging. Annu Rev Pharmacol Toxicol.
64:339–357. 2024. View Article : Google Scholar
|
|
73
|
Boada-Romero E, Martinez J, Heckmann BL
and Green DR: The clearance of dead cells by efferocytosis. Nat Rev
Mol Cell Biol. 21:398–414. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Kourtzelis I, Hajishengallis G and
Chavakis T: Phagocytosis of apoptotic cells in resolution of
inflammation. Front Immunol. 11:5532020. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Shi H, Wang X, Li F, Gerlach BD, Yurdagul
A Jr, Moore MP, Zeldin S, Zhang H, Cai B, Zheng Z, et al:
CD47-SIRPα axis blockade in NASH promotes necroptotic hepatocyte
clearance by liver macrophages and decreases hepatic fibrosis. Sci
Transl Med. 14:eabp83092022. View Article : Google Scholar
|
|
76
|
Hu H, Cheng X, Li F, Guan Z, Xu J, Wu D,
Gao Y, Zhan X, Wang P, Zhou H, et al: Defective efferocytosis by
aged macrophages promotes STING signaling mediated inflammatory
liver injury. Cell Death Discov. 9:2362023. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Pegg AE: Functions of polyamines in
mammals. J Biol Chem. 291:14904–14912. 2016. View Article : Google Scholar : PubMed/NCBI
|