Introduction
Epithelial-mesenchymal transition (EMT) is an
essential process for tumor invasion and metastasis (1–5). EMT
is the conversion of epithelial cells into more invasive
mesenchymal cells and characterized by a loss of cell-cell contact
and apical-basal polarity through repression of the expression of
E-cadherin and other epithelial markers and activation of the
expression of mesenchymal markers (such as vimentin and
fibronectin). TGF-β and Wnt are known to induce EMT during cancer
development and progression (1–3,5–8). The
transcription factor Snail is implicated in the repression of
E-cadherin expression in response to TGF-β and Wnt. We previously
showed that Wnt/Snail signaling induces the Warburg effect (also
termed as glycolytic switch) (9).
The Warburg effect is that cancer cells mainly use glycolysis for
ATP production instead of mitochondrial oxidative phosphorylation,
even in the presence of oxygen. The glycolytic switch increases the
availability of biosynthetic precursors for nucleotides, lipids and
amino acids needed for tumor cell proliferation (10–14).
Mitochondrial dysfunction is closely linked to the induction of
glycolytic switch (15–18). In cancer cells, inhibition of the
glycolytic switch results in growth failure; thus, molecules
implicated in glycolytic switch are regarded as potential target
for cancer therapies.
Dlx-2 is one of the human distal-less (Dlx) gene
family proteins that play an important role(s) in the embryonic
development (19,20). Increased Dlx-2 expression is
observed in a number of tumor tissues, suggesting an essential role
of Dlx-2 in carcinogenesis (21–23).
Recently, Dlx-2 was shown to be induced by TGF-β and involved in
the shift of the TGF-β tumor suppressive activity to its tumor
promoting activity (23).
Inhibition of Dlx-2 expression has been shown to impair the
metastasis ability of B16 melanoma cells (23). In addition, we previously showed
that Dlx-2 is induced by reactive oxygen species (ROS) and is
implicated in metabolic stress-induced necrosis (21). ROS contribute to cancer development
and progression (24,25). However, the precise mechanism of
Dlx-2 in tumor progression remains to be elucidated.
In this study, we show that Dlx-2 is implicated in
TGF-β- and Wnt-induced EMT and glycolytic switch via Snail
induction. We also show that TGF-β/Wnt suppressed mitochondrial
respiration through inhibiting cytochrome c oxidase (COX),
the terminal enzyme of the mitochondrial respiratory chain, by
Dlx-2/Snail cascade. COXVIc appeared to be a common target of
Dlx-2, Snail, TGF-β and Wnt. Taken together, our results show that
Dlx-2 plays an important role in TGF-β/Wnt-induced EMT, glycolytic
switch and mitochondrial repression and COX inhibition.
Materials and methods
Cell culture
MCF-7, Madin Darby Canine Kidney (MDCK) and L cells
were obtained from the American Type Culture Collection (ATCC;
authenticated by short tandem repeat profiling). Wnt3a-secreting L
cells and HCT116 cells were provided by Dr D.S. Min and Dr Y.J.
Kim, respectively (Pusan National University, Pusan, Korea). The
cell lines were passaged two times per week and low-passage
cultures (passages 5–25) were used for the experiments. The cells
were routinely tested negative for mycoplasma using the Mycoplasma
PCR Detection kit (iNtRON Biotechnology). MCF-7 and MDCK cells were
cultured in Eagle’s minimal essential medium (EMEM; Hyclone, Logan,
UT, USA); and L cells and Wnt3a secreting L cells in Dulbecco’s
modified Eagle’s medium (DMEM; Hyclone); HCT116 cells were cultured
RPMI supplemented with 10% (v/v) heat-inactivated fetal bovine
serum (FBS, Hyclone) and 1% penicillin-streptomycin (PS, Hyclone)
in a 37°C humidified incubator with 5% CO2. Recombinant
TGF-β (R&D Systems, MN, USA) was applied to cells at a
concentration of 10 ng/ml.
Transfection and short hairpin RNA
(shRNA) interference
The expression vectors pCAGGS-Dlx-2 (provided by Dr
John L.R. Rubenstein, University of California at San Francisco)
and pCR3.1-Snail-Flg (provided by J.I. Yook, Yonsei University,
Korea) were transfected into MCF-7 cells using jetPEI (Polyplus
transfection). pSUPER vectors for shRNA against control, Dlx-2,
Snail, Smad2, Smad3, Smad4, β-catenin, TCF4, Axin1, Axin2 and
COXVIc (abbreviations; shCon, shDlx-2, etc.) were produced and
transfected as described previously (9).
Immunoblotting and quantitative real-time
PCR (qRT-PCR)
Immunoblotting and qRT-PCR were performed as
described previously (9,26). Immunoblotting was performed with
the following antibodies: Dlx-2 (Millipore, Billerca, MA, USA);
Snail (Abgent, San Diego, CA, USA); E-cadherin, vimentin, COXVIIc
and COX19 (Santa Cruz, CA, USA); COXVIc, COXVIIa, and COXVIIb
(Mitoscience, Eugene, OR, USA); SCO2 and COX18 (Abcam, Cambridge,
MA, UK); α-tubulin (Biogenex, CA, USA). Total mRNA was isolated
from cells by using the TRIzol (Invitrogen, Carlsbad, CA, USA)
according to the supplier’s instructions. Transcript levels were
assessed with qRT-PCR with primers for Dlx-2, Snail, E-cadherin and
β-actin. Values are normalized to β-actin.
Immunofluorescence (IF) microscopy
MCF-7 cells were fixed for 10 min in 3.7%
formaldehyde in PBS, permeabilized in PBS containing 0.2% Triton
X-100 for 30 min, and blocked with 2% BSA in 0.1% PBST for 3 h. For
E-cadherin staining, cells were incubated with mouse
anti-E-cadherin (Santa Cruz) antibody for overnight at 4°C and
immunostained with Alexa-Fluoro-488-labeled anti-mouse secondary
antibody (Molecular Probes, NY, USA). Hoechst 33342 (Molecular
Probes) was used to stain cell nuclei.
Chromatin immunoprecipitation (ChIP)
assay
ChIP assays were conducted using a ChIP Assay kit
(Millipore). IgG, anti-Dlx-2 or anti-Snail (Santa Cruz) was used to
immunoprecipitate DNA-containing complexes. ChIP-enriched DNA was
analyzed by PCR using primers complementary to the promoter
regions.
Assays for mitochondrial respiration, COX
activity, glucose (Glc) consumption, lactate (Lac) production and
ATP production
Mitochondrial respiration and COX activity were
measured as described previously (9,27).
For mitochondrial respiration assay, exponentially growing cells
(1.5×106) were washed with TD buffer (137 mM NaCl, 5 mM
KCl, 0.7 mM Na2HPO4, 25 mM Tris-HCl, pH 7.4),
and were collected and resuspended in complete medium without
phenol red. The cells (5×105) were transferred to the
Mitocell chamber equipped with a Clark oxygen electrode (782 Oxygen
Meter, Strathkelvin Instruments, Glasgow, UK). Oxygen consumption
rates were measured after adding 30 μM DNP to obtain maximum
respiration rate and its specificity for mitochondrial respiration
was confirmed by adding 5 mM KCN (29). COX activity was determined by
measuring the KCN-sensitive COX-dependent O2 consumption
rate by adding 3 mM TMPD in the presence of 30 μM DNP and 20 μM
antimycin A. Glc, Lac and intracellular ATP levels were determined
using a glucose oxidation assay kit (Sigma, MO, USA), a
colorimetric and fluorescence-based lactate assay kit (BioVision,
CA, USA), and an ATP Bioluminescence Assay kit (Roche,
Switzerland), respectively. The level of ATP produced by aerobic
respiration and glycolysis was determined by measuring Lac
production and oxygen consumption (9,28).
Human tumor samples
All human tissues from patients #70331 (infiltrating
ductal carcinoma), #69965 (invasive ductal carcinoma), #69941
(metaplastic carcinoma) and #70168 (pleomorphic lobular carcinoma)
with breast cancer and normal matched tissue pairs from the same
individuals were provided by the National Biobank of Korea, PNUH in
compliance with all the regulations related to biomedical research
with human samples, including informed consent of the patients for
the use of their samples. We performed qRT-PCR and immunoblotting
with cancer tissues. TRIzol extraction of total RNA and subsequent
extraction of protein was carried out essentially according to the
manufacturer’s specifications (Invitrogen Corp.). To a 50–100 mg
tissue segment, 1 ml of TRIzol was added, and the sample was
homogenized with 2–3 min homogenisation with a tissue lyser
(Qiagen, Hilden, Germany) at 30 Hz.
Measurement of circularity
For circularity, microscopic images were analyzed
with Axiovision LE software (Release 4.8 version). Circularity was
measured with the Axiovision LE software Measure command that
calculates object circularity using the formula circularity =
4π(area/perimeter2). Circularity value of 1.0 indicates
a perfect circle. As the value approaches 0.0, it indicates an
increasingly elongated polygon.
Statistical analysis
qRT-PCR and assays for mitochondrial respiration,
COX activity, Glc consumption, Lac production and ATP production
were performed at least in triplicate and most experiments were
repeated more than twice. Data were analyzed by the Student’s
t-test (unpaired, two-tailed) and results were expressed as mean ±
SE. P<0.05 was considered statistically significant.
Results and Discussion
Dlx-2 induces EMT via Snail
activation
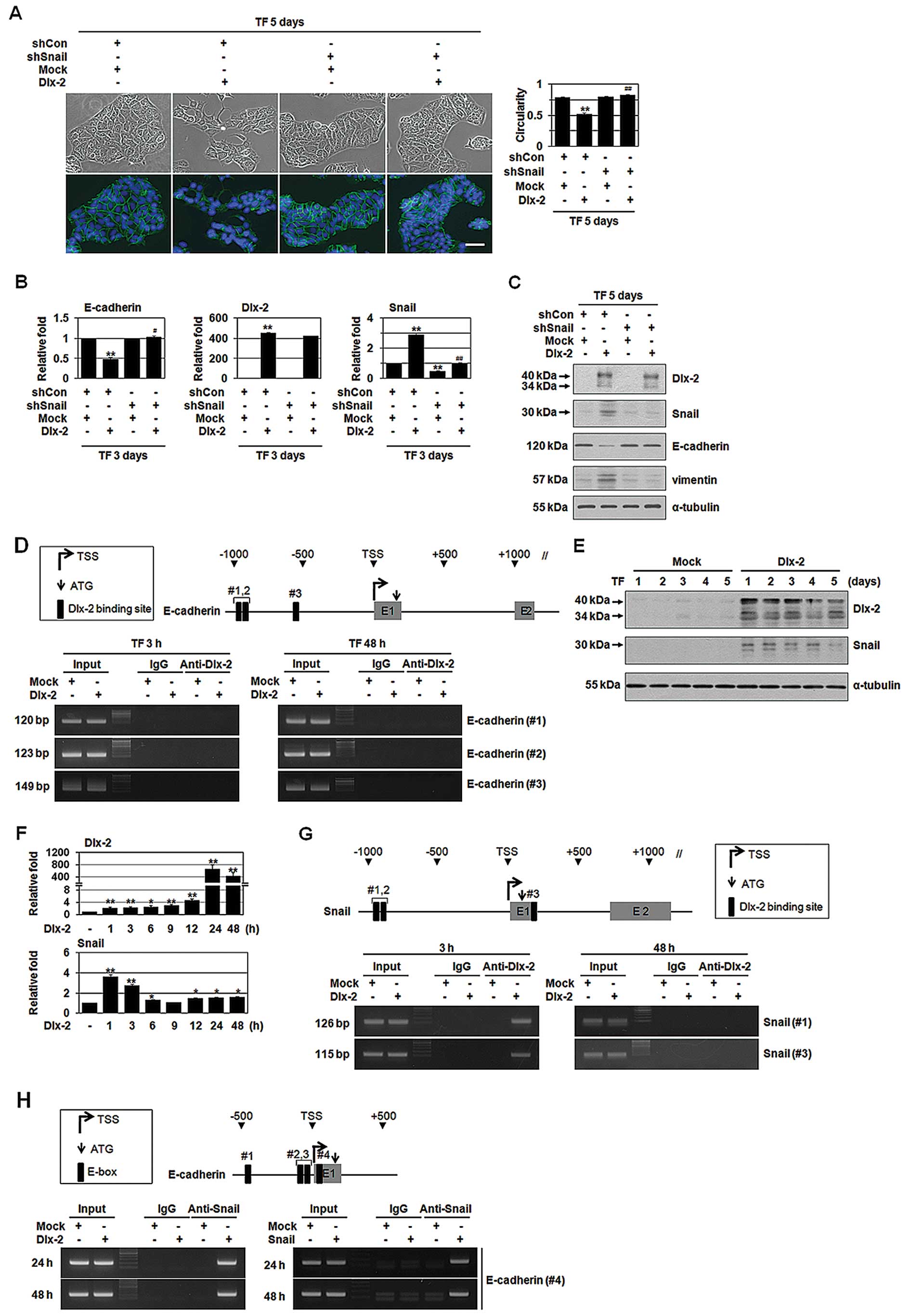
We examined the effects of Dlx-2 overexpression in
non-invasive breast cancer cell line MCF-7. Dlx-2 overexpression in
MCF-7 cells induced the loss of cell polarity and the formation of
elongated morphology with pseudopodia, which are a characteristic
of mesenchymal cells, indicating that Dlx-2 may induce EMT
(Fig. 1A). Spindle quantification
also supported Dlx-2-induced EMT (Fig.
1A). The phenotypical change was accompanied by a decreased
expression of an epithelial marker E-cadherin, as revealed by IF,
qRT-PCR and immunoblotting (Fig.
1A–C). In addition, Dlx-2 increased the levels of a mesenchymal
marker vimentin (Fig. 1C).
We examined whether Dlx-2 directly regulates
E-cadherin expression. We found three putative Dlx-2 binding sites
between −1,000 and +1,000 from the transcription start site (TSS)
in the promoter of E-cadherin. However, Dlx-2 binding to the
E-cadherin promoter was not detected in ChIP analysis
(Fig. 1D). Thus, we thought that
Dlx-2 may indirectly reduce E-cadherin by activating other
E-cadherin repressor. Snail is a typical E-cadherin repressor for
EMT (2). Therefore, we examined
whether Snail is involved in Dlx-2-induced EMT. Dlx-2
overexpression prominently increased Snail protein (Fig. 1E) and mRNA levels (Fig. 1F). Three putative Dlx-2 binding
sites were found in the Snail promoter (Fig. 1G). ChIP assay showed Dlx-2 binding
to the Snail promoter (Fig.
1G), indicating that Dlx-2 regulates Snail expression. Note
that Dlx-2 binding to the Snail promoters was detected only
at an early time-point (3 h) after transfection (Fig. 1G). We examined whether Snail is
involved in Dlx-2-induced EMT. shSnail (hereafter, shSnail)
prevented Dlx-2-induced EMT and E-cadherin downregulation and
vimentin upregulation (Fig. 1A–C),
indicating that Dlx-2 induces EMT via Snail activation. We
performed ChIP assay to examine Snail binding to the
E-cadherin promoter. 4 E-boxes for Snail binding are found
in human E-cadherin promoter; but Snail has been shown to
bind to the E-boxes 1, 3 and 4 with the most strong binding
activity for E-box 4 (6,29). Dlx-2 and Snail overexpression
enhanced Snail binding to the E-box 4 of the E-cadherin
promoter (Fig. 1H), indicating
that Dlx-2-induced Snail can interact with E-box sites (including
E-box 4) in the E-cadherin promoter to repress E-cadherin
expression.
Dlx-2/Snail cascade is implicated in
TGF-β- and Wnt3a-induced EMT
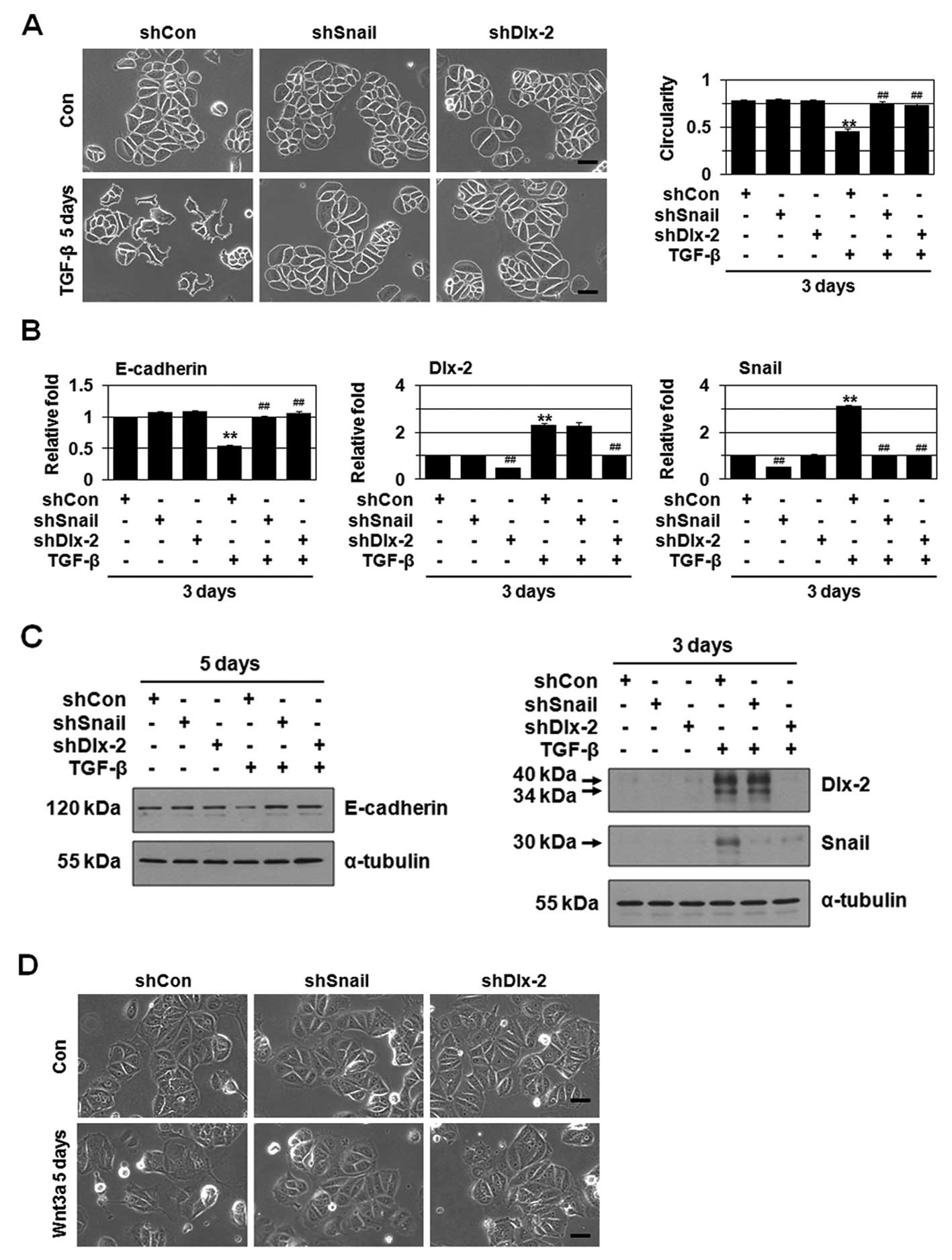
TGF-β and Wnt signaling pathways are known to induce
EMT through Snail activation (2,3,8).
Thus, we examined if the Dlx-2/Snail cascade is involved in TGF-β-
and Wnt-induced EMT. TGF-β or Wnt3a-conditioned medium (CM,
obtained from Wnt3a-secreting L cells) induced EMT and E-cadherin
downregulation (Fig. 2A–F). TGF-β
and Wnt3a also increased expression of Dlx-2 and Snail (Fig. 2B, C, E and F). Note that TGF-β
increased Snail expression at both mRNA and protein levels, whereas
Wnt induced Snail at the level of protein but not mRNA (Fig. 2B, C, E and F). In the Wnt
signaling, Axin2 is one of the Wnt target genes and regulates EMT
by acting as a chaperone for nuclear export of GSK3β that is the
dominant kinase responsible for Snail protein turnover and
activity, in human breast cancer cells, thereby increasing Snail
protein stability in the nucleus (30). Thus, in Wnt signaling, Dlx-2 may be
involved in GSK3β-mediated Snail protein turnover; although it
remained to be elucidated. shDlx-2 suppressed Snail expression,
whereas shSnail had no effect on Dlx-2 expression (Fig. 2B, C, E and F), indicating that
Dlx-2 acts upstream of Snail. shDlx-2 or shSnail appeared to block
TGF-β- and Wnt3a-induced EMT and E-cadherin downregulation
(Fig. 2A–F).
TGF-β induces EMT through activation of Smad
signaling pathways (8). Smad2/3/4
shRNA suppressed TGF-β-induced EMT and E-cadherin downregulation
(Fig. 2G) as well as Dlx-2 and
Snail expression (Fig. 2G and H).
Wnt induces EMT via canonical pathways, which includes β-catenin,
TCF4 and Axin1/2. shRNA for β-catenin, TCF4 and Axin1/2 suppressed
Wnt3a-induced EMT/E-cadherin downregulation (Fig. 2I) as well as Dlx-2 (but not Snail)
expression (Fig. 2I and J). These
results supported that the Dlx-2/Snail cascade is implicated in
TGF-β- and Wnt3a-induced EMT.
We further examined the effects of shDlx-2 and
shSnail on the EMT in HCT116 and MDCK cells. shDlx-2 and shSnail
suppressed Wnt3a-induced EMT and E-cadherin downregulation in
HCT116 cells (Fig. 2K and L).
Similar inhibitory effects of shDlx-2 and shSnail on TGF-β-induced
EMT were observed in MDCK cells. shDlx-2 and shSnail prevented
TGF-β-induced EMT and E-cadherin downregulation (Fig. 2M and N).
Dlx-2/Snail signaling is involved in
TGF-β- and Wnt3a-induced glycolytic switch and mitochondrial
repression
Wnt3a/Snail cascade has been shown to induce
glycolytic switch and mitochondrial repression (9). Therefore, we examined whether Dlx-2
is involved in the Snail-induced glycolytic switch. Dlx-2
overexpression significantly increased Glc consumption and Lac
production (Fig. 3A). In addition,
Dlx-2 overexpression decreased O2 consumption (Fig. 3A). ATP levels were similar in both
control and Dlx-2 transfected cells (data not shown). By measuring
oxygen consumption and Lac production, we estimated the relative
contributions of glycolysis and aerobic respiration in total ATP
production. Dlx-2 increased the ratio of ATP produced by glycolysis
versus ATP produced by aerobic respiration (Fig. 3A), indicating that Dlx-2 induces
glycolytic switch. Dlx-2-induced glycolytic switch and
mitochondrial repression were prevented by shSnail (Fig. 3A), indicating that Dlx-2 induces
glycolytic switch/mitochondrial repression via Snail.
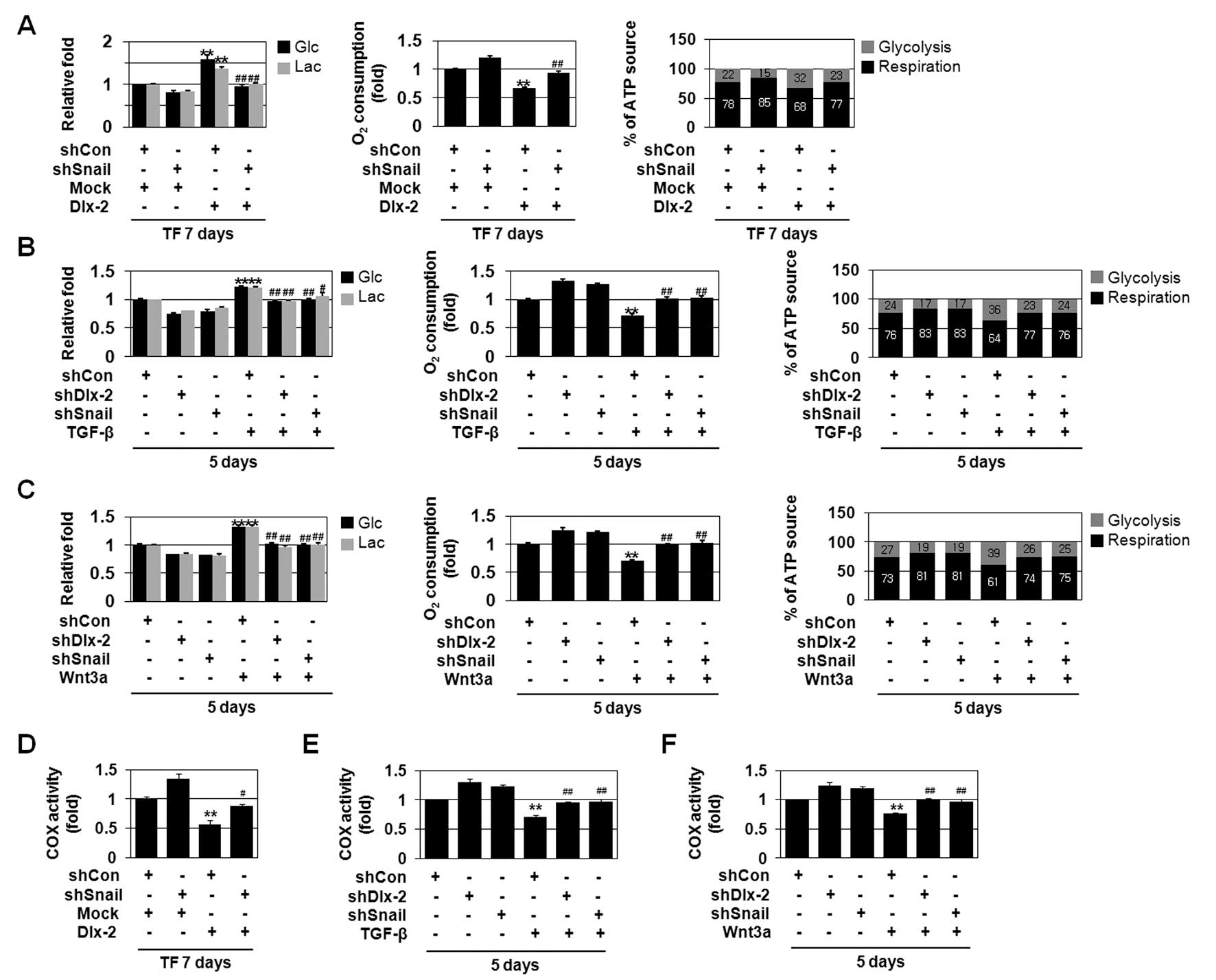 | Figure 3The Dlx-2/Snail cascade is implicated
in TGF-β- and Wnt3a-induced glycolytic switch, mitochondrial
repression and COX inhibition. (A) MCF-7 cells were co-transfected
with Dlx-2 and shSnail. The cells were analyzed for glucose (Glc)
consumption, lactate (Lac) production, mitochondrial respiration
and percentage (%) of ATP source. **P<0.01 versus
mock, ##P<0.01 versus Dlx-2. (B and C) MCF-7 cells
were transfected with shDlx-2 or shSnail and then treated with
TGF-β (B) or Wnt3a CM (C). The cells were analyzed by Glc
consumption, Lac production, mitochondrial respiration and
percentage (%) of ATP source. **P<0.01 versus
untreated, #P<0.05; ##P<0.01 versus
shCon. The amount of ATP produced by aerobic respiration (black
bars) and glycolysis (gray bars) was calculated by measuring oxygen
consumption and Lac production in the cells (right panels in A–C).
(D) COX activity was measured in MCF-7 cells co-transfected with
Dlx-2 and shSnail. **P<0.01 versus control (mock and
shCon), #P<0.05 versus Dlx-2. (E and F) MCF-7 cells
transfected with shDlx-2 or shSnail were treated with TGF-β (E) or
Wnt3a CM (F), and COX activity was measured. **P<0.01
versus untreated, ##P<0.01 versus shCon. All error
bars represent SE. |
Then, we examined if TGF-β and Wnt3a induce
glycolytic switch/mitochondrial repression via the Dlx-2/Snail
cascade. TGF-β and Wnt3a induced Glc consumption and Lac production
(Fig. 3B and C). In addition,
TGF-β and Wnt3a reduced O2 consumption (Fig. 3B and C). Although total ATP
concentrations remained the same in all cells, TGF-β and Wnt3a
increased the ratio of ATP produced by glycolysis versus ATP
produced by aerobic respiration (Fig.
3B and C), indicating that TGF-β and Wnt3a induce glycolytic
switch. In addition, shDlx-2 or shSnail decreased TGF-β- and
Wnt3a-induced increase of Glc consumption and Lac production and
impairment of O2 consumption (Fig. 3B and C), indicating that the
Dlx-2-Snail axis is involved in TGF-β- and Wnt3a-induced glycolytic
switch and mitochondrial repression.
Dlx-2/Snail signaling is involved in
TGF-β/Wnt-induced COX inhibition
Changes in the activity of COX, the terminal enzyme
of the mitochondrial respiratory chain, are closely associated with
decreased mitochondrial respiratory activity. Therefore, we
examined the effects of Dlx-2 on COX activity. Dlx-2 overexpression
reduced COX enzymatic activity (Fig.
3D). Dlx-2-induced COX inhibition was prevented by shSnail
(Fig. 3D), indicating that Dlx-2
induces COX inhibition via Snail activation.
We also found that TGF-β and Wnt3a reduce COX
activity (Fig. 3E and F). We
examined if Dlx-2-Snail axis is implicated in TGF-β- and
Wnt3a-induced COX inhibition. shDlx-2 or shSnail decreased TGF-β-
and Wnt3a-induced impairment of COX activity (Fig. 3E and F), indicating that the
Dlx-2-Snail axis is involved in TGF-β- and Wnt3a-induced
mitochondrial repression.
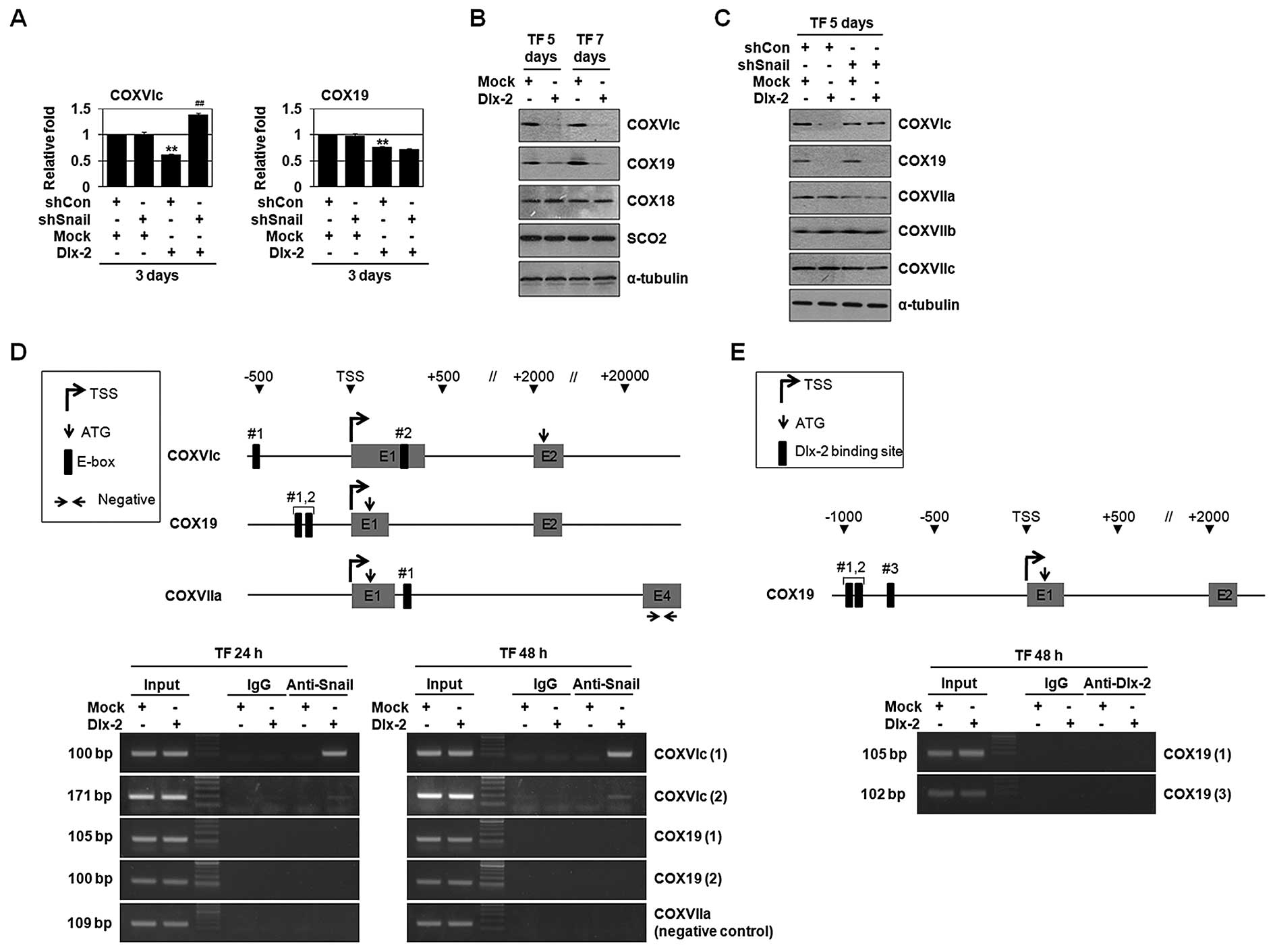
Dlx-2/Snail signaling is implicated in
TGF-β/Wnt-induced downregulation of multiple COX subunits and
assembly factors
Eukaryotic COX is composed of 13 different subunits
and its assembly is regulated by a sequential action of several
nucleus-encoded assembly factors. We examined the effects of Dlx-2
and Snail on the gene expression of COX subunits and assembly
factors using real-time PCR (Table
IV). Dlx-2 downregulated the expression of COXVIc and COX19
(Table IV and Fig. 4A–C). Snail has been shown to
decrease mRNA levels of COXVIc, COXVIIa and COXVIIc (9). Note that Snail-mediated COXVIIa and
COXVIIc repression was not observed in Dlx-2 expressing cells.
shSnail suppressed Dlx-2-induced reduction in the levels of COXVIc,
but not COX19 (Fig. 4A and C),
suggesting that Snail is implicated in Dlx-2-mediated COXVIc gene
repression, but not in COX19 gene repression. Dlx-2 overexpression
enhanced Snail binding to the COXVIc promoter, but not COX19
promoter (Fig. 4D), confirming
that COXVIc, but not COX19, is regulated by a Snail-dependent
mechanism. As expected, Dlx-2 also did not bind to the COX19
promoter (Fig. 4E).
 | Table IVRegulation of COX subunits and
assembly factors by Dlx-2 and TGF-β. |
Table IV
Regulation of COX subunits and
assembly factors by Dlx-2 and TGF-β.
| Genes | Dlx-2
(n=5–13)
48 h | TGF-β
(n=5–7)
48 h |
|---|
| E-cadherin | 0.697a | 0.571a |
| COX subunits |
| COXIV | 0.697a | 0.571a |
| COXVa | 1.044 | 1.152 |
| COXVb | 1.079 | 1.102 |
| COXVIa | 1.029 | 1.167 |
| COXVIb | 0.992 | 2.371 |
| COXVIc | 0.959 | 0.953 |
| COXVIIa | 0.778a | 0.380a |
| COXVIIb | 0.900 | 0.636a |
| COXVIIc | 1.031 | 0.920 |
| COXVIII | 1.016 | 1.055 |
| Assembly
factors |
| COX10 | 1.090 | 0.905 |
| COX11 | 0.993 | 1.125 |
| COX15 | 0.927 | 0.680a |
| COX17 | 0.986 | 1.305 |
| COX18 | 1.080 | 1.156 |
| COX19 | 1.150 | 0.928 |
| LRPPRC | 0.679a | 0.994 |
| SURF1 | 1.072 | 1.049 |
| SCO1 | 1.078 | 1.152 |
| SCO2 | 1.034 | 1.035 |
We examined the effects of TGF-β and Wnt on the gene
expression of COX subunits and assembly factors using real-time
PCR. TGF-β decreased mRNA levels of COXVIc, COXVIIa and COX11
(Table IV and Fig. 4F). Snail-mediated COXVIIc
repression was not observed in TGF-β-treated cells by unknown
mechanism. shSnail suppressed TGF-β-induced reduction in the levels
of COXVIc and COXVIIa, but not COX11 (Fig. 4F). shDlx-2 suppressed TGF-β-induced
reduction in the levels of COXVIc (Fig. 4G). In case of Wnt3a, it decreased
mRNA levels of COXVIc, COXVIIa and COXVIIc (Fig. 4H) (9). shSnail suppressed Wnt3a-induced
reduction in the levels of COXVIc, COXVIIa and COXVIIc (Fig. 4H). shDlx-2 also suppressed
Wnt3a-induced reduction in the levels of COXVIc (Fig. 4I).
COXVIc was a common target of TGF-β, Wnt, Dlx-2 and
Snail. Because TGF-β- and Wnt-induced COX inhibition was suppressed
by shDlx-2, COXVIc levels seem to be more important. Thus, TGF-β-
and Wnt-induced COX inhibition is thought to be mediated by COXVIc
inhibition by the Dlx-2/Snail-mediated pathway.
We examined the effects of shCOXVIc on mitochondrial
respiration and COX activity. Without affecting the cell morphology
(Fig. 4J), shCOXVIc inhibited
mitochondrial respiration and COX activity (Fig. 4K).
The expression of Dlx-2, Snail and COXVIc
in human tumors
To further examine the physiological relevance of
Dlx-2/Snail/COXVIc cascade, we analyzed human tumor samples. We
examined the expression of Dlx-2, Snail and COXVIc by qRT-PCR using
RNAs extracted from paired biopsy of breast cancer and the
corresponding normal tissues. Dlx-2 and Snail expression were
higher and COXVIc expression was lower irrespective of the stage in
breast cancer tissues compared with matched normal tissues
(Fig. 4L). We also examined the
expression of Dlx-2 and Snail protein using immunoblotting. Dlx-2
and Snail expression were higher in breast cancer tissues than in
matched non-tumorigenic tissues (Fig.
4L). These results further support an important role of Dlx-2
and Snail in tumor development.
In this study, we show novel functions of Dlx-2 that
contribute to tumor development and progression; to induce EMT and
glycolytic switch. Dlx-2 induced EMT and glycolytic switch via
Snail activation. The Dlx-2/Snail cascade was involved in
TGF-β/Wnt-induced EMT and glycolytic switch. Furthermore, we found
that TGF-β/Wnt suppressed COX in a Dlx-2/Snail-dependent manner.
COXVIc downregulation appeared to play an important role in
TGF-β/Wnt-induced COX inhibition. Taken together, our findings
suggest that Dlx-2 plays an important role in TGF-β- and
Wnt-induced tumor progression and aggressiveness.
Acknowledgements
This study was supported by the National Research
Foundation of Korea (NRF) grant funded by the Korea government
(MSIP) (nos. 2011-0011084, 2013M2B2A9A03050902 and
2012R1A1A2044246) and by a grant from the National R&D Program
for Cancer Control, Ministry of Health and Welfare, Republic of
Korea (1320040). We thank Drs. K.L. Jang, Y.H. Moon and D. S. Min
for providing their qRT-PCR machines.
References
|
1
|
De Craene B and Berx G: Regulatory
networks defining EMT during cancer initiation and progression. Nat
Rev Cancer. 13:97–110. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nieto MA: The snail superfamily of
zinc-finger transcription factors. Nat Rev Mol Cell Biol.
3:155–166. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Puisieux A, Brabletz T and Caramel J:
Oncogenic roles of EMT-inducing transcription factors. Nat Cell
Biol. 16:488–494. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Thiery JP and Sleeman JP: Complex networks
orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell
Biol. 7:131–142. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zheng H and Kang Y: Multilayer control of
the EMT master regulators. Oncogene. 33:1755–1763. 2014. View Article : Google Scholar
|
|
6
|
Liu YN, Lee WW, Wang CY, Chao TH, Chen Y
and Chen JH: Regulatory mechanisms controlling human E-cadherin
gene expression. Oncogene. 24:8277–8290. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Polyak K and Weinberg RA: Transitions
between epithelial and mesenchymal states: acquisition of malignant
and stem cell traits. Nat Rev Cancer. 9:265–273. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee SY, Jeon HM, Ju MK, et al: Wnt/Snail
signaling regulates cytochrome c oxidase and glucose metabolism.
Cancer Res. 72:3607–3617. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cairns RA, Harris IS and Mak TW:
Regulation of cancer cell metabolism. Nat Rev Cancer. 11:85–95.
2011. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dang CV: Links between metabolism and
cancer. Genes Dev. 26:877–890. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hsu PP and Sabatini DM: Cancer cell
metabolism: Warburg and beyond. Cell. 134:703–707. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Vander Heiden MG, Cantley LC and Thompson
CB: Understanding the Warburg effect: the metabolic requirements of
cell proliferation. Science. 324:1029–1033. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Warburg O: On the origin of cancer cells.
Science. 123:309–314. 1956. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Alirol E and Martinou JC: Mitochondria and
cancer: is there a morphological connection? Oncogene.
25:4706–4716. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brandon M, Baldi P and Wallace DC:
Mitochondrial mutations in cancer. Oncogene. 25:4647–4662. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kroemer G: Mitochondria in cancer.
Oncogene. 25:4630–4632. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gogvadze V, Orrenius S and Zhivotovsky B:
Mitochondria in cancer cells: what is so special about them? Trends
Cell Biol. 18:165–173. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Merlo GR, Zerega B, Paleari L, Trombino S,
Mantero S and Levi G: Multiple functions of Dlx genes. Int J Dev
Biol. 44:619–626. 2000.PubMed/NCBI
|
|
20
|
Panganiban G and Rubenstein JL:
Developmental functions of the Distal-less/Dlx homeobox genes.
Development. 129:4371–4386. 2002.PubMed/NCBI
|
|
21
|
Lee SY, Jeon HM, Kim CH, et al: Homeobox
gene Dlx-2 is implicated in metabolic stress-induced necrosis. Mol
Cancer. 10:1132011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tang P, Huang H, Chang J, Zhao GF, Lu ML
and Wang Y: Increased expression of DLX2 correlates with advanced
stage of gastric adenocarcinoma. World J Gastroenterol.
19:2697–2703. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yilmaz M, Maass D, Tiwari N, et al:
Transcription factor Dlx2 protects from TGFbeta-induced cell-cycle
arrest and apoptosis. EMBO J. 30:4489–4499. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hussain SP, Hofseth LJ and Harris CC:
Radical causes of cancer. Nat Rev Cancer. 3:276–285. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Weinberg F and Chandel NS: Reactive oxygen
species-dependent signaling regulates cancer. Cell Mol Life Sci.
66:3663–3673. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kim CH, Jeon HM, Lee SY, et al:
Implication of snail in metabolic stress-induced necrosis. PLoS
One. 6:e180002011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yoon YS, Lee JH, Hwang SC, Choi KS and
Yoon G: TGF beta1 induces prolonged mitochondrial ROS generation
through decreased complex IV activity with senescent arrest in
Mv1Lu cells. Oncogene. 24:1895–1903. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sariban-Sohraby S, Magrath IT and Balaban
RS: Comparison of energy metabolism in human normal and neoplastic
(Burkitt’s lymphoma) lymphoid cells. Cancer Res. 43:4662–4664.
1983.PubMed/NCBI
|
|
29
|
Batlle E, Sancho E, Franci C, et al: The
transcription factor snail is a repressor of E-cadherin gene
expression in epithelial tumour cells. Nat Cell Biol. 2:84–89.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yook JI, Li XY, Ota I, et al: A
Wnt-Axin2-GSK3beta cascade regulates Snail1 activity in breast
cancer cells. Nat Cell Biol. 8:1398–1406. 2006. View Article : Google Scholar : PubMed/NCBI
|