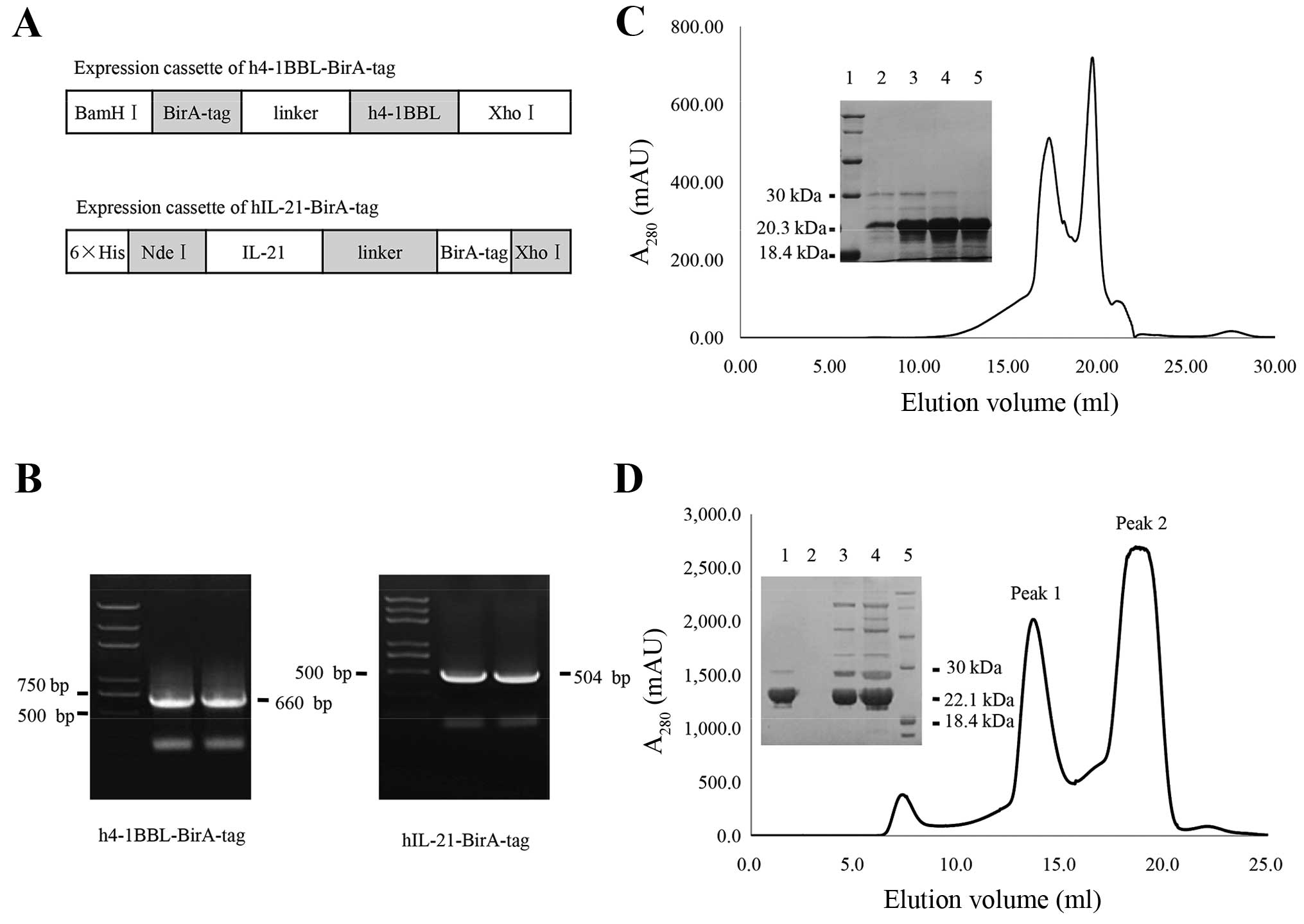
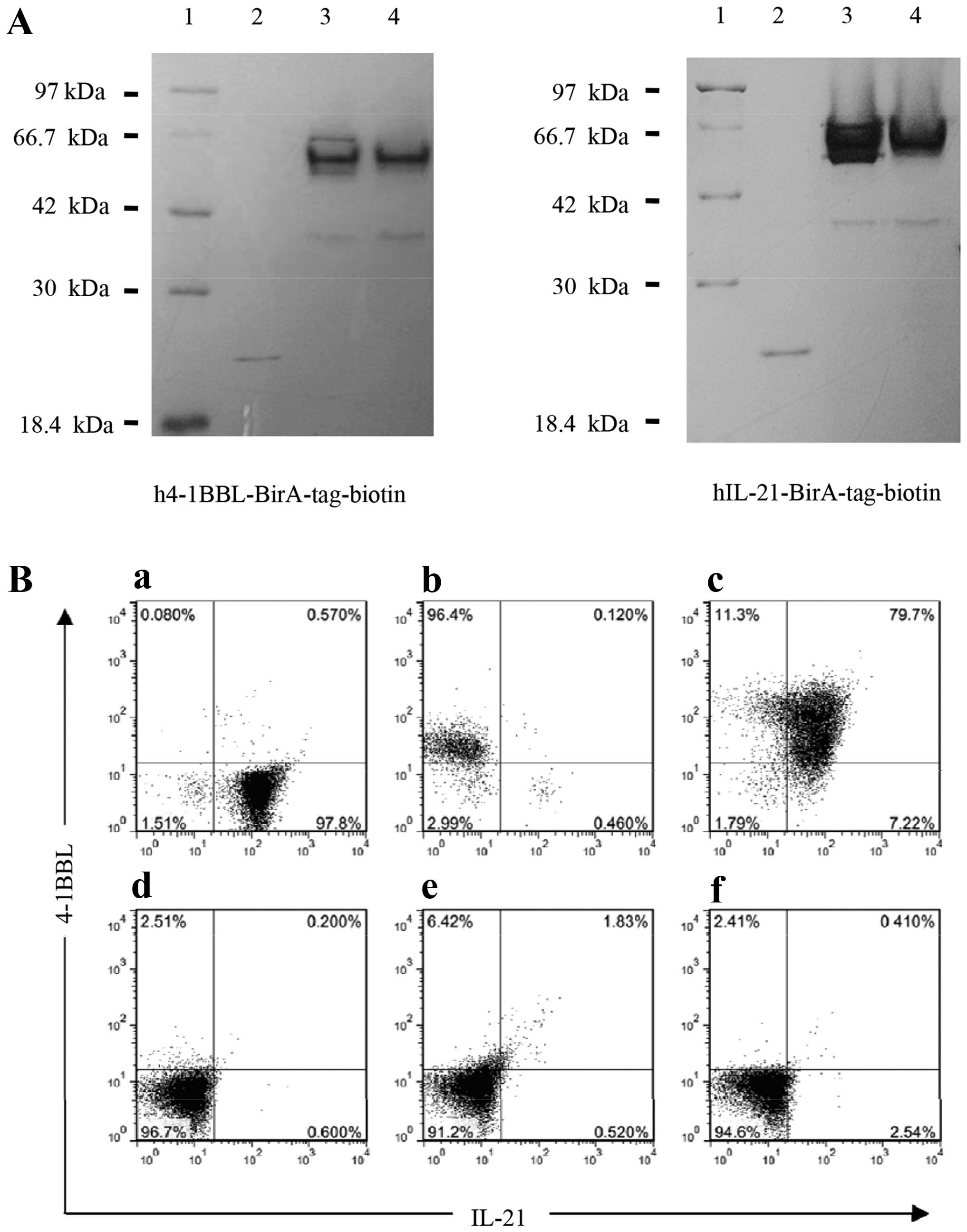
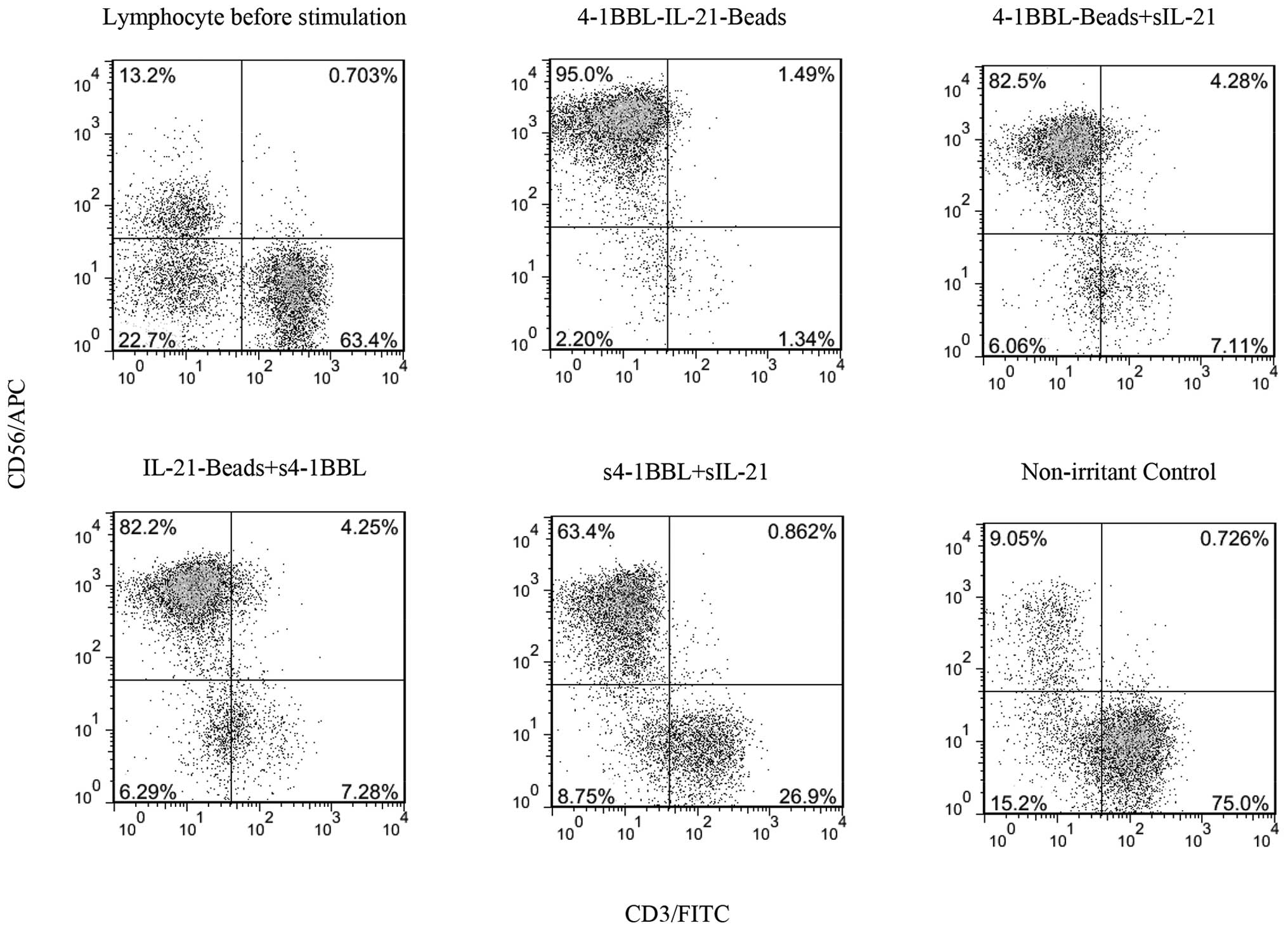
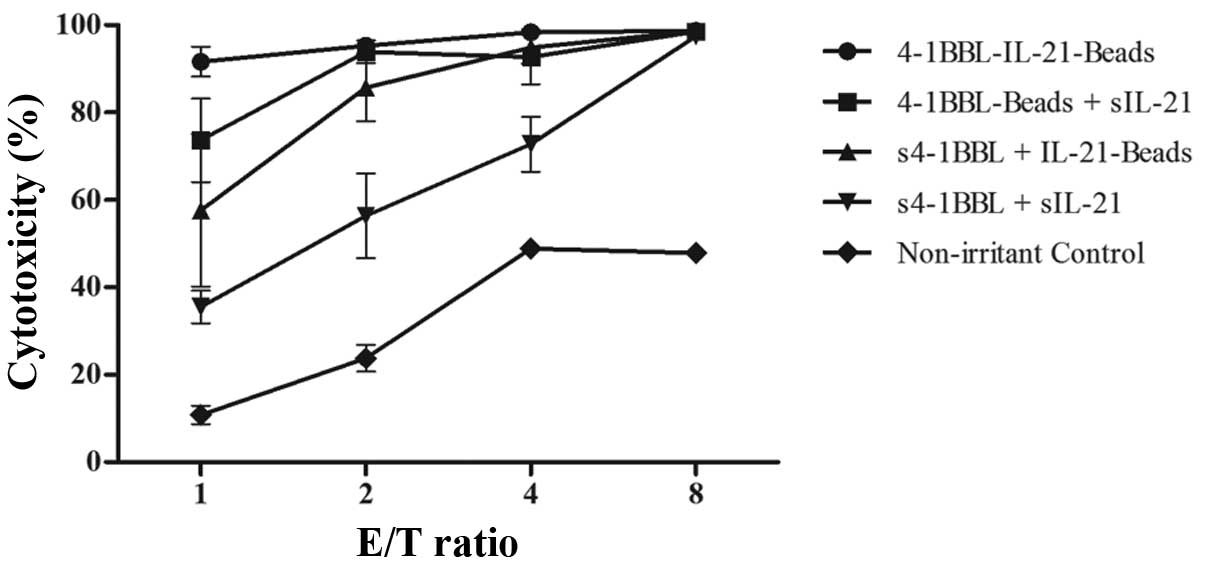
|
1
|
Nagler A, Lanier LL, Cwirla S and Phillips
JH: Comparative studies of human FcRIII-positive and negative
natural killer cells. J Immunol. 143:3183–3191. 1989.PubMed/NCBI
|
|
2
|
Lanier LL, Le AM, Civin CI, Loken MR and
Phillips JH: The relationship of CD16 (Leu-11) and Leu-19 (NKH-1)
antigen expression on human peripheral blood NK cells and cytotoxic
T lymphocytes. J Immunol. 136:4480–4486. 1986.PubMed/NCBI
|
|
3
|
Davies JO, Stringaris K, Barrett AJ and
Rezvani K: Opportunities and limitations of natural killer cells as
adoptive therapy for malignant disease. Cytotherapy. 16:1453–1466.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Childs RW and Berg M: Bringing natural
killer cells to the clinic: Ex vivo manipulation. Hematology (Am
Soc Hematol Educ Program). 2013:234–246. 2013. View Article : Google Scholar
|
|
5
|
Vivier E, Raulet DH, Moretta A, Caligiuri
MA, Zitvogel L, Lanier LL, Yokoyama WM and Ugolini S: Innate or
adaptive immunity? The example of natural killer cells. Science.
331:44–49. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ljunggren H-G and Malmberg K-J: Prospects
for the use of NK cells in immunotherapy of human cancer. Nat Rev
Immunol. 7:329–339. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Srivastava S, Lundqvist A and Childs RW:
Natural killer cell immunotherapy for cancer: A new hope.
Cytotherapy. 10:775–783. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Miller JS, Soignier Y,
Panoskaltsis-Mortari A, McNearney SA, Yun GH, Fautsch SK, McKenna
D, Le C, Defor TE, Burns LJ, et al: Successful adoptive transfer
and in vivo expansion of human haploidentical NK cells in patients
with cancer. Blood. 105:3051–3057. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Klingemann H and Boissel L: Targeted
cellular therapy with natural killer cells. Horm Metab Res.
40:122–125. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sutlu T, Stellan B, Gilljam M, Quezada HC,
Nahi H, Gahrton G and Alici E: Clinical-grade, large-scale,
feeder-free expansion of highly active human natural killer cells
for adoptive immunotherapy using an automated bioreactor.
Cytotherapy. 12:1044–1055. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Herberman RB: Cancer immunotherapy with
natural killer cells. Seminars in Oncology. Elsevier; pp. 27–30.
2002, View Article : Google Scholar
|
|
12
|
Klingemann H-G and Martinson J: Ex vivo
expansion of natural killer cells for clinical applications.
Cytotherapy. 6:15–22. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Berg M, Lundqvist A, McCoy P Jr, Samsel L,
Fan Y, Tawab A and Childs R: Clinical-grade ex vivo-expanded human
natural killer cells up-regulate activating receptors and death
receptor ligands and have enhanced cytolytic activity against tumor
cells. Cytotherapy. 11:341–355. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Imai C, Iwamoto S and Campana D: Genetic
modification of primary natural killer cells overcomes inhibitory
signals and induces specific killing of leukemic cells. Blood.
106:376–383. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Denman CJ, Senyukov VV, Somanchi SS,
Phatarpekar PV, Kopp LM, Johnson JL, Singh H, Hurton L, Maiti SN,
Huls MH, et al: Membrane-bound IL-21 promotes sustained ex vivo
proliferation of human natural killer cells. PLoS One.
7:e302642012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Carlens S, Gilljam M, Chambers BJ, Aschan
J, Guven H, Ljunggren HG, Christensson B and Dilber MS: A new
method for in vitro expansion of cytotoxic human
CD3-CD56+ natural killer cells. Hum Immunol.
62:1092–1098. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Smith CA, Farrah T and Goodwin RG: The TNF
receptor superfamily of cellular and viral proteins: Activation,
costimulation, and death. Cell. 76:959–962. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
DeBenedette MA, Shahinian A, Mak TW and
Watts TH: Costimulation of CD28− T lymphocytes by 4-1BB
ligand. J Immunol. 158:551–559. 1997.PubMed/NCBI
|
|
19
|
Hurtado JC, Kim SH, Pollok KE, Lee ZH and
Kwon BS: Potential role of 4-1BB in T cell activation. Comparison
with the costimulatory molecule CD28. J Immunol. 155:3360–3367.
1995.PubMed/NCBI
|
|
20
|
Vinay DS and Kwon BS: 4-1BB signaling
beyond T cells. Cell Mol Immunol. 8:281–284. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Melero I, Johnston JV, Shufford WW,
Mittler RS and Chen L: NK1.1 cells express 4-1BB (CDw137)
costimulatory molecule and are required for tumor immunity elicited
by anti-4-1BB monoclonal antibodies. Cell Immunol. 190:167–172.
1998. View Article : Google Scholar
|
|
22
|
Wu C, Guo H, Wang Y, Gao Y, Zhu Z and Du
Z: Extracellular domain of human 4-1BBL enhanced the function of
cytotoxic T-lymphocyte induced by dendritic cell. Cell Immunol.
271:118–123. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guo H, Jiang W, Liu W, Gao Y, Yang M, Zhou
Y, Wang J, Qi J, Cheng X, Zhu Z, et al: Extracellular domain of
4-1BBL enhanced the antitumoral efficacy of peripheral blood
lymphocytes mediated by anti-CD3 × anti-Pgp bispecific diabody
against human multidrug-resistant leukemia. Cell Immunol.
251:102–108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rabu C, Quéméner A, Jacques Y,
Echasserieau K, Vusio P and Lang F: Production of recombinant human
trimeric CD137L (4-1BBL). Cross-linking is essential to its T cell
co-stimulation activity. J Biol Chem. 280:41472–41481. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Parrish-Novak J, Dillon SR, Nelson A,
Hammond A, Sprecher C, Gross JA, Johnston J, Madden K, Xu W, West
J, et al: Interleukin 21 and its receptor are involved in NK cell
expansion and regulation of lymphocyte function. Nature. 408:57–63.
2000. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Parrish-Novak J, Foster DC, Holly RD and
Clegg CH: Interleukin-21 and the IL-21 receptor: Novel effectors of
NK and T cell responses. J Leukoc Biol. 72:856–863. 2002.PubMed/NCBI
|
|
27
|
Zeng R, Spolski R, Casas E, Zhu W, Levy DE
and Leonard WJ: The molecular basis of IL-21-mediated
proliferation. Blood. 109:4135–4142. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Burgess SJ, Marusina AI, Pathmanathan I,
Borrego F and Coligan JE: IL-21 down-regulates NKG2D/DAP10
expression on human NK and CD8+ T cells. J Immunol.
176:1490–1497. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mehta DS, Wurster AL and Grusby MJ:
Biology of IL-21 and the IL-21 receptor. Immunol Rev. 202:84–95.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang X, Lee DA, Wang Y, Wang L, Yao Y, Lin
Z, Cheng J and Zhu S: Membrane-bound interleukin-21 and CD137
ligand induce functional human natural killer cells from peripheral
blood mononuclear cells through STAT-3 activation. Clin Exp
Immunol. 172:104–112. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Goodwin RG, Din WS, Davis-Smith T,
Anderson DM, Gimpel SD, Sato TA, Maliszewski CR, Brannan CI,
Copeland NG, Jenkins NA, et al: Molecular cloning of a ligand for
the inducible T cell gene 4-1BB: A member of an emerging family of
cytokines with homology to tumor necrosis factor. Eur J Immunol.
23:2631–2641. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fa P, Zhang Z, Li J, Hu Z and Gao J:
Expression, purification and bioactivity evaluation of
streptavidin-tagged human interleukin-21 fusion protein. Nan Fang
Yi Ke Da Xue Xue Bao. 30:1240–1243. 12492010.(In Chinese).
|
|
33
|
Lee CM, McGuire H, Basten A, King C and
Christ D: Expression, purification and characterization of
recombinant interleukin-21. J Immunol Methods. 362:185–189. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Asano R, Kudo T, Makabe K, Tsumoto K and
Kumagai I: Antitumor activity of interleukin-21 prepared by novel
refolding procedure from inclusion bodies expressed in Escherichia
coli. FEBS Lett. 528:70–76. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Garboczi DN, Utz U, Ghosh P, Seth A, Kim
J, VanTienhoven EA, Biddison WE and Wiley DC: Assembly, specific
binding, and crystallization of a human TCR-alphabeta with an
antigenic Tax peptide from human T lymphotropic virus type 1 and
the class I MHC molecule HLA-A2. J Immunol. 157:5403–5410.
1996.PubMed/NCBI
|
|
36
|
Fu X, Tao L, Rivera A, Williamson S, Song
XT, Ahmed N and Zhang X: A simple and sensitive method for
measuring tumor-specific T cell cytotoxicity. PLoS One.
5:e118672010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Brown CE, Wright CL, Naranjo A, Vishwanath
RP, Chang WC, Olivares S, Wagner JR, Bruins L, Raubitschek A,
Cooper LJ, et al: Biophotonic cytotoxicity assay for
high-throughput screening of cytolytic killing. J Immunol Methods.
297:39–52. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ma J, Han H, Liu D, Li W, Feng H, Xue X,
Wu X, Niu G, Zhang G, Zhao Y, et al: HER2 as a promising target for
cytotoxicity T cells in human melanoma therapy. PLoS One.
8:e732612013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang H, Cui Y, Voong N, et al: Activating
signals dominate inhibitory signals in CD137L/IL-15 activated
natural killer cells. J Immunother. 34:187–195. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Alici E, Sutlu T, Björkstrand B, Gilljam
M, Stellan B, Nahi H, Quezada HC, Gahrton G, Ljunggren HG and
Dilber MS: Autologous antitumor activity by NK cells expanded from
myeloma patients using GMP-compliant components. Blood.
111:3155–3162. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fujisaki H, Kakuda H, Shimasaki N, Imai C,
Ma J, Lockey T, Eldridge P, Leung WH and Campana D: Expansion of
highly cytotoxic human natural killer cells for cancer cell
therapy. Cancer Res. 69:4010–4017. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lapteva N, Durett AG, Sun J, Rollins LA,
Huye LL, Fang J, Dandekar V, Mei Z, Jackson K, Vera J, et al:
Large-scale ex vivo expansion and characterization of natural
killer cells for clinical applications. Cytotherapy. 14:1131–1143.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Luhm J, Brand J-M, Koritke P, Höppner M,
Kirchner H and Frohn C: Large-scale generation of natural killer
lymphocytes for clinical application. J Hematother Stem Cell Res.
11:651–657. 2002. View Article : Google Scholar : PubMed/NCBI
|