1. Introduction
Monoterpenes, specifically the C10
isoprenoids, are dietary compounds that are found in the essential
oils of many fruits, vegetables and herbs. Monoterpenes have been
considered to play a physiological role in the mediation of
plant-environment interactions (1). Monoterpenes have been widely used as
flavorings and fragrances in the food and cosmetic industries, and
as active ingredients in many pharmaceutical products due to their
analgesic, anti-inflammatory, anti-pain and wound healing
properties (2–9). Several dietary monoterpenes, such as
d-limonene and perillyl alcohol, have been known to exert
chemotherapeutic or chemopreventive effects against cancer
(4,10–15).
Geraniol is an acyclic monoterpene alcohol that is
derived from the essential oils of aromatic plants (see ref.
16 for the detailed
physicochemical and toxicological properties of geraniol). In
addition to its use in various commercial products, including
cosmetics and fine fragrances, geraniol exerts a broad spectrum of
pharmacological activities, such as anti-microbial,
anti-inflammatory, anti-oxidant, anti-ulcer and neuroprotective
activities (17–20). Geraniol has garnered attention for
its potential role in the treatment of a range of cancers,
including breast, lung, colon, prostate, pancreatic and hepatic
cancers (21–26). In addition, geraniol has been shown
to sensitize tumor cells to commonly used chemotherapy agents, such
as 5-fluorouracil (5-FU) and docetaxel (25,27,28).
Because geraniol is classified into the
generally-recognized-as-safe (GRAS) category by the Flavor and
Extract Manufacturers Association (FEMA) and the Food and Drug
Administration (FDA) of the United States (16), its therapeutic effects against
cancer have important clinical and translational implications.
Geraniol has been demonstrated to control multiple
signaling molecules and pathways that are involved in various
biological processes, such as the cell cycle, cell survival and
proliferation, apoptosis, autophagy and metabolism (22,23,25,29–31).
Multi-target therapeutic agents are more efficacious for the
treatment of complex diseases, such as cancer, and are less
vulnerable to adaptive resistance (32–36).
Therefore, geraniol may be a promising resource that may be used as
an active ingredient, a lead compound, or a chemical moiety for
future drug development. In the present review, we highlight the
antitumor activity of geraniol against various types of cancer. We
also summarize the current knowledge with regards to the effects of
geraniol on target molecules or pathways. Our review provides novel
insight into the challenges and perspectives of geraniol research
as it pertains to future clinical investigations.
2. The effects of geraniol on cancer
hallmarks
Successive genetic lesions promote the transition of
a cell from a normal to a neoplastic state because they confer
relative fitness benefits, which ensure a high degree of
adaptability to altered environmental cues. When tumor cells evolve
progressively under selective pressures, they acquire certain
cancer hallmarks that enable the tumor to survive and grow and
ultimately become malignant (37–42).
Therefore, the molecules or pathways that are responsible for the
acquisition of these hallmarks have been considered promising
targets for the development of effective anticancer agents.
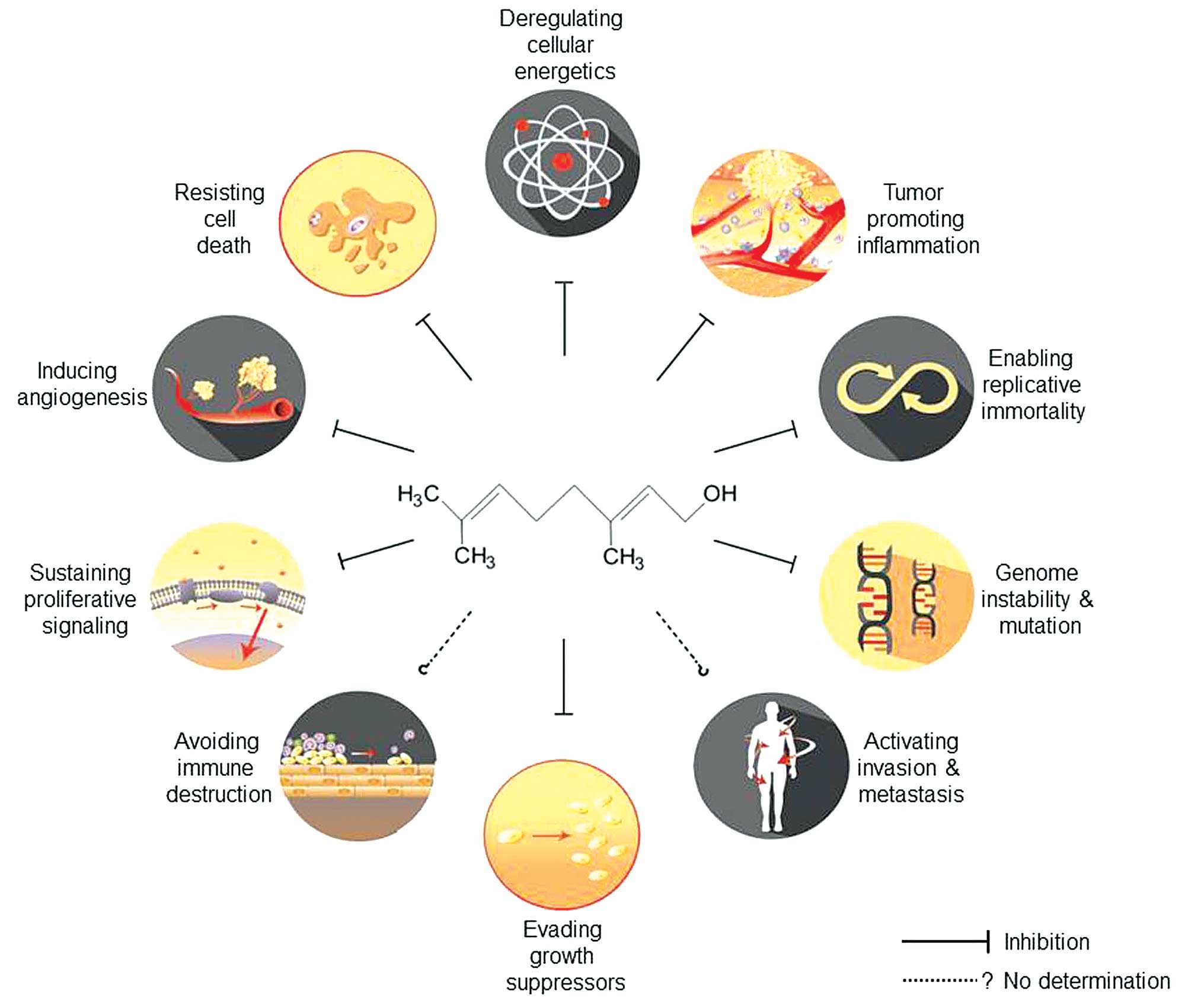
Geraniol has been demonstrated to affect many
signaling molecules and pathways that represent cancer hallmarks,
such as sustaining proliferative signaling (26,43–47),
evading growth suppressors (30),
enabling replicative immortality (22,23,25,28,46,48–50),
tumor-promoting inflammation (6–8,45,46,50–53),
inducing angiogenesis (46,54),
genome instability and mutation (49), and resisting cell death (25,26,30,45,46,49,50,54)
and deregulating cellular energetics (23,24,26,29–31,47,52).
The pleiotropic effects of geraniol on cancer hallmarks are
schematically illustrated in Fig.
1 and are summarized in Table
I. However, the detailed molecular mechanisms through which
geraniol functions will be described in the next section. These
results suggest that geraniol effectively abolishes various
properties of tumor cells that allow them to adapt and survive
under selective pressure.
 | Table IThe effects of geraniol on cancer
hallmarks. |
Table I
The effects of geraniol on cancer
hallmarks.
| Cancer
hallmark | The affected
molecule or pathway (ref.) |
|---|
| Sustaining
proliferative signaling | c-fos↓ (46), cholesterol↓ (26,44),
ERK1/2↓ (43,45), PKC↓ (43), Ras↓ (26,45),
Raf↓ (45), RhoA↓ (47) |
| Evading growth
suppressors | pAKT↓, pmTOR↓
(30) |
| Avoiding immune
destruction | N.D.a |
| Enabling
replicative immortality | PCNA↓ (46,50),
cyclin-A↓ (23,25,48),
cyclin-B↓ (25,48), cyclin-D1↓ (25,46,48),
cyclin-E↓ (23,25), CDK1↓, p27Kip1↑ (25,48),
CDK2↓ (48), CDK4↓ (23,25),
p21Cip1↑ (25,48), ODC↓ (22), TS↓, TK↓ (28) |
| Tumor-promoting
inflammation | CAT↑ (51,52),
COX-2↓ (45,46,52),
GPX↑ (45,50,52),
reduced-GSH↑ (45,50,52),
GST↑ (45,50), GR↑, MDA↓ (45,52),
NF-κB↓ (50,52), SOD↑ (51,52),
vitamin E↑ (51), IL-1β↓, IL-6↓,
TNF-α↓, NO↓, pp38↓, QR↑ (52),
n-Nrf-2↑ (53) |
| Activating invasion
and metastasis | N.D. |
| Inducing
angiogenesis | VEGF↓ (46), VEGFR-2↓ (54) |
| Genome instability
and mutation | DNA damage↓
(49) |
| Resisting cell
death | ATG5↑, LC3↑
(30), Bax↑ (25,45,46),
Bcl-2↓ (25,30,45,46,49),
Bax↑, Bcl-xL↓ (30), Bcl-w↓,
BNIP3↑ (25), caspase-3↑ (25,26,30,46,50,54),
caspase-8↑ (50), caspase-9↑
(46,50), Kim-1↓ (50), p53↓ (46,50) |
| Deregulating
cellular energetics | G6PD↑ (52), HMG-CoA reductase↓ (23,26,29,31,47),
pAMPK↑ (30) |
3. The effects of geraniol on various types
of cancer
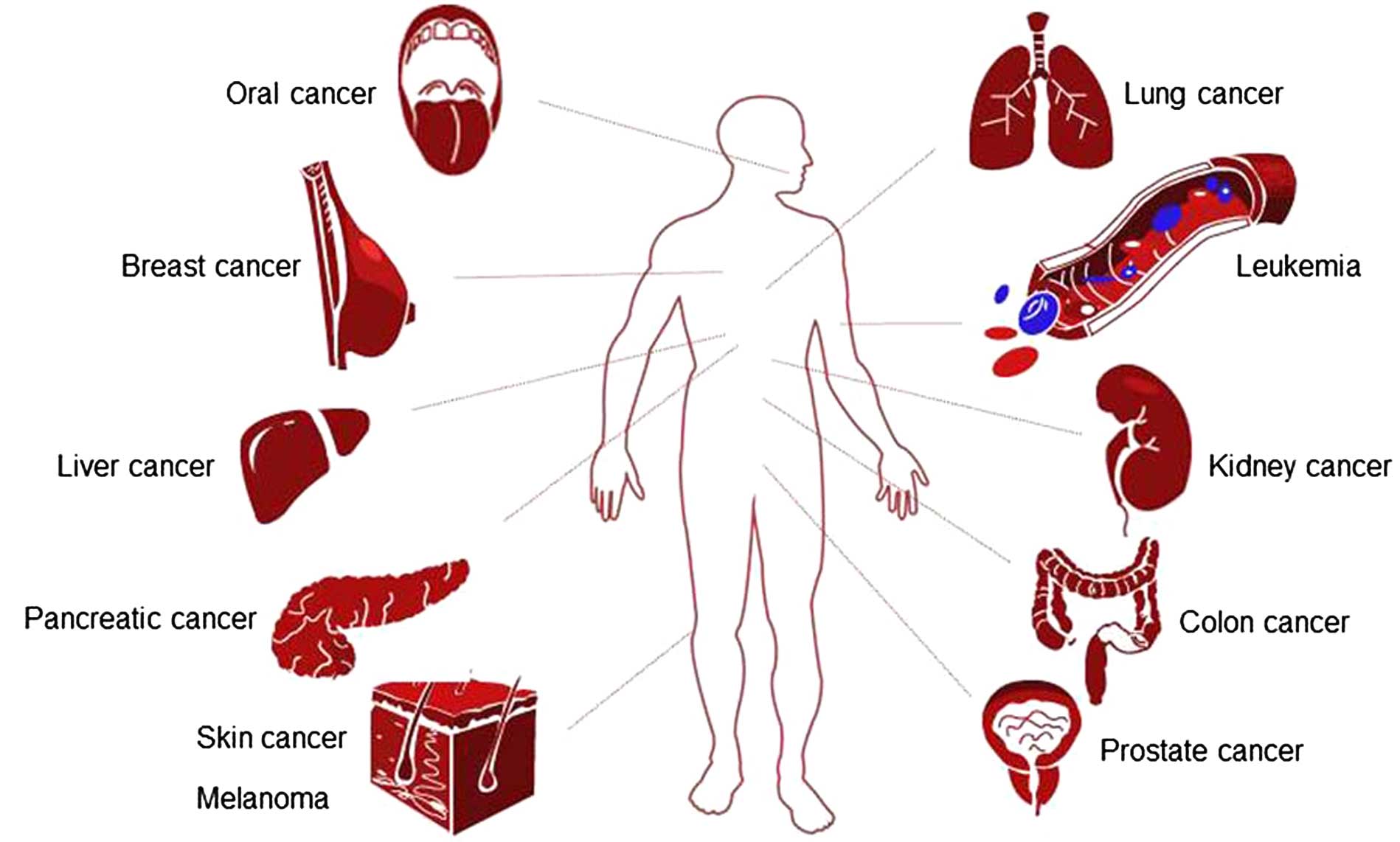
Geraniol has been found to be effective against a
broad range of cancers, including breast, lung, colon, prostate,
pancreatic, skin, liver, kidney and oral cancers (Fig. 2). The molecular mechanisms that
underlie the therapeutic effects of geraniol on various types of
cancer are summarized in Table
II. Of cancer hallmarks, enabling replicative immortality and
resisting cell death appear to be the most central targets of
geraniol (Table II). However, it
is emphasized that this can be research or publication bias: for
example, it is still not reported whether geraniol affects
avoidance of immune destruction or activating invasion and
metastasis. The effective dose range of geraniol in vitro
(cell culture), and in in vivo (xenograft or chemical
carcinogenesis) models are presented in Table III.
 | Table IIThe antitumor activity of geraniol
against cancer. |
Table II
The antitumor activity of geraniol
against cancer.
|
Cancer
hallmarks (ref.) |
|---|
|
|
|---|
| Sustaining
proliferative signaling | Evading growth
suppressors | Avoiding immune
destruction | Enabling
replicative immortality | Tumor-promoting
inflammation | Activating invasion
and metastasis | Inducing
angiogenesis | Genome instability
and mutation | Resisting cell
death | Deregulating
cellular energetics |
|---|
| Breast cancer | N.D.a | N.D. | N.D. | CDK2↓, CDK4↓,
cyclin-D1↓, cyclin-E↓, cyclin-A↓, p27Kip1↑ (23) | N.D. | N.D. | N.D. | N.D. | N.D. | HMG-CoA reductase↓
(23) |
| Lung cancer | Ras↓ (26) | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | Caspase-3↑
(26) | HMG-CoA reductase↓
(26) |
| Colon cancer | ERK1/2↓, PKC↓
(43) | N.D. | N.D. | ODC↓ (22), TS↓, TK↓ (28) | N.D. | N.D. | VEGFR-2↓ (54) | DNA damage↓
(49 | Bcl-2↓ (49), caspase-3↑ (54) | N.D. |
| Prostate
cancer | N.D. | pAKT↓, pmTOR↓
(30) | N.D. | CDK1↓, CDK4↓,
cyclin-A↓, cyclin-B↓, cyclin-D↓, cyclin-E↓, p21Cip1↑,
p27Kip1↑ (25) | N.D. | N.D. | N.D. | N.D. | ATG5↑, LC3↑,
Bcl-xL↓ (30), Bax↑, Bcl-2↓,
caspase-3↑ (25,30), Bcl-w↓, Bax↑, BNIP3↑ (25) | pAMPK↑ (30) |
| Pancreatic
cancer | N.D. | N.D. | N.D. | CDK2↓, cyclin-A↓,
cyclin-B1↓, cyclin-D1↓, p21Cip1↑, p27Kip1↑
(48) | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
| Skin cancer | ERK1/2↓, Raf↓, Ras↓
(45) | N.D. | N.D. | N.D. | CAT↑, COX-2↓, GPX↑,
reduced-GSH↑, GR↑, MDA↓, (45,52),
GST↑ (45), GR↓, IL-1β↓, IL-6↓,
NF-κB↓, NO↓, pp38↓, QR↓, SOD↓, TNF-α↓ (52) | N.D. | N.D. | N.D. | Bax↑, Bcl-2↓
(45) | G6PD↑ (52) |
| Liver cancer | RhoA↓ (47) | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | HMG-CoA reductase↓
(29,31,47) |
| Kidney cancer | N.D. | N.D. | N.D. | PCNA↓ (50) | CAT↑, GPX↑, GST↑,
reduced-GSH↑, NF-κB↓ (50) | N.D. | N.D. | N.D. | Caspase-3↑,
caspase-8↑, caspase-9↑, Kim-1↓, p53↓ (50) | N.D. |
| Oral cancer | c-fos↓ (46) | N.D. | N.D. | Cyclin-D1↓, PCNA↓
(46) | CAT↑, GPX↑, GSH↑,
SOD↑ vitamin E↑ (51), COX-2↓,
NF-κB↓, p53↓ (46), n-Nrf-2↑
(53) | N.D. | VEGF↓ (46) | N.D. | Bax↑, Bcl-2↓,
caspase-3↑, caspase-9↑ (46) | N.D. |
 | Table IIIThe effective doses of geraniol in
cancer models. |
Table III
The effective doses of geraniol in
cancer models.
| Cancer | Model | In vivo
route | Effective dose
(ref.) |
|---|
| Breast | MCF-7 cell
culture | | 300, 500 and 700 μM
(23) |
| Lung | A549 cell
culture | | 727.2 μM (26) |
| A549 xenograft | Oral | 25, 50 and 75
mmol/kg (26) |
| Colon | Caco-2 cell
culture | | 150 and 400 μM
(22,27,28,43) |
| SW620 cell
culture | | 150 μM (28) |
| TC118
xenograft | I.P.a | 150 mg/kg (28) |
|
Dimethylhydrazine-treated Wistar rat | I.P. | 250 mg/kg (49) |
| Prostate | PC-3 cell
culture | | 250, 500 and 1,000
μM (25,30) |
| PC-3 xenograft | I.T.a | 60 mg/kg (25) |
| Pancreatic | MIA PaCa-2 cell
culture | | 100, 200, 400 and
500 μM (21,48) |
| BxPC-3 cell
culture | | 10, 20, 40, 80,
160, 400 and 800 μM (48,60) |
| PC-1 xenograft | Oral | 20 g/kg (21) |
| Skin |
12-O-tetradecanoylphorbol | Dermal | 10, 50 and 100
mg/kg (45,52) |
| 13-acetate-treated
mice | | |
| Liver | HepG2 cell
culture | | 100, 200 and 400 μM
(29,31,61) |
| Morris hepatoma
7777 xenograft | Oral | 23 mmol/kg
(62) |
|
2-acetylaminofluorene-treated Wistar
rat | Oral | 250 mg/kg (24,47) |
| Kidney | Ferric
nitrilotriacetate-treated Wistar rat | Oral | 100 and 200 mg/kg
(50) |
| Oral |
7,12-dimethylbenz[α]anthracene-treated
hamster | Oral | 250 and 500 mg/kg
(46,51) |
|
4-niroquinoline-1-oxide-treated mice | Oral | 200 mg/kg (53) |
| Other cancers | B16 melanoma cell
culture | | 150 and 160 μM
(64,66) |
| B16 xenograft | Oral | 6.5 and 65 mmol/kg
(62) |
| P388 leukemia cell
culture | | 900 μM (63) |
Breast cancer
Geraniol suppressed the growth of MCF-7 breast
cancer cells via the induction of cell cycle arrest in the G1 phase
(23). The protein levels of
cyclin D1, cyclin-dependent kinase 4 (CDK4), cyclin E, and cyclin A
were reduced in geraniol-treated MCF-7 cells, whereas those of
p27Kip1 were elevated by geraniol (23). In contrast, geraniol did not affect
the growth of MCF-10F normal breast epithelial cells (23), which indicates that its activity is
tumor-specific. Although geraniol inhibited
3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase activity,
geraniol-induced cell cycle arrest was not rescued by
supplementation with mevalonate, which is a reaction product of
HMG-CoA reductase (23). These
results indicate that the anti-proliferative effect of geraniol on
MCF-7 cells is not related to a reduction in HMG-CoA reductase
activity or the limitation of cellular mevalonate levels, but
rather, that it is linked to other molecular mechanisms.
Lung cancer
Experiments in xenograft models revealed that
geraniol reduced tumor weight and volume in mice bearing tumors
that formed from A549 cells (26),
which showed an increased TUNEL-positive tumor cell population and
elevated caspase-3 activity. Geraniol also inhibited cholesterol
synthesis in A549 cells and reduced the serum levels of cholesterol
in mice with A549 cell xenografts through the suppression of
HMG-CoA reductase activity (26).
In addition, geraniol reduced the expression levels of
membrane-bound Ras in A549 cell-bearing mice without corresponding
changes in the total protein levels of Ras (26). This was probably due to the actions
of geraniol that are related to the inhibition of mevalonate
pathways and the resultant reduction in Ras prenylation (55). However, it is unclear whether
mevalonate supplementation can rescue geraniol-induced
phenotypes.
Colon cancer
Geraniol exerted anti-proliferative activity against
Caco-2 colorectal adenocarcinoma cells through the induction of S
phase cell cycle arrest (22). In
addition, geraniol elicited apoptosis in mouse xenograft models
using CT26 colon carcinoma cells (54). Geraniol affected polyamine
metabolism in Caco-2 cells through the reduction of ornithine
decarboxylase (ODC) activity (22). This is clinically relevant because
polyamine metabolism is a promising target of chemotherapeutic and
chemopreventive drug development strategies (56). Geraniol also induced the
depolarization of the plasma membrane potential with a decrease in
membrane resistance in Caco-2 cells (43), which led to the reduction in the
activities of protein kinase C (PKC) and extracellular
signal-regulated protein kinases (ERK) (43). Under the experimental conditions,
mevalonate supplementation did not reverse geraniol-mediated
inhibition of cell growth (43),
which indicates that HMG-CoA reductase is not linked to the
antitumor actions of geraniol.
Geraniol sensitized cultured Caco-2 cells and SW620
colorectal adenocarcinoma cells to 5-FU and also sensitized mice
with xenografts of TC118 colon cancer cells to 5-FU (27,28).
In addition, geraniol elevated the uptake rate of 5-FU in colon
cancer cells (27). Geraniol
effectively decreased the expression levels of thymidine kinase and
thymidylate synthase (28), which
are important targets for the development of anticancer drugs and
chemosensitizing agents (57–59).
These results suggest that geraniol might serve as a
chemosensitizer.
Geraniol was found to elicit chemopreventive
activities (49). It decreased the
total number of aberrant crypt foci in the colons of Wistar rats
that were treated with dimethylhydrazine (49). Under this condition, geraniol
reduced DNA damage in the colonic mucosa. In addition, apoptosis in
the distal colon was significantly higher in the geraniol-treated
group compared with the control group. Moreover, the expression
levels of Bcl-2 were significantly decreased in the
geraniol-treated group (49).
Prostate cancer
Geraniol inhibited tumor cell growth via the
induction of apoptosis in cultured and grafted PC-3 prostate cancer
cells (25,30) due to the depolarization of
mitochondrial membrane potential, the activation of caspase-3, the
reduction in the expression levels of Bcl-2 and Bcl-w, and the
increased expression levels of Bax and BNIP3 (25). Geraniol also potently induced
autophagy, which is crucial for the effective stimulation of cell
death (30). The combined
inhibition of apoptosis and autophagy restored cell growth in
geraniol-treated PC-3 cells. In addition, cell cycle arrest at G1
phase, and a slight arrest at G2 phase, were observed in
geraniol-treated PC-3 cells (25,30),
which was confirmed by the decreased expression levels of cyclins
A, B, D and E and CDK1 and CDK4, as well as by the elevated levels
of p21Cip1 and p27Kip1.
At the molecular level, geraniol has been found to
inhibit AKT-mTOR signaling without affecting mitogen-activated
protein kinase (MAPK) activity (30). For example, the PP2A inhibitor
okadaic acid, but not the PP1 inhibitor tautomycin, reversed
geraniol-mediated inhibition of AKT. Concomitantly, geraniol
activated AMP-activated protein kinase (AMPK). The combined
treatment with the AKT inhibitor perifosine and the AMPK activator
AICAR synergistically inhibited tumor cell growth (30). However, under the condition,
geraniol did not affect the activity of transient receptor
potential melastatin type 8 (TRPM8) channel or calcium mobilization
(25,30). In addition, mevalonate
supplementation did not reverse the geraniol-mediated inhibition of
cell growth (30).
Geraniol increased the therapeutic response of PC-3
cells to chemotherapy agents, such as docetaxel, doxorubicin,
paclitaxel, etoposide, 5-FU and cisplatin (25). Particularly, the combination of the
sub-optimal doses of geraniol and docetaxel noticeably suppressed
the growth of prostate cancer cells in culture and those in mouse
xenografts; the percentage of active caspase-3-positive cells was
markedly increased in tumor tissues that were treated with geraniol
and docetaxel compared with the treatment of cells with either drug
alone.
Pancreatic cancer
Geraniol inhibited the proliferation of MIA PaCa-2
pancreatic carcinoma cells and BxPC-3 pancreatic adenocarcinoma
cells and reduced tumor volume in hamsters injected with PC-1
pancreatic ductal adenocarcinoma cells (21,48,60).
In addition, geraniol induced G1 phase cell cycle arrest in MIA
PaCa-2 cells (48) and elevated
the expression levels of p21Cip1 and p27Kip1
but reduced those of cyclin A, cyclin B1 and CDK2. siRNAs against
p21Cip1 and p27Kip1 subsequently alleviated
geraniol-induced cell cycle arrest (48). Finally, the protein levels of
cyclin D1 were reduced by geraniol in MIA PaCa-2 cells, but not in
BxPC-3 cells (48).
When the tumor cells were incubated with geraniol
before they were treated with gemcitabine, which is one of the
first-line treatments for advanced pancreatic cancer, proliferation
of BxPC-3 cells was significantly inhibited compared with when the
cells were treated with either drug alone (60). In addition, geraniol markedly
enhanced the apoptosis-inducing effect of gemcitabine on BxPC-3
cells (60).
Skin cancer
Geraniol inhibited skin edema and epidermal
hyperplasia in mice that were stimulated with
12-O-tetradecanolyphorbal-13-acetate (TPA)/7,
12-dimethyl-benzanthracene (DMBA) (45,52).
The chemopreventive effects of geraniol on skin tumorigenesis are
ascribed to the activation of apoptotic pathways and the
attenuation of Ras-mediated cell proliferation pathways.
Particularly, geraniol reduced the levels of membrane-bound Ras in
DMBA/TPA-stimulated mice (45) and
also decreased Raf expression and ERK activity. However, it is
unclear whether geraniol inhibits HMG-CoA reductase activity in
this setting and whether mevalonate rescues the geraniol-induced
phenotypic changes. In addition, geraniol suppressed the
TPA-induced increase in cutaneous ODC activity and the thymidine
incorporation rate in mouse skin (45).
Geraniol suppressed skin inflammation through the
inhibition of cyclooxygenase (COX)-2 induction in DMBA/TPA-treated
mice (45). Geraniol also
inhibited the altered expression of nuclear factor (NF)-κB; the
production of cutaneous proinflammatory cytokines, such as tumor
necrosis factor (TNF)-α, interleukin (IL)-6, IL-1β and pp38; and
the formation of superoxide dismutase (SOD) and nitric oxide (NO)
(52). In addition, geraniol
exerted antioxidant activity to reduce TPA-induced malondialdehyde
(MDA) production through modulation of the following: glutathione
(GSH), glutathione reductase (GR), glutathione peroxidase (GPX),
glutathione-S-transferase (GST), catalase (CAT), lipid
peroxidation, glucose 6-phosphate dehydrogenase (G6PD) and quinone
reductase (QR) (45,52).
Liver cancer
In one study, geraniol exerted anti-proliferative
activity in HepG2 hepatocarcinoma cells (29,31,61),
and G1 phase cell cycle arrest and apoptosis were observed in
geraniol-treated cells (31). In
addition, geraniol suppressed tumor growth in mice that received
transplanted Morris 7777 hepatoma cells (62). Geraniol inhibited HMG-CoA reductase
activity in HepG2 cells and 2-acetylaminofluorene-treated Wistar
rat (29,47) and reduced the level of RhoA in
hepatic membranes (47). Although
geraniol affected various lipid metabolic pathways, including
HMG-CoA reductase or Δ5-desaturase (29,31),
it is uncertain whether its effects are linked to tumor
suppression.
Geraniol exhibited chemopreventive activity against
hepatocarcinogenesis in rats that were stimulated with
diethylnitrosamine and 2-acetylaminofluorene. It also inhibited
cell proliferation and DNA damage in preneoplastic lesions (PNLs)
and caused an elevation in apoptosis in PNLs (24,47).
Kidney cancer
Geraniol demonstrated chemopreventive activity in
Wistar rats treated with ferric nitrilotriacetate (Fe-NTA)
(50) in that it effectively
reduced renal oxidative stress and tumor incidence via the
modulation of multiple molecular targets that are involved in
proliferation, apoptosis, or inflammation. Geraniol restored the
activities of GSH-dependent antioxidant enzymes, such as
glutathione S-transferase, glutathione peroxidase, and catalase in
Fe-NTA-injected rats, which suppressed Fe-NTA-induced renal
toxicity (50). In the rats that
were treated with Fe-NTA and N-nitrosodiethylamine, geraniol
downregulated kidney injury molecule-1 (Kim-1), NF-κB,
proliferating cell nuclear antigen (PCNA), and p53 and upregulated
caspase-3, -8 and -9 (50).
Oral squamous cell carcinoma
Geraniol prevented DMBA-induced buccal pouch
carcinogenesis in the hamster (46,51).
Its anti-inflammatory, anti-angiogenic, anti-proliferative, and
apoptosis-inducing properties contributed to the suppression of
histological changes, such as keratosis, dysplasia and hyperplasia,
in the buccal pouch of DMBA-treated hamsters. Geraniol also exerted
its chemopreventive potential against 4-nitroquinoline-1-oxide
(4NQO)-induced oral carcinogenesis in Wistar rats (53).
Geraniol reduced the plasma concentrations of lipid
peroxidation by-products in DMBA-stimulated hamsters (51). Geraniol also restored the
DMBA-induced disturbance in the status of antioxidants in terms of
glutathione peroxidase, reduced glutathione, vitamin E, superoxide
dismutase, and catalase in the plasma and in the buccal mucosa
(51). In addition, the
chemopreventive potential of geraniol relies on modulatory effects
on phase I (cytochrome P450 and cytochrome b5) and II
(glutathione-S-transferase, glutathione reductase and
DT-diaphorase) detoxification enzymes to excrete carcinogenic
metabolites during DMBA-induced buccal pouch carcinogenesis in the
hamster (51). Similar effects of
geraniol were observed in 4NQO-stimulated models of oral squamous
cell carcinoma (53).
Geraniol reduced the expression levels of Bcl-2,
PCNA, mutant p53, VEGF, c-fos, COX-2, NF-κB, and cyclin D1, whereas
it elevated Bax, caspase-3 and -9 activity in DMBA-treated hamsters
(46). In addition, geraniol
increased the nuclear levels of nuclear factor erythroid 2-related
factor 2 (Nrf-2), a redox-sensitive transcription factor, in
4NQO-stimulated rats (53).
Other cancers
Geraniol inhibited cell growth in P388 leukemia
cells (63). Geraniol exerted
anti-proliferative activity against B16 melanoma cells in culture
and in xenograft models (63–66).
Mevalonate supplementation rescued geraniol-induced phenotypes of
P388 leukemia cells, but not those of B16 melanoma cells (63). These results suggest that
limitations in mevalonate levels suppress tumor growth in a cancer
type-specific manner.
4. Discussion and future perspective
Geraniol has been approved as GRAS by FDA and FEMA.
The acute oral LD50 was calculated as 3.6 to 4.8 g/kg in
rats and the intramuscular LD50 was reported to be 4
g/kg. In addition, dose toxicity was only slightly observed in
repeated administration of geraniol at 1,000 mg/kg/day in the diet
for 16 weeks (see the ref. 16 for
the detailed toxicological data of geraniol). The recent
experimental observations obtained from in vitro and in
vivo models have supported the idea that geraniol exerts
therapeutic, chemosensitizing, or preventive effects against
different cancer types by modulating multiple cancer hallmark
pathways (Tables I and II). Particularly, the antitumor effects
of geraniol were achieved by oral delivery in different cancer
types (Table III), which is
unlike poor oral bioavailability of many phytochemicals (67). These results suggest that geraniol
might be beneficial for achieving a balance between therapeutic
outcome and adverse drug responses and useful for improvement in
quality of life of patients and palliative treatment. Therefore,
geraniol might be promising for further development as a lead
compound or a chemical moiety.
The non-toxic properties of geraniol (16) and the experimental results
summarized in this review have demonstrated that geraniol
specifically suppresses tumor growth, but does not affect normal
physiology at an individual level. Geraniol commonly affects the
signaling molecules that are involved in the acquisition of cancer
hallmarks, particularly those that involve the cell cycle and
apoptosis, across various types of cancer, as summarized in
Tables I and II. These findings suggest that geraniol
elicits tumor-specific effects through common molecular mechanisms.
However, several geraniol-reactive signaling pathways are also
important physiological cell functions. For example, geraniol has
been shown to inhibit angiogenesis, which does not only occur
during tumor growth but also during embryo development, wound
healing and other physiological conditions (68). Therefore, the cancer type-specific
actions and the adverse effects of geraniol need to be carefully
evaluated.
HMG-CoA reductase catalyzes the synthesis of
mevalonate, which is the committed step in the synthesis of prenyl
groups (55) and is the limiting
step in cholesterol biosynthesis (69). The expression levels of HMG-CoA
reductase are frequently upregulated in various types of cancer in
humans (23,26,29,31,47).
It has been shown that geraniol inhibits HMG-CoA reductase in most
types of tumor cells, which raises the possibility that the reduced
prenylation of small G-proteins, such as Ras or RhoA, accounts for
the antitumor effects of geraniol. However, mevalonate
supplementation did not reverse geraniol-induced growth suppression
in any type of cancer tested (23,26)
except leukemia (63). These
results suggest that geraniol-induced molecular changes do not
necessarily induce phenotypic outcomes and that the inhibition of
HMG-CoA reductase might be effective against specific types of
cancer, including leukemia.
The molecular mechanisms by which geraniol exerts
its anticancer effects are still largely unknown. Therefore, much
effort is required to obtain additional information on the
effectiveness and efficacy of geraniol. In this regard,
computational approaches using omics technology-driven data should
be emphasized to broaden our understanding of the pharmacological
mechanisms of action of geraniol and for the translation of this
biological knowledge into clinical practice (70–76).
Data-driven approaches allow us novel insights into
geraniol-induced changes in terms of the molecular makeup of
biological systems, which contributes to the efficient discovery of
the latent target molecules of geraniol.
There seem to be no doubt that geraniol inhibits
cancer growth in vitro and in vivo models. However,
in vitro models, submillimolar concentrations of geraniol
have been used to define the therapeutic effects and the mechanism
of action (Table III). This may
be a weakness of geraniol studies because these concentrations are
not clinically relevant. Nonetheless, geraniol effectively and
efficiently suppresses tumor growth in in vivo models. These
findings are similar to those of metformin, an anti-diabetic drug.
Although milli-molar concentrations of metformin usually exerts
anticancer activity in in vitro models, there is consensus
that metformin is a clinically promising cancer therapeutic agent
(77,78). Therefore, it is crucial that the
mechanisms of action of geraniol are elucidated in in vivo
studies.
The present review has highlighted the pleiotropic
mechanisms of action of geraniol in various types of cancer.
Geraniol is therefore useful in the development of multi-target
therapies against cancer, and our review provides a scientific
basis for future basic and translational research.
Acknowledgements
We are very grateful to Ms. Da-Kyung Lee, Ms. Young
Hyun Yun, and Prof. Jeong Han Kim (Bio-Medical Arts and Design
Education Center, Department of Contemporary Art and Ceramics and
Fiber Arts, College of Fine Art, Seoul Women's University) for
preparation of the figures. This research was supported by the
National Research Foundation of Korea (NRF) funded by the Ministry
of Science, ICT and Future Planning (2014R1A2A1A11050616) and the
Education and Research Encouragement Fund of Seoul National
University Hospital.
References
|
1
|
Mahmoud SS and Croteau RB: Strategies for
transgenic manipulation of monoterpene biosynthesis in plants.
Trends Plant Sci. 7:366–373. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Guimarães AG, Quintans JS and Quintans LJ
Jr: Monoterpenes with analgesic activity: a systematic review.
Phytother Res. 27:1–15. 2013. View
Article : Google Scholar
|
|
3
|
de Cássia da Silveira e Sá R, Andrade LN
and de Sousa DP: A review on anti-inflammatory activity of
monoterpenes. Molecules. 18:1227–1254. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bhalla Y, Gupta VK and Jaitak V:
Anticancer activity of essential oils: A review. J Sci Food Agric.
93:3643–3653. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Barreto RS, Albuquerque-Júnior RL, Araújo
AA, Almeida JR, Santos MR, Barreto AS, DeSantana JM, Siqueira-Lima
PS, Quintans JS and Quintans-Júnior LJ: A systematic review of the
wound-healing effects of monoterpenes and iridoid derivatives.
Molecules. 19:846–862. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hasan SK and Sultana S: Geraniol
attenuates 2-acetylamino-fluorene induced oxidative stress,
inflammation and apoptosis in the liver of wistar rats. Toxicol
Mech Methods. 25:559–573. 2015.
|
|
7
|
Medicherla K, Sahu BD, Kuncha M, Kumar JM,
Sudhakar G and Sistla R: Oral administration of geraniol
ameliorates acute experimental murine colitis by inhibiting
pro-inflammatory cytokines and NF-κB signaling. Food Funct.
6:2984–2995. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Soubh AA, Abdallah DM and El-Abhar HS:
Geraniol ameliorates TNBS-induced colitis: Involvement of
Wnt/β-catenin, p38MAPK, NF-κB, and PPARγ signaling pathways. Life
Sci. 136:142–150. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kozioł A, Stryjewska A, Librowski T, Sałat
K, Gaweł M, Moniczewski A and Lochyński S: An overview of the
pharmacological properties and potential applications of natural
monoterpenes. Mini Rev Med Chem. 14:1156–1168. 2014. View Article : Google Scholar
|
|
10
|
Gould MN: Cancer chemoprevention and
therapy by monoterpenes. Environ Health Perspect. 105(Suppl 4):
977–979. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Crowell PL: Prevention and therapy of
cancer by dietary monoterpenes. J Nutr. 129:775S–778S.
1999.PubMed/NCBI
|
|
12
|
Chen TC, Fonseca CO and Schönthal AH:
Preclinical development and clinical use of perillyl alcohol for
chemoprevention and cancer therapy. Am J Cancer Res. 5:1580–1593.
2015.PubMed/NCBI
|
|
13
|
Samaila D, Toy BJ, Wang RC and Elegbede
JA: Monoterpenes enhanced the sensitivity of head and neck cancer
cells to radiation treatment in vitro. Anticancer Res.
24:3089–3095. 2004.PubMed/NCBI
|
|
14
|
Bardon S, Foussard V, Fournel S and Loubat
A: Monoterpenes inhibit proliferation of human colon cancer cells
by modulating cell cycle-related protein expression. Cancer Lett.
181:187–194. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sobral MV, Xavier AL, Lima TC and de Sousa
DP: Antitumor activity of monoterpenes found in essential oils. Sci
World J. 2014:9534512014. View Article : Google Scholar
|
|
16
|
Lapczynski A, Bhatia SP, Foxenberg RJ,
Letizia CS and Api AM: Fragrance material review on geraniol. Food
Chem Toxicol. 46(Suppl 11): S160–S170. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Solórzano-Santos F and Miranda-Novales MG:
Essential oils from aromatic herbs as antimicrobial agents. Curr
Opin Biotechnol. 23:136–141. 2012. View Article : Google Scholar
|
|
18
|
Tiwari M and Kakkar P: Plant derived
antioxidants - Geraniol and camphene protect rat alveolar
macrophages against t-BHP induced oxidative stress. Toxicol In
Vitro. 23:295–301. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
de Carvalho KI, Bonamin F, Dos Santos RC,
Périco LL, Beserra FP, de Sousa DP, Filho JM, da Rocha LR and
Hiruma-Lima CA: Geraniol - a flavoring agent with multifunctional
effects in protecting the gastric and duodenal mucosa. Naunyn
Schmiedebergs Arch Pharmacol. 387:355–365. 2014. View Article : Google Scholar
|
|
20
|
Rekha KR, Selvakumar GP, Sethupathy S,
Santha K and Sivakamasundari RI: Geraniol ameliorates the motor
behavior and neurotrophic factors inadequacy in MPTP-induced mice
model of Parkinson's disease. J Mol Neurosci. 51:851–862. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Burke YD, Stark MJ, Roach SL, Sen SE and
Crowell PL: Inhibition of pancreatic cancer growth by the dietary
isoprenoids farnesol and geraniol. Lipids. 32:151–156. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Carnesecchi S, Schneider Y, Ceraline J,
Duranton B, Gosse F, Seiler N and Raul F: Geraniol, a component of
plant essential oils, inhibits growth and polyamine biosynthesis in
human colon cancer cells. J Pharmacol Exp Ther. 298:197–200.
2001.PubMed/NCBI
|
|
23
|
Duncan RE, Lau D, El-Sohemy A and Archer
MC: Geraniol and beta-ionone inhibit proliferation, cell cycle
progression, and cyclin-dependent kinase 2 activity in MCF-7 breast
cancer cells independent of effects on HMG-CoA reductase activity.
Biochem Pharmacol. 68:1739–1747. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ong TP, Heidor R, de Conti A, Dagli ML and
Moreno FS: Farnesol and geraniol chemopreventive activities during
the initial phases of hepatocarcinogenesis involve similar actions
on cell proliferation and DNA damage, but distinct actions on
apoptosis, plasma cholesterol and HMGCoA reductase. Carcinogenesis.
27:1194–1203. 2006. View Article : Google Scholar
|
|
25
|
Kim SH, Bae HC, Park EJ, Lee CR, Kim BJ,
Lee S, Park HH, Kim SJ, So I, Kim TW, et al: Geraniol inhibits
prostate cancer growth by targeting cell cycle and apoptosis
pathways. Biochem Biophys Res Commun. 407:129–134. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Galle M, Crespo R, Kladniew BR, Villegas
SM, Polo M and de Bravo MG: Suppression by geraniol of the growth
of A549 human lung adenocarcinoma cells and inhibition of the
mevalonate pathway in culture and in vivo: Potential use in cancer
chemotherapy. Nutr Cancer. 66:888–895. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Carnesecchi S, Langley K, Exinger F, Gosse
F and Raul F: Geraniol, a component of plant essential oils,
sensitizes human colonic cancer cells to 5-Fluorouracil treatment.
J Pharmacol Exp Ther. 301:625–630. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Carnesecchi S, Bras-Gonçalves R, Bradaia
A, Zeisel M, Gossé F, Poupon MF and Raul F: Geraniol, a component
of plant essential oils, modulates DNA synthesis and potentiates
5-fluorouracil efficacy on human colon tumor xenografts. Cancer
Lett. 215:53–59. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Polo MP and de Bravo MG: Effect of
geraniol on fatty-acid and mevalonate metabolism in the human
hepatoma cell line Hep G2. Biochem Cell Biol. 84:102–111. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim SH, Park EJ, Lee CR, Chun JN, Cho NH,
Kim IG, Lee S, Kim TW, Park HH, So I, et al: Geraniol induces
cooperative interaction of apoptosis and autophagy to elicit cell
death in PC-3 prostate cancer cells. Int J Oncol. 40:1683–1690.
2012.
|
|
31
|
Crespo R, Montero Villegas S, Abba MC, de
Bravo MG and Polo MP: Transcriptional and posttranscriptional
inhibition of HMGCR and PC biosynthesis by geraniol in 2 Hep-G2
cell proliferation linked pathways. Biochem Cell Biol. 91:131–139.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zimmermann GR, Lehár J and Keith CT:
Multi-target therapeutics: When the whole is greater than the sum
of the parts. Drug Discov Today. 12:34–42. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Efferth T and Koch E: Complex interactions
between phytochemicals. The multi-target therapeutic concept of
phytotherapy. Curr Drug Targets. 12:122–132. 2011. View Article : Google Scholar
|
|
34
|
Rather MA, Bhat BA and Qurishi MA:
Multicomponent phyto-therapeutic approach gaining momentum: Is the
‘one drug to fit all’ model breaking down? Phytomedicine. 21:1–14.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tao L, Zhu F, Xu F, Chen Z, Jiang YY and
Chen YZ: Co-targeting cancer drug escape pathways confers clinical
advantage for multi-target anticancer drugs. Pharmacol Res.
102:123–131. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Huang S and Kauffman S: How to escape the
cancer attractor: Rationale and limitations of multi-target drugs.
Semin Cancer Biol. 23:270–278. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hanahan D and Weinberg RA: The hallmarks
of cancer. Cell. 100:57–70. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Floor SL, Dumont JE, Maenhaut C and Raspe
E: Hallmarks of cancer: Of all cancer cells, all the time? Trends
Mol Med. 18:509–515. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ellenbroek SI and van Rheenen J: Imaging
hallmarks of cancer in living mice. Nat Rev Cancer. 14:406–418.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Horne SD, Pollick SA and Heng HH:
Evolutionary mechanism unifies the hallmarks of cancer. Int J
Cancer. 136:2012–2021. 2015. View Article : Google Scholar
|
|
42
|
Sonnenschein C and Soto AM: The aging of
the 2000 and 2011 Hallmarks of Cancer reviews: A critique. J
Biosci. 38:651–663. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Carnesecchi S, Bradaia A, Fischer B,
Coelho D, Schöller-Guinard M, Gosse F and Raul F: Perturbation by
geraniol of cell membrane permeability and signal transduction
pathways in human colon cancer cells. J Pharmacol Exp Ther.
303:711–715. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hoshi D, Nakajima A, Inoue E, Shidara K,
Sato E, Kitahama M, Seto Y, Tanaka E, Urano W, Ichikawa N, et al:
Incidence of serious respiratory infections in patients with
rheumatoid arthritis treated with tocilizumab. Mod Rheumatol.
22:122–127. 2012. View Article : Google Scholar
|
|
45
|
Chaudhary SC, Siddiqui MS, Athar M and
Alam MS: Geraniol inhibits murine skin tumorigenesis by modulating
COX-2 expression, Ras-ERK1/2 signaling pathway and apoptosis. J
Appl Toxicol. 33:828–837. 2013. View Article : Google Scholar
|
|
46
|
Vinothkumar V, Manoharan S, Sindhu G,
Nirmal MR and Vetrichelvi V: Geraniol modulates cell proliferation,
apoptosis, inflammation, and angiogenesis during
7,12-dimethylbenz[a] anthracene-induced hamster buccal pouch
carcinogenesis. Mol Cell Biochem. 369:17–25. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Cardozo MT, de Conti A, Ong TP, Scolastici
C, Purgatto E, Horst MA, Bassoli BK and Moreno FS: Chemopreventive
effects of β-ionone and geraniol during rat hepatocarcinogenesis
promotion: Distinct actions on cell proliferation, apoptosis,
HMGCoA reductase, and RhoA. J Nutr Biochem. 22:130–135. 2011.
View Article : Google Scholar
|
|
48
|
Wiseman DA, Werner SR and Crowell PL: Cell
cycle arrest by the isoprenoids perillyl alcohol, geraniol, and
farnesol is mediated by p21Cip1 and p27Kip1
in human pancreatic adenocarcinoma cells. J Pharmacol Exp Ther.
320:1163–1170. 2007. View Article : Google Scholar
|
|
49
|
Vieira A, Heidor R, Cardozo MT, Scolastici
C, Purgatto E, Shiga TM, Barbisan LF, Ong TP and Moreno FS:
Efficacy of geraniol but not of β-ionone or their combination for
the chemoprevention of rat colon carcinogenesis. Braz J Med Biol
Res. 44:538–545. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ahmad ST, Arjumand W, Seth A, Nafees S,
Rashid S, Ali N and Sultana S: Preclinical renal cancer
chemopreventive efficacy of geraniol by modulation of multiple
molecular pathways. Toxicology. 290:69–81. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Vinothkumar V and Manoharan S:
Chemopreventive efficacy of geraniol against
7,12-dimethylbenz[a]anthracene-induced hamster buccal pouch
carcinogenesis. Redox Rep. 16:91–100. 2011. View Article : Google Scholar
|
|
52
|
Khan AQ, Khan R, Qamar W, Lateef A, Rehman
MU, Tahir M, Ali F, Hamiza OO, Hasan SK and Sultana S: Geraniol
attenuates 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced
oxidative stress and inflammation in mouse skin: Possible role of
p38 MAP Kinase and NF-κB. Exp Mol Pathol. 94:419–429. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Madankumar A, Jayakumar S, Gokuladhas K,
Rajan B, Raghunandhakumar S, Asokkumar S and Devaki T: Geraniol
modulates tongue and hepatic phase I and phase II conjugation
activities and may contribute directly to the chemopreventive
activity against experimental oral carcinogenesis. Eur J Pharmacol.
705:148–155. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wittig C, Scheuer C, Parakenings J, Menger
MD and Laschke MW: Geraniol suppresses angiogenesis by
down-regulating vascular endothelial growth factor (VEGF)/VEGFR-2
signaling. PLoS One. 10:e01319462015. View Article : Google Scholar
|
|
55
|
Gysin S, Salt M, Young A and McCormick F:
Therapeutic strategies for targeting ras proteins. Genes Cancer.
2:359–372. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wallace HM: Targeting polyamine
metabolism: A viable therapeutic/preventative solution for cancer?
Expert Opin Pharmacother. 8:2109–2116. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Peters GJ, Backus HH, Freemantle S, van
Triest B, Codacci-Pisanelli G, van der Wilt CL, Smid K, Lunec J,
Calvert AH, Marsh S, et al: Induction of thymidylate synthase as a
5-fluorouracil resistance mechanism. Biochim Biophys Acta.
1587:194–205. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Popat S, Matakidou A and Houlston RS:
Thymidylate synthase expression and prognosis in colorectal cancer:
A systematic review and meta-analysis. J Clin Oncol. 22:529–536.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Topolcan O and Holubec L Jr: The role of
thymidine kinase in cancer diseases. Expert Opin Med Diagn.
2:129–141. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Jin X, Sun J, Miao X, Liu G and Zhong D:
Inhibitory effect of geraniol in combination with gemcitabine on
proliferation of BXPC-3 human pancreatic cancer cells. J Int Med
Res. 41:993–1001. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Polo MP, Crespo R and de Bravo MG:
Geraniol and simvastatin show a synergistic effect on a human
hepatocarcinoma cell line. Cell Biochem Funct. 29:452–458. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Yu SG, Hildebrandt LA and Elson CE:
Geraniol, an inhibitor of mevalonate biosynthesis, suppresses the
growth of hepatomas and melanomas transplanted to rats and mice. J
Nutr. 125:2763–2767. 1995.PubMed/NCBI
|
|
63
|
Shoff SM, Grummer M, Yatvin MB and Elson
CE: Concentration-dependent increase of murine P388 and B16
population doubling time by the acyclic monoterpene geraniol.
Cancer Res. 51:37–42. 1991.PubMed/NCBI
|
|
64
|
He L, Mo H, Hadisusilo S, Qureshi AA and
Elson CE: Isoprenoids suppress the growth of murine B16 melanomas
in vitro and in vivo. J Nutr. 127:668–674. 1997.PubMed/NCBI
|
|
65
|
Mo H, Tatman D, Jung M and Elson CE:
Farnesyl anthranilate suppresses the growth, in vitro and in vivo,
of murine B16 melanomas. Cancer Lett. 157:145–153. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
McAnally JA, Jung M and Mo H:
Farnesyl-O-acetylhydroquinone and geranyl-O-acetylhydroquinone
suppress the proliferation of murine B16 melanoma cells, human
prostate and colon adenocarcinoma cells, human lung carcinoma
cells, and human leukemia cells. Cancer Lett. 202:181–192. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Aqil F, Munagala R, Jeyabalan J and
Vadhanam MV: Bioavailability of phytochemicals and its enhancement
by drug delivery systems. Cancer Lett. 334:133–141. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Chung AS and Ferrara N: Developmental and
pathological angiogenesis. Annu Rev Cell Dev Biol. 27:563–584.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
DeBose-Boyd RA: Feedback regulation of
cholesterol synthesis: Sterol-accelerated ubiquitination and
degradation of HMG CoA reductase. Cell Res. 18:609–621. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Chun JN, Lim JM, Kang Y, Kim EH, Shin YC,
Kim HG, Jang D, Kwon D, Shin SY, So I, et al: A network perspective
on unraveling the role of TRP channels in biology and disease.
Pflugers Arch. 466:173–182. 2014. View Article : Google Scholar
|
|
71
|
Barabási AL, Gulbahce N and Loscalzo J:
Network medicine: A network-based approach to human disease. Nat
Rev Genet. 12:56–68. 2011. View Article : Google Scholar :
|
|
72
|
Furlong LI: Human diseases through the
lens of network biology. Trends Genet. 29:150–159. 2013. View Article : Google Scholar
|
|
73
|
Ideker T and Krogan NJ: Differential
network biology. Mol Syst Biol. 8:5652012. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Grossglauser M and Saner H: Data-driven
healthcare: From patterns to actions. Eur J Prev Cardiol.
21(Suppl): 14–17. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Lusher SJ, McGuire R, van Schaik RC,
Nicholson CD and de Vlieg J: Data-driven medicinal chemistry in the
era of big data. Drug Discov Today. 19:859–868. 2014. View Article : Google Scholar
|
|
76
|
Quan Y, Wang ZY, Xiong M, Xiao ZT and
Zhang HY: Dissecting traditional Chinese medicines by omics and
bioinformatics. Nat Prod Commun. 9:1391–1396. 2014.
|
|
77
|
Quinn BJ, Kitagawa H, Memmott RM, Gills JJ
and Dennis PA: Repositioning metformin for cancer prevention and
treatment. Trends Endocrinol Metab. 24:469–480. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Anisimov VN: Do metformin a real
anticarcinogen? A critical reappraisal of experimental data. Ann
Transl Med. 2:602014.PubMed/NCBI
|