Phosphatidylinositol 3-kinase (PI3K)/AKT pathway is
been known as an important signaling pathway in a variety of human
cancers (1–3), which also plays critical roles in
normal cell survival and proliferation under physiological
condition (4,5). For example, prostate cancer
development is often associated with silencing of the tumor
suppressor phosphatase and tensin homolog (PTEN), a negative
regulator of the PI3K/AKT signaling pathway (6,7). The
PTEN is a multi-functional enzyme inhibiting the PI3K/AKT signaling
in the cytosol and stabilizing genomes in the nucleus. Various
extracellular signals including growth factors and/or cell
nutrients could act as modulators for the PI3K/AKT/PTEN signaling
axis. Once activated PI3K recruits cellular protein kinases such as
the serine/threonine kinase AKT that consecutively transduces a
signal to several downstream molecules. In general, relationship of
the intracellular signaling networks coordinates cellular function
containing cell cycle progression, proliferation and cell
protection. Mutation in some of the effectors may result in
consequent activation and/or inhibition of the signaling. This
dysregulation of the signal occurs commonly in many malignancies,
thereby rendering the cancer cells proliferative and with survival
advantage. Accordingly, both nuclear and cytosolic PTEN/PI3K/AKT
activity should be reflected for correlation with
clinicopathological parameters of cancer (8). In addition, the PI3K/AKT signaling
has been shown to induce the gene expression of multidrug
resistance protein that is considerably associated with cancer
therapy-opposition (9).
Development of this multidrug resistance seems the major obstacle
for chemotherapy of cancers. Therefore, it seems a remarkable
target for molecular therapy in various diseases including cancer
and related diseases. To date, many pharmacological inhibitors
acting on the PI3K/AKT pathway have been established (10).
Overall, cancer treatment could have secondary
negative effects such as neurocognitive deficits, serious sequelae
that follow the therapy used to treat patients especially with
brain neoplasms (11,12). The pathogenesis of the
neurocognitive deficits involves apoptosis of neurons and other
neuronal cells in a region in brain for learning and memory. After
and during the intensive cancer therapy, brain damage has emerged
as an important clinical problem (13,14).
In regards to this, some reports show that nuclear trafficking of
PTEN following brain injury leads to neuronal survival (15), indicating hope for new treatment
options. The present review focuses on the recent advance in
PI3K/AKT/PTEN-mediated neuroprotection and neurogenesis after
several cancer therapies, highlighting its potential molecular and
cellular mechanisms on the therapeutic advantages.
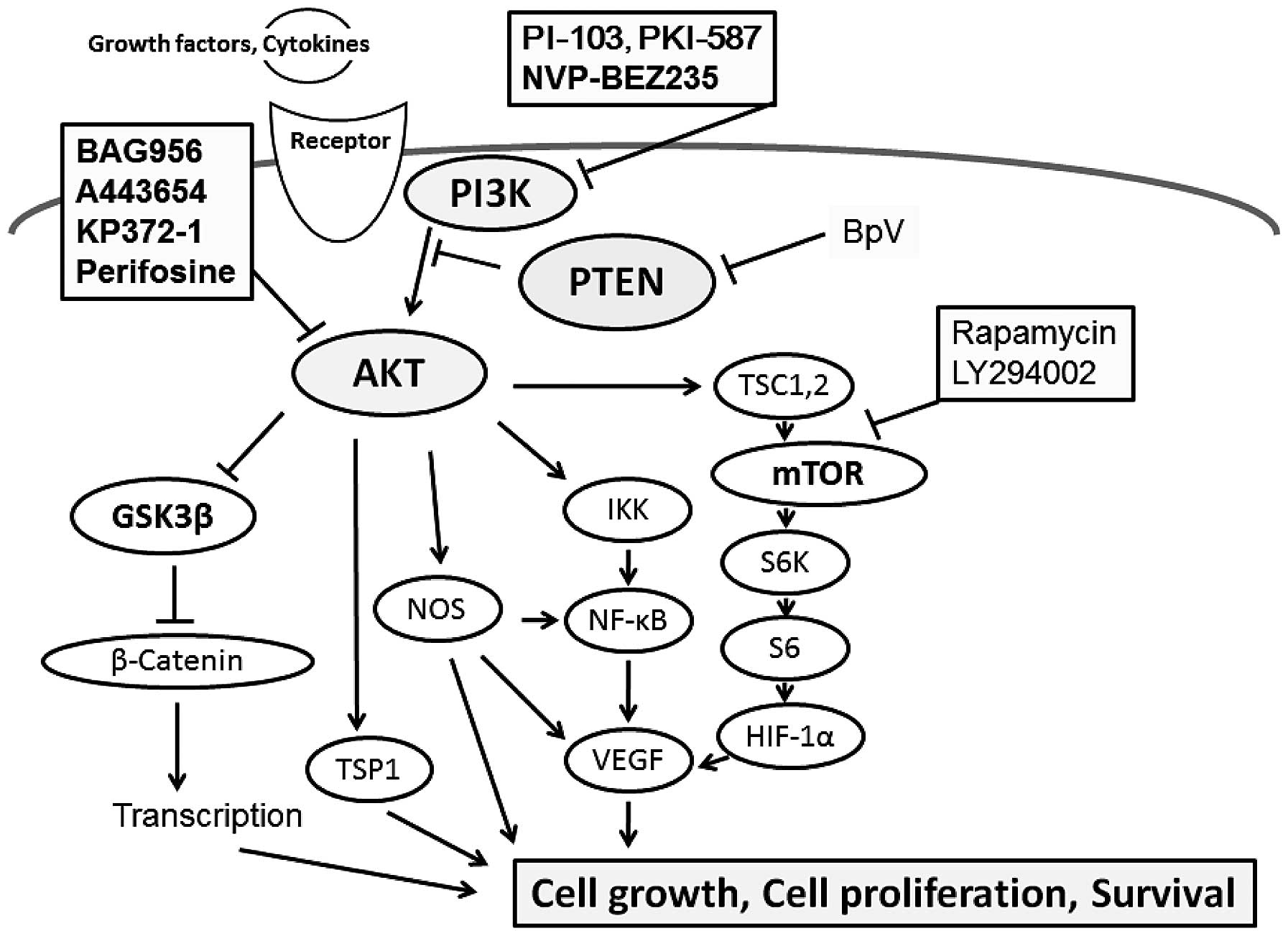
Increased proliferation and cell motility are common
cellular consequences associated with the high levels of
intra-cellular phosphatidylinositol 3,4,5-triphosphate (PIP3) that
give the metastatic characteristics to cancer (16). A feedback mechanism may increase
the PIP3 further activating AKT and metastasis. Therefore, several
studies have advocated that the PI3K/AKT signaling pathway is
concerned with the progression and the prognosis of various types
of cancer (17,18). Accordingly, suppression of PI3K
and/or AKT inhibits proliferation and induces apoptosis in cancer
cells (Fig. 1). On the contrary
for example, dysregulation of the PI3K/AKT pathway seems to
contribute to the malignant tumor activity with RET proto-oncogene
mutations (19), which are
involved in the pathogenesis of some forms of a neuroendocrine
medullary thyroid cancer. In addition, it has also been shown that
PI3K/AKT/mTOR signaling may play an important role in the biology
of papillary tumors (20).
Positive mTOR expression and PTEN loss may have a synergic effect
on tumorigenesis and cancer cell proliferation (21). The evidence may lead to possible
procedures of PI3K/AKT inhibitors in the therapy for patients with
certain cancers (Fig. 1). In fact,
targeting PI3K/AKT signaling pathway through specific inhibitors
may represent an attractive potential therapeutic approach for the
patients (19), actually
exhibiting potent antitumor efficacy, providing rationale for
clinical investigation of this inhibition in cancers of
PI3K-mediated cells (22).
Classically, wortmannin and LY294002 are the most characterized
PI3K/AKT pathway inhibitors which prevent ATP from binding to the
PI3Ks (23,24), both of which are cell-permeable and
low molecular weight compounds. Wortmannin irreversibly inhibits
PI3Ks, while the inhibition with LY294002 is reversible (10). Generally, PI3K inhibitors have been
discovered to affect cell growth, proliferation and survival of
cancer cells, as predicted before. The PI3K/AKT and its downstream
inhibitors, when combined with other pharmacological agents would
appear to be a more promising therapeutic modality. Particularly,
PI3K/AKT inhibitors are prominently effective in PTEN null cells
(25).
ROS play a pivotal role in the pathogenesis both of
neuroinflammatory and of neuro-degenerative diseases (26). Inflammatory conditions generally
lead to the chronic neurodegenerative disease such as Parkinson's
disease, Huntington's disease and Alzheimer's disease (27). Studies have indicated that cancer
therapy commonly induces ROS production, which accelerates
blood-brain barrier disruption and the neuronal cell death
(28). Cancer therapy may induce
apoptosis both for cancer cells and for normal neuronal cells. In
this meaning, reduction of ROS may have a high potential to
diminish brain damage induced during cancer therapy. It has been
demonstrated that upregulation of PTEN causes modulation of
PI3K/AKT signaling pathway to reduce the ROS generation in cells
(29). As described before, the
PTEN is one of the most prominent tumor suppressor genes
that is frequently deleted or mutated in a variety of human cancers
(30). In addition, PTEN is highly
expressed in neurons and it has been described to be related to
critical neuroprotection (31,32).
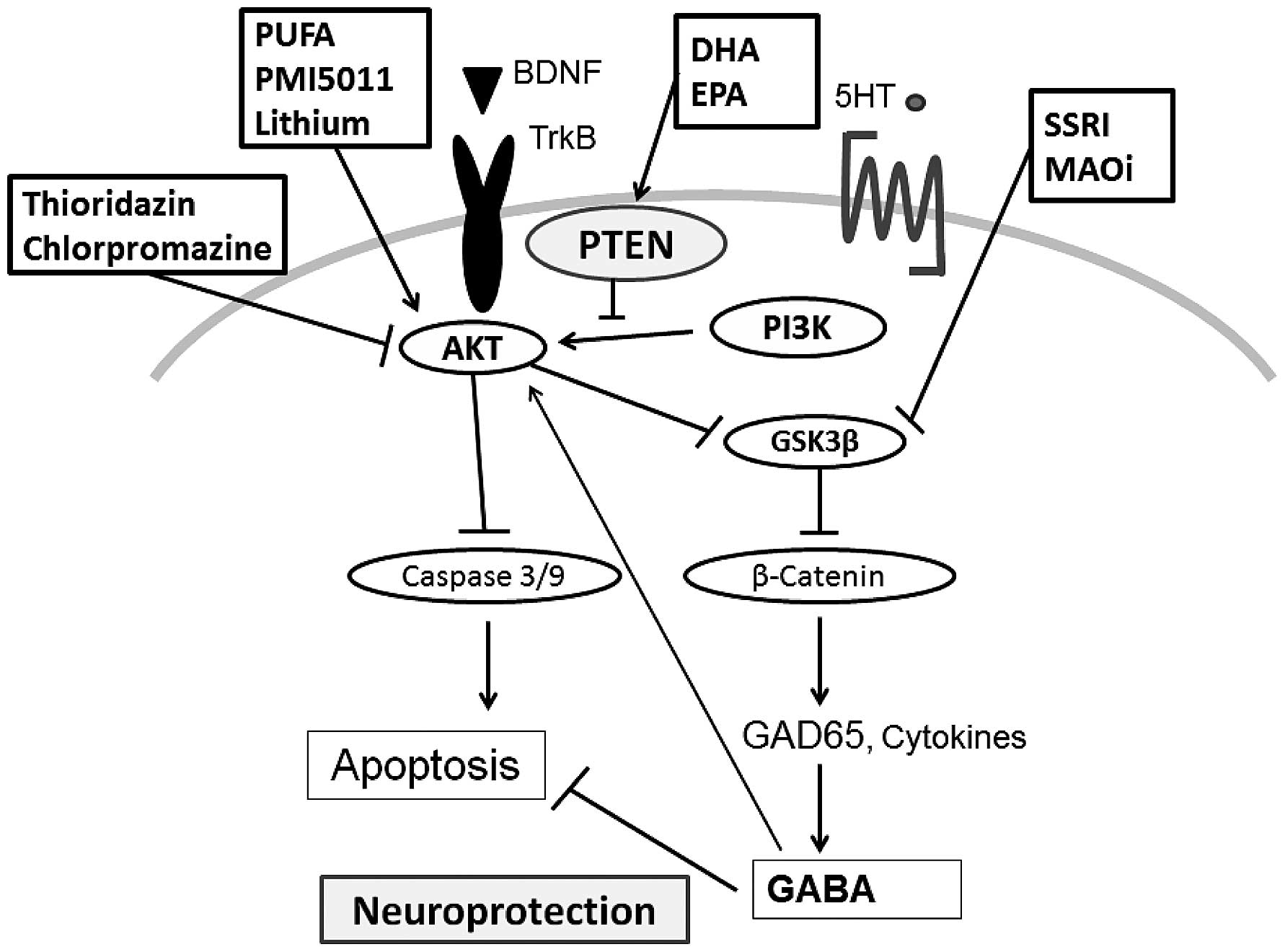
Recently, it has been shown that AKT activation may likely play a
therapeutic role in neurodegenerative diseases (33,34).
Schematic structures of the AKT and PTEN protein are shown in
Fig. 2. Accordingly, the tumor
suppressor PTEN, which antagonizes the PI3K/AKT pathway, has been
recognized to play a crucial role in neural functions.
Neuroprotection by inhibiting PTEN has been reported by activating
the PI3K/AKT pathway in primary neurons (35,36).
Deletion of PTEN gene has been shown to result in cognitive
impairment (37). Ischemic stroke
induces rapid PTEN degradation in both neurons and astrocytes which
play both protective and detrimental action to neurons in a
spatiotemporal- and cell-type-dependent manner (38). Simultaneous deletion of PTEN
promotes significant nerve regeneration after the crush injury with
enhanced axon regeneration (39).
However, there is somewhat controversy whether accumulation of the
tumor suppressor PTEN protein under stress conditions such as
trauma and stroke causes neuronal cell death. A number of studies
have reported enhanced apoptosis in neurons possessing increased
amount of nuclear PTEN, with the interpretation that its nuclear
phosphatase activity leads to the reduction of survival AKT
signaling activity. One interpretation is that brain trauma may
modify the nucleo-cytoplasmic distribution of PTEN, resulting in
the increased nuclear PTEN, but only in surviving neurons near the
traumatic lesion (15,40).
Cognitive failures after brain tumor and its
treatments are appropriately consistent, although there is only a
small worldwide body of research describing them (41). Chemotherapy against cancer is often
associated with cognitive deficits which may remain after the end
of the treatment (42). In the
central nervous system, PI3K/AKT signaling modifies synaptic
plasticity underlying the memory-processes suggesting that PI3K/AKT
signaling contributes to improvement in the cognitive development.
Inhibition of PTEN is expected to promote neural cell survival,
neuroprotection and neuroregeneration, which can also promote
myelination of axons through the AKT activation (43). Actually, a PTEN inhibitor reduces
tissue damage, neuronal cell death and promotes the functional
recovery (43). In addition, it
has been shown that PTEN-induced putative kinase 1 (PINK1) induces
mitophagy promoting neuroprotection on Huntington's disease
(44). Furthermore,
anti-depressants acting on serotonin neurotransmission generally
activate AKT then inhibit the downstream GSK3β in neuronal cells.
Several psychoactive medicines have been shown to modulate the
activity of the PI3K/AKT signaling (45) (Fig.
2). AKT has downstream substrates such as the GSK3β (46). MAO inhibitors and SSRI
antidepressants which promote serotonin synaptic transmission may
inhibit the GSK3β activity (47).
Atypical antipsychotics may inhibit the regulation of the GSK3β
signaling (48). Similar results
have been reported after the treatment with haloperidol (49,50).
Chlorpromazine is a prominent antipsychotic agent initially
developed to control several psychotic disorders, which also
inhibits cell proliferation and cell survival by suppressing
PI3K/AKT signaling pathway (51).
Whereas AKT pathway is regulated by different types of psychiatric
drugs in this way, lithium activates PI3K and PI3K-dependent
downstream AKT signaling (52).
These activities eventually protect neuronal cells against neuronal
toxicity. Therefore, treatment with inhibitors of PI3K may
exacerbate neuronal cell death after brain damage in the area of
injury. On the contrary, inhibition of PTEN expression diminishes
the neurological damage after brain injury. In addition, it has
been revealed that pharmacological inhibition of PTEN protects
against brain injury in a dose-dependent manner, whose protective
effect might be induced via the upregulation of PI3K/AKT signaling
(53,54). Furthermore, PTEN has some crosstalk
effects with serotonin receptor signaling (55). By blocking dopamine D2 receptors,
classic antipsychotics can prevent the inhibition of AKT signaling
(56,57). In addition, reductions in AKT
activation in neurons may increase excitability through decreases
in GABA neurotransmission (58).
It seems important to exploit the potential benefits of these
optimal treatments and/or combination with the PI3K/AKT modulators
for the actual neuroprotection against neurological damage.
The PI3K/AKT and PTEN signaling plays various
cellular key roles under normal and/or pathological conditions.
Activation of both pathways can impact functional consequences in
cancer therapy and the subsequent multiple CNS disorders. Of
course, pharmacological interference of signaling molecules
provides convenient method. For example, bisperoxovanadium
compounds inhibit PTEN signaling, and have been used for elevation
of neuroprotection in many CNS damage studies (43,59,60).
In addition, small molecules have prevalent use by means of
experimental tools as well as therapeutics (61). Now, it is challenging to describe
appropriate strategies to achieve cost effective benefits from easy
diets to control the PTEN signaling molecules. Dietary and/or
therapeutic interventions could not only contribute to the
prevention of diseases but reduce the rate of its development.
Actually, several herbs had been established to have
characteristics for antitumor activity (62). In addition, dietary regulation of
PI3K/AKT signaling via the modulating PTEN expression might be
effective both for cancer inhibition and for a neuroprotection. For
example, dietary intake of isothiocyanate sulforaphane, a dietary
isothiocyanate derived from broccoli, modulates PTEN gene
expression (63). Dietary exposure
to the soy isoflavone such as genistein also induces PTEN
expression at physiologically relevant concentrations (64). Phytoestrogen exposure might result
in an increase in PTEN expression and the subsequent decrease in
cellular responses including AKT phosphorylation. Accordingly,
consumption of moderate levels of soy, vegetables, some fruits, and
red wine can be protective against cancers. A high-fat diet raises
circulating fatty acids, which also increases PTEN expression in
prostate epithelial cells (65).
In addition, dietary intake of indole-3-carbinol upregulates PTEN
in an animal model (66).
Indole-3-carbinol is a promising cancer-preventive phytochemical
found in some vegetables such as broccoli. PTEN expression at the
level of mRNA and protein is elevated in experimental animals fed
with whey protein (67) which has
been shown to possess multiple health benefits (67). In addition, it has been reported
that DHA and EPA raise the level of PTEN in breast cancer cells,
providing a mechanism for the beneficial effects of fish oils on
breast cancer cells (68,69). Fish oil rich in polyunsaturated
fatty acids may induce the PTEN expression by activation of
peroxisome proliferator-activated receptor (PPAR) (68,69),
which also attenuate neuron cellular damage after a brain ischemia
and appear to play an important role in the activation of
anti-apoptotic signaling (70).
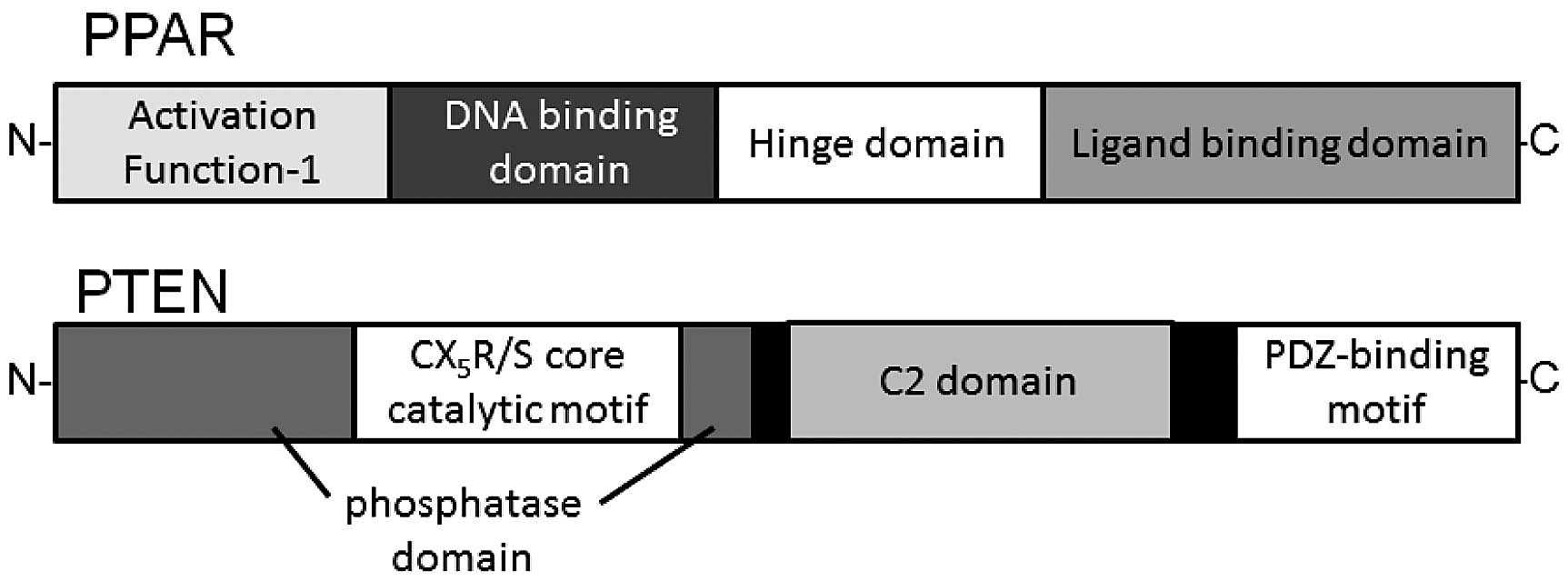
Schematic structures of human PPAR and PTEN are shown in Fig. 3. PUFA ethanolamides DHA and EPA
induce autophagy through PPAR activation in cancer cells (68). In contrast, high fat diet
attenuates the neuroprotection because of decreased survival of AKT
signaling (71). A lignan Honokiol
isolated from the bark of Magnolia officinalis could
attenuate PI3K/AKT signaling by upregulation of PTEN expression
(72), which is a potential
antitumor compound. Honokiol has been reported to improve the
learning and memory impairments in experimental animals (73). A food ingredient curcumin, derived
from the root of the plant Curcuma longa, repairs PTEN
expression (74). Curcumin
inhibits cell proliferation in human osteoclastoma cells (75). Furthermore, curcumin is a potential
therapeutic mediator for neuro-cognition (76). In contrast, certain component of
rosemary herb decreases the expression of PTEN in K562 human
myeloid cells (77). Resveratrol,
an ingredient in grapes, has been reported to exhibit anticancer
activity, anti-inflammatory activity and cardiovascular protection
property. Notably, resveratrol has been recently reported to have
neuroprotective effect (78).
Supplementation of these natural compounds may provide an
innovative therapeutic approach to brain disorders after cancer
therapy (31).
Increasing evidence pointing to AKT
pathway-modification in psychotic disorders offer a novel
implication of the treatment mechanisms for rescuing brain damage
after cancer therapy. Although PTEN has been discovered as a tumor
suppressor, PTEN is also involved in several other diseases, and
may be regulated at multiple levels including transcription,
protein stability and phosphorylation. Potential synergy with other
targeted inhibitors and/or with conventional chemotherapy may
provide additional therapeutic options to optimize the therapeutic
efficacy. In addition, abnormalities in the PI3K/AKT signaling
pathway and in the cross-talk pathways provide a clear rationale
for the development of signal transduction inhibitor-based
approaches aimed for both cancer therapy and the subsequent
neurological disorders. Understanding the important roles of the
intracellular signaling and the intricate internet work
communication is an important direction for making effective and
specific therapies. However, much more work is needed to
effectively produce and validate therapies that target pathways.
Particularly, it will be now a challenge to seek out how to use
medicinal herbs for the correction in critical processes required
for maintaining neuronal homeostasis linking to a brain injury
characterized by cancer therapy. Current evidence, as described
here, suggests that work toward understanding these complex
signaling cascades is not only promising, but may be critical for
achieving the goal of improved neurological outcome after brain and
spinal cord damage. This relationship has similarities with another
relationship between methotrexate (MTX) therapy and a leucovorin
(LV) rescue (79). We think the
order of the usage of medicine and/or compound for the treatment is
the most important. Of course, precise understanding of these
regulations is crucial for therapeutic intervention and the
effective design of novel therapeutics. A comprehensive description
of the PI3K/AKT signaling network has to be evolved. Studies may be
ongoing to identify the most optimal inhibitors for cancer
subtypes.
The present study was supported by the JSPS KAKENHI
grant number 26-12035, 24240098.
|
1
|
Lee JJ, Loh K and Yap YS: PI3K/Akt/mTOR
inhibitors in breast cancer. Cancer Biol Med. 12:342–354. 2015.
|
|
2
|
Robbins HL and Hague A: The PI3K/Akt
pathway in tumors of endocrine tissues. Front Endocrinol
(Lausanne). 6:1882016.
|
|
3
|
Faes S and Dormond O: PI3K and AKT:
Unfaithful partners in cancer. Int J Mol Sci. 16:21138–21152. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gesmundo I, Villanova T, Gargantini E,
Arvat E, Ghigo E and Granata R: The mineralocorticoid agonist
fludrocortisone promotes survival and proliferation of adult
hippocampal progenitors. Front Endocrinol (Lausanne). 7:662016.
|
|
5
|
Wahane SD, Hellbach N, Prentzell MT, Weise
SC, Vezzali R, Kreutz C, Timmer J, Krieglstein K, Thedieck K and
Vogel T: PI3K-p110-alpha-subtype signalling mediates survival,
proliferation and neurogenesis of cortical progenitor cells via
activation of mTORC2. J Neurochem. 130:255–267. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Simper NB, Jones CL, MacLennan GT,
Montironi R, Williamson SR, Osunkoya AO, Wang M, Zhang S, Grignon
DJ, Eble JN, et al: Basal cell carcinoma of the prostate is an
aggressive tumor with frequent loss of PTEN expression and
overexpression of EGFR. Hum Pathol. 46:805–812. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fallahabadi ZR, Noori Daloii MR, Mahdian
R, Behjati F, Shokrgozar MA, Abolhasani M, Asgari M and Shahrokh H:
Frequency of PTEN alterations, TMPRSS2-ERG fusion and their
association in prostate cancer. Gene. 575:755–760. 2016. View Article : Google Scholar
|
|
8
|
Collaud S, Tischler V, Atanassoff A, Wiedl
T, Komminoth P, Oehlschlegel C, Weder W and Soltermann A: Lung
neuroendocrine tumors: Correlation of ubiquitinylation and
sumoylation with nucleo-cytosolic partitioning of PTEN. BMC Cancer.
15:742015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen SY, Zheng XW, Cai JX, Zhang WP, You
HS, Xing JF and Dong YL: Histone deacetylase inhibitor reverses
multidrug resistance by attenuating the nucleophosmin level through
PI3K/Akt pathway in breast cancer. Int J Oncol. 49:294–304.
2016.PubMed/NCBI
|
|
10
|
Sinagra T, Tamburella A, Urso V, Siarkos
I, Drago F, Bucolo C and Salomone S: Reversible inhibition of
vasoconstriction by thiazolidinediones related to PI3K/Akt
inhibition in vascular smooth muscle cells. Biochem Pharmacol.
85:551–559. 2013. View Article : Google Scholar
|
|
11
|
Day J, Gillespie DC, Rooney AG, Bulbeck
HJ, Zienius K, Boele F and Grant R: Neurocognitive deficits and
neurocognitive rehabilitation in adult brain tumors. Curr Treat
Options Neurol. 18:222016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cheung YT and Krull KR: Neurocognitive
outcomes in long-term survivors of childhood acute lymphoblastic
leukemia treated on contemporary treatment protocols: A systematic
review. Neurosci Biobehav Rev. 53:108–120. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zheng Q, Yang L, Tan LM, Qin LX, Wang CY
and Zhang HN: Stroke-like migraine attacks after radiation therapy
syndrome. Chin Med J (Engl). 128:2097–2101. 2015. View Article : Google Scholar
|
|
14
|
Ricard D, Taillia H and Renard JL: Brain
damage from anticancer treatments in adults. Curr Opin Oncol.
21:559–565. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Goh CP, Putz U, Howitt J, Low LH,
Gunnersen J, Bye N, Morganti-Kossmann C and Tan SS: Nuclear
trafficking of Pten after brain injury leads to neuron survival not
death. Exp Neurol. 252:37–46. 2014. View Article : Google Scholar
|
|
16
|
Qin J, Xie Y, Wang B, Hoshino M, Wolff DW,
Zhao J, Scofield MA, Dowd FJ, Lin MF and Tu Y: Upregulation of
PIP3-dependent Rac exchanger 1 (P-Rex1) promotes prostate cancer
metastasis. Oncogene. 28:1853–1863. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen F, Chen X, Yang D, Che X, Wang J, Li
X, Zhang Z, Wang Q, Zheng W, Wang L, et al: Isoquercitrin inhibits
bladder cancer progression in vivo and in vitro by regulating the
PI3K/Akt and PKC signaling pathways. Oncol Rep. 36:165–172.
2016.PubMed/NCBI
|
|
18
|
Zhang J, Yu XH, Yan YG, Wang C and Wang
WJ: PI3K/Akt signaling in osteosarcoma. Clin Chim Acta.
444:182–192. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Manfredi GI, Dicitore A, Gaudenzi G,
Caraglia M, Persani L and Vitale G: PI3K/Akt/mTOR signaling in
medullary thyroid cancer: A promising molecular target for cancer
therapy. Endocrine. 48:363–370. 2015. View Article : Google Scholar
|
|
20
|
Goschzik T, Gessi M, Denkhaus D and
Pietsch T: PTEN mutations and activation of the PI3K/Akt/mTOR
signaling pathway in papillary tumors of the pineal region. J
Neuropathol Exp Neurol. 73:747–751. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Han X, Ji Y, Zhao J, Xu X and Lou W:
Expression of PTEN and mTOR in pancreatic neuroendocrine tumors.
Tumour Biol. 34:2871–2879. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee M, Wiedemann T, Gross C, Leinhäuser I,
Roncaroli F, Braren R and Pellegata NS: Targeting PI3K/mTOR
signaling displays potent antitumor efficacy against nonfunctioning
pituitary adenomas. Clin Cancer Res. 21:3204–3215. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bony C, Roche S, Shuichi U, Sasaki T,
Crackower MA, Penninger J, Mano H and Pucéat M: A specific role of
phosphatidylinositol 3-kinase gamma. A regulation of autonomic
Ca2+ oscillations in cardiac cells. J Cell Biol.
152:717–728. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yanamandra M, Mitra S and Giri A:
Development and application of PI3K assays for novel drug
discovery. Expert Opin Drug Discov. 10:171–186. 2015. View Article : Google Scholar
|
|
25
|
Lassen A, Atefi M, Robert L, Wong DJ,
Cerniglia M, Comin-Anduix B and Ribas A: Effects of AKT inhibitor
therapy in response and resistance to BRAF inhibition in melanoma.
Mol Cancer. 13:832014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schilling T and Eder C: Stimulus-dependent
requirement of ion channels for microglial NADPH oxidase-mediated
production of reactive oxygen species. J Neuroimmunol. 225:190–194.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Clark LF and Kodadek T: The immune system
and neuroinflammation as potential sources of blood-based
biomarkers for Alzheimer's disease, Parkinson's disease, and
Huntington's disease. ACS Chem Neurosci. 7:520–527. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lam M, Oleinick NL and Nieminen AL:
Photodynamic therapy-induced apoptosis in epidermoid carcinoma
cells. Reactive oxygen species and mitochondrial inner membrane
permeabilization. J Biol Chem. 276:47379–47386. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhu Y, Hoell P, Ahlemeyer B, Sure U,
Bertalanffy H and Krieglstein J: Implication of PTEN in production
of reactive oxygen species and neuronal death in in vitro models of
stroke and Parkinson's disease. Neurochem Int. 50:507–516. 2007.
View Article : Google Scholar
|
|
30
|
Lim HJ, Crowe P and Yang JL: Current
clinical regulation of PI3K/PTEN/Akt/mTOR signalling in treatment
of human cancer. J Cancer Res Clin Oncol. 141:671–689. 2015.
View Article : Google Scholar
|
|
31
|
Kitagishi Y and Matsuda S: Diets involved
in PPAR and PI3K/AKT/PTEN pathway may contribute to neuroprotection
in a traumatic brain injury. Alzheimers Res Ther. 5:422013.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu NK and Xu XM: Neuroprotection and its
molecular mechanism following spinal cord injury. Neural Regen Res.
7:2051–2062. 2012.PubMed/NCBI
|
|
33
|
Cheng B, Martinez AA, Morado J, Scofield
V, Roberts JL and Maffi SK: Retinoic acid protects against
proteasome inhibition associated cell death in SH-SY5Y cells via
the AKT pathway. Neurochem Int. 62:31–42. 2013. View Article : Google Scholar
|
|
34
|
Ermak G, Hench KJ, Chang KT, Sachdev S and
Davies KJ: Regulator of calcineurin (RCAN1-1L) is deficient in
Huntington disease and protective against mutant huntingtin
toxicity in vitro. J Biol Chem. 284:11845–11853. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Walker CL, Liu NK and Xu XM: PTEN/PI3K and
MAPK signaling in protection and pathology following CNS injuries.
Front Biol (Beijing). 8:421–433. 2013. View Article : Google Scholar
|
|
36
|
Delgado-Esteban M, Martin-Zanca D,
Andres-Martin L, Almeida A and Bolaños JP: Inhibition of PTEN by
peroxynitrite activates the phosphoinositide-3-kinase/Akt
neuroprotective signaling pathway. J Neurochem. 102:194–205. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Takeuchi K, Gertner MJ, Zhou J, Parada LF,
Bennett MV and Zukin RS: Dysregulation of synaptic plasticity
precedes appearance of morphological defects in a Pten conditional
knockout mouse model of autism. Proc Natl Acad Sci USA.
110:4738–4743. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li W, Huang R, Chen Z, Yan LJ, Simpkins JW
and Yang SH: PTEN degradation after ischemic stroke: A double-edged
sword. Neuroscience. 274:153–161. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sun F, Park KK, Belin S, Wang D, Lu T,
Chen G, Zhang K, Yeung C, Feng G, Yankner BA, et al: Sustained axon
regeneration induced by co-deletion of PTEN and SOCS3. Nature.
480:372–375. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li Y, Low LH, Putz U, Goh CP, Tan SS and
Howitt J: Rab5 and Ndfip1 are involved in Pten ubiquitination and
nuclear trafficking. Traffic. 15:749–761. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mandelblatt JS, Hurria A, McDonald BC,
Saykin AJ, Stern RA, VanMeter JW, McGuckin M, Traina T, Denduluri
N, Turner S, et al; Thinking and Living With Cancer Study.
Cognitive effects of cancer and its treatments at the intersection
of aging: What do we know; what do we need to know? Semin Oncol.
40:709–725. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Evenden J: Cognitive impairments and
cancer chemotherapy: Translational research at a crossroads. Life
Sci. 93:589–595. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Walker CL and Xu XM: PTEN inhibitor
bisperoxovanadium protects oligodendrocytes and myelin and prevents
neuronal atrophy in adult rats following cervical hemicontusive
spinal cord injury. Neurosci Lett. 573:64–68. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Khalil B, El Fissi N, Aouane A,
Cabirol-Pol MJ, Rival T and Liévens JC: PINK1-induced mitophagy
promotes neuroprotection in Huntington's disease. Cell Death Dis.
6:e16172015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ozaita A, Puighermanal E and Maldonado R:
Regulation of PI3K/Akt/GSK-3 pathway by cannabinoids in the brain.
J Neurochem. 102:1105–1114. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Li S, Zhou Y, Wang R, Zhang H, Dong Y and
Ip C: Selenium sensitizes MCF-7 breast cancer cells to
doxorubicin-induced apoptosis through modulation of phospho-Akt and
its downstream substrates. Mol Cancer Ther. 6:1031–1038. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Hui J, Zhang J, Kim H, Tong C, Ying Q, Li
Z, Mao X, Shi G, Yan J, Zhang Z, et al: Fluoxetine regulates
neurogenesis in vitro through modulation of GSK-3β/β-catenin
signaling. Int J Neuropsychopharmacol. 18:pyu0992014. View Article : Google Scholar
|
|
48
|
Park SW, Phuong VT, Lee CH, Lee JG, Seo
MK, Cho HY, Fang ZH, Lee BJ and Kim YH: Effects of antipsychotic
drugs on BDNF, GSK-3β, and β-catenin expression in rats subjected
to immobilization stress. Neurosci Res. 71:335–340. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Emamian ES, Hall D, Birnbaum MJ,
Karayiorgou M and Gogos JA: Convergent evidence for impaired
AKT1-GSK3beta signaling in schizophrenia. Nat Genet. 36:131–137.
2004. View
Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kozlovsky N, Amar S, Belmaker RH and Agam
G: Psychotropic drugs affect Ser9-phosphorylated GSK-3 beta protein
levels in rodent frontal cortex. Int J Neuropsychopharmacol.
9:337–342. 2006. View Article : Google Scholar
|
|
51
|
Shin SY, Lee KS, Choi YK, Lim HJ, Lee HG,
Lim Y and Lee YH: The antipsychotic agent chlorpromazine induces
autophagic cell death by inhibiting the Akt/mTOR pathway in human
U-87MG glioma cells. Carcinogenesis. 34:2080–2089. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liu KJ, Lee YL, Yang YY, Shih NY, Ho CC,
Wu YC, Huang TS, Huang MC, Liu HC, Shen WW, et al: Modulation of
the development of human monocyte-derived dendritic cells by
lithium chloride. J Cell Physiol. 226:424–433. 2011. View Article : Google Scholar
|
|
53
|
Wu J, Li J, Hu H, Liu P, Fang Y and Wu D:
Rho-kinase inhibitor, fasudil, prevents neuronal apoptosis via the
Akt activation and PTEN inactivation in the ischemic penumbra of
rat brain. Cell Mol Neurobiol. 32:1187–1197. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Liu B, Li L, Zhang Q, Chang N, Wang D,
Shan Y, Li L, Wang H, Feng H, Zhang L, et al: Preservation of GABAA
receptor function by PTEN inhibition protects against neuronal
death in ischemic stroke. Stroke. 41:1018–1026. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Cai J, Yi Z, Lu W, Fang Y and Zhang C:
Crosstalk between 5-HT2cR and PTEN signaling pathway in atypical
antipsychotic-induced metabolic syndrome and cognitive dysfunction.
Med Hypotheses. 80:486–489. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Mirones I, Angel Rodríguez-Milla M,
Cubillo I, Mariñas-Pardo L, de la Cueva T, Zapata A, González C,
Ramírez M and García-Castro J: Dopamine mobilizes mesenchymal
progenitor cells through D2-class receptors and their PI3K/AKT
pathway. Stem Cells. 32:2529–2538. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Stavarache MA, Musatov S, McGill M, Vernov
M and Kaplitt MG: The tumor suppressor PTEN regulates motor
responses to striatal dopamine in normal and Parkinsonian animals.
Neurobiol Dis. 82:487–494. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Souza BR, Romano-Silva MA and Tropepe V:
Dopamine D2 receptor activity modulates Akt signaling and alters
GABAergic neuron development and motor behavior in zebrafish
larvae. J Neurosci. 31:5512–5525. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Walker CL, Walker MJ, Liu NK, Risberg EC,
Gao X, Chen J and Xu XM: Systemic bisperoxovanadium activates
Akt/mTOR, reduces autophagy, and enhances recovery following
cervical spinal cord injury. PLoS One. 7:e300122012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Mao D and Sun X: Reactivation of the
PI3K/Akt signaling pathway by the bisperoxovanadium compound
bpV(pic) attenuates photoreceptor apoptosis in experimental retinal
detachment. Invest Ophthalmol Vis Sci. 56:5519–5532. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Spinelli L, Lindsay YE and Leslie NR: PTEN
inhibitors: an evaluation of current compounds. Adv Biol Regul.
57:102–111. 2015. View Article : Google Scholar
|
|
62
|
Tian HY, Li ZX, Li HY, Wang HJ, Zhu XW and
Dou ZH: Effects of 14 single herbs on the induction of caspase-3 in
tumor cells: A brief review. Chin J Integr Med. 19:636–640. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Traka MH, Spinks CA, Doleman JF, Melchini
A, Ball RY, Mills RD and Mithen RF: The dietary isothiocyanate
sulforaphane modulates gene expression and alternative gene
splicing in a PTEN null preclinical murine model of prostate
cancer. Mol Cancer. 9:1892010. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
de la Parra C, Castillo-Pichardo L,
Cruz-Collazo A, Cubano L, Redis R, Calin GA and Dharmawardhane S:
Soy isoflavone genistein-mediated downregulation of miR-155
contributes to the anticancer effects of genistein. Nutr Cancer.
68:154–164. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Kwon OJ, Zhang B, Zhang L and Xin L: High
fat diet promotes prostatic basal-to-luminal differentiation and
accelerates initiation of prostate epithelial hyperplasia
originated from basal cells. Stem Cell Res (Amst). 16:682–691.
2016. View Article : Google Scholar
|
|
66
|
Aronchik I, Kundu A, Quirit JG and
Firestone GL: The antiproliferative response of indole-3-carbinol
in human melanoma cells is triggered by an interaction with NEDD4-1
and disruption of wild-type PTEN degradation. Mol Cancer Res.
12:1621–1634. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Eason RR, Velarde MC, Chatman L Jr, Till
SR, Geng Y, Ferguson M, Badger TM and Simmen RC: Dietary exposure
to whey proteins alters rat mammary gland proliferation, apoptosis,
and gene expression during postnatal development. J Nutr.
134:3370–3377. 2004.PubMed/NCBI
|
|
68
|
Rovito D, Giordano C, Vizza D, Plastina P,
Barone I, Casaburi I, Lanzino M, De Amicis F, Sisci D, Mauro L, et
al: Omega-3 PUFA ethanolamides DHEA and EPEA induce autophagy
through PPARγ activation in MCF-7 breast cancer cells. J Cell
Physiol. 228:1314–1322. 2013. View Article : Google Scholar
|
|
69
|
Ghosh-Choudhury T, Mandal CC, Woodruff K,
St Clair P, Fernandes G, Choudhury GG and Ghosh-Choudhury N: Fish
oil targets PTEN to regulate NFkappaB for downregulation of
anti-apoptotic genes in breast tumor growth. Breast Cancer Res
Treat. 118:213–228. 2009. View Article : Google Scholar
|
|
70
|
Moreira JD, Knorr L, Thomazi AP, Simão F,
Battú C, Oses JP, Gottfried C, Wofchuk S, Salbego C, Souza DO, et
al: Dietary omega-3 fatty acids attenuate cellular damage after a
hippocampal ischemic insult in adult rats. J Nutr Biochem.
21:351–356. 2010. View Article : Google Scholar
|
|
71
|
Yu H, Deng J and Zuo Z: High-fat diet
reduces neuroprotection of isoflurane post-treatment: Role of
carboxyl-terminal modulator protein-Akt signaling. Obesity (Silver
Spring). 22:2396–2405. 2014. View Article : Google Scholar
|
|
72
|
Liu H, Zang C, Emde A, Planas-Silva MD,
Rosche M, Kühnl A, Schulz CO, Elstner E, Possinger K and Eucker J:
Anti-tumor effect of honokiol alone and in combination with other
anticancer agents in breast cancer. Eur J Pharmacol. 591:43–51.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Xian YF, Ip SP, Mao QQ, Su ZR, Chen JN,
Lai XP and Lin ZX: Honokiol improves learning and memory
impairments induced by scopolamine in mice. Eur J Pharmacol.
760:88–95. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Wong TF, Takeda T, Li B, Tsuiji K,
Kitamura M, Kondo A and Yaegashi N: Curcumin disrupts uterine
leiomyosarcoma cells through AKT-mTOR pathway inhibition. Gynecol
Oncol. 122:141–148. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Cao F, Liu T, Xu Y, Xu D and Feng S:
Curcumin inhibits cell proliferation and promotes apoptosis in
human osteoclastoma cell through MMP-9, NF-κB and JNK signaling
pathways. Int J Clin Exp Pathol. 8:6037–6045. 2015.
|
|
76
|
Hoppe JB, Coradini K, Frozza RL, Oliveira
CM, Meneghetti AB, Bernardi A, Pires ES, Beck RC and Salbego CG:
Free and nano-encapsulated curcumin suppress β-amyloid-induced
cognitive impairments in rats: Involvement of BDNF and Akt/GSK-3β
signaling pathway. Neurobiol Learn Mem. 106:134–144. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Yoshida H, Okumura N, Kitagishi Y,
Nishimura Y and Matsuda S: Ethanol extract of Rosemary repressed
PTEN expression in K562 culture cells. Int J appl Biol Pharm
Technol. 2:316–322. 2011.
|
|
78
|
Choi SY, Kim YO, Son D, Lee J, Kim S, Kim
H, Kim S and Hur J: 3-[2-(3,5-Dimethoxyphenyl)vinyl]furan protects
hippocampal neurons against ischemic damage. Brain Res. 1472:32–37.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Tedeschi PM, Kathari YK, Farooqi IN and
Bertino JR: Leucovorin rescue allows effective high-dose
pralatrexate treatment and an increase in therapeutic index in
mesothelioma xenografts. Cancer Chemother Pharmacol. 74:1029–1032.
2014. View Article : Google Scholar : PubMed/NCBI
|