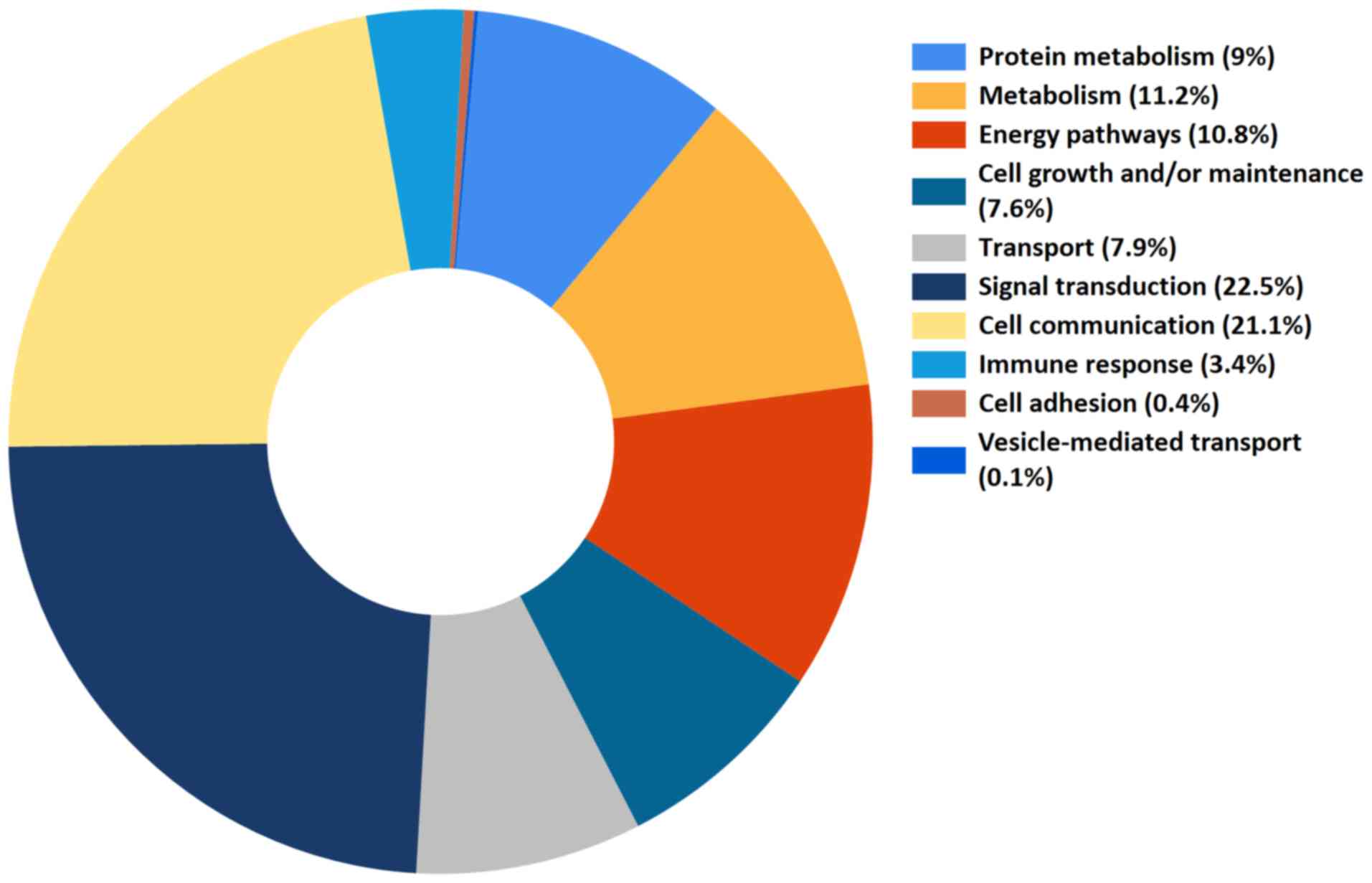
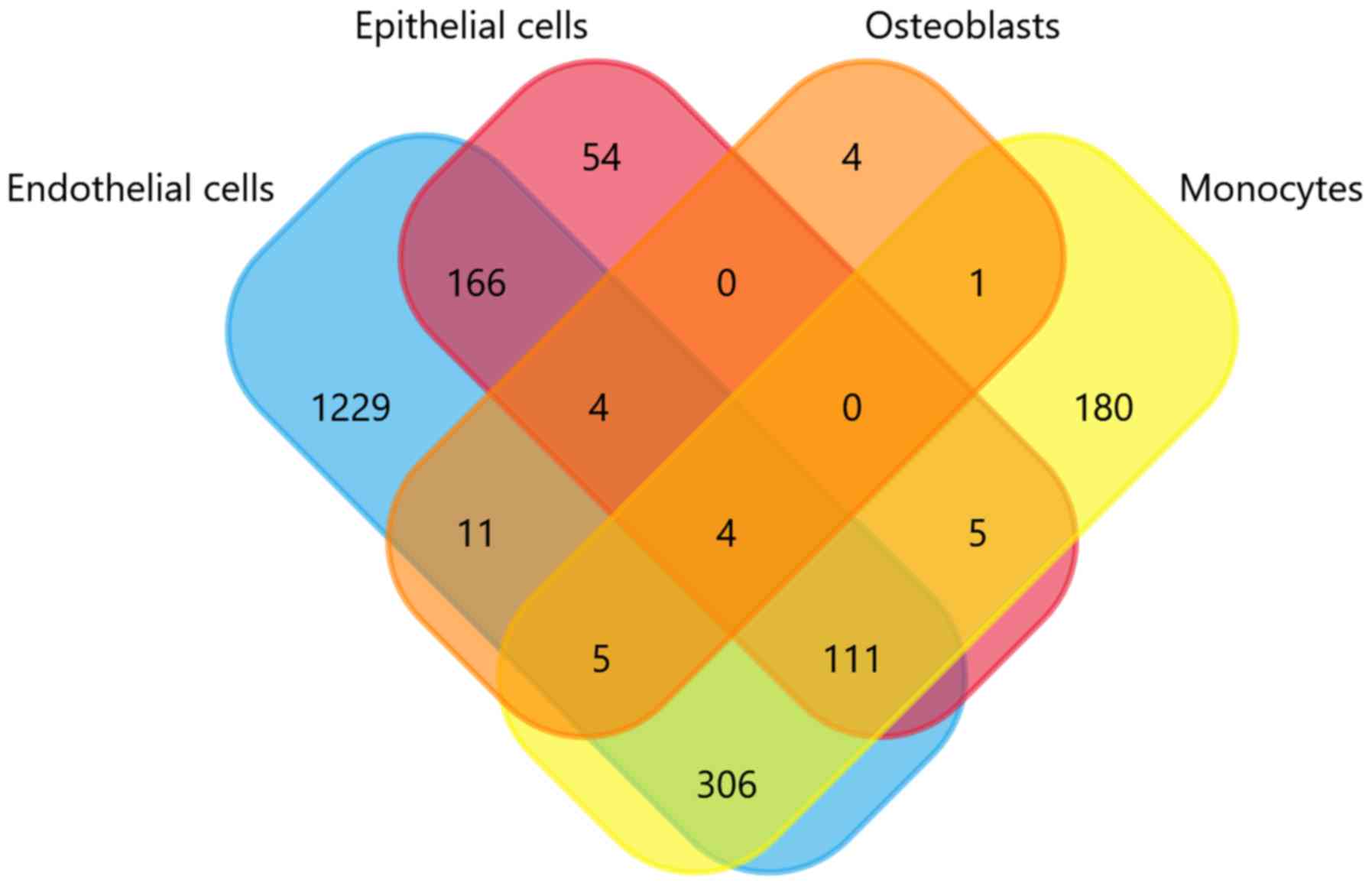
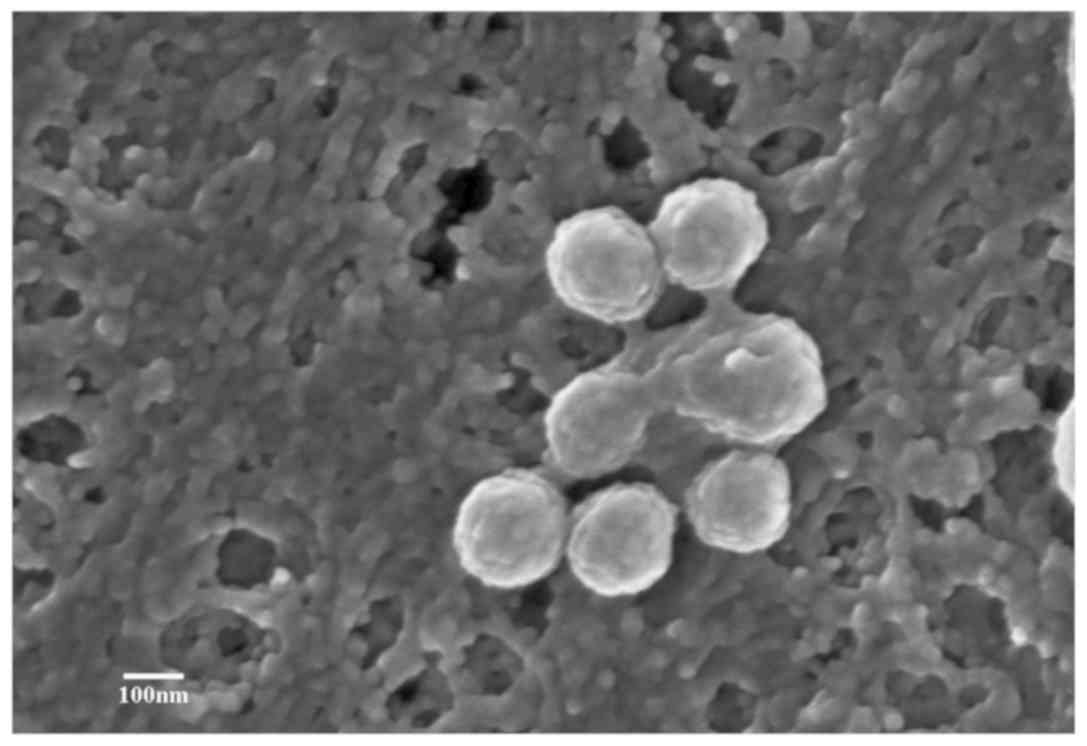
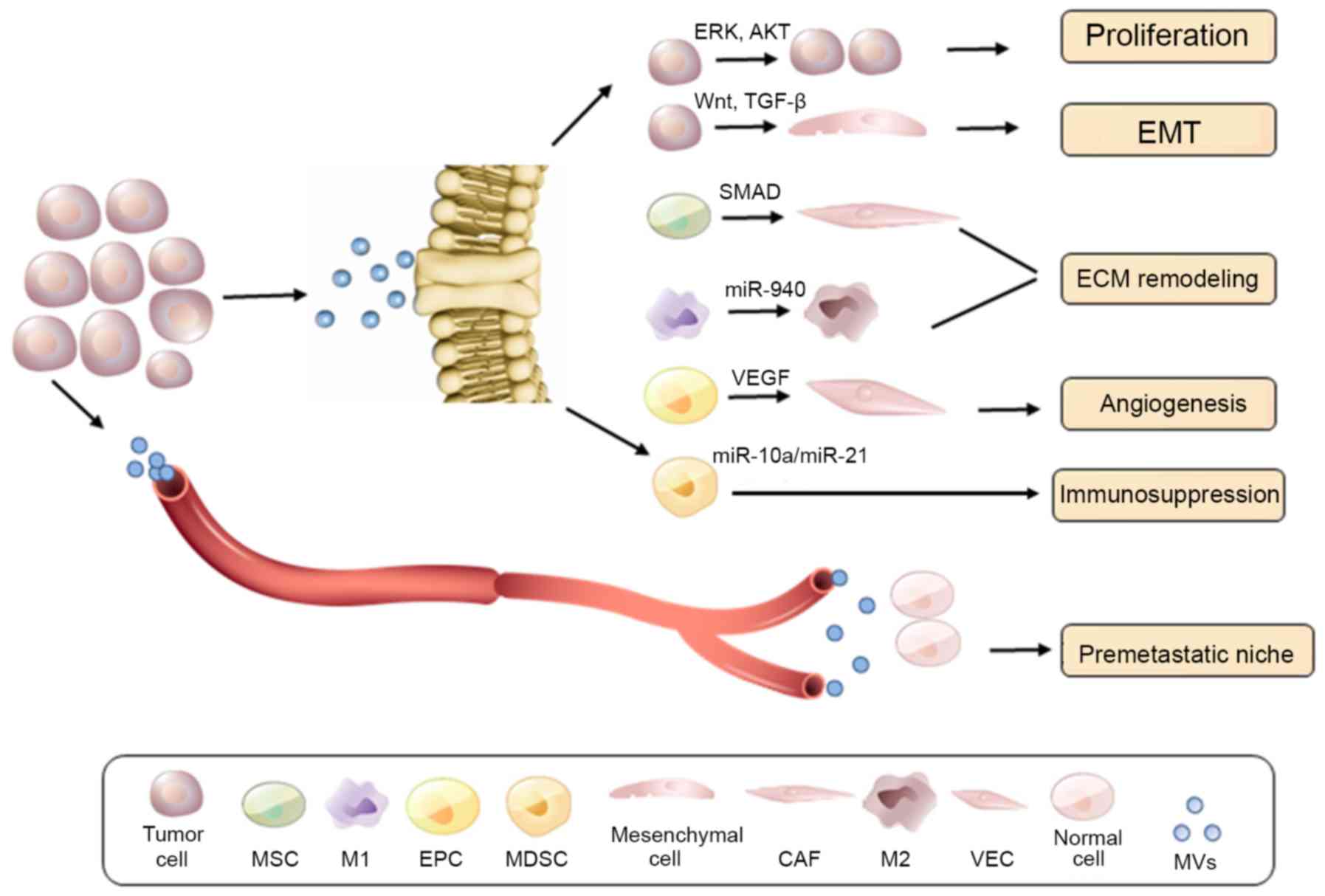
|
1
|
Lee TH, D’Asti E, Magnus N, Al-Nedawi K,
Meehan B and Rak J: Microvesicles as mediators of intercellular
communication in cancer–the emerging science of cellular ‘debris’.
Semin Immunopathol. 33:455–467. 2011.
|
|
2
|
Meckes DG Jr and Raab-Traub N:
Microvesicles and viral infection. J Virol. 85:12844–12854.
2011.
|
|
3
|
Raposo G and Stoorvogel W: Extracellular
vesicles: Exosomes, microvesicles, and friends. J Cell Biol.
200:373–383. 2013.
|
|
4
|
Barteneva NS, Maltsev N and Vorobjev IA:
Microvesicles and intercellular communication in the context of
parasitism. Front Cell Infect Microbiol. 3:492013.
|
|
5
|
Kalra H, Simpson RJ, Ji H, Aikawa E,
Altevogt P, Askenase P, Bond VC, Borràs FE, Breakefield X, Budnik
V, et al: Vesiclepedia: A compendium for extracellular vesicles
with continuous community annotation. PLoS Biol.
10:e10014502012.
|
|
6
|
Ratajczak J, Wysoczynski M, Hayek F,
Janowska-Wieczorek A and Ratajczak MZ: Membrane-derived
microvesicles: Important and underappreciated mediators of
cell-to-cell communication. Leukemia. 20:1487–1495. 2006.
|
|
7
|
Martínez MC, Larbret F, Zobairi F,
Coulombe J, Debili N, Vainchenker W, Ruat M and Freyssinet JM:
Transfer of differentiation signal by membrane microvesicles
harboring hedgehog morphogens. Blood. 108:3012–3020. 2006.
|
|
8
|
Akers JC, Gonda D, Kim R, Carter BS and
Chen CC: Biogenesis of extracellular vesicles (EV): Exosomes,
microvesicles, retrovirus-like vesicles, and apoptotic bodies. J
Neurooncol. 113:1–11. 2013.
|
|
9
|
Lawson C, Vicencio JM, Yellon DM and
Davidson SM: Microvesicles and exosomes: New players in metabolic
and cardiovascular disease. J Endocrinol. 228:R57–R71. 2016.
|
|
10
|
Jansa R, Sustar V, Frank M, Susanj P,
Bester J, Mancek-Keber M, Krzan M and Iglic A: Number of
microvesicles in peripheral blood and ability of plasma to induce
adhesion between phos-pholipid membranes in 19 patients with
gastrointestinal diseases. Blood Cells Mol Dis. 41:124–132.
2008.
|
|
11
|
Loyer X, Vion AC, Tedgui A and Boulanger
CM: Microvesicles as cell-cell messengers in cardiovascular
diseases. Circ Res. 114:345–353. 2014.
|
|
12
|
Sullivan R: Epididymosomes: A
heterogeneous population of microvesicles with multiple functions
in sperm maturation and storage. Asian J Androl. 17:726–729.
2015.
|
|
13
|
Didiot MC, Hall LM, Coles AH, Haraszti RA,
Godinho BM, Chase K, Sapp E, Ly S, Alterman JF, Hassler MR, et al:
Exosome-mediated Delivery of Hydrophobically Modified siRNA for
Huntingtin mRNA Silencing. Mol Ther. 24:1836–1847. 2016.
|
|
14
|
Cao FY, Fan JX, Long Y, Zeng X and Zhang
XZ: A smart fluorescence nanoprobe for the detection of cellular
alkaline phosphatase activity and early osteogenic differentiation.
Nanomedicine (Lond). 12:1313–1322. 2016.
|
|
15
|
Lau KH and Baylink DJ: Osteoblastic
tartrate-resistant acid phosphatase: Its potential role in the
molecular mechanism of osteogenic action of fluoride. J Bone Miner
Res. 18:1897–1900. 1900.
|
|
16
|
Aliotta JM, Pereira M, Johnson KW, de Paz
N, Dooner MS, Puente N, Ayala C, Brilliant K, Berz D and Lee D:
Microvesicle entry into marrow cells mediates tissue-specific
changes in mRNA by direct delivery of mRNA and induction of
transcription. Exp Hematol. 38:233–245. 2010.
|
|
17
|
Collino F, Deregibus MC, Bruno S, Sterpone
L, Aghemo G, Viltono L, Tetta C and Camussi G: Microvesicles
derived from adult human bone marrow and tissue specific
mesenchymal stem cells shuttle selected pattern of miRNAs. PLoS
One. 5:e118032010.
|
|
18
|
Waldenström A, Gennebäck N, Hellman U and
Ronquist G: Cardiomyocyte microvesicles contain DNA/RNA and convey
biological messages to target cells. PLoS One. 7:e346532012.
|
|
19
|
Haraszti RA, Didiot MC, Sapp E, Leszyk J,
Shaffer SA, Rockwell HE, Gao F, Narain NR, DiFiglia M, Kiebish MA,
et al: High-resolution proteomic and lipidomic analysis of exosomes
and microvesicles from different cell sources. J Extracell
Vesicles. 5:325702016.
|
|
20
|
Zheng T, Pu J, Chen Y, Mao Y, Guo Z, Pan
H, Zhang L, Zhang H, Sun B and Zhang B: Plasma Exosomes Spread and
Cluster Around β-Amyloid Plaques in an Animal Model of Alzheimer’s
Disease. Front Aging Neurosci. 9:122017.
|
|
21
|
Hwang DW, Choi H, Jang SC, Yoo MY, Park
JY, Choi NE, Oh HJ, Ha S, Lee YS, Jeong JM, et al: Noninvasive
imaging of radio-labeled exosome-mimetic nanovesicle using
(99m)Tc-HMPAO. Sci Rep. 5:156362015.
|
|
22
|
Zhao JY, Chen G, Gu YP, Cui R, Zhang ZL,
Yu ZL, Tang B, Zhao YF and Pang DW: Ultrasmall Magnetically
Engineered Ag2Se Quantum Dots for Instant Efficient Labeling and
Whole-Body High-Resolution Multimodal Real-Time Tracking of
Cell-Derived Microvesicles. J Am Chem Soc. 138:1893–1903. 2016.
|
|
23
|
Abels ER and Breakefield XO: Introduction
to Extracellular Vesicles: Biogenesis, RNA Cargo Selection,
Content, Release, and Uptake. Cell Mol Neurobiol. 36:301–312.
2016.
|
|
24
|
Datta A, Chen CP and Sze SK: Discovery of
prognostic biomarker candidates of lacunar infarction by
quantitative proteomics of microvesicles enriched plasma. PLoS One.
9:e946632014.
|
|
25
|
Tricarico C, Clancy J and D’Souza-Schorey
C: Biology and biogenesis of shed microvesicles. Small GTPases.
8:220–232. 2017.
|
|
26
|
Al-Nedawi K, Meehan B, Micallef J, Lhotak
V, May L, Guha A and Rak J: Intercellular transfer of the oncogenic
receptor EGFRvIII by microvesicles derived from tumour cells. Nat
Cell Biol. 10:619–624. 2008.
|
|
27
|
Principe S, Hui AB, Bruce J, Sinha A, Liu
FF and Kislinger T: Tumor-derived exosomes and microvesicles in
head and neck cancer: Implications for tumor biology and biomarker
discovery. Proteomics. 13:1608–1623. 2013.
|
|
28
|
Lee K, Shao H, Weissleder R and Lee H:
Acoustic purification of extracellular microvesicles. ACS Nano.
9:2321–2327. 2015.
|
|
29
|
Tickoo SK, Lee MW, Eble JN, Amin M,
Christopherson T, Zarbo RJ and Amin MB: Ultrastructural
observations on mitochondria and microvesicles in renal oncocytoma,
chromophobe renal cell carcinoma, and eosinophilic variant of
conventional (clear cell) renal cell carcinoma. Am J Surg Pathol.
24:1247–1256. 2000.
|
|
30
|
Silverman JM and Reiner NE: Exosomes and
other microvesicles in infection biology: Organelles with
unanticipated phenotypes. Cell Microbiol. 13:1–9. 2011.
|
|
31
|
Mathivanan S, Lim JW, Tauro BJ, Ji H,
Moritz RL and Simpson RJ: Proteomics analysis of A33
immunoaffinity-purified exosomes released from the human colon
tumor cell line LIM1215 reveals a tissue-specific protein
signature. Mol Cell Proteomics. 9:197–208. 2010.
|
|
32
|
Wenzel D, Schauermann G, von Lüpke A and
Hinz G: The cargo in vacuolar storage protein transport vesicles is
stratified. Traffic. 6:45–55. 2005.
|
|
33
|
Miranda KC, Bond DT, Levin JZ, Adiconis X,
Sivachenko A, Russ C, Brown D, Nusbaum C and Russo LM: Massively
parallel sequencing of human urinary exosome/microvesicle RNA
reveals a predominance of non-coding RNA. PLoS One.
9:e960942014.
|
|
34
|
Arendt BK, Walters DK, Wu X, Tschumper RC
and Jelinek DF: Multiple myeloma dell-derived microvesicles are
enriched in CD147 expression and enhance tumor cell proliferation.
Oncotarget. 5:5686–5699. 2014.
|
|
35
|
Liao CF, Lin SH, Chen HC, Tai CJ, Chang
CC, Li LT, Yeh CM, Yeh KT, Chen YC, Hsu TH, et al: CSE1L, a novel
microvesicle membrane protein, mediates Ras-triggered microvesicle
generation and metastasis of tumor cells. Mol Med. 18:1269–1280.
2012.
|
|
36
|
Dong Y, Pan Q, Jiang L, Chen Z, Zhang F,
Liu Y, Xing H, Shi M, Li J, Li X, et al: Tumor endothelial
expression of P-glycoprotein upon microvesicular transfer of TrpC5
derived from adriamycin-resistant breast cancer cells. Biochem
Biophys Res Commun. 446:85–90. 2014.
|
|
37
|
Aubertin K, Silva AKA, Luciani N, Espinosa
A, Djemat A, Charue D, Gallet F, Blanc-Brude O and Wilhelm C:
Massive release of extracellular vesicles from cancer cells after
photo-dynamic treatment or chemotherapy. Sci Rep. 6:353762016.
|
|
38
|
Qiu J, Yang G, Feng M, Zheng S, Cao Z, You
L, Zheng L, Zhang T and Zhao Y: Extracellular vesicles as mediators
of the progression and chemoresistance of pancreatic cancer and
their potential clinical applications. Mol Cancer. 17:22018.
|
|
39
|
Muralidharan-Chari V, Clancy JW, Sedgwick
A and D’Souza-Schorey C: Microvesicles: Mediators of extracellular
communication during cancer progression. J Cell Sci. 123:1603–1611.
2010.
|
|
40
|
Wang X, Xu C, Hua Y, Sun L, Cheng K, Jia
Z, Han Y, Dong J, Cui Y and Yang Z: Exosomes play an important role
in the process of psoralen reverse multidrug resistance of breast
cancer. J Exp Clin Cancer Res. 35:1862016.
|
|
41
|
Kholia S, Jorfi S, Thompson PR, Causey CP,
Nicholas AP, Inal JM and Lange S: A novel role for peptidylarginine
deiminases in microvesicle release reveals therapeutic potential of
PAD inhibition in sensitizing prostate cancer cells to
chemotherapy. J Extracell Vesicles. 4:261922015.
|
|
42
|
Al-Nedawi K, Meehan B and Rak J:
Microvesicles: Messengers and mediators of tumor progression. Cell
Cycle. 8:2014–2018. 2009.
|
|
43
|
Wendler F, Favicchio R, Simon T,
Alifrangis C, Stebbing J and Giamas G: Extracellular vesicles swarm
the cancer micro-environment: From tumor-stroma communication to
drug intervention. Oncogene. 36:877–884. 2017.
|
|
44
|
Balaj L, Lessard R, Dai L, Cho YJ, Pomeroy
SL, Breakefield XO and Skog J: Tumour microvesicles contain
retrotransposon elements and amplified oncogene sequences. Nat
Commun. 2:1802011.
|
|
45
|
Qu JL, Qu XJ, Zhao MF, Teng YE, Zhang Y,
Hou KZ, Jiang YH, Yang XH and Liu YP: Gastric cancer exosomes
promote tumour cell proliferation through PI3K/Akt and MAPK/ERK
activation. Dig Liver Dis. 41:875–880. 2009.
|
|
46
|
Salnikov AV, Liu L, Platen M, Gladkich J,
Salnikova O, Ryschich E, Mattern J, Moldenhauer G, Werner J,
Schemmer P, et al: Hypoxia induces EMT in low and highly aggressive
pancreatic tumor cells but only cells with cancer stem cell
characteristics acquire pronounced migratory potential. PLoS One.
7:e463912012.
|
|
47
|
Zhou Y, Xiong M, Fang L, Jiang L, Wen P,
Dai C, Zhang CY and Yang J: miR-21-containing microvesicles from
injured tubular epithelial cells promote tubular phenotype
transition by targeting PTEN protein. Am J Pathol. 183:1183–1196.
2013.
|
|
48
|
Kim J, Kim TY, Lee MS, Mun JY, Ihm C and
Kim SA: Exosome cargo reflects TGF-β1-mediated
epithelial-to-mesenchymal transition (EMT) status in A549 human
lung adenocarcinoma cells. Biochem Biophys Res Commun. 478:643–648.
2016.
|
|
49
|
Ribatti D, Mangialardi G and Vacca A:
Stephen Paget and the ‘seed and soil’ theory of metastatic
dissemination. Clin Exp Med. 6:145–149. 2006.
|
|
50
|
Witz IP: The tumor microenvironment: The
making of a paradigm. Cancer Microenviron. 2(Suppl 1): 9–17.
2009.
|
|
51
|
Wiig H, Tenstad O, Iversen PO, Kalluri R
and Bjerkvig R: Interstitial fluid: The overlooked component of the
tumor micro-environment? Fibrogenesis Tissue Repair. 3:122010.
|
|
52
|
Chouaib S, Kieda C, Benlalam H, Noman MZ,
Mami-Chouaib F and Rüegg C: Endothelial cells as key determinants
of the tumor microenvironment: Interaction with tumor cells,
extracellular matrix and immune killer cells. Crit Rev Immunol.
30:529–545. 2010.
|
|
53
|
Vlodavsky I, Korner G, Ishai-Michaeli R,
Bashkin P, Bar-Shavit R and Fuks Z: Extracellular matrix-resident
growth factors and enzymes: Possible involvement in tumor
metastasis and angio-genesis. Cancer Metastasis Rev. 9:203–226.
1990.
|
|
54
|
Micke P and Ostman A: Exploring the tumour
environment: Cancer-associated fibroblasts as targets in cancer
therapy. Expert Opin Ther Targets. 9:1217–1233. 2005.
|
|
55
|
Cho JA, Park H, Lim EH and Lee KW:
Exosomes from breast cancer cells can convert adipose
tissue-derived mesenchymal stem cells into myofibroblast-like
cells. Int J Oncol. 40:130–138. 2012.
|
|
56
|
Dings R, Vang K, Griffioen A, Farrar M and
Mayo K: Angiogenesis inhibitors promote T-cell mediated anti-tumor
response by diminishing tumor counter immuno-surveillance. Cancer
Res. 68:11192008.
|
|
57
|
Caillou B, Talbot M, Weyemi U,
Pioche-Durieu C, Al Ghuzlan A, Bidart JM, Chouaib S, Schlumberger M
and Dupuy C: Tumor-associated macrophages (TAMs) form an
interconnected cellular supportive network in anaplastic thyroid
carcinoma. PLoS One. 6:e225672011.
|
|
58
|
Yang M, Chen J, Su F, Yu B, Su F, Lin L,
Liu Y, Huang JD and Song E: Microvesicles secreted by macrophages
shuttle invasion-potentiating microRNAs into breast cancer cells.
Mol Cancer. 10:1172011.
|
|
59
|
Ying X, Wu Q, Wu X, Zhu Q and Wang X,
Jiang L, Chen X and Wang X: Epithelial ovarian cancer-secreted
exosomal miR-222-3p induces polarization of tumor-associated
macrophages. Oncotarget. 7:43076–43087. 2016.
|
|
60
|
Hsieh CH, Tai SK and Yang MH:
Snail-overexpressing Cancer Cells Promote M2-Like Polarization of
Tumor-Associated Macrophages by Delivering MiR-21-Abundant
Exosomes. Neoplasia. 20:775–788. 2018.
|
|
61
|
Huang Q, Duan L, Qian X, Fan J, Lv Z,
Zhang X, Han J, Wu F, Guo M, Hu G, et al: IL-17 Promotes Angiogenic
Factors IL-6, IL-8, and Vegf Production via Stat1 in Lung
Adenocarcinoma. Sci Rep. 6:365512016.
|
|
62
|
Szajnik M, Czystowska M, Szczepanski MJ,
Mandapathil M and Whiteside TL: Tumor-derived microvesicles induce,
expand and up-regulate biological activities of human regulatory T
cells (Treg). PLoS One. 5:e114692010.
|
|
63
|
Blonska M, Agarwal NK and Vega F: Shaping
of the Tumor Microenvironment: Stromal Cells and Vessels. Semin
Cancer Biol. 34:3–13. 2015.
|
|
64
|
Atala A: Re: Microvesicles released from
human renal cancer stem cells stimulate angiogenesis and formation
of lung premeta-static niche. J Urol. 187:1506–1507. 2012.
|
|
65
|
Martins VR, Dias MS and Hainaut P:
Tumor-cell-derived microvesicles as carriers of molecular
information in cancer. Curr Opin Oncol. 25:66–75. 2013.
|
|
66
|
Alipoor SD, Mortaz E, Varahram M,
Movassaghi M, Kraneveld AD, Garssen J and Adcock IM: The Potential
Biomarkers and Immunological Effects of Tumor-Derived Exosomes in
Lung Cancer. Front Immunol. 9:8192018.
|
|
67
|
Heinrich LF, Andersen DK, Cleasby ME and
Lawson C: Long-term high fat feeding of rats results in increased
numbers of circulating microvesicles with pro-inflammatory effects
on endothelial cells. Br J Nutr. 113:1704–1711. 2015.
|
|
68
|
Aguado BA, Bushnell GG, Rao SS, Jeruss JS
and Shea LD: Engineering the pre-metastatic niche. Nat Biomed Eng.
1:1–28. 2017.
|
|
69
|
Hood JL, San RS and Wickline SA: Exosomes
released by melanoma cells prepare sentinel lymph nodes for tumor
metastasis. Cancer Res. 71:3792–3801. 2011.
|
|
70
|
Melo SA, Luecke LB, Kahlert C, Fernandez
AF, Gammon ST, Kaye J, LeBleu VS, Mittendorf EA, Weitz J, Rahbari
N, et al: Glypican-1 identifies cancer exosomes and detects early
pancreatic cancer. Nature. 523:177–182. 2015.
|
|
71
|
Semenza GL: The hypoxic tumor
microenvironment: A driving force for breast cancer progression.
Biochim Biophys Acta. 1863:382–391. 2016.
|
|
72
|
Svensson K: Mechanistic Studies On the
Role of Polyamines and Microvesicles in Tumor Growth and
Hypoxia-mediated Angiogenesis. Scand J Dent Res. 80:139–154.
2012.
|
|
73
|
Lima LG, Chammas R, Monteiro RQ, Moreira
ME and Barcinski MA: Tumor-derived microvesicles modulate the
establishment of metastatic melanoma in a
phosphatidyl-serine-dependent manner. Cancer Lett. 283:168–175.
2009.
|
|
74
|
Mineo M, Garfield SH, Taverna S, Flugy A,
De Leo G, Alessandro R and Kohn EC: Exosomes released by K562
chronic myeloid leukemia cells promote angiogenesis in a
Src-dependent fashion. Angiogenesis. 15:33–45. 2012.
|
|
75
|
Rahbari M, Rahbari N, Reissfelder C, Weitz
J and Kahlert C: Exosomes: Novel implications in diagnosis and
treatment of gastrointestinal cancer. Langenbecks Arch Surg.
401:1097–1110. 2016.
|
|
76
|
Diaz LA Jr and Bardelli A: Liquid
biopsies: Genotyping circulating tumor DNA. J Clin Oncol.
32:579–586. 2014.
|
|
77
|
Armstrong D and Wildman DE: Extracellular
Vesicles and the Promise of Continuous Liquid Biopsies. J Pathol
Transl Med. 52:1–8. 2018.
|
|
78
|
Wieckowski EU, Kim JW, Stanson JD, Taylor
DD, Reichert TE and Whiteside TL: FasL+ and FasL-microvesicles in
the circulation of patients with cancer induce apoptosis of
activated T lymphocytes. Cancer Res. 64:Issue 72004.
|
|
79
|
Hosseinibeheshti E, Pham S, Adomat H, Li N
and Tomlinson Guns ES: Exosomes as biomarker enriched
microvesicles: characterization of exosomal proteins derived from a
panel of prostate cell lines with distinct AR phenotypes. Mol Cell
Proteomics. 11:863–885. 2012.
|
|
80
|
Baran J, Baj-Krzyworzeka M, Weglarczyk K,
Szatanek R and Zembala M, Barbasz J, Czupryna A, Szczepanik A and
Zembala M: Circulating tumour-derived microvesicles in plasma of
gastric cancer patients. Cancer Immunol Immunother. 59:841–850.
2010.
|
|
81
|
Valenti R, Huber V, Filipazzi P, Pilla L,
Sovena G, Villa A, Corbelli A, Fais S, Parmiani G and Rivoltini L:
Human tumor-released microvesicles promote the differentiation of
myeloid cells with transforming growth factor-beta-mediated
suppressive activity on T lymphocytes. Cancer Res. 66:9290–9298.
2006.
|
|
82
|
Kimura S, Naganuma S, Susuki D, Hirono Y,
Yamaguchi A, Fujieda S, Sano K and Itoh H: Expression of microRNAs
in squamous cell carcinoma of human head and neck and the
esophagus: miR-205 and miR-21 are specific markers for HNSCC and
ESCC. Oncol Rep. 23:1625–1633. 2010.
|
|
83
|
Kachakova D, Mitkova A, Popov E, Popov I,
Vlahova A, Dikov T, Christova S, Mitev V, Slavov C and Kaneva R:
Combinations of serum prostate-specific antigen and plasma
expression levels of let-7c, miR-30c, miR-141, and miR-375 as
potential better diagnostic biomarkers for prostate cancer. DNA
Cell Biol. 34:189–200. 2015.
|
|
84
|
Kahlert C, Melo SA, Protopopov A, Tang J,
Seth S, Koch M, Zhang J, Weitz J, Chin L, Futreal A, et al:
Identification of double-stranded genomic DNA spanning all
chromosomes with mutated KRAS and p53 DNA in the serum exosomes of
patients with pancreatic cancer. J Biol Chem. 289:3869–3875.
2014.
|
|
85
|
Borecka M, Zemankova P, Vocka M, Soucek P,
Soukupova J, Kleiblova P, Sevcik J, Kleibl Z and Janatova M:
Mutation analysis of the PALB2 gene in unselected pancreatic cancer
patients in the Czech Republic. Cancer Genet. 209:199–204.
2016.
|
|
86
|
Vader P, Breakefield XO and Wood MJ:
Extracellular vesicles: Emerging targets for cancer therapy. Trends
Mol Med. 20:385–393. 2014.
|
|
87
|
Sidhu SS, Mengistab AT, Tauscher AN,
LaVail J and Basbaum C: The microvesicle as a vehicle for EMMPRIN
in tumor-stromal interactions. Oncogene. 23:956–963. 2004.
|
|
88
|
Jorfi S and Inal JM: The role of
microvesicles in cancer progression and drug resistance. Biochem
Soc Trans. 41:293–298. 2013.
|
|
89
|
Chen WX, Zhong SL, Ji MH, Pan M, Hu Q, Lv
MM, Luo Z, Zhao JH and Tang JH: MicroRNAs delivered by
extracellular vesicles: An emerging resistance mechanism for breast
cancer. Tumour Biol. 35:2883–2892. 2014.
|
|
90
|
Esteva FJ: Novel strategies for
HER-2-positive metastatic disease: Mechanisms and therapeutic
options to overcome trastuzumab resistance. Breast Cancer Res.
9(Suppl 1): 1–2. 2007.
|
|
91
|
Silva AK, Luciani N, Gazeau F, Aubertin K,
Bonneau S, Chauvierre C, Letourneur D and Wilhelm C: Combining
magnetic nanoparticles with cell derived microvesicles for drug
loading and targeting. Nanomedicine. 11:645–655. 2015.
|
|
92
|
Vader P, Mol EA, Pasterkamp G and
Schiffelers RM: Extracellular vesicles for drug delivery. Adv Drug
Deliv Rev. 106(Pt A): 148–156. 2016.
|
|
93
|
Tian Y, Li S, Song J, Ji T, Zhu M,
Anderson GJ, Wei J and Nie G: A doxorubicin delivery platform using
engineered natural membrane vesicle exosomes for targeted tumor
therapy. Biomaterials. 35:2383–2390. 2014.
|
|
94
|
Mizrak A, Bolukbasi MF, Ozdener GB,
Brenner GJ, Madlener S, Erkan EP, Ströbel T, Breakefield XO and
Saydam O: Genetically engineered microvesicles carrying suicide
mRNA/protein inhibit schwannoma tumor growth. Mol Ther. 21:101–108.
2013.
|
|
95
|
Kim HO, Choi SM and Kim HS: Mesenchymal
stem cell-derived secretome and microvesicles as a cell-free
therapeutics for neuro-degenerative disorders. Tissue Eng Regen
Med. 10:93–101. 2013.
|
|
96
|
Miller VM, Lahr BD, Bailey KR, Hodis HN,
Mulvagh SL and Jayachandran M: Specific cell-derived microvesicles:
Linking endothelial function to carotid artery intima-media
thickness in low cardiovascular risk menopausal women.
Atherosclerosis. 246:21–28. 2016.
|
|
97
|
Kahlert C and Kalluri R: Exosomes in tumor
microenvironment influence cancer progression and metastasis. J Mol
Med (Berl). 91:431–437. 2013.
|