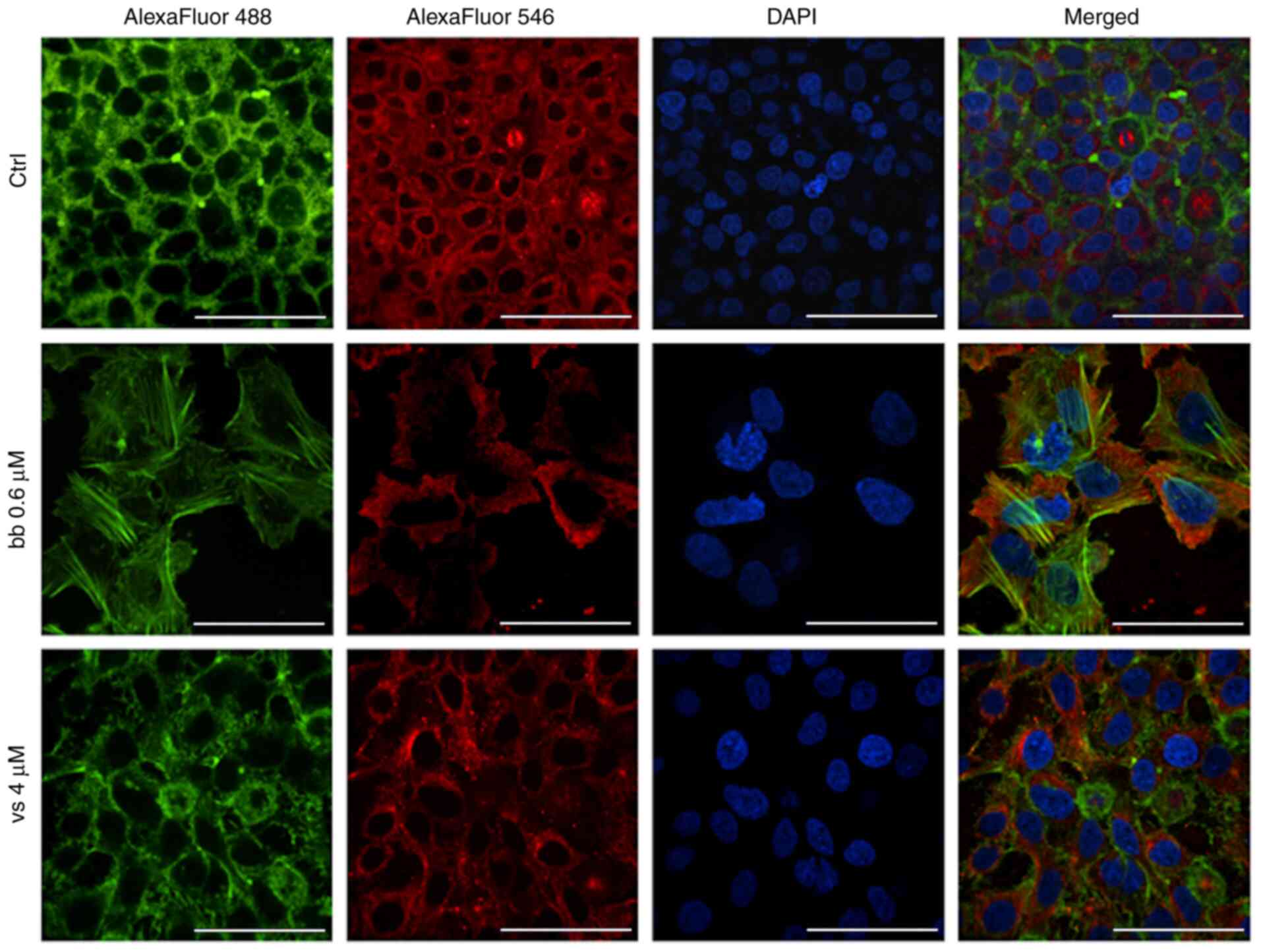
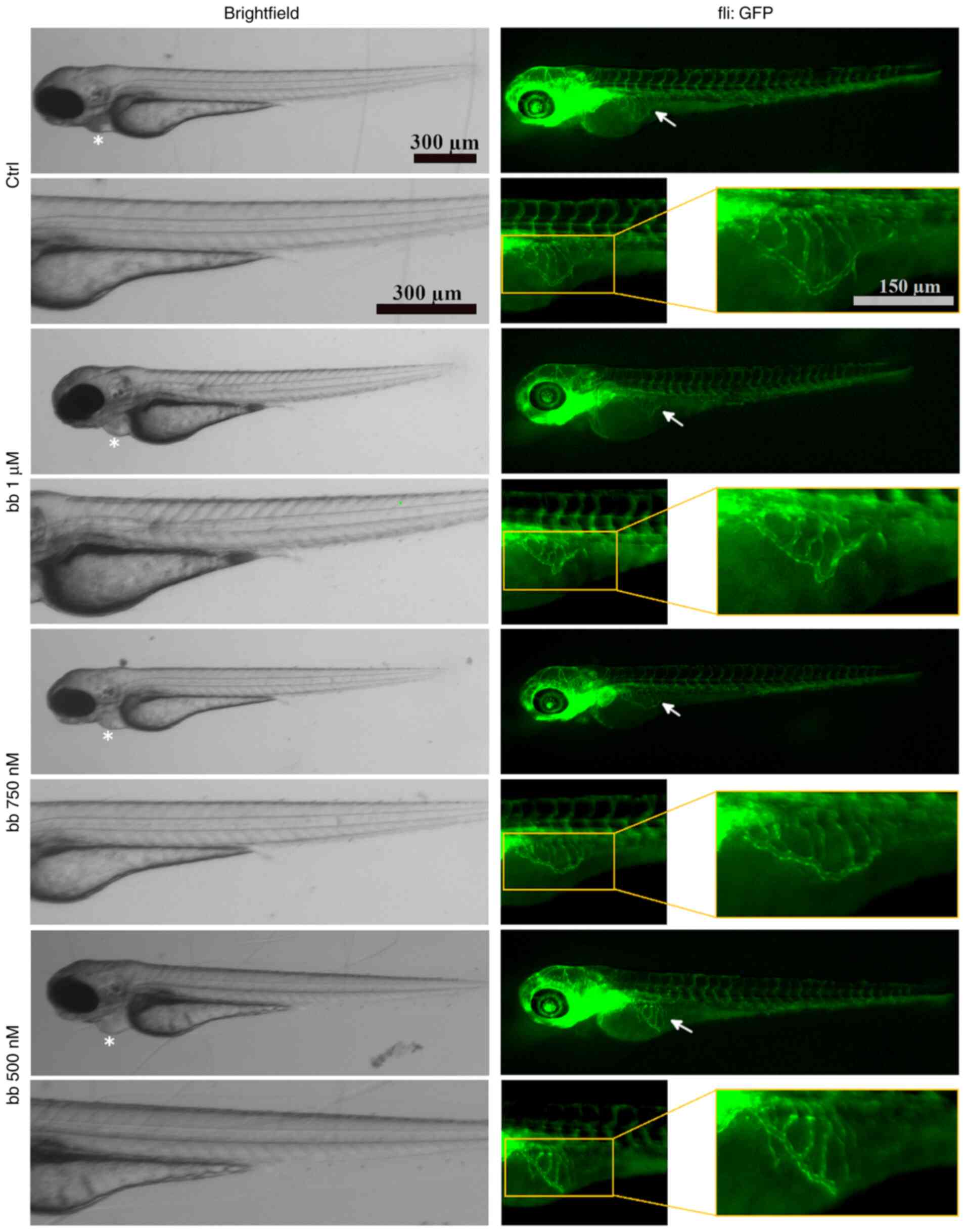
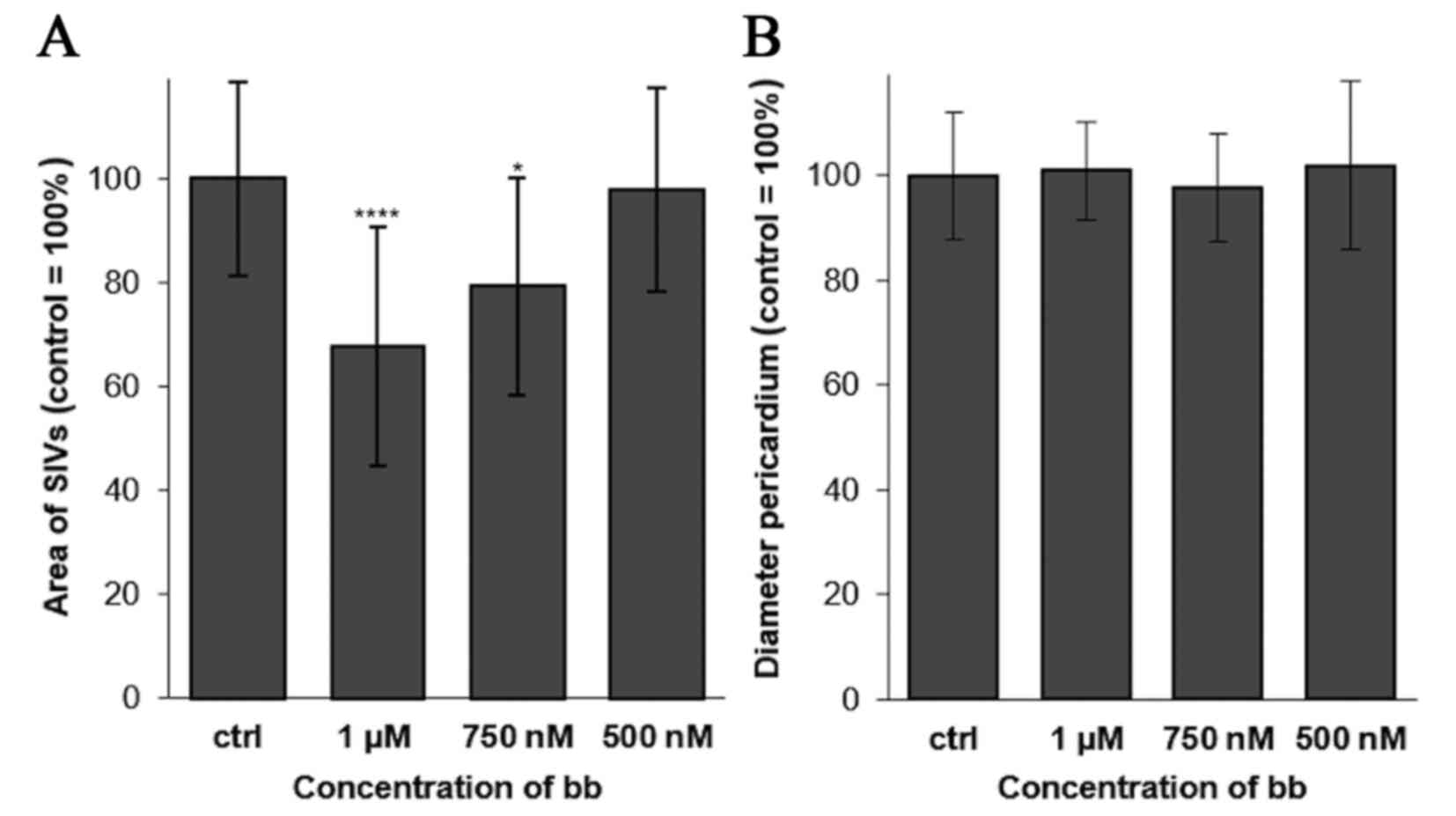
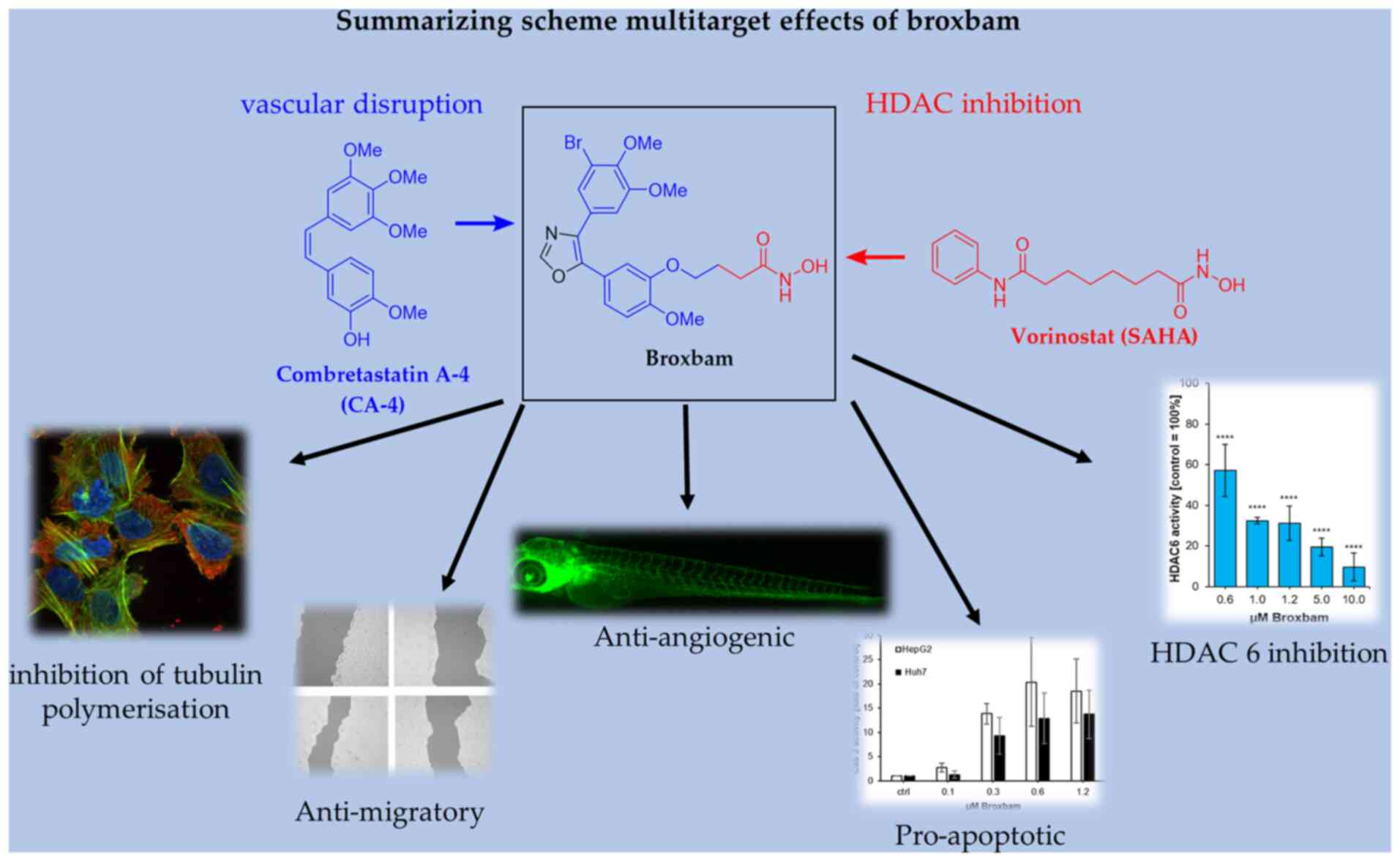
|
1
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer statistics, 2021. CA Cancer J Clin. 71:7–33. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mosconi S, Beretta GD, Labianca R, Zampino
MG, Gatta G and Heinemann V: Cholangiocarcinoma. Crit Rev Oncol
Hematol. 69:259–270. 2009. View Article : Google Scholar
|
|
3
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
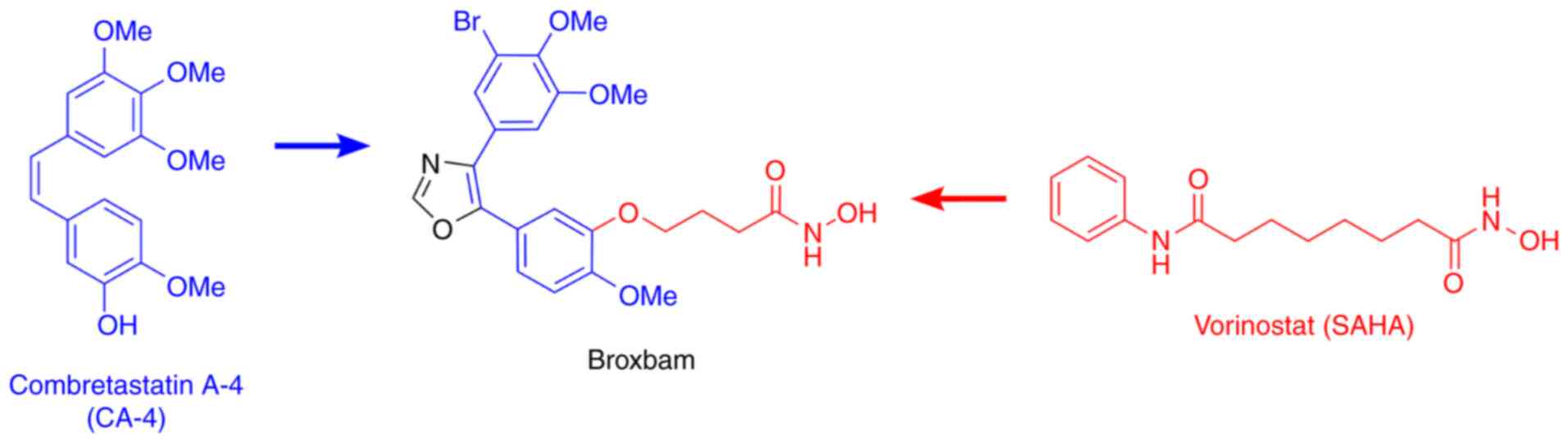
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Vogel A, Cervantes A, Chau I, Daniele B,
Llovet JM, Meyer T, Nault JC, Neumann U, Ricke J, Sangro B, et al:
Hepatocellular carcinoma: ESMO clinical practice guidelines for
diagnosis, treatment and follow-up. Ann Oncol. 29(Suppl 4):
iv238–iv255. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lim H, Ramjeesingh R, Liu D, Tam VC, Knox
JJ, Card PB and Meyers BM: Optimizing survival and the changing
landscape of targeted therapy for intermediate and advanced
hepatocellular carcinoma: A systematic review. J Natl Cancer Inst.
113:123–136. 2021. View Article : Google Scholar :
|
|
6
|
Li Y, Gao ZH and Qu XJ: The adverse
effects of sorafenib in patients with advanced cancers. Basic Clin
Pharmacol Toxicol. 116:216–221. 2015. View Article : Google Scholar
|
|
7
|
Tang W, Chen Z, Zhang W, Cheng Y, Zhang B,
Wu F, Wang Q, Wang S, Rong D, Reiter FP, et al: The mechanisms of
sorafenib resistance in hepatocellular carcinoma: Theoretical basis
and therapeutic aspects. Sig Transduct Target Ther. 5:872020.
View Article : Google Scholar
|
|
8
|
Doherty B, Nambudiri VE and Palmer WC:
Update on the diagnosis and treatment of cholangiocarcinoma. Curr
Gastroenterol Rep. 19:22017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Morise Z, Sugioka A, Tokoro T, Tanahashi
Y, Okabe Y, Kagawa T and Takeura C: Surgery and chemotherapy for
intrahepatic cholangiocarcinoma. World J Hepatol. 2:58–64. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Goehringer N, Biersack B, Peng Y, Schobert
R, Herling M, Ma A, Nitzsche B and Höpfner M: Anticancer activity
and mechanisms of action of new chimeric EGFR/HDAC-inhibitors. Int
J Mol Sci. 22:84322021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Biersack B, Polat S and Höpfner M:
Anticancer properties of chimeric HDAC and kinase inhibitors. Semin
Cancer Biol. Nov 12–2020.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Park SY and Kim JS: A short guide to
histone deacetylases including recent progress on class II enzymes.
Exp Mol Med. 52:204–212. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kaletsch A, Pinkerneil M, Hoffmann MJ,
Jaguva Vasudevan AA, Wang C, Hansen FK, Wiek C, Hanenberg H,
Gertzen C, Gohlke H, et al: Effects of novel HDAC inhibitors on
urothelial carcinoma cells. Clin Epigenetics. 10:1002018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gong D, Zeng Z, Yi F and Wu J: Inhibition
of histone deacetylase 11 promotes human liver cancer cell
apoptosis. Am J Transl Res. 11:983–990. 2019.PubMed/NCBI
|
|
15
|
Steinemann G, Dittmer A, Kuzyniak W,
Hoffmann B, Schrader M, Schobert R, Biersack B, Nitzsche B and
Höpfner M: Animacroxam, a novel dual-mode compound targeting
histone deacetylases and cytoskeletal integrity of testicular germ
cell cancer cells. Mol Cancer Ther. 16:2364–2374. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Manal M, Chandrasekar MJN, Gomathi Priya J
and Nanjan MJ: Inhibitors of histone deacetylase as antitumor
agents: A critical review. Bioorg Chem. 67:18–42. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Juan LJ, Shia WJ, Chen MH, Yang WM, Seto
E, Lin YS and Wu CW: Histone deacetylases specifically
down-regulate p53-dependent gene activation. J Biol Chem.
275:20436–20443. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mann BS, Johnson JR, Cohen MH, Justice R
and Pazdur R: FDA approval summary: Vorinostat for treatment of
advanced primary cutaneous T-cell lymphoma. Oncologist.
12:1247–1252. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Streubel G, Schrepfer S, Kallus H,
Parnitzke U, Wulff T, Hermann F, Borgmann M and Hamm S: Histone
deacetylase inhibitor resminostat in combination with sorafenib
counteracts platelet-mediated protumoral effects in hepatocellular
carcinoma. Sci Rep. 11:95872021. View Article : Google Scholar
|
|
20
|
Schmitt F, Gosch LC, Dittmer A, Rothemund
M, Mueller T, Schobert R, Biersack B, Volkamer A and Höpfner M:
Oxazole-bridged combretastatin A-4 derivatives with tethered
hydroxamic acids: Structure-activity relations of new inhibitors of
HDAC and/or tubulin function. Int J Mol Sci. 20:3832019. View Article : Google Scholar
|
|
21
|
Méndez-Callejas GM, Leone S, Tanzarella C
and Antoccia A: Combretastatin A-4 induces p53
mitochondrial-relocalisation independent-apoptosis in non-small
lung cancer cells. Cell Biol Int. 38:296–308. 2014. View Article : Google Scholar
|
|
22
|
Kim JH, Yoon EK, Chung HJ, Park SY, Hong
KM, Lee CH, Lee YS, Choi K, Yang Y, Kim K and Kim IH: p53
acetylation enhances Taxol-induced apoptosis in human cancer cells.
Apoptosis. 18:110–120. 2013. View Article : Google Scholar
|
|
23
|
Tron GC, Pirali T, Sorba G, Pagliai F,
Busacca S and Genazzani AA: Medicinal chemistry of combretastatin
A4: present and future directions. J Med Chem. 49:3033–3044. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hesham HM, Lasheen DS and Abouzid KAM:
Chimeric HDAC inhibitors: Comprehensive review on the HDAC-based
strategies developed to combat cancer. Med Res Rev. 38:2058–2109.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Griggs J, Metcalfe JC and Hesketh R:
Targeting tumour vasculature: The development of combretastatin A4.
Lancet Oncol. 2:82–87. 2001. View Article : Google Scholar
|
|
26
|
Gaspari R, Prota AE, Bargsten K, Cavalli A
and Steinmetz MO: Structural basis of cis-and trans-combretastatin
binding to tubulin. Chem. 2:102–113. 2017. View Article : Google Scholar
|
|
27
|
Schaller E, Ma A, Gosch LC, Klefenz A,
Schaller D, Goehringer N, Kaps L, Schuppan D, Volkamer A, Schobert
R, et al: New 3-Aryl-2-(2-thienyl)acrylonitriles with high activity
against hepatoma cells. Int J Mol Sci. 22:22432021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nwosu ZC, Battello N, Rothley M, Piorońska
W, Sitek B, Ebert MP, Hofmann U, Sleeman J, Wölfl S, Meyer C, et
al: Liver cancer cell lines distinctly mimic the metabolic gene
expression pattern of the corresponding human tumours. J Exp Clin
Cancer Res. 37:2112018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kasai F, Hirayama N, Ozawa M, Satoh M and
Kohara A: HuH-7 reference genome profile: Complex karyotype
composed of massive loss of heterozygosity. Hum Cell. 31:261–267.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nakabayashi H, Taketa K, Miyano K, Yamane
T and Sato J: Growth of human hepatoma cells lines with
differentiated functions in chemically defined medium. Cancer Res.
42:3858–3863. 1982.PubMed/NCBI
|
|
31
|
Monteil M, Migianu-Griffoni E,
Sainte-Catherine O, Di Benedetto M and Lecouvey M: Bisphosphonate
prodrugs: Synthesis and biological evaluation in HuH7
hepatocarcinoma cells. Eur J Med Chem. 77:56–64. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xu L, Hausmann M, Dietmaier W, Kellermeier
S, Pesch T, Stieber-Gunckel M, Lippert E, Klebl F and Rogler G:
Expression of growth factor receptors and targeting of EGFR in
cholangio-carcinoma cell lines. BMC Cancer. 10:3022010. View Article : Google Scholar
|
|
33
|
Jo JR, An S, Ghosh S, Nedumaran B and Kim
YD: Growth hormone promotes hepatic gluconeogenesis by enhancing
BTG2-YY1 signaling pathway. Sci Rep. 11:189992021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kuete V, Sandjo LP, Ouete JLN, Fouotsa H,
Wiench B and Efferth T: Cytotoxicity and modes of action of three
naturally occurring xanthones (8-hydroxycudraxanthone G, morusignin
I and cudraxanthone I) against sensitive and multidrug-resistant
cancer cell lines. Phytomedicine. 21:315–322. 2014. View Article : Google Scholar
|
|
35
|
Gillies RJ, Didier N and Denton M:
Determination of cell number in monolayer cultures. Anal Biochem.
159:109–113. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Korzeniewski C and Callewaert DM: An
enzyme-release assay for natural cytotoxicity. J Immunol Methods.
64:313–320. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Nitzsche B, Gloesenkamp C, Schrader M,
Ocker M, Preissner R, Lein M, Zakrzewicz A, Hoffmann B and Höpfner
M: Novel compounds with antiangiogenic and antiproliferative
potency for growth control of testicular germ cell tumours. Br J
Cancer. 103:18–28. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sivandzade F, Bhalerao A and Cucullo L:
Analysis of the mito-chondrial membrane potential using the
cationic JC-1 dye as a sensitive fluorescent probe. Bio Protoc.
9:e31282019. View Article : Google Scholar
|
|
39
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
40
|
Bobadilla AV, Arévalo J, Sarró E, Byrne
HM, Maini PK, Carraro T, Balocco S, Meseguer A and Alarcón T: In
vitro cell migration quantification method for scratch assays. J R
Soc Interface. 16:201807092019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Almeida VM, Bezerra MA Jr, Nascimento JC
and Amorim LMF: Anticancer drug screening: Standardization of in
vitro wound healing assay. J Bras Patol Med Lab. 55:606–619. 2019.
View Article : Google Scholar
|
|
42
|
Serbedzija GN, Flynn E and Willett CE:
Zebrafish angiogenesis: A new model for drug screening.
Angiogenesis. 3:353–359. 1999. View Article : Google Scholar
|
|
43
|
Warburg O: On the origin of cancer cells.
Science. 123:309–314. 1956. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Thorens B: GLUT2, glucose sensing and
glucose homeostasis. Diabetologia. 58:221–232. 2015. View Article : Google Scholar
|
|
45
|
Kaufmann SH and Earnshaw WC: Induction of
apoptosis by cancer chemotherapy. Exp Cell Res. 256:42–49. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Reers M, Smiley ST, Mottola-Hartshorn C,
Chen A, Lin M and Chen LB: Mitochondrial membrane potential
monitored by JC-1 dye. Methods Enzymol. 260:406–417. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kim R, Emi M and Tanabe K: Role of
mitochondria as the gardens of cell death. Cancer Chemother
Pharmacol. 57:545–553. 2006. View Article : Google Scholar
|
|
48
|
Fridman JS and Lowe SW: Control of
apoptosis by p53. Oncogene. 22:9030–9040. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kim R, Tanabe K, Uchida Y, Emi M, Inoue H
and Toge T: Current status of the molecular mechanisms of
anticancer drug-induced apoptosis. The contribution of
molecular-level analysis to cancer chemotherapy. Cancer Chemother
Pharmacol. 50:343–352. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Giaginis C, Damaskos C, Koutsounas I,
Zizi-Serbetzoglou A, Tsoukalas N, Patsouris E, Kouraklis G and
Theocharis S: Histone deacetylase (HDAC)-1, -2, -4 and -6
expression in human pancreatic adenocarcinoma: Associations with
clinicopathological parameters, tumor proliferative capacity and
patients' survival. BMC Gastroenterol. 15:1482015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Garmpis N, Damaskos C, Garmpi A,
Georgakopoulou VE, Sarantis P, Antoniou EA, Karamouzis MV, Nonni A,
Schizas D, Diamantis E, et al: Histone deacetylase inhibitors in
the treatment of hepatocellular carcinoma: Current evidence and
future opportunities. J Pers Med. 11:2232021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Scholzen T and Gerdes J: The Ki-67
protein: From the known and the unknown. J Cell Physiol.
182:311–322. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kim HR, Rahman FU, Kim KS, Kim EK, Cho SM,
Lee K, Moon OS, Seo YW, Yoon WK, Won YS, et al: Critical roles of
E2F3 in growth and musculo-skeletal phenotype in mice. Int J Med
Sci. 16:1557–1563. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Hubbert C, Guardiola A, Shao R, Kawaguchi
Y, Ito A, Nixon A, Yoshida M, Wang XF and Yao TP: HDAC6 is a
microtubule-associated deacetylase. Nature. 417:455–458. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Garcin C and Straube A: Microtubules in
cell migration. Essays Biochem. 63:509–520. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Schaks M, Giannone G and Rottner K: Actin
dynamics in cell migration. Essays Biochem. 63:483–495. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Norouzzadeh M, Kalikias Y, Mohammadpour Z,
Sharifi L and Mahmoudi M: Determining population doubling time and
the appropriate number of HepG2 cells for culturing in 6-well
plate. IJSBAR. 10:299–303. 2016.
|
|
58
|
Deng B, Luo Q, Halim A, Liu Q, Zhang B and
Song G: The antiangiogenesis role of histone deacetylase
inhibitors: Their potential application to tumor therapy and tissue
repair. DNA Cell Biol. 39:167–176. 2020. View Article : Google Scholar
|
|
59
|
Tomaszewska B, Muzolf M, Grabysa R and
Bodnar L: Cardiotoxicity of antiangiogenic drugs: Causes and
mechanisms. OncoReview. 11:12–18. 2021. View Article : Google Scholar
|
|
60
|
Oda K, Arakawa H, Tanaka T, Matsuda K,
Tanikawa C, Mori T, Nishimori H, Tamai K, Tokino T, Nakamura Y and
Taya Y: p53AIP1, a potential mediator of p53-dependent apoptosis,
and its regulation by Ser-46-phosphorylated p53. Cell. 102:849–862.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Matsuda K, Yoshida K, Taya Y, Nakamura K,
Nakamura Y and Arakawa H: p53AIP1 regulates the mitochondrial
apoptotic pathway. Cancer Res. 62:2883–2889. 2002.PubMed/NCBI
|
|
62
|
Porter AG and Jänicke RU: Emerging roles
of caspase-3 in apoptosis. Cell Death Differ. 6:99–104. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Lee YR and Park SY: P53 expression in
hepatocellular carcinoma: Influence on the radiotherapeutic
response of the hepatocellular carcinoma. Clin Mol Hepatol.
21:230–231. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Li T, Zhang C, Hassan S, Liu X, Song F,
Chen K, Zhang W and Yang J: Histone deacetylase 6 in cancer. J
Hematol Oncol. 11:1112018. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Aldana-Masangkay GI and Sakamoto KM: The
role of HDAC6 in cancer. J Biomed Biotechnol. 2011:8758242011.
View Article : Google Scholar
|
|
66
|
Gao Y, Hubbert CC, Lu J, Lee YS, Lee JY
and Yao TP: Histone deacetylase 6 regulates growth factor-induced
actin remodeling and endocytosis. Mol Cell Biol. 27:8637–8647.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Tran AD-A, Marmo TP, Salam AA, Che S,
Finkelstein E, Kabarriti R, Xenias HS, Mazitschek R, Hubbert C,
Kawaguchi Y, et al: HDAC6 deacetylation of tubulin modulates
dynamics of cellular adhesions. J Cell Sci. 120:1469–1479. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Enomoto T: Microtubule disruption induces
the formation of actin stress fibers and focal adhesions in
cultured cells: Possible involvement of the rho signal cascade.
Cell Struct Funct. 21:317–326. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Liu BP, Chrzanowska-Wodnicka M and
Burridge K: Microtubule depolymerization induces stress fibers,
focal adhesions, and DNA synthesis via the GTP-binding protein Rho.
Cell Adhes Commun. 5:249–255. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zhang C, Willett C and Fremgen T:
Zebrafish: An animal model for toxicological studies. Curr Protoc
Toxicol. 17:1.7.1–1.7. 2003.
|
|
71
|
Deroanne CF, Bonjean K, Servotte S, Devy
L, Colige A, Clausse N, Blacher S, Verdin E, Foidart JM, Nusgens BV
and Castronovo V: Histone deacetylases inhibitors as
anti-angiogenic agents altering vascular endothelial growth factor
signaling. Oncogene. 21:427–436. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Su M, Huang J, Liu S, Xiao Y, Qin X, Liu
J, Pi C, Luo T, Li J, Chen X and Luo Z: The anti-angiogenic effect
and novel mechanisms of action of Combretastatin A-4. Sci Rep.
6:281392016. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhou J, Cheng M, Wu M, Boriboun C, Jujo K,
Xu S, Zhao TC, Tang YL, Kishore R and Qin G: Contrasting roles of
E2F2 and E2F3 in endothelial cell growth and ischemic angiogenesis.
J Mol Cell Cardiol. 60:68–71. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Heo YA and Syed YY: Regorafenib: A review
in hepatocellular carcinoma. Drugs. 78:951–958. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Labeur TA, Hofsink Q, Takkenberg RB, van
Delden OM, Mathôt RAA, Schinner R, Malfertheiner P, Amthauer H,
Schütte K, Basu B, et al: The value of sorafenib trough levels in
patients with advanced hepatocellular carcinoma-a substudy of the
SORAMIC trial. Acta Oncol. 59:1028–1035. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Yuan H, Li AJ, Ma SL, Cui LJ, Wu B, Yin L
and Wu MC: Inhibition of autophagy significantly enhances
combination therapy with sorafenib and HDAC inhibitors for human
hepa-toma cells. World J Gastroenterol. 20:4953–4962. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Bitzer M, Horger M, Giannini EG, Ganten
TM, Wörns MA, Siveke JT, Dollinger MM, Gerken G, Scheulen ME, Wege
H, et al: Resminostat plus sorafenib as second-line therapy of
advanced hepatocellular carcinoma-The SHELTER study. J Hepatol.
65:280–288. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Freese K, Seitz T, Dietrich P, Lee SML,
Thasler WE, Bosserhoff A and Hellerbrand C: Histone deacetylase
expressions in hepato-cellular carcinoma and functional effects of
histone deacetylase inhibitors on liver cancer cells in vitro.
Cancers (Basel). 11:15872019. View Article : Google Scholar
|
|
79
|
Gordon SW, McGuire WP III, Shafer DA,
Sterling RK, Lee HM, Matherly SC, Roberts JD, Bose P, Tombes MB,
Shrader EE, et al: Phase I study of sorafenib and vorinostat in
advanced hepatocellular carcinoma. Am J Clin Oncol. 42:649–654.
2019. View Article : Google Scholar : PubMed/NCBI
|