According to recent statistics, prostate cancer
(PCa) continues to have a high incidence, and it is the most
prevalent type of cancer among adult males in developed nations; it
is also associated with the second-highest rate of cancer-related
mortality among males, and poses a significant global public health
burden (1). Age, race and a family
history of the disease have been identified as the main risk
factors for PCa (2). Studies have
demonstrated that a high-fat diet is a preventable factor linked to
disease progression (3).
In tumorigenesis and progression, in addition to
genetic mutations, epigenetic alterations and altered cellular
signaling pathways, tumor cells spontaneously generate metabolic
reprogramming, which is also a key feature that distinguishes them
from normal tissues (4,5). In 2017, Flavahan et al
(6) introduced the notion of
energy metabolic reprogramming, which includes three primary
abnormal metabolic pathways: Sugar, lipid and amino acid
metabolism. The Warburg effect is the lack of typical oxidative
phosphorylation in the mitochondria, and tumor cells continue to
generate energy mostly through anaerobic glycolysis, even in an
environment with abundant oxygen. The Warburg effect is the
biological enhancement of tumor cell proliferation, migration and
invasion caused by the absence of typical oxidative phosphorylation
in the mitochondria (7). Fatty
acid metabolism plays a crucial role in maintaining membrane
structure formation, the post-translational modification of
oncoproteins, energy storage and supply, and signaling in tumor
cells; it is also closely linked to the onset, progression, drug
resistance and recurrent metastasis of PCa. Lipid metabolism is one
of the main energy sources of tumor cells.
It has been established that obesity is a risk
factor for the development of PCa in the genetic context of
phosphatase and tensin homolog (PTEN) (8) deficiency and that a high-fat diet
causes lipid buildup that is sufficient to promote metastasis
(9). The prostate is surrounded by
periprostatic adipose tissue (PPAT), whose adipocytes secrete the
chemokine, CCL7. This chemokine moves from PPAT to the
periprostatic region and promotes the migration of tumor cells that
express CCR3 (10). PCa has an
active lipophagy mechanism (11).
Additionally, PCa exhibits a number of abnormalities in lipid
metabolism, including the increased uptake of circulating lipids
(12), the increased ab
initio synthesis of fatty acids and phospholipids (13), the increased transfer of fatty
acids from stromal adipocytes into PCa cells (14), and increased phospholipids
(15) in contrast to cholesterol
stored in cytoplasmic lipid droplets as cholesterol esters
(16).
In general, lipid metabolism is closely related to
the pathogenesis and progression of PCa, and its molecular
mechanisms and signaling pathways are relatively complex. the
present review focuses on certain new proteins that regulate lipid
metabolism and discusses the relevance of fatty acid, cholesterol
and phospholipid metabolic processes to PCa, as well as the
clinical application values of these novel markers in the diagnosis
and prognosis of PCa. The present review aims to lay the foundation
for subsequent studies on the role and mechanisms of lipid
metabolism in human cancer.
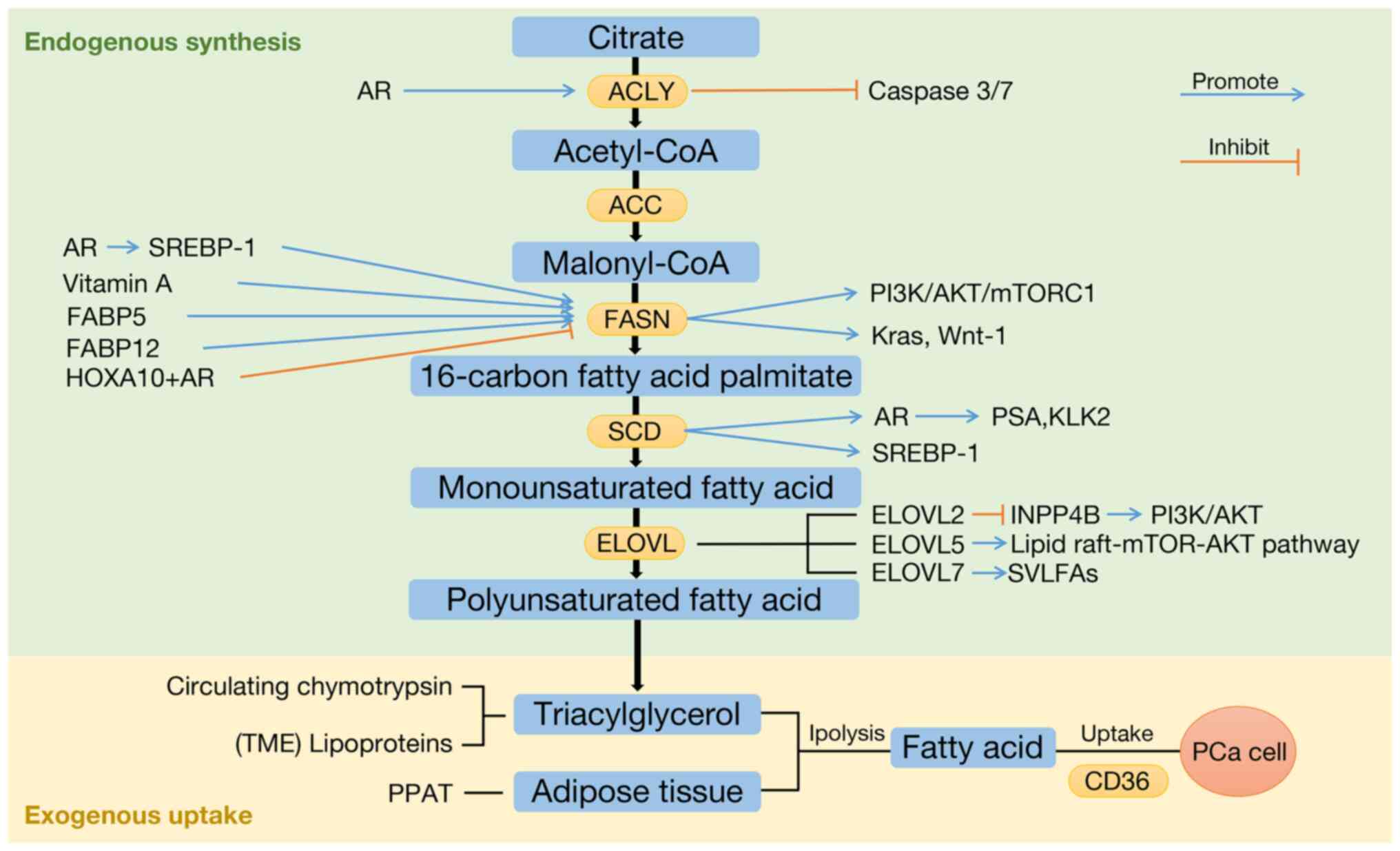
Fatty acid metabolism is a critical part of lipid
metabolism. The altered metabolic activity of fatty acids can
contribute to the malignant properties of cancer cells (17). Some fatty acids are produced by
adipose tissue lipolysis or triglyceride breakdown in circulating
chymotrypsin and lipoproteins. These exogenous fatty acids are the
preferred source of adenosine 5'-triphosphate production, membrane
biosynthesis, energy storage and the production of a wide range of
signaling molecules in the majority of non-tumor cells (Fig. 1) (18).
Adipose-derived fatty acids are also strongly linked
to cancer, and when adipocytes are located near tumor lesions,
their secretory products can influence disease progression, as
observed in ovarian and breast cancers (19,20).
The prostate is surrounded by PPAT, which provides a high
concentration of fatty acids and alters the prostate tumor
microenvironment (TME) (11).
Lipoproteins in the TME can also be taken up by cancer cells,
providing them with cholesterol and fatty acids (21). In PCa cells, fatty acid uptake
increases and serves as a direct raw material for substance
production. CD36 has been described as a carrier that mediates
fatty acid transport. It has been shown that CD36 knockdown in
cancer-susceptible PTEN−/− mice reduces fatty
acid uptake and the lipid abundance of oncogenic signals, thereby
inhibiting tumor progression (22). These data suggest that the
inhibition of fatty acid uptake may be a promising therapeutic
approach for the treatment of PCa.
The rate-limiting enzyme of FASN is ACC, which
catalyzes the conversion of acetyl-CoA to malonyl-CoA. ACC has two
isoforms: ACC1 (ACCα) and ACC2 (ACCβ) (27). The ACACA gene encodes ACC1, while
the ACACB gene encodes ACC2. ACC1 and ACACA have been linked to the
development and progression of numerous types of cancers, including
breast (28), ovarian (29), liver (30) and colon cancer (31). The ACACA gene is upregulated in PCa
tissues, and silencing the ACACA gene can inhibit PCa cell
proliferation and induce apoptosis (32). The expression of the ACACA gene is
associated with the local infiltration of tumor cells, lymph node
metastasis and distant metastasis, and its expression level is
positively associated with the Gleason score of PCa (33).
SCD is a key enzyme for the synthesis of
monounsaturated fatty acids. Human PCa has a higher ratio of
monounsaturated to saturated fatty acids than normal prostate
tissue, and SCD is highly expressed in PCa. SCD promotes the
proliferation of AR-positive LNCaP cells, increases
dihydrotestosterone (DHT)-induced AR transcriptional activity and
increases prostate-specific antigen (PSA) and kallikrein-related
peptidase 2 expression, and the inhibition of SCD attenuates the
progression of PCa (44,45). A previous study discovered that
inhibiting SCD activity with sterculic oil reduced LNCaP and PC3
cell viability, blocked the G2 cell cycle, decreased cell
proliferation and promoted apoptosis (46). Furthermore, SCD1 is a
transcriptional target of sterol regulatory element binding protein
(SREBP)1 that mediates the ferroptosis-suppressing activity of
SREBP1 by producing monounsaturated fatty acids, rendering
PI3K/AKT/mTOR pathway-mutant PCa cells ferroptosis-resistant
(47). SREBP1 is a central
transcription factor regulating lipid metabolism, and SREBPs are
discussed in more detail below.
Members of the ELOVL protein family are involved in
the production of polyunsaturated fatty acids, which are critical
components of cell membranes and are involved in the composition of
the cytoskeleton, the regulation of cell membrane fluidity,
signaling between the cell membrane and the cytoplasm, and the
regulation of ferroptosis. ELOVL is also linked to ferroptosis
regulation and plays a key role in tumorigenesis, development and
drug resistance (48-50).
ELOVL7 is required for the synthesis of saturated
very long-chain fatty acids and their derivatives, and its level is
negatively associated with the survival of patients with PCa.
ELOVL7 is promising as a novel molecular target for the treatment
or prevention of PCa (51).
Compared to normal tissues, PCa tissues have higher
levels of ELOVL2. A high expression of ELOVL2 indicates a better
prognosis for patients with PCa, while ELOVL2 expression is
adversely associated with the Gleason score. Low levels of ELOVL2
expression stimulate the growth of subcutaneous xenografts, colony
formation, migration, invasion and PCa by downregulating inositol
polyphosphate-4-phosphatase type II B to activate the PI3K/AKT
signaling pathway. EVOVL2 may thus be a predictive biomarker and
treatment target for PCa (52).
ELOVL5 is the main ELOVL expressed in primary and
metastatic PCa, and the level of ELOVL5 in PCa is higher than that
in non-malignant prostate tissue. When ELOVL5 is not present,
mitochondrial function is disrupted, and oxidative stress is
induced, inhibiting PCa cell proliferation and metastasis (15). When ELOVL5 is overexpressed, PCa
cells exhibit an increased resistance to enzalutamide treatment,
whereas ELOVL5 downregulation renderes PCa cells more responsive to
enzalutamide treatment. The lipid raft/mTOR/AKT pathway is
responsible for this effect, which has significant therapeutic
implications for CRPC (53).
SREBP overexpression is associated with aggressive
pathological features of human PCa (36). The set of genes activated by SREBP
transcription factors is significantly upregulated in PML and PTEN
double-null PCa (54). SREBPs are
spliced into two biologically active products, SREBP-1 and SREBP-2
(55).
SREBP-1 is a master transcription factor that
controls lipid metabolism. By binding to the FASN promoter region
and either directly or indirectly activating FASN transcription,
SREBP-1 can also participate in the transcriptional regulation of
AR and fatty acid synthesis. This in turn can promote PCa growth,
migration, invasion and depot resistance (36,56)
and is positively associated with the clinical Gleason
classification of human PCa (56).
SREBP-1/FASN inhibition reduces fatty acid levels and lipid droplet
accumulation in PCa cells (57).
In healthy cells, there are two proteins SREBP-1, SREBP-1a and
SREBP-1c, the latter of which is involved in controlling the ab
initio production of endogenous fatty acids (55). SREBP-1 is transcriptionally
regulated by microRNA-21 in vitro in cultured cells and
mouse models (58) and can
increase the production of reactive oxygen species, and NADPH
oxidase 5 expression induces oxidative stress in PCa cells
(56).
SREBP2 controls cholesterol production in healthy
cells, and studies have shown that PTEN/p53-deficient cancers
depend on cholesterol metabolism. Through the activation of SREBP2,
PTEN/p53 deficiency transcriptionally elevates squalene
epoxidase/monooxygenase (SQLE), and SQLE boosts cholesterol
production and encourages tumor cell proliferation and survival
(59).
In terms of medicine, the inhibition of SREBP is a
possible novel strategy for the treatment of PCa. The SREBP pathway
and AR signaling network can be targeted and blocked in
vitro and in vivo by lipoinhibitors, new SREBP
inhibitors, to decrease tumor development and distant metastasis
with anti-prostate cancer action (36,54).
In addition, drugs targeting the SREBP-2 pathway, such as
tocotrienols, which can lower cholesterol levels, are also
potential treatment options for PCa (60).
FABPs are multifunctional proteins that regulate
fatty acid uptake, transport, signal transduction and intracellular
lipid droplet formation (61) and
regulate metabolic and inflammatory pathways that have been closely
linked to obesity, metabolic diseases, cardiac dysfunction and
cancer (62).
All five FABP genes, FABP4, FABP5, FABP12, FABP9 and
FABP8, located on chromosome 8q21.13, are linked to PCa, and
patients with high Gleason scores have higher levels of all five
FABP mRNAs. Chromosome 8q21 is the most often amplified area in
metastatic PCa (63,64).
The promotion of PCa metastasis by FASN is largely
dependent on the expression of vitamin A and FABP5 in vivo
(74). By enhancing FA oxidation,
the tricarboxylic acid cycle and oxidative phosphorylation, FABP5
deficiency may rewire metabolic pathways and increase ATP
production by activating the PPAR signaling pathway (75). Vascular endothelial growth factor
(VEGF) expression is controlled by androgens in androgen-dependent
PCa cells; however, when PCa cells are no longer
androgen-dependent, this route is replaced by the FABP5/PPAR/VEGF
signaling pathway. Angiogenesis is another key element in the
evolution of PCa (76). In the
absence of FABP5, VEGF levels and microvessel density are reduced
(77), PCa cells are less
proliferative and invasive in vitro, and tumor growth and
metastasis are decreased in vivo (78).
In terms of other FABPs, FABP1 and FABP2 levels are
higher in PCa cells than in normal prostate cells, while FABP3
expression is lower (66).
HOX genes are a group of highly conserved genes that
control cell and tissue differentiation, morphogenesis, and
homeostasis during development. A number of human tumors have an
aberrant HOX gene expression (80,81).
Different HOX genes are linked to the development of various
prostate lobes, seminal vesicles and epididymis (80). HOXA10 is required for prostate
development and can affect PCa progression by regulating fatty acid
metabolism (82).
Human PCa frequently exhibits an abnormal HOXA10
expression, and HOXA10 levels are inversely associated with PCa
cell differentiation, the Gleason score and clinical stage
(83). It has been established
that HOXA10 plays a crucial role in regulating AR signaling and
adipogenesis. HOXA10 can bind AR to form a protein complex that
inhibits AR from entering the FASN gene promoter, hence suppressing
FASN gene transcription and preventing the progression of PCa to
CRPC. By contrast, the downregulation of HOXA10 activates the FASN
gene through AR signaling, promoting adipogenesis and the
progression of PCa (82).
Animals primarily catabolize fatty acids through
β-oxidation. Fatty acid oxidation has been shown to be crucial in
maintaining the malignant phenotype (84). In PCa cells, fatty acid β-oxidation
is one of the main forms of energy supply (85,86),
and the dysregulation of mitochondrial fatty acid β-oxidation
promotes the pathogenesis of PCa (87). The mitochondria are the primary
sites for fatty acid oxidation and sugar oxidative phosphorylation.
PLC is required for fatty acid binding to cell surface receptors,
and the PLC pathway increases intracellular calcium
(Ca2+) levels, which are involved in the
dephosphorylation of Drp-1 protein, resulting in Drp-1 protein
activation and mitochondrial division (88). The interaction between adipocytes
and cancer cells is thought to mediate the regulation of
mitochondrial dynamics via changes in intracellular
Ca2+, which affects fatty acid oxidation in
mitochondria.
The fatty acid β-oxidation and fatty acid uptake
capacity of cells are positively associated, whereas both are
negatively associated with the process of fatty acid ab
initio synthesis, indicating that fatty acid β-oxidation and
fatty acid carbon chain lengthening are two processes that are
mutually antagonistic (89). CPT1
is a key enzyme in fatty acid β-oxidation, and three CPT1 homologs
have been identified, namely CPT1A, CPT1B and CPT1C (90). CPT1A can regulate the entry of
fatty acids into mitochondria for β-oxidation (91), and the downregulation of CPT1A can
attenuate the growth of PCa cells (92). In PCa samples, CPT1B, a crucial
protein for rate limitation during mitochondrial β-oxidation, is
increased and linked to prognostically unfavorable outcomes. Cell
proliferation, S-phase distribution and invasive potential are all
affected by CPT1B silencing. Conversely, CPT1B overexpression
boosts AKT expression and phosphorylation, and markedly increases
enzalutamide resistance in C4-2R cells (93). CPT1C is also involved in fatty acid
catabolism and is a key gene for intracellular homeostasis
(90).
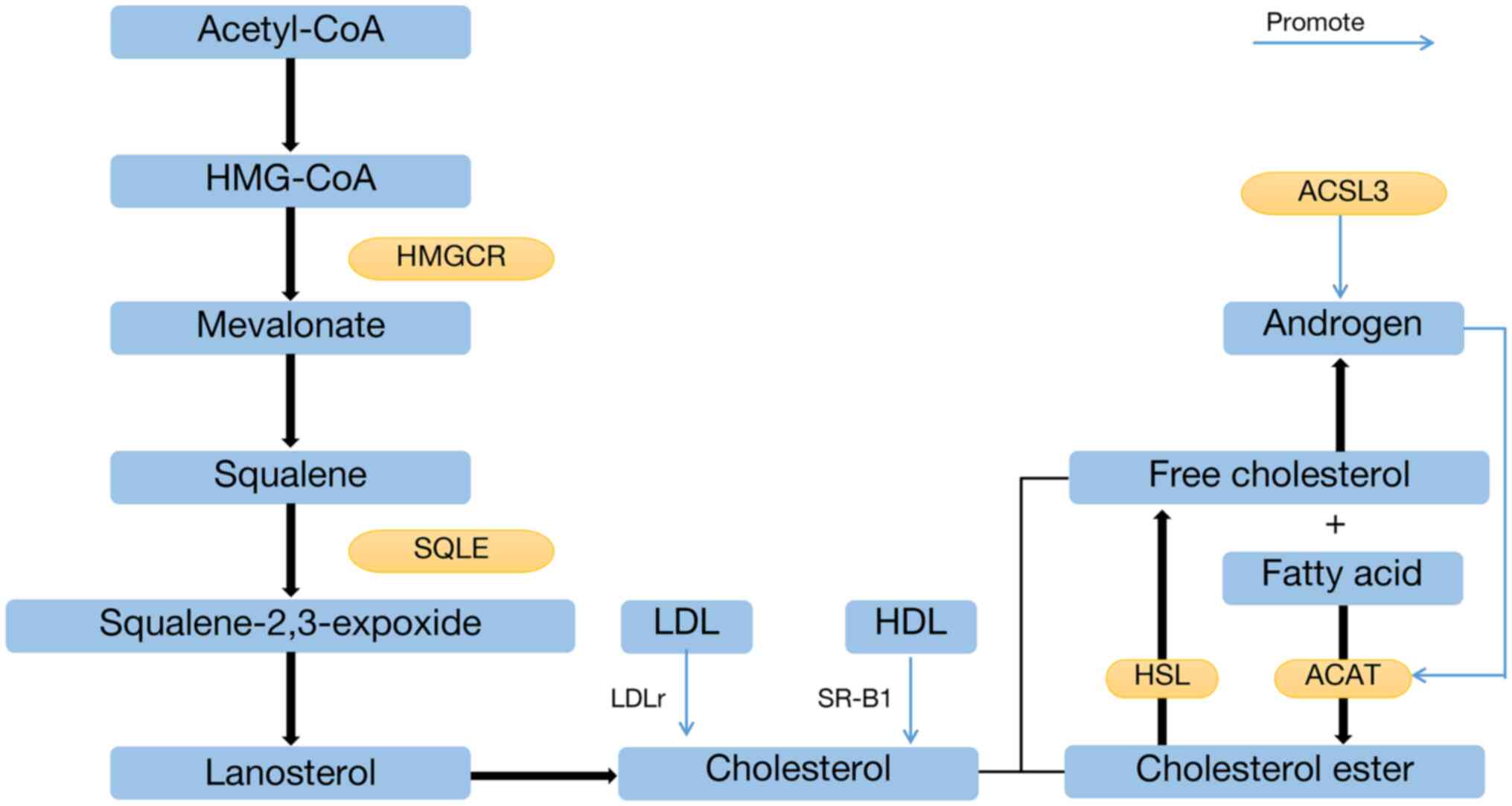
The most prevalent steroid substance in the body is
cholesterol, a steroidal lipid that makes up approximately one
third of the plasma membrane's lipid content and is crucial for
maintaining membrane fluidity and structural integrity (94). Additionally, cholesterol plays a
key role in the metabolism of certain types of cancer and is a
precursor to five key steroid hormones (glucocorticoid,
mineralocorticoid, androgen, estrogen and vitamin D) (95).
Cholesterol in normal cells of the body is usually
derived from two forms: Endogenous in situ synthesis and
exogenous uptake (96). Only the
liver and adipose tissue can normally generate cholesterol
endogenously; all other tissues and organs primarily obtain
cholesterol through external absorption. Low-density lipoprotein
(LDL) and high-density lipoprotein (HDL) are involved in the
exogenous uptake of cholesterol, which is primarily absorbed from
food through the small intestine. LDL is transported into the cells
by the LDL receptor (LDLr), and HDL is transported into the cells
by scavenger receptor class B type 1 (SR-B1) (Fig. 2) (97,98).
The key signaling pathways of human steroid-producing cells depend
heavily on SR-B1, which has been linked to the entry and exit of
cholesterol from cells (99).
The synthesis of cholesterol in the human body is
very complex, and cells use acetyl-CoA as a raw material for ab
initio synthesis, going through a total of ~30 steps, which can
be broadly divided into four stages: The production of
β-hydroxy-β-methylglutaryl-CoA, the production of mevalonate, the
production of squalene and the production of cholesterol.
Acetyl-CoA undergoes a series of stages consisting of
β-hydroxy-β-methylglutaryl-CoA reductase, 2,3-oxidosqualene
cyclase, squalene synthetase, SQLE, lanosterol synthase and
farnesyl-diphosphate synthase-mediated enzymatic reactions
(100,101), resulting in the synthesis of
cholesterol (Fig. 2).
The key signaling pathways of human
steroid-producing cells depend heavily on SR-B1, which has been
linked to the entry and exit of cholesterol from cells (102). PCa cells can regulate
intracellular cholesterol levels through various pathways, such as
endocytosis, exocytosis, synthesis and degradation, and certain
transcription factors play a critical role in this process. SREBP-2
promotes endogenous cholesterol synthesis and increases cholesterol
levels (103); SR-B1 promotes
cholesterol influx from lipoproteins in the body circulation into
cells (104) and is necessary to
drive cholesterol uptake required for steroidal and nonsteroidal
biological pathways (102), while
liver X receptor promotes cholesterol efflux (60) and down-regulates AKT survival
signaling in lipid rafts to induce the apoptosis of PCa cells
(105).
To avoid the cytotoxicity of high cholesterol
concentrations, the accumulation of cholesteryl esters (CEs) is
common in high-grade PCa and metastases (16). Free cholesterol and fatty acids
catalyzed by acyl coenzyme A-cholesterol acyltransferase (ACAT) can
generate non-toxic CEs stored intracellularly (Fig. 2) (84,106,107), which serve as precursors for
androgen synthesis and as raw materials for energy metabolism. In
the absence of androgens, CEs are catalyzed by hormone-sensitive
triglyceride lipase to produce free cholesterol (Fig. 2) (106,108), which in turn synthesizes
androgens and promotes the proliferation of PCa cells (99). CE translocation out of the cell is
mainly mediated by ATP-binding cassette subfamily A and ATP-binding
cassette subfamily G member 1 proteins (109,110).
In addition to being closely related to PTEN
deficiency and the activation of the PI3K/AKT/mTOR/SREBP signaling
pathway, the accumulation of CEs in PCa cells may be caused by the
anaerobic metabolism of tumor cells, which produces significant
amounts of raw materials for cholesterol synthesis and the
increased uptake of exogenous lipoproteins (16). The synthesis of CEs from free
cholesterol and long-chain fatty acids is only catalyzed by the
intracellular enzyme ACAT, and both of its major enzymes, ACAT1 and
ACAT2, are controlled by androgens (108,111). This alteration in lipid
metabolism is primarily caused by the activation of SREBP and LDLr,
which boosts the esterification of ACAT and raises the uptake of
foreign lipoproteins, increasing the accumulation of CEs in tumor
cells. Reduced levels of specific essential amino acids and
lipoproteins can prevent the buildup of CEs, which reduces tumor
cell proliferation and invasiveness and attenuates tumor growth
(16,107).
Cholesterol metabolism plays a critical role in cell
membrane generation and cell proliferation, and it is linked to
tumor cell survival and proliferation (112). As regards one of the primary
elements of cell membranes, lipid rafts, cholesterol plays a role
in the composition of these structures. Cholesterol also plays a
role in cell signaling and has the ability to control particular
proteins that are crucial for PCa cell growth and survival
(113,114). Membrane cholesterol
concentrations have a direct impact on the composition of signaling
proteins and the transmission of signals. On the one hand,
excessive cholesterol may cause abnormal signaling and alter the
lipid-protein balance. On the other hand, lower cholesterol levels
alter lipid raft integrity and prevent oncogenic signaling
complexes from functioning (114).
Lipid rafts have numerous functions and are involved
in the translocation and sorting of intracellular molecules, the
downregulation and recycling of receptors, and the targeted export
of proteins and lipids. Lipid rafts are also signaling platforms
that are associated with a large number of signaling proteins
(115), including epithelial
growth factor receptor (116),
other tyrosine kinase receptors (117), estrogen receptor (118), AR (119) and fatty acid synthase receptor
(120). The function of AKT is
controlled by the amount of cholesterol in the membrane, and the
AKT subpopulation within lipid rafts has different substrate
specificity from non-lipid raft AKT, is involved in cell growth and
survival, and controls crucial genes related to lipid and
cholesterol synthesis at the transcriptional level (121). Increased cholesterol inhibits
apoptosis in cells by working with lipid rafts. Additionally, since
cholesterol synthesis and the cell cycle are tightly connected,
reducing cholesterol levels with blockers may result in the
inhibition of cell growth, which in turn causes PCa cells to
undergo apoptosis (122).
Androgens are steroidal compounds, cholesterol is an
essential androgen synthesis precursor (95), and PCa cells can use cholesterol
and adrenal androgens to produce testosterone and DHT.
Intracellular cholesterol promotes PCa progression as a substrate
for de novo androgen synthesis and through the regulation of
AKT signaling (123). By
controlling steroidogenic genes, the acyl-CoA synthetase long-chain
family member 3 (ACSL3) participates in the synthesis of fatty
acyl-CoA ester, limits the catabolism of active androgens and
stimulates steroid biosynthesis in tumors, all of which contribute
to the progression of PCa. Since ACSL3 is substantially more highly
expressed in CRPC than in hormone-sensitive PCa, ACSL3 may be a
viable target for CRPC therapy (124).
Blood cholesterol levels are also associated with
the progression of PCa, and it has been shown that
hypercholesterolemia caused by high cholesterol and high-fat diets
increases the risk of developing PCa in older males (125-127) and may also promote the growth and
metastasis of PCa (128), whereas
low blood cholesterol levels slow the growth of PCa (123,129), and the risk of high-grade PCa is
lower in patients with lower blood cholesterol levels than in those
with high blood cholesterol levels (130). Statins have been shown to be
effective in reducing the risk of PCa (123,125,131,132).
The phospholipids in the plasma membrane mainly
include phosphatidylcholine (PC), phosphatidylethanolamine (PE),
phosphatidylinositol (PI), phosphatidylserine (PS) and
sphingomyelin (SM). Current research on phospholipids associated
with PCa is focused on diagnostic and prognostic aspects.
There is a significant difference between PE and
glycerophosphatidylethanolamine (and their ratios) between PCa and
benign prostatic hyperplasia (136), and in vitro 31P
nuclear magnetic resonance can be used to detect phospholipid
metabolites to assist in the diagnosis of PCa (137). Moreover, phospholipids can be
radiolabeled and developed as PET imaging agents for PCa (138). Gradients of changes in the
intensity of various lipids, such as PC, PS, PI, phosphatidic acid
and cardiolipin, are associated with increases in Gleason scores
(139). In CRPC, high levels of
sphingolipids are associated with a poor prognosis, and PC, SM and
ceramide are associated with a shorter survival (140).
Annexin (ANX) is a family of intracellular proteins
that binds membrane phospholipids using calcium ions and is
important in the diagnosis and prognostic monitoring of PCa. ANX1
expression is reduced in PCa and high-grade prostatic
intraepithelial neoplasia (PIN) and is associated with a lower
Gleason score (141). ANX2
expression decreases with the progression of PCa and is
significantly and negatively associated with the Gleason score
(142). Both benign prostatic
epithelium and high-grade PIN samples contain ANXA3; however, the
staining intensity is lower in PIN lesions than it is in benign
prostatic epithelium. In addition, ANXA3 is negatively associated
with the Gleason score and is a stand-alone poor prognostic marker
in PCa (143). ANXA7 is a
suppressor of tumorigenesis and metastasis in PCa, and activated
ANXA7 GTPase promotes apoptosis in PCa cells (144). Statins inhibit the proliferation,
migration and invasion of androgen-dependent PCa cells by
upregulating ANXA10 (145). The
combined detection of ANX and serum PSA levels may help to improve
the accuracy of the early diagnosis of PCa.
Phospholipids have been linked to PCa treatment in
addition to functioning as biomarkers. PS is normally anchored to
the inner side of the cell membrane; however, when complex
conditions, such as phospholipid translocator protease inactivation
in tumor cells occur, PS is translocated to the outer side of the
cell membrane (146). In response
to this feature of PS, a number of molecules targeting tumor PS
have been developed to provide new insight into for tumor therapy.
Mitochondrial 2,4-dienoyl-CoA reductase 1 (DECR1) can participate
in the dynamic balance of redox by controlling the balance between
saturated and unsaturated phospholipids. The knockdown of DECR1
induces endoplasmic reticulum stress and increases the sensitivity
of CRPC cells to ferroptosis, and DECR1 deficiency in vivo
impairs lipid metabolism and inhibits CRPC tumor growth (147). It has been reported that DECR1
and medication resistance in CRPC are closely connected. To shield
siRNA from enzymatic degradation and to increase siRNA release with
gene silencing and anticancer effects for the treatment of CRPC,
amphiphilic phospholipid peptide dendrimers can facilitate the
efficient delivery of siRNA targeting heat shock protein 27
(148).
The mechanism by which PCa develops into malignant
cancer is significantly influenced by abnormalities in lipid
metabolism. It is anticipated that several of the aforementioned
proteins that are involved in the control of lipid metabolism will
serve as novel targets for the detection and treatment of PCa.
These implications are summarized and presented in Table I.
Additionally, there is a strong association between
lipid concentrations and PCa. Lipoprotein A [Lp(a)] is a lipid
biomarker, and Lp(a) concentrations are associated with an
increased risk of developing PCa; lowering Lp(a) levels may prevent
the development of PCa (149). An
elevated Gleason score and likelihood of lymph node metastases are
both associated with elevated cholesterol levels (150). A higher likelihood of PCa
recurrence is also linked to elevated levels of triglycerides and
cholesterol (151). Extracellular
vesicles from PCa have been identified to mediate intercellular
communication with bone marrow cells in a cholesterol-dependent
manner, promoting PCa cell metastasis (152).
Statins are currently the primary treatment agents
for aberrant lipid metabolism; however, it is uncertain whether the
use of statins increases the risk of developing PCa (153). Some studies (154-157) have demonstrated that there is no
association between statin use and the risk of developing PCa,
while other studies (158-160)
have indicated that statin use reduces the risk of developing
advanced PCa and the risk of fatal PCa. Statin use has been
reported to attenuate the increased aggressiveness of PCa caused by
a high intake of saturated fats (161); combining statins has been shown
to improve PCa sensitivity to specific chemotherapeutic drugs
(162), and the use of statins
following ADT initiation has been found to improve the prognosis of
patients with PCa (163). From
another perspective, traditional PCa treatment drugs also
significantly affect lipid metabolism. Butler et al
(164) discovered that the
treatment of primary tumor 'explants' with the AR antagonist,
enzalutamide, caused significant changes in lipid subsets within
only 48 h. The targeted inhibition of tumor-associated lipid
profiles caused significantly decreased cell proliferation and
induced apoptosis in tissue explants (164).
Lipid metabolism is closely related to the
development and progression of PCa and has become a hot research
topic in recent years. The metabolism of fatty acids, cholesterol
and phospholipids, particularly key genes and proteins in various
lipid metabolism pathways, play a crucial role in the growth,
invasion, migration and malignant transformation of PCa and may
potentially be good diagnostic and prognostic markers or even
potential therapeutic targets (Table
I).
Abnormalities in lipid metabolism appear to be
associated with the malignant transformation and poor prognosis of
PCa. It may be possible to target abnormal lipid metabolic pathways
to modify this vulnerability. For example, in the setting of
increased obesity, PCa is more likely to progress to advanced-stage
or more aggressive PCa, and it may be possible to PCa or reduce the
risk of developing more malignant PCa by avoiding obesity and the
use of statins. PPAT provides high levels of fatty acids to alter
the TME and promote PCa progression, and altering the TME through
interactions between lipids and immune cells may be an option.
Although it may currently not be practical to replace conventional
PCa therapy with medications that target lipid metabolic pathways,
perhaps the use of lipid-targeted medications in combination with
other medications could increase the treatment efficacy and
prognosis of patients with PCa. At this time, it is unlikely that
lipid biomarkers will completely replace classical PCa markers;
however, they are more likely to be a component of a combined
diagnostic strategy that will help with diagnosis and prognosis.
However, further prospective studies are required for
validation.
Currently however, identifying strategies to
accurately assess the efficacy and biosafety of these drugs, to
mitigate the toxic side-effects of the combination of related drugs
and to determine the variability of lipid biomarkers in different
populations, as well as the elucidation of the mechanisms of action
of these key regulatory molecules are all urgent scientific
challenges that need to be addressed. It is considered that these
issues may soon be resolved, bringing new advances to the diagnosis
and treatment of PCa.
Not applicable.
ZZ and WW wrote and completed the manuscript and
abstract. PK and KF consulted the relevant literature and completed
the English revisions. CL TS, YS and XD completed the design of the
framework of the manuscript, and completed the figures and tables.
WL and ZT provided constructive feedback and guidance. WL completed
critical revisions and proofread the manuscript. All authors have
read and approved the final manuscript. Data authentication is not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present study was supported by grants from the National
Natural Science Foundation of China Youth Science Foundation
Project (no. 81802571), and the Zhejiang Medical and Health Science
and Technology Project (no. 2019RC039).
|
1
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer statistics, 2022. CA Cancer J Clin. 72:7–33. 2022.
|
|
2
|
Culp MB, Soerjomataram I, Efstathiou JA,
Bray F and Jemal A: Recent global patterns in prostate cancer
incidence and mortality rates. Eur Urol. 77:38–52. 2020.
|
|
3
|
Milliron BJ, Bruneau M, Obeid E, Gross L,
Bealin L, Smaltz C and Giri VN: Diet assessment among men
undergoing genetic counseling and genetic testing for inherited
prostate cancer: Exploring a teachable moment to support diet
intervention. Prostate. 79:778–783. 2019.
|
|
4
|
Hanahan D: Hallmarks of cancer: New
dimensions. Cancer Discov. 12:31–46. 2022.
|
|
5
|
Pavlova NN, Zhu J and Thompson CB: The
hallmarks of cancer metabolism: Still emerging. Cell Metab.
34:355–377. 2022.
|
|
6
|
Flavahan WA, Gaskell E and Bernstein BE:
Epigenetic plasticity and the hallmarks of cancer. Science.
357:eaal23802017.
|
|
7
|
Liberti MV and Locasale JW: The Warburg
effect: How does it benefit cancer cells? Trends Biochem Sci.
41:211–218. 2016.
|
|
8
|
Chaudagar K, Hieromnimon HM, Khurana R,
Labadie B, Hirz T, Mei S, Hasan R, Shafran J, Kelley A, Apostolov
E, et al: Reversal of lactate and PD-1-mediated macrophage
immunosuppression controls growth of PTEN/p53-deficient prostate
cancer. Clin Cancer Res. Mar;2023:2023.Epub ahead of print.
|
|
9
|
Poulose N, Amoroso F, Steele RE, Singh R,
Ong CW and Mills IG: Genetics of lipid metabolism in prostate
cancer. Nat Genet. 50:169–171. 2018.
|
|
10
|
Laurent V, Guérard A, Mazerolles C, Le
Gonidec S, Toulet A, Nieto L, Zaidi F, Majed B, Garandeau D,
Socrier Y, et al: Periprostatic adipocytes act as a driving force
for prostate cancer progression in obesity. Nat Commun.
7:102302016.
|
|
11
|
Fontaine A, Bellanger D, Guibon R, Bruyère
F, Brisson L and Fromont G: Lipophagy and prostate cancer:
Association with disease aggressiveness and proximity to
periprostatic adipose tissue. J Pathol. 255:166–176. 2021.
|
|
12
|
Kuemmerle NB, Rysman E, Lombardo PS,
Flanagan AJ, Lipe BC, Wells WA, Pettus JR, Froehlich HM, Memoli VA,
Morganelli PM, et al: Lipoprotein lipase links dietary fat to solid
tumor cell proliferation. Mol Cancer Ther. 10:427–436. 2011.
|
|
13
|
De Piano M, Manuelli V, Zadra G, Otte J,
Edqvist PD, Pontén F, Nowinski S, Niaouris A, Grigoriadis A, Loda
M, et al: Lipogenic signalling modulates prostate cancer cell
adhesion and migration via modification of Rho GTPases. Oncogene.
39:3666–3679. 2020.
|
|
14
|
Gazi E, Gardner P, Lockyer NP, Hart CA,
Brown MD and Clarke NW: Direct evidence of lipid translocation
between adipocytes and prostate cancer cells with imaging FTIR
microspectroscopy. J Lipid Res. 48:1846–1856. 2007.
|
|
15
|
Centenera MM, Scott JS, Machiels J, Nassar
ZD, Miller DC, Zinonos I, Dehairs J, Burvenich IJG, Zadra G, Chetta
PM, et al: ELOVL5 is a critical and targetable fatty acid elongase
in prostate cancer. Cancer Res. 81:1704–1718. 2021.
|
|
16
|
Yue S, Li J, Lee SY, Lee HJ, Shao T, Song
B, Cheng L, Masterson TA, Liu X, Ratliff TL and Cheng JX:
Cholesteryl ester accumulation induced by PTEN loss and PI3K/AKT
activation underlies human prostate cancer aggressiveness. Cell
Metab. 19:393–406. 2014.
|
|
17
|
DeBerardinis RJ and Chandel NS:
Fundamentals of cancer metabolism. Sci Adv. 2:e16002002016.
|
|
18
|
Weiss L, Hoffmann GE, Schreiber R, Andres
H, Fuchs E, Körber E and Kolb HJ: Fatty-acid biosynthesis in man, a
pathway of minor importance. Purification, optimal assay
conditions, and organ distribution of fatty-acid synthase. Biol
Chem Hoppe Seyler. 367:905–912. 1986.
|
|
19
|
Dirat B, Bochet L, Dabek M, Daviaud D,
Dauvillier S, Majed B, Wang YY, Meulle A, Salles B, Le Gonidec S,
et al: Cancer-associated adipocytes exhibit an activated phenotype
and contribute to breast cancer invasion. Cancer Res. 71:2455–2465.
2011.
|
|
20
|
Nieman KM, Kenny HA, Penicka CV, Ladanyi
A, Buell-Gutbrod R, Zillhardt MR, Romero IL, Carey MS, Mills GB,
Hotamisligil GS, et al: Adipocytes promote ovarian cancer
metastasis and provide energy for rapid tumor growth. Nat Med.
17:1498–1503. 2011.
|
|
21
|
Gomaraschi M: Role of lipoproteins in the
microenvironment of hormone-dependent cancers. Trends Endocrinol
Metab. 31:256–268. 2020.
|
|
22
|
Watt MJ, Clark AK, Selth LA, Haynes VR,
Lister N, Rebello R, Porter LH, Niranjan B, Whitby ST, Lo J, et al:
Suppressing fatty acid uptake has therapeutic effects in
preclinical models of prostate cancer. Sci Transl Med.
11:eaau57582019.
|
|
23
|
Batchuluun B, Pinkosky SL and Steinberg
GR: Lipogenesis inhibitors: Therapeutic opportunities and
challenges. Nat Rev Drug Discov. 21:283–305. 2022.
|
|
24
|
Zhao S, Torres A, Henry RA, Trefely S,
Wallace M, Lee JV, Carrer A, Sengupta A, Campbell SL, Kuo YM, et
al: ATP-citrate lyase controls a glucose-to-acetate metabolic
switch. Cell Rep. 17:1037–1052. 2016.
|
|
25
|
Galbraith L, Leung HY and Ahmad I: Lipid
pathway deregulation in advanced prostate cancer. Pharmacol Res.
131:177–184. 2018.
|
|
26
|
Gao Y, Islam MS, Tian J, Lui VWY and Xiao
D: Inactivation of ATP citrate lyase by Cucurbitacin B: A bioactive
compound from cucumber, inhibits prostate cancer growth. Cancer
Lett. 349:15–25. 2014.
|
|
27
|
Hunkeler M, Hagmann A, Stuttfeld E, Chami
M, Guri Y, Stahlberg H and Maier T: Structural basis for regulation
of human acetyl-CoA carboxylase. Nature. 558:470–474. 2018.
|
|
28
|
Rios Garcia M, Steinbauer B, Srivastava K,
Singhal M, Mattijssen F, Maida A, Christian S, Hess-Stumpp H,
Augustin HG, Müller-Decker K, et al: Acetyl-CoA carboxylase
1-dependent protein acetylation controls breast cancer metastasis
and recurrence. Cell Metab. 26:842–855.e5. 2017.
|
|
29
|
Zhao S, Cheng L, Shi Y, Li J, Yun Q and
Yang H: MIEF2 reprograms lipid metabolism to drive progression of
ovarian cancer through ROS/AKT/mTOR signaling pathway. Cell Death
Dis. 12:182021.
|
|
30
|
Lally JSV, Ghoshal S, DePeralta DK, Moaven
O, Wei L, Masia R, Erstad DJ, Fujiwara N, Leong V, Houde VP, et al:
Inhibition of acetyl-CoA carboxylase by phosphorylation or the
inhibitor ND-654 suppresses lipogenesis and hepatocellular
carcinoma. Cell Metab. 29:174–182.e5. 2019.
|
|
31
|
Raimondo S, Saieva L, Cristaldi M,
Monteleone F, Fontana S and Alessandro R: Label-free quantitative
proteomic profiling of colon cancer cells identifies acetyl-CoA
carboxylase alpha as antitumor target of Citrus limon-derived
nanovesicles. J Proteomics. 173:1–11. 2018.
|
|
32
|
Brusselmans K, De Schrijver E, Verhoeven G
and Swinnen JV: RNA interference-mediated silencing of the
acetyl-CoA-carboxylase-alpha gene induces growth inhibition and
apoptosis of prostate cancer cells. Cancer Res. 65:6719–6725.
2005.
|
|
33
|
O'Malley J, Kumar R, Kuzmin AN, Pliss A,
Yadav N, Balachandar S, Wang J, Attwood K, Prasad PN and Chandra D:
Lipid quantification by Raman microspectroscopy as a potential
biomarker in prostate cancer. Cancer Lett. 397:52–60. 2017.
|
|
34
|
Nguyen PL, Ma J, Chavarro JE, Freedman ML,
Lis R, Fedele G, Fiore C, Qiu W, Fiorentino M, Finn S, et al: Fatty
acid synthase polymorphisms, tumor expression, body mass index,
prostate cancer risk, and survival. J Clin Oncol. 28:3958–3964.
2010.
|
|
35
|
Rossi S, Graner E, Febbo P, Weinstein L,
Bhattacharya N, Onody T, Bubley G, Balk S and Loda M: Fatty acid
synthase expression defines distinct molecular signatures in
prostate cancer. Mol Cancer Res. 1:707–715. 2003.
|
|
36
|
Li X, Chen YT, Hu P and Huang WC:
Fatostatin displays high antitumor activity in prostate cancer by
blocking SREBP-regulated metabolic pathways and androgen receptor
signaling. Mol Cancer Ther. 13:855–866. 2014.
|
|
37
|
Migita T, Ruiz S, Fornari A, Fiorentino M,
Priolo C, Zadra G, Inazuka F, Grisanzio C, Palescandolo E, Shin E,
et al: Fatty acid synthase: A metabolic enzyme and candidate
oncogene in prostate cancer. J Natl Cancer Inst. 101:519–532.
2009.
|
|
38
|
Wu X, Dong Z, Wang CJ, Barlow LJ, Fako V,
Serrano MA, Zou Y, Liu JY and Zhang JT: FASN regulates cellular
response to genotoxic treatments by increasing PARP-1 expression
and DNA repair activity via NF-κB and SP1. Proc Natl Acad Sci USA.
113:E6965–E6973. 2016.
|
|
39
|
Ventura R, Mordec K, Waszczuk J, Wang Z,
Lai J, Fridlib M, Buckley D, Kemble G and Heuer TS: Inhibition of
de novo palmitate synthesis by fatty acid synthase induces
apoptosis in tumor cells by remodeling cell membranes, inhibiting
signaling pathways, and reprogramming gene expression.
EBioMedicine. 2:808–824. 2015.
|
|
40
|
Zadra G, Ribeiro CF, Chetta P, Ho Y,
Cacciatore S, Gao X, Syamala S, Bango C, Photopoulos C, Huang Y, et
al: Inhibition of de novo lipogenesis targets androgen receptor
signaling in castration-resistant prostate cancer. Proc Natl Acad
Sci USA. 116:631–640. 2019.
|
|
41
|
Agostini M, Almeida LY, Bastos DC, Ortega
RM, Moreira FS, Seguin F, Zecchin KG, Raposo HF, Oliveira HC,
Amoêdo ND, et al: The fatty acid synthase inhibitor orlistat
reduces the growth and metastasis of orthotopic tongue oral
squamous cell carcinomas. Mol Cancer Ther. 13:585–595. 2014.
|
|
42
|
Menendez JA, Vellon L and Lupu R:
Antitumoral actions of the anti-obesity drug orlistat (XenicalTM)
in breast cancer cells: Blockade of cell cycle progression,
promotion of apoptotic cell death and PEA3-mediated transcriptional
repression of Her2/neu (erbB-2) oncogene. Ann Oncol. 16:1253–1267.
2005.
|
|
43
|
Wright C, Iyer AKV, Kaushik V and Azad N:
Anti-tumorigenic potential of a novel orlistat-AICAR combination in
prostate cancer cells. J Cell Biochem. 118:3834–3845. 2017.
|
|
44
|
Fritz V, Benfodda Z, Rodier G, Henriquet
C, Iborra F, Avancès C, Allory Y, de la Taille A, Culine S, Blancou
H, et al: Abrogation of de novo lipogenesis by stearoyl-CoA
desaturase 1 inhibition interferes with oncogenic signaling and
blocks prostate cancer progression in mice. Mol Cancer Ther.
9:1740–1754. 2010.
|
|
45
|
Kim SJ, Choi H, Park SS, Chang C and Kim
E: Stearoyl CoA desaturase (SCD) facilitates proliferation of
prostate cancer cells through enhancement of androgen receptor
transactivation. Mol Cells. 31:371–377. 2011.
|
|
46
|
Contreras-López EF, Cruz-Hernández CD,
Cortés-Ramírez SA, Ramírez-Higuera A, Peña-Montes C,
Rodríguez-Dorantes M and Oliart-Ros RM: Inhibition of stearoyl-CoA
desaturase by sterculic oil reduces proliferation and induces
apoptosis in prostate cancer cell lines. Nutr Cancer. 74:1308–1321.
2022.
|
|
47
|
Yi J, Zhu J, Wu J, Thompson CB and Jiang
X: Oncogenic activation of PI3K-AKT-mTOR signaling suppresses
ferroptosis via SREBP-mediated lipogenesis. Proc Natl Acad Sci USA.
117:31189–31197. 2020.
|
|
48
|
Berquin IM, Edwards IJ, Kridel SJ and Chen
YQ: Polyunsaturated fatty acid metabolism in prostate cancer.
Cancer Metastasis Rev. 30:295–309. 2011.
|
|
49
|
Staubach S and Hanisch FG: Lipid rafts:
Signaling and sorting platforms of cells and their roles in cancer.
Expert Rev Proteomics. 8:263–277. 2011.
|
|
50
|
Yang WS, Kim KJ, Gaschler MM, Patel M,
Shchepinov MS and Stockwell BR: Peroxidation of polyunsaturated
fatty acids by lipoxygenases drives ferroptosis. Proc Natl Acad Sci
USA. 113:E4966–E4975. 2016.
|
|
51
|
Tamura K, Makino A, Hullin-Matsuda F,
Kobayashi T, Furihata M, Chung S, Ashida S, Miki T, Fujioka T,
Shuin T, et al: Novel lipogenic enzyme ELOVL7 is involved in
prostate cancer growth through saturated long-chain fatty acid
metabolism. Cancer Res. 69:8133–8140. 2009.
|
|
52
|
Hu T, Zhang H, Du Y, Luo S, Yang X, Zhang
H, Feng J, Chen X, Tu X, Wang C and Zhang Y: ELOVL2 restrains cell
proliferation, migration, and invasion of prostate cancer via
regulation of the tumor suppressor INPP4B. Cell Signal.
96:1103732022.
|
|
53
|
Xu H, Li S, Sun Y, Xu L, Hong X, Wang Z
and Hu H: ELOVL5-mediated long chain fatty acid elongation
contributes to enzalutamide resistance of prostate cancer. Cancers
(Basel). 13:39572021.
|
|
54
|
Chen M, Zhang J, Sampieri K, Clohessy JG,
Mendez L, Gonzalez-Billalabeitia E, Liu XS, Lee YR, Fung J, Katon
JM, et al: An aberrant SREBP-dependent lipogenic program promotes
metastatic prostate cancer. Nat Genet. 50:206–218. 2018.
|
|
55
|
Lee HJ, Jung YH, Choi GE, Ko SH, Lee SJ,
Lee SH and Han HJ: BNIP3 induction by hypoxia stimulates
FASN-dependent free fatty acid production enhancing therapeutic
potential of umbilical cord blood-derived human mesenchymal stem
cells. Redox Biol. 13:426–443. 2017.
|
|
56
|
Huang WC, Li X, Liu J, Lin J and Chung
LWK: Activation of androgen receptor, lipogenesis, and oxidative
stress converged by SREBP-1 is responsible for regulating growth
and progression of prostate cancer cells. Mol Cancer Res.
10:133–142. 2012.
|
|
57
|
Hsieh PF, Jiang WP, Basavaraj P, Huang SY,
Ruangsai P, Wu JB, Huang GJ and Huang WC: Cell suspension culture
extract of Eriobotrya japonica attenuates growth and induces
apoptosis in prostate cancer cells via targeting
SREBP-1/FASN-driven metabolism and AR. Phytomedicine.
93:1538062021.
|
|
58
|
Kanagasabai T, Li G, Shen TH, Gladoun N,
Castillo-Martin M, Celada SI, Xie Y, Brown LK, Mark ZA, Ochieng J,
et al: MicroRNA-21 deficiency suppresses prostate cancer
progression through downregulation of the IRS1-SREBP-1 signaling
pathway. Cancer Lett. 525:46–54. 2022.
|
|
59
|
Shangguan X, Ma Z, Yu M, Ding J, Xue W and
Qi J: Squalene epoxidase metabolic dependency is a targetable
vulnerability in castration-resistant prostate cancer. Cancer Res.
82:3032–3044. 2022.
|
|
60
|
Krycer JR, Phan L and Brown AJ: A key
regulator of cholesterol homoeostasis, SREBP-2, can be targeted in
prostate cancer cells with natural products. Biochem J.
446:191–201. 2012.
|
|
61
|
Storch J and Corsico B: The emerging
functions and mechanisms of mammalian fatty acid-binding proteins.
Annu Rev Nutr. 28:73–95. 2008.
|
|
62
|
Li B, Hao J, Zeng J and Sauter ER:
SnapShot: FABP functions. Cell. 182:1066–1066.e1. 2020.
|
|
63
|
Cher ML, Bova GS, Moore DH, Small EJ,
Carroll PR, Pin SS, Epstein JI, Isaacs WB and Jensen RH: Genetic
alterations in untreated metastases and androgen-independent
prostate cancer detected by comparative genomic hybridization and
allelotyping. Cancer Res. 56:3091–3102. 1996.
|
|
64
|
Taylor BS, Schultz N, Hieronymus H,
Gopalan A, Xiao Y, Carver BS, Arora VK, Kaushik P, Cerami E, Reva
B, et al: Integrative genomic profiling of human prostate cancer.
Cancer Cell. 18:11–22. 2010.
|
|
65
|
Yang PB, Hou PP, Liu FY, Hong WB, Chen HZ,
Sun XY, Li P, Zhang Y, Ju CY, Luo LJ, et al: Blocking PPARγ
interaction facilitates Nur77 interdiction of fatty acid uptake and
suppresses breast cancer progression. Proc Natl Acad Sci USA.
117:27412–27422. 2020.
|
|
66
|
Liu RZ and Godbout R: An amplified fatty
acid-binding protein gene cluster in prostate cancer: Emerging
roles in lipid metabolism and metastasis. Cancers (Basel).
12:38232020.
|
|
67
|
Guo Y, Liu Y, Zhao S, Xu W, Li Y, Zhao P,
Wang D, Cheng H, Ke Y and Zhang X: Oxidative stress-induced FABP5
S-glutathionylation protects against acute lung injury by
suppressing inflammation in macrophages. Nat Commun.
12:70942021.
|
|
68
|
Montaigne D, Butruille L and Staels B:
PPAR control of metabolism and cardiovascular functions. Nat Rev
Cardiol. 18:809–823. 2021.
|
|
69
|
Aguilar-Recarte D, Barroso E, Gumà A,
Pizarro-Delgado J, Peña L, Ruart M, Palomer X, Wahli W and
Vázquez-Carrera M: GDF15 mediates the metabolic effects of PPARβ/δ
by activating AMPK. Cell Rep. 36:1095012021.
|
|
70
|
Ahmad I, Mui E, Galbraith L, Patel R, Tan
EH, Salji M, Rust AG, Repiscak P, Hedley A, Markert E, et al:
Sleeping Beauty screen reveals Pparg activation in metastatic
prostate cancer. Proc Natl Acad Sci USA. 113:8290–8295. 2016.
|
|
71
|
Prentice KJ, Saksi J, Robertson LT, Lee
GY, Inouye KE, Eguchi K, Lee A, Cakici O, Otterbeck E, Cedillo P,
et al: A hormone complex of FABP4 and nucleoside kinases regulates
islet function. Nature. 600:720–726. 2021.
|
|
72
|
Massillo C, Dalton GN, Porretti J, Scalise
GD, Farré PL, Piccioni F, Secchiari F, Pascuali N, Clyne C, Gardner
K, et al: CTBP1/CYP19A1/estradiol axis together with adipose tissue
impacts over prostate cancer growth associated to metabolic
syndrome. Int J Cancer. 144:1115–1127. 2019.
|
|
73
|
Harraz AM, Atia N, Ismail A, Shady A, Farg
H, Gabr H, Fouda M, Abol-Enein H and Abdel-Aziz AF: Evaluation of
serum fatty acid binding protein-4 (FABP-4) as a novel biomarker to
predict biopsy outcomes in prostate biopsy naïve patients. Int Urol
Nephrol. 52:1483–1490. 2020.
|
|
74
|
Carbonetti G, Wilpshaar T, Kroonen J,
Studholme K, Converso C, d'Oelsnitz S and Kaczocha M: FABP5
coordinates lipid signaling that promotes prostate cancer
metastasis. Sci Rep. 9:189442019.
|
|
75
|
Hou Y, Wei D, Zhang Z, Guo H, Li S, Zhang
J, Zhang P, Zhang L and Zhao Y: FABP5 controls macrophage
alternative activation and allergic asthma by selectively
programming long-chain unsaturated fatty acid metabolism. Cell Rep.
41:1116682022.
|
|
76
|
Carbonetti G, Converso C, Clement T, Wang
C, Trotman LC, Ojima I and Kaczocha M: Docetaxel/cabazitaxel and
fatty acid binding protein 5 inhibitors produce synergistic
inhibition of prostate cancer growth. Prostate. 80:88–98. 2020.
|
|
77
|
Adamson J, Morgan EA, Beesley C, Mei Y,
Foster CS, Fujii H, Rudland PS, Smith PH and Ke Y: High-level
expression of cutaneous fatty acid-binding protein in prostatic
carcinomas and its effect on tumorigenicity. Oncogene.
22:2739–2749. 2003.
|
|
78
|
O'Sullivan SE and Kaczocha M: FABP5 as a
novel molecular target in prostate cancer. Drug Discov Today. Sep
20–2020.Epub ahead of print.
|
|
79
|
Liu RZ, Choi WS, Jain S, Dinakaran D, Xu
X, Han WH, Yang XH, Glubrecht DD, Moore RB, Lemieux H and Godbout
R: The FABP12/PPARγ pathway promotes metastatic transformation by
inducing epithelial-to-mesenchymal transition and lipid-derived
energy production in prostate cancer cells. Mol Oncol.
14:3100–3120. 2020.
|
|
80
|
Javed S and Langley SEM: Importance of HOX
genes in normal prostate gland formation, prostate cancer
development and its early detection. BJU Int. 113:535–540.
2014.
|
|
81
|
Xu F, Shangguan X, Pan J, Yue Z, Shen K,
Ji Y, Zhang W, Zhu Y, Sha J, Wang Y, et al: HOXD13 suppresses
prostate cancer metastasis and BMP4-induced epithelial-mesenchymal
transition by inhibiting SMAD1. Int J Cancer. 148:3060–3070.
2021.
|
|
82
|
Long Z, Li Y, Gan Y, Zhao D, Wang G, Xie
N, Lovnicki JM, Fazli L, Cao Q, Chen K and Dong X: Roles of the
HOXA10 gene during castrate-resistant prostate cancer progression.
Endocr Relat Cancer. 26:279–292. 2019.
|
|
83
|
Hatanaka Y, de Velasco MA, Oki T, Shimizu
N, Nozawa M, Yoshimura K, Yoshikawa K, Nishio K and Uemura H:
HOXA10 expression profiling in prostate cancer. Prostate.
79:554–563. 2019.
|
|
84
|
Carracedo A, Cantley LC and Pandolfi PP:
Cancer metabolism: Fatty acid oxidation in the limelight. Nat Rev
Cancer. 13:227–232. 2013.
|
|
85
|
Liu Y: Fatty acid oxidation is a dominant
bioenergetic pathway in prostate cancer. Prostate Cancer Prostatic
Dis. 9:230–234. 2006.
|
|
86
|
Tennakoon JB, Shi Y, Han JJ, Tsouko E,
White MA, Burns AR, Zhang A, Xia X, Ilkayeva OR, Xin L, et al:
Androgens regulate prostate cancer cell growth via an
AMPK-PGC-1α-mediated metabolic switch. Oncogene. 33:5251–5261.
2014.
|
|
87
|
Bramhecha YM, Guérard KP, Audet-Walsh É,
Rouzbeh S, Kassem O, Pernet E, Scarlata E, Hamel L, Brimo F,
Divangahi M, et al: Fatty acid oxidation enzyme Δ3, Δ2-enoyl-CoA
isomerase 1 (ECI1) drives aggressive tumor phenotype and predicts
poor clinical outcome in prostate cancer patients. Oncogene.
41:2798–2810. 2022.
|
|
88
|
Bravo-Sagua R, Parra V, López-Crisosto C,
Díaz P, Quest AF and Lavandero S: Calcium transport and signaling
in mitochondria. Compr Physiol. 7:623–634. 2017.
|
|
89
|
Butler LM, Centenera MM and Swinnen JV:
Androgen control of lipid metabolism in prostate cancer: Novel
insights and future applications. Endocr Relat Cancer.
23:R219–R227. 2016.
|
|
90
|
Adamopoulos PG, Kontos CK and Scorilas A:
Molecular characterization, genomic structure and expression
analysis of a gene (CATL1/CPT1C) encoding a third member of the
human carnitine acyltransferase family. Genomics. May 22–2019.Epub
ahead of print.
|
|
91
|
Fondevila MF, Fernandez U, Heras V,
Parracho T, Gonzalez-Rellan MJ, Novoa E, Porteiro B, Alonso C, Mayo
R, da Silva Lima N, et al: Inhibition of carnitine
palmitoyltransferase 1A in hepatic stellate cells protects against
fibrosis. J Hepatol. 77:15–28. 2022.
|
|
92
|
Joshi M, Stoykova GE, Salzmann-Sullivan M,
Dzieciatkowska M, Liebman LN, Deep G and Schlaepfer IR: CPT1A
supports castration-resistant prostate cancer in androgen-deprived
conditions. Cells. 8:11152019.
|
|
93
|
Abudurexiti M, Zhu W, Wang Y, Wang J, Xu
W, Huang Y, Zhu Y, Shi G, Zhang H, Zhu Y, et al: Targeting CPT1B as
a potential therapeutic strategy in castration-resistant and
enzalutamide-resistant prostate cancer. Prostate. 80:950–961.
2020.
|
|
94
|
Simons K and Ikonen E: How cells handle
cholesterol. Science. 290:1721–1726. 2000.
|
|
95
|
El-Kenawi A, Dominguez-Viqueira W, Liu M,
Awasthi S, Abraham-Miranda J, Keske A, Steiner KK, Noel L, Serna
AN, Dhillon J, et al: Macrophage-derived cholesterol contributes to
therapeutic resistance in prostate cancer. Cancer Res.
81:5477–5490. 2021.
|
|
96
|
Garcia-Bermudez J, Baudrier L, Bayraktar
EC, Shen Y, La K, Guarecuco R, Yucel B, Fiore D, Tavora B,
Freinkman E, et al: Squalene accumulation in cholesterol
auxotrophic lymphomas prevents oxidative cell death. Nature.
567:118–122. 2019.
|
|
97
|
Revilla G, Cedó L, Tondo M, Moral A, Pérez
JI, Corcoy R, Lerma E, Fuste V, Reddy ST, Blanco-Vaca F, et al:
LDL, HDL and endocrine-related cancer: From pathogenic mechanisms
to therapies. Semin Cancer Biol. 73:134–157. 2021.
|
|
98
|
Shen WJ, Azhar S and Kraemer FB: SR-B1: A
unique multifunctional receptor for cholesterol influx and efflux.
Annu Rev Physiol. 80:95–116. 2018.
|
|
99
|
Leon CG, Locke JA, Adomat HH, Etinger SL,
Twiddy AL, Neumann RD, Nelson CC, Guns ES and Wasan KM: Alterations
in cholesterol regulation contribute to the production of
intratumoral androgens during progression to castration-resistant
prostate cancer in a mouse xenograft model. Prostate. 70:390–400.
2010.
|
|
100
|
Ediriweera MK: Use of cholesterol
metabolism for anti-cancer strategies. Drug Discov Today. 27:Sep
7–2022.Epub ahead of print.
|
|
101
|
Hilvo M, Denkert C, Lehtinen L, Müller B,
Brockmöller S, Seppänen-Laakso T, Budczies J, Bucher E, Yetukuri L,
Castillo S, et al: Novel theranostic opportunities offered by
characterization of altered membrane lipid metabolism in breast
cancer progression. Cancer Res. 71:3236–3245. 2011.
|
|
102
|
Gordon JA, Noble JW, Midha A, Derakhshan
F, Wang G, Adomat HH, Tomlinson Guns ES, Lin YY, Ren S, Collins CC,
et al: Upregulation of scavenger receptor B1 is required for
steroidogenic and nonsteroidogenic cholesterol metabolism in
prostate cancer. Cancer Res. 79:3320–3331. 2019.
|
|
103
|
Wang B, Rong X, Palladino END, Wang J,
Fogelman AM, Martín MG, Alrefai WA, Ford DA and Tontonoz P:
Phospholipid remodeling and cholesterol availability regulate
intestinal stemness and tumorigenesis. Cell Stem Cell.
22:206–220.e4. 2018.
|
|
104
|
Pandey M, Cuddihy G, Gordon JA, Cox ME and
Wasan KM: Inhibition of scavenger receptor class B type 1 (SR-B1)
expression and activity as a potential novel target to disrupt
cholesterol availability in castration-resistant prostate cancer.
Pharmaceutics. 13:15092021.
|
|
105
|
Pommier AJC, Alves G, Viennois E, Bernard
S, Communal Y, Sion B, Marceau G, Damon C, Mouzat K, Caira F, et
al: Liver X receptor activation downregulates AKT survival
signaling in lipid rafts and induces apoptosis of prostate cancer
cells. Oncogene. 29:2712–2723. 2010.
|
|
106
|
Locke JA, Wasan KM, Nelson CC, Guns ES and
Leon CG: Androgen-mediated cholesterol metabolism in LNCaP and PC-3
cell lines is regulated through two different isoforms of
acyl-coenzyme A: Cholesterol acyltransferase (ACAT). Prostate.
68:20–33. 2008.
|
|
107
|
Raftopulos NL, Washaya TC, Niederprüm A,
Egert A, Hakeem-Sanni MF, Varney B, Aishah A, Georgieva ML, Olsson
E, Dos Santos DZ, et al: Prostate cancer cell proliferation is
influenced by LDL-cholesterol availability and cholesteryl ester
turnover. Cancer Metab. 10:12022.
|
|
108
|
Cai C and Balk SP: Intratumoral androgen
biosynthesis in prostate cancer pathogenesis and response to
therapy. Endocr Relat Cancer. 18:R175–R182. 2011.
|
|
109
|
An T, Zhang X, Li H, Dou L, Huang X, Man
Y, Zhang X, Shen T, Li G, Li J and Tang W: GPR120 facilitates
cholesterol efflux in macrophages through activation of AMPK
signaling pathway. FEBS J. 287:5080–5095. 2020.
|
|
110
|
Hu YW, Yang JY, Ma X, Chen ZP, Hu YR, Zhao
JY, Li SF, Qiu YR, Lu JB, Wang YC, et al: A
lincRNA-DYNLRB2-2/GPR119/GLP-1R/ABCA1-dependent signal transduction
pathway is essential for the regulation of cholesterol homeostasis.
J Lipid Res. 55:681–697. 2014.
|
|
111
|
Locke JA, Nelson CC, Adomat HH, Hendy SC,
Gleave ME and Guns ES: Steroidogenesis inhibitors alter but do not
eliminate androgen synthesis mechanisms during progression to
castration-resistance in LNCaP prostate xenografts. J Steroid
Biochem Mol Biol. 115:126–136. 2009.
|
|
112
|
Stopsack KH, Gerke TA, Sinnott JA, Penney
KL, Tyekucheva S, Sesso HD, Andersson SO, Andrén O, Cerhan JR,
Giovannucci EL, et al: Cholesterol metabolism and prostate cancer
lethality. Cancer Res. 76:4785–4790. 2016.
|
|
113
|
Dambal S, Alfaqih M, Sanders S, Maravilla
E, Ramirez-Torres A, Galvan GC, Reis-Sobreiro M, Rotinen M, Driver
LM, Behrove MS, et al: 27-Hydroxycholesterol impairs plasma
membrane lipid raft signaling as evidenced by inhibition of
IL6-JAK-STAT3 signaling in prostate cancer cells. Mol Cancer Res.
18:671–684. 2020.
|
|
114
|
Zhuang L, Lin J, Lu ML, Solomon KR and
Freeman MR: Cholesterol-rich lipid rafts mediate akt-regulated
survival in prostate cancer cells. Cancer Res. 62:2227–2231.
2002.
|
|
115
|
Freeman MR and Solomon KR: Cholesterol and
prostate cancer. J Cell Biochem. 91:54–69. 2004.
|
|
116
|
Jiang S, Wang X, Song D, Liu X, Gu Y, Xu
Z, Wang X, Zhang X, Ye Q, Tong Z, et al: Cholesterol induces
epithelial-to-mesenchymal transition of prostate cancer cells by
suppressing degradation of EGFR through APMAP. Cancer Res.
79:3063–3075. 2019.
|
|
117
|
Waugh MG, Lawson D and Hsuan JJ: Epidermal
growth factor receptor activation is localized within low-buoyant
density, non-caveolar membrane domains. Biochem J. 337:591–597.
1999.
|
|
118
|
Márquez DC, Chen HW, Curran EM, Welshons
WV and Pietras RJ: Estrogen receptors in membrane lipid rafts and
signal transduction in breast cancer. Mol Cell Endocrinol.
246:91–100. 2006.
|
|
119
|
Freeman MR, Cinar B and Lu ML: Membrane
rafts as potential sites of nongenomic hormonal signaling in
prostate cancer. Trends Endocrinol Metab. 16:273–279. 2005.
|
|
120
|
Legembre P, Daburon S, Moreau P, Ichas F,
de Giorgi F, Moreau JF and Taupin JL: Amplification of Fas-mediated
apoptosis in type II cells via microdomain recruitment. Mol Cell
Biol. 25:6811–6820. 2005.
|
|
121
|
Priolo C, Pyne S, Rose J, Regan ER, Zadra
G, Photopoulos C, Cacciatore S, Schultz D, Scaglia N, McDunn J, et
al: AKT1 and MYC induce distinctive metabolic fingerprints in human
prostate cancer. Cancer Res. 74:7198–7204. 2014.
|
|
122
|
Dong P, Flores J, Pelton K and Solomon KR:
Prohibitin is a cholesterol-sensitive regulator of cell cycle
transit. J Cell Biochem. 111:1367–1374. 2010.
|
|
123
|
Lee BH, Taylor MG, Robinet P, Smith JD,
Schweitzer J, Sehayek E, Falzarano SM, Magi-Galluzzi C, Klein EA
and Ting AH: Dysregulation of cholesterol homeostasis in human
prostate cancer through loss of ABCA1. Cancer Res. 73:1211–1218.
2013.
|
|
124
|
Migita T, Takayama KI, Urano T, Obinata D,
Ikeda K, Soga T, Takahashi S and Inoue S: ACSL3 promotes
intratumoral steroidogenesis in prostate cancer cells. Cancer Sci.
108:2011–2021. 2017.
|
|
125
|
Locke JA, Guns ES, Lehman ML, Ettinger S,
Zoubeidi A, Lubik A, Margiotti K, Fazli L, Adomat H, Wasan KM, et
al: Arachidonic acid activation of intratumoral steroid synthesis
during prostate cancer progression to castration resistance.
Prostate. 70:239–251. 2010.
|
|
126
|
Masko EM, Allott EH and Freedland SJ: The
relationship between nutrition and prostate cancer: Is more always
better? Eur Urol. 63:810–820. 2013.
|
|
127
|
Pardo JC, Ruiz de Porras V, Gil J, Font A,
Puig-Domingo M and Jordà M: Lipid metabolism and epigenetics
crosstalk in prostate cancer. Nutrients. 14:8512022.
|
|
128
|
Allott EH and Freedland SJ: Words of
wisdom. Re: Impact of circulating cholesterol levels on growth and
intratumoral androgen concentration of prostate tumors. Eur Urol.
63:178–179. 2013.
|
|
129
|
Zhuang L, Kim J, Adam RM, Solomon KR and
Freeman MR: Cholesterol targeting alters lipid raft composition and
cell survival in prostate cancer cells and xenografts. J Clin
Invest. 115:959–968. 2005.
|
|
130
|
Platz EA, Till C, Goodman PJ, Parnes HL,
Figg WD, Albanes D, Neuhouser ML, Klein EA, Thompson IM Jr and
Kristal AR: Men with low serum cholesterol have a lower risk of
high-grade prostate cancer in the placebo arm of the prostate
cancer prevention trial. Cancer Epidemiol Biomarkers Prev.
18:2807–2813. 2009.
|
|
131
|
Magura L, Blanchard R, Hope B, Beal JR,
Schwartz GG and Sahmoun AE: Hypercholesterolemia and prostate
cancer: A hospital-based case-control study. Cancer Causes Control.
19:1259–1266. 2008.
|
|
132
|
Platz EA, Leitzmann MF, Visvanathan K,
Rimm EB, Stampfer MJ, Willett WC and Giovannucci E: Statin drugs
and risk of advanced prostate cancer. J Natl Cancer Inst.
98:1819–1825. 2006.
|
|
133
|
Sherwin RW, Wentworth DN, Cutler JA,
Hulley SB, Kuller LH and Stamler J: Serum cholesterol levels and
cancer mortality in 361,662 men screened for the multiple risk
factor intervention trial. JAMA. 257:943–948. 1987.
|
|
134
|
Ribas V, García-Ruiz C and Fernández-Checa
JC: Mitochondria, cholesterol and cancer cell metabolism. Clin
Transl Med. 5:222016.
|
|
135
|
Solomon KR and Freeman MR: The complex
interplay between cholesterol and prostate malignancy. Urol Clin
North Am. 38:243–259. 2011.
|
|
136
|
Komoroski RA, Holder JC, Pappas AA and
Finkbeiner AE: 31P NMR of phospholipid metabolites in prostate
cancer and benign prostatic hyperplasia. Magn Reson Med.
65:911–913. 2011.
|
|
137
|
Philips BWJ, van Uden MJ, Rietsch SHG,
Orzada S and Scheenen TWJ: A multitransmit external body array
combined with a 1 H and 31 P endorectal coil
to enable a multiparametric and multimetabolic MRI examination of
the prostate at 7T. Med Phys. 46:3893–3905. 2019.
|
|
138
|
Kwan KH, Burvenich IJG, Centenera MM, Goh
YW, Rigopoulos A, Dehairs J, Swinnen JV, Raj GV, Hoy AJ, Butler LM,
et al: Synthesis and fluorine-18 radiolabeling of a phospholipid as
a PET imaging agent for prostate cancer. Nucl Med Biol. 93:37–45.
2021.
|
|
139
|
Randall EC, Zadra G, Chetta P, Lopez BGC,
Syamala S, Basu SS, Agar JN, Loda M, Tempany CM, Fennessy FM and
Agar NYR: Molecular characterization of prostate cancer with
associated gleason score using mass spectrometry imaging. Mol
Cancer Res. 17:1155–1165. 2019.
|
|
140
|
Lin HM, Mahon KL, Weir JM, Mundra PA,
Spielman C, Briscoe K, Gurney H, Mallesara G, Marx G, Stockler MR,
et al: A distinct plasma lipid signature associated with poor
prognosis in castration-resistant prostate cancer. Int J Cancer.
141:2112–2120. 2017.
|
|
141
|
Patton KT, Chen HM, Joseph L and Yang XJ:
Decreased annexin I expression in prostatic adenocarcinoma and in
high-grade prostatic intraepithelial neoplasia. Histopathology.
47:597–601. 2005.
|
|
142
|
Beyene DA, Naab TJ, Kanarek NF, Apprey V,
Esnakula A, Khan FA, Blackman MR, Brown CA and Hudson TS:
Differential expression of annexin 2, SPINK1, and Hsp60 predict
progression of prostate cancer through bifurcated WHO Gleason score
categories in African American men. Prostate. 78:801–811. 2018.
|
|
143
|
Köllermann J, Schlomm T, Bang H, Schwall
GP, von Eichel-Streiber C, Simon R, Schostak M, Huland H, Berg W,
Sauter G, et al: Expression and prognostic relevance of annexin A3
in prostate cancer. Eur Urol. 54:1314–1323. 2008.
|
|
144
|
Liu S, Li X, Lin Z, Su L, Yan S, Zhao B
and Miao J: SEC-induced activation of ANXA7 GTPase suppresses
prostate cancer metastasis. Cancer Lett. 416:11–23. 2018.
|
|
145
|
Miyazawa Y, Sekine Y, Kato H, Furuya Y,
Koike H and Suzuki K: Simvastatin up-regulates annexin A10 that can
inhibit the proliferation, migration, and invasion in
androgen-independent human prostate cancer cells. Prostate.
77:337–349. 2017.
|
|
146
|
Sharma B and Kanwar SS:
Phosphatidylserine: A cancer cell targeting biomarker. Semin Cancer
Biol. 52:17–25. 2018.
|
|
147
|
Blomme A, Ford CA, Mui E, Patel R, Ntala
C, Jamieson LE, Planque M, McGregor GH, Peixoto P, Hervouet E, et
al: 2,4-dienoyl-CoA reductase regulates lipid homeostasis in
treatment-resistant prostate cancer. Nat Commun. 11:25082020.
|
|
148
|
Dong Y, Chen Y, Zhu D, Shi K, Ma C, Zhang
W, Rocchi P, Jiang L and Liu X: Self-assembly of amphiphilic
phospholipid peptide dendrimer-based nanovectors for effective
delivery of siRNA therapeutics in prostate cancer therapy. J
Control Release. 322:416–425. 2020.
|
|
149
|
Ioannidou A, Watts EL, Perez-Cornago A,
Platz EA, Mills IG, Key TJ, Travis RC; PRACTICAL consortium, CRUK,
C3; et al: The relationship between lipoprotein A and other lipids
with prostate cancer risk: A multivariable Mendelian randomisation
study. PLoS Med. 19:e10038592022.
|
|
150
|
Liu Y, Wang Y, Hao S, Qin Y and Wu Y:
Knockdown of sterol O-acyltransferase 1 (SOAT1) suppresses
SCD1-mediated lipogenesis and cancer procession in prostate cancer.
Prostaglandins Other Lipid Mediat. 153:1065372021.
|
|
151
|
Freedland SJ, Howard LE, Ngo A,
Ramirez-Torres A, Csizmadi I, Cheng S, Mack A and Lin PH: Low
carbohydrate diets and estimated cardiovascular and metabolic
syndrome risk in prostate cancer. J Urol. 206:1411–1419. 2021.
|
|
152
|
Henrich SE, McMahon KM, Plebanek MP,
Calvert AE, Feliciano TJ, Parrish S, Tavora F, Mega A, De Souza A,
Carneiro BA and Thaxton CS: Prostate cancer extracellular vesicles
mediate intercellular communication with bone marrow cells and
promote metastasis in a cholesterol-dependent manner. J Extracell
Vesicles. 10:e120422020.
|
|
153
|
Scheinberg T, Mak B, Butler L, Selth L and
Horvath LG: Targeting lipid metabolism in metastatic prostate
cancer. Ther Adv Med Oncol. 15:175883592311528392023.
|
|
154
|
Kaulanjan K, Lavigne D, Saad F,
Karakiewicz PI, Flammia RS, Kluth LA, Mandel P, Chun FK, Taussky D
and Hoeh B: Impact of statin use on localized prostate cancer
outcomes after radiation therapy: Long-term follow-up. Cancers
(Basel). 14:36062022.
|
|
155
|
Chan JM, Litwack-Harrison S, Bauer SR,
Daniels NA, Wilt TJ, Shannon J and Bauer DC: Statin use and risk of
prostate cancer in the prospective osteoporotic fractures in men
(MrOS) study. Cancer Epidemiol Biomarkers Prev. 21:1886–1888.
2012.
|
|
156
|
Jacobs EJ, Newton CC, Thun MJ and Gapstur
SM: Long-term use of cholesterol-lowering drugs and cancer
incidence in a large United States cohort. Cancer Res.
71:1763–1771. 2011.
|
|
157
|
Alfaqih MA, Allott EH, Hamilton RJ,
Freeman MR and Freedland SJ: The current evidence on statin use and
prostate cancer prevention: are we there yet? Nat Rev Urol.
14:107–119. 2017.
|
|
158
|
Kafka M, Gruber R, Neuwirt H, Ladurner M
and Eder IE: Long-term treatment with simvastatin leads to reduced
migration capacity of prostate cancer cells. Biomedicines.
11:292022.
|
|
159
|
Mak B, Lin HM, Duong T, Mahon KL, Joshua
AM, Stockler MR, Gurney H, Parnis F, Zhang A, Scheinberg T, et al:
Modulation of plasma lipidomic profiles in metastatic
castration-resistant prostate cancer by simvastatin. Cancers
(Basel). 14:47922022.
|
|
160
|
Joshua AM, Armstrong A, Crumbaker M, Scher
HI, de Bono J, Tombal B, Hussain M, Sternberg CN, Gillessen S,
Carles J, et al: Statin and metformin use and outcomes in patients
with castration-resistant prostate cancer treated with
enzalutamide: A meta-analysis of AFFIRM, PREVAIL and PROSPER. Eur J
Cancer. 170:285–295. 2022.
|
|
161
|
Allott EH, Arab L, Su LJ, Farnan L,
Fontham ET, Mohler JL, Bensen JT and Steck SE: Saturated fat intake
and prostate cancer aggressiveness: Results from the
population-based North Carolina-Louisiana prostate cancer project.
Prostate Cancer Prostatic Dis. 20:48–54. 2017.
|
|
162
|
Iannelli F, Roca MS, Lombardi R,
Ciardiello C, Grumetti L, De Rienzo S, Moccia T, Vitagliano C,
Sorice A, Costantini S, et al: Synergistic antitumor interaction of
valproic acid and simvastatin sensitizes prostate cancer to
docetaxel by targeting CSCs compartment via YAP inhibition. J Exp
Clin Cancer Res. 39:2132020.
|
|
163
|
Peltomaa AI, Raittinen P, Talala K, Taari
K, Tammela TLJ, Auvinen A and Murtola TJ: Prostate cancer prognosis
after initiation of androgen deprivation therapy among statin
users. A population-based cohort study. Prostate Cancer Prostatic
Dis. 24:917–924. 2021.
|
|
164
|
Butler LM, Mah CY, Machiels J, Vincent AD,
Irani S, Mutuku SM, Spotbeen X, Bagadi M, Waltregny D, Moldovan M,
et al: Lipidomic profiling of clinical prostate cancer reveals
targetable alterations in membrane lipid composition. Cancer Res.
81:4981–4993. 2021.
|
|
165
|
Rhodes DR, Yu J, Shanker K, Deshpande N,
Varambally R, Ghosh D, Barrette T, Pandey A and Chinnaiyan AM:
ONCOMINE: A cancer microarray database and integrated data-mining
platform. Neoplasia. 6:1–6. 2004.
|