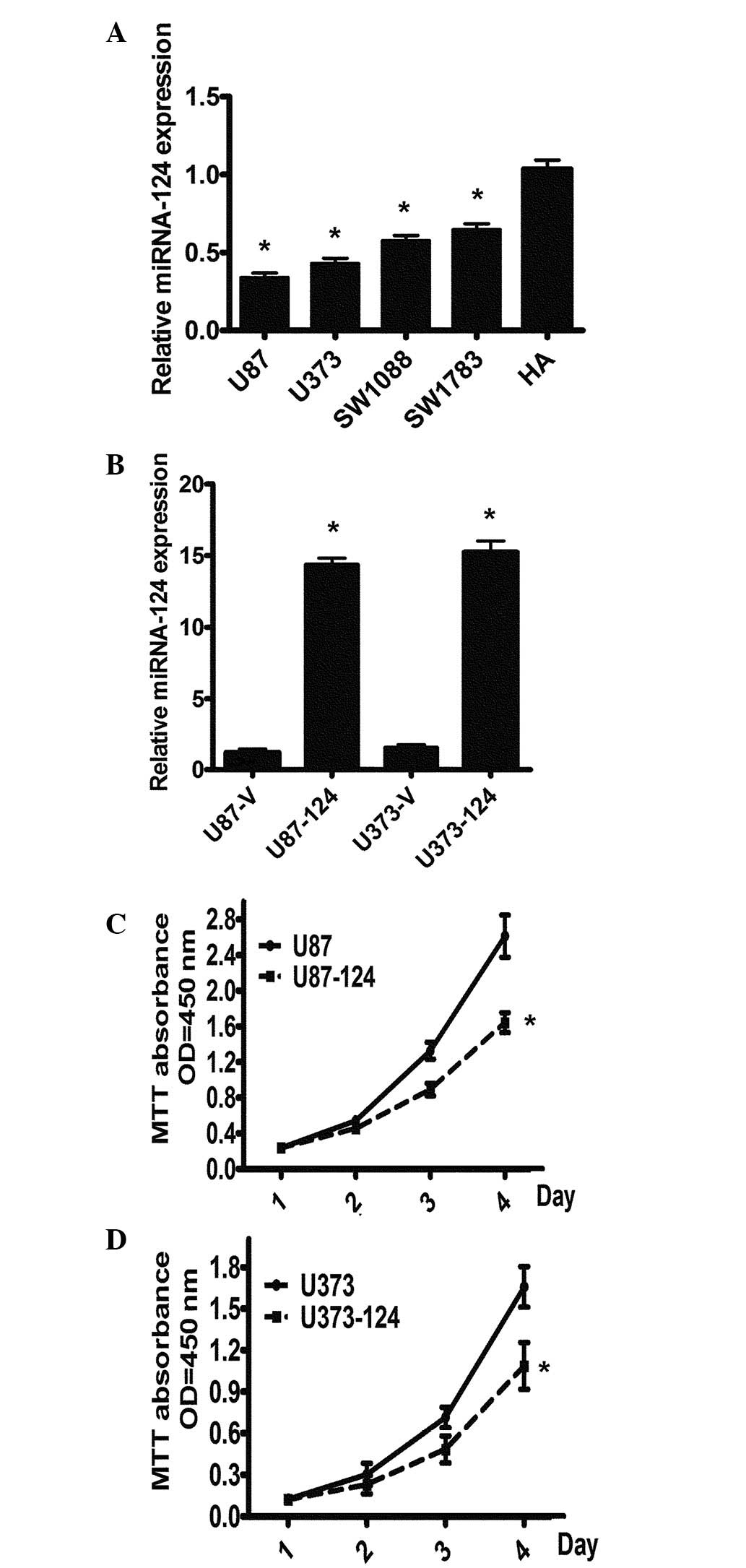
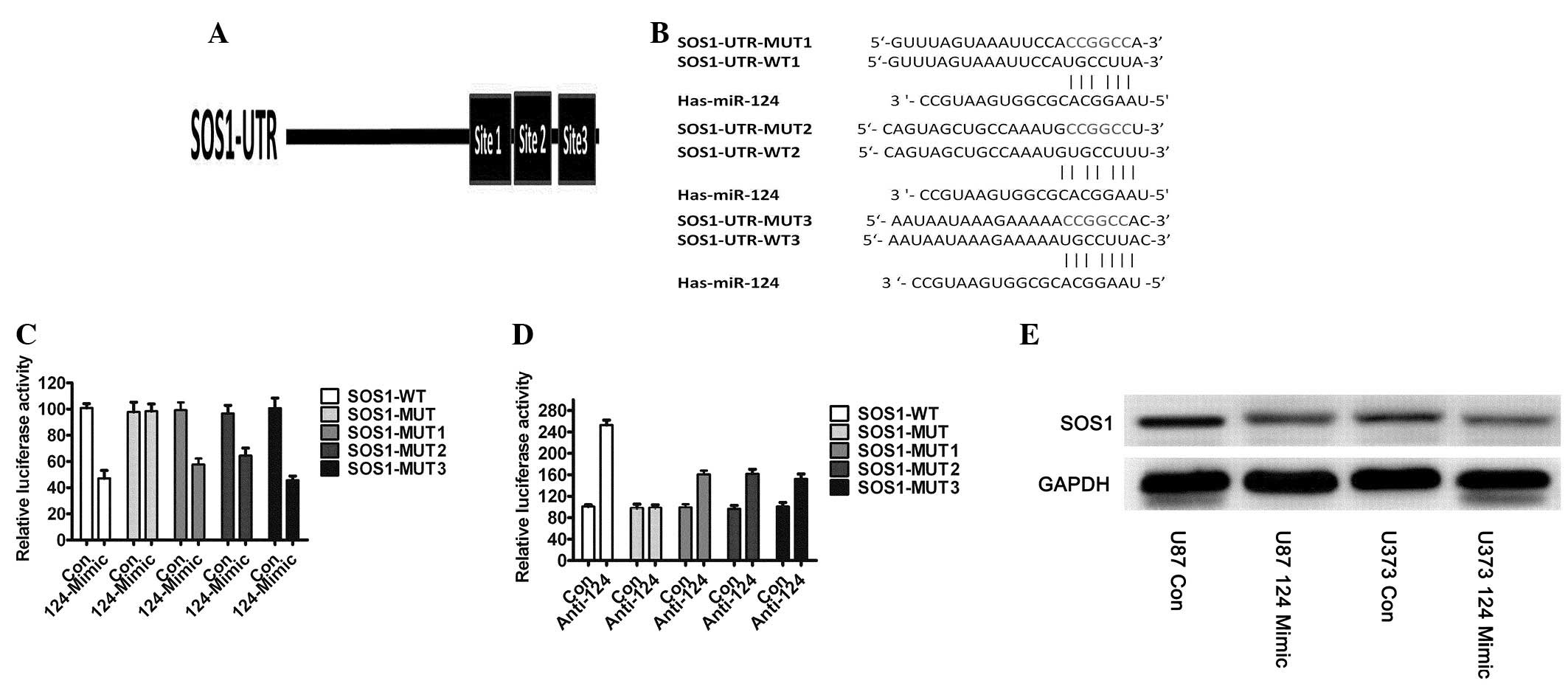
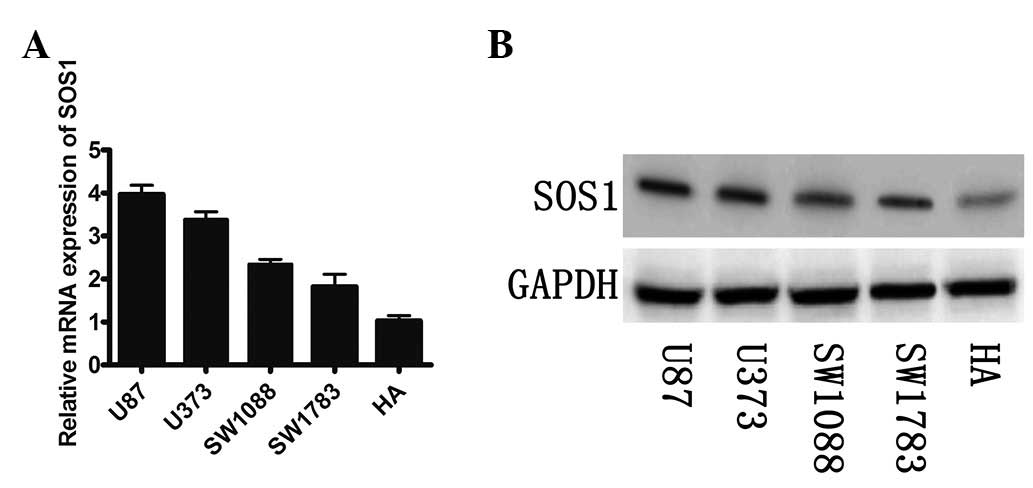
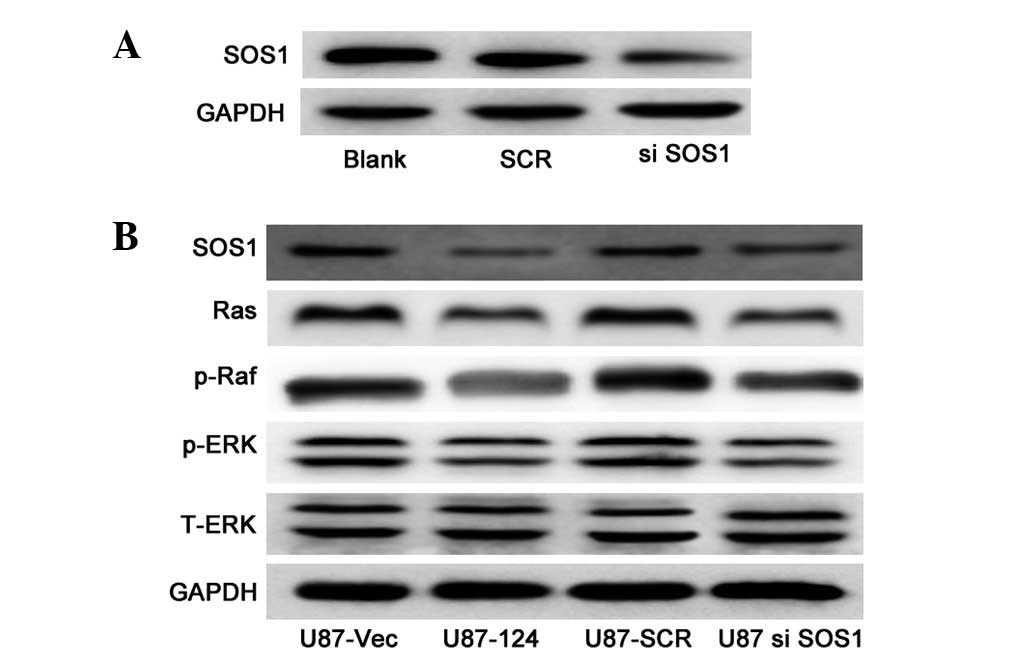
|
1
|
Karsy M, Arslan E and Moy F: Current
progress on understanding microRNAs in glioblastoma multiforme.
Genes Cancer. 3:3–15. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Omuro AM, Faivre S and Raymond E: Lessons
learned in the development of targeted therapy for malignant
gliomas. Mol Cancer Ther. 6:1909–1919. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Garofalo M and Croce CM: microRNAs: master
regulators as potential therapeutics in cancer. Annu Rev Pharmacol
Toxicol. 51:25–43. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bartel DP: MicroRNAs: genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kwak HJ, Kim YJ, Chun KR, et al:
Downregulation of Spry2 by miR-21 triggers malignancy in human
gliomas. Oncogene. 30:2433–2442. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhou X, Ren Y, Moore L, et al:
Downregulation of miR-21 inhibits EGFR pathway and suppresses the
growth of human glioblastoma cells independent of PTEN status. Lab
Invest. 90:144–155. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhu XC, Dong QZ, Zhang XF, et al:
microRNA-29a suppresses cell proliferation by targeting SPARC in
hepatocellular carcinoma. Int J Mol Med. 30:1321–1326.
2012.PubMed/NCBI
|
|
8
|
Tang H, Liu X, Wang Z, et al: Interaction
of hsa-miR-381 and glioma suppressor LRRC4 is involved in glioma
growth. Brain Res. 1390:21–32. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ernst A, Campos B, Meier J, et al:
De-repression of CTGF via the miR-17–92 cluster upon
differentiation of human glioblastoma spheroid cultures. Oncogene.
29:3411–3422. 2010.PubMed/NCBI
|
|
10
|
Huse JT, Brennan C, Hambardzumyan D, et
al: The PTEN-regulating microRNA miR-26a is amplified in high-grade
glioma and facilitates gliomagenesis in vivo. Genes Dev.
23:1327–1337. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang H, Kong W, He L, et al: MicroRNA
expression profiling in human ovarian cancer: miR-214 induces cell
survival and cisplatin resistance by targeting PTEN. Cancer Res.
68:425–433. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim H, Huang W, Jiang X, Pennicooke B,
Park PJ and Johnson MD: Integrative genome analysis reveals an
oncomir/oncogene cluster regulating glioblastoma survivorship. Proc
Natl Acad Sci USA. 107:2183–2188. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gureasko J, Galush WJ, Boykevisch S, et
al: Membrane-dependent signal integration by the Ras activator Son
of sevenless. Nat Struct Mol Biol. 15:452–461. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nimnual AS, Yatsula BA and Bar-Sagi D:
Coupling of Ras and Rac guanosine triphosphatases through the Ras
exchanger Sos. Science. 279:560–563. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Margarit SM, Sondermann H, Hall BE, et al:
Structural evidence for feedback activation by Ras. GTP of the
Ras-specific nucleotide exchange factor SOS. Cell. 112:685–695.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bar-Sagi D: The Sos (Son of sevenless)
protein. Trends Endocrinol Metab. 5:165–169. 1994. View Article : Google Scholar
|
|
17
|
Vetter IR and Wittinghofer A: The guanine
nucleotide-binding switch in three dimensions. Science.
294:1299–1304. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dhillon AS and Kolch W: Untying the
regulation of the Raf-1 kinase. Arch Biochem Biophys. 404:3–9.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yamamoto T, Ebisuya M, Ashida F, Okamoto
K, Yonehara S and Nishida E: Continuous ERK activation
downregulates antiproliferative genes throughout G1 phase to allow
cell-cycle progression. Curr Biol. 16:1171–1182. 2006. View Article : Google Scholar
|
|
20
|
Cheng LC, Pastrana E, Tavazoie M and
Doetsch F: miR-124 regulates adult neurogenesis in the
subventricular zone stem cell niche. Nat Neurosci. 12:399–408.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yoo AS, Sun AX, Li L, et al:
MicroRNA-mediated conversion of human fibroblasts to neurons.
Nature. 476:228–231. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen C, Ridzon DA, Broomer AJ, et al:
Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic
Acids Res. 33:e1792005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lewis BP, Burge CB and Bartel DP:
Conserved seed pairing, often flanked by adenosines, indicates that
thousands of human genes are microRNA targets. Cell. 120:15–20.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Krek A, Grün D, Poy MN, et al:
Combinatorial microRNA target predictions. Nat Genet. 37:495–500.
2005. View
Article : Google Scholar
|
|
25
|
Cordes KR, Sheehy NT, White MP, et al:
miR-145 and miR-143 regulate smooth muscle cell fate and
plasticity. Nature. 460:705–710. 2009.PubMed/NCBI
|
|
26
|
Chen CZ, Li L, Lodish HF and Bartel DP:
MicroRNAs modulate hematopoietic lineage differentiation. Science.
303:83–86. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Makeyev EV, Zhang J, Carrasco MA and
Maniatis T: The MicroRNA miR-124 promotes neuronal differentiation
by triggering brain-specific alternative pre-mRNA splicing. Mol
Cell. 27:435–448. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
King IN, Qian L, Liang J, et al: A
genome-wide screen reveals a role for microRNA-1 in modulating
cardiac cell polarity. Dev Cell. 20:497–510. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Silber J, Lim DA, Petritsch C, et al:
miR-124 and miR-137 inhibit proliferation of glioblastoma
multiforme cells and induce differentiation of brain tumor stem
cells. BMC Med. 6:142008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xia H, Cheung WK, Ng SS, et al: Loss of
brain-enriched miR-124 microRNA enhances stem-like traits and
invasiveness of glioma cells. J Biol Chem. 287:9962–9971. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Timofeeva OA, Zhang X, Ressom HW, et al:
Enhanced expression of SOS1 is detected in prostate cancer
epithelial cells from African-American men. Int J Oncol.
35:751–760. 2009.PubMed/NCBI
|
|
32
|
Daniels MA, Teixeiro E, Gill J, et al:
Thymic selection threshold defined by compartmentalization of
Ras/MAPK signalling. Nature. 444:724–729. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Roose JP, Mollenauer M, Ho M, Kurosaki T
and Weiss A: Unusual interplay of two types of Ras activators,
RasGRP and SOS, establishes sensitive and robust Ras activation in
lymphocytes. Mol Cell Biol. 27:2732–2745. 2007. View Article : Google Scholar : PubMed/NCBI
|