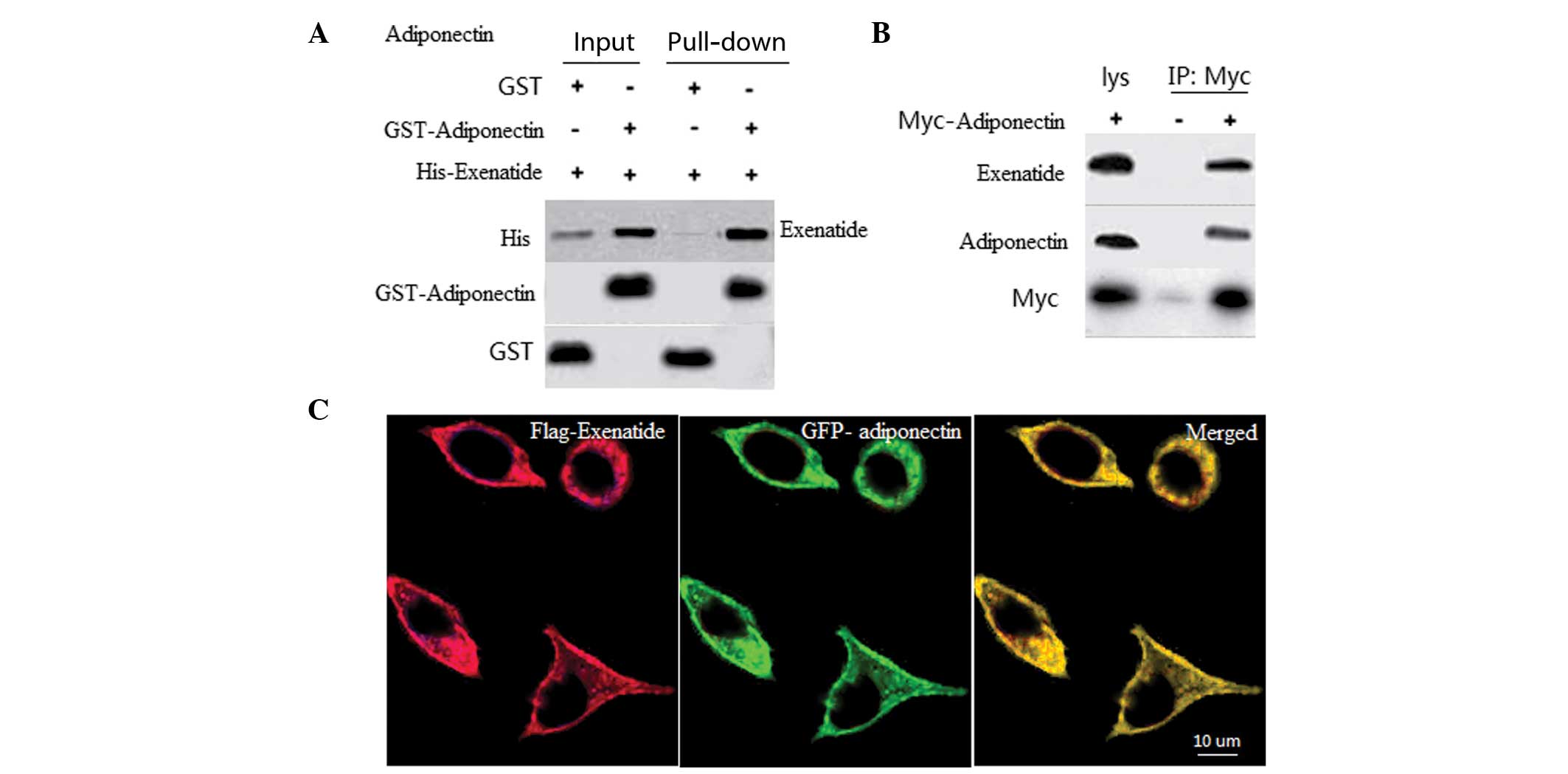
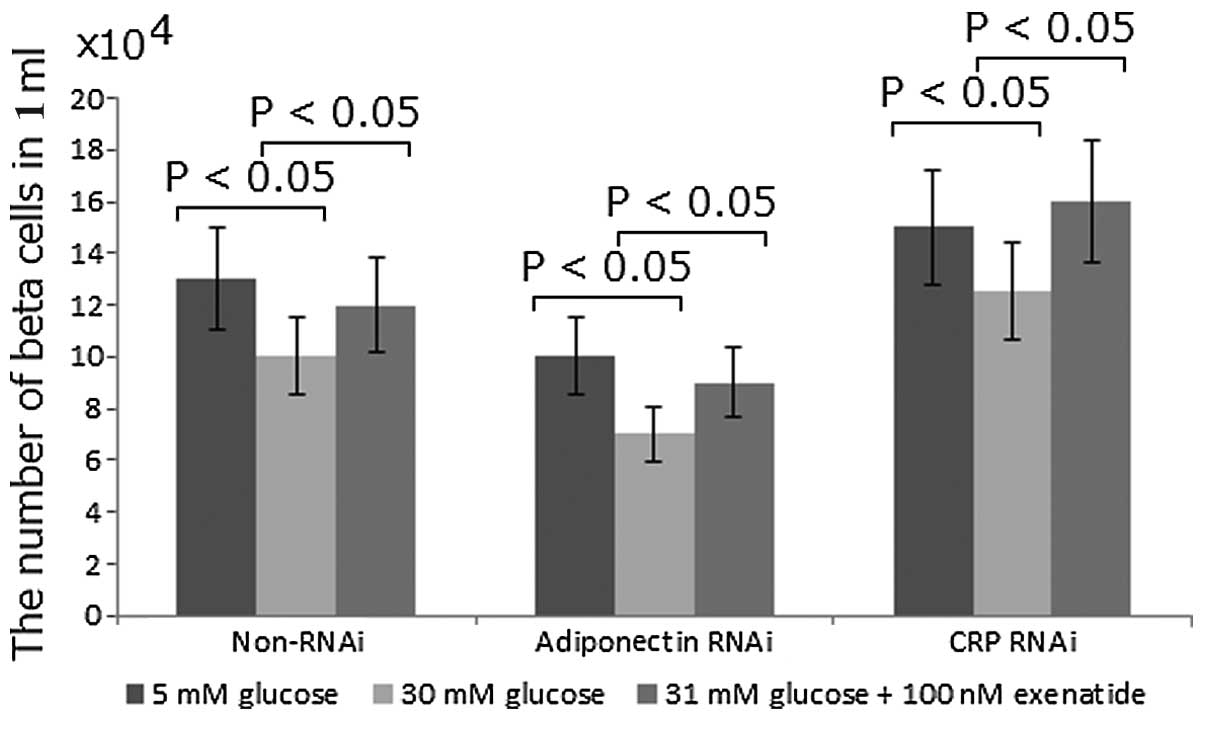
|
1
|
Anvari M: Use of metabolic surgery for the
treatment of type 2 diabetes. Can J Diabetes. 35:99–108. 2011.
View Article : Google Scholar
|
|
2
|
Srinivasan K and Ramarao P: Animal models
in type 2 diabetes research: an overview. Indian J Med Res.
125:451–472. 2007.PubMed/NCBI
|
|
3
|
Bennett WL, Maruthur NM, Singh S, et al:
Comparative effectiveness and safety of medications for type 2
diabetes: an update including new drugs and 2-drug combinations.
Ann Intern Med. 154:602–613. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pathania S, Randhawa V and Bagler G:
Prospecting for novel plant-derived molecules of Rauvolfia
serpentina as inhibitors of Aldose Reductase, a potent drug
target for diabetes and its complications. PloS one.
8:e613272013.PubMed/NCBI
|
|
5
|
Sugimoto T and Kashiwagi A: The
cutting-edge of medicine; novel therapeutic agents for the
treatment of diabetes sodium-glucose co-transporter (SGLT) 2
inhibitors. Nihon Naika Gakkai Zasshi. 102:1474–1483. 2013.(In
Japanese).
|
|
6
|
Meier JJ: GLP-1 receptor agonists for
individualized treatment of type 2 diabetes mellitus. Nat Rev
Endocrinol. 8:728–742. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gedulin BR, Nikoulina SE, Smith PA, et al:
Exenatide (exendin-4) improves insulin sensitivity and {beta}-cell
mass in insulin-resistant obese fa/fa Zucker rats independent of
glycemia and body weight. Endocrinology. 146:2069–2076. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bunck MC, Diamant M, Cornér A, et al:
One-year treatment with exenatide improves beta-cell function,
compared with insulin glargine, in metformin-treated type 2
diabetic patients: a randomized, controlled trial. Diabetes Care.
32:762–768. 2009.
|
|
9
|
Fehse F, Trautmann M, Holst JJ, et al:
Exenatide augments first- and second-phase insulin secretion in
response to intravenous glucose in subjects with type 2 diabetes. J
Clin Endocrinol Metab. 90:5991–5997. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tripathy NR, Basha S, Jain R, Shetty S and
Ramachandran A: Exenatide and acute pancreatitis. J Assoc
Physicians India. 56:987–988. 2008.PubMed/NCBI
|
|
11
|
Kendall DM, Riddle MC, Rosenstock J, et
al: Effects of exenatide (exendin-4) on glycemic control over 30
weeks in patients with type 2 diabetes treated with metformin and a
sulfonylurea. Diabetes Care. 28:1083–1091. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gutzwiller JP, Drewe J, Göke B, et al:
Glucagon-like peptide-1 promotes satiety and reduces food intake in
patients with diabetes mellitus type 2. Am J Physiol.
276:R1541–R1544. 1999.PubMed/NCBI
|
|
13
|
Zander M, Madsbad S, Madsen JL and Holst
JJ: Effect of 6-week course of glucagon-like peptide 1 on glycaemic
control, insulin sensitivity, and beta-cell function in type 2
diabetes: a parallel-group study. Lancet. 359:824–830. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Navarro M, Rodriquez de Fonseca F, Alvarez
E, et al: Colocalization of glucagon-like peptide-1 (GLP-1)
receptors, glucose transporter GLUT-2, and glucokinase mRNAs in rat
hypothalamic cells: evidence for a role of GLP-1 receptor agonists
as an inhibitory signal for food and water intake. J Neurochem.
67:1982–1991. 1996. View Article : Google Scholar
|
|
15
|
Nir T, Melton DA and Dor Y: Recovery from
diabetes in mice by beta cell regeneration. J Clin Invest.
117:2553–2561. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Iwaki M, Matsuda M, Maeda N, et al:
Induction of adiponectin, a fat-derived antidiabetic and
antiatherogenic factor, by nuclear receptors. Diabetes.
52:1655–1663. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Freeman DJ, Norrie J, Caslake MJ, et al:
C-reactive protein is an independent predictor of risk for the
development of diabetes in the West of Scotland Coronary Prevention
Study. Diabetes. 51:1596–1600. 2002. View Article : Google Scholar
|
|
18
|
Bergenstal RM, Wysham C, Macconell L, et
al: DURATION-2 Study Group: Efficacy and safety of exenatide once
weekly versus sitagliptin or pioglitazone as an adjunct to
metformin for treatment of type 2 diabetes (DURATION-2): a
randomised trial. Lancet. 376:431–439. 2010. View Article : Google Scholar
|
|
19
|
Derosa G, Putignano P, Bossi AC, et al:
Exenatide or glimepiride added to metformin on metabolic control
and on insulin resistance in type 2 diabetic patients. Eur J
Pharmacol. 666:251–256. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wijesekara N, Krishnamurthy M,
Bhattacharjee A, Suhail A, Sweeney G and Wheeler MB:
Adiponectin-induced ERK and Akt phosphorylation protects against
pancreatic beta cell apoptosis and increases insulin gene
expression and secretion. J Biol Chem. 285:33623–33631. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rakatzi I, Mueller H, Ritzeler O,
Tennagels N and Eckel J: Adiponectin counteracts cytokine-and fatty
acid-induced apoptosis in the pancreatic beta-cell line INS-1.
Diabetologia. 47:249–258. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kahn SE, Zinman B, Haffner SM, et al:
ADOPT Study Group: Obesity is a major determinant of the
association of C-reactive protein levels and the metabolic syndrome
in type 2 diabetes. Diabetes. 55:2357–2364. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Boden G and Shulman GI: Free fatty acids
in obesity and type 2 diabetes: defining their role in the
development of insulin resistance and beta-cell dysfunction. Eur J
Clin Invest. 32(Suppl 3): 14–23. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Federici M, Hribal M, Perego L, et al:
High glucose causes apoptosis in cultured human pancreatic islets
of Langerhans: a potential role for regulation of specific Bcl
family genes toward an apoptotic cell death program. Diabetes.
50:1290–1301. 2001. View Article : Google Scholar
|
|
25
|
Moretto TJ, Milton DR, Ridge TD, et al:
Efficacy and tolerability of exenatide monotherapy over 24 weeks in
antidiabetic drug-naive patients with type 2 diabetes: a
randomized, double-blind, placebo-controlled, parallel-group study.
Clin Ther. 30:1448–1460. 2008. View Article : Google Scholar
|
|
26
|
Gentilella R, Bianchi C, Rossi A and
Rotella CM: Exenatide: a review from pharmacology to clinical
practice. Diabetes Obes Metab. 11:544–556. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fineman MS, Shen LZ, Taylor K, Kim DD and
Baron AD: Effectiveness of progressive dose-escalation of exenatide
(exendin-4) in reducing dose-limiting side effects in subjects with
type 2 diabetes. Diabetes Metab Res Rev. 20:411–417. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Brubaker PL: Incretin-based therapies:
mimetics versus protease inhibitors. Trends Endocrinol Metab.
18:240–245. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen C, Constantinou A and Deonarain M:
Modulating antibody pharmacokinetics using hydrophilic polymers.
Expert Opin Drug Deliv. 8:1221–1236. 2011. View Article : Google Scholar : PubMed/NCBI
|