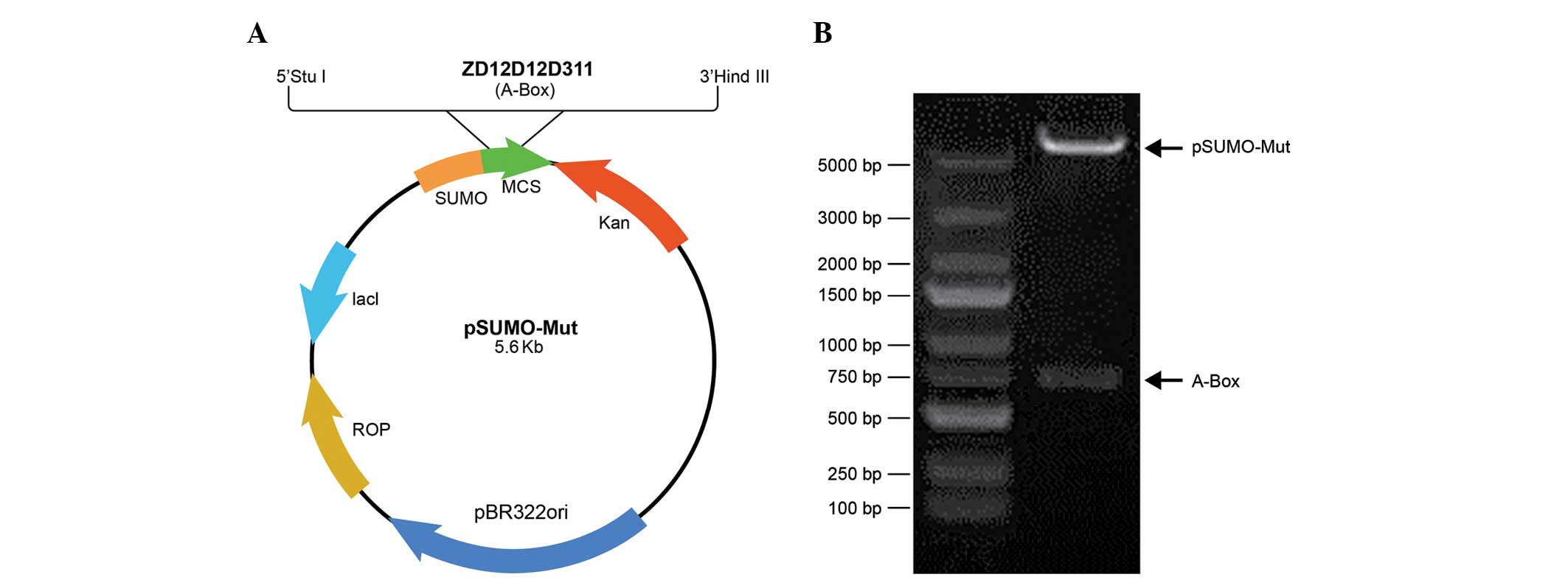
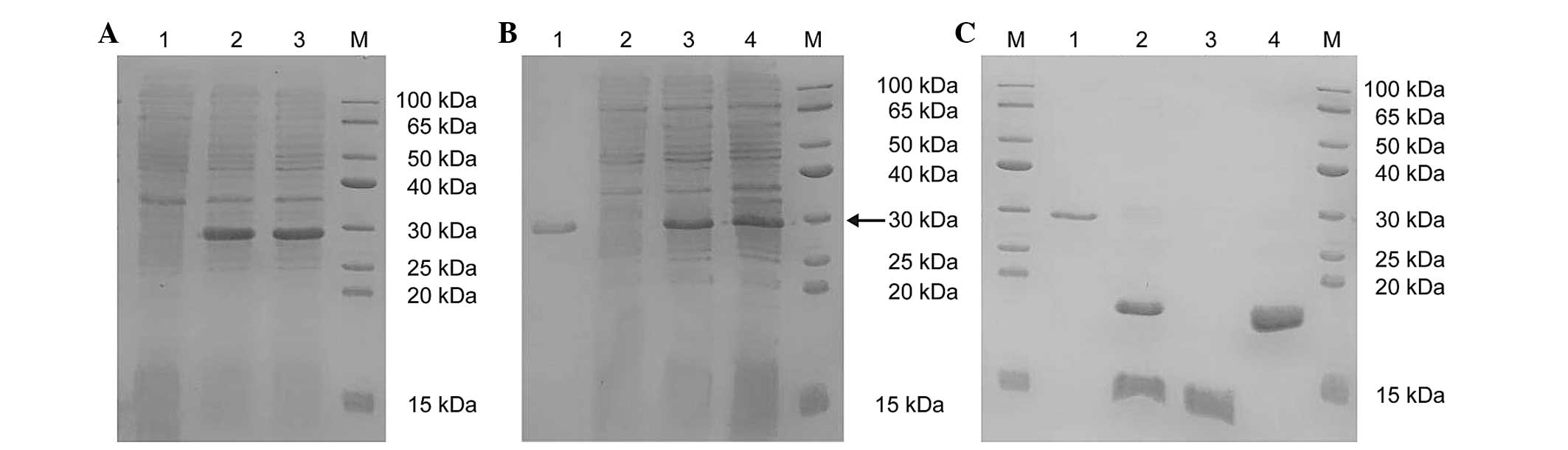
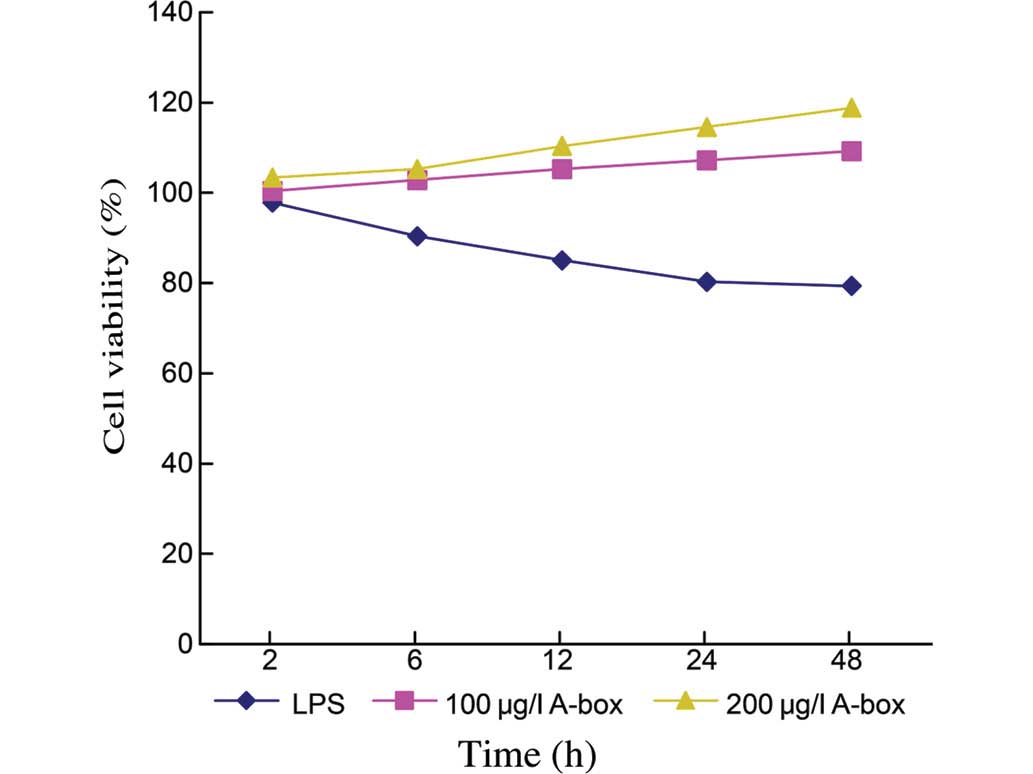
|
1
|
Bustin M, Lehn DA and Landsman D:
Structural features of the HMG chromosomal proteins and their
genes. Biochim Biophys Acta. 1049:231–243. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Boonyaratanakornkit V, Melvin V,
Prendergast P, Altmann M, Ronfani L, Bianchi ME, Taraseviciene L,
Nordeen SK, Allegretto EA and Edwards DP: High-mobility group
chromatin proteins 1 and 2 functionally interact with steroid
hormone receptors to enhance their DNA binding in vitro and
transcriptional activity in mammalian cells. Mol Cell Biol.
18:4471–4487. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Melvin VS and Edwards DP: Coregulatory
proteins in steroid hormone receptor action: The role of chromatin
high mobility group proteins HMG-1 and -2. Steroids. 64:576–586.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Vaccari T, Beltrame M, Ferrari S and
Bianchi ME: Hmg4, a new member of the Hmg1/2 gene family. Genomics.
49:247–252. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Funayama A, Shishido T, Netsu S, Narumi T,
Kadowaki S, Takahashi H, Miyamoto T, Watanabe T, Woo CH, Abe J, et
al: Cardiac nuclear high mobility group box 1 prevents the
development of cardiac hypertrophy and heart failure. Cardiovasc
Res. 99:657–664. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tsung A, Tohme S and Billiar TR: High
mobility group box-1 in sterile inflammation. J Intern Med.
276:425–443. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Dumitriu IE, Baruah P, Manfredi AA,
Bianchi ME and Rovere-Querini P: HMGB1: Guiding immunity from
within. Trends Immunol. 26:381–387. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang D, Chen Q, Yang H, Tracey KJ, Bustin
M and Oppenheim JJ: High mobility group box-1 protein induces the
migration and activation of human dendritic cells and acts as an
alarmin. J Leukoc Biol. 81:59–66. 2007. View Article : Google Scholar
|
|
9
|
Yang H, Wang H, Czura CJ and Tracey KJ:
HMGB1 as a cytokine and therapeutic target. J Endotoxin Res.
8:469–472. 2002. View Article : Google Scholar
|
|
10
|
Andersson U and Tracey KJ: HMGB1 in
sepsis. Scand J Infect Dis. 35:577–584. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kokkola R, Li J, Sundberg E, Aveberger AC,
Palmblad K, Yang H, Tracey KJ, Andersson U and Harris HE:
Successful treatment of collagen-induced arthritis in mice and rats
by targeting extracellular high mobility group box chromosomal
protein 1 activity. Arthritis Rheum. 48:2052–2058. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang H, Bloom O, Zhang M, Vishnubhakat JM,
Ombrellino M, Che J, Frazier A, Yang H, Ivanova S, Borovikova L, et
al: HMG-1 as a late mediator of endotoxin lethality in mice.
Science. 285:248–251. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang Y, Yin H, Han J, Huang B, Xu J,
Zheng F, Tan Z, Fang M, Rui L, Chen D, et al: Extracellular hmgb1
functions as an innate immune-mediator implicated in murine cardiac
allograft acute rejection. Am J Transplant. 7:799–808. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Andrassy M, Volz HC, Igwe JC, Funke B,
Eichberger SN, Kaya Z, Buss S, Autschbach F, Pleger ST, Lukic IK,
et al: High-mobility group box-1 in ischemia-reperfusion injury of
the heart. Circulation. 117:3216–3226. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Andersson U and Tracey KJ: HMGB1 is a
therapeutic target for sterile inflammation and infection. Annu Rev
Immunol. 29:139–162. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yang H, Ochani M, Li J, Qiang X, Tanovic
M, Harris HE, Susarla SM, Ulloa L, Wang H, DiRaimo R, et al:
Reversing established sepsis with antagonists of endogenous
high-mobility group box 1. Proc Natl Acad Sci USA. 101:296–301.
2004. View Article : Google Scholar :
|
|
17
|
Butt TR, Edavettal SC, Hall JP and Mattern
MR: SUMO fusion technology for difficult-to-express proteins.
Protein Expr Purif. 43:1–9. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Johnson ES: Protein modification by SUMO.
Annu Rev Biochem. 73:355–382. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hochstrasser M: Evolution and function of
ubiquitin-like protein-conjugation systems. Nat Cell Biol.
2:E153–E157. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jentsch S and Pyrowolakis G: Ubiquitin and
its kin: How close are the family ties? Trends Cell Biol.
10:335–342. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Müller S, Hoege C, Pyrowolakis G and
Jentsch S: SUMO, ubiquitin's mysterious cousin. Nat Rev Mol Cell
Biol. 2:202–210. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Malakhov MP, Mattern MR, Malakhova OA,
Drinker M, Weeks SD and Butt TR: SUMO fusions and SUMO-specific
protease for efficient expression and purification of proteins. J
Struct Funct Genomics. 5:75–86. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zuo X, Mattern MR, Tan R, Li S, Hall J,
Sterner DE, Shoo J, Tran H, Lim P, Sarafianos SG, et al: Expression
and purification of SARS coronavirus proteins using SUMO-fusions.
Protein Expr Purif. 42:100–110. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang Z, Zhang L, Zhou C and Wu H:
Ketamine inhibits LPS-induced HGMB1 release in vitro and in vivo.
Int Immunopharmacol. 23:14–26. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kong X, Yuan H, Wu X, Zhang J, Zhou H,
Wang M, Liu Y and Jin X: High-mobility-group box protein 1A box
reduces development of sodium laurate-induced thromboangiitis
obliterans in rats. J Vasc Surg. 57:194–204. 2013. View Article : Google Scholar
|