Introduction
Diabetic nephropathy (DN) is a common complication
of diabetes mellitus (DM), which is a major health problem
worldwide (1). Chronic
inflammation and oxidative stress are crucial contributors to the
progression of DN (2). Various
preventable factors, including hypertension, alcoholism and
smoking, also accelerate DN progression. Epidemiological studies
suggest that cigarette smoking increases the progression rate of
renal failure among insulin-and non-insulin-dependent patients with
DM (3), and is associated with
elevated serum creatinine levels (4). A dose-dependent relationship between
the number of cigarettes smoked and the development of albuminuria
was reported in type 1 DM (5).
Additionally, the prevalence of microalbuminuria and
macroalbuminuria was significantly higher in patients with type 2
DM that were smokers. A possible role of smoking in the
acceleration of renal remodeling and damage was postulated to
operate via a mechanism independent of the damage caused by DM
(6). Evidence from the European
population indicates that smoking may be associated with the
progression of chronic kidney disease (CKD), particularly in men
(7).
Previous studies have focused on anti-inflammatory
and anti-oxidative treatments as a method to delay the progression
of glomerulopathy (2,8). Curcumin, derived from the rhizome of
the herb Curcuma longa, has been indicated to possess a
variety of beneficial effects, including anti-inflammatory and
anti-oxidative activities (9).
Curcumin has been demonstrated to be protective against nephropathy
and islet damage in streptozotocin (STZ)-induced diabetic rats
(10,11). Curcumin was previously reported to
be effective in preventing glucose-induced oxidative stress in the
endothelial cells and hearts of diabetic animals (12).
The current study aimed to investigate the potential
protective effects of curcumin against the combined oxidative
stress of DM and nicotine (NC), a major component of cigarette
smoke.
Materials and methods
Animals
A total of 24 adult male Wistar rats (65–70 days
old, 190–220 g) were purchased from the Experimental Animal Center,
Faculty of Pharmacy, King Saud University (Riyadh, Saudi Arabia).
Animals were housed in the animal facility of the College of
Science, Taif University (Taif, Saudi Arabia) at 22°C and 55%
humidity with a 12 h light/dark cycle. The rats were fed a standard
pellet diet and provided with water ad libitum. The present
study was approved by the ethical committee of Taif University.
Reformation of animal food with the
addition of curcumin
As curcumin is water insoluble, it was added to the
food of the animals at a concentration of 1.5 g/kg of food. The
food pellet was crushed to a powder and mixed thoroughly with
curcumin powder (Nature's Purest, West Los Angeles, CA, USA). A
small volume of water was added to create a paste and the pellets
were reformed. The pellets were left to air dry for 4 days. The
concentration of curcumin was selected based on the average body
weight of the rats (~200 g) and the estimated average daily feed
consumption (~25–30 g/day). The NC (Merck Millipore, Darmstadt,
Germany) concentration was also selected according to the average
body weight and the average daily water consumption (~25
ml/day).
Experimental design
Following 1-week acclimatization, the rats were
divided into 2 groups as follows: 18 rats were fasted for 8 hrs,
after which they were injected intraperitonealy with a single dose
of streptozotocin (STZ; 60 mg/kg body weight; Sigma-Aldrich, St.
Louis, MO, USA) freshly dissolved in citrate buffer (pH 4.5); and a
negative control group (6 rats) were injected with an equivalent
volume of vehicle (citrate buffer, pH 4.5). Following injection,
the rats were provided with free access to food and water, and were
given a 5% glucose solution to drink overnight to counteract
hypoglycemic shock. DM in the rats was identified by moderate
polydipsia and marked polyuria. After 3 days, the fasting blood
glucose levels were measured in tail blood samples (Accu-Chek Aviva
Blood Glucose Meter; Roche Diagnostics, Indianapolis, IN, USA). The
rats with glucose levels >200 mg/100 ml were considered to be
diabetic and selected for further experimentation. The diabetic
rats were divided into three groups of six rats each as follows: i)
The DM group; ii) the DM + NC group; and iii) the DM + NC +
curcumin group. NC was added to the drinking water of the DM + NC
and the DM + NC + curcumin group rats at a concentration of 12
µg/ml to attain a treatment dose of 1.5 mg/kg body weight.
The food of the DM + NC + curcumin group rats was additionally
supplemented with curcumin at a concentration of 1.5 g/kg body
weight to obtain a final concentration of 1.5 mg/kg body weight.
Treatments were continued for the 8 weeks following induction of
DM. Random blood glucose levels were measured from tail blood
samples every week to ensure the rats remained diabetic. At the end
of the experiment, the rats were fasted for 8 h and anesthetized
with diethyl ether (≥99.0%; Sigma-Aldrich), while blood samples
were taken from the medial canthus of the eyes into heparinized
tubes. The rats were sacrificed by head dislocation and tissue
samples were taken for RNA extraction and stored at −80°C prior to
use. Samples for histological examination were stored in 10%
formalin (Sigma-Aldrich) until processing.
Biochemical analysis
Urea and plasma creatinine assays were performed
using the UREA liquicolor (cat. no. 10505) and CREATININE
liquicolor (cat. no. 10051) Complete Test kits purchased from HUMAN
Gesellschaft für Biochemica und Diagnostica mbH (Wiesbaden,
Germany). Cholesterol, triacylglycerol (TG) and high density
lipoprotein (HDL) levels were measured using kits (cat. nos.
024-100, 059L-050 and 041-050, respectively) purchased from United
Diagnostics Industry (Dammam, Saudi Arabia). Commercial kits were
purchased from Nanjing Jiancheng Bioengineering Research Institute
(Nanjing, China) to measure the levels of plasma malondialdehyde
(MDA; A003-1), and the activities of γ-glutamyltranspeptidase
(GGT), superoxide dismutase (SOD; A001-1) and glutathione
peroxidase (GPx; A005). The plasma phospholipid levels were
determined using the Phospholipid Assay kit (MAK122;
Sigma-Aldrich), according to the manufacturer's protocol. The
concentrations of the measured biochemical constituents were
calculated according to the manufacturer's instructions.
RNA extraction
For preparation of total RNA, 50 mg of kidney tissue
sample was homogenized in 1 ml QIAzol (Qiagen, Inc., Valencia, CA,
USA) and 0.3 ml chloroform (Nanjing Jiancheng Bioengineering
Research Institute) was added to the homogenate. The mixtures were
then shaken for 30 sec followed by centrifugation at 4°C and 12,000
× g for 20 min. The supernatant layers were transferred to a new
set of tubes, and an equal volume of isopropanol (Sigma-Aldrich)
was added to the samples. The samples were shaken for 15 sec and
centrifuged at 4°C and 12,000 x g for 15 min. The RNA pellets were
washed with 70% ethanol, briefly dried and dissolved in
diethylpyrocarbonate (DEPC) water. The prepared RNA integrity was
determined by 1.5% agarose gel electrophoresis. The RNA
concentration and purity were determined spectrophotometrically at
260 nm using the GelDoc-It Imaging System (UVP, Inc., Upland, CA,
USA). The ratio of the 260/280 nm optical density of all RNA
samples was 1.7–1.9.
cDNA synthesis
DNase treatment of the extracted RNA was performed
prior to cDNA synthesis using the RQ1 RNase-Free DNase kit (M6101;
Promega Corporation, Madison, WI, USA). For synthesis of cDNA, a
mixture of 2 µg total RNA and 0.5 ng Oligo (dT)15 primers
(Macrogen, Inc., Seoul, Korea) in a total volume of 11 µl
sterilized DEPC water was incubated in the Thermo Hybaid PXE 0.2
Thermal Cycler (Thermo Fisher Scientific, Inc., Waltham, MA, USA)
at 70°C for 10 min for denaturing. Subsequently, 4 µl 5X
RT-buffer, 2 µl 10 mM dNTPs and 100 units RevertAid Premium
reverse transcriptase (Fermentas; Thermo Fisher Scientific, Inc.,
Pittsburgh, PA, USA) were added to the reaction mixture and DEPC
water was used to bring the total volume to 20 µl. The
mixture was then incubated in the thermal cycler at 30°C for 10
min, 42°C for 1 h and 90°C for 10 min. The resulting cDNA was then
preserved at −20°C until use.
Semi-quantitative polymerase chain
reaction (PCR)
The mRNA expression levels of gene markers of kidney
damage, kidney protection and integrity, and inflammation and
fibrosis were analyzed by semi-quantitative PCR using the
corresponding gene-specific primers (Table I). The genes analyzed were
vimentin, desmin, synaptopodin (SPD), connexin 43 (Cx43), sterol
regulatory element-binding protein-1c (SREBP-1c), transforming
growth factor β1 (TGF-β1) and erythropoietin (EPO). As a reference
gene, the expression of GAPDH mRNA was determined. The primers were
designed using the Oligo Primer Analysis software, version 4
(www.oligo.net) and nucleotide sequences published
in Genbank (www.ncbi.nlm.nih.gov/genbank; Table I). The primers were synthesized by
Macrogen, Inc. PCR was conducted in a final reaction volume of 25
µl consisting of 1 µl cDNA, 1 µl of each 10 pM
forward and reverse primers, 12.5 µl PCR master mix (Promega
Corporation) and nuclease-free deionized water. PCR was conducted
using the thermal cycler with a cycle sequence of denaturing at
94°C for 4 min for 1 cycle, followed by 25–32 cycles of
denaturation at 94°C for 1 min, annealing at the specific
temperature corresponding to each primer (see Table I) and extension at 72°C for 1 min,
with an additional final extension step at 72°C for 5 min. A pilot
study was performed to optimize the specific number of cycles for
each gene according to its expression level. The PCR products were
electrophoresed on a 1% agarose A (Bio Basic Canada, Inc., Markham,
ON, Canada) gel in Tris-EDTA buffer at 100 V for 30 min, and
stained with ethidium bromide. The PCR products were visualized
under UV light and images were captured using the GelDoc-It Imaging
System. The intensities of the bands were densitometrically
quantified using NIH Image software (National Institutes of Health,
Bethesda, MD, USA; rsb.info.nih.gov/nih-image).
 | Table IPrimers and polymerase chain reaction
conditions used for the tested genes. |
Table I
Primers and polymerase chain reaction
conditions used for the tested genes.
| Gene | Product size
(bp) | Annealing
temperature (°C) | Cycles | Primer | Sequence
(5′-3′) |
|---|
| GAPDH | 309 | 52 | 25 | Sense |
AGATCCACAACGGATACATT |
| | | | Antisense |
TCCCTCAAGATTGTCAGCAA |
| Vimentin | 190 | 55 | 30 | Sense |
GAGTCAAACGAATACCGGAGAC |
| | | | Antisense |
GTGGTGCTGAGAAGTCTCATTG |
| Desmin | 420 | 54 | 28 | Sense |
TCACAATCACCTCTTTGTGGTC |
| | | | Antisense |
CAGCACCTTCCAGTTCTCTCTT |
| Synaptopodin | 457 | 59 | 32 | Sense |
ACGCCCACTAAGGTGTATAGTGA |
| | | | Antisense |
CTTCCAAAATTCCTGTCTTGTTG |
| Connexin 43 | 435 | 59 | 30 | Sense |
TACTTGGCCCATGTGTTCTATG |
| | | | Antisense |
AACGCCTTTGAAGAAGACGTAG |
| SREBP-1c | 191 | 58 | 28 | Sense |
GGAGCCATGGATTGCACATT |
| | | | Antisense |
AGGAAGGCTTCCAGAGAGGA |
| TGF-β1 | 304 | 58 | 30 | Sense |
TGAGTGGCTGTCTTTTGACG |
| | | | Antisense |
TGGTTGTAGAGGGCAAGGAC |
| Erythropoietin | 530 | 60 | 29 | Sense |
TACGTAGCCTCACTTCACTGCTT |
| | | | Antisense |
GCAGAAAGTATCCGCTGTGAGTGTTC |
| iNOS | 378 | 59 | 32 | Sense |
GGTGTTCTTTGCTTCTGTGCTA |
| | | | Antisense |
TGAGACAGTTTCTGGTCGATGT |
Histological examination
Small specimens (1.5×0.5 cm) from the pancreas and
kidneys were fixed in 10% neutral-buffered formalin (Sigma-Aldrich)
for 24 h, then washed under a running tap and preserved in 70%
ethanol. The samples were dehydrated in ascending grades of
ethanol, cleared in xylene (Sigma-Aldrich), embedded in Paraplast
Plus (Sigma-Aldrich) and sectioned to 5 µm thickness. Tissue
sections were mounted onto positively charged, coated slides
(Thermo Fisher Scientific, Inc.). Sections were stained with
hematoxylin and eosin (Sigma-Aldrich) for studying the general
histological analyses (13).
Immunohistochemistry
For immunohistochemical detection of insulin
granules, paraffin-embedded sections of the pancreas were
deparaffinized in xylene, rehydrated in descending grades of
alcohol and then immersed in 0.3% hydrogen peroxide (Sigma-Aldrich)
for 30 min to inactivate endogenous peroxidase activity. The
samples were heated in 10 mM citrate buffer (Sigma-Aldrich) at
121°C for 30 min for antigen retrieval, blocked in 5% normal serum
(Sigma-Aldrich) for 20 min, and incubated with a rabbit polyclonal
anti-insulin [dilution, 1:100 in phosphate-buffered saline (PBS);
sc-9168; Santa Cruz Biotechnology Inc., Dallas, TX, USA] antibody
overnight at 4°C. Subsequently, three PBS washes were performed and
the sections were incubated with a goat anti-rabbit
biotin-conjugated secondary antibody (dilution, 1:2,000 in PBS;
sc-2040; Santa Cruz Biotechnology, Inc.) for 20 min at room
temperature. After further incubation with horseradish
peroxidase-labeled streptavidin (Vector Laboratories, Inc.,
Burlingame, CA, USA), the antibody binding was visualized with
diaminobenzidine (Sigma-Aldrich) and sections were counterstained
with hematoxylin. Negative controls, in which the primary or
secondary antibodies, or the avidin-biotin complex reagent was
omitted, produced no positive staining. Positive controls were used
according to the instructions provided by the manufacturer's of the
primary antibodies.
Photomicrography
Photomicrographs were captured using a Leica DM LB
light microscope (Leica Microsystems, Inc., Buffalo Grove, IL, USA)
and a Leica EC3 digital camera (Leica Microsystems, Inc.).
Statistic analysis
Statistical analysis was performed using analysis of
variance and Scheffe's protected least-significant difference test
on SPSS software, version 13.0 (SPSS, Inc., Chicago, IL, USA).
P<0.05 was considered to indicate a statistically significant
difference. Results are expressed as the mean ± standard error.
Results
Effect of curcumin on the plasma levels
of biochemical parameters in diabetic rats
In the present study, plasma levels of the
anti-oxidant enzymes, SOD and GPx, were significantly decreased in
diabetic rats compared with controls (Table II), which may be caused by
oxidative stress generated by the development of DM. Further
reduction of these anti-oxidant enzyme levels was observed in
diabetic rats treated with NC, however, the levels were increased
to a level similar to that of the control by curcumin
supplementation (Table II). The
plasma concentrations of the oxidative markers, MDA and
γ-glutamyltranspeptidase (GGT), and the lipid profile, including
cholesterol, TG and phospholipids, were increased in diabetic rats
compared with control rats. This increase was enhanced by NC
treatment. Curcumin supplementation reversed these effects of DM
and NC. The diabetic nephropathy markers, urea and creatinine, were
increased in rats of the diabetic group compared with levels in the
control group. These were further enhanced in the DM + NC group,
while almost normalized with curcumin supplementation.
 | Table IISerum biochemical analysis. |
Table II
Serum biochemical analysis.
| Parameter | Control | DM | DM + NC | DM + NC + Cr |
|---|
| Glucose
(mg/dl) | 101.00±0.70 |
479.30±50.00a |
416.00±49.50a |
483.30±47.10a |
| Superoxide
dismutase (U/ml) | 11.67±0.16 | 7.94±0.17a | 5.80±2.00a,b | 10.73±0.30c |
| Glutathione
peroxidase (U/ml) | 115.67±3.80 | 85.67±3.30a | 69.00±0.71a,b | 105.30±9.40c |
| Malondialdehyde
(nmol/ml) | 3.00±0.80 | 4.40±2.10 | 8.74±1.20b | 3.63±0.16c |
|
γ-Glutamyltranspeptidase (U/l) | 4.91±0.50 | 7.53±0.41a | 10.50±0.71a,b | 6.10±1.10c |
| Cholesterol
(mg/dl) | 72.30±2.59 | 95.30±0.47a | 114.00±0.71a,b | 79.70±0.94c |
| Triacylglycerol
(mg/dl) | 52.30±8.00 | 63.30±12.50 | 81.70±4.50a,b | 61.70±14.00c |
| High density
lipoprotein (mg/dl) | 18.70±1.20 | 15.30±0.50a | 11.30±1.20a,b | 21.00±1.01c |
| Phospholipid
(mg/dl) | 91.00±2.80 | 94.70±3.80 | 122.30±4.00a,b | 92.70±12.90c |
| Urea (mg/dl) | 68.30±9.33 | 95.00±4.95a |
138.00±24.75a,b | 85.33±21.50b |
| Creatinine
(mg/dl) | 1.09±0.07 | 2.30±0.061a | 2.88±0.18a,b | 1.60±0.23c |
| Nitric oxide
(µmol/l) | 22.70±1.70 | 62.00±4.70a | 39.00±0.70b | 28.70±0.30c |
Effect of curcumin on the mRNA expression
of gene markers of kidney damage
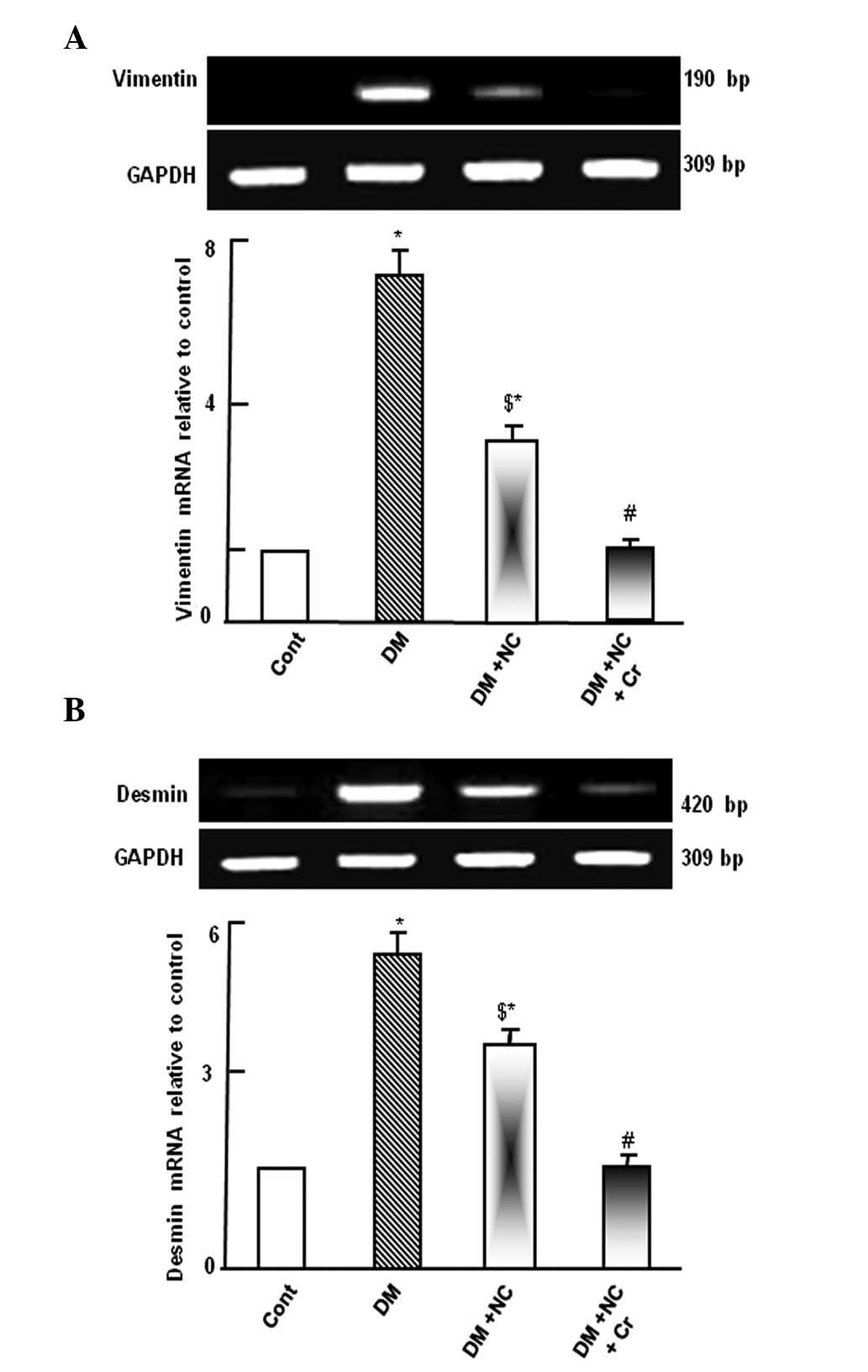
Diabetic rats demonstrated upregulation of vimentin
and desmin mRNA expression levels compared with control rats
(P<0.05; Fig. 1). Treatment of
diabetic rats with NC decreased the expression levels of these
genes compared with untreated diabetic rats (P<0.05), however,
the expression remained increased compared with the control group
(P<0.05). Following curcumin treatment, the DM and NC-induced
upregulation of vimentin and desmin mRNA expression levels were
comparable to the control group (Fig.
1).
Effect of curcumin on the mRNA expression
of gap junction and podocytes integrity gene markers
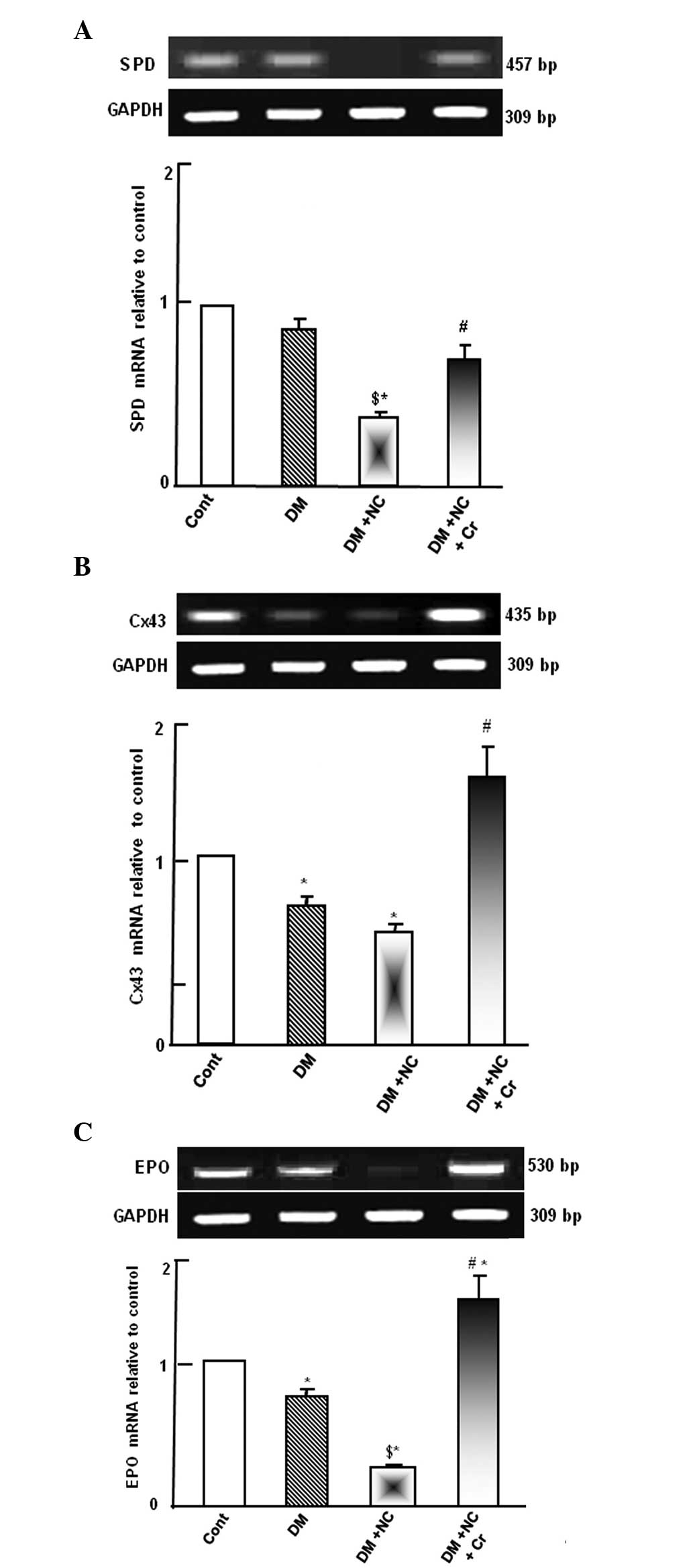
Diabetic rats demonstrated no changes to SPD mRNA
expression level. However, diabetic rats treated with NC exhibited
significantly reduced SPD mRNA expression levels compared with
diabetic or control rats (P<0.05; Fig. 2A). Diabetic rats treated with NC
and curcumin exhibited increased levels of SPD mRNA expression
compared with the diabetic rats with NC treatment (P<0.05;
Fig. 2A).
DM significantly decreased the Cx43 mRNA expression
compared with the control levels (P<0.05; Fig. 2B). Administration of NC to DM rats
resulted in a further reduction of the Cx43 mRNA expression level.
However, curcumin supplementation abrogated the combined inhibitory
effect of DM and NC on the Cx43 mRNA, restoring its expression, as
compared with the DM + NC group (P<0.05; Fig. 2B).
Diabetic rats exhibited a downregulation of EPO mRNA
expression levels compared with the control rats (P<0.05;
Fig. 2C). NC treatment of diabetic
rats caused further suppression of EPO mRNA expression compared
with the diabetic group (P<0.05). Curcumin supplementation
prevented the diabetic and NC-induced suppression of EPO
(P<0.05) and further increased EPO expression levels compared
with control levels (P<0.05; Fig.
2C).
Effect of curcumin on mRNA expression of
kidney oxidative stress markers
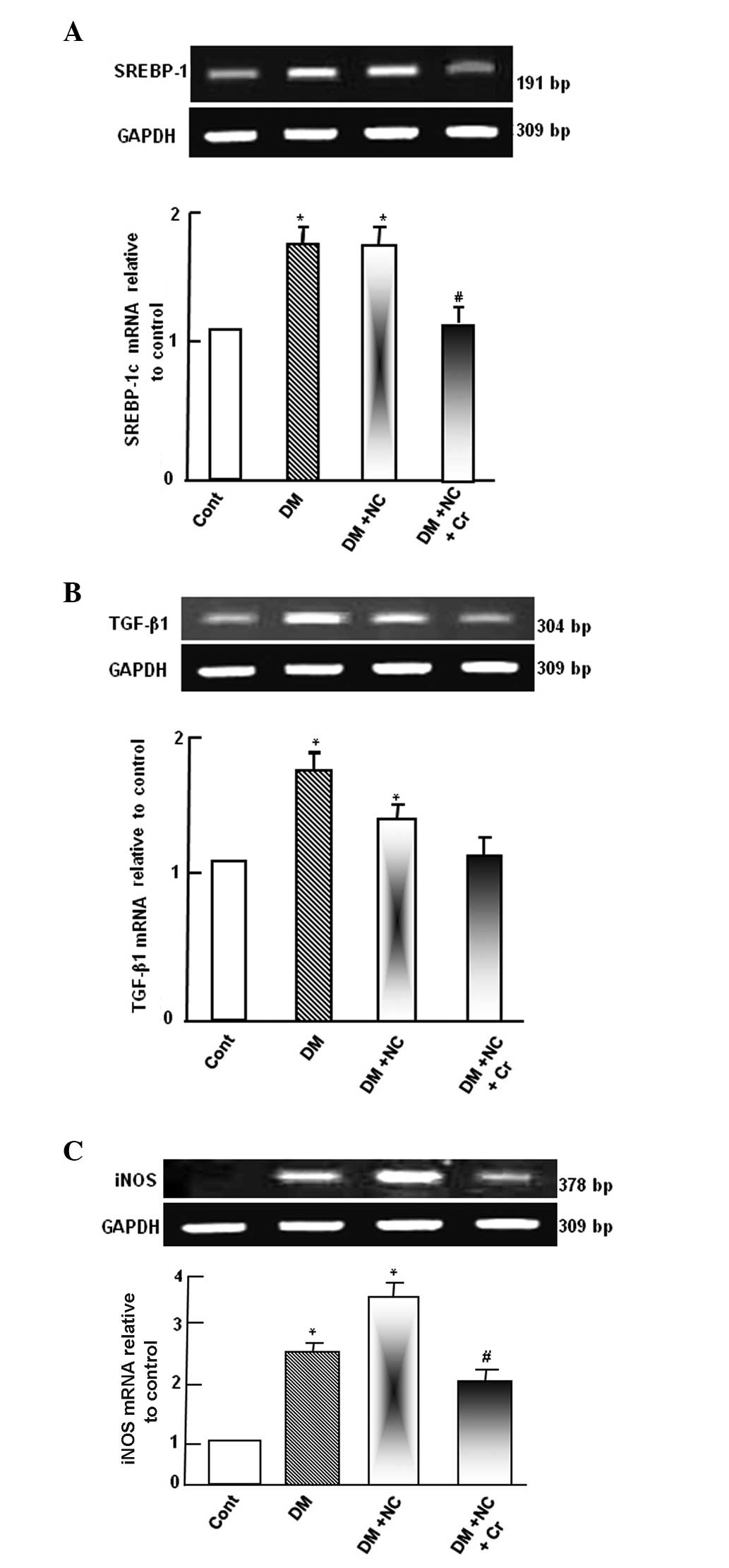
Diabetic rats exhibited upregulated SREBP-1c mRNA
expression levels compared with control rats (P<0.05; Fig. 3A). NC treatment of diabetic rats
did not demonstrate any significant change to the SREBP-1c mRNA
expression compared with diabetic rats. However, the DM-induced
SREBP-1c mRNA expression was abolished by curcumin supplementation,
as compared with the DM + NC group (P<0.05; Fig. 3A).
Diabetic rats exhibited a significant increase of
TGF-β1 mRNA expression levels compared with the control group
(P<0.05; Fig. 3B). NC treatment
of diabetic rats decreased TGF-β1 mRNA expression levels compared
with diabetic rats, however, the levels remained significantly
higher compared with the control rats (P<0.05). The DM-induced
increases in the TGF-β1 mRNA expression level was significantly
inhibited by curcumin supplementation, as compared with the DM + NC
group (P<0.05), and was normalized to control levels (Fig. 3B).
Diabetic rats exhibited upregulated iNOS mRNA
expression levels compared with control rats. NC treatment of
diabetic rats further increased iNOS mRNA expression levels, as
compared with diabetic rats. However, the increased level of iNOS
mRNA was inhibited by curcumin supplementation, compared with the
DM and NC group (P<0.05; Fig.
3C).
Histopathological findings
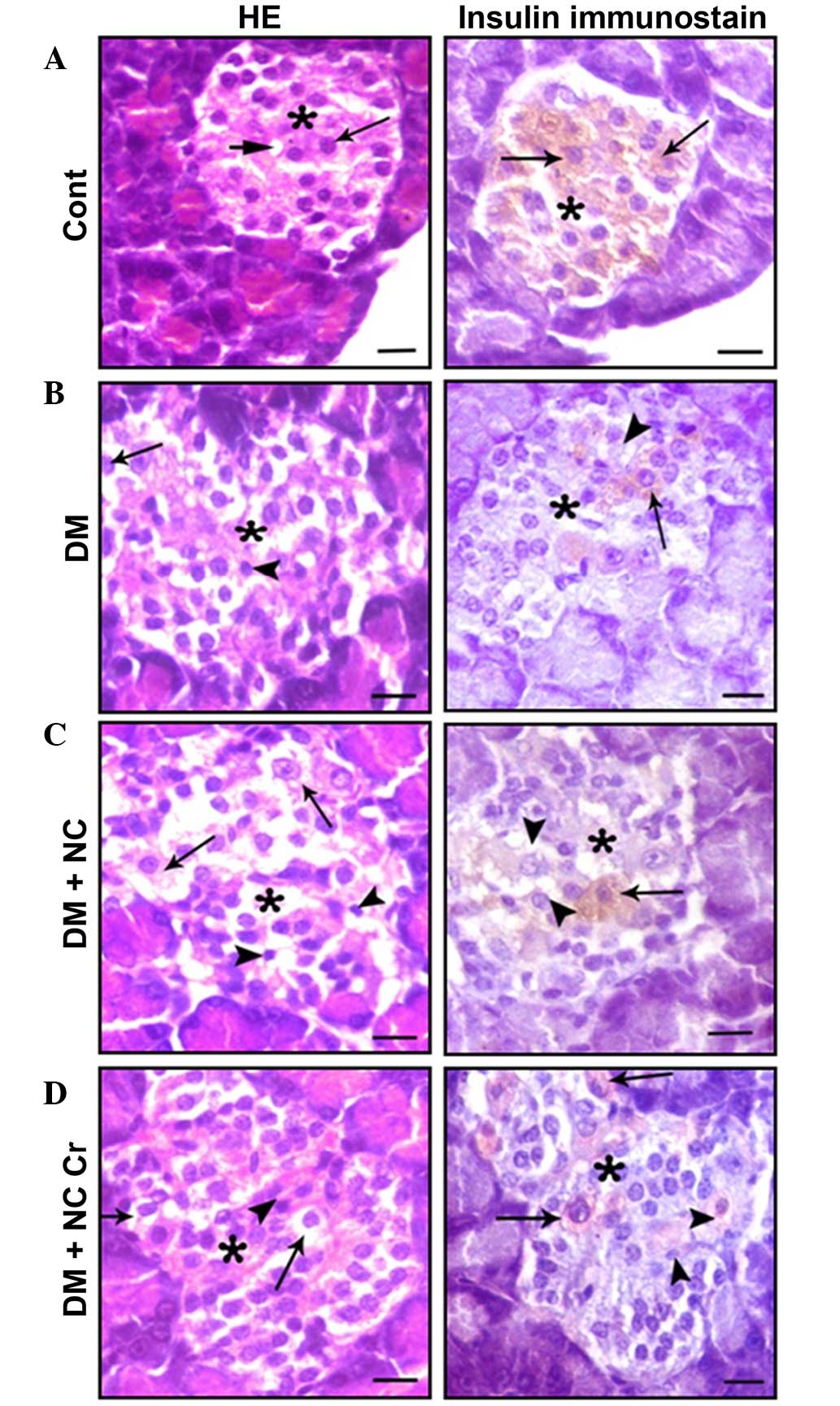
The effect of curcumin on the histological structure
of the pancreas was investigated. Pancreatic sections from control
rats exhibited islets of Langerhans cells, and contained lightly
stained acidophilic cells arranged in branching and anastomosing
cords (Fig. 4A). Sections
immunostained for the presence of insulin displayed positive
β-cells within the islets (Fig.
4A). Sections from diabetic rats displayed damaged islets,
represented by cytoplasmic vacuolations, pyknotic nuclei and a
decrease in insulin antibody reaction (Fig. 4B). β-Cells from diabetic rats that
received NC exhibited more severe islet alterations (Fig. 4C). Sections from diabetic rats that
received NC and curcumin exhibited decreased β-cell damage and
increased numbers of positive insulin-reacted cells (Fig. 4D).
Effect of curcumin on the kidney
histological structure
Kidney sections of control rats displayed the cortex
to be occupied by renal corpuscles and surrounding proximal and
distal convoluted tubules (Fig.
5A, left panel), and the medulla tissue exhibited straight
proximal and distal tubules, collecting ducts and thin tubules
distinguished from medullary blood capillaries (Fig. 5A, right panel). Kidney tissues from
DM rats displayed tissue damage, including glomerular hypertrophy,
hypercellularity, mesangial expansion, pronounced glomerular
sclerosis, severe tubulo-interstitial changes represented by lipid
accumulation in the cortical tubules, tubular dilatation,
interstitial mononuclear cell infiltration and fibroplasia
(Fig. 5B). NC treatment further
enhanced tissue damage, which was represented by increases in
glomerular hypertrophy, sclerosis, mesangial expansion and severe
tubulo-interstitial changes (Fig.
5C). However, curcumin treatment reduced the incidence of
glomerular sclerosis, lipid accumulation and tubular dilatation
compared with that of diabetic rats receiving NC without curcumin
treatment (Fig. 5D).
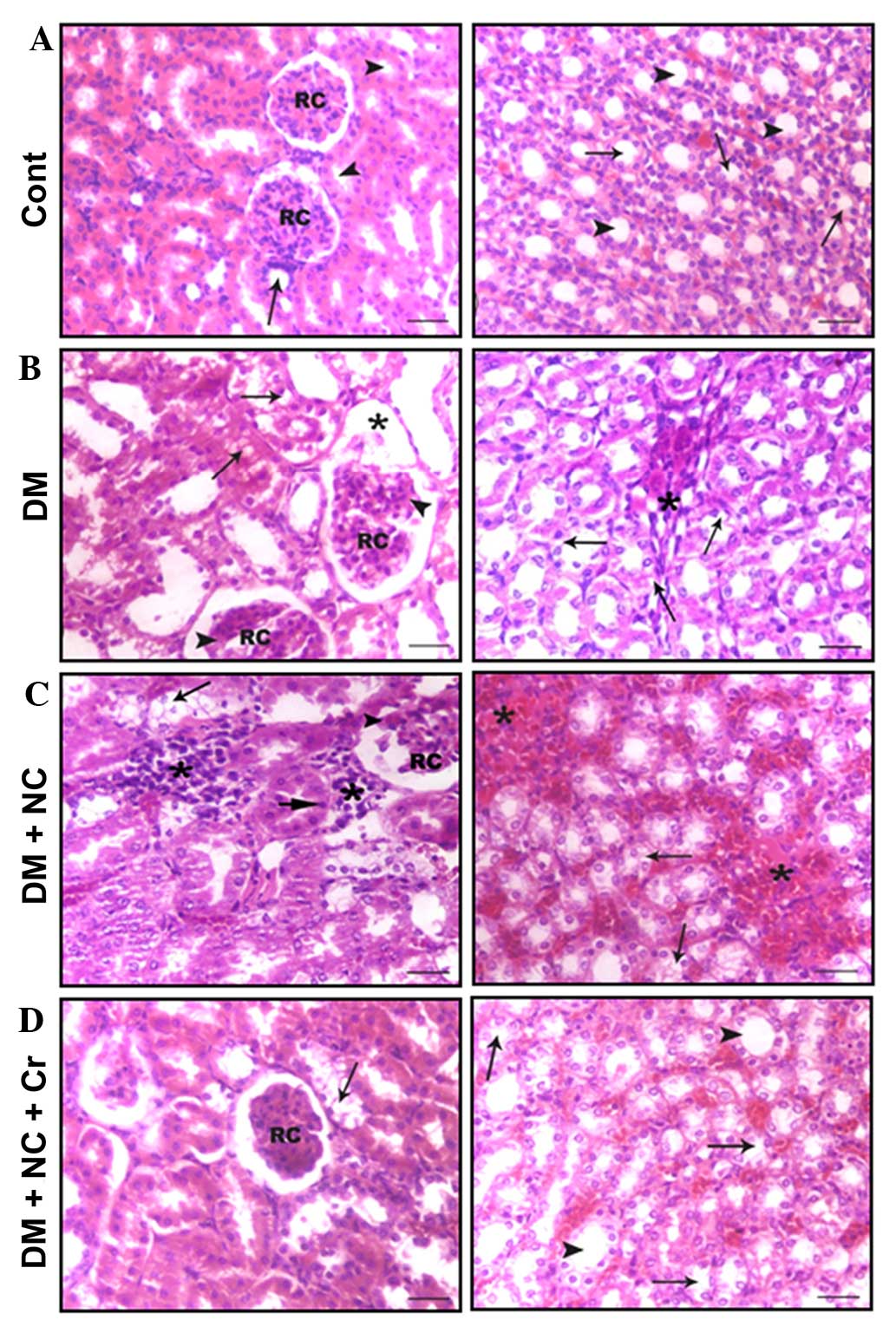 | Figure 5Effect of curcumin on the
histological structure of the cortex (left panel) and medulla
(right panel) of the rat kidneys (hematoxylin and eosin staining).
(A) The cortex of the control rats had RC and proximal (arrowheads)
and distal (arrow) convoluted tubules, and the medulla had a normal
structure with thin tubules (arrows) and collecting ducts
(arrowheads). (B) The cortex of the DM rats had enlarged RC, with
glomerular sclerosis (arrowheads) and expansion (asterisk), and
tubular lipid deposition (arrows) and dilatation. The medulla
exhibited tubular dilatation, lipid deposition (arrows),
interstitial nodular sclerosis and fibroplasia (asterisk). (C) The
cortex from DM + NC rats had enlarged RC, with glomerular sclerosis
and expansion (arrowheads), tubular lipid deposition (arrows) and
pyknosis of a number of tubular cells (long-head arrow), and
interstitial leukocytic infiltration (asterisk); the medulla
exhibited tubular lipid deposition (arrows) and pronounced
interstitial nodular sclerosis and fibroplasia (asterisks). (D) The
cortex from DM + NC + Cr rats exhibited normal sized RC and a lower
incidence of tubular lipid deposition (arrows), and the medulla
displayed a normal histological structure of thin tubules (arrows)
and collecting ducts (arrowheads). Scale bar = 30 µm. RC,
renal corpuscles; DM, diabetes mel-litus; NC, nicotine; Cr,
curcumin. |
Discussion
The results of the present study demonstrated
vimentin and desmin mRNA levels to be upregulated in diabetic rats.
This indicates tissue injury, as vimentin is an important marker of
tubular epithelial-mesenchymal transition (EMT) (14). Desmin upregulation is considered to
be a marker of podocyte injury (15). The downregulation of vimentin and
desmin in diabetic rats by NC indicated that NC-induced DN
exaggeration may require vimentin and desmin upregulation. However,
their normalization following curcumin supplementation suggested
that curcumin protects against DN. The results of the current study
are in agreement with a previous finding demonstrating that fasudil
(a renal anti-fibrotic drug) prevented high glucose-induced EMT via
decreased vimentin expression (16).
The slit diaphragm is important in the prevention of
glomerular protein leakage during physiological and pathological
processes (17). The slit
diaphragm consists of proteins specifically expressed by podocytes,
including nephrin, podocin, and SPD (18). The observation of reduced
expression of SPD and Cx43 mRNA levels in diabetic rats in the
present study corroborates previous studies that have demonstrated
Cx43 reduction in the kidneys of STZ-diabetic rats (19) and reduced SPD expression in the
glomeruli of patients with DM (20). The further downregulation of SPD
and Cx43 mRNA levels by NC exposure implies that the NC-induced
progression of DN may act via gap junctions and perturbation of
podocytes. The reduction of SPD mRNA expression levels in diabetic
rats may indicate the loss of podocytes in the urine, and its
further reduction in diabetic rats treated with NC may indicate
that NC increases the loss of podocytes in urine. Previous studies
have demonstrated increased gene expression levels of podocyte
markers, including SPD, in the urine of patients with DN, implying
increased excretion of podocytes (21). The prevention of the suppressive
effect of DM and NC on SPD and Cx43 mRNA levels by curcumin
indicates that curcumin may protect podocytes and renal gap
junctions against the oxidative stress caused by DM and
smoking.
The downregulation of EPO mRNA expression levels in
diabetic rats may be associated with the high risk of anemia, as
patients with DN have a greater risk of developing anemia (22). DM-associated events in DN, such as
tubulo-interstitial damage, result in the impairment of EPO
production by peritubular fibroblasts, leading to the development
of anemia (23). In the present
study, the further suppression of EPO mRNA levels in diabetic rats
exposed to NC indicated the ability of NC to increase cumulative
oxidative stress and suppress EPO. Smoking during pregnancy was
previously reported to be associated with a high risk of
compromised child development due to maternal anemia and fetal
hypoxia (24). Curcumin protected
EPO mRNA expression levels from the combined inhibition by DM and
NC, indicating that curcumin may be able to protect the kidney from
anemia caused by DM and NC. A previous study suggested that EPO
administration may reduce anemia and slow the progression of CKD
(25). EPO upregulation by
curcumin in the current study may also represent a pathway via
which curcumin mediates its reported protective activity against
obesity-induced oxidative stress in mice (26).
Renal SREBP-1c upregulation is an important
mechanism leading to renal TG accumulation and DN (27). In the current study, SREBP-1c mRNA
upregulation was indicated, consistent with trends observed in
previous studies on mesangial cells (28) and rats with type 1 DM (29). TGF-β1 is considered as one of the
main cytokines that aggravates DN (30). ECM accumulation is one of the
characteristic structural abnormalities associated with DN, in
addition to kidney hypertrophy and increased thickness of the
glomerular basement membrane (31). In the present study, the
upregulation TGF-β1 in diabetic rats may be caused by SREBP-1c
activation. Previously, high glucose-induced TGF-β1 upregulation
was demonstrated to be caused by the binding of activated SREBP-1c
to the TGF promoter (28). In the
current study, the partial downregulation of TGF-β1 caused by NC in
DM rats may indicate that NC is able to partially inactivate the
SREBP-1c protein, as the mRNA expression level of SREBP-1c was not
affected. NC was previously demonstrated to improve the serum lipid
profile, decrease insulin serum levels, and to reduce steatosis and
inflammation (32). The
normalization of the SREBP-1 mRNA expression levels by curcumin
supplementation in diabetic rats may be important for the
normalization of TGF-β1 and may represent a pathway by which
curcumin protects the kidney from DN. Zhenqing recipe (a Chinese
herbal prescription for improvement of renal function) has been
reported to improve DN through the downregulation of SREBP-1c mRNA,
which is overexpressed in rats with type 2 DM (33). Additionally, TEMPOL (a
SOD-mimicking drug) was reported to ameliorate the pathological
changes in diabetic glomeruli by reducing the TGF-β1 expression
levels (34). The results of the
current study indicated that NC has 2 opposing effects on the
kidney as follows: A protective effect represented by the
downregulation of vimentin and desmin expression; and a detrimental
effect represented by the downregulation of SPD, Cx43 and EPO and
the upregulation of iNOS mRNA expression levels. However, the net
effects of NC on the kidney demonstrate an increase in the markers
of kidney function (creatinine, urea and MDA), further reductions
in the levels of GPx and SOD in the serum, and the
histopathological changes to the kidney structure. These changes
demonstrate the overwhelmingly destructive effects of NC. Notably,
destructive effects of combined DM and NC were ameliorated by
curcumin supplementation.
The morphological changes in pancreatic islets from
STZ-induced diabetic rats, including cytoplasmic vacuolations,
pyknotic nuclei and the decrease in insulin-positive granules, are
in agreement with previous reports (35). STZ-induced DM is caused by
oxidative stress on β-cells, which are particularly susceptible to
oxidative insult due to their relatively low levels of antioxidants
(36). NC-induced oxidative stress
(37) increases the insult on the
cells and may cause NC-induced β-cell apoptosis and permanent
β-cell loss (38,39). Abnormal lipid deposits have been
previously reported in the renal cortical tubules of patients with
DM (40) and have been proposed to
participate in the pathogenesis of DN in STZ-induced diabetic rats
(41). Curcumin amelioration of
Langerhans lesions in rat pancreatic islets may be due to the
anti-oxidative activity of curcumin, which may be mediated by
decreased iNOS and/or increased EPO expression levels.
Additionally, curcumin may exert its anti-oxidative effect via
increased ROS scavenging activity through the upregulation of SOD
and GPx mRNA levels. The infiltration of macrophages and mesangial
accumulation of ECM proteins are commonly associated with DN
(42). In a previous study,
curcumin exhibited renoprotective effects in STZ-induced type 1 DN,
it reduced the proteinuria and pathological changes associated with
DM through the inhibition of macrophage infiltration. Curcumin was
suggested to have antifibrotic and anti-inflammatory effects
(43).
Increased lipid deposition in the kidney tubules of
diabetic rats indicated disturbance in lipid metabolism caused by
oxidative stress (44). Additional
lipid deposition due to NC exposure indicated that NC increases
oxidative stress further in DM. The increased lipid deposition in
the kidney contributes to cellular damage and to the progression of
DN (41). The ameliorative effect
of curcumin against DM and the effects of NC may be due to its
effects on a wide range of molecular targets that control lipid
accumulation (45). Curcumin was
reported to protect against DN progression through the
phosphorylation of AMP-activated kinase and SREBP-1c suppression,
and the reduction of renal TG accumulation (41).
In conclusion, the current study investigated the
mechanisms through which curcumin protects against the combined
oxidative stress of DM and NC exposure. Curcumin acts through
normalizing the expression levels of various factors important
during DN progression. This includes normalization of the levels of
the suppressed antioxidants, SPD, Cx43 and EPO, and the DM-induced
factors, MDA, GGT, vimintin, desmin, SREBP-1c, TGF-β1 and iNOS.
Clarification of the underlying mechanisms of curcumin activity may
provide novel therapeutic targets for the treatment of DN.
Acknowledgments
The present work was supported in part by a research
project grant (no. 1/434/2799) from Taif University. The authors
are thankful to Associate Professor Samir Ahmed El-shzely, at the
Department of Biotechnology, College of Science (Taif University)
and to Dr Mohamed Abdo Nassan (Department of Pathology, Faculty of
Veterinary Medicine, Zagazig University, Egypt) for their help with
the practical part of the study.
Abbreviations:
|
DN
|
diabetic nephropathy
|
|
STZ
|
streptozotocin
|
|
HDL
|
high density lipoprotein
|
|
SOD
|
superoxide dismutase
|
|
GPx
|
glutathione peroxidase
|
|
SPD
|
synaptopodin
|
|
Cx43
|
connexin 43
|
|
EPO
|
erythropoietin
|
|
TG
|
triacylglycerol
|
|
MDA
|
malondialdehyde
|
|
GGT
|
γ-glutamyltranspeptidase or
glutathione hydrolase
|
|
SREBP-1
|
sterol regulatory element binding
protein 1
|
|
iNOS
|
inducible nitric oxide synthase
|
|
TGF-β1
|
transforming growth factor β1
|
|
DM
|
diabetes mellitus
|
|
EMT
|
epithelial-mesenchymal transition
|
|
ECM
|
extracellular matrix
|
References
|
1
|
Gao Q, Shen W, Qin W, Zheng C, Zhang M,
Zeng C, Wang S, Wang J, Zhu X and Liu Z: Treatment of db/db
diabetic mice with triptolide: A novel therapy for diabetic
nephropathy. Nephrol Dial Transplant. 25:3539–3547. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Forbes JM, Coughlan MT and Cooper ME:
Oxidative stress as a major culprit in kidney disease in diabetes.
Diabetes. 57:1446–1454. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rossing P, Hougaard P and Parving HH: Risk
factors for development of incipient and overt diabetic nephropathy
in type 1 diabetic patients: A 10-year prospective observational
study. Diabetes Care. 25:859–864. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sawicki PT, Didjurgeit U, Mühlhauser I,
Bender R, Heinemann L and Berger M: Smoking is associated with
progression of diabetic nephropathy. Diabetes Care. 17:126–131.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mühlhauser I, Bender R, Bott U, Jörgens V,
Grüsser M, Wagener W, Overmann H and Berger M: Cigarette smoking
and progression of retinopathy and nephropathy in type 1 diabetes.
Diabet Med. 13:536–543. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
De Cosmo S, Lamacchia O, Rauseo A, Viti R,
Gesualdo L, Pilotti A, Trischitta V and Cignarelli M: Cigarette
smoking is associated with low glomerular filtration rate in male
patients with type 2 diabetes. Diabetes Care. 11:2467–2470. 2006.
View Article : Google Scholar
|
|
7
|
Hallan SI and Orth SR: Smoking is a risk
factor in the progression to kidney failure. Kidney Int.
80:516–523. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Winer S, Chan Y, Paltser G, Truong D, Tsui
H, Bahrami J, Dorfman R, Wang Y, Zielenski J, Mastronardi F, et al:
Normalization of obesity-associated insulin resistance through
immunotherapy. Nat Med. 15:921–929. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Aggarwal BB, Sundaram C, Malani N and
Ichikawa H: Curcumin: The Indian solid gold. Adv Exp Med Biol.
595:1–75. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sharma S, Kulkarni SK and Chopra K:
Curcumin, the active principle of turmeric (Curcuma longa),
ameliorates diabetic nephropathy in rats. Clin Exp Pharmacol
Physiol. 33:940–945. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Meghana K, Sanjeev G and Ramesh B:
Curcumin prevents streptozotocin-induced islet damage by scavenging
free radicals: A prophylactic and protective role. Eur J Pharmacol.
577:183–191. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chiu J, Khan ZA, Farhangkhoee H and
Chakrabarti S: Curcumin prevents diabetes-associated abnormalities
in the kidneys by inhibiting p300 and nuclear factor-kappaB.
Nutrition. 25:964–972. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Suvarna SK, Layton C and Bancroft JD: The
hematoxylins and eosin. Bancroft's Theory and Practice of
Histological Techniques. 7th edition. Churchill Livingstone;
London: pp. 172–186. 2013
|
|
14
|
Liu DG and Wang TM: Role of connective
tissue growth factor in experimental radiation nephropathy in rats.
Chin Med J (Engl). 121:1925–1931. 2008.
|
|
15
|
Zheng CX, Chen ZH, Zeng CH, Qin WS, Li LS
and Liu ZH: Triptolide protects podocytes from puromycin
aminonucleoside induced injury in vivo and in vitro. Kidney Int.
74:596–612. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gu L, Gao Q, Ni L, Wang M and Shen F:
Fasudil inhibits epithelial-myofibroblast transdifferentiation of
human renal tubular epithelial HK-2 cells induced by high glucose.
Chem Pharm Bull (Tokyo). 61:688–694. 2013. View Article : Google Scholar
|
|
17
|
Lan X, Rai P, Chandel N, Cheng K, Lederman
R, Saleem MA, Mathieson PW, Husain M, Crosson JT, Gupta K, et al:
Morphine induces albuminuria by compromising podocyte integrity.
PLoS One. 8:e557482013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Benzing T: Signaling at the slit
diaphragm. J Am Soc Nephrol. 15:1382–1391. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Satriano J, Mansoury H, Deng A, Sharma K,
Vallon V, Blantz RC and Thomson SC: Transition of kidney tubule
cells to a senescent phenotype in early experimental diabetes. Am J
Physiol Cell Physiol. 299:C374–C380. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Miyauchi M, Toyoda M, Kobayashi K, Abe M,
Kobayashi T, Kato M, Yamamoto N, Kimura M, Umezono T and Suzuki D:
Hypertrophy and loss of podocytes in diabetic nephropathy. Intern
Med. 48:1615–1620. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zheng M1, Lv LL, Ni J, Ni HF, Li Q, Ma KL
and Liu BC: Urinary podocyte-associated mRNA profile in various
stages of diabetic nephropathy. PLoS One. 6:e204312011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dousdampanis P, Trigka K and Fourtounas C:
Prevalence of anemia in patients with type II diabetes and mild to
moderate chronic kidney disease and the impact of anti-RAS
medications. Saudi J Kidney Dis Transpl. 25:552–557. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Singh DK, Winocour P and Farrington K:
Erythropoietic stress and anemia in diabetes mellitus. Nat Rev
Endocrinol. 5:204–210. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Habek D, Habek JC, Ivanisević M and
Djelmis J: Fetal tobacco syndrome and perinatal outcome. Fetal
Diagn Ther. 6:367–371. 2002. View Article : Google Scholar
|
|
25
|
Gouva C, Nikolopoulos P, Ioannidis JP and
Siamopoulos KC: Treating anemia early in renal failure patients
slows the decline of renal function: A randomized controlled trial.
Kidney Int. 66:753–760. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Martínez-Morúa A, Soto-Urquieta MG,
Franco-Robles E, Zúñiga-Trujillo I, Campos-Cervantes A,
Pérez-Vázquez V and Ramírez-Emiliano J: Curcumin decreases
oxidative stress in mitochondria isolated from liver and kidneys of
high-fat diet-induced obese mice. J Asian Nat Prod Res. 15:905–915.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ishigaki N, Yamamoto T, Shimizu Y,
Kobayashi K, Yatoh S, Sone H, Takahashi A, Suzuki H, Yamagata K,
Yamada N and Shimano H: Involvement of glomerular SREBP-1c in
diabetic nephropathy. Biochem Biophys Res Commun. 364:502–508.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Uttarwar L, Gao B, Ingram AJ and Krepinsky
JC: SREBP-1 activation by glucose mediates TGF-β upregulation in
mesangial cells. Am J Physiol Renal Physiol. 302:F329–F341. 2012.
View Article : Google Scholar
|
|
29
|
Jun H, Song Z, Chen W, Zanhua R, Yonghong
S, Shuxia L and Huijun D: In vivo and in vitro effects of SREBP-1
on diabetic renal tubular lipid accumulation and RNAi-mediated gene
silencing study. Histochem Cell Biol. 3:327–345. 2009. View Article : Google Scholar
|
|
30
|
Basile DP: Transforming growth factor-beta
as a target for treatment in diabetic nephropathy. Am J Kidney Dis.
38:887–892. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lv S, Liu G, Sun AG, Wang J, Cheng J, Wang
W, Liu X, Nie H and Guan G: Mesenchymal stem cells ameliorate
diabetic glomerular fibrosis in vivo and in vitro by inhibiting
TGF-β signalling via secretion of bone morphogenetic protein 7.
Diab Vasc Dis Res. 11:251–261. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Seoane-Collazo P, Martínez de Morentin PB,
Fernø J, Diéguez C, Nogueiras R and López M: Nicotine improves
obesity and hepatic steatosis and ER stress in diet-induced obese
male rats. Endocrinology. 155:1679–1689. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wen X, Zeng Y, Liu L, Zhang H, Xu W, Li N
and Jia X: Zhenqing recipe alleviates diabetic nephropathy in
experimental type 2 diabetic rats through suppression of SREBP-1c.
J Ethnopharmacol. 142:144–150. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Luan J, Li W, Han J, Zhang W, Gong H and
Ma R: Renal protection of in vivo administration of tempol in
streptozotocin-induced diabetic rats. J Pharmacol Sci. 119:167–176.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Simsek N, Kaya M, Kara A, Can I, Karadeniz
A and Kalkan Y: Effects of melatonin on islet neogenesis and beta
cell apoptosis in streptozotocin-induced diabetic rats: An
immunohistochemical study. Domest Anim Endocrinol. 43:47–57. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Akinola OB, Caxton Martins EA and Dini L:
Chronic treatment with ethanolic extract of the leaves of
Azadirachta indica ameliorates lesions of pancreatic islets in
streptozotocin diabetes. Int J Morphol. 28:291–302. 2010.
|
|
37
|
Bruin JE, Kellenberger LD, Gerstein HC,
Morrison KM and Holloway AC: Fetal and neonatal nicotine exposure
and postnatal glucose homeostasis: Identifying critical windows of
exposure. Journal of Endocrinology. 194:171–178. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bruin JE, Petre MA, Lehman MA, Raha S,
Gerstein HC, Morrison KM and Holloway AC: Maternal nicotine
exposure increases oxidative stress in the offspring. Free Radic
Biol Med. 44:1919–1925. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Abdul-Hamid M and Moustafa N: Protective
effect of curcumin on histopathology and ultrastructure of pancreas
in the alloxan treated rats for induction of diabetes. J Appl Zool.
66:169–179. 2013. View Article : Google Scholar
|
|
40
|
Ong AC, Jowett TP, Firth JD, Burton S,
Kitamura M and Fine LG: Human tubular-derived endothelin in the
paracrine regulation of renal interstitial fibroblast function. Exp
Nephrol. 2:1341994.PubMed/NCBI
|
|
41
|
Soetikno V, Suzuki K, Veeraveedu PT,
Arumugam S, Lakshmanan AP, Sone H and Watanabe K: Molecular
understanding of curcumin in diabetic nephropathy. Drug Discov
Today. 18:756–63. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sassy-Prigent C, Heudes D, Mandet C,
Bélair MF, Michel O, Perdereau B, Bariéty J and Bruneval P: Early
glomerular macrophage recruitment in streptozotocin-induced
diabetic rats. Diabetes. 49:466–475. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Soetikno V, Sari FR, Veeraveedu PT,
Thandavarayan RA, Harima M, Sukumaran V, Lakshmanan AP, Suzuki K,
Kawachi H and Watanabe K: Curcumin ameliorates macrophage
infiltration by inhibiting NF-κB activation and proinflammatory
cytokines in streptozotocin induced-diabetic nephropathy. Nutr
Metab (Lond). 8:352001. View Article : Google Scholar
|
|
44
|
Asaba K, Tojo A, Onozato ML, Goto A, Quinn
MT, Fujita T and Wilcox CS: Effects of NADPH oxidase inhibitor in
diabetic nephropathy. Kidney Int. 67:1890–1898. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Goel A, Kunnumakkara AB and Aggarwal BB:
Curcumin as 'Curecumin': From kitchen to clinic. Biochem Pharmacol.
75:787–809. 2008. View Article : Google Scholar
|