|
1
|
Raab MS, Podar K, Breitkreutz I,
Richardson PG and Anderson KC: Multiple myeloma. Lancet.
374:324–339. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Di Martino MT, Gullà A, Cantafio ME,
Lionetti M, Leone E, Amodio N, Guzzi PH, Foresta U, Conforti F,
Cannataro M, et al: In vitro and in vivo anti-tumor activity of
miR-221/222 inhibitors in multiple myeloma. Oncotarget. 4:242–255.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Huang EW, Xue SJ, Li XY, Xu SW, Cheng JD,
Zheng JX, Shi H, Lv GL, Li ZG, Li Y, et al: EEN regulates the
proliferation and survival of multiple myeloma cells by
potentiating IGF-1 secretion. Biochem Biophys Res Commun.
447:271–277. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
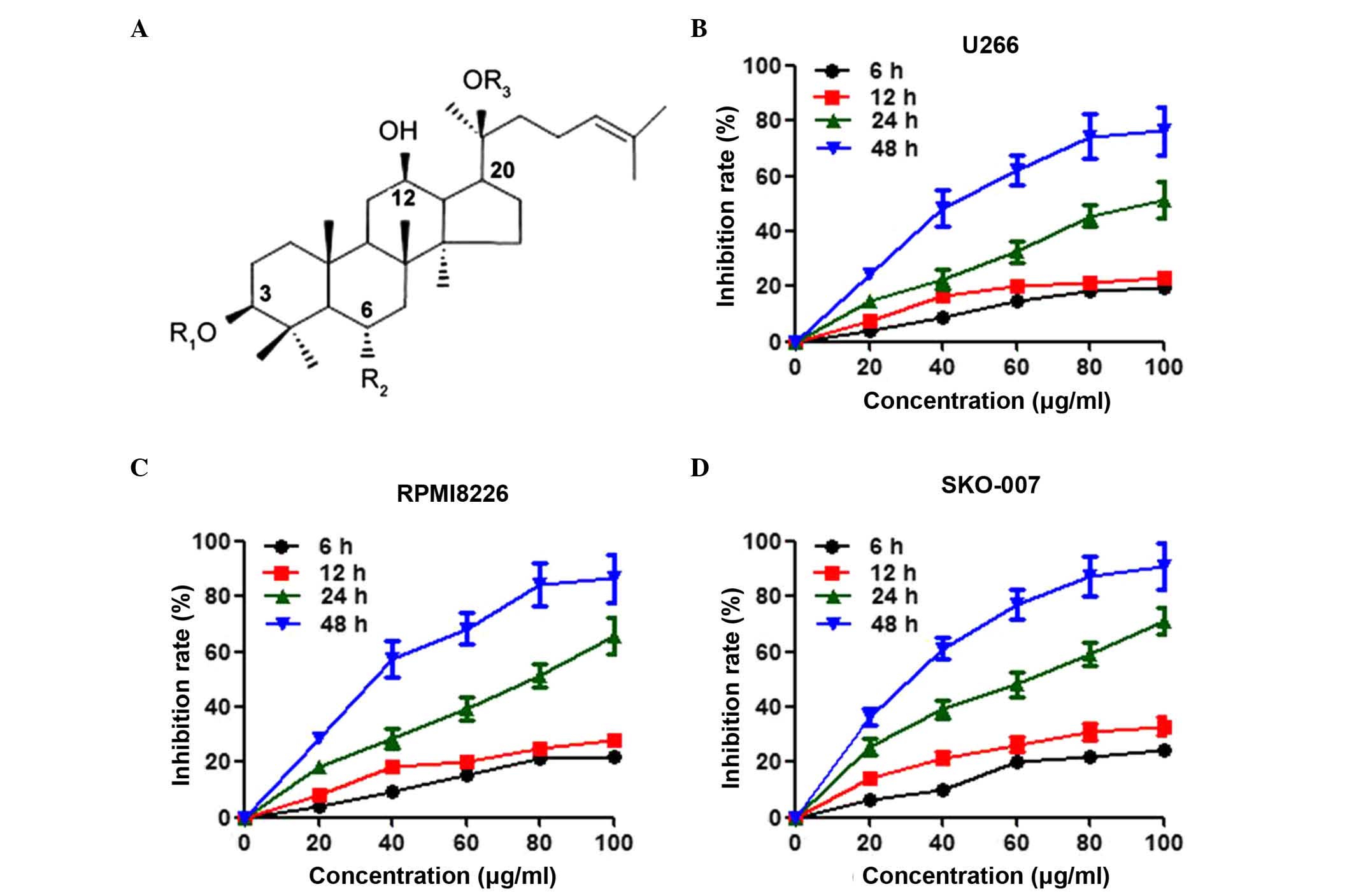
|
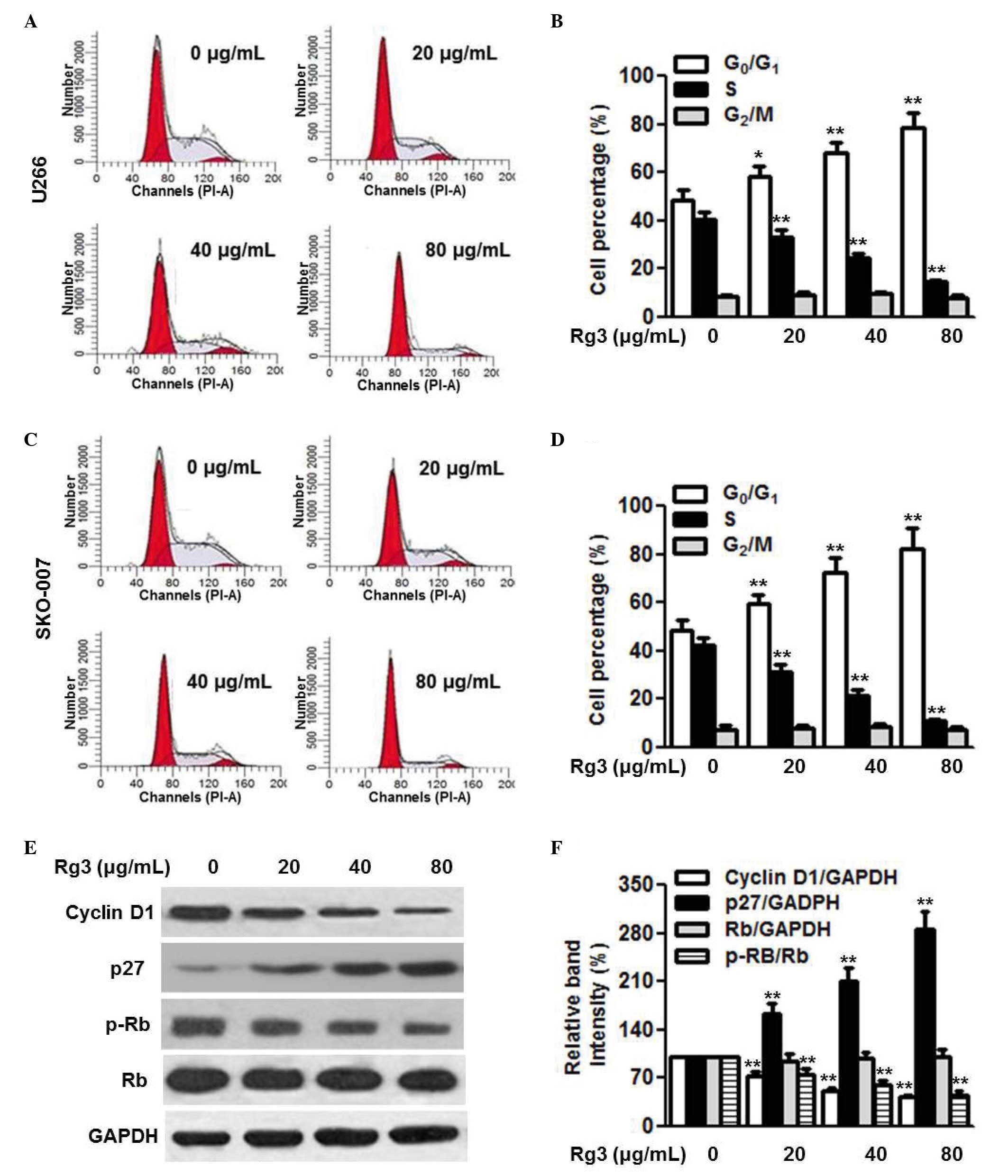
Kumar S and Rajkumar SV: Thalidomide and
lenalidomide in the treatment of multiple myeloma. Eur J Cancer.
42:1612–1622. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kyle RA and Rajkumar SV: Multiple myeloma.
Blood. 111:2962–2972. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Richardson PG, Mitsiades C, Schlossman R,
Ghobrial I, Hideshima T, Munshi N and Anderson KC: Bortezomib in
the front-line treatment of multiple myeloma. Expert Rev Anticancer
Ther. 8:1053–1072. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hideshima T, Chauhan D, Shima Y, Raje N,
Davies FE, Tai YT, Treon SP, Lin B, Schlossman RL, Richardson P, et
al: Thalidomide and its analogs overcome drug resistance of human
multiple myeloma cells to conventional therapy. Blood.
96:2943–2950. 2000.PubMed/NCBI
|
|
8
|
Brown RE, Stern S, Dhanasiri S and Schey
S: Lenalidomide for multiple myeloma: Cost-effectiveness in
patients with one prior therapy in England and Wales. Eur J Health
Econ. 14:507–514. 2013. View Article : Google Scholar
|
|
9
|
Lee JY, Jung KH, Morgan MJ, Kang YR, Lee
HS, Koo GB, Hong SS, Kwon SW and Kim YS: Sensitization of
TRAIL-induced cell death by 20(S)-ginsenoside Rg3 via CHOP-mediated
DR5 upregulation in human hepatocellular carcinoma cells. Mol
Cancer Ther. 12:274–285. 2013. View Article : Google Scholar
|
|
10
|
Park EH, Kim YJ, Yamabe N, Park SH, Kim
HK, Jang HJ, Kim JH, Cheon GJ, Ham J and Kang KS: Stereospecific
anticancer effects of ginsenoside Rg3 epimers isolated from
heat-processed American ginseng on human gastric cancer cell. J
Ginseng Res. 38:22–27. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yuan HD, Quan HY, Zhang Y, Kim SH and
Chung SH: 20(S)-Ginsenoside Rg3-induced apoptosis in HT-29 colon
cancer cells is associated with AMPK signaling pathway. Mol Med
Rep. 3:825–831. 2010.
|
|
12
|
Min JK, Kim JH, Cho YL, Maeng YS, Lee SJ,
Pyun BJ, Kim YM, Park JH and Kwon YG: 20(S)-Ginsenoside Rg3
prevents endothelial cell apoptosis via inhibition of a
mitochondrial caspase pathway. Biochem Biophys Res Commun.
349:987–994. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shan X, Fu YS, Aziz F, Wang XQ, Yan Q and
Liu JW: Ginsenoside Rg3 inhibits melanoma cell proliferation
through down-regulation of histone deacetylase 3 (HDAC3) and
increase of p53 acetylation. PLoS One. 9:e1154012014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim BM, Kim DH, Park JH, Surh YJ and Na
HK: Ginsenoside Rg3 inhibits constitutive activation of NF-kB
signaling in human breast cancer (MDA-MB-231) cells: ERK and Akt as
potential upstream targets. J Cancer Prev. 19:23–30. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zeng D, Wang J, Kong P, Chang C and Li J
and Li J: Ginsenoside Rg3 inhibits HIF-1α and VEGF expression in
patient with acute leukemia via inhibiting the activation of
PI3K/Akt and ERK1/2 pathways. Int J Clin Exp Pathol. 7:2172–2178.
2014.
|
|
16
|
Sin S, Kim SY and Kim SS: Chronic
treatment with ginsenoside Rg3 induces Akt-dependent senescence in
human glioma cells. Int J Oncol. 41:1669–1674. 2012.PubMed/NCBI
|
|
17
|
Jiang JW, Chen XM, Chen XH and Zheng SS:
Ginsenoside Rg3 inhibit hepatocellular carcinoma growth via
intrinsic apoptotic pathway. World J Gastroenterol. 17:3605–3613.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sedding DG, Hermsen J, Seay U, Eickelberg
O, Kummer W, Schwencke C, Strasser RH, Tillmanns H and
Braun-Dullaeus RC: Caveolin-1 facilitates mechanosensitive protein
kinase B (Akt) signaling in vitro and in vivo. Circ Res.
96:635–642. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Héron-Milhavet L, Franckhauser C, Rana V,
Berthenet C, Fisher D, Hemmings BA, Fernandez A and Lamb NJ: Only
Akt1 is required for proliferation, while Akt2 promotes cell cycle
exit through p21 binding. Mol Cell Biol. 26:8267–8280. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kuwabara I, Kuwabara Y, Yang RY, Schuler
M, Green DR, Zuraw BL, Hsu DK and Liu FT: Galectin-7 (PIG1)
exhibits pro-apoptotic function through JNK activation and
mitochondrial cytochrome c release. J Biol Chem. 277:3487–3497.
2002. View Article : Google Scholar
|
|
21
|
Cea M, Cagnetta A, Fulciniti M, Tai YT,
Hideshima T, Chauhan D, Roccaro A, Sacco A, Calimeri T, Cottini F,
et al: Targeting NAD+ salvage pathway induces autophagy in multiple
myeloma cells via mTORC1 and extracellular signal-regulated kinase
(ERK1/2) inhibition. Blood. 120:3519–3529. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Su F, Viros A, Milagre C, Trunzer K,
Bollag G, Spleiss O, Reis-Filho JS, Kong X, Koya RC, Flaherty KT,
et al: RAS mutations in cutaneous squamous-cell carcinomas in
patients treated with BRAF inhibitors. N Engl J Med. 366:207–215.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wajapeyee N, Serra RW, Zhu X, Mahalingam M
and Green MR: Oncogenic BRAF induces senescence and apoptosis
through pathways mediated by the secreted protein IGFBP7. Cell.
132:363–374. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Palazzolo I, Stack C, Kong L, Musaro A,
Adachi H, Katsuno M, Sobue G, Taylor JP, Sumner CJ, Fischbeck KH
and Pennuto M: Overexpression of IGF-1 in muscle attenuates disease
in a mouse model of spinal and bulbar muscular atrophy. Neuron.
63:316–328. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sun Y, Lin H, Zhu Y, Feng J, Chen Z, Li G,
Zhang X, Zhang Z, Tang J, Shi M, et al: A randomized, prospective,
multi-centre clinical trial of NP regimen (vinorelbine+cisplatin)
plus Gensing Rg3 in the treatment of advanced non-small cell lung
cancer patients. Zhongguo Fei Ai Za Zhi. 9:254–258. 2006.In
Chinese. PubMed/NCBI
|
|
26
|
Luo Y, Zhang P, Zeng HQ, Lou SF and Wang
DX: Ginsenoside Rg3 induces apoptosis in human multiple myeloma
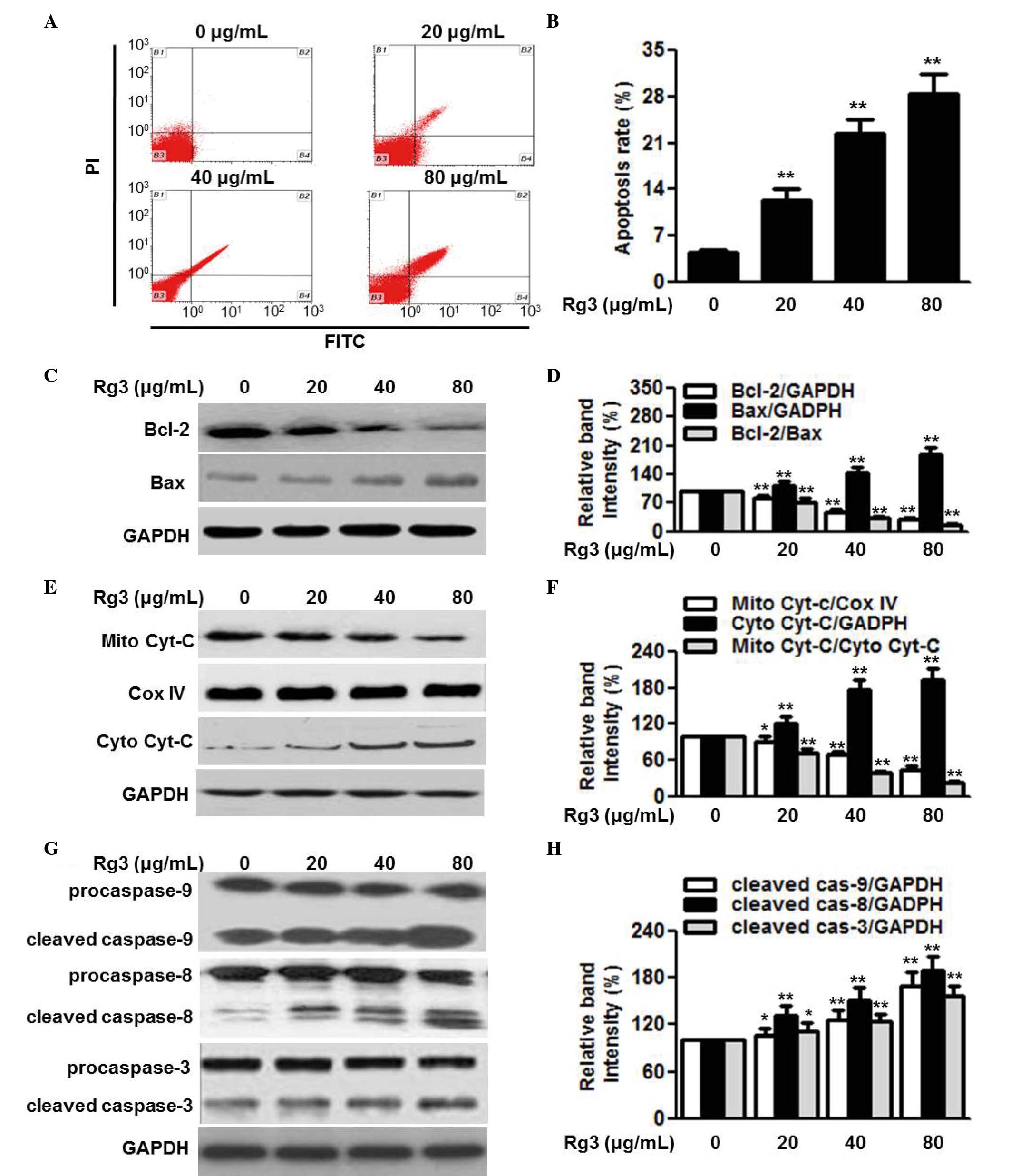
cells via the activation of Bcl-2-associated X protein. Mol Med
Rep. 12:3557–3562. 2015.PubMed/NCBI
|
|
27
|
Song Y, Hou J, Kang L and Gao S: Effect of
20 (S)-ginsenoside Rg3 on the proliferation inhibition and
secretion of vascular endothelial growth factor of multiple myeloma
cell line U266. Zhonghua Xue Ye Xue Za Zhi. 35:519–523. 2014.In
Chinese. PubMed/NCBI
|
|
28
|
Woo RA and Poon RY: Cyclin-dependent
kinases and S phase control in mammalian cells. Cell Cycle.
2:316–324. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Misiti F, Orsini F, Clementi ME, Lattanzi
W, Giardina B and Michetti F: Mitochondrial oxygen consumption
inhibition importance for TMT-dependent cell death in
undifferentiated PC12 cells. Neurochem Int. 52:1092–1099. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ott M, Gogvadze V, Orrenius S and
Zhivotovsky B: Mitochondria, oxidative stress and cell death.
Apoptosis. 12:913–922. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mignotte B and Vayssiere JL: Mitochondria
and apoptosis. Eur J Biochem. 252:1–15. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sharp ZD and Bartke A: Evidence for
down-regulation of phosphoinositide 3-kinase/Akt/mammalian target
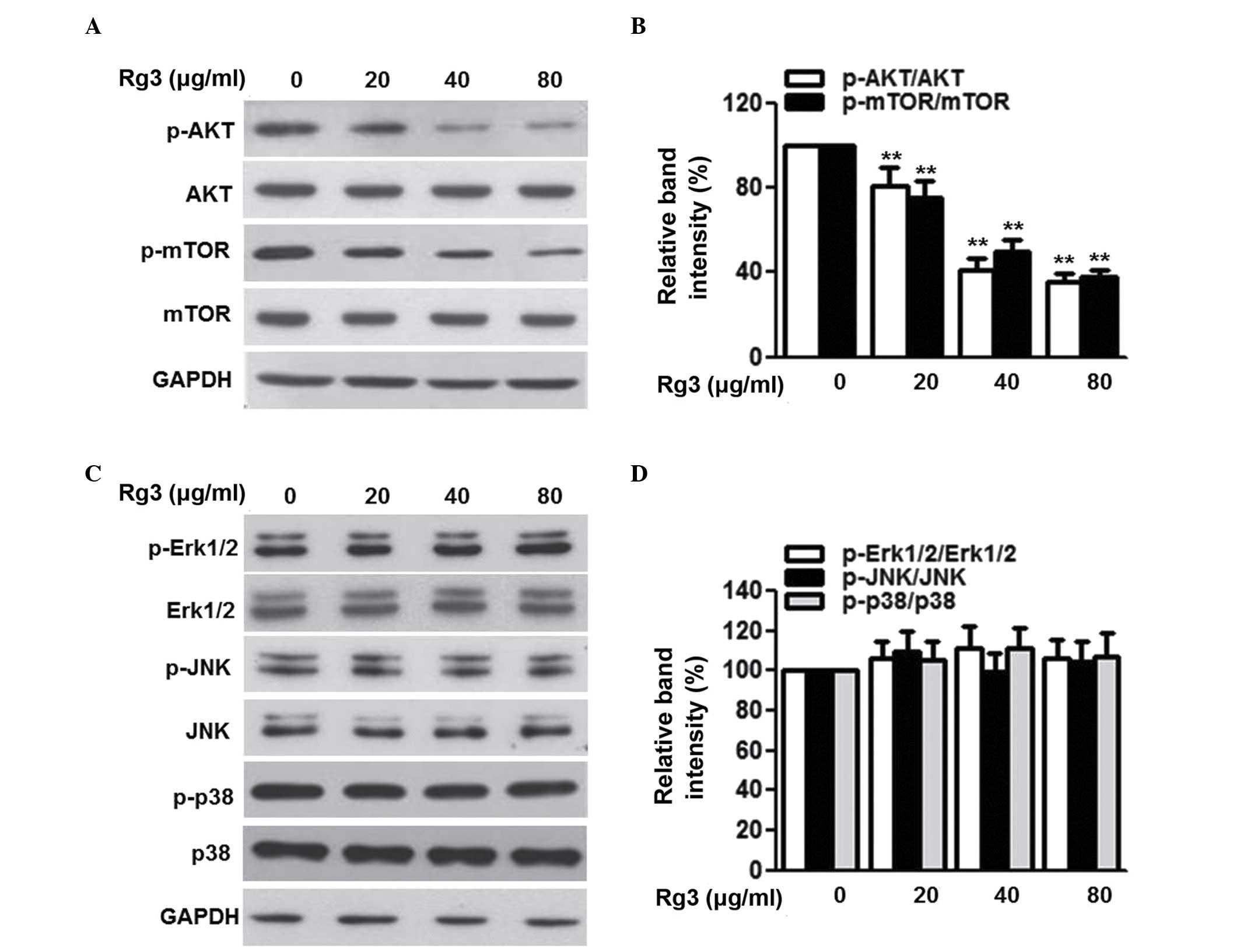
of rapamycin (PI3K/Akt/mTOR)-dependent translation regulatory
signaling pathways in Ames dwarf mice. J Gerontol A Biol Sci Med
Sci. 60:293–300. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Morgensztern D and McLeod HL:
PI3K/Akt/mTOR pathway as a target for cancer therapy. Anticancer
Drugs. 16:797–803. 2005. View Article : Google Scholar : PubMed/NCBI
|