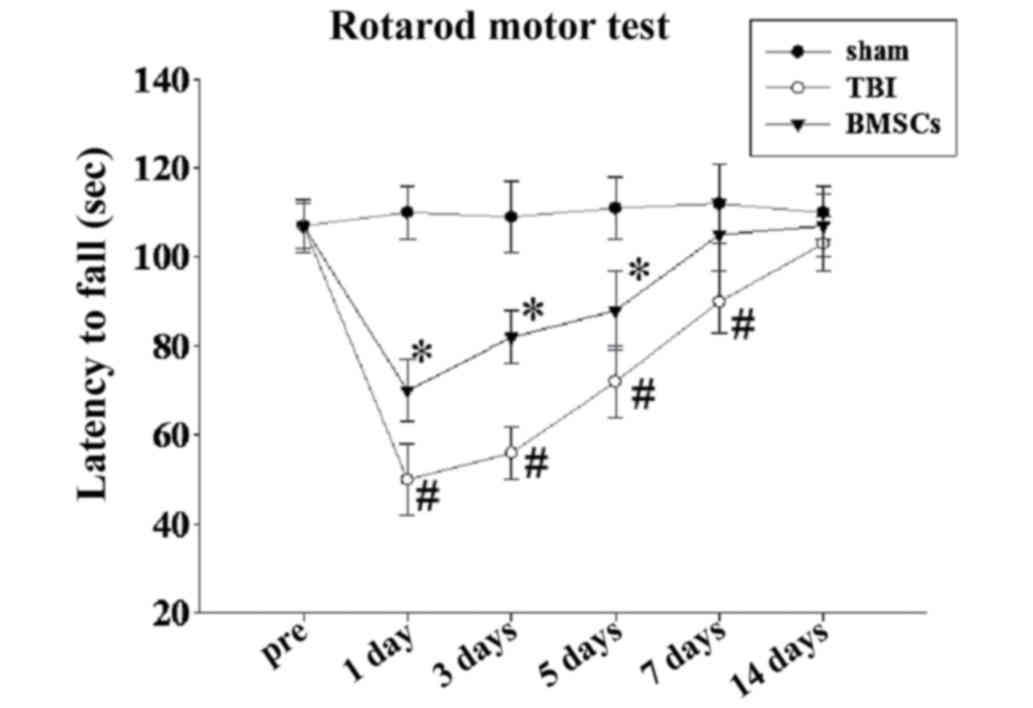
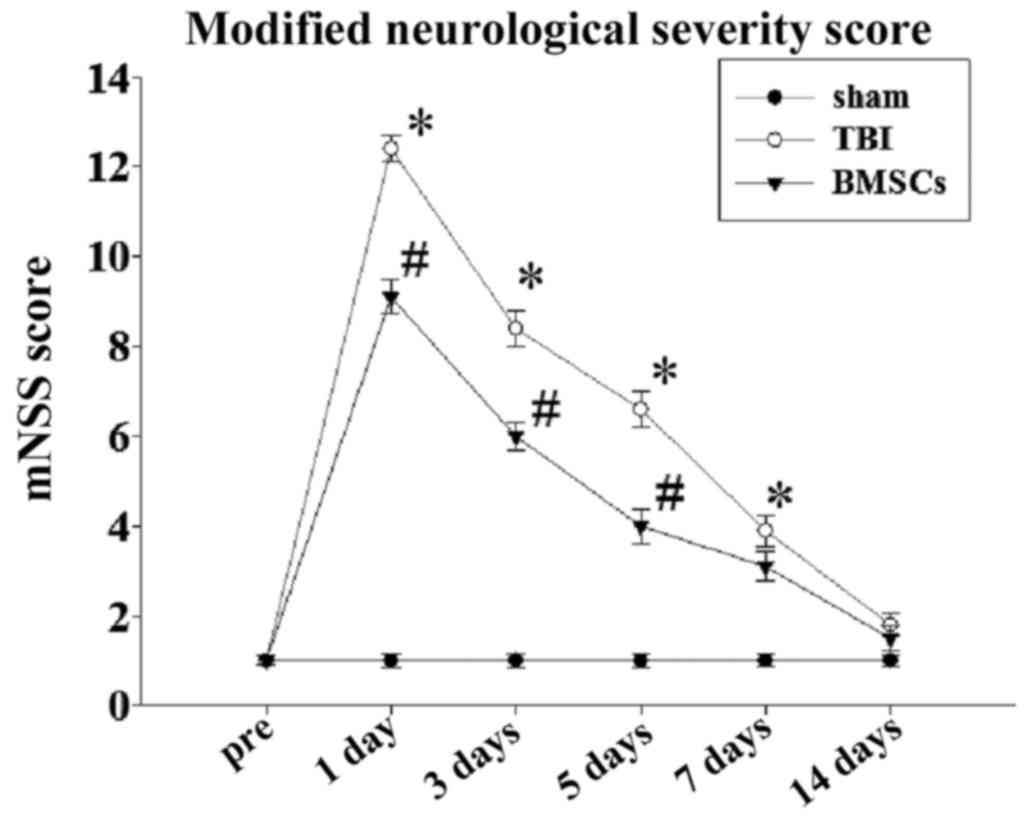
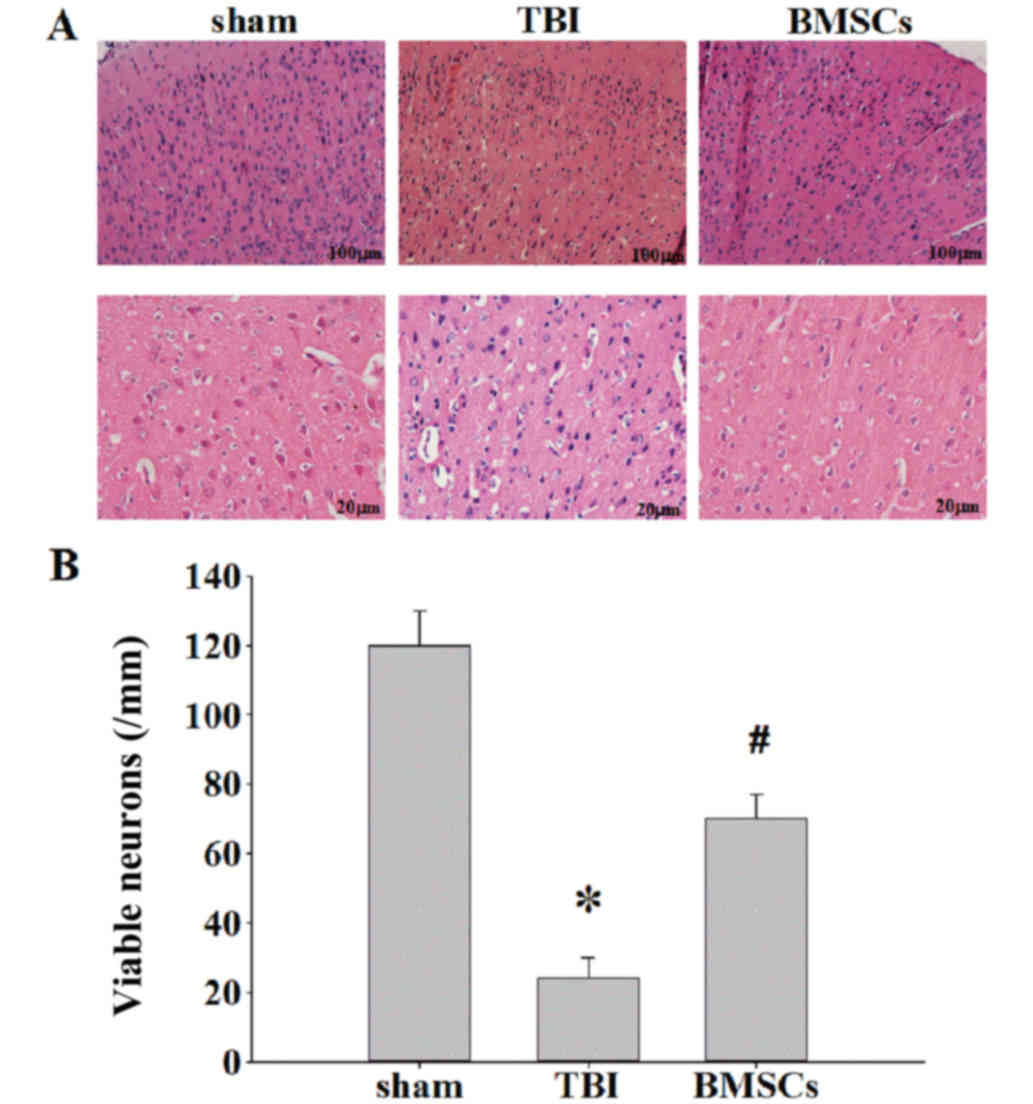
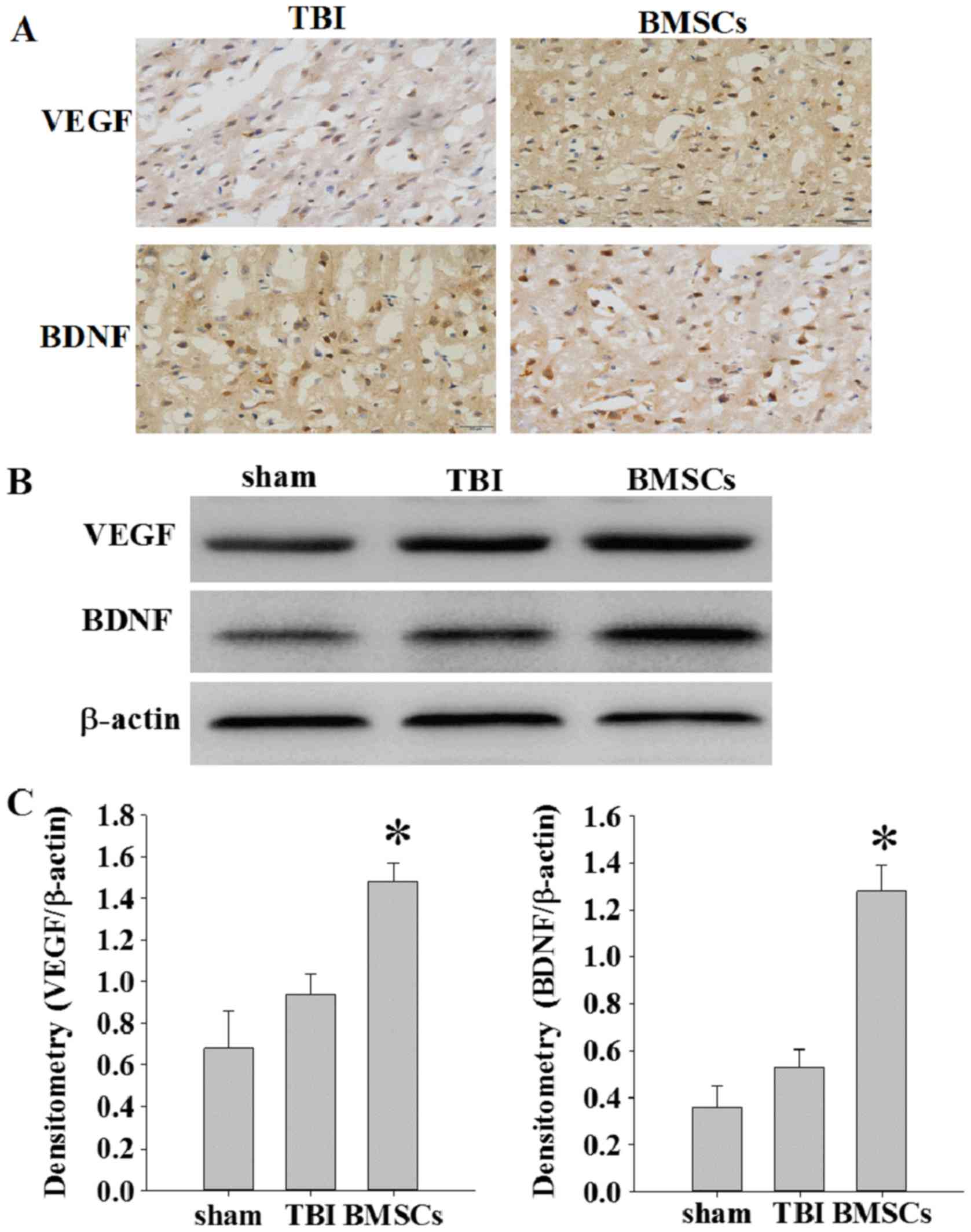
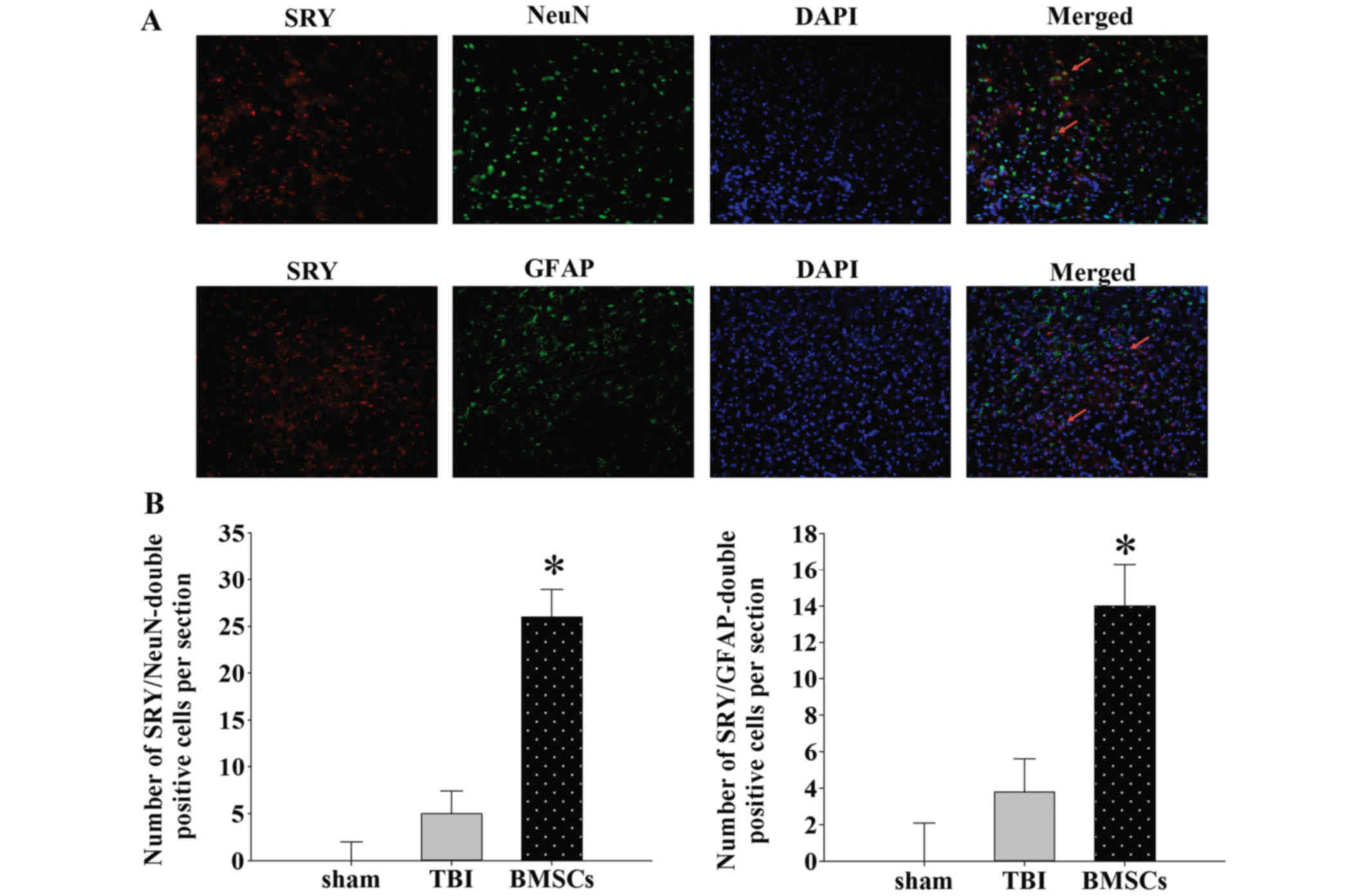
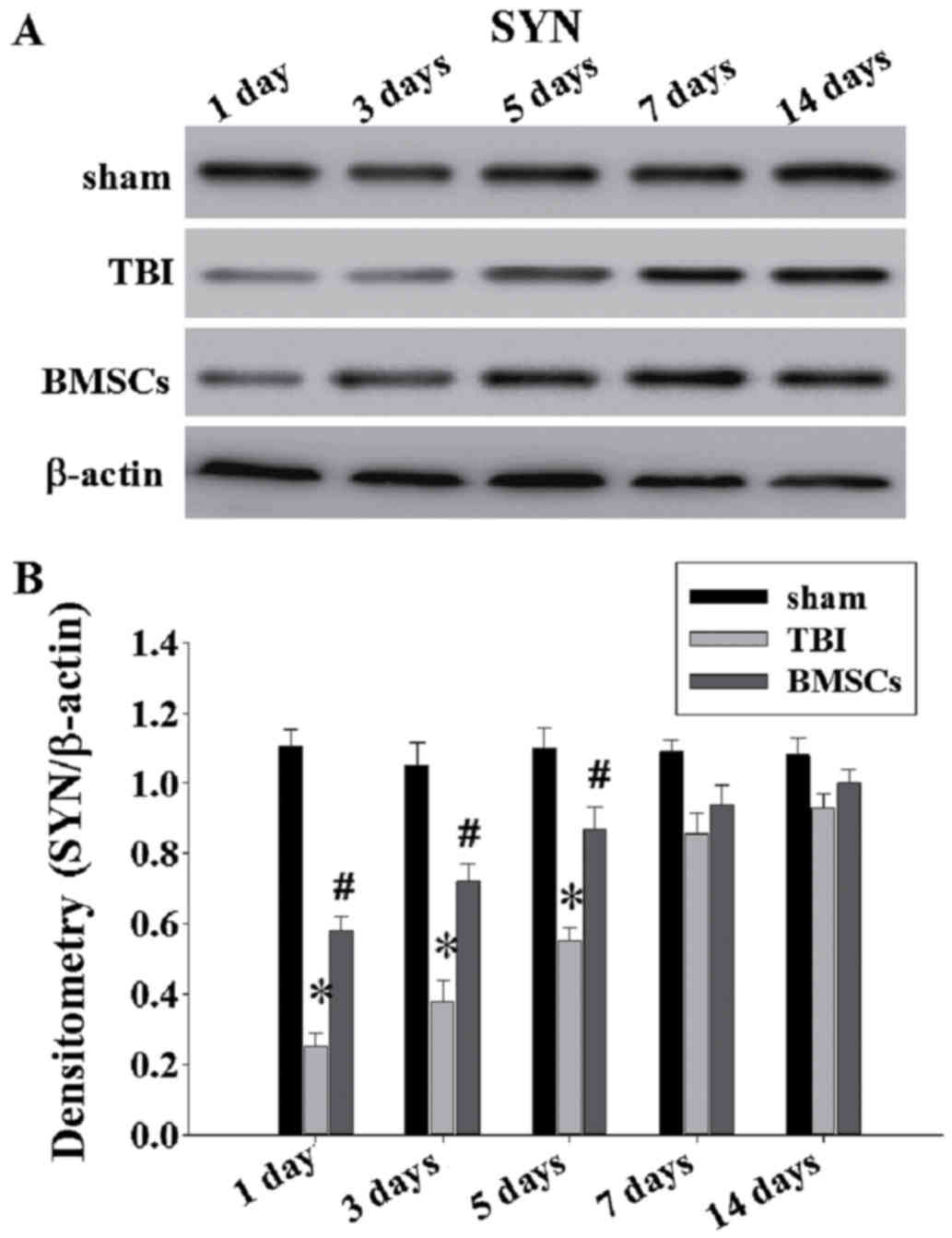
|
1
|
Logan TT, Villapol S and Symes AJ: TGF-β
superfamily gene expression and induction of the Runx1
transcription factor in adult neurogenic regions after brain
injury. PLoS One. 8:e592502013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dharmasaroja P: Bone marrow-derived
mesenchymal stem cells for the treatment of ischemic stroke. J Clin
Neurosci. 16:12–20. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nishida H, Nakayama M, Tanaka H, Kitamura
M, Hatoya S, Sugiura K, Harada Y, Suzuki Y, Ide C and Inaba T:
Safety of autologous bone marrow stromal cell transplantation in
dogs with acute spinal cord injury. Vet Surg. 41:437–442. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Islam MN, Das SR, Emin MT, Wei M, Sun L,
Westphalen K, Rowlands DJ, Quadri SK, Bhattacharya S and
Bhattacharya J: Mitochondrial transfer from bone-marrow-derived
stromal cells to pulmonary alveoli protects against acute lung
injury. Nat Med. 18:759–765. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhao L, Feng Z, Hu B, Chi X and Jiao S: Ex
vivo-expanded bone marrow mesenchymal stem cells facilitate
recovery from chemically induced acute liver damage.
Hepatogastroenterology. 59:2389–2394. 2012.PubMed/NCBI
|
|
6
|
Li Y, Chen J, Chen XG, Wang L, Gautam SC,
Xu YX, Katakowski M, Zhang LJ, Lu M, Janakiraman N and Chopp M:
Human marrow stromal cell therapy for stroke in rat: Neurotrophins
and functional recovery. Neurology. 59:514–523. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Osanai T, Kuroda S, Sugiyama T, Kawabori
M, Ito M, Shichinohe H, Kuge Y, Houkin K, Tamaki N and Iwasaki Y:
Therapeutic effects of intra-arterial delivery of bone marrow
stromal cells in traumatic brain injury of rats-in vivo cell
tracking study by near-infrared fluorescence imaging. Neurosurgery.
70:435–444. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bae KS, Park JB, Kim HS, Kim DS, Park DJ
and Kang SJ: Neuron-like differentiation of bone marrow-derived
mesenchymal stem cells. Yonsei Med J. 52:401–412. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tohill M, Mantovani C, Wiberg M and
Terenghi G: Rat bone marrow mesenchymal stem cells express glial
markers and stimulate nerve regeneration. Neurosci Lett.
362:200–203. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gao X, Deng P, Xu ZC and Chen J: Moderate
traumatic brain injury causes acute dendritic and synaptic
degeneration in the hippocampa dentate gyrus. PLoS One.
6:e245662011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ye X, Yan T, Chopp M, Zacharek A, Ning R,
Venkat P, Roberts C and Chen J: Combination BMSC and Niaspan
treatment of stroke enhances white matter remodeling and synaptic
protein expression in diabetic rats. Int J Mol Sci. 14:22221–22232.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Aizawa-Kohama M, Endo T, Kitada M, Wakao
S, Sumiyoshi A, Matsuse D, Kuroda Y, Morita T, Riera JJ, Kawashima
R, et al: Transplantation of bone marrow stromal cell-derived
neural precursor cells ameliorates deficits in a rat model of
complete spinal cord transaction. Cell Transplant. 22:1613–1625.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cui X, Chopp M, Zacharek A, Roberts C, Lu
M, Savant-Bhonsale S and Chen J: Chemokine, vascular and
therapeutic effects of combination Simvastatin and BMSCs treatment
of stroke. Neurobiol Dis. 36:35–41. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Marmarou A, Foda MA, van den Brink W,
Campbell J, Kita H and Demetriadou K: A new model of diffuse brain
injury in rats. Part I: Pathophysiology and biomechanics. J
Neurosurg. 80:291–300. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Vonder Haar C, Emery MA and Hoane MR:
Chronic folic acid administration confers no treatment effects in
either a high or low dose following unilateral controlled cortical
impact injury in the rat. Restor Neurol Neurosci. 30:291–302.
2012.PubMed/NCBI
|
|
16
|
Gutiérrez-Fernández M, Rodríguez-Frutos B,
Ramos-Cejudo J, Teresa Vallejo-Cremades M, Fuentes B, Cerdán S and
Díez-Tejedor E: Effects of intravenous administration of allogenic
bone marrow-and adipose tissue-derived mesenchymal stem cells on
functional recovery and brain repair markers in experimental
ischemic stroke. Stem Cell Res Ther. 4:112013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen J, Li Y, Wang L, Zhang Z, Lu D, Lu M
and Chopp M: Therapeutic benefit of intravenous administration of
bone marrow stromal cells after cerebral ischemia in rats. Stroke.
32:1005–1011. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pavlichenko N, Sokolova I, Vijde S,
Shvedova E, Alexandrov G, Krouglyakov P, Fedotova O, Gilerovich EG,
Polyntsev DG and Otellin VA: Mesenchymal stem cells transplantation
could be beneficial for treatment of experimental ischemic stroke
in rats. Brain Res. 1233:203–213. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bhakta S, Hong P and Koc O: The surface
adhesion molecule CXCR4 stimulates mesenchymal stem cell migration
to stromal cell-derived factor-1 in vitro but does not decrease
apoptosis under serum deprivation. Cardiovasc Revasc Med. 7:19–24.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Guan J, Bennet L, Gluckman PD and Gunn AJ:
Insulin-like growth factor-1 and post-ischemic brain injury. Progr
Neurobiol. 70:443–462. 2003. View Article : Google Scholar
|
|
21
|
Zhang HY, Jin XB and Lue TF: Three
important components in the regeneration of the cavernous nerve:
Brain-derived neurotrophic factor, vascular endothelial growth
factor and the JAK/STAT signaling pathway. Asian J Androl.
13:231–235. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim JH, Choi KH, Jang YJ, Kim HN, Bae SS,
Choi BT and Shin HK: Electroacupuncture preconditioning reduces
cerebral ischemic injury via BDNF and SDF-1α in mice. BMC
Complement Altern Med. 13:222013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Han X, Yang N, Cui Y, Xu Y, Dang G and
Song C: Simvastatin mobilizes bone marrow stromal cells migrating
to injured areas and promotes functional recovery after spinal cord
injury in the rat. Neurosci Lett. 521:136–141. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Song M, Mohamad O, Gu X, Wei L and Yu SP:
Restoration of intracortical and thalamocortical circuits after
transplantation of bone marrow mesenchymal stem cells into the
ischemic brain of mice. Cell Transplant. 22:2001–2015. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Brock TO and O'Callaghan JP: Quantitative
changes in the synaptic vesicle proteins synapsin I and p38 and the
astrocyte-specific protein glial fibrillary acidic protein are
associated with chemical-induced injury to the rat central nervous
system. J Neurosci. 7:931–942. 1987.PubMed/NCBI
|
|
26
|
Pati S, Muthuraju S, Hadi RA, Huat TJ,
Singh S, Maletic-Savatic M, Abdullah JM and Jaafar H: Neurogenic
plasticity of mesenchymal stem cell, an alluring cellular
replacement for traumatic brain injury. Curr Stem Cell Res Ther.
11:149–157. 2016. View Article : Google Scholar : PubMed/NCBI
|