Introduction
Ovarian cancer (OC) is the most lethal malignant
tumor type in gynecology and a common cause of cancer-associated
mortality in females worldwide. According to the statistics, more
than two-thirds of OC cases are diagnosed at an advanced stage,
with a five-year survival rate of ~30% (1,2).
Despite substantial advances in OC research, the
mortality-to-incidence ratio and overall survival rate remains low
(3). To gain further insights into
the pathogenic mechanisms and to improve the treatment of OC, it is
necessary to further explore the molecular biology that
characterizes OC cells.
High-mobility group protein B1 (HMGB1) is a highly
conserved, non-histone chromatin-binding extracellular nuclear
protein. HMGB1 binds chromatin, stabilizes nucleosomes, regulates
nuclear transcription and acts as an extracellular signaling
molecule (4,5). It is thought that HMGB1 is actively
secreted by tumor cells, and passively released from necrotic cells
to the tumor microenvironment (6).
Recent studies have demonstrated that HMGB1 is closely associated
with tumorigenesis, angiogenesis and metastasis in a number of
malignancies (3,7,8).
Increased expression of HMGB1 has been reported in various tumor
types, including ovarian, breast, prostate, colorectal and gastric
cancer, and is associated with poor survival (9). In addition, high HMGB1 expression is
associated with poor differentiation, a high stage and a positive
lymph node status in OC (10).
Toll-like receptors (TLRs) are essential components of innate
immunity that enhance the function of HMGB1 in cancer, by creating
a procancerous environment through inflammation, angiogenesis and
cell death (11). As an important
damage-associated molecular pattern (DAMP), HMGB1 activation of
TLRs expressed on tumor cells initiates pro-inflammatory signaling
pathways and mediates the release of cytokines and chemokines from
tumor cells. These molecules recruit immune cells that subsequently
release additional cytokines, pro-angiogenic mediators and growth
factors that facilitate tumor growth (12). However, a limited number of studies
have addressed the association between HMGB1/TLR4 signaling and the
clinicopathological characteristics of OC. Serous
cystadenocarcinoma and endometrioid carcinoma are the most common
histological types of epithelial OC. In the present study, 20
patients with epithelial OC were examined and the mRNA and protein
expressions of HMGB1, TLR4, NF-κB and TNF-α in patients with
epithelial OC were detected. The aim of the present study was to
investigate the expression of HMGB1/TLR4 signaling pathway proteins
to further elucidate if they were associated with the
clinicopathological characteristics of malignant epithelial ovarian
cancer (MEOC), and to further elucidate the mechanisms underlying
the development and progression of epithelial OC.
Materials and methods
Ethics statement
The present study was approved by the Institutional
Review Board of Yangpu Hospital, Tongji University School of
Medicine (Shanghai, China). All biopsy specimens were collected,
according to the guidelines of the Declaration of Helsinki. Written
informed consent was obtained from all participants.
Participants
From March 2016 to March 2017, 20 patients who
underwent comprehensive staging operation or tumor reductive
surgery for MEOC at the Gynecological Department of Yangpu
Hospital, Tongji University School of Medicine were included in the
present study. Patients with MEOC were clinically staged, according
to the International Federation of Gynecology and Obstetrics (FIGO)
staging system (13), and were
diagnosed for the first time during the enrollment period. Tissue
samples were collected prior to treatment including surgery,
chemotherapy and radiotherapy. Patients who underwent surgery for a
benign ovarian condition (BOC; all of them had been identified as
serous cystadenoma by histological examination) within the same
time period were recruited as the control group. All patients
enrolled in the present study had not taken any hormonal therapy in
the last three months, including oral contraceptive pills,
progestins, gonadotropin-releasing hormone agonists or
levonorgestrel intrauterine system. Individuals with other
malignant tumors, cardiovascular, autoimmune, endocrine, metabolic,
pelvic inflammatory or infectious disease were excluded.
Tissue collection
Tissue samples were collected from patients who
underwent surgery for ovarian tumors at the Gynecological
Department of Yangpu Hospital (Shanghai, China). MEOC tissue
samples were collected from areas that had been macroscopically
identified as cancer by pathologists, and the final diagnosis
predominantly depended on histological examination of the biopsy.
Under strict asepsis, fresh MEOC and BOC tissue specimens were
frozen in liquid nitrogen and stored at −80°C for RNA and protein
extraction.
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from the frozen tissue
samples using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA). Complementary DNA was
synthesized with RevertAid First Strand cDNA Synthesis kit
(Fermentas; Thermo Fisher Scientific, Inc.) at 42°C for 1 h and
75°C for 5 min. Primer sequences specific for HMGB1, TLR4, nuclear
factor (NF)-κB and tumor necrosis factor (TNF)-α are presented in
Table I. qPCR was performed using
SYBR Green Master mix on an ABI7300 platform (Thermo Fisher
Scientific, Inc.). β-actin was used as an internal control. The PCR
thermocycling conditions were as follows: 94°C for 7 min, followed
by 40 cycles of 15 sec at 94°C and 45 sec at 60°C. ΔCq was defined
as the difference in the cycle threshold between the target gene
and internal control, and ΔΔCq was defined as the difference
between the ΔCq values of the test sample and control. The relative
expression of the target genes was calculated as 2−ΔΔCq
(14).
 | Table I.Primers used in reverse
transcription-quantitative polymerase chain reaction. |
Table I.
Primers used in reverse
transcription-quantitative polymerase chain reaction.
| Gene | Primer sequences
(5′-3′) | Annealing temperature
(°C) | Product size
(bp) |
|---|
| HMGB1 |
| F |
GTGGCTCACGCCTGTAATCC | 61 | 230 |
| R |
GGCACAATCTCGGCTCACTG |
|
|
| TLR4 |
| F |
CCGCTTTCACTTCCTCTCAC | 58 | 182 |
| R |
CATCCTGGCATCATCCTCAC |
|
|
| NF-κB |
| F |
GAATGGCTCGTCTGTAGTG | 56 | 232 |
| R |
TGGTATCTGTGCTCCTCTC |
|
|
| TNF-α |
| F |
CCTGGTATGAGCCCATCTATC | 57 | 218 |
| R |
AGGTTGAGGGTGTCTGAAG |
|
|
| β-actin |
| F |
CAAGATCATTGCTCCTCCTG | 56 | 90 |
| R |
ATCCACATCTGCTGGAAGG |
|
|
Western blot analysis
Protein was extracted with efficient
radioimmunoprecipitation assay histological/cell lysis fluid
(Beijing Solarbio Science & Technology Co., Ltd., Beijing,
China), and centrifuged at 12,000 × g for 15 min at 4°C. Protein
concentration was determined with a bicinchoninic acid protein
assay (Thermo Fisher Scientific, Inc., Waltham, MA, USA) and stored
at −80°C. Total lysates, 15 µl per lane were resolved by 10 or 12%
SDS-PAGE, according to the protein molecular weight. Proteins were
blotted onto a nitrocellulose membrane and blocked at room
temperature for 1 h with 5% skim milk powder, and incubated at 4°C
overnight with the following primary antibodies: HMGB1 (1:300; cat.
no. ab77302), TLR4 (1:500; cat. no. ab22048; both Abcam, Cambridge,
MA, USA), NF-κB p65 (1:1,000, cat. no. 6956s; Cell Signaling
Technology, Inc., Danvers, MA, USA), TNF-α (1:2,000; cat. no.
ab9739; Abcam), GAPDH (1:2,000, cat. no. 5174; Cell Signaling
Technology, Inc.), diluted in Tris-buffered saline (TBS) containing
1% skimmed milk. The blots were subsequently washed with TBS,
incubated with horseradish peroxidase (HRP)-labeled goat
anti-rabbit IgG (1:1,000; cat. no. a0208) HRP-labeled donkey
anti-goat IgG (1:1,000; cat. no. a0181) and HRP-labeled goat
anti-mouse IgG (1:1,000; cat. no. a0216) secondary antibodies
(Beyotime Institute of Biotechnology, Haimen, China) at room
temperature for 1 h. Proteins were visualized using an enhanced
chemiluminescence western blotting system (Bio-Rad Laboratories,
Inc., Hercules, CA, USA). Densitometry was performed using ImageJ
bundled with 64-bit Java 1.8.0_112 (National Institutes of Health,
Bethesda, MD, USA) to quantify protein expression. GAPDH served as
an internal control.
Statistical analysis
All statistical analyses were performed using SPSS
software 16.0 (SPSS, Inc., Chicago, IL, USA). P<0.05 was
considered to indicate a statistically significant difference.
Diagrams were drawn with GraphPad Prism 5 (GraphPad Software, Inc.,
La Jolla, CA, USA). Data normality was determined with the kurtosis
and skewness measures, as well as the Shapiro-Wilk test. Normally
distributed data were presented as the mean ± standard deviation,
and intra-group differences were investigated using the Student's
t-test. Non-normally distributed data were presented as the median
(quartiles), and intra-group differences were determined using the
Mann-Whitney U test. Categorical variables were expressed as the
number of cases and percentages (%). Differences between
categorical data were evaluated using the χ2 test or
Fisher's exact test. A Pearson's correlation test was conducted to
investigate the association between the expression levels of HMGB1,
TLR4, NF-κB and TNF-α.
Results
General patient data
The sociodemographic and clinical characteristics of
the groups are presented in Table
II. There were no statistically significant differences in age,
BMI, fertility or history of prior surgery between the groups
(P>0.05).
 | Table II.General patient data. |
Table II.
General patient data.
| Patient
characteristics | MEOC (n=20) | BOC (n=20) | P-value |
|---|
| Age, y (range) | 57.85±9.60
(38–76) | 53.95±12.62
(22–70) | 0.278a |
| BMI, kg/m2
(range) | 23.07±3.04
(18.82–28.74) | 22.21±2.87
(18.14–27.24) | 0.363a |
| Median parity (lower
quartile, upper quartile) | 1 (1, 1) | 1 (1, 1.75) | 0.768b |
| Median abortion
(lower quartile, upper quartile) | 1.5 (1, 2) | 1 (1, 2) | 0.489b |
| Previous abdominal
surgery (%) |
|
| 1.000c |
| General
surgery | 4 (20) | 4 (20) |
|
|
Gynecological surgery | 7 (35) | 6 (30) |
|
Clinicopathological characteristics of
patients with MEOC
The 20 patients with MEOC were pathologically
diagnosed, and included 11 (55.00%) cases of serous
cystadenocarcinoma and nine (45.00%) cases of endometrioid
carcinoma. According to the FIGO staging system, there were nine
(45.00%) cases of early-stage (stage I or II) MEOC and 11 (55.00%)
cases of advanced-stage (stage III or IV) MEOC. In addition,
according to the degree of differentiation, the tumors were
classified into six (30.00%) cases of poorly differentiated MEOC
and 14 (70.00%) cases of moderately and well-differentiated
MEOC.
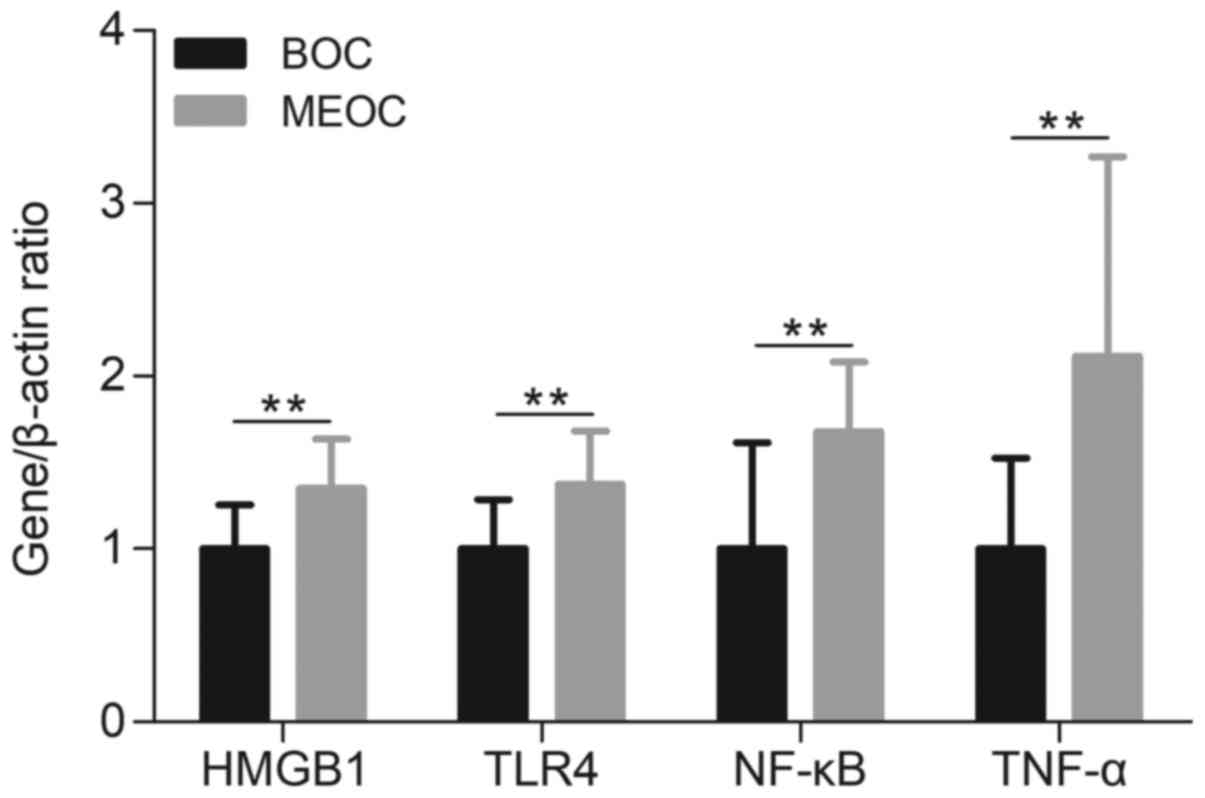
HMGB1, TLR4, NF-κB and TNF-α mRNA and
protein expression is increased in MEOC
RT-qPCR analysis demonstrated that the mRNA
expression of HMGB1, TLR4, NF-κB and TNF-α was significantly
increased in the MEOC group, compared with the BOC group
(P<0.01; Fig. 1). In addition,
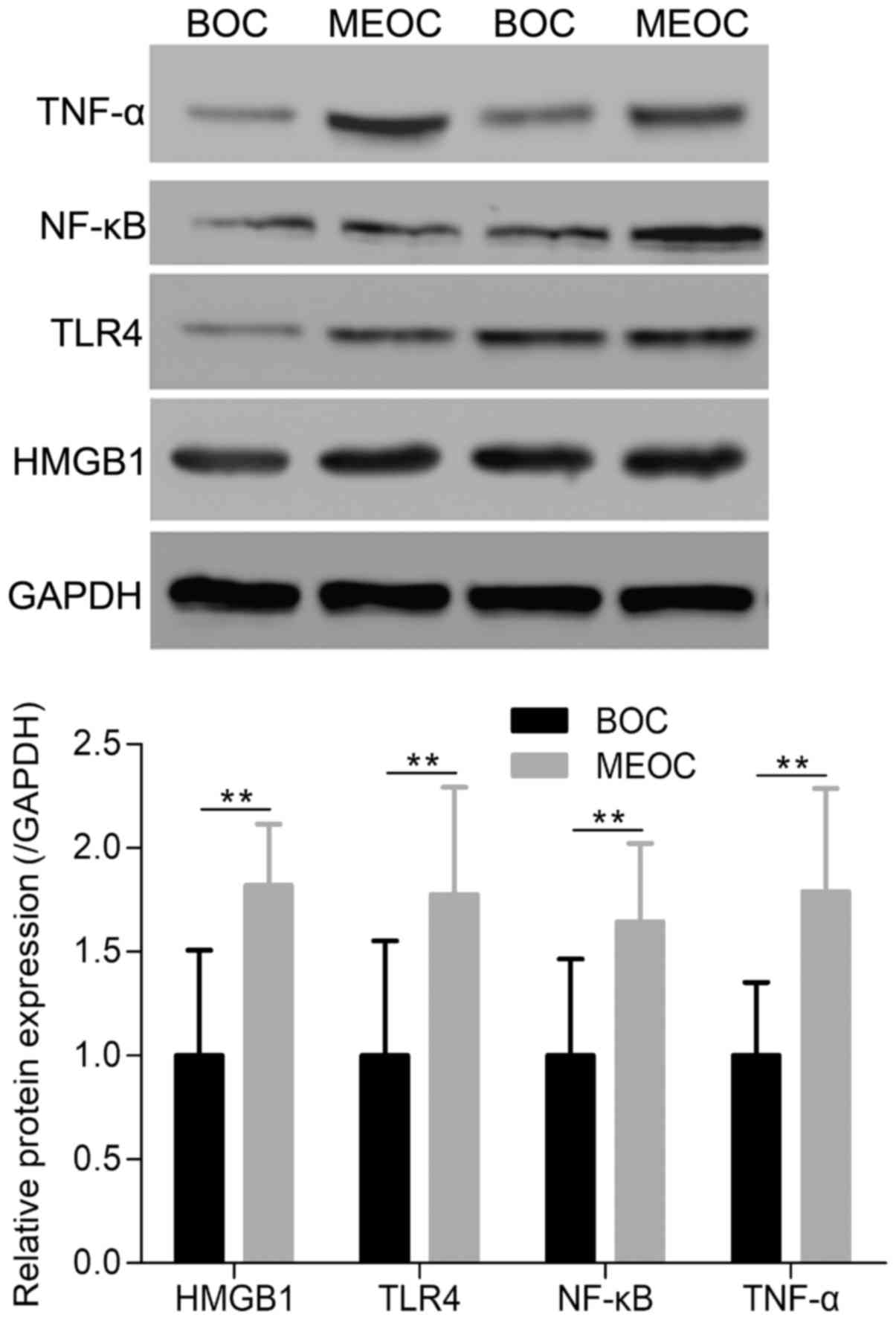
western blot analysis was performed to detect the protein
expression of HMGB1, TLR4, NF-κB and TNF-α in the MEOC and BOC
groups. As presented in Fig. 2,
the protein expression of HMGB1, TLR4, NF-κB and TNF-α was
significantly increased in the MEOC group, compared with the BOC
group (P<0.01).
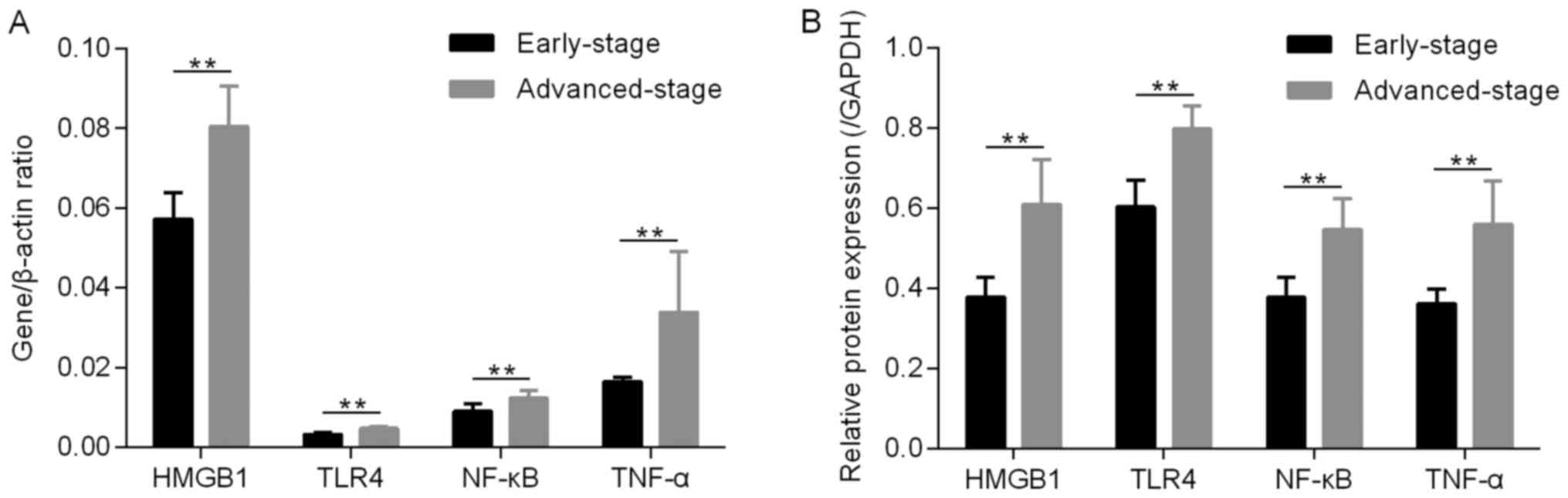
HMGB1, TLR4, NF-κB and TNF-α
expression levels are increased in advanced-stage MEOC
In the present study, patients with MEOC were
divided into two subgroups according to the tumor stage. The gene
expression levels of HMGB1, TLR4, NF-κB and TNF-α in the
advanced-stage group were significantly increased, compared with
the early-stage group (P<0.01; Fig.
3A). Similar results were obtained at the protein level,
demonstrating that the protein expression of HMGB1, TLR4, NF-κB and
TNF-α in the advanced-stage group was significantly increased,
compared with the early-stage group (P<0.01; Fig. 3B).
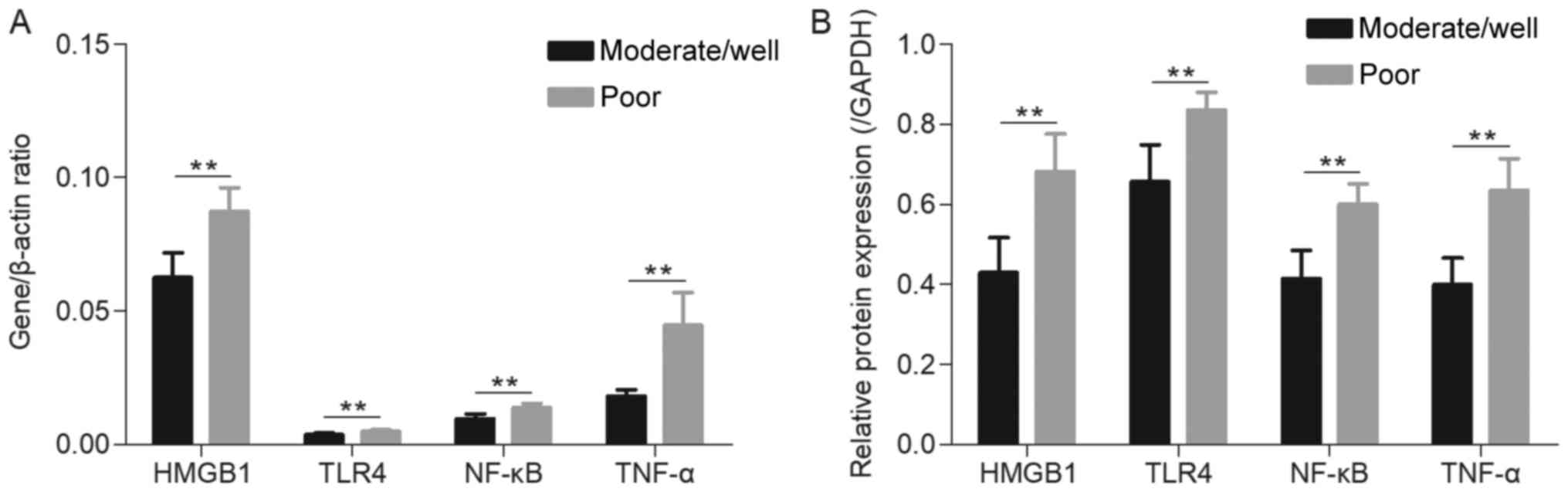
HMGB1, TLR4, NF-κB and TNF-α
expression levels are increased in MEOC with poor
differentiation
In addition, patients with MEOC were divided into
two subgroups according to the degree of differentiation. As
presented in Fig. 4, the gene
(Fig. 4A) and protein (Fig. 4B) expression of HMGB1, TLR4, NF-κB
and TNF-α in the poorly differentiated group was significantly
increased, compared with the moderately and well-differentiated
group (P<0.01).
Correlations between the expression
levels of HMGB1, TLR4, NF-κB and TNF-α in MEOC group
Data analysis revealed that there was a significant
positive correlation between the mRNA and protein expression levels
of HMGB1, TLR4, NF-κB and TNF-α in MEOC (P<0.01). The values of
r are presented in Tables III
and IV.
 | Table III.Correlation between the mRNA
expression levels of HMGB1, TLR4, NF-κB and TNF-α in malignant
epithelial ovarian cancer. |
Table III.
Correlation between the mRNA
expression levels of HMGB1, TLR4, NF-κB and TNF-α in malignant
epithelial ovarian cancer.
|
| HMGB1 | TLR4 | NF-κB | TNF-α |
|---|
| HMGB1 | 1 | 0.983a | 0.973a | 0.989a |
| TLR4 | 0.983a | 1 | 0.953a | 0.967a |
| NF-κB | 0.973a | 0.953a | 1 | 0.944a |
| TNF-α | 0.989a | 0.967a | 0.944a | 1 |
 | Table IV.Correlation between the protein
expression levels of HMGB1, TLR4, NF-κB and TNF-α in malignant
epithelial ovarian cancer. |
Table IV.
Correlation between the protein
expression levels of HMGB1, TLR4, NF-κB and TNF-α in malignant
epithelial ovarian cancer.
|
| HMGB1 | TLR4 | NF-κB | TNF-α |
|---|
| HMGB1 | 1 | 0.968a | 0.970a | 0.971a |
| TLR4 | 0.968a | 1 | 0.935a | 0.971a |
| NF-κB | 0.970a | 0.935a | 1 | 0.950a |
| TNF-α | 0.971a | 0.971a | 0.950a | 1 |
Discussion
Ovarian cancer is the leading cause of
cancer-associated mortality in gynecology worldwide. MEOC is
successfully treated in <40% of women, due to the lack of
effective screening strategies and the non-specific nature of early
signs and symptoms associated with this disease, resulting in an
advanced stage diagnosis for the majority of patients (15,16).
Tumor recurrence and metastasis are considered the major reasons
for poor clinical outcomes and mortality. Therefore, clarification
of the mechanism underlying the invasion and metastasis of MEOC
will provide further insight into the development and progression
of OC (10). Inflammation is an
essential element in tumorigenesis. HMGB1 is a key DAMP. DAMPs are
molecules released from necrotic cells as intrinsic danger signals,
which induce inflammation and trigger innate immunity (17). TLRs trigger an inflammatory
response and cell survival in the tumor microenvironment, and TLR4
is known to be a receptor of HMGB1 (12). A number of previous studies have
confirmed the tumor-facilitating effect of HMGB1. For example,
HMGB1 is increased in ovarian, colorectal, lung and gastric cancer,
indicating that HMGB1 is an important mediator for cancer
transformation, proliferation and invasion (7,8,10).
Furthermore, it has recently been demonstrated that activation of
TLR4/NF-κB signaling by DAMPs may contribute to an inflammatory
microenvironment that drives a more aggressive phenotype with
poorer clinical outcomes in patients with MEOC (11,18).
Wang et al (3) performed a meta-analysis that revealed
that HMGB1 levels in the tissue and serum of patients with OC are
significantly higher compared with those detected in benign tumor
and normal ovarian samples. The effect of HMGB1 on cancer types may
be mediated by multiple surface receptors, including TLRs (3). In the present study, the mRNA
expression levels of HMGB1, TLR4, NF-κB and TNF-α were determined
in 20 patients with MEOC and compared with the expression in 20
patients with BOC. It was demonstrated that the mRNA expression of
HMGB1 in MEOC was significantly increased compared with the BOC
group. The same result was obtained at the protein expression
level, which was consistent with a previous study (3), suggesting that HMGB1 may serve a
pivotal role in the development of OC. Furthermore, the expression
levels of TLR4, a HMGB1 receptor, in addition to the expression of
downstream effectors NF-κB and TNF-α, were significantly increased
in the MEOC group, compared with in the BOC group. These results
indicated that the HMGB1/TLR4 signaling pathway may be implicated
in the development of the tumor-associated inflammatory
microenvironment, which subsequently may serve a pivotal role in
MEOC carcinogenesis and clinical outcomes.
HMGB1 has recently been identified to be
overexpressed in malignant OC, and associated with poor
clinicopathological features and prognosis (8). Chen et al (10) reported that knockdown of HMGB1
suppresses OC cell proliferation and inhibits cell migration and
invasion, which is accompanied by decreased cyclin D1,
proliferating cell nuclear antigen, matrix metalloproteinase (MMP)2
and MMP9 mRNA expression and enzymatic activity. In order to
elucidate whether the HMGB1/TLR4 signaling pathway was associated
with the clinicopathological characteristics of MEOC, the
expression levels of HMGB1, TLR4, NF-κB and TNF-α at different
tumor stages and differentiation grades were evaluated. The results
revealed that the gene and protein expression levels of HMGB1,
TLR4, NF-κB and TNF-α in the advanced-stage and poorly
differentiated groups were significantly higher compared with those
in the early-stage and well/moderately differentiated group,
respectively, indicating that the HMGB1/TLR4 signaling pathway was
associated with the tumor stage and degree of tumor cell
differentiation in MEOC. In addition, the correlation between the
expression of HMGB1, TLR4, NF-κB and TNF-α in MEOC group was
evaluated and it was demonstrated that all the parameters were
positively correlated with each other. It is known that the
HMGB1/TLR4 signaling pathway regulates cell proliferation and
survival, and creates a tumor microenvironment that facilitates
tumor growth by inducing immune cell expansion and integrating
inflammatory responses (19).
Considering these previous findings and those obtained from the
present study, it was therefore concluded that increased HMGB1/TLR4
in the inflammatory immune signaling system may be involved in MEOC
tumor stage and differentiation. Furthermore, this may have been
mediated via the downstream NF-κB signaling pathway.
However, there are certain limitations in the
present study. First, as a result of the ethical and experimental
limitations, only 20 patients with BOC were recruited as the
control group, with no comparison to normal ovarian tissue.
Secondly, patient data during hospitalization only were obtained
for analysis, which did not contain information concerning
postoperative recurrence and disease-free survival. In addition,
cytology associated experiments were not conducted in the present
study. Therefore, further studies with larger sample sizes are
required to avoid these limitations and verify the findings of the
present study.
In conclusion, it was demonstrated that components
involved in the HMGB1/TLR4 signaling pathway and its downstream
effectors were overexpressed and associated with MEOC tumor stage
and differentiation. This may have be mediated via the NF-κB
signaling pathway. These findings further elucidated the mechanisms
underlying the development and progression of MEOC.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Fundamental
Research Funds for the Central Universities (grant no.
1516219014).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HK, RC and WG conceived and designed the
experiments. CJ performed the experiments, created the figures and
wrote the manuscript. XQ and WY contributed to statistical analysis
and revision of the manuscript. HK participated in sample
collection. ZC made substantial contributions to the conception and
design of the study, revised the manuscript critically for
important intellectual content and gave final approval of the
version to be published. All authors reviewed the manuscript and
contributed to patient management. All authors read and approved
the final version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Institutional
Review Board of Yangpu Hospital, Tongji University School of
Medicine. All biopsy specimens were collected, according to the
guidelines of the Declaration of Helsinki. Written informed consent
was obtained from all participants.
Patient consent for publication
Written informed consent was obtained from all
participants.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jayson GC, Kohn EC, Kitchener HC and
Ledermann JA: Ovarian cancer. Lancet. 384:1376–1388. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Luo N, Guo J, Chen L, Yang W, Qu X and
Cheng Z: ARHGAP10, downregulated in ovarian cancer, suppresses
tumorigenicity of ovarian cancer cells. Cell Death Dis.
7:e21572016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang H, Li Z, Sun Y, Xu Z, Han J, Song B,
Song W, Qin C and Yin L: Relationship between high-mobility group
box 1 overexpression in ovarian cancer tissue and serum: A
meta-analysis. Onco Targets Ther. 8:3523–3531. 2015.PubMed/NCBI
|
|
4
|
Li Y, Tian J, Fu X, Chen Y, Zhang W, Yao H
and Hao Q: Serum high mobility group box protein 1 as a clinical
marker for ovarian cancer. Neoplasma. 61:579–584. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhou LY, Shi LY and Xiao Y: Changes of
HMGB1 expression on angiogenesis of ovarian cancer and its
mechanism. J Biol Regul Homeost Agents. 30:233–238. 2016.PubMed/NCBI
|
|
6
|
Zhang W, Tian J and Hao Q: HMGB1 combining
with tumor-associated macrophages enhanced lymphangiogenesis in
human epithelial ovarian cancer. Tumour Biol. 35:2175–2186. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Paek J, Lee M, Nam EJ, Kim SW and Kim YT:
Clinical impact of high mobility group box 1 protein in epithelial
ovarian cancer. Arch Gynecol Obstet. 293:645–650. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li S and Wei Y: Association of HMGB1,
BRCA1 and P62 expression in ovarian cancer and chemotherapy
sensitivity. Oncol Lett. 15:9572–9576. 2018.PubMed/NCBI
|
|
9
|
Ju LL, Zhao CY, Ye KF, Yang H and Zhang J:
Expression and clinical implication of Beclin1, HMGB1, p62,
survivin, BRCA1 and ERCC1 in epithelial ovarian tumor tissues. Eur
Rev Med Pharmacol Sci. 20:1993–2003. 2016.PubMed/NCBI
|
|
10
|
Chen J, Liu X, Zhang J and Zhao Y:
Targeting HMGB1 inhibits ovarian cancer growth and metastasis by
lentivirus-mediated RNA interference. J Cell Physiol. 230:25792015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li Z, Block MS, Vierkant RA, Fogarty ZC,
Winham SJ, Visscher DW, Kalli KR, Wang C and Goode EL: The
inflammatory microenvironment in epithelial ovarian cancer: A role
for TLR4 and MyD88 and related proteins. Tumor Biol.
37:13279–13286. 2016. View Article : Google Scholar
|
|
12
|
Husseinzadeh N and Davenport SM: Role of
toll-like receptors in cervical, endometrial and ovarian cancers: A
review. Gynecol Oncol. 135:359–363. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Prat J: FIGO Committee on Gynecologic
Oncology: Staging classification for cancer of the ovary, fallopian
tube, and peritoneum. Int J Gynaecol Obstet. 124:1–5. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cheng Z, Guo J, Chen L, Luo N, Yang W and
Qu X: Overexpression of TMEM158 contributes to ovarian
carcinogenesis. J Exp Clin Cancer Res. 34:752015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kobayashi M, Sawada K and Kimura T:
Potential of integrin inhibitors for treating ovarian cancer: A
literature review. Cancers (Basel). 9:pii: E83. 2017.
|
|
17
|
Waki K, Kawano K, Tsuda N, Ushijima K,
Itoh K and Yamada A: Plasma levels of high-mobility group box 1
during peptide vaccination in patients with recurrent ovarian
cancer. J Immunol Res. 2017:14236832017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sun NK, Huang SL, Chang TC and Chao CC:
TLR4 and NFκB signaling is critical for taxol resistance in ovarian
carcinoma cells. J Cell Physiol. 233:2489–2501. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jiang C, Liu C, Guo J, Chen L, Luo N, Qu
X, Yang W, Ren Q and Cheng Z: The expression of Toll-like receptors
in eutopic and ectopic endometrium and its implication in the
inflammatory pathogenesis of adenomyosis. Sci Rep. 7:73652017.
View Article : Google Scholar : PubMed/NCBI
|