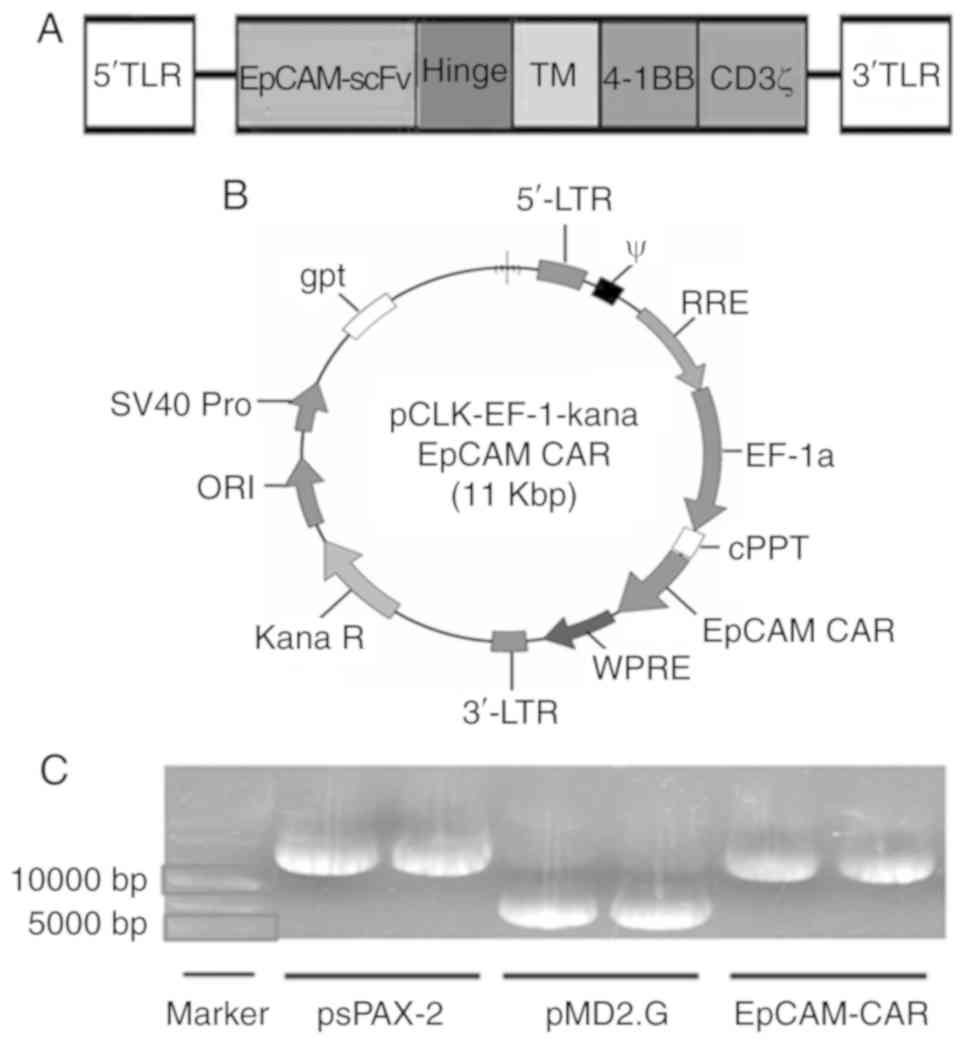
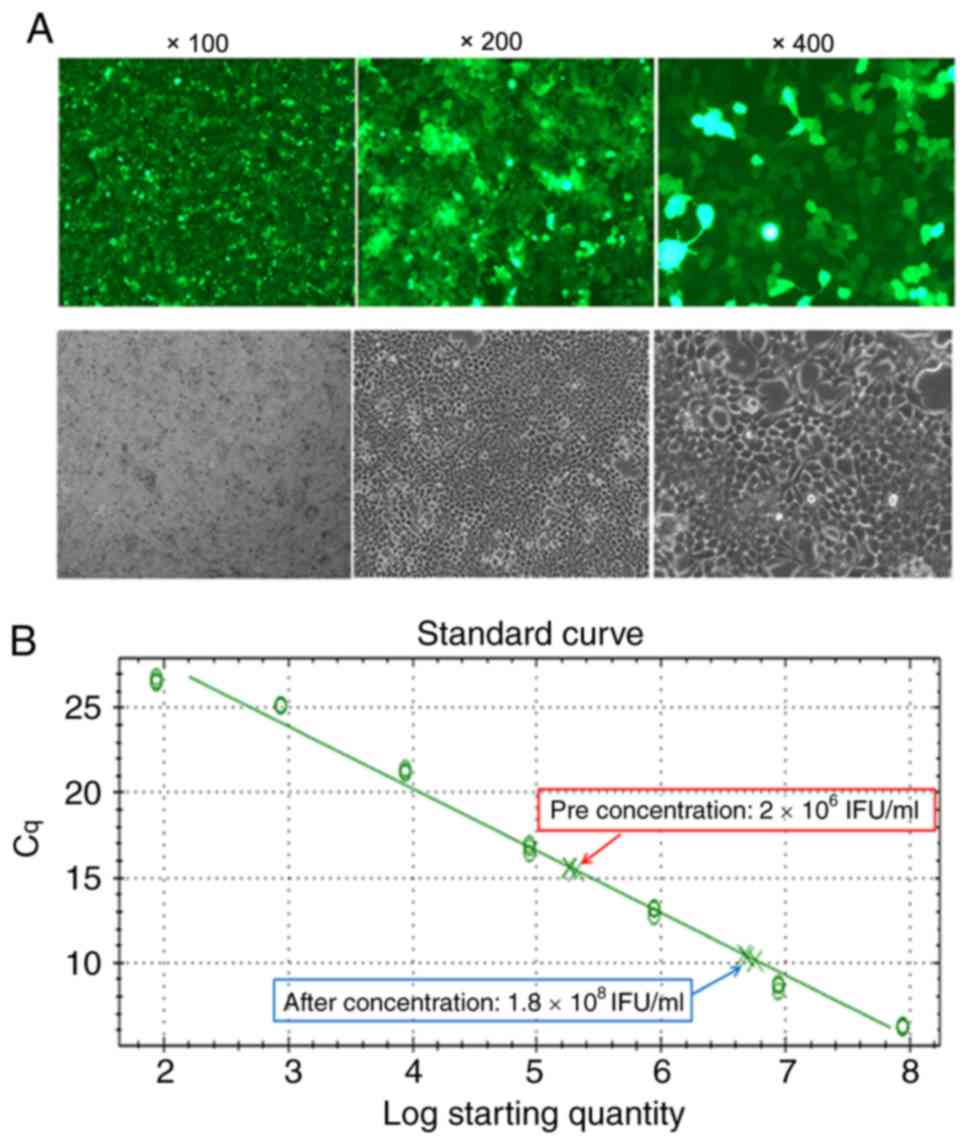
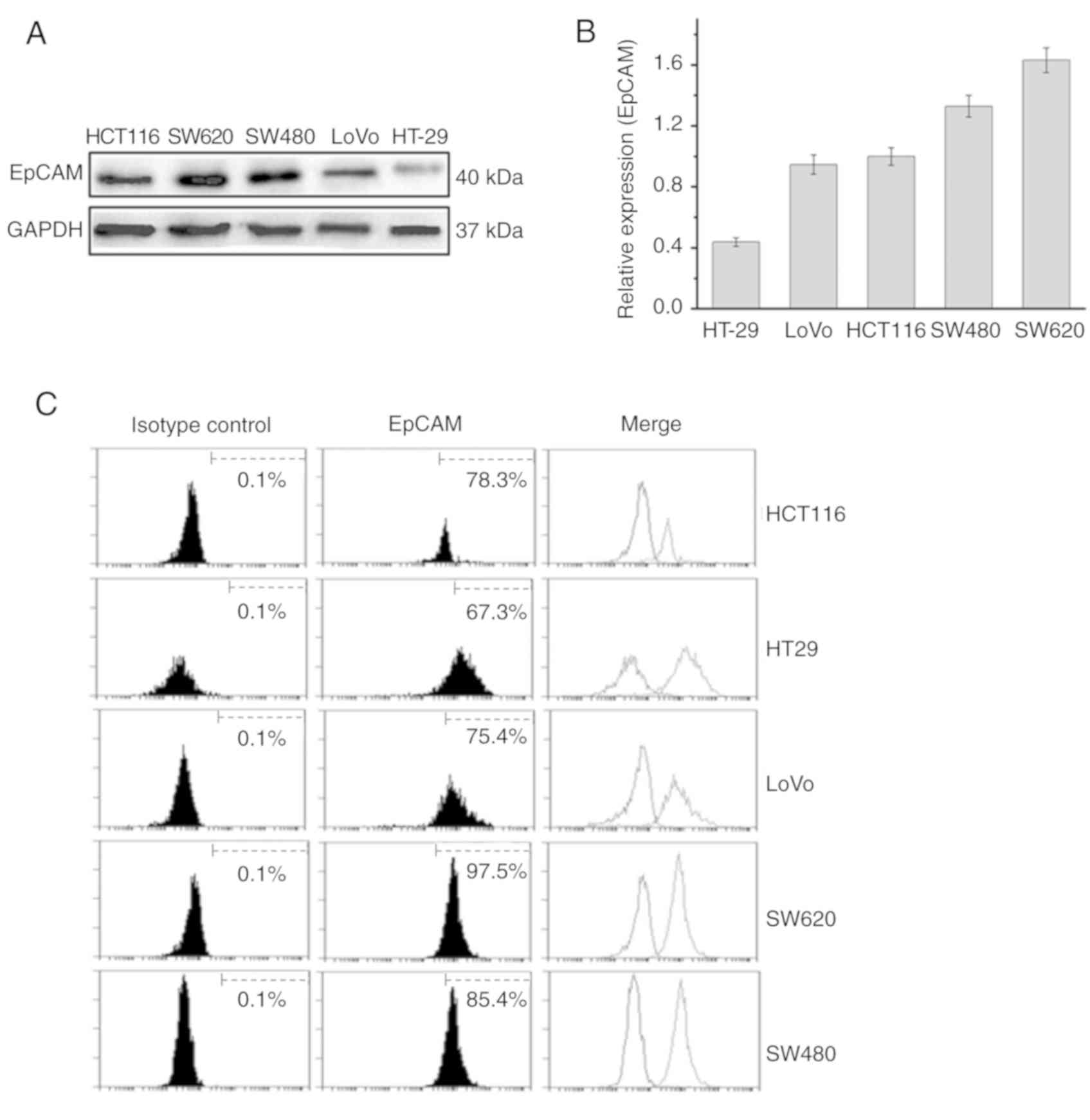
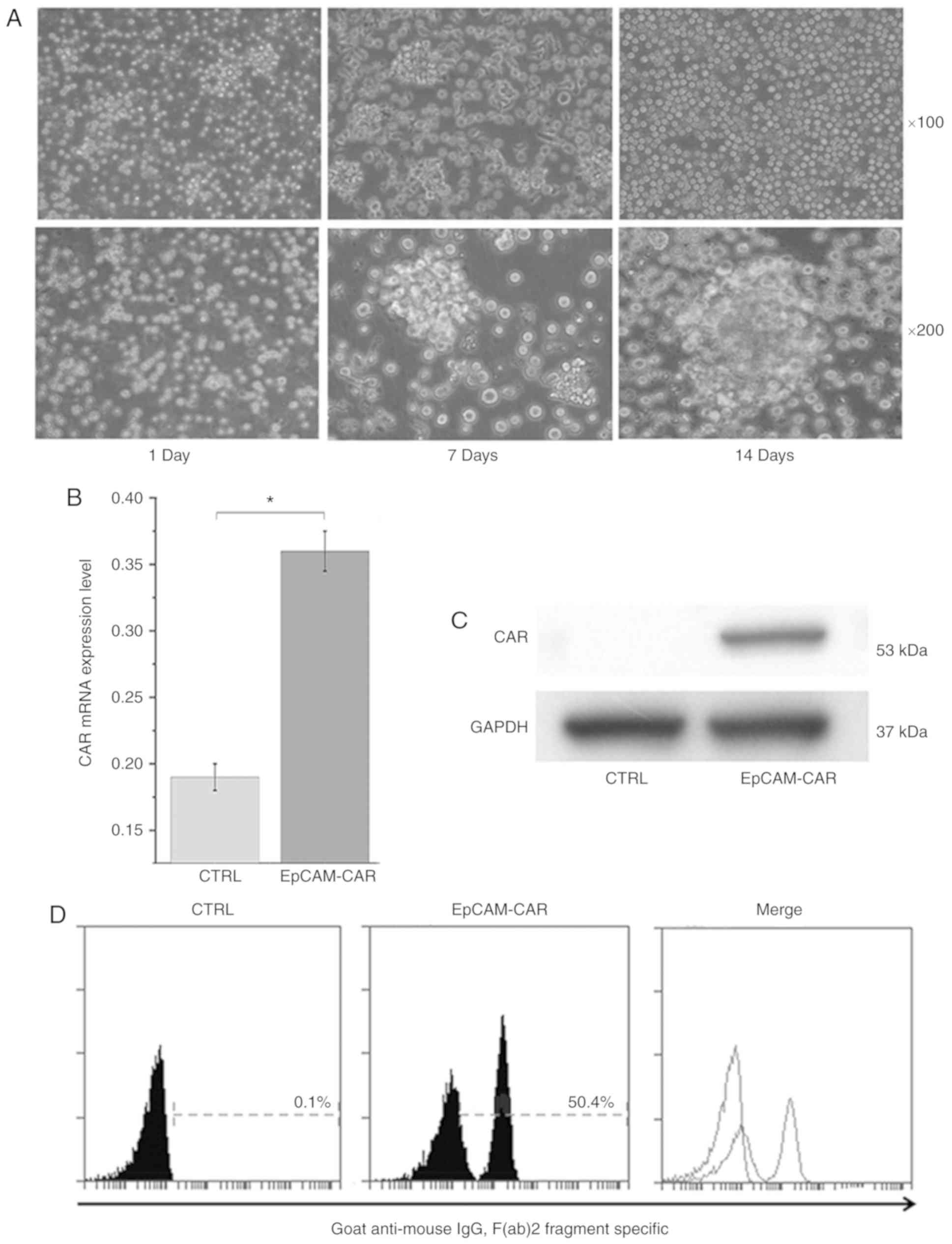
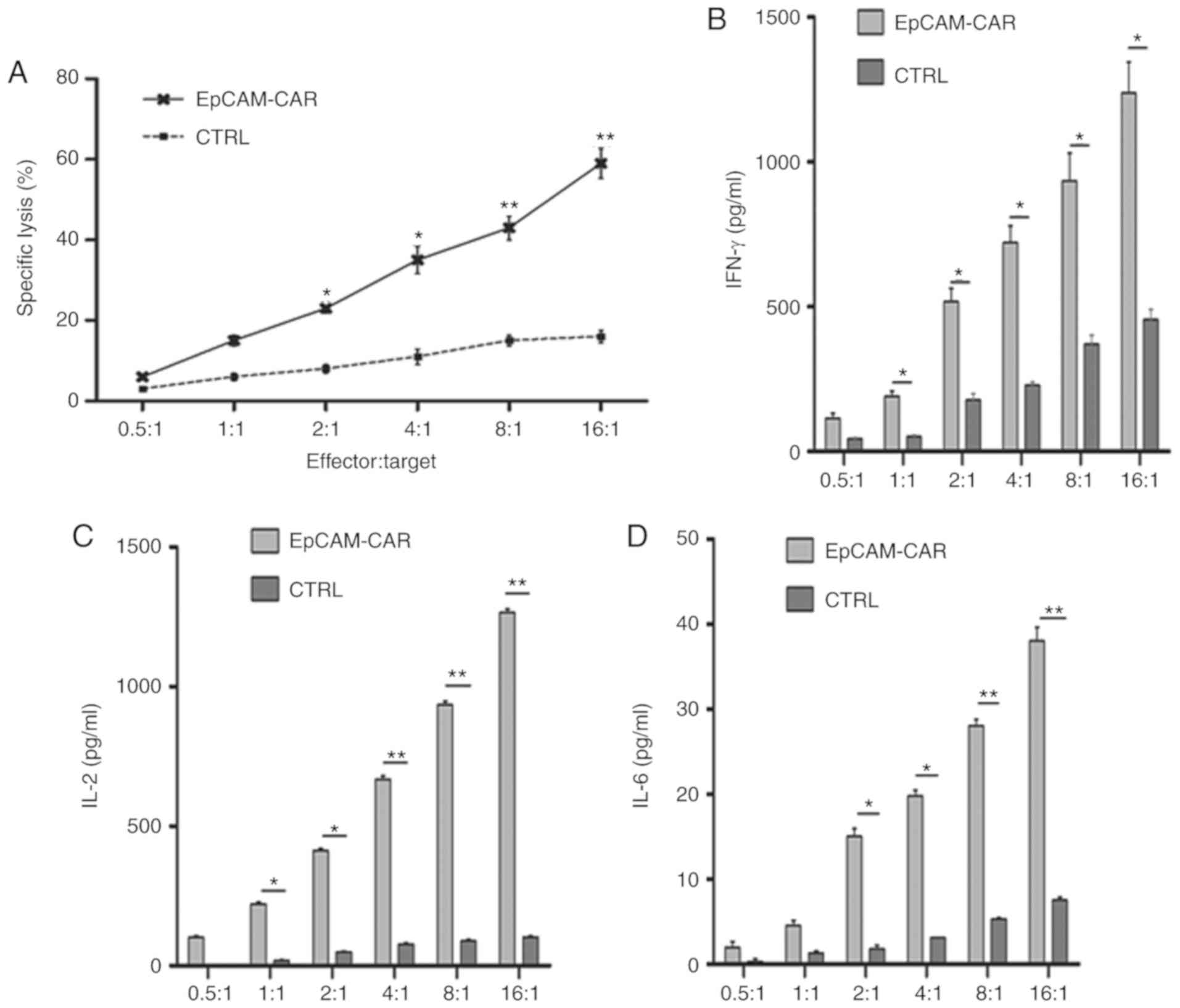
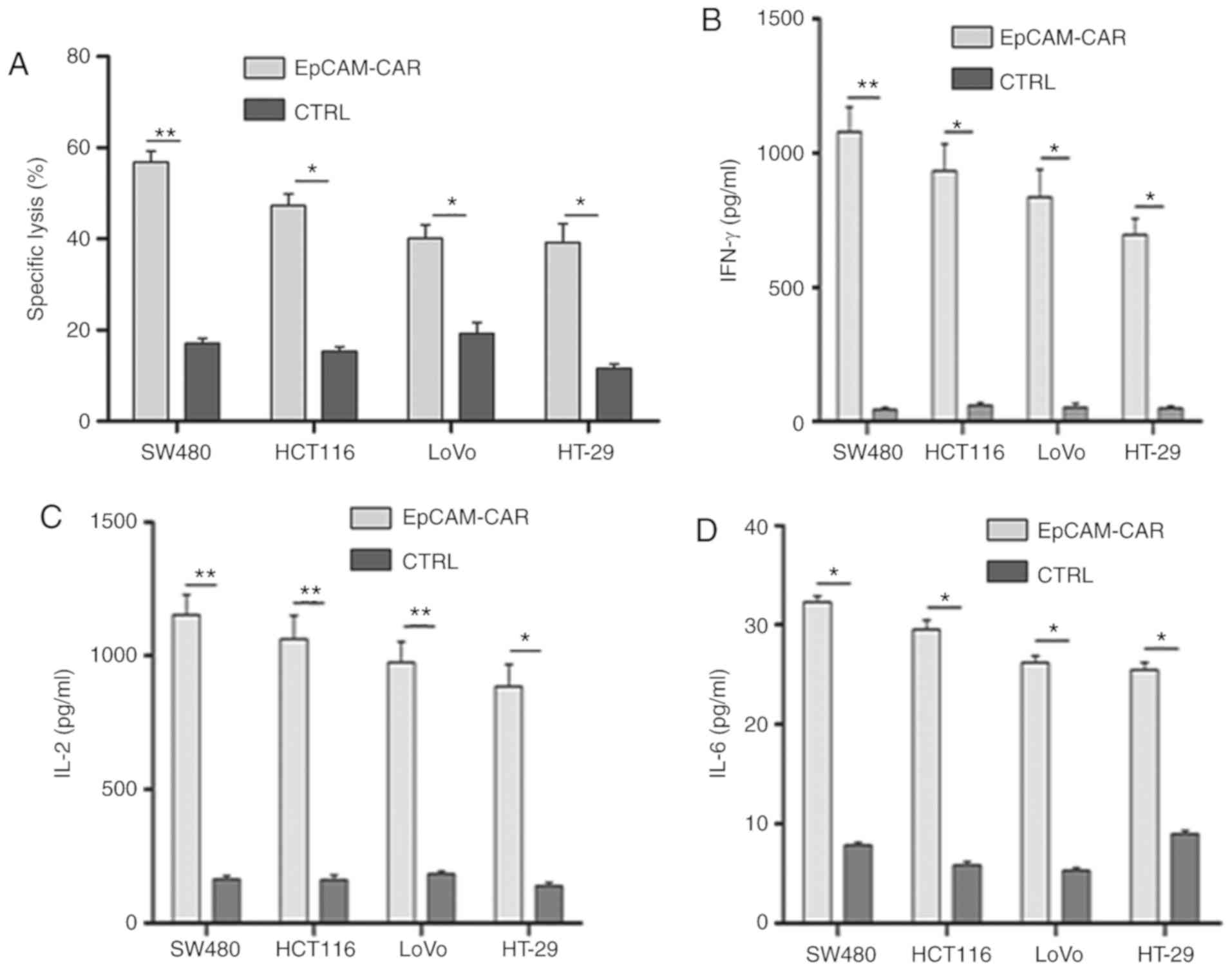
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics, 2009. CA Cancer J Clin. 59:225–249.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ,
Meester RGS, Barzi A and Jemal A: Colorectal cancer statistics,
2017. CA Cancer J Clin. 67:177–193. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pang Y, Hou X, Yang C, Liu Y and Jiang G:
Advances on chimeric antigen receptor-modified T-cell therapy for
oncotherapy. Mol Cancer. 17:912018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Couzin-Frankel J: Breakthrough of the year
2013. Cancer immunotherapy. Science. 342:1432–1433. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mellman I, Coukos G and Dranoff G: Cancer
immunotherapy comes of age. Nature. 480:480–490. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rosenberg SA and Restifo NP: Adoptive cell
transfer as personalized immunotherapy for human cancer. Science.
348:62–68. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tran E, Robbins PF and Rosenberg SA:
‘Final common pathway’ of human cancer immunotherapy: Targeting
random somatic mutations. Nat Immunol. 18:255–262. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Crompton JG, Klemen N and Kammula US:
Metastasectomy for tumor-infiltrating lymphocytes: An emerging
operative indication in surgical oncology. Ann Surg Oncol.
25:565–572. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kunert A, Obenaus M, Lamers CHJ,
Blankenstein T and Debets R: T-cell receptors for clinical therapy:
In vitro assessment of toxicity risk. Clin Cancer Res.
23:6012–6020. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hay KA and Turtle CJ: Chimeric antigen
receptor (CAR) T cells: Lessons learned from targeting of CD19 in
B-cell malignancies. Drugs. 77:237–245. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dominguez G: The CART gene: Structure and
regulation. Peptides. 27:1913–1918. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang C, Liu J, Zhong JF and Zhang X:
Engineering CAR-T cells. Biomark Res. 5:222017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Martowicz A, Seeber A and Untergasser G:
The role of EpCAM in physiology and pathology of the epithelium.
Histol Histopathol. 31:349–355. 2016.PubMed/NCBI
|
|
15
|
Jin Z, Maiti S, Huls H, Singh H, Olivares
S, Mátés L, Izsvák Z, Ivics Z, Lee DA, Champlin RE and Cooper LJ:
The hyperactive sleeping beauty transposase SB100X improves the
genetic modification of T cells to express a chimeric antigen
receptor. Gene Therapy. 18:849–856. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hao H, Zhen Y, Wang Z, Chen F and Xie X: A
novel therapeutic drug for colon cancer: EpCAM scFv-truncated
protamine (tp)-siRNA. Cell Biol Int. 37:860–864. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mala J, Puthong S, Maekawa H, Kaneko Y,
Palaga T, Komolpis K and Sooksai S: Construction and sequencing
analysis of scFv antibody fragment derived from monoclonal antibody
against norfloxacin (Nor155). J Genet Eng Biotechnol. 15:69–76.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Carpenito C, Milone MC, Hassan R, Simonet
JC, Lakhal M, Suhoski MM, Varela-Rohena A, Haines KM, Heitjan DF,
Albelda SM, et al: Control of large, established tumor xenografts
with genetically retargeted human T cells containing CD28 and CD137
domains. Proc Natl Acad Sci USA. 106:3360–3365. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Savoldo B, Ramos CA, Liu E, Mims MP,
Keating MJ, Carrum G, Kamble RT, Bollard CM, Gee AP, Mei Z, et al:
CD28 costimulation improves expansion and persistence of chimeric
antigen receptor-modified T cells in lymphoma patients. J Clin
Invest. 121:1822–1826. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ang WX, Li Z, Chi Z, Du SH, Chen C, Tay
JC, Toh HC, Connolly JE, Xu XH and Wang S: Intraperitoneal
immunotherapy with T cells stably and transiently expressing
anti-EpCAM CAR in xenograft models of peritoneal carcinomatosis.
Oncotarget. 8:13545–13559. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fisher DT, Appenheimer MM and Evans SS:
The two faces of IL-6 in the tumor microenvironment. Semin Immunol.
26:38–47. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nakajima C, Uekusa Y, Iwasaki M, Yamaguchi
N, Mukai T, Gao P, Tomura M, Ono S, Tsujimura T, Fujiwara H and
Hamaoka T: A role of interferon-gamma (IFN-gamma) in tumor
immunity: T cells with the capacity to reject tumor cells are
generated but fail to migrate to tumor sites in IFN-gamma-deficient
mice. Cancer Res. 61:3399–3405. 2001.PubMed/NCBI
|
|
24
|
Rosenberg SA: IL-2: The first effective
immunotherapy for human cancer. J Immunol. 192:5451–5458. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lichty BD, Breitbach CJ, Stojdl DF and
Bell JC: Going viral with cancer immunotherapy. Nat Rev Cancer.
14:559–567. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rodgers DT, Mazagova M, Hampton EN, Cao Y,
Ramadoss NS, Hardy IR, Schulman A, Du J, Wang F, Singer O, et al:
Switch-mediated activation and retargeting of CAR-T cells for
B-cell malignancies. Proc Natl Acad Sci USA. 113:E459–E468. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Grupp SA, Kalos M, Barrett D, Aplenc R,
Porter DL, Rheingold SR, Teachey DT, Chew A, Hauck B, Wright JF, et
al: Chimeric antigen receptor-modified T cells for acute lymphoid
leukemia. N Engl J Med. 368:1509–1518. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Porter DL, Levine BL, Kalos M, Bagg A and
June CH: Chimeric antigen receptor-modified T cells in chronic
lymphoid leukemia. N Engl J Med. 365:725–733. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Schuster SJ, Svoboda J, Chong EA, Nasta
SD, Mato AR, Anak Ö, Brogdon JL, Pruteanu-Malinici I, Bhoj V,
Landsburg D, et al: Chimeric antigen receptor T cells in refractory
B-cell lymphomas. N Engl J Med. 377:2545–2554. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Singh N, Shi J, June CH and Ruella M:
Genome-editing technologies in adoptive T cell immunotherapy for
cancer. Curr Hematol Malig Rep. 12:522–529. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Schmelzer E and Reid LM: EpCAM expression
in normal, non-pathological tissues. Front Biosci. 13:3096–3100.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dai M, Yuan F, Fu C and Shen G, Hu S and
Shen G: Relationship between epithelial cell adhesion molecule
(EpCAM) overexpression and gastric cancer patients: A systematic
review and meta-analysis. PLoS One. 12:e01753572017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Fong D, Moser P, Kasal A, Seeber A, Gastl
G, Martowicz A, Wurm M, Mian C, Obrist P, Mazzoleni G and Spizzo G:
Loss of membranous expression of the intracellular domain of EpCAM
is a frequent event and predicts poor survival in patients with
pancreatic cancer. Histopathology. 64:683–692. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Seeber A, Untergasser G, Spizzo G,
Terracciano L, Lugli A, Kasal A, Kocher F, Steiner N, Mazzoleni G,
Gastl G and Fong D: Predominant expression of truncated EpCAM is
associated with a more aggressive phenotype and predicts poor
overall survival in colorectal cancer. Int J Cancer. 139:657–663.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yahyazadeh Mashhadi SM, Kazemimanesh M,
Arashkia A, Azadmanesh K, Meshkat Z, Golichenari B and Sahebkar A:
Shedding light on the EpCAM: An overview. J Cell Physiol.
234:12569–12580. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cooper LJ, Topp MS, Serrano LM, Gonzalez
S, Chang WC, Naranjo A, Wright C, Popplewell L, Raubitschek A,
Forman SJ and Jensen MC: T-cell clones can be rendered specific for
CD19: Toward the selective augmentation of the
graft-versus-B-lineage leukemia effect. Blood. 101:1637–1644. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Davila ML, Riviere I, Wang X, Bartido S,
Park J, Curran K, Chung SS, Stefanski J, Borquez-Ojeda O, Olszewska
M, et al: Efficacy and toxicity management of 19-28z CAR T cell
therapy in B cell acute lymphoblastic leukemia. Sci Transl Med.
6:224ra252014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang Y, Zhang WY, Han QW, Liu Y, Dai HR,
Guo YL, Bo J, Fan H, Zhang Y, Zhang YJ, et al: Effective response
and delayed toxicities of refractory advanced diffuse large B-cell
lymphoma treated by CD20-directed chimeric antigen
receptor-modified T cells. Clin Immunol. 155:160–175. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Till BG, Jensen MC, Wang J, Qian X, Gopal
AK, Maloney DG, Lindgren CG, Lin Y, Pagel JM, Budde LE, et al:
CD20-specific adoptive immunotherapy for lymphoma using a chimeric
antigen receptor with both CD28 and 4-1BB domains: Pilot clinical
trial results. Blood. 119:3940–3950. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Dholaria BR, Bachmeier CA and Locke F:
Mechanisms and management of chimeric antigen receptor T-cell
therapy-related toxicities. BioDrugs. 33:45–60. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lee DW, Gardner R, Porter DL, Louis CU,
Ahmed N, Jensen M, Grupp SA and Mackall CL: Current concepts in the
diagnosis and management of cytokine release syndrome. Blood.
2:188–195. 2014. View Article : Google Scholar
|
|
42
|
Lee DW, Kochenderfer JN, Stetler-Stevenson
M, Cui YK, Delbrook C, Feldman SA, Fry TJ, Orentas R, Sabatino M,
Shah NN, et al: T-cells expressing CD19 chimeric antigen receptors
for acute lymphoblastic leukaemia in children and young adults: A
phase 1 dose-escalation trial. Lancet. 385:517–528. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Oluwole OO and Davila ML: At the bedside:
Clinical review of chimeric antigen receptor (CAR) T cell therapy
for B cell malignancies. J Leukoc Biol. 100:1265–1272. 2016.
View Article : Google Scholar : PubMed/NCBI
|