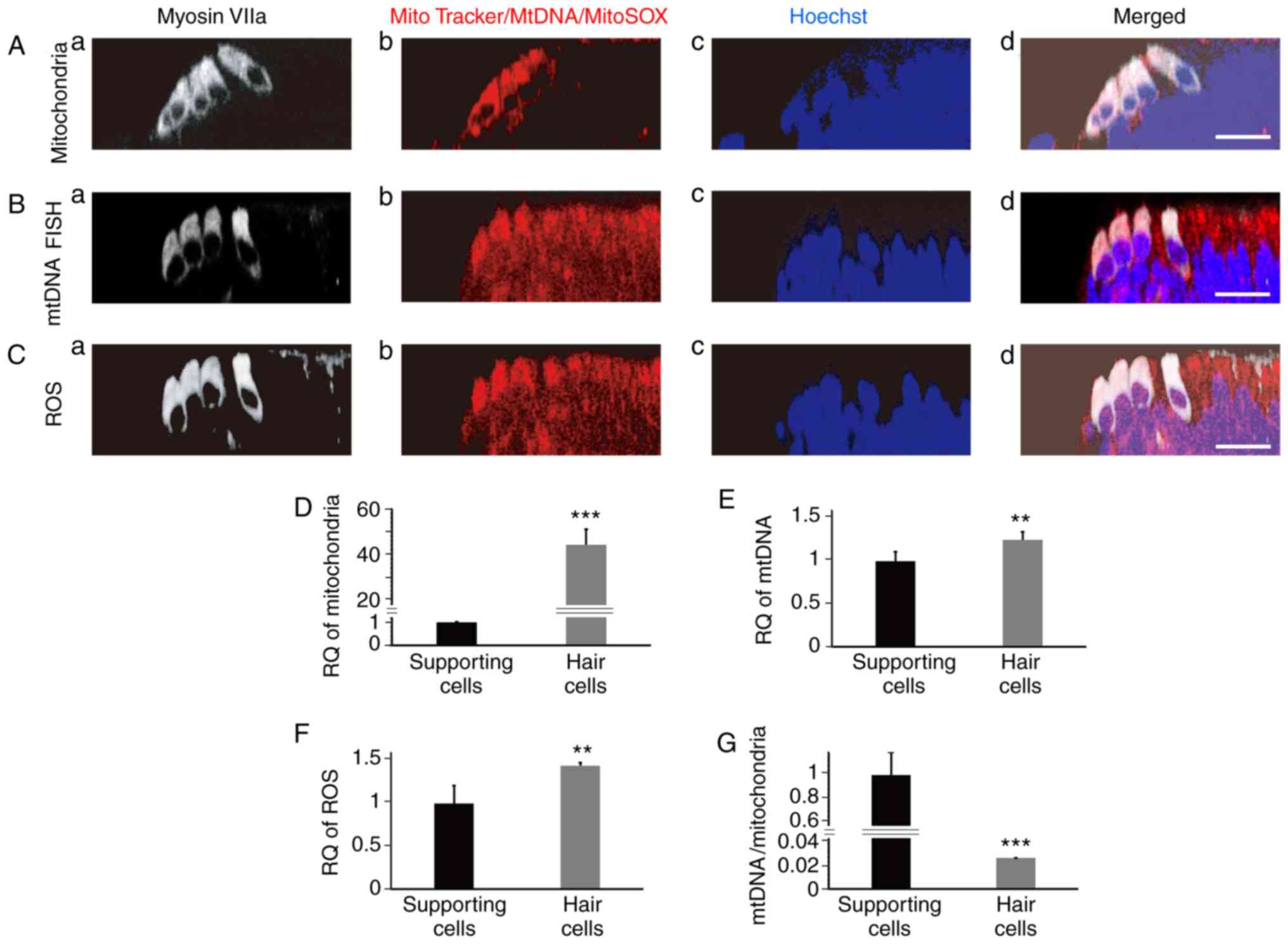
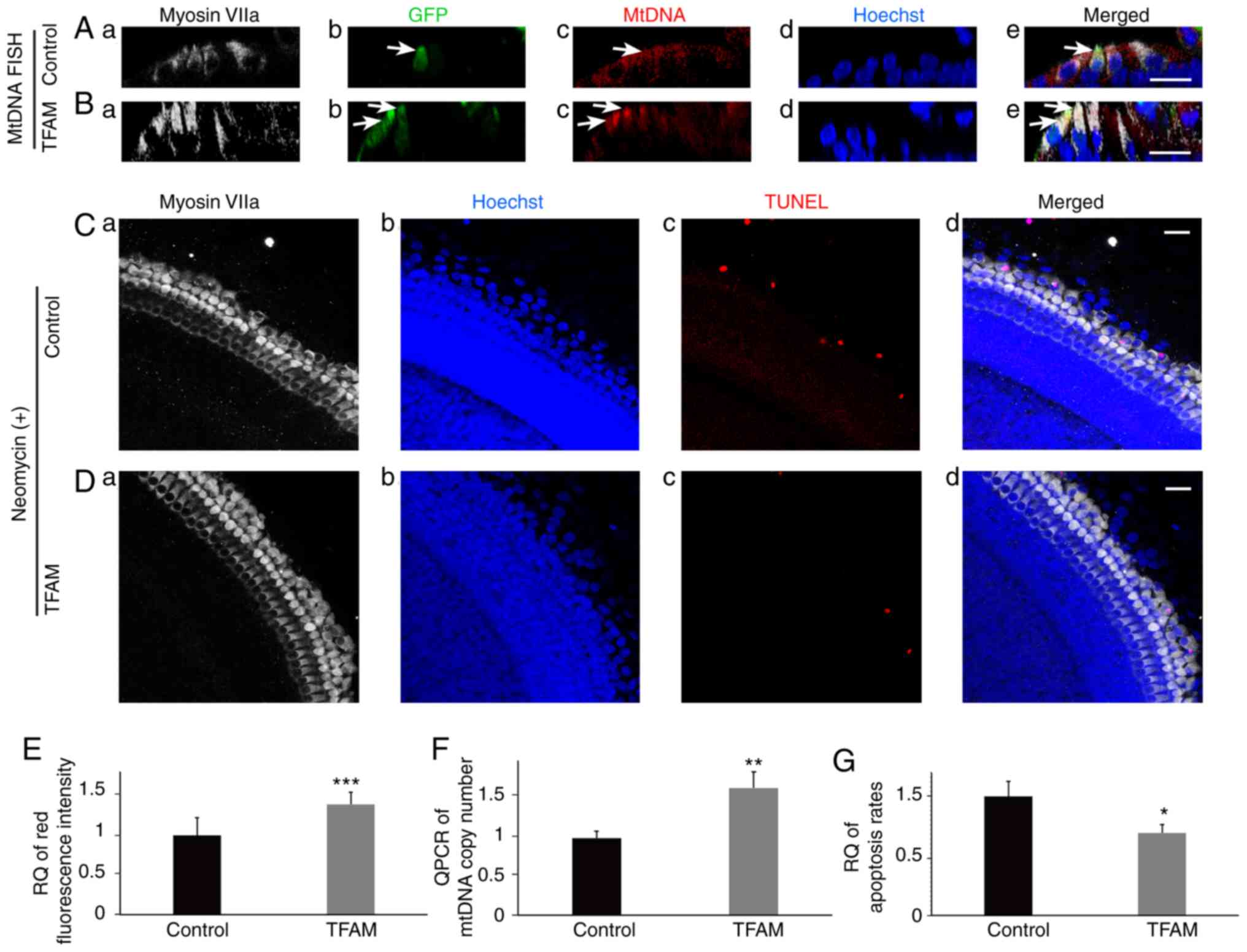
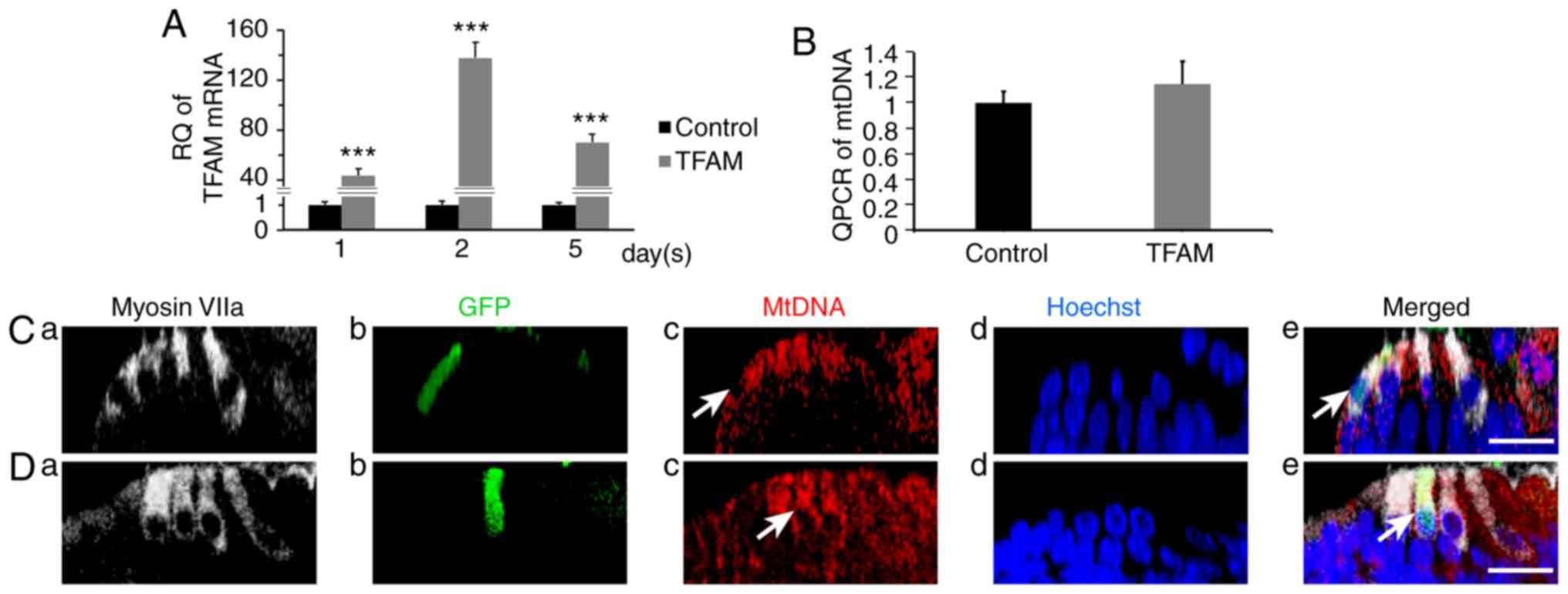
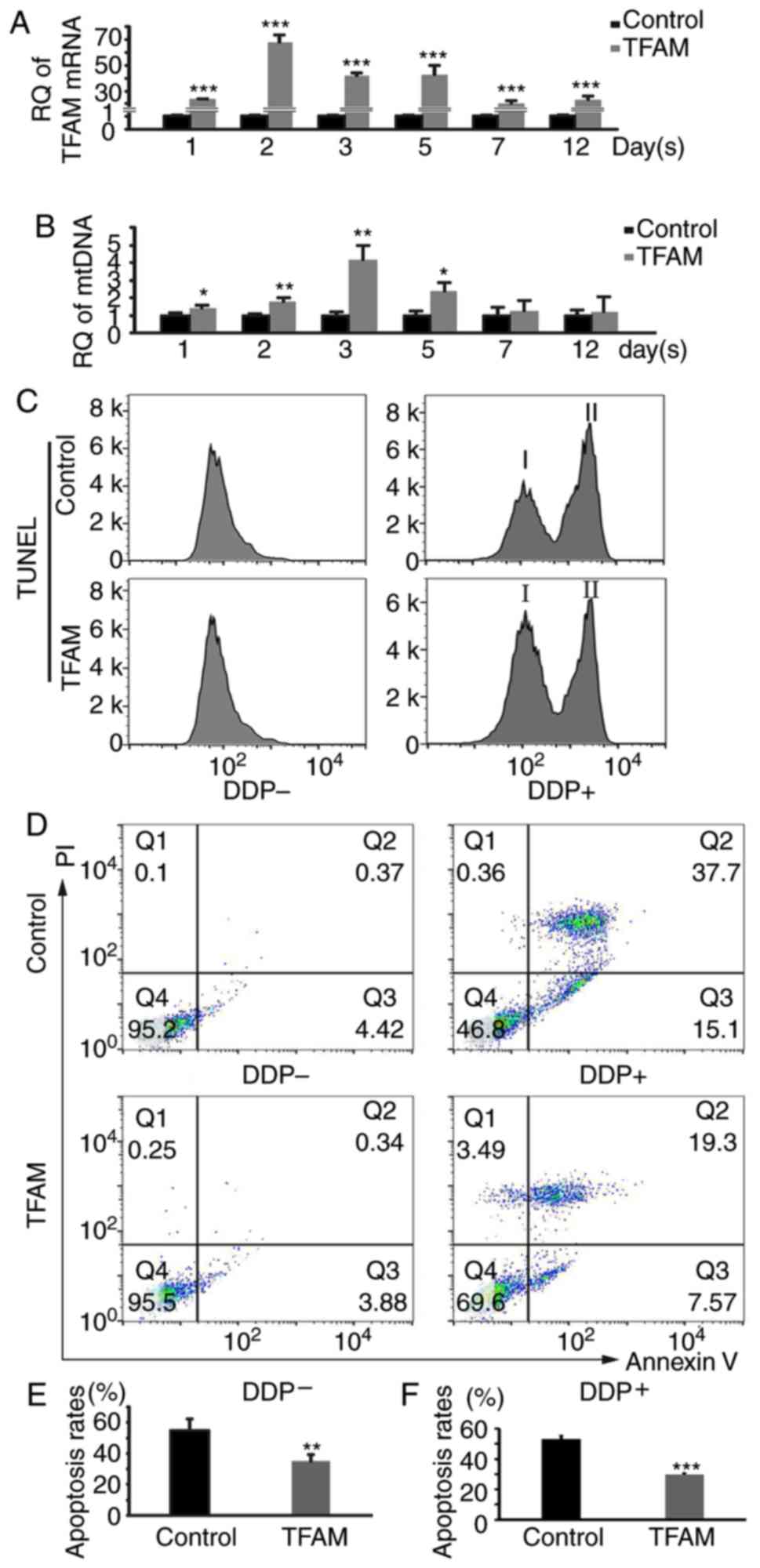
|
1
|
Zhang W, Kim SM, Wang W, Cai C, Feng Y,
Kong W and Lin X: Cochlear gene therapy for sensorineural hearing
loss: Current status and major remaining hurdles for translational
success. Front Mol Neurosci. 11:2212018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Monroe JD, Rajadinakaran G and Smith ME:
Sensory hair cell death and regeneration in fishes. Front Cell
Neurosci. 9:1312015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Atkinson PJ, Huarcaya Najarro E, Sayyid ZN
and Cheng AG: Sensory hair cell development and regeneration:
Similarities and differences. Development. 142:1561–1571. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hardie DG: Keeping the home fires burning:
AMP-activated protein kinase. J R Soc Interface. 15:201707742018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cerritelli SM, Frolova EG, Feng C,
Grinberg A, Love PE and Crouch RJ: Failure to produce mitochondrial
DNA results in embryonic lethality in Rnaseh1 null mice. Mol Cell.
11:807–815. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mei H, Sun S, Bai Y, Chen Y, Chai R and Li
H: Reduced mtDNA copy number increases the sensitivity of tumor
cells to chemotherapeutic drugs. Cell Death Dis. 6:e17102015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kalinec GM, Webster P, Lim DJ and Kalinec
F: A cochlear cell line as an in vitro system for drug ototoxicity
screening. Audiol Neurotol. 8:177–189. 2003. View Article : Google Scholar
|
|
8
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ekstrand MI, Falkenberg M, Rantanen A,
Park CB, Gaspari M, Hultenby K, Rustin P, Gustafsson CM and Larsson
NG: Mitochondrial transcription factor A regulates mtDNA copy
number in mammals. Hum Mol Genet. 13:935–944. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Goodall-Copestake WP: nrDNA:mtDNA copy
number ratios as a comparative metric for evolutionary and
conservation genetics. Heredity (Edinb). 121:105–111. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hashimoto S, Morimoto N, Yamanaka M,
Matsumoto H, Yamochi T, Goto H, Inoue M, Nakaoka Y, Shibahara H and
Morimoto Y: Quantitative and qualitative changes of mitochondria in
human preimplantation embryos. J Assist Reprod Genet. 34:573–580.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Birket MJ, Orr AL, Gerencser AA, Madden
DT, Vitelli C, Swistowski A, Brand MD and Zeng X: A reduction in
ATP demand and mitochondrial activity with neural differentiation
of human embryonic stem cells. J Cell Sci. 124:348–358. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rusecka J, Kaliszewska M, Bartnik E and
Tońska K: Nuclear genes involved in mitochondrial diseases caused
by instability of mitochondrial DNA. J Appl Genet. 59:43–57. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Foote K, Reinhold J, Yu EPK, Figg NL,
Finigan A, Murphy MP and Bennett MR: Restoring mitochondrial DNA
copy number preserves mitochondrial function and delays vascular
aging in mice. Aging Cell. 17:e127732018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Warren EB, Aicher AE, Fessel JP and
Konradi C: Mitochondrial DNA depletion by ethidium bromide
decreases neuronal mitochondrial creatine kinase: Implications for
striatal energy metabolism. PLoS One. 12:e01904562017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gustafsson CM, Falkenberg M and Larsson
NG: Maintenance and expression of mammalian mitochondrial DNA. Annu
Rev Biochem. 85:133–160. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
King GA, Hashemi Shabestari M, Taris KH,
Pandey AK, Venkatesh S, Thilagavathi J, Singh K, Krishna Koppisetti
R, Temiakov D, Roos WH, et al: Acetylation and phosphorylation of
human TFAM regulate TFAM-DNA interactions via contrasting
mechanisms. Nucleic Acids Res. 46:3633–3642. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kukat C, Davies KM, Wurm CA, Spåhr H,
Bonekamp NA, Kühl I, Joos F, Polosa PL, Park CB, Posse V, et al:
Cross-strand binding of TFAM to a single mtDNA molecule forms the
mitochondrial nucleoid. Proc Natl Acad Sci USA. 112:11288–11293.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lin SCY, Thorne PR, Housley GD and
Vlajkovic SM: Resistance to neomycin ototoxicity in the extreme
basal (hook) region of the mouse cochlea. Histochem Cell Biol.
150:281–289. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jiang M, Karasawa T and Steyger PS:
Aminoglycoside-induced cochleotoxicity: A review. Front Cell
Neurosci. 11:3082017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen Y, Yu H, Zhang Y, Li W, Lu N, Ni W,
He Y, Li J, Sun S, Wang Z and Li H: Cotransfection of Pax2 and
Math1 promote in situ cochlear hair cell regeneration after
neomycin insult. Sci Rep. 3:29962013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee SR and Han J: Mitochondrial nucleoid:
Shield and switch of the mitochondrial genome. Oxid Med Cell
Longev. 2017:80609492017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gilkerson R, Bravo L, Garcia I, Gaytan N,
Herrera A, Maldonado A and Quintanilla B: The mitochondrial
nucleoid: Integrating mitochondrial DNA into cellular homeostasis.
Cold Spring Harb Perspect Biol. 5:a0110802013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kukat C, Wurm CA, Spåhr H, Falkenberg M,
Larsson NG and Jakobs S: Super-resolution microscopy reveals that
mammalian mitochondrial nucleoids have a uniform size and
frequently contain a single copy of mtDNA. Proc Natl Acad Sci USA.
108:13534–13539. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gerhold JM, Cansiz-Arda Ş, Lõhmus M,
Engberg O, Reyes A, van Rennes H, Sanz A, Holt IJ, Cooper HM and
Spelbrink JN: Human mitochondrial DNA-protein complexes attach to a
cholesterol-rich membrane structure. Sci Rep. 5:152922015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Desai R, Frazier AE, Durigon R, Patel H,
Jones AW, Dalla Rosa I, Lake NJ, Compton AG, Mountford HS, Tucker
EJ, et al: ATAD3 gene cluster deletions cause cerebellar
dysfunction associated with altered mitochondrial DNA and
cholesterol metabolism. Brain. 140:1595–1610. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Rottenberg H and Hoek JB: The path from
mitochondrial ROS to aging runs through the mitochondrial
permeability transition pore. Aging Cell. 16:943–955. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Treulen F, Uribe P, Boguen R and Villegas
JV: Mitochondrial permeability transition increases reactive oxygen
species production and induces DNA fragmentation in human
spermatozoa. Hum Reprod. 30:767–776. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zorov DB, Juhaszova M and Sollott SJ:
Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS
release. Physiol Rev. 94:909–950. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Baines CP and Gutiérrez-Aguilar M: The
still uncertain identity of the channel-forming unit(s) of the
mitochondrial permeability transition pore. Cell Calcium.
73:121–130. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bernardi P and Di Lisa F: The
mitochondrial permeability transition pore: Molecular nature and
role as a target in cardioprotection. J Mol Cell Cardiol.
78:100–106. 2015. View Article : Google Scholar : PubMed/NCBI
|