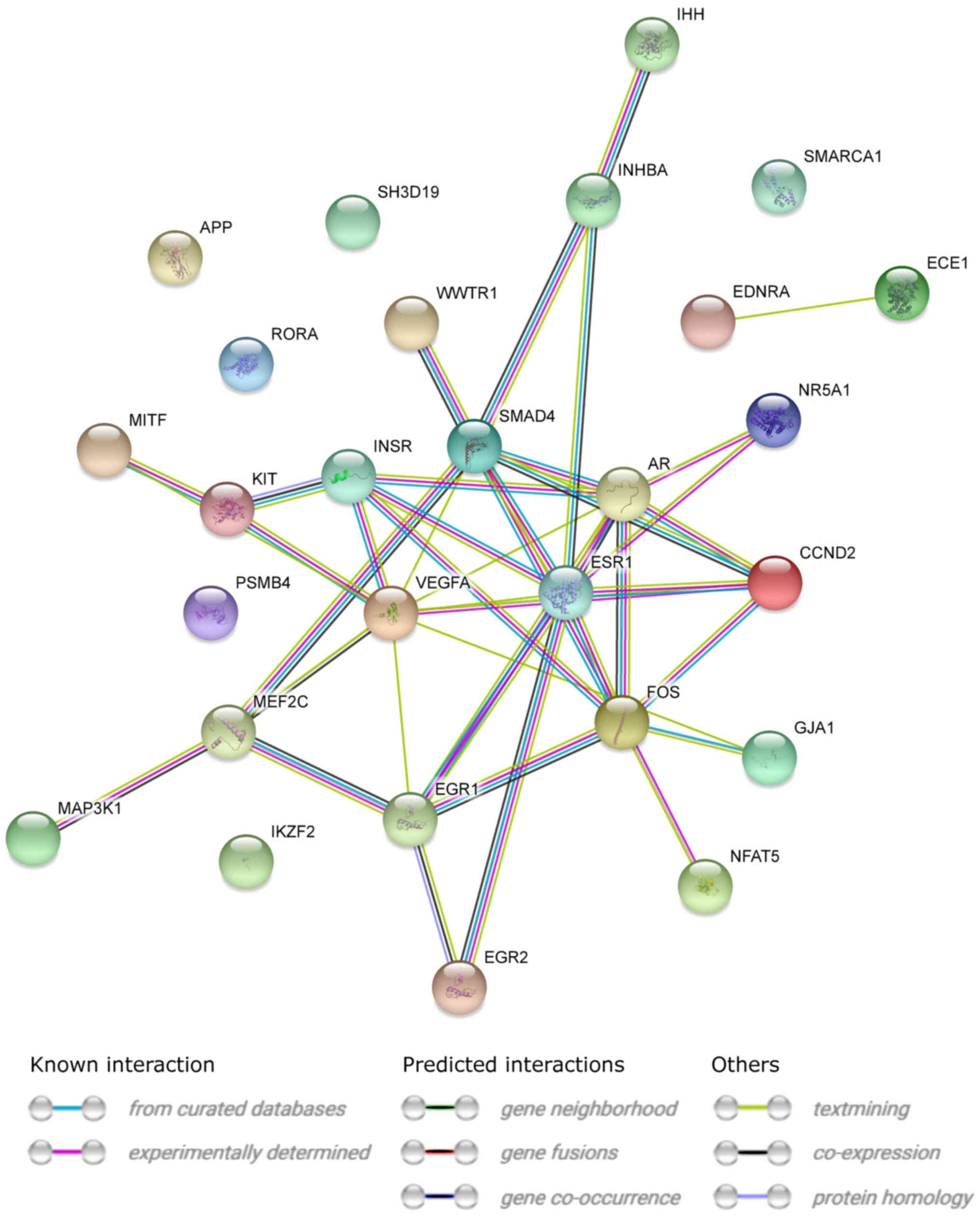
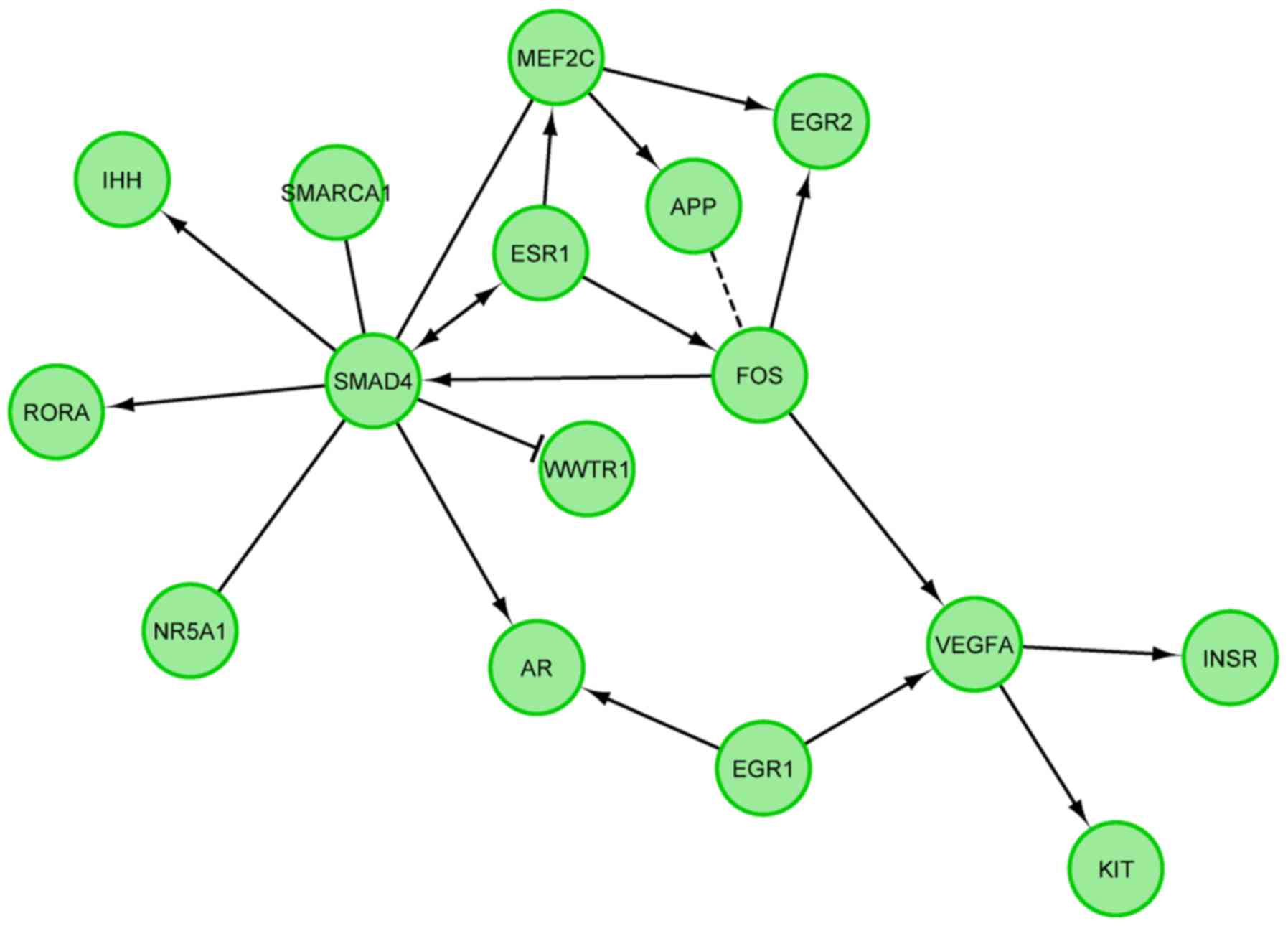
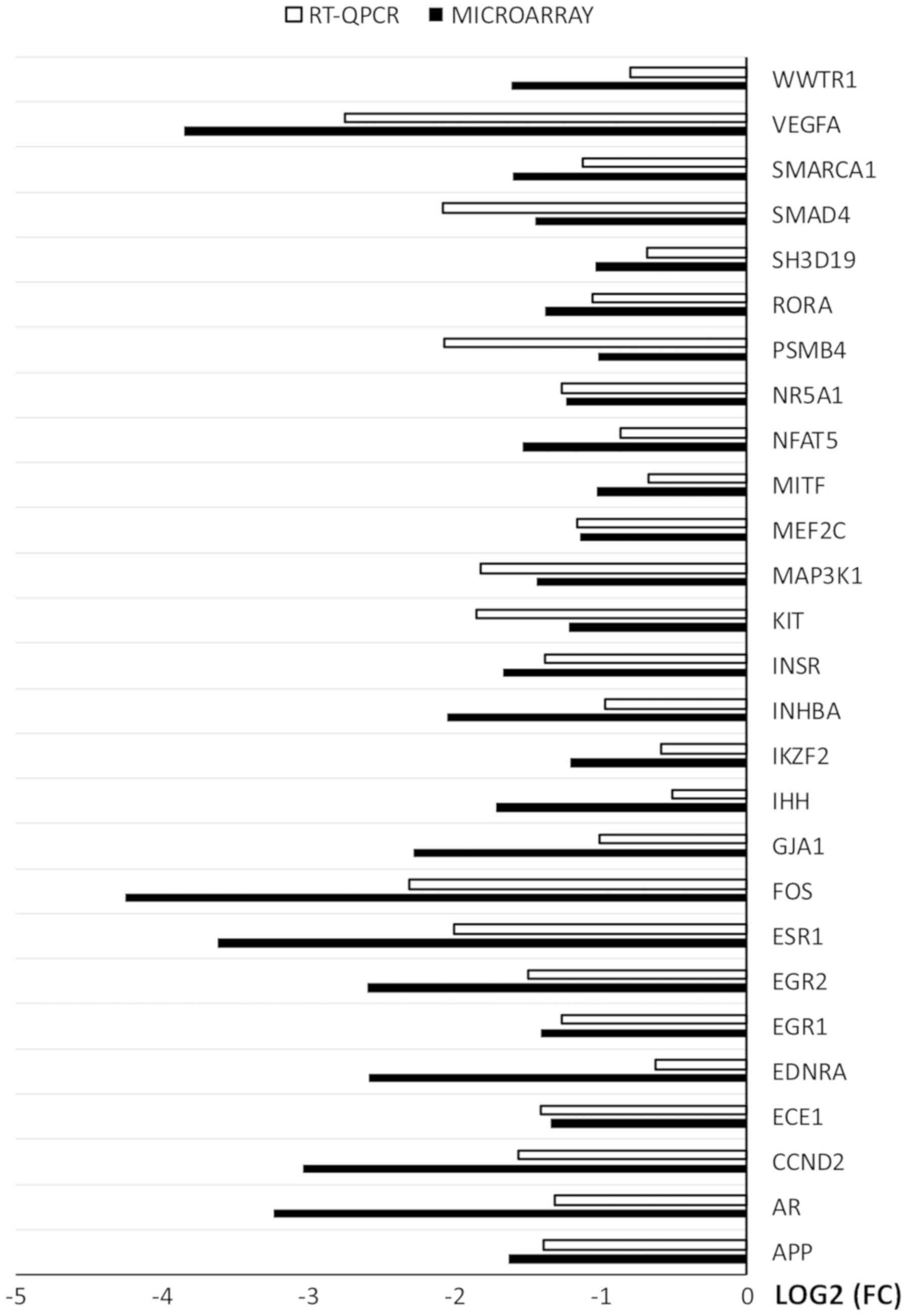
|
1
|
Eppig JJ: Oocyte control of ovarian
follicular development and function in mammals. Reproduction.
122:829–838. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gilchrist RB, Ritter LJ and Armstrong DT:
Oocyte-somatic cell interactions during follicle development in
mammals. Anim Reprod Sci. 82-83:431–446. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Matzuk MM, Burns KH, Viveiros MM and Eppig
JJ: Intercellular communication in the mammalian ovary: Oocytes
carry the conversation. Science. 296:2178–2180. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rybska M, Knap S, Jankowski M, Jeseta M,
Bukowska D, Antosik P, Nowicki M, Zabel M, Kempisty B and Jaśkowski
JM: Cytoplasmic and nuclear maturation of oocytes in mammals-living
in the shadow of cells developmental capability. Med J Cell Biol.
6:13–17. 2018. View Article : Google Scholar
|
|
5
|
Rybska M, Knap S, Stefańska K, Jankowski
M, Gliszczyńska AC, Popis M, Jeseta M, Bukowska D, Antosik P,
Kempisty B and Jaśkowski JM: Transforming growth factor (TGF)-is it
a key protein in mammalian reproductive biology? Med J Cell Biol.
6:125–130. 2018. View Article : Google Scholar
|
|
6
|
Rybska M, Knap S, Jankowski M, Jeseta M,
Bukowska D, Antosik P, Nowicki M, Zabel M, Kempisty B and Jaśkowski
JM: Characteristic of factors influencing the proper course of
folliculogenesis in mammals. Med J Cell Biol. 6:33–38. 2018.
View Article : Google Scholar
|
|
7
|
Regassa A, Rings F, Hoelker M, Cinar U,
Tholen E, Looft C, Schellander K and Tesfaye D: Transcriptome
dynamics and molecular cross-talk between bovine oocyte and its
companion cumulus cells. BMC Genomics. 12:572011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Borys-Wójcik S, Kocherova I, Celichowski
P, Popis M, Jeseta M, Bukowska D, Antosik P, Nowicki M and Kempisty
B: Protein oligomerization is the biochemical process highly
up-regulated in porcine oocytes before in vitro maturation (IVM).
Med J Cell Biol. 6:155–162. 2018. View Article : Google Scholar
|
|
9
|
Budna J, Celichowski P, Bryja A, Jeseta M,
Jankowski M, Bukowska D, Antosik P, Nowicki A, Brüssow KP, Bruska
M, et al: Expression changes in fatty acid metabolic processrelated
genes in porcine oocytes during in vitro maturation. Med J Cell
Biol. 6:48–54. 2018. View Article : Google Scholar
|
|
10
|
Kranc W, Brązert M, Ożegowska K,
Budna-Tukan J, Celichowski P, Jankowski M, Bryja A, Nawrocki MJ,
Popis M, Jeseta M, et al: Response to abiotic and organic
substances stimulation belongs to ontologic groups significantly
up-regulated in porcine immature oocytes. Med J Cell Biol.
6:91–100. 2018. View Article : Google Scholar
|
|
11
|
Chermuła B, Brązert M, Jeseta M, Ożegowska
K, Sujka-Kordowska P, Konwerska A, Bryja A, Kranc W, Jankowski M,
Nawrocki MJ, et al: The unique mechanisms of cellular
proliferation, migration and apoptosis are regulated through oocyte
maturational development-A complete transcriptomic and
histochemical study. Int J Mol Sci. 20:E842018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ożegowska K, Dyszkiewicz-Konwińska M,
Celichowski P, Nawrocki MJ, Bryja A, Jankowski M, Kranc W, Brązert
M, Knap S, Jeseta M, et al: Expression pattern of new genes
regulating female sex differentiation and in vitro maturational
status of oocytes in pigs. Theriogenology. 121:122–133. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Borys S, Brązert M, Jankowski M, Kocherova
I, Ożegowska K, Celichowski P, Nawrocki MJ, Kranc W, Bryja A, Kulus
M, et al: Enzyme linked receptor protein signaling pathway is one
of the ontology groups that are highly up-regulated in porcine
oocytes before in vitro maturation. J Biol Regul Homeost Agents.
32:1089–1103. 2018.PubMed/NCBI
|
|
14
|
Pujol M, López-Béjar M and Paramio MT:
Developmental competence of heifer oocytes selected using the
brilliant cresyl blue (BCB) test. Theriogenology. 61:735–744. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Le Guienne B: Small atlas of bovine
oocyte. Elevage et Insemination (France). 24–30. 1998.[In
French].
|
|
16
|
Chomczynski P and Sacchi N: Single-step
method of RNA isolation by acid guanidinium
thiocyanate-phenol-chloroform extraction. Anal Biochem.
162:156–159. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nawrocki MJ, Celichowski P, Jankowski M,
Kranc W, Bryja A, Borys-Wójcik S, Jeseta M, Antosik P, Bukowska D,
Bruska M, et al: Ontology groups representing angiogenesis and
blood vessels development are highly up-regulated during porcine
oviductal epithelial cells long-term real-time proliferation-a
primary cell culture approach. Med J Cell Biol. 6:186–194. 2018.
View Article : Google Scholar
|
|
18
|
Budna J, Celichowski P, Karimi P, Kranc W,
Bryja A, Ciesiółka S, Rybska M, Borys S, Jeseta M, Bukowska D, et
al: Does porcine oocytes maturation in vitro is regulated by genes
involved in transforming growth factor beta receptor signaling
pathway? Adv Cell Biol. 5:1–14. 2017. View Article : Google Scholar
|
|
19
|
Curran T and Franza BR Jr: Fos and jun:
The AP-1 connection. Cell. 55:395–397. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Angel P and Karin M: The role of Jun, Fos
and the AP-1 complex in cell-proliferation and transformation.
Biochim Biophys Acta. 1072:129–157. 1991.PubMed/NCBI
|
|
21
|
Rusovici R and LaVoie HA: Expression and
distribution of AP-1 transcription factors in the porcine ovary.
Biol Reprod. 69:64–74. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Johnson RS, Spiegelman BM and Papaioannou
V: Pleiotropic effects of a null mutation in the c-fos
proto-oncogene. Cell. 71:577–586. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Müller R, Tremblay JM, Adamson ED and
Verma IM: Tissue and cell type-specific expression of two human
c-onc genes. Nature. 304:454–456. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hattori MA, Kato Y and Fujihara N:
Retinoic acid suppression of endothelial nitric oxide synthase in
porcine oocyte. Can J Physiol Pharmacol. 80:777–782. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Blaha M, Nemcova L, Kepkova KV, Vodicka P
and Prochazka R: Gene expression analysis of pig cumulus-oocyte
complexes stimulated in vitro with follicle stimulating hormone or
epidermal growth factor-like peptides. Reprod Biol Endocrinol.
13:1132015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Leung DW, Cachianes G, Kuang WJ, Goeddel
DV and Ferrara N: Vascular endothelial growth factor is a secreted
angiogenic mitogen. Science. 246:1306–1309. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ferrara N and Henzel WJ: Pituitary
follicular cells secrete a novel heparin-binding growth factor
specific for vascular endothelial cells. Biochem Biophys Res
Commun. 161:851–858. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ferrara N and Davis-Smyth T: The biology
of vascular endothelial growth factor. Endocr Rev. 18:4–25. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sousa LM, Campos DB, Fonseca VU, Viau P,
Kfoury JR Jr, Oliveira CA, Binelli M, Buratini J Jr and Papa PC:
Vascular endothelial growth factor A (VEGFA) modulates bovine
placenta steroidogenesis in vitro. Placenta. 33:788–794. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shimizu T: Promotion of ovarian follicular
development by injecting vascular endothelial growth factor (VEGF)
and growth differentiation factor 9 (GDF-9) genes. J Reprod Dev.
52:23–32. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
McFee RM, Rozell TG and Cupp AS: The
balance of proangiogenic and antiangiogenic VEGFA isoforms regulate
follicle development. Cell Tissue Res. 349:635–647. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bui TMT, Nguyễn KX, Karata A, Ferré P,
Trần MT, Wakai T and Funahashi H: Presence of vascular endothelial
growth factor during the first half of IVM improves the meiotic and
developmental competence of porcine oocytes from small follicles.
Reprod Fertil Dev. 29:1902–1909. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ravindranath N, Little-Ihrig L, Phillips
HS, Ferrara N and Zeleznik AJ: Vascular endothelial growth factor
messenger ribonucleic acid expression in the primate ovary.
Endocrinology. 131:254–260. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yamamoto S, Konishi I, Tsuruta Y, Nanbu K,
Mandai M, Kuroda H, Matsushita K, Hamid AA, Yura Y and Mori T:
Expression of vascular endothelial growth factor (VEGF) during
folliculogenesis and corpus luteum formation in the human ovary.
Gynecol Endocrinol. 11:371–381. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xu J, Xu M, Bernuci MP, Fisher TE, Shea
LD, Woodruff TK, Zelinski MB and Stouffer RL: Primate follicular
development and oocyte maturation in vitro. Adv Exp Med Biol.
761:43–67. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Silva CM, Matos MH, Rodrigues GQ, Faustino
LR, Pinto LC, Chaves RN, Araújo VR, Campello CC and Figueiredo JR:
In vitro survival and development of goat preantral follicles in
two different oxygen tensions. Anim Reprod Sci. 117:83–89. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Walters KA, Simanainen U and Handelsman
DJ: Molecular insights into androgen actions in male and female
reproductive function from androgen receptor knockout models. Hum
Reprod Update. 16:543–558. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gleicher N, Weghofer A and Barad DH: The
role of androgens in follicle maturation and ovulation induction:
Friend or foe of infertility treatment? Reprod Biol Endocrinol.
9:1162011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lenie S and Smitz J: Functional AR
signaling is evident in an in vitro mouse follicle culture bioassay
that encompasses most stages of folliculogenesis. Biol Reprod.
80:685–695. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li M, Ai JS, Xu BZ, Xiong B, Yin S, Lin
SL, Hou Y, Chen DY, Schatten H and Sun QY: Testosterone potentially
triggers meiotic resumption by activation of intra-oocyte SRC and
MAPK in porcine oocytes. Biol Reprod. 79:897–905. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Van Nieuwerburgh F, Stoop D, Cabri P,
Dhont M, Deforce D and De Sutter P: Shorter CAG repeats in the
androgen receptor gene may enhance hyperandrogenicity in polycystic
ovary syndrome. Gynecol Endocrinol. 24:669–673. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
O'Donovan KJ, Tourtellotte WG, Millbrandt
J and Baraban JM: The EGR family of transcription-regulatory
factors: Progress at the interface of molecular and systems
neuroscience. Trends Neurosci. 22:167–173. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Shin H, Seol DW, Nam M, Song H, Lee DR and
Lim HJ: Expression of Egr3 in mouse gonads and its localization and
function in oocytes. Asian-Australas J Anim Sci. 30:781–787. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Thiel G, Müller I and Rössler OG:
Expression, signaling and function of Egr transcription factors in
pancreatic β-cells and insulin-responsive tissues. Mol Cell
Endocrinol. 388:10–19. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Safford M, Collins S, Lutz MA, Allen A,
Huang CT, Kowalski J, Blackford A, Horton MR, Drake C, Schwartz RH
and Powell JD: Egr-2 and Egr-3 are negative regulators of T cell
activation. Nat Immunol. 6:472–480. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Fang F, Ooka K, Bhattachyya S, Wei J, Wu
M, Du P, Lin S, Del Galdo F, Feghali-Bostwick CA and Varga J: The
early growth response gene Egr2 (alias Krox20) is a novel
transcriptional target of transforming growth factor-β that is
up-regulated in systemic sclerosis and mediates profibrotic
responses. Am J Pathol. 178:2077–2090. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Nagarajan R, Svaren J, Le N, Araki T,
Watson M and Milbrandt J: EGR2 mutations in inherited neuropathies
dominant-negatively inhibit myelin gene expression. Neuron.
30:355–368. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Hu TM, Chen CH, Chuang YA, Hsu SH and
Cheng MC: Resequencing of early growth response 2 (EGR2) gene
revealed a recurrent patient-specific mutation in schizophrenia.
Psychiatry Res. 228:958–960. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Le N, Nagarajan R, Wang JY, Araki T,
Schmidt RE and Milbrandt J: Analysis of congenital hypomyelinating
Egr2Lo/Lo nerves identifies Sox2 as an inhibitor of schwann cell
differentiation and myelination. Proc Natl Acad Sci USA.
102:2596–2601. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Li X, Zhang Z, Yu M, Li L, Du G, Xiao W
and Yang H: Involvement of miR-20a in promoting gastric cancer
progression by targeting early growth response 2 (EGR2). Int J Mol
Sci. 14:16226–16239. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Barbeau DJ, La KT, Kim DS, Kerpedjieva SS,
Shurin GV and Tamama K: Early growth response-2 signaling mediates
immunomodulatory effects of human multipotential stromal cells.
Stem Cells Dev. 23:155–166. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jin H, Won M, Shin E, Kim HM, Lee K and
Bae J: EGR2 is a gonadotropin-induced survival factor that controls
the expression of IER3 in ovarian granulosa cells. Biochem Biophys
Res Commun. 482:877–882. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Burger HG: Inhibin: Definition and
nomenclature, including related substances. J Endocrinol.
117:159–160. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Seder CW, Hartojo W, Lin L, Silvers AL,
Wang Z, Thomas DG, Giordano TJ, Chen G, Chang AC, Orringer MB and
Beer DG: Upregulated INHBA expression may promote cell
proliferation and is associated with poor survival in lung
adenocarcinoma. Neoplasia. 11:388–396. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Howley BV, Hussey GS, Link LA and Howe PH:
Translational regulation of inhibin βa by TGFβ via the RNA-binding
protein hnRNP E1 enhances the invasiveness of epithelial-to-
mesenchymal transitioned cells. Oncogene. 35:1725–1735. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Katayama Y, Oshima T, Sakamaki K, Aoyama
T, Sato T, Masudo K, Shiozawa M, Yoshikawa T, Rino Y, Imada T and
Masuda M: Clinical significance of INHBA gene expression in
patients with gastric cancer who receive curative resection
followed by adjuvant s-1 chemotherapy. In Vivo. 31:565–571. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Donovan P, Dubey OA, Kallioinen S, Rogers
KW, Muehlethaler K, Müller P, Rimoldi D and Constam DB: Paracrine
activin-A signaling promotes melanoma growth and metastasis through
immune evasion. J Invest Dermatol. 137:2578–2587. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Locci M, Wu JE, Arumemi F, Mikulski Z,
Dahlberg C, Miller AT and Crotty S: Activin A programs the
differentiation of human TFH cells. Nat Immunol. 17:976–984. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
McKenzie LJ, Pangas SA, Carson SA, Kovanci
E, Cisneros P, Buster JE, Amato P and Matzuk MM: Human cumulus
granulosa cell gene expression: A predictor of fertilization and
embryo selection in women undergoing IVF. Hum Reprod. 19:2869–2874.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Assidi M, Dufort I, Ali A, Hamel M,
Algriany O, Dielemann S and Sirard MA: Identification of potential
markers of oocyte competence expressed in bovine cumulus cells
matured with follicle-stimulating hormone and/or phorbol myristate
acetate in vitro. Biol Reprod. 79:209–222. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Bristol-Gould SK, Kreeger PK, Selkirk CG,
Kilen SM, Cook RW, Kipp JL, Shea LD, Mayo KE and Woodruff TK:
Postnatal regulation of germ cells by activin: The establishment of
the initial follicle pool. Dev Biol. 298:132–148. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Myers M, Middlebrook BS, Matzuk MM and
Pangas SA: Loss of inhibin alpha uncouples oocyte-granulosa cell
dynamics and disrupts postnatal folliculogenesis. Dev Biol.
334:458–467. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Maguire JJ and Davenport AP: Endothelin
receptors and their antagonists. Semin Nephrol. 35:125–136. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Asai R, Kurihara Y, Fujisawa K, Sato T,
Kawamura Y, Kokubo H, Tonami K, Nishiyama K, Uchijima Y,
Miyagawa-Tomita S and Kurihara H: Endothelin receptor type A
expression defines a distinct cardiac subdomain within the heart
field and is later implicated in chamber myocardium formation.
Development. 137:3823–3833. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Kawamura K, Ye Y, Liang CG, Kawamura N,
Gelpke MS, Rauch R, Tanaka T and Hsueh AJ: Paracrine regulation of
the resumption of oocyte meiosis by endothelin-1. Dev Biol.
327:62–70. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Cui L, Shen J, Fang L, Mao X, Wang H and
Ye Y: Endothelin-1 promotes human germinal vesicle-stage oocyte
maturation by downregulating connexin-26 expression in cumulus
cells. Mol Hum Reprod. 24:27–36. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Cheng JC, Chang HM, Fang L, Sun YP and
Leung PC: TGF-β1 up-regulates connexin43 expression: A potential
mechanism for human trophoblast cell differentiation. J Cell
Physiol. 230:1558–1566. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Salameh A, Haunschild J, Bräuchle P, Peim
O, Seidel T, Reitmann M, Kostelka M, Bakhtiary F, Dhein S and
Dähnert I: On the role of the gap junction protein Cx43 (GJA1) in
human cardiac malformations with fallot-pathology. A study on
paediatric cardiac specimen. PLoS One. 9:e953442014. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Edry I, Sela-Abramovich S and Dekel N:
Meiotic arrest of oocytes depends on cell-to-cell communication in
the ovarian follicle. Mol Cell Endocrinol. 252:102–106. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Li SH, Lin MH, Hwu YM, Lu CH, Yeh LY, Chen
YJ and Lee RK: Correlation of cumulus gene expression of GJA1,
PRSS35, PTX3, and SERPINE2 with oocyte maturation, fertilization,
and embryo development. Reprod Biol Endocrinol. 13:932015.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Hasegawa J, Yanaihara A, Iwasaki S,
Mitsukawa K, Negishi M and Okai T: Reduction of connexin 43 in
human cumulus cells yields good embryo competence during ICSI. J
Assist Reprod Genet. 24:463–466. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Wang HX, Tong D, El-Gehani F, Tekpetey FR
and Kidder GM: Connexin expression and gap junctional coupling in
human cumulus cells: Contribution to embryo quality. J Cell Mol
Med. 13:972–984. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Amberger JS, Bocchini CA, Schiettecatte F,
Scott AF and Hamosh A: OMIM.org: Online mendelian inheritance in
man (OMIM®), an online catalog of human genes and
genetic disorders. Nucleic Acids Res. 43:D789–D798. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Calatayud NE, Pask AJ, Shaw G, Richings
NM, Osborn S and Renfree MB: Ontogeny of the oestrogen receptors
ESR1 and ESR2 during gonadal development in the tammar wallaby,
Macropus eugenii. Reproduction. 139:599–611. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Jameson JL, DeGroot LJ, De Kretser DM,
Giudice LC, Grossman AB, Melmed S, Potts JT Jr and Weir GC:
Endocrinology: Adult & Pediatric. 7th. Elsevier; Philadelphia,
PA: 2016
|
|
76
|
Labrie F, Luu-The V, Lin SX, Simard J and
Labrie C: Role of 17β-hydroxysteroid dehydrogenases in sex steroid
formation in peripheral intracrine tissues. Trends Endocrinol
Metab. 11:421–427. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Kuiper GG, Carlsson B, Grandien K, Enmark
E, Häggblad J, Nilsson S and Gustafsson JA: Comparison of the
ligand binding specificity and transcript tissue distribution of
estrogen receptors alpha and beta. Endocrinology. 138:863–870.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Bondesson M, Hao R, Lin CY, Williams C and
Gustafsson JÅ: Estrogen receptor signaling during vertebrate
development. Biochim Biophys Acta. 1849:142–151. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Paterni I, Granchi C, Katzenellenbogen JA
and Minutolo F: Estrogen receptors alpha (ERα) and beta (ERβ):
Subtype-selective ligands and clinical potential. Steroids.
90:13–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Pelletier G and El-Alfy M:
Immunocytochemical localization of estrogen receptors alpha and
beta in the human reproductive organs. J Clin Endocrinol Metab.
85:4835–4840. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Nathan MR and Schmid P: A review of
fulvestrant in breast cancer. Oncol Ther. 5:17–29. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Artini PG, Tatone C, Sperduti S, D'Aurora
M, Franchi S, Di Emidio G, Ciriminna R, Vento M, Di Pietro C,
Stuppia L, et al: Cumulus cells surrounding oocytes with high
developmental competence exhibit down-regulation of phosphoinositol
1, 3 kinase/protein kinase B (PI3K/AKT) signalling genes involved
in proliferation and survival. Hum Reprod. 32:2474–2484. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Musgrove EA, Caldon CE, Barraclough J,
Stone A and Sutherland RL: Cyclin D as a therapeutic target in
cancer. Nat Rev Cancer. 11:558–572. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Robker RL and Richards JS: Hormone-induced
proliferation and differentiation of granulosa cells: A coordinated
balance of the cell cycle regulators cyclin D2 and p27Kip1. Mol
Endocrinol. 12:924–940. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Han Y, Xia G and Tsang BK: Regulation of
cyclin D2 expression and degradation by follicle-stimulating
hormone during rat granulosa cell proliferation in vitro. Biol
Reprod. 88:572013. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
François CM, Petit F, Giton F, Gougeon A,
Ravel C, Magre S, Cohen-Tannoudji J and Guigon CJ: A novel action
of follicle-stimulating hormone in the ovary promotes estradiol
production without inducing excessive follicular growth before
puberty. Sci Rep. 7:462222017. View Article : Google Scholar : PubMed/NCBI
|