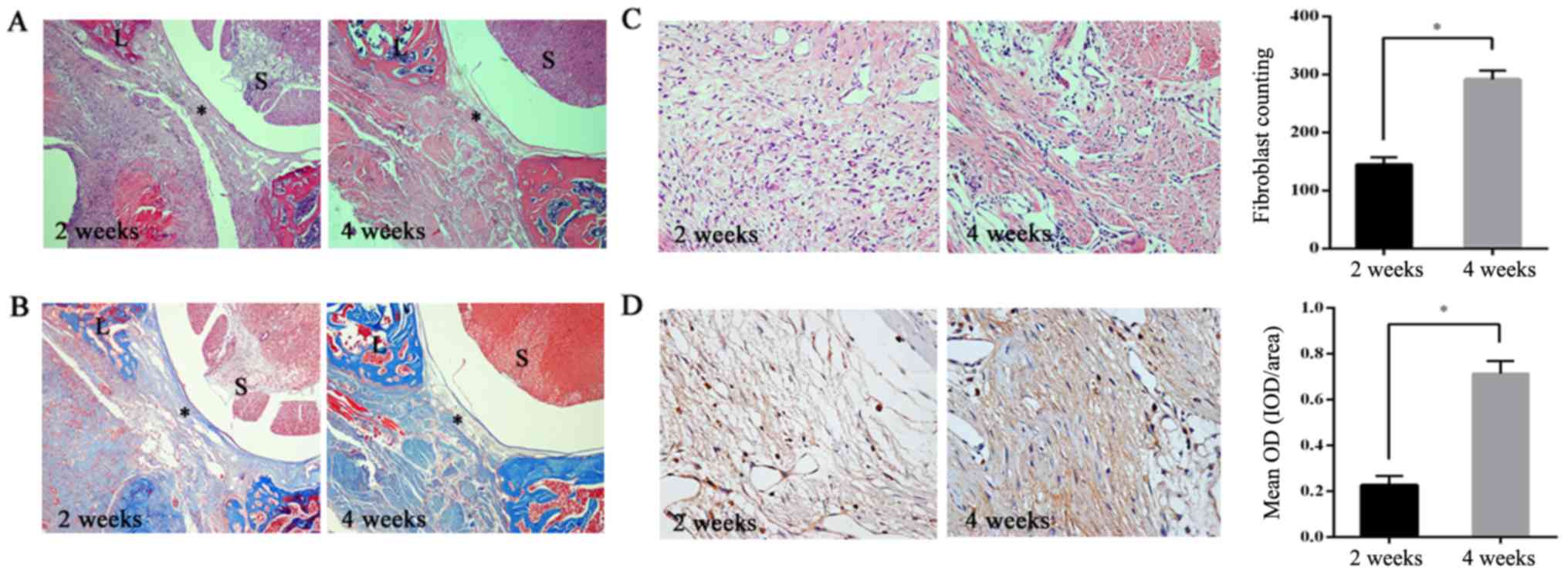
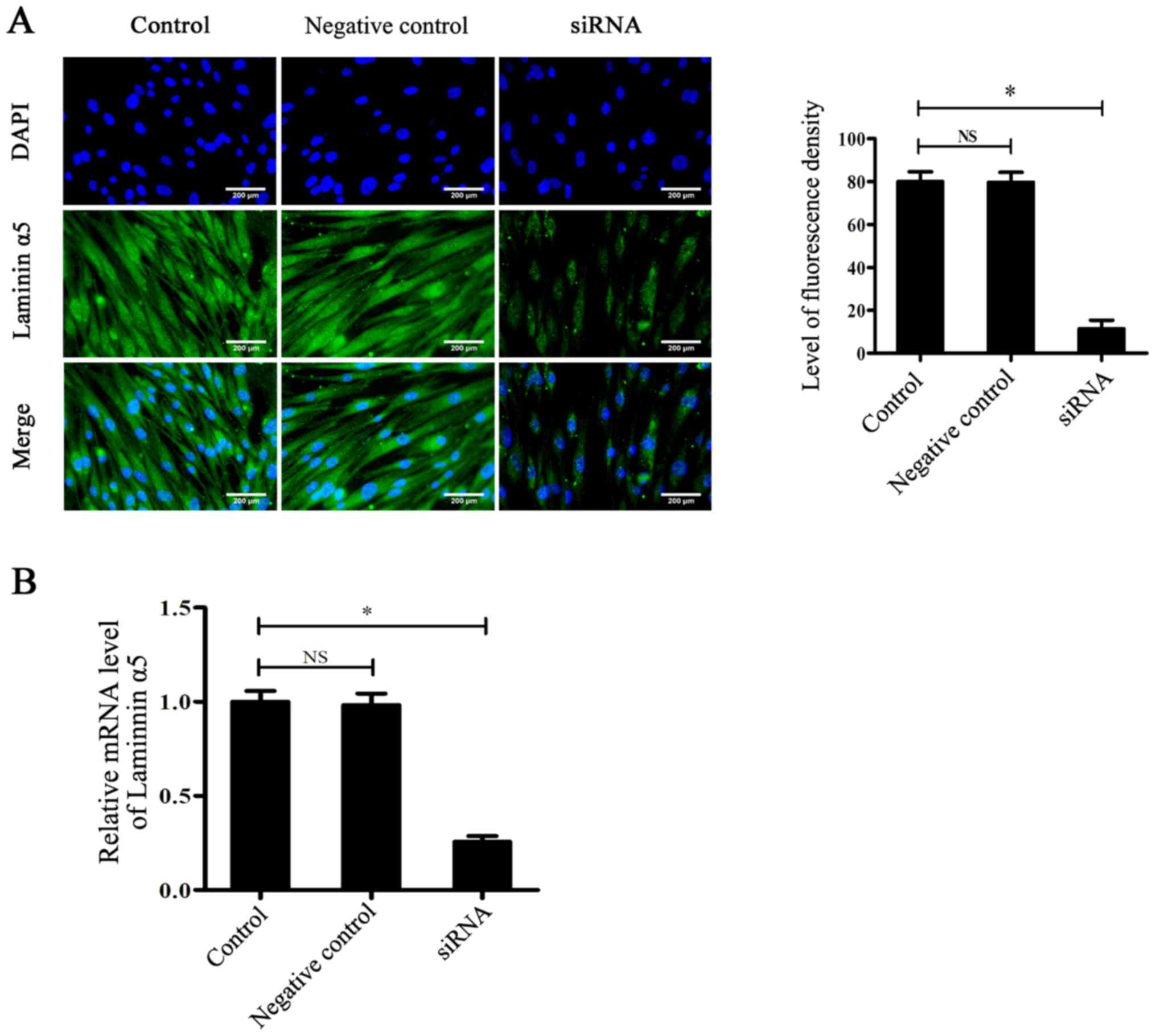
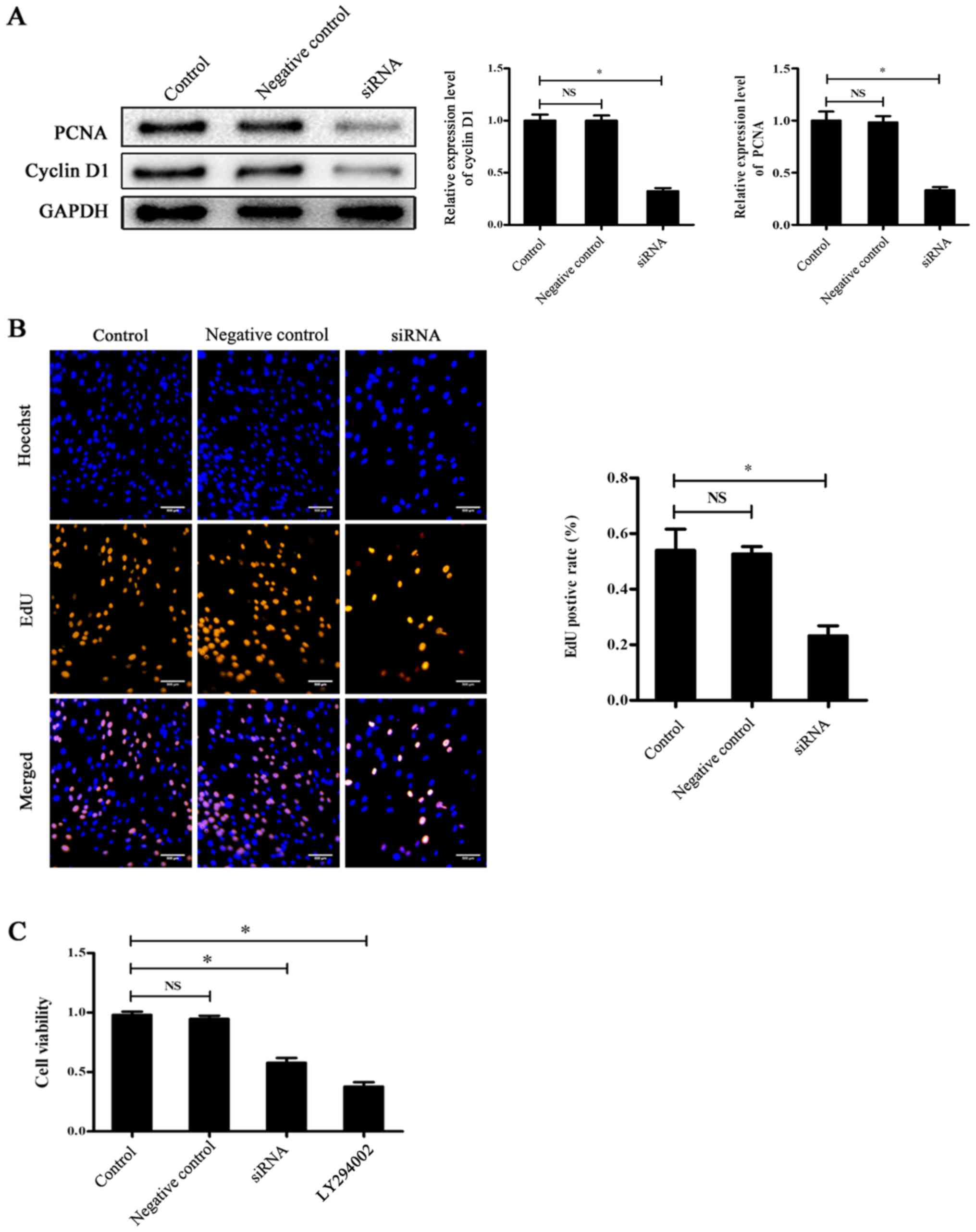
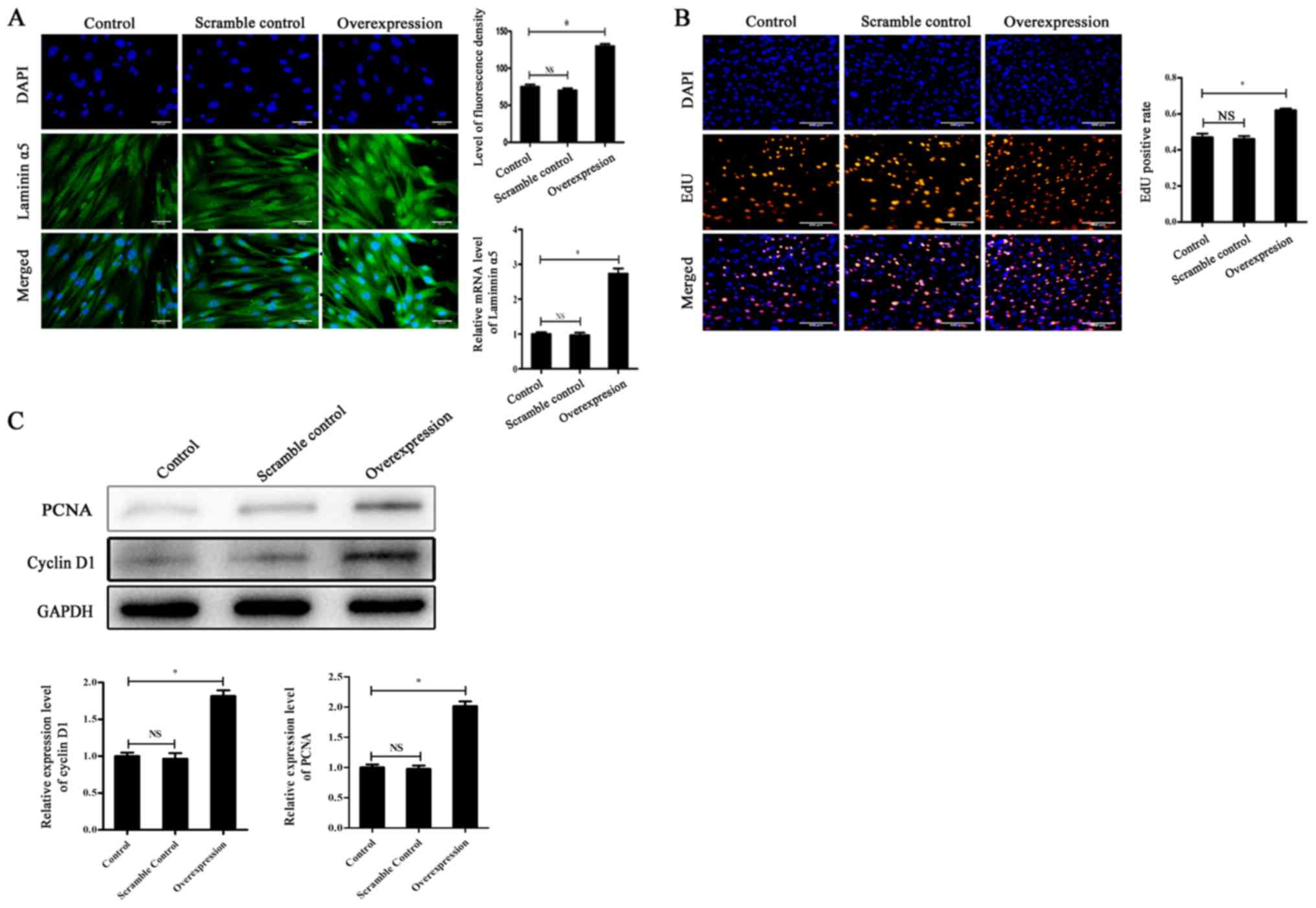
|
1
|
Burton CV, Kirkaldy-Willis WH, Yong-Hing K
and Heithoff KB: Causes of failure of surgery on the lumbar spine.
Clin Orthop Relat Res. 157:191–199. 1981.
|
|
2
|
Songer MN, Rauschning W, Carson EW and
Pandit SM: Analysis of peridural scar formation and its prevention
after lumbar laminotomy and discectomy in dogs. Spine (Phila Pa
1976). 20:571–580. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sen O, Kizilkilic O, Aydin MV, Yalcin O,
Erdogan B, Cekinmez M, Caner H and Altinors N: The role of
closed-suction drainage in preventing epidural fibrosis and its
correlation with a new grading system of epidural fibrosis based on
MRI. Eur Spine J. 14:409–414. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mirzai H, Eminoglu M and Orguc S: Are
drains useful for lumbar disc surgery? A prospective, randomized
clinical study. J Spinal Disord Tech. 19:171–177. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cekinmez M, Erdogan B, Tufan K, Sarica FB,
Ozen O and Caner H: Is topical tissue plasminogen activator
application effective on prevention of post-laminectomy epidural
fibrosis? An experimental study. Neurol Res. 31:322–326. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sun Y, Zhao S, Li X, Yan L, Wang J, Wang
D, Chen H, Dai J and He J: Local application of rapamycin reduces
epidural fibrosis after laminectomy via inhibiting fibroblast
proliferation and prompting apoptosis. J Orthop Surg Res.
11:582016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen H, Yan L, Wang J, Sun Y, Li X, Zhao
S, Wang D, Zhu G and Liang Y: Methotrexate prevents epidural
fibrosis through endoplasmic reticulum stress signalling pathway.
Eur J Pharmacol. 796:131–138. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dai J, Li X, Yan L, Chen H, He J, Wang S,
Wang J and Sun Y: The effect of suramin on inhibiting fibroblast
proliferation and preventing epidural fibrosis after laminectomy in
rats. J Orthop Surg Res. 11:1082016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Atsuta I, Yamaza T, Yoshinari M, Goto T,
Kido MA, Kagiya T, Mino S, Shimono M and Tanaka T: Ultrastructural
localization of laminin-5 (gamma2 chain) in the rat peri-implant
oral mucosa around a titanium-dental implant by immuno-electron
microscopy. Biomaterials. 26:6280–6287. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Durbeej M: Laminins. Cell Tissue Res.
339:259–268. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Burgeson RE, Chiquet M, Deutzmann R,
Ekblom P, Engel J, Kleinman H, Martin GR, Meneguzzi G, Paulsson M,
Sanes J, et al: A new nomenclature for the laminins. Matrix Biol.
14:209–211. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Domogatskaya A, Rodin S and Tryggvason K:
Functional diversity of laminins. Annu Rev Cell Dev Biol.
28:523–553. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Petz M, Them N, Huber H, Beug H and
Mikulits W: La enhances IRES-mediated translation of laminin B1
during malignant epithelial to mesenchymal transition. Nucleic
Acids Res. 40:290–302. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Aumailley M and Rousselle P: Laminins of
the dermo-epidermal junction. Matrix Biol. 18:19–28. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Petz M, Them NC, Huber H and Mikulits W:
PDGF enhances IRES-mediated translation of Laminin B1 by
cytoplasmic accumulation of La during epithelial to mesenchymal
transition. Nucleic Acids Res. 40:9738–9749. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sun Y, Wang TL, Toh WS and Pei M: The role
of laminins in cartilaginous tissues: From development to
regeneration. Eur Cell Mater. 34:40–54. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Savino W, Mendes-da-Cruz DA, Golbert DC,
Riederer I and Cotta-de-Almeida V: Laminin-mediated interactions in
thymocyte migration and development. Front Immunol. 6:5792015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Laperle A, Hsiao C, Lampe M, Mortier J,
Saha K, Palecek SP and Masters KS: α-5 Laminin synthesized by human
pluripotent stem cells promotes self-renewal. Stem Cell Rep.
5:195–206. 2015. View Article : Google Scholar
|
|
19
|
Miner JH: Laminins and their roles in
mammals. Microsc Res Tech. 71:349–356. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gao J, DeRouen MC, Chen CH, Nguyen M,
Nguyen NT, Ido H, Harada K, Sekiguchi K, Morgan BA, Miner JH, et
al: Laminin-511 is an epithelial message promoting dermal papilla
development and function during early hair morphogenesis. Genes
Dev. 22:2111–2124. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sampaolo S, Napolitano F, Tirozzi A,
Reccia MG, Lombardi L, Farina O, Barra A, Cirillo F, Melone MAB,
Gianfrancesco F, et al: Identification of the first dominant
mutation of LAMA5 gene causing a complex multisystem syndrome due
to dysfunction of the extracellular matrix. J Med Genet.
54:710–720. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Napolitano F, Di Iorio V, Di Iorio G,
Melone MAB, Gianfrancesco F, Simonelli F, Esposito T, Testa F and
Sampaolo S: Early posterior vitreous detachment is associated with
LAMA5 dominant mutation. Ophthalmic Genet. 40:39–42. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Engelman JA, Luo J and Cantley LC: The
evolution of phosphatidylinositol 3-kinases as regulators of growth
and metabolism. Nat Rev Genet. 7:606–619. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lam L, Hu X, Aktary Z, Andrews DW and
Pasdar M: Tamoxifen and ICI 182,780 increase Bcl-2 levels and
inhibit growth of breast carcinoma cells by modulating PI3K/AKT,
ERK and IGF-1R pathways independent of ERalpha. Breast Cancer Res
Treat. 118:605–621. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang S, Li X, Yan L, Nie Q, Dai J, Chen H,
Wang J and Sun Y: Tamoxifen inhibits fibroblast proliferation and
prevents epidural fibrosis by regulating the AKT pathway in rats.
Biochem Biophys Res Commun. 497:937–942. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sun Y, Wang LX, Wang L, Sun SX, Cao XJ,
Wang P and Feng L: A comparison of the effectiveness of mitomycin C
and 5-fluorouracil in the prevention of peridural adhesion after
laminectomy. J Neurosurg Spine. 7:423–428. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dai J, Sun Y, Yan L, Wang J, Li X and He
J: Upregulation of NOXA by 10-Hydroxycamptothecin plays a key role
in inducing fibroblasts apoptosis and reducing epidural fibrosis.
PeerJ. 5:e28582017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jiao R, Chen H, Wan Q, Zhang X, Dai J, Li
X, Yan L and Sun Y: Apigenin inhibits fibroblast proliferation and
reduces epidural fibrosis by regulating Wnt3a/β-catenin signaling
pathway. J Orthop Surg Res. 14:2582019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li X, Chen H, Wang S, Dai J, Yan L, Wang J
and Sun Y: Tacrolimus induces fibroblasts apoptosis and reduces
epidural fibrosis by regulating miR-429 and its target of RhoE.
Biochem Biophys Res Commun. 490:1197–1204. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li X, Wang S, Dai J, Yan L, Zhao S, Wang J
and Sun Y: Homoharringtonine prevents surgery-induced epidural
fibrosis through endoplasmic reticulum stress signaling pathway.
Eur J Pharmacol. 815:437–445. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sandoval MA and Hernandez-Vaquero D:
Preventing peridural fibrosis with nonsteroidal anti-inflammatory
drugs. Eur Spine J. 17:451–455. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pozzi A, Yurchenco PD and Iozzo RV: The
nature and biology of basement membranes. Matrix Biol. 57-58:1–11.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hynes RO: The evolution of metazoan
extracellular matrix. J Cell Biol. 196:671–679. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Spenle C, Simon-Assmann P, Orend G and
Miner JH: Laminin α5 guides tissue patterning and organogenesis.
Cell Adh Migr. 7:90–100. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Fukumoto S, Miner JH, Ida H, Fukumoto E,
Yuasa K, Miyazaki H, Hoffman MP and Yamada Y: Laminin alpha5 is
required for dental epithelium growth and polarity and the
development of tooth bud and shape. J Biol Chem. 281:5008–5016.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sharma N, Nanta R, Sharma J, Gunewardena
S, Singh KP, Shankar S and Srivastava RK: PI3K/AKT/mTOR and sonic
hedgehog pathways cooperate together to inhibit human pancreatic
cancer stem cell characteristics and tumor growth. Oncotarget.
6:32039–32060. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Su JS, Woods SM and Ronen SM: Metabolic
consequences of treatment with AKT inhibitor perifosine in breast
cancer cells. NMR Biomed. 25:379–388. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Bundschuh CV, Modic MT, Ross JS, Masaryk
TJ and Bohlman H: Epidural fibrosis and recurrent disk herniation
in the lumbar spine: MR imaging assessment. AJR Am J Roentgenol.
150:923–932. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ritie L, Spenle C, Lacroute J,
Bolcato-Bellemin AL, Lefebvre O, Bole-Feysot C, Jost B, Klein A,
Arnold C, Kedinger M, et al: Abnormal Wnt and PI3Kinase signaling
in the malformed intestine of lama5 deficient mice. PLoS One.
7:e377102012. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xia H, Nho RS, Kahm J, Kleidon J and Henke
CA: Focal adhesion kinase is upstream of phosphatidylinositol
3-kinase/Akt in regulating fibroblast survival in response to
contraction of type I collagen matrices via a beta 1 integrin
viability signaling pathway. J Biol Chem. 279:33024–33034. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Santos AR, Corredor RG, Obeso BA,
Trakhtenberg EF, Wang Y, Ponmattam J, Dvoriantchikova G, Ivanov D,
Shestopalov VI, Goldberg JL, et al: β1 integrin-focal adhesion
kinase (FAK) signaling modulates retinal ganglion cell (RGC)
survival. PLoS One. 7:e483322012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wegner J, Loser K, Apsite G, Nischt R,
Eckes B, Krieg T, Werner S and Sorokin L: Laminin α5 in the
keratinocyte basement membrane is required for epidermal-dermal
intercommunication. Matrix Biol. 56:24–41. 2016. View Article : Google Scholar : PubMed/NCBI
|