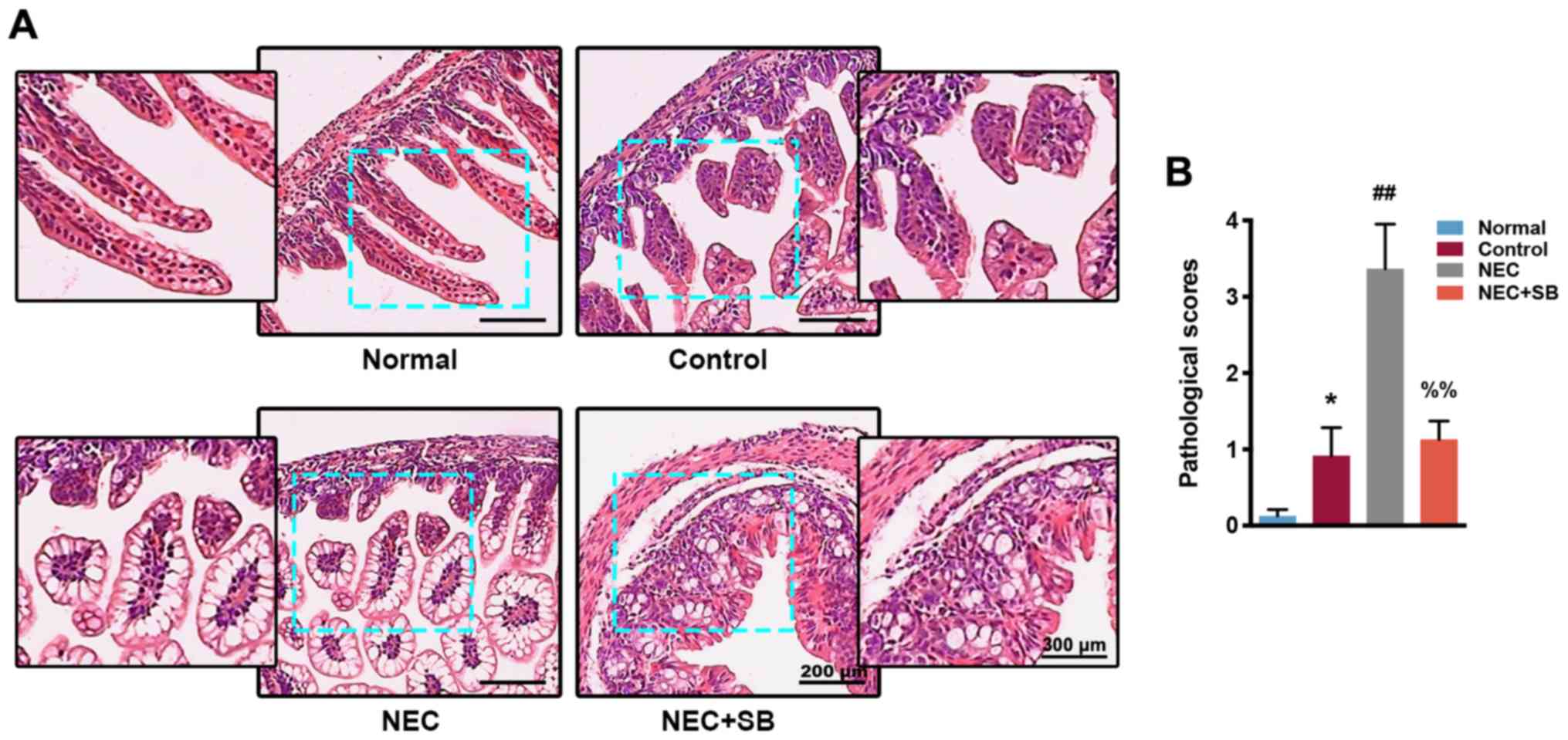
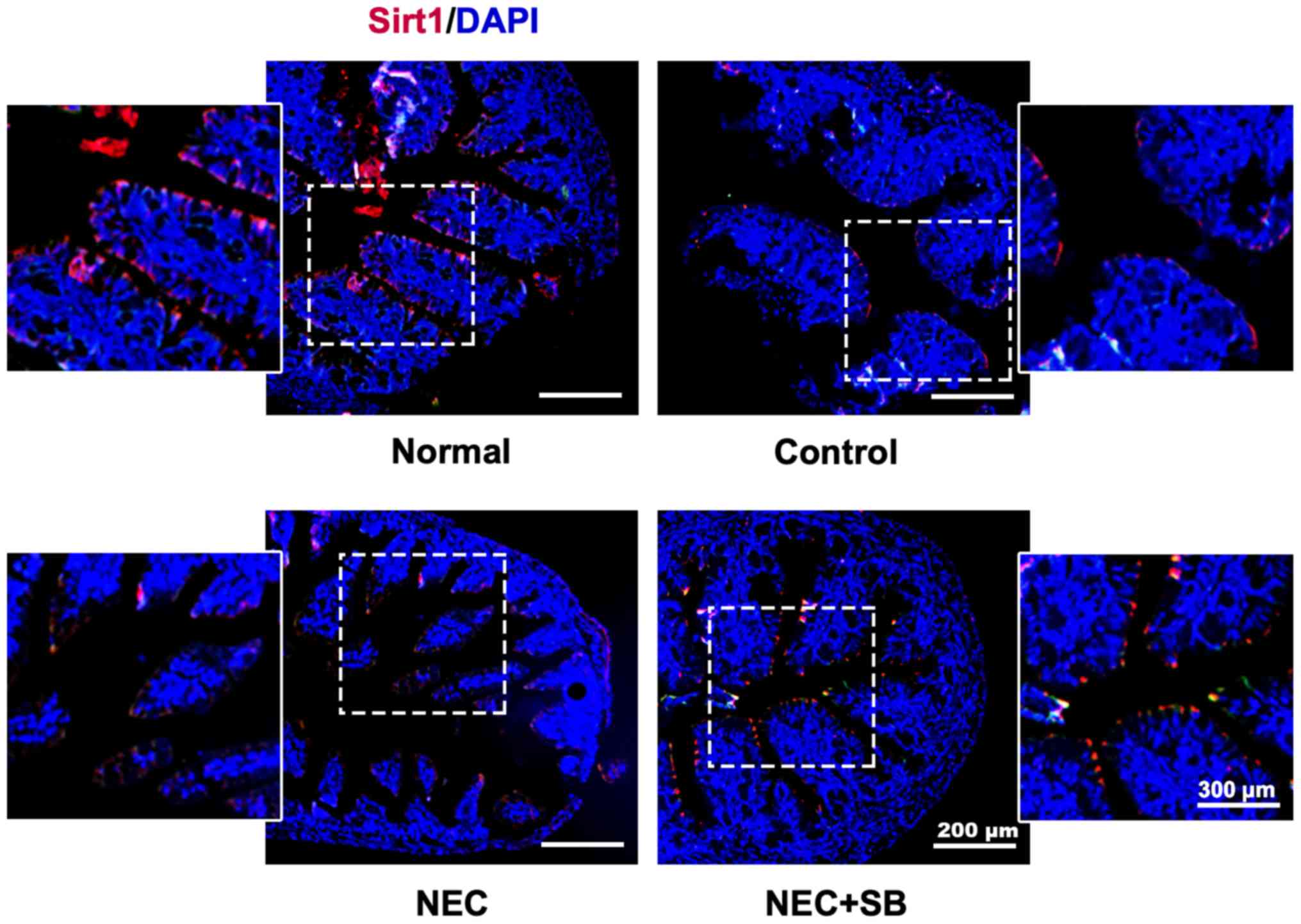
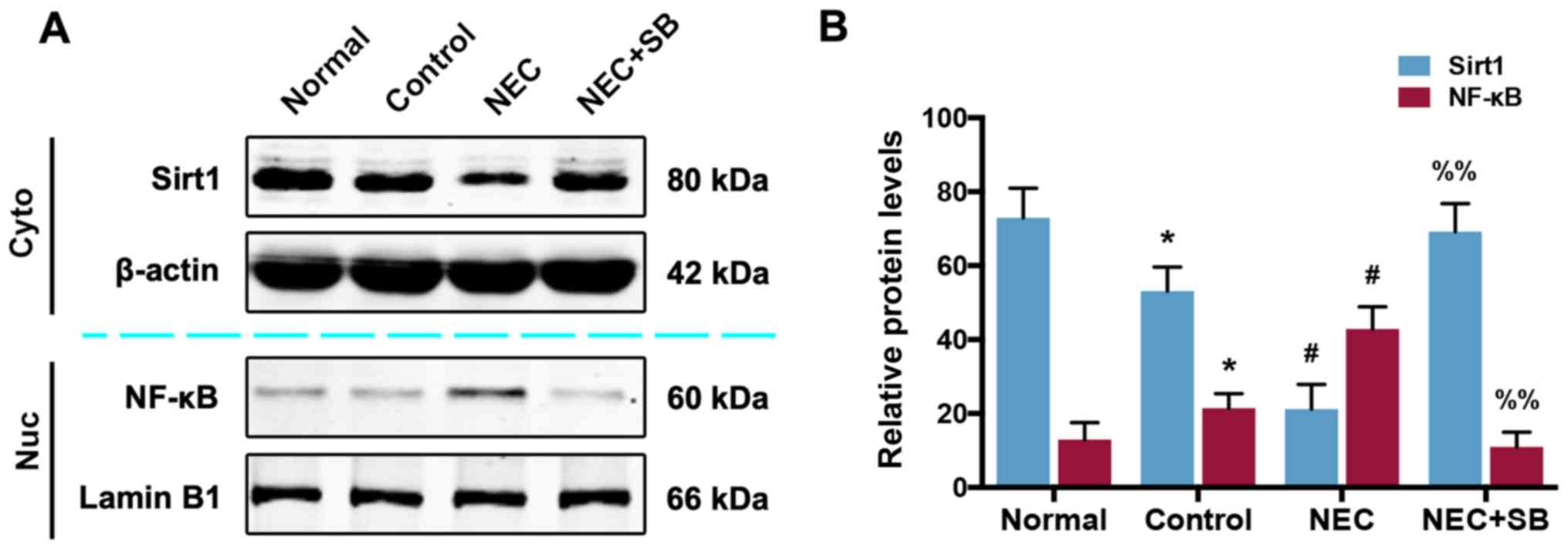
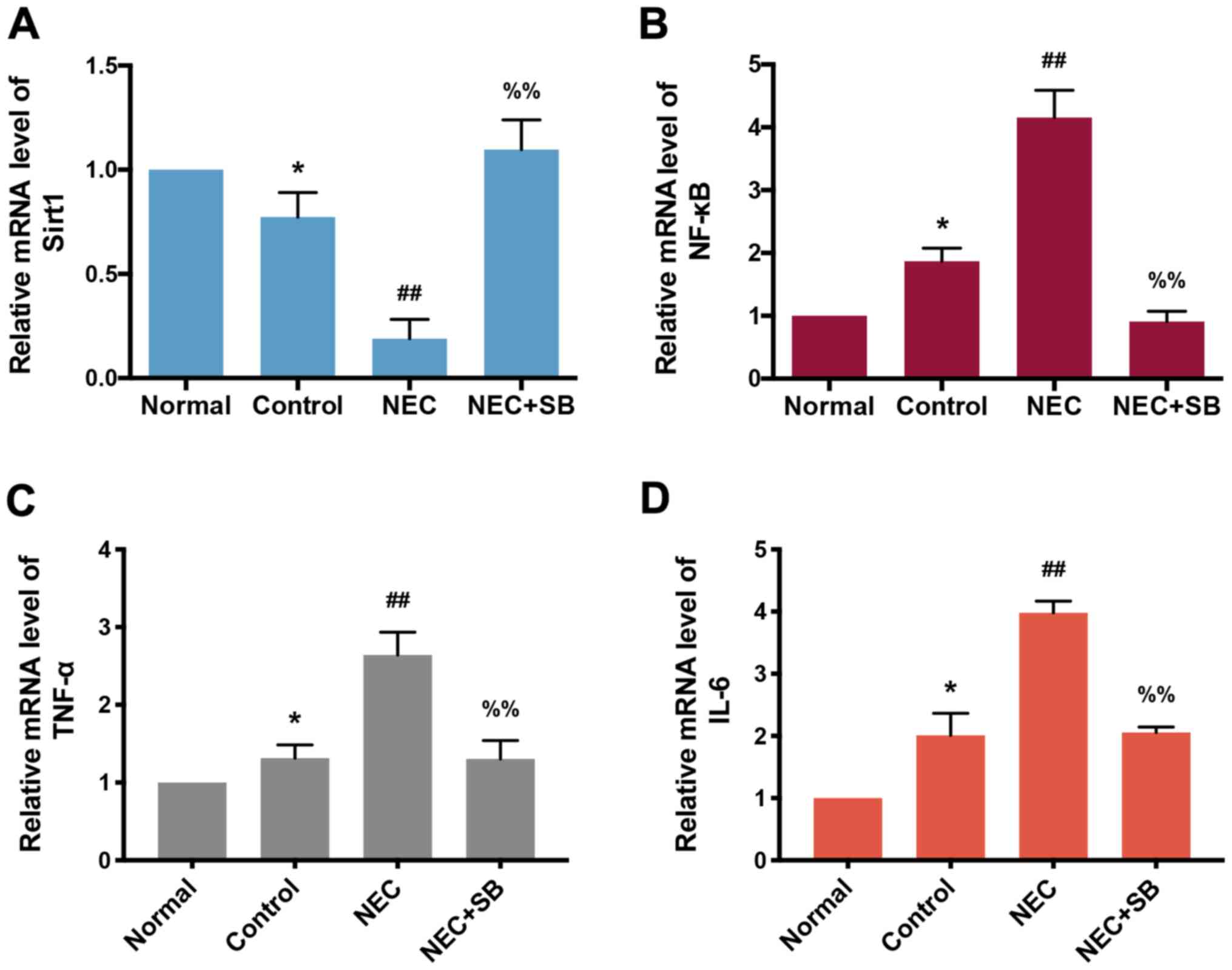
|
1
|
Neu J and Walker WA: Necrotizing
enterocolitis. N Engl J Med. 364:255–264. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kafetzis DA, Skevaki C and Costalos C:
Neonatal necrotizing enterocolitis: An overview. Curr Opin Infect
Dis. 16:349–355. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bell MJ, Ternberg JL, Feigin RD, Keating
JP, Marshall R, Barton L and Brotherton T: Neonatal necrotizing
enterocolitis. Therapeutic decisions based upon clinical staging.
Ann Surg. 187:1–7. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Guthrie SO, Gordon PV, Thomas V, Thorp JA,
Peabody J and Clark RH: Necrotizing enterocolitis among neonates in
the United States. J Perinatol. 23:278–285. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Holman RC, Stoll BJ, Clarke MJ and Glass
RI: The epidemiology of necrotizing enterocolitis infant mortality
in the United States. Am J Public Health. 87:2026–2031. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Holman RC, Stoll BJ, Curns AT, Yorita KL,
Steiner CA and Schonberger LB: Necrotising enterocolitis
hospitalisations among neonates in the United States. Paediatr
Perinat Epidemiol. 20:498–506. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Horbar JD, Carpenter JH, Badger GJ, Kenny
MJ, Soll RF, Morrow KA and Buzas JS: Mortality and neonatal
morbidity among infants 501 to 1500 grams from 2000 to 2009.
Pediatrics. 129:1019–1026. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Aceti A, Gori D, Barone G, Callegari ML,
Di Mauro A, Fantini MP, Indrio F, Maggio L, Meneghin F, Morelli L,
et al: Probiotics for prevention of necrotizing enterocolitis in
preterm infants: Systematic review and meta-analysis. Ital J
Pediatr. 41:892015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hatoum R, Labrie S and Fliss I:
Antimicrobial and probiotic properties of yeasts: From fundamental
to novel applications. Front Microbiol. 3:4212012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Generoso SV, Viana M, Santos R, Martins
FS, Machado JA, Arantes RM, Nicoli JR, Correia MI and Cardoso VN:
Saccharomyces cerevisiae strain UFMG 905 protects against bacterial
translocation, preserves gut barrier integrity and stimulates the
immune system in a murine intestinal obstruction model. Arch
Microbiol. 192:477–484. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Buts JP and De Keyser N: Effects of
saccharomyces boulardii on intestinal mucosa. Dig Dis Sci.
51:1485–1492. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Serce O, Benzer D, Gursoy T, Karatekin G
and Ovali F: Efficacy of saccharomyces boulardii on necrotizing
enterocolitis or sepsis in very low birth weight infants: A
randomised controlled trial. Early Hum Dev. 89:1033–1036. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Demirel G, Erdeve O, Celik IH and Dilmen
U: Saccharomyces boulardii for prevention of necrotizing
enterocolitis in preterm infants: A randomized, controlled study.
Acta Paediatr. 102:e560–e565. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Akisu M, Baka M, Yalaz M, Huseyinov A and
Kultursay N: Supplementation with saccharomyces boulardii
ameliorates hypoxia/reoxygenation-induced necrotizing enterocolitis
in young mice. Eur J Pediatr Surg. 13:319–323. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Haigis MC and Sinclair DA: Mammalian
sirtuins: Biological insights and disease relevance. Annu Rev
Pathol. 5:253–295. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rajendrasozhan S, Yang SR, Kinnula VL and
Rahman I: SIRT1, an antiinflammatory and antiaging protein, is
decreased in lungs of patients with chronic obstructive pulmonary
disease. Am J Respir Crit Care Med. 177:861–870. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Deng Z, Jin J, Wang Z, Wang Y, Gao Q and
Zhao J: The metal nanoparticle-induced inflammatory response is
regulated by SIRT1 through NF-kappaB deacetylation in aseptic
loosening. Int J Nanomedicine. 12:3617–3636. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wen X, Chen X, Liang X, Zhao H, Li Y, Sun
X and Lu J: The small molecule NSM00191 specifically represses the
TNF-α/NF-кB axis in foot and ankle rheumatoid arthritis. Int J Biol
Sci. 14:1732–1744. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wellman AS, Metukuri MR, Kazgan N, Xu X,
Xu Q, Ren NSX, Czopik A, Shanahan MT, Kang A, Chen W, et al:
Intestinal epithelial sirtuin 1 regulates intestinal inflammation
during aging in mice by altering the intestinal microbiota.
Gastroenterology. 153:772–786. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kazgan N, Metukuri MR, Purushotham A, Lu
J, Rao A, Lee S, Pratt-Hyatt M, Lickteig A, Csanaky IL, Zhao Y, et
al: Intestine-Specific deletion of SIRT1 in mice impairs
DCoH2-HNF-1α-FXR signaling and alters systemic bile acid
homeostasis. Gastroenterology. 146:1006–1016. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Barna J, Renner E, Arszovszki A, Cservenák
M, Kovács Z, Palkovits M and Dobolyi A: Suckling induced activation
pattern in the brain of rat pups. Nutr Neurosci. 21:317–327. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Premkumar MH, Sule G, Nagamani SC,
Chakkalakal S, Nordin A, Jain M, Ruan MZ, Bertin T, Dawson B, Zhang
J, et al: Argininosuccinate lyase in enterocytes protects from
development of necrotizing enterocolitis. Am J Physiol Gastrointest
Liver Physiol. 307:G347–G354. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Radulescu A, Zhang HY, Yu X, Olson JK,
Darbyshire AK, Chen Y and Besner GE: Heparin-Binding epidermal
growth factor-like growth factor overexpression in transgenic mice
increases resistance to necrotizing enterocolitis. J Pediatr Surg.
45:1933–1939. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nadler EP, Dickinson E, Knisely A, Zhang
XR, Boyle P, Beer-Stolz D, Watkins SC and Ford HR: Expression of
inducible nitric oxide synthase and interleukin-12 in experimental
necrotizing enterocolitis. J Surg Res. 92:71–77. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yee WH, Soraisham AS, Shah VS, Aziz K,
Yoon W and Lee SK; Canadian Neonatal Network, : Incidence and
timing of presentation of necrotizing enterocolitis in preterm
infants. Pediatrics. 129:e298–e304. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Niemarkt HJ, De Meij TG, van Ganzewinkel
CJ, de Boer NKH, Andriessen P, Hütten MC and Kramer BW: Necrotizing
enterocolitis, gut microbiota, and brain development: Role of the
brain-gut axis. Neonatology. 115:423–431. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hagen PC and Skelley JW: Efficacy of
bifidobacterium species in prevention of necrotizing enterocolitis
in very-low birth weight infants. A systematic review. J Pediatr
Pharmacol Ther. 24:10–15. 2019.PubMed/NCBI
|
|
29
|
Gephart SM, McGrath JM, Effken JA and
Halpern MD: Necrotizing enterocolitis risk: State of the science.
Adv Neonatal Care. 12:77–87. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Czerucka D, Piche T and Rampal P: Review
article: Yeast as probiotics-saccharomyces boulardii. Aliment
Pharmacol Ther. 26:767–778. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rodrigues AC, Cara DC, Fretez SH, Cunha
FQ, Vieira EC, Nicoli JR and Vieira LQ: Saccharomyces boulardii
stimulates sIgA production and the phagocytic system of gnotobiotic
mice. J Appl Microbiol. 89:404–414. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Qamar A, Aboudola S, Warny M, Michetti P,
Pothoulakis C, LaMont JT and Kelly CP: Saccharomyces boulardii
stimulates intestinal immunoglobulin A immune response to
clostridium difficile toxin A in mice. Infect Immun. 69:2762–2765.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Stier H and Bischoff SC: Influence of
saccharomyces boulardii CNCM I-745on the gut-associated immune
system. Clin Exp Gastroenterol. 9:269–279. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kelesidis T and Pothoulakis C: Efficacy
and safety of the probiotic saccharomyces boulardii for the
prevention and therapy of gastrointestinal disorders. Therap Adv
Gastroenterol. 5:111–125. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Haigis MC and Guarente LP: Mammalian
sirtuins-emerging roles in physiology, aging, and calorie
restriction. Genes Dev. 20:2913–2921. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Liu TF, Vachharajani VT, Yoza BK and
McCall CE: NAD+-dependent sirtuin 1 and 6 proteins coordinate a
switch from glucose to fatty acid oxidation during the acute
inflammatory response. J Biol Chem. 287:25758–25769. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yoshizaki T, Milne JC, Imamura T, Schenk
S, Sonoda N, Babendure JL, Lu JC, Smith JJ, Jirousek MR and Olefsky
JM: SIRT1 exerts anti-inflammatory effects and improves insulin
sensitivity in adipocytes. Mol Cell Biol. 29:1363–1374. 2009.
View Article : Google Scholar : PubMed/NCBI
|