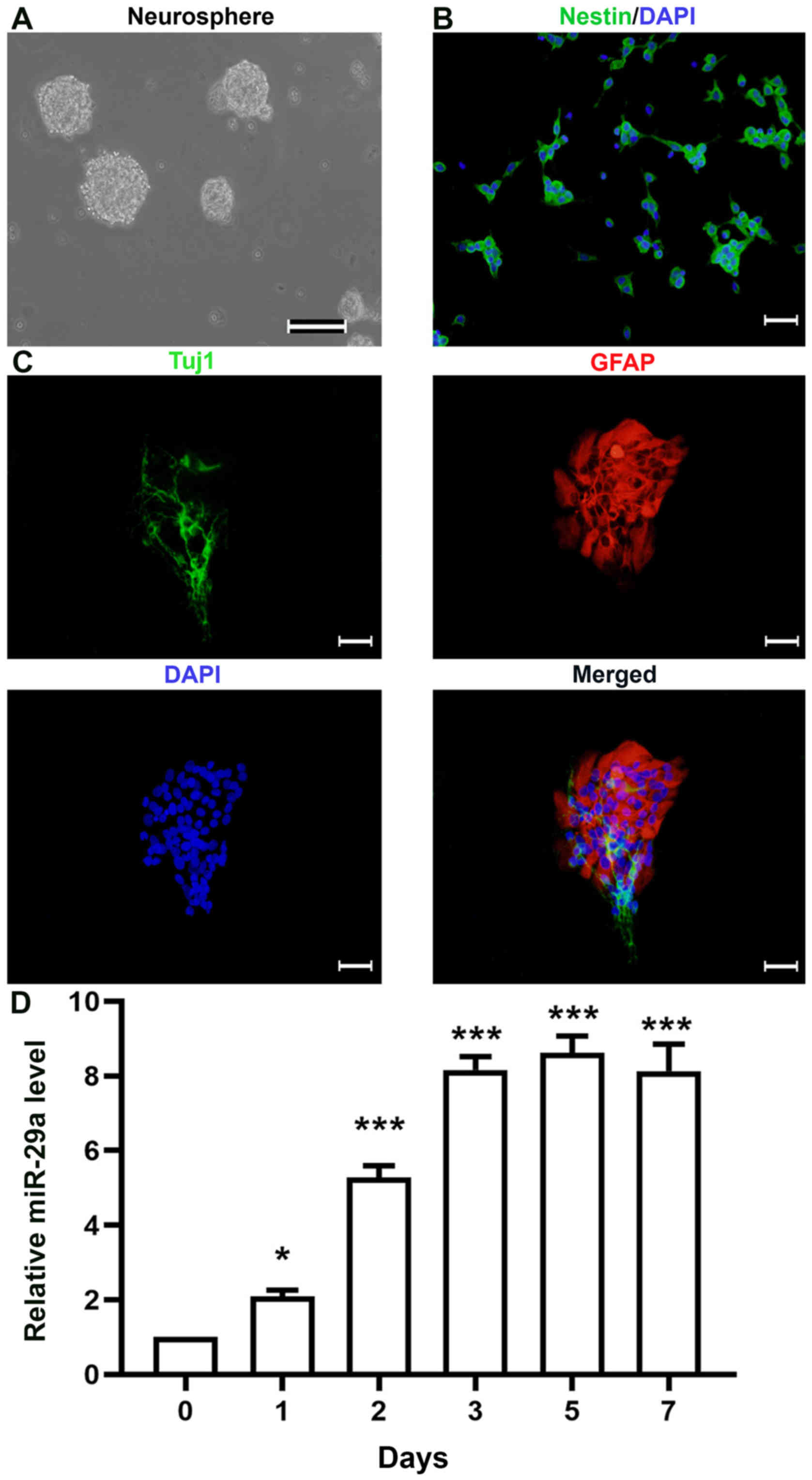
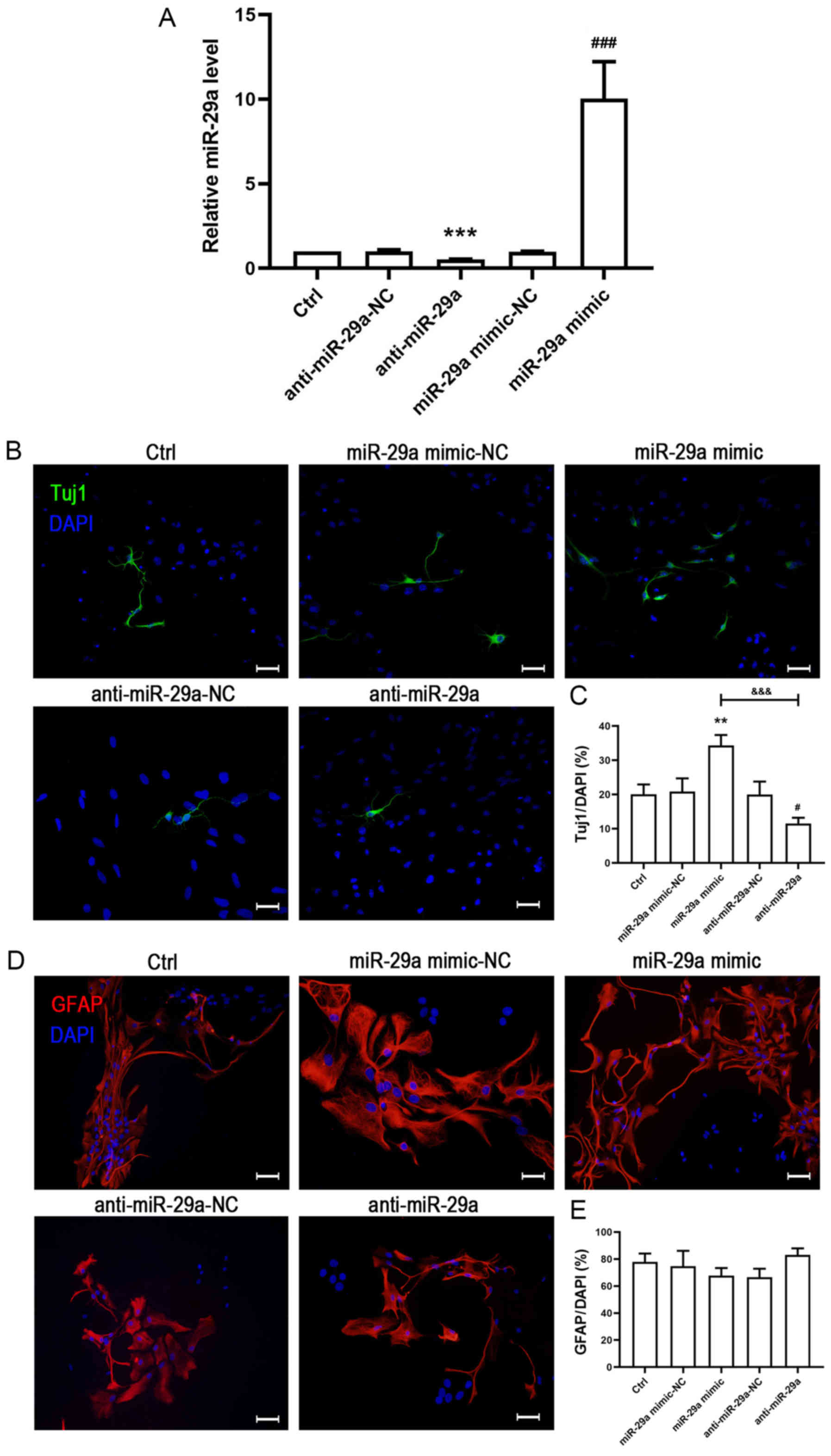
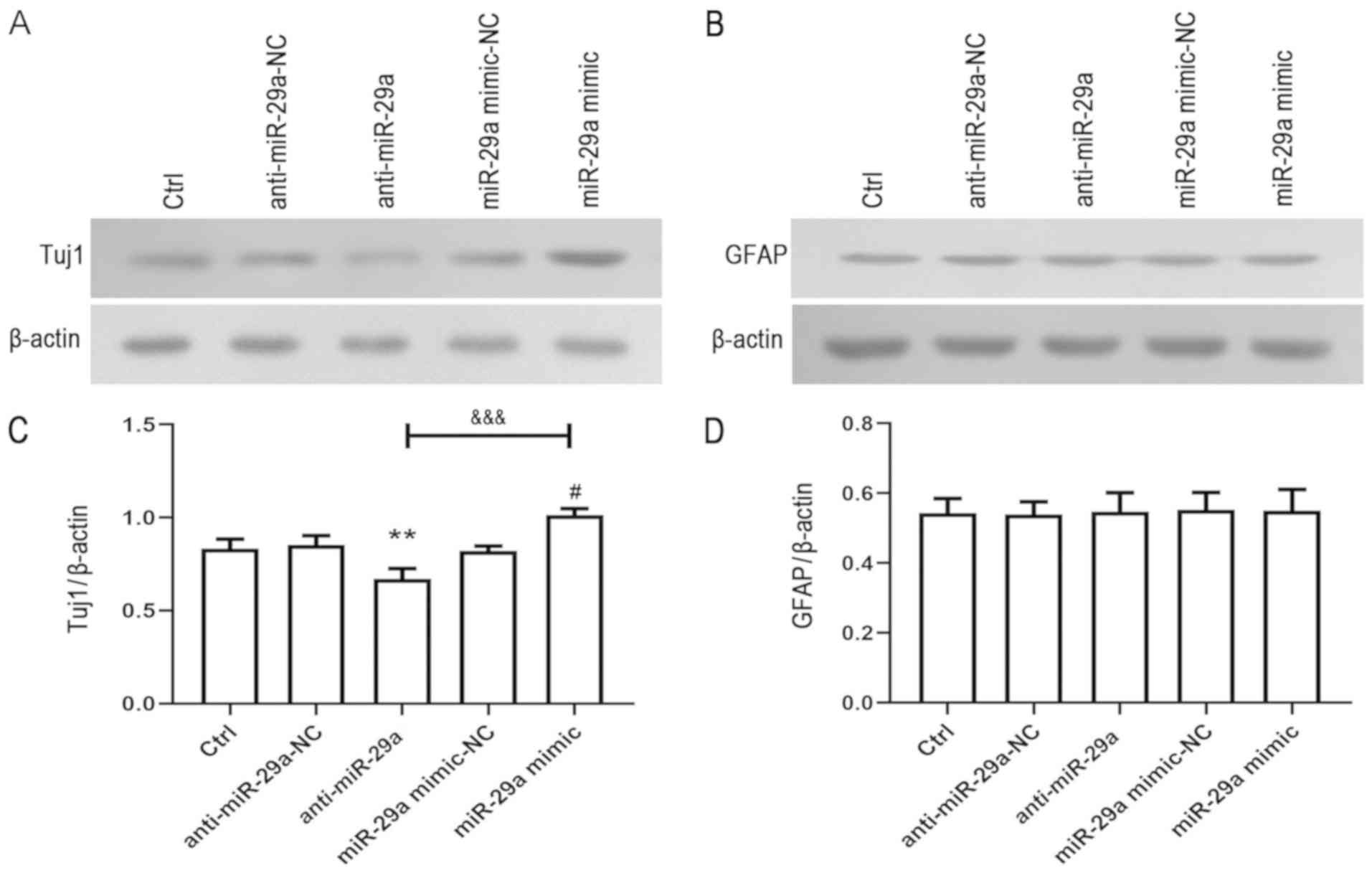
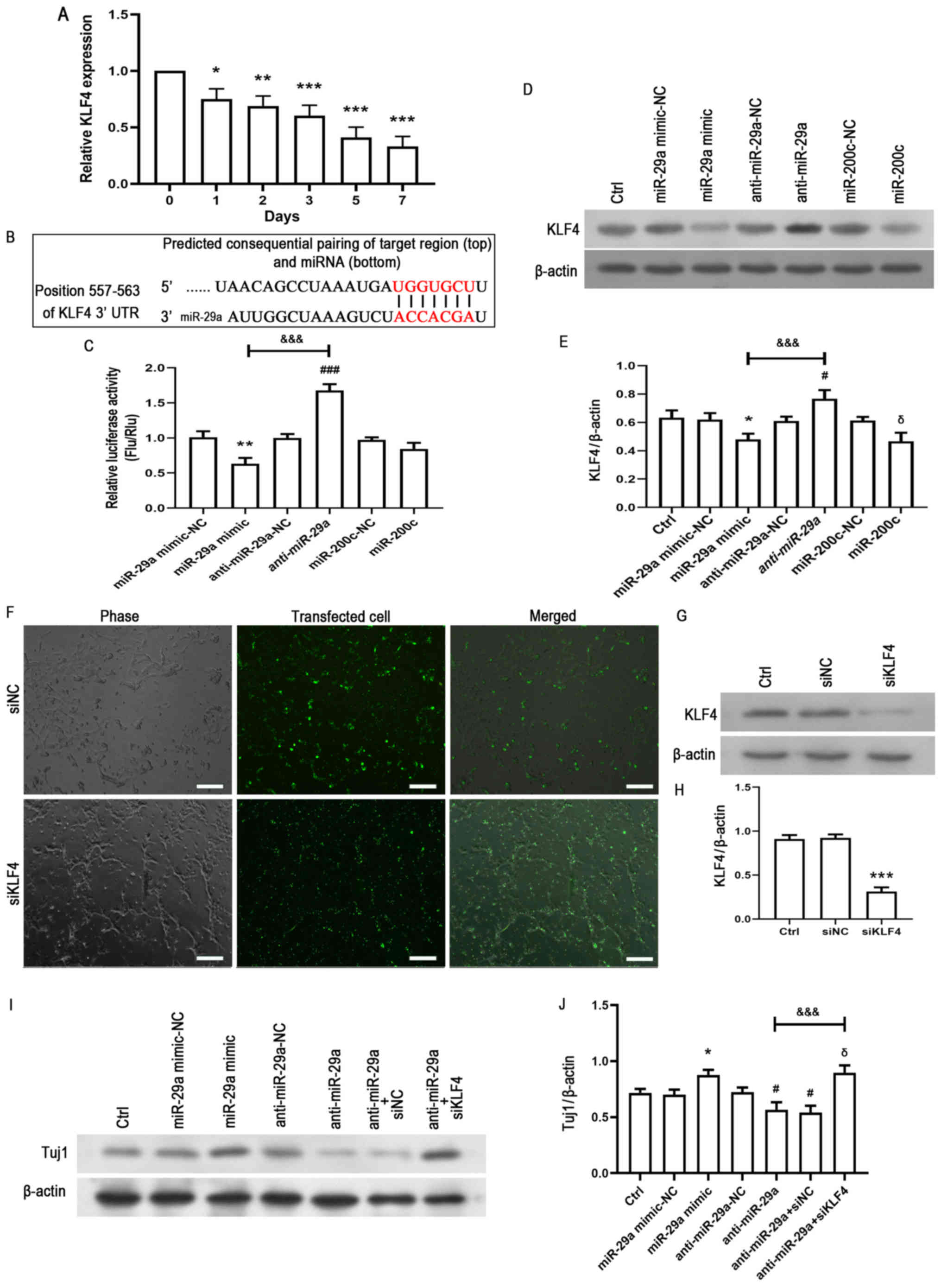
|
1
|
Ming GL and Song H: Adult neurogenesis in
the mammalian brain: Significant answers and significant questions.
Neuron. 70:687–702. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhao C, Deng W and Gage FH: Mechanisms and
functional implications of adult neurogenesis. Cell. 132:645–660.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sierra A, Encinas JM and Maletic-Savatic
M: Adult human neurogenesis: From microscopy to magnetic resonance
imaging. Front Neurosci. 5:472011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Abati E, Bresolin N, Comi GP and Corti S:
Preconditioning and cellular engineering to increase the survival
of transplanted neural stem cells for motor neuron disease therapy.
Mol Neurobiol. 56:3356–3367. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bellenchi GC, Volpicelli F, Piscopo V,
Perrone-Capano C and di Porzio U: Adult neural stem cells: An
endogenous tool to repair brain injury? J Neurochem. 124:159–167.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Encinas JM and Fitzsimons CP: Gene
regulation in adult neural stem cells. Current challenges and
possible applications. Adv Drug Deliv Rev. 120:118–132. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Imayoshi I and Kageyama R: The role of
Notch signaling in adult neurogenesis. Mol Neurobiol. 44:7–12.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ming GL and Song H: Adult neurogenesis in
the mammalian central nervous system. Annu Rev Neurosci.
28:223–250. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Meza-Sosa KF, Pedraza-Alva G and
Pérez-Martínez L: microRNAs: Key triggers of neuronal cell fate.
Front Cell Neurosci. 8:1752014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Alizadeh M, Safarzadeh A, Beyranvand F,
Ahmadpour F, Hajiasgharzadeh K, Baghbanzadeh A and Baradaran B: The
potential role of miR-29 in health and cancer diagnosis, prognosis,
and therapy. J Cell Physiol. 234:19280–19297. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rajendran G, Dutta D, Hong J, Paul A, Saha
B, Mahato B, Ray S, Home P, Ganguly A, Weiss ML and Paul S:
Inhibition of protein kinase C signaling maintains rat embryonic
stem cell pluripotency. J Biol Chem. 288:24351–24362. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gao Y, Qiao H, Lu Z and Hou Y: miR29
promotes the proliferation of cultured rat neural stem/progenitor
cells via the PTEN/AKT signaling pathway. Mol Med Rep.
20:2111–2118. 2019.PubMed/NCBI
|
|
13
|
Zhang Z, Zheng X and Liu Y, Luan Y, Wang
L, Zhao L, Zhang J, Tian Y, Lu H, Chen X and Liu Y: Activation of
metabotropic glutamate receptor 4 regulates proliferation and
neural differentiation in neural stem/progenitor cells of the rat
subventricular zone and increases phosphatase and tensin homolog
protein expression. J Neurochem. Feb 12–2020.doi:
10.1111/jnc.14984. Online ahead of print. View Article : Google Scholar
|
|
14
|
Chen X, Tian Y, Yao L, Zhang J and Liu Y:
Hypoxia stimulates proliferation of rat neural stem cells with
influence on the expression of cyclin D1 and c-Jun N-terminal
protein kinase signaling pathway in vitro. Neuroscience.
165:705–714. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jia L, Chopp M, Wang L, Lu X, Zhang Y,
Szalad A and Zhang ZG: MiR-34a regulates axonal growth of dorsal
root ganglia neurons by targeting FOXP2 and VAT1 in postnatal and
adult mouse. Mol Neurobiol. 55:9089–9099. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang Z, Hu F, Liu Y, Ma B, Chen X, Zhu K,
Shi Y, Wei T, Xing Y, Gao Y, et al: Activation of type 5
metabotropic glutamate receptor promotes the proliferation of rat
retinal progenitor cell via activation of the PI-3-K and MAPK
signaling pathways. Neuroscience. 322:138–151. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Suzuki S, Namiki J, Shibata S, Mastuzaki Y
and Okano H: The neural stem/progenitor cell marker nestin is
expressed in proliferative endothelial cells, but not in mature
vasculature. J Histochem Cytochem. 58:721–730. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Takahashi K and Yamanaka S: A decade of
transcription factor-mediated reprogramming to pluripotency. Nat
Rev Mol Cell Biol. 17:183–193. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Black AR, Black JD and Azizkhan-Clifford
J: Sp1 and krüppel-like factor family of transcription factors in
cell growth regulation and cancer. J Cell Physiol. 188:143–160.
2001. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wellner U, Schubert J, Burk UC,
Schmalhofer O, Zhu F, Sonntag A, Waldvogel B, Vannier C, Darling D,
zur Hausen A, et al: The EMT-activator ZEB1 promotes tumorigenicity
by repressing stemness-inhibiting microRNAs. Nat Cell Biol.
11:1487–1495. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
van Rooij E and Olson EN: MicroRNA
therapeutics for cardiovascular disease: Opportunities and
obstacles. Nat Rev Drug Discov. 11:860–872. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jin M, Wu Y, Wang J, Ye W, Wang L, Yin P,
Liu W, Pan C and Hua X: MicroRNA-29 facilitates transplantation of
bone marrow-derived mesenchymal stem cells to alleviate pelvic
floor dysfunction by repressing elastin. Stem Cell Res Ther.
7:1672016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
O'Connell RM: Endogenous miR-29a regulates
HSC function in mammals. Blood. 125:2180–2181. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Barkho BZ and Zhao X: Adult neural stem
cells: Response to stroke injury and potential for therapeutic
applications. Curr Stem Cell Res Ther. 6:327–338. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Müller M, Jäkel L, Bruinsma IB, Claassen
JA, Kuiperij HB and Verbeek MM: MicroRNA-29a is a candidate
biomarker for Alzheimer's disease in cell-free cerebrospinal fluid.
Mol Neurobiol. 53:2894–2899. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bai X, Tang Y, Yu M, Wu L, Liu F, Ni J,
Wang Z, Wang J, Fei J, Wang W, et al: Downregulation of blood serum
microRNA 29 family in patients with Parkinson's disease. Sci Rep.
7:54112017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hébert SS, Horré K, Nicolaï L,
Papadopoulou AS, Mandemakers W, Silahtaroglu AN, Kauppinen S,
Delacourte A and De Strooper B: Loss of microRNA cluster
miR-29a/b-1 in sporadic Alzheimer's disease correlates with
increased BACE1/beta-secretase expression. Proc Natl Acad Sci USA.
105:6415–6420. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pandi G, Nakka VP, Dharap A, Roopra A and
Vemuganti R: MicroRNA miR-29c down-regulation leading to
de-repression of its target DNA methyltransferase 3a promotes
ischemic brain damage. PLoS One. 8:e580392013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rowland BD and Peeper DS: KLF4, p21 and
context-dependent opposing forces in cancer. Nat Rev Cancer.
6:11–23. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ghaleb AM and Yang VW: Krüppel-like factor
4 (KLF4): What we currently know. Gene. 611:27–37. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Simmen RCM, Pabona JMP, Velarde MC,
Simmons C, Rahal O and Simmen FA: The emerging role of Krüppel-like
factors in endocrine-responsive cancers of female reproductive
tissues. J Endocrinol. 204:223–231. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tamanini S, Comi GP and Corti S: In vivo
transient and partial cell reprogramming to pluripotency as a
therapeutic tool for neurodegenerative diseases. Mol Neurobiol.
55:6850–6862. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vangapandu H and Ai W: Krüppel like factor
4 (KLF4): A transcription factor with diverse context-dependent
functions. Gene Ther Mol Biol 13a. 194–204. 2009.
|