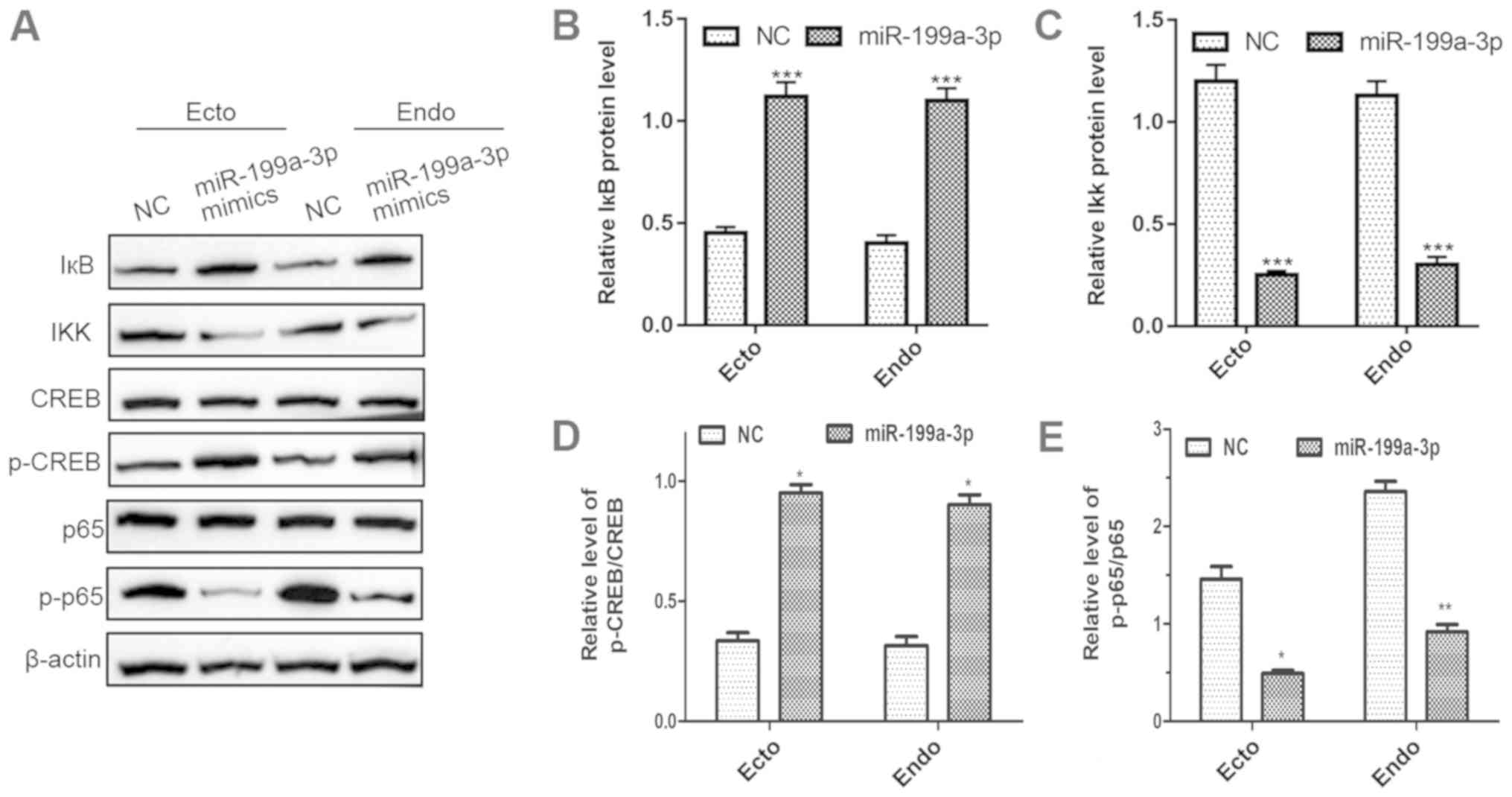
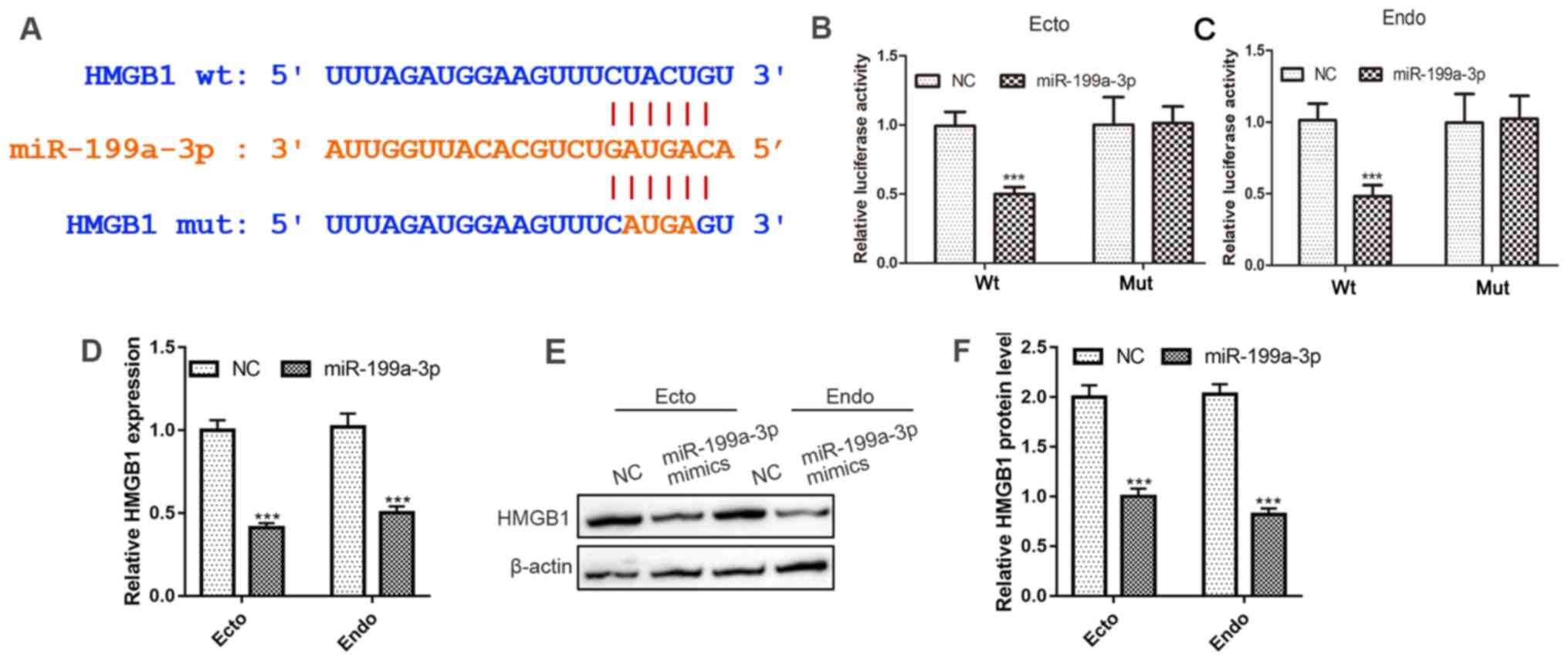
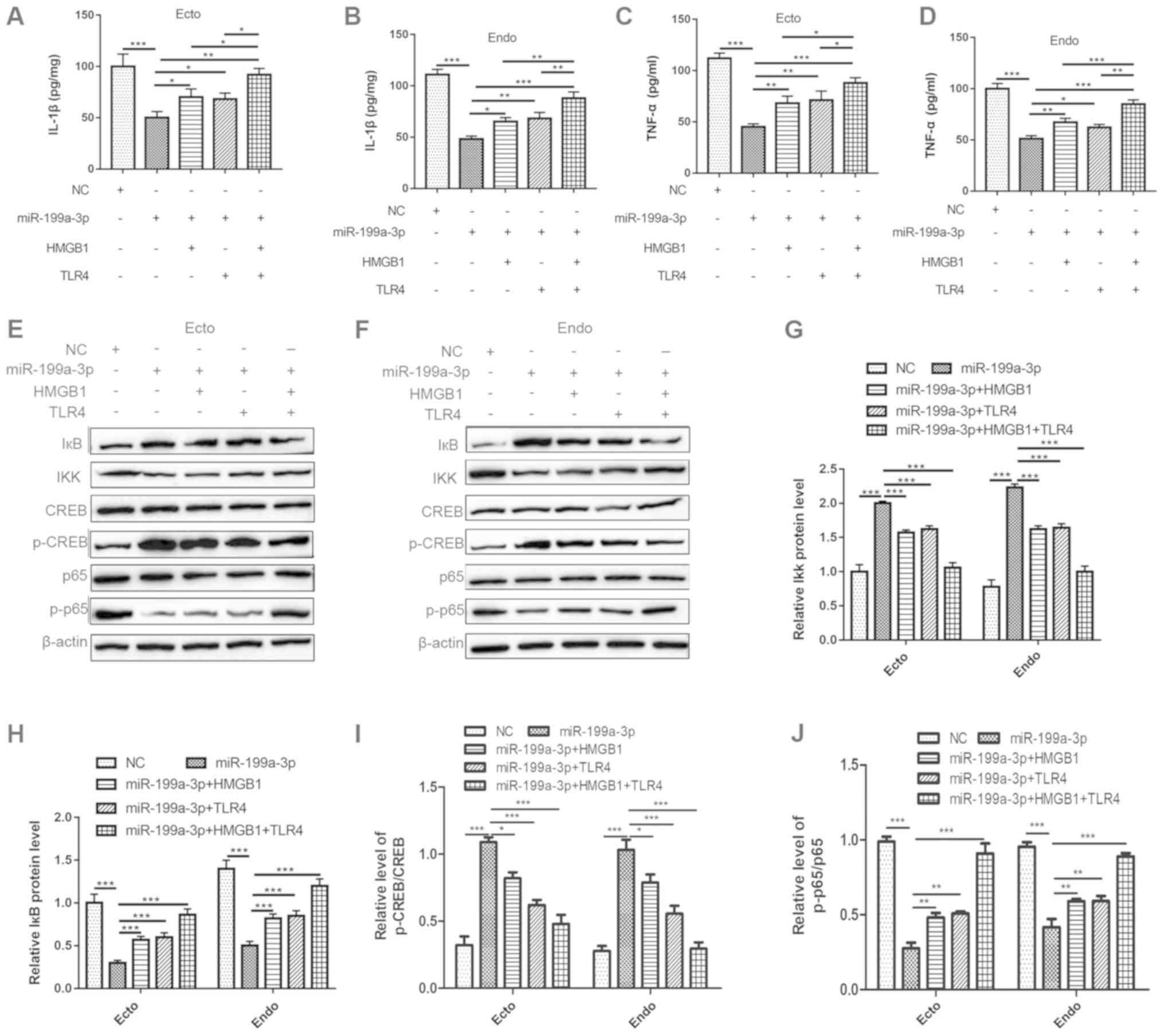
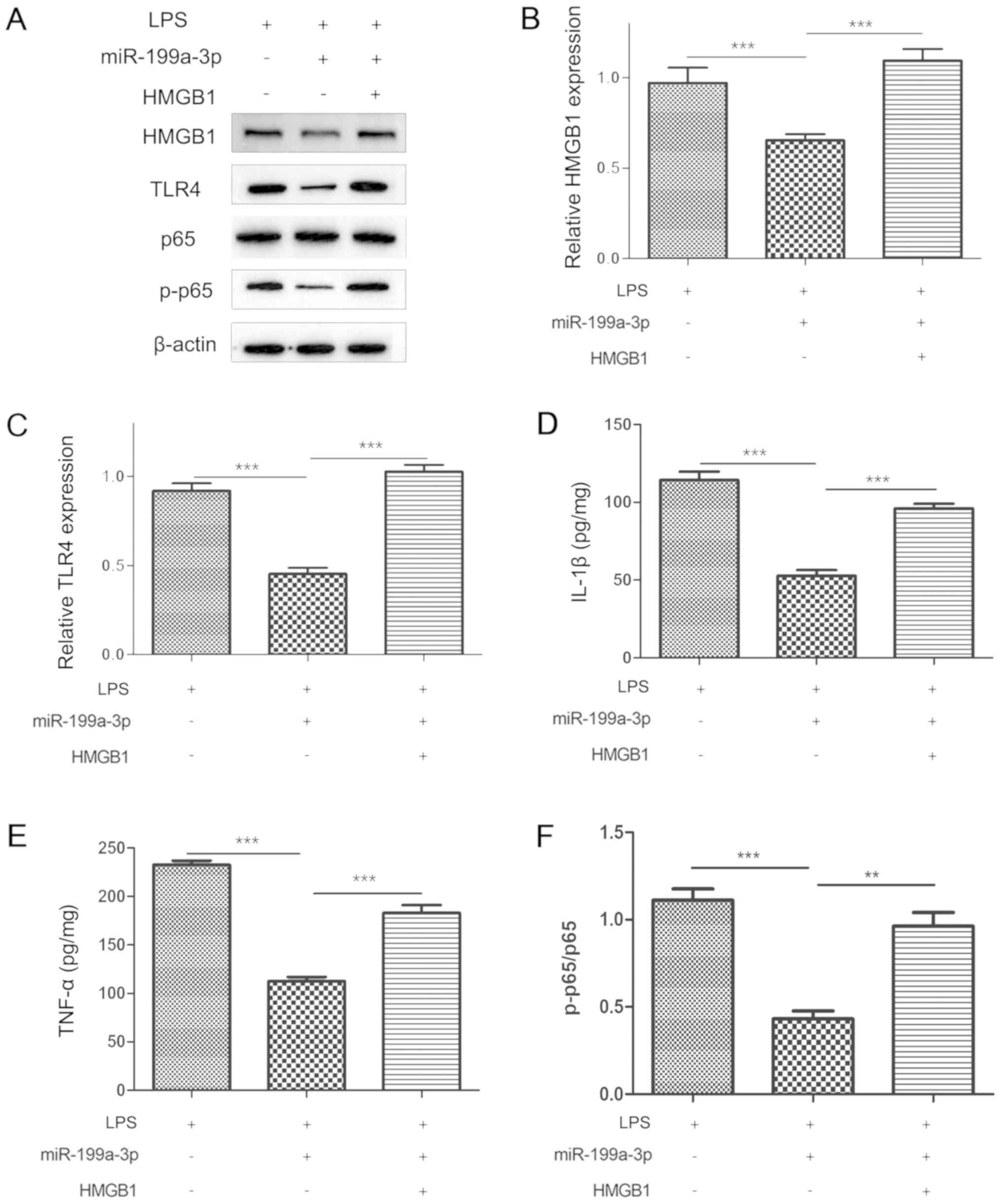
|
1
|
McCall EM, Alderdice F, Halliday HL, Vohra
S and Johnston L: Interventions to prevent hypothermia at birth in
preterm and/or low birth weight infants. Cochrane Database Syst
Rev. 2:CD0042102018.PubMed/NCBI
|
|
2
|
Son M and Miller ES: Predicting preterm
birth: Cervical length and fetal fibronectin. Semin Perinatol.
41:445–451. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gomez-Lopez N, Romero R, Xu Y, Plazyo O,
Unkel R, Leng Y, Than NG, Chaiworapongsa T, Panaitescu B, Dong Z,
et al: A role for the inflammasome in spontaneous preterm labor
with acute histologic chorioamnionitis. Reprod Sci. 24:1382–1401.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Boyle AK, Rinaldi SF, Norman JE and Stock
SJ: Preterm birth: Inflammation, fetal injury and treatment
strategies. J Reprod Immunol. 119:62–66. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vink J and Mourad M: The pathophysiology
of human premature cervical remodeling resulting in spontaneous
preterm birth: Where are we now? Semin Perinatol. 41:427–437. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Carter Moylan HE, Caitlyn Nguyen-Ngo C,
Ratana Lim R and Lappas M: The short chain fatty acids butyrate and
propionate protect against inflammation induced activation of
mediators involved in active labor: implications for preterm birth.
Mol Hum Reprod. April 1–2020.(Epub ahead of print).
|
|
7
|
Canan Unal C, Esra Karatas E, Fadıloglu E,
Portakal O and Beksac MS: Comparison of term and preterm labor
procalcitonin and leukocyte cell volume, conductivity and light
scatter (VCS) parameters in order to demonstrate the impact of
inflammation on the triggering mechanisms of preterm uterin
contractions. J Obstet Gynaecol Res. 46:694–698. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Challis JR, Lockwood CJ, Myatt L, Norman
JE, Strauss JF III and Petraglia F: Inflammation and pregnancy.
Reprod Sci. 16:206–215. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mendelson CR: Minireview: Fetal-maternal
hormonal signaling in pregnancy and labor. Mol Endocrinol.
23:947–954. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lu HY, Ma JL, Shan JY, Zhang J, Wang QX
and Zhang Q: High-mobility group box-1 and receptor for advanced
glycation end products in preterm infants with brain injury. World
J Pediatr. 13:228–235. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Splichalova A, Slavikova V, Splichalova Z
and Splichal I: Preterm life in sterile conditions: A study on
preterm, germ-free piglets. Front Immunol. 9:2202018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Andersson U and Tracey KJ: HMGB1 is a
therapeutic target for sterile inflammation and infection. Annu Rev
Immunol. 29:139–162. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Baumbusch MA, Buhimschi CS, Oliver EA,
Zhao G, Thung S, Rood K and Buhimschi IA: High Mobility Group-Box 1
(HMGB1) levels are increased in amniotic fluid of women with
intra-amniotic inflammation-determined preterm birth, and the
source may be the damaged fetal membranes. Cytokine. 81:82–87.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Plazyo O, Romero R, Unkel R, Balancio A,
Mial TN, Xu Y, Dong Z, Hassan SS and Gomez-Lopez N: HMGB1 induces
an inflammatory response in the chorioamniotic membranes that is
partially mediated by the inflammasome. Biol Reprod. 95:1302016.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Menon R, Behnia F, Polettini J, Saade GR,
Campisi J and Velarde M: Placental membrane aging and HMGB1
signaling associated with human parturition. Aging (Albany NY).
8:216–230. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mukherjee S, Huda S and Sinha Babu SP:
Toll-like receptor polymorphism in host immune response to
infectious diseases: A review. Scand J Immunol. 90:e127712019.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tsung A, Klune JR, Zhang X, Jeyabalan G,
Cao Z, Peng X, Stolz DB, Geller DA, Rosengart MR and Billiar TR:
HMGB1 release induced by liver ischemia involves Toll-like receptor
4-dependent reactive oxygen species production and calcium-mediated
signaling. J Exp Med. 204((12)): 2913–2923. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Park JS, Svetkauskaite D, He Q, Kim JY,
Strassheim D, Ishizaka A and Abraham E: Involvement of toll-like
receptors 2 and 4 in cellular activation by high mobility group box
1 protein. J Biol Chem. 279:7370–7377. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tsung A, Zheng N, Jeyabalan G, Izuishi K,
Klune JR, Geller DA, Lotze MT, Lu L and Billiar TR: Increasing
numbers of hepatic dendritic cells promote HMGB1-mediated
ischemia-reperfusion injury. J Leukoc Biol. 81:119–128. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fan J, Li Y, Levy RM, Fan JJ, Hackam DJ,
Vodovotz Y, Yang H, Tracey KJ, Billiar TR and Wilson MA:
Hemorrhagic shock induces NAD(P)H oxidase activation in
neutrophils: Role of HMGB1-TLR4 signaling. J Immunol.
178:6573–6580. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kumar N, Nandula P, Menden H, Jarzembowski
J and Sampath V: Placental TLR/NLR expression signatures are
altered with gestational age and inflammation. J Matern Fetal
Neonatal Med. 30:1588–1595. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhong X, Jiang YZ, Liu P, He W, Xiong Z,
Chang W, Zhu J and Cui Q: Toll-like 4 receptor /NFκB
inflammatory/miR-146a pathway contributes to the ART-correlated
preterm birth outcome. Oncotarget. 7:72475–72485. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Elovitz MA, Anton L, Bastek J and Brown
AG: Can microRNA profiling in maternal blood identify women at risk
for preterm birth? Am J Obstet Gynecol. 212:782.e1–782.e5. 2015.
View Article : Google Scholar
|
|
24
|
Elovitz MA, Brown AG, Anton L, Gilstrop M,
Heiser L and Bastek J: Distinct cervical microRNA profiles are
present in women destined to have a preterm birth. Am J Obstet
Gynecol. 210:221.e1–11. 2014. View Article : Google Scholar
|
|
25
|
Wommack JC, Trzeciakowski JP, Miranda RC,
Stowe RP and Ruiz RJ: Micro RNA clusters in maternal plasma are
associated with preterm birth and infant outcomes. PLoS One.
13:e01990292018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Williams KC, Renthal NE, Gerard RD and
Mendelson CR: The microRNA (miR)-199a/214 cluster mediates opposing
effects of progesterone and estrogen on uterine contractility
during pregnancy and labor. Mol Endocrinol. 26:1857–1867. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bardin P, Marchal-Duval E, Sonneville F,
Blouquit-Laye S, Rousselet N, Le Rouzic P, Corvol H and Tabary O:
Small RNA and transcriptome sequencing reveal the role of
miR-199a-3p in inflammatory processes in cystic fibrosis airways. J
Pathol. 245:410–420. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen Z, Dong WH, Chen Q, Li QG and Qiu ZM:
NLRP1Downregulation of miR-199a-3p mediated by the
CtBP2-HDAC1-FOXP3 transcriptional complex contributes to acute lung
injury by targeting. Int J Biol Sci. 15:2627–2640. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Δ Δ C(T)) Method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Renthal NE, Chen CC, Williams KC, Gerard
RD, Prange-Kiel J and Mendelson CR: miR-200 family and targets,
ZEB1 and ZEB2, modulate uterine quiescence and contractility during
pregnancy and labor. Proc Natl Acad Sci USA. 107:20828–20833. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Organization WH: WHO: Recommended
definitions, terminology and format for statistical tables related
to the perinatal period and use of a new certificate for cause of
perinatal deaths. Modifications recommended by FIGO as amended
October 14, 1976. Acta Obstet Gynecol Scand. 56:247–253.
1977.PubMed/NCBI
|
|
32
|
Goldenberg RL, Culhane JF, Iams JD and
Romero R: Epidemiology and causes of preterm birth. Lancet.
371:75–84. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu L, Johnson HL, Cousens S, Perin J,
Scott S, Lawn JE, Rudan I, Campbell H, Cibulskis R, Li M, et al
Child Health Epidemiology Reference Group of WHO and UNICEF, :
Global, regional, and national causes of child mortality: An
updated systematic analysis for 2010 with time trends since 2000.
Lancet. 379:2151–2161. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Paquette AG, Shynlova O, Kibschull M,
Price ND and Lye SJ; Global Alliance to Prevent Prematurity and
Stillbirth Systems Biology of Preterm Birth Team, : Comparative
analysis of gene expression in maternal peripheral blood and
monocytes during spontaneous preterm labor. Am J Obstet Gynecol.
218:345.e341–345.e330. 2018. View Article : Google Scholar
|
|
35
|
Romero R, Espinoza J, Kusanovic JP, Gotsch
F, Hassan S, Erez O, Chaiworapongsa T and Mazor M: The preterm
parturition syndrome. BJOG. 113 (Suppl 3):17–42. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gonçalves LF, Chaiworapongsa T and Romero
R: Intrauterine infection and prematurity. Ment Retard Dev Disabil
Res Rev. 8:3–13. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
The Preterm Labor Syndrome, . Biochemical,
Cytologic, Immunologic, Pathologic, Microbiologic, and Clinical
Evidence That Preterm Labor Is a Heterogeneous Disease. Am J Obstet
Gynecol. 168:2881993. View Article : Google Scholar
|
|
38
|
Ludmir J, Samuels P, Brooks S and Mennuti
MT: Pregnancy outcome of patients with uncorrected uterine
anomalies managed in a high-risk obstetric setting. Obstet Gynecol.
75:906–910. 1990.PubMed/NCBI
|
|
39
|
Lallar M and Nandal R and Nandal R; Anam
Ul Haq, : Perinatal outcome in idiopathic polyhydramnios. J Obstet
Gynaecol India. 65:310–314. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Colditz P: Multiple Pregnancy:
Epidemiology, Gestation, and Perinatal Outcome. Twin Res Hum Genet.
9:183–184. 2006. View Article : Google Scholar
|
|
41
|
Choi J, Park JW, Kim BJ, Choi YJ, Hwang JH
and Lee SM: Funisitis is more common in cervical insufficiency than
in preterm labor and preterm premature rupture of membranes. J
Perinat Med. 44:523–529. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Hobel CJ: Stress and preterm birth. Clin
Obstet Gynecol. 47:856–880, discussion 881–882. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Romero R, Dey SK and Fisher SJ: Preterm
labor: One syndrome, many causes. Science. 345:760–765. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Romero R, Gómez R, Chaiworapongsa T,
Conoscenti G, Kim JC and Kim YM: The role of infection in preterm
labour and delivery. Paediatr Perinat Epidemiol. 15 (Suppl
2):41–56. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gilman-Sachs A, Dambaeva S, Salazar Garcia
MD, Hussein Y, Kwak-Kim J and Beaman K: Inflammation induced
preterm labor and birth. J Reprod Immunol. 129:53–58. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chen GY and Nuñez G: Sterile inflammation:
Sensing and reacting to damage. Nat Rev Immunol. 10:826–837. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Cappelletti M, Della Bella S, Ferrazzi E,
Mavilio D and Divanovic S: Inflammation and preterm birth. J Leukoc
Biol. 99:67–78. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Timmons BC, Mitchell SM, Gilpin C and
Mahendroo MS: Dynamic changes in the cervical epithelial tight
junction complex and differentiation occur during cervical ripening
and parturition. Endocrinology. 148:1278–1287. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Gonzalez JM, Xu H, Chai J, Ofori E and
Elovitz MA: Preterm and term cervical ripening in CD1 Mice (Mus
musculus): Similar or divergent molecular mechanisms? Biol Reprod.
81:1226–1232. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Timmons B, Akins M and Mahendroo M:
Cervical remodeling during pregnancy and parturition. Trends
Endocrinol Metab. 21:353–361. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Read CP, Word RA, Ruscheinsky MA, Timmons
BC and Mahendroo MS: Cervical remodeling during pregnancy and
parturition: Molecular characterization of the softening phase in
mice. Reproduction. 134:327–340. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Nold C, Anton L, Brown A and Elovitz M:
Inflammation promotes a cytokine response and disrupts the cervical
epithelial barrier: a possible mechanism of premature cervical
remodeling and preterm birth. Am J Obstet Gynecol 206: 208.e1-7,
2012.
|
|
53
|
Akgul Y, Word RA, Ensign LM, Yamaguchi Y,
Lydon J, Hanes J and Mahendroo M: Hyaluronan in cervical epithelia
protects against infection-mediated preterm birth. J Clin Invest.
124:5481–5489. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Timmons BC and Mahendroo M: Processes
regulating cervical ripening differ from cervical dilation and
postpartum repair: Insights from gene expression studies. Reprod
Sci. 14 (Suppl):53–62. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gu N, You L, Shi C, Yang L, Pang L, Cui X,
Ji C, Zheng W and Guo X: Expression of miR-199a-3p in human
adipocytes is regulated by free fatty acids and adipokines. Mol Med
Rep. 14:1180–1186. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lotze MT and Tracey KJ: High-mobility
group box 1 protein (HMGB1): Nuclear weapon in the immune arsenal.
Nat Rev Immunol. 5:331–342. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Bellussi LM, Iosif C, Sarafoleanu C, Jianu
E, Duda R, Panaitescu E, Passali FM and Passali D: Are HMGB1
protein expression and secretion markers of upper airways
inflammatory diseases? J Biol Regul Homeost Agents. 27:791–804.
2013.PubMed/NCBI
|
|
58
|
Tian J, Avalos AM, Mao SY, Chen B, Senthil
K, Wu H, Parroche P, Drabic S, Golenbock D, Sirois C, et al:
Toll-like receptor 9-dependent activation by DNA-containing immune
complexes is mediated by HMGB1 and RAGE. Nat Immunol. 8:487–496.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Dong W, Zhu Q, Yang B, Qin Q, Wang Y, Xia
X, Zhu X, Liu Z, Song E and Song Y: Polychlorinated biphenyl
quinone induces caspase 1-mediated pyroptosis through induction of
pro-inflammatory HMGB1-TLR4-NLRP3-GSDMD signal axis. Chem Res
Toxicol. 32:1051–1057. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Thankam FG, Roesch ZK, Dilisio MF, Radwan
MM, Kovilam A, Gross RM and Agrawal DK: Association of inflammatory
responses and ECM disorganization with HMGB1 upregulation and NLRP3
inflammasome activation in the injured rotator cuff tendon. Sci
Rep. 8:89182018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Kim EJ, Park SY, Baek SE, Jang MA, Lee WS,
Bae SS, Kim K and Kim CD: HMGB1 increases IL-1β production in
vascular smooth muscle cells via NLRP3 inflammasome. Front Physiol.
9:3132018. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Subramanian S, Pallati PK, Sharma P,
Agrawal DK and Nandipati KC: Significant association of TREM-1 with
HMGB1, TLRs and RAGE in the pathogenesis of insulin resistance in
obese diabetic populations. Am J Transl Res. 9:3224–3244.
2017.PubMed/NCBI
|
|
63
|
Son GH, Kim Y, Lee JJ, Lee KY, Ham H, Song
JE, Park ST and Kim YH: MicroRNA-548 regulates high mobility group
box 1 expression in patients with preterm birth and
chorioamnionitis. Sci Rep. 9:197462019. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Kumar N, Nandula P, Menden H, Jarzembowski
J and Sampath V: Placental TLR/NLR expression signatures are
altered with gestational age and inflammation. J Matern Fetal
Neonatal Med. 30:1588–1595. 2017. View Article : Google Scholar : PubMed/NCBI
|