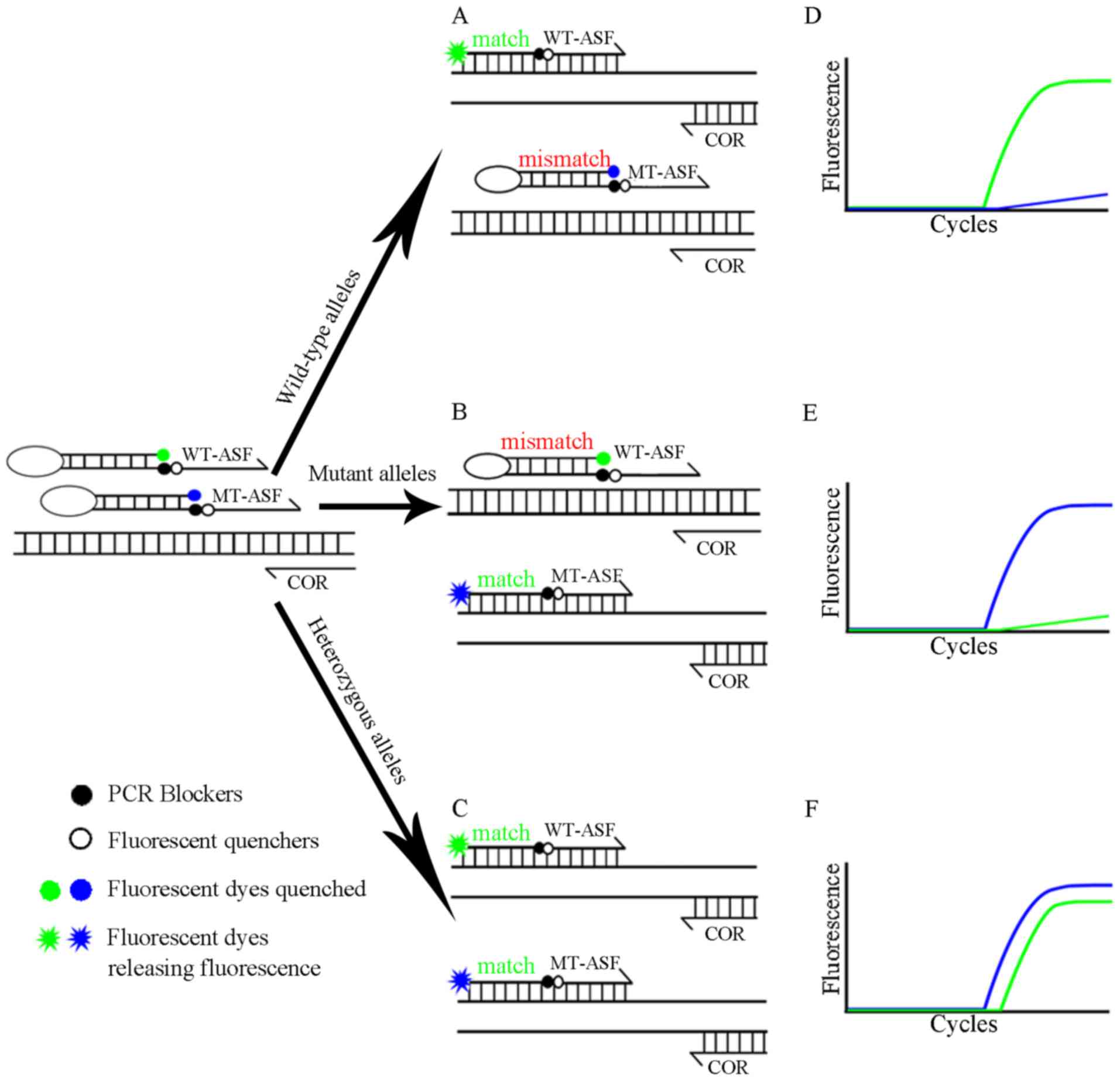
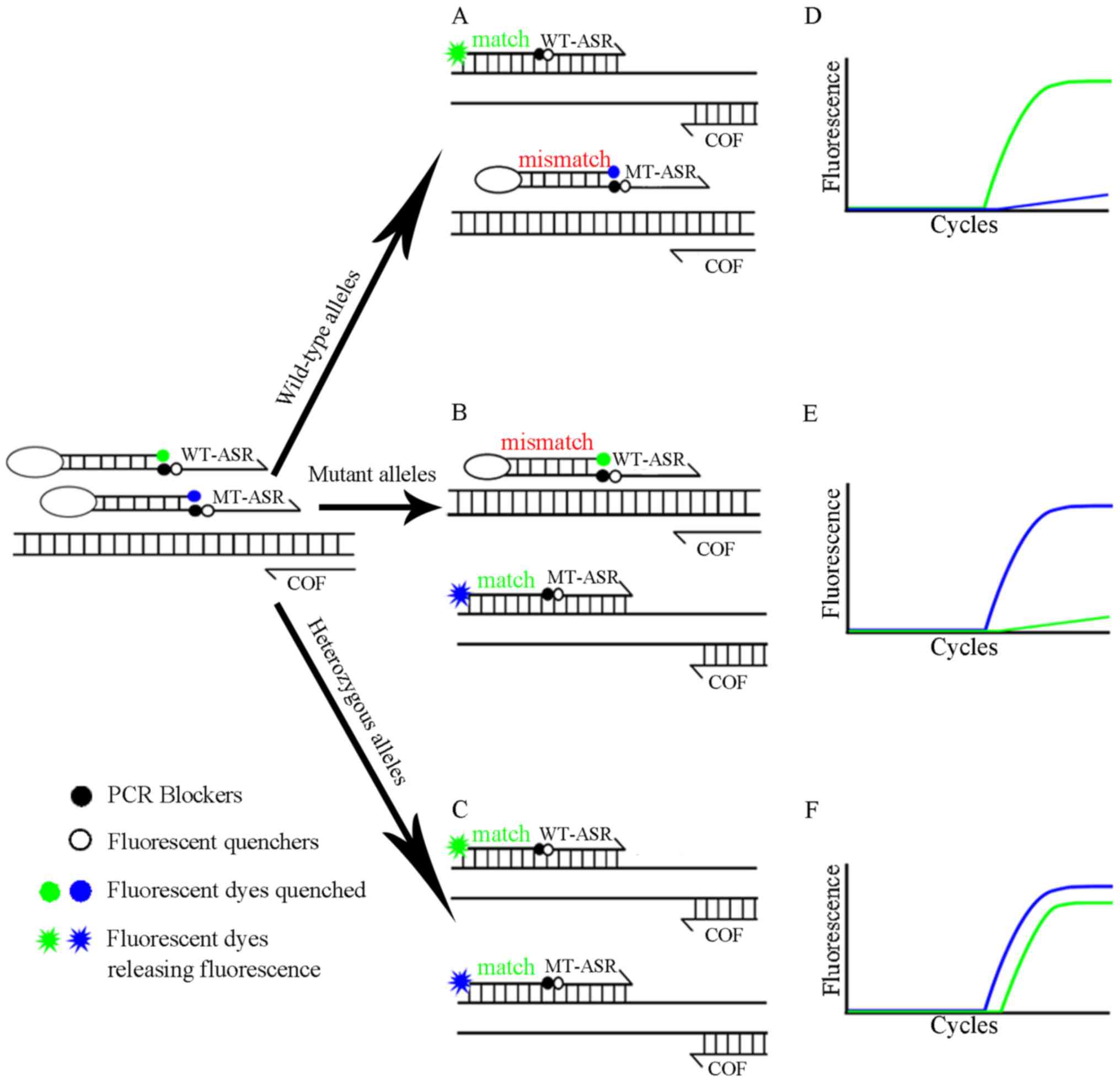
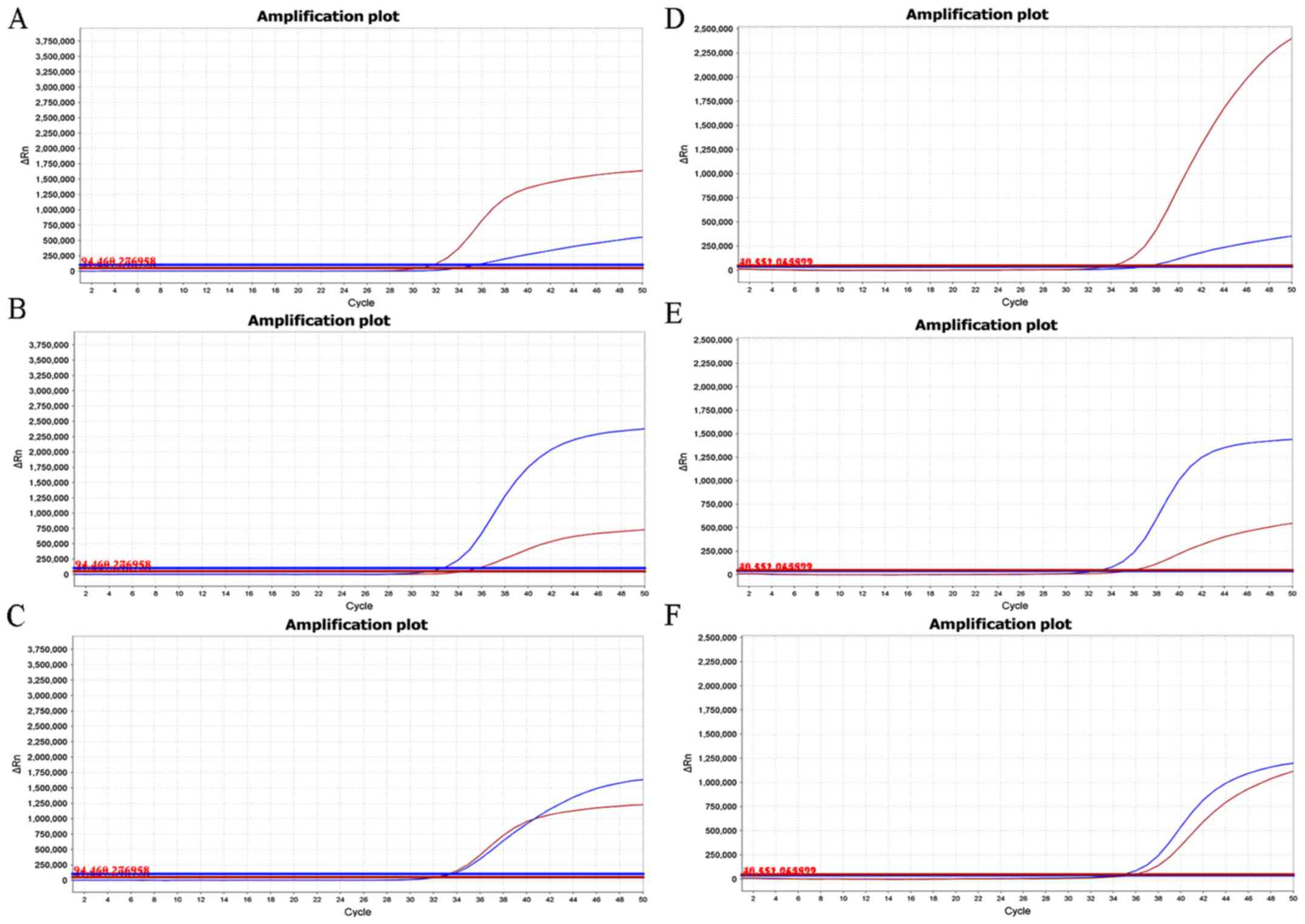
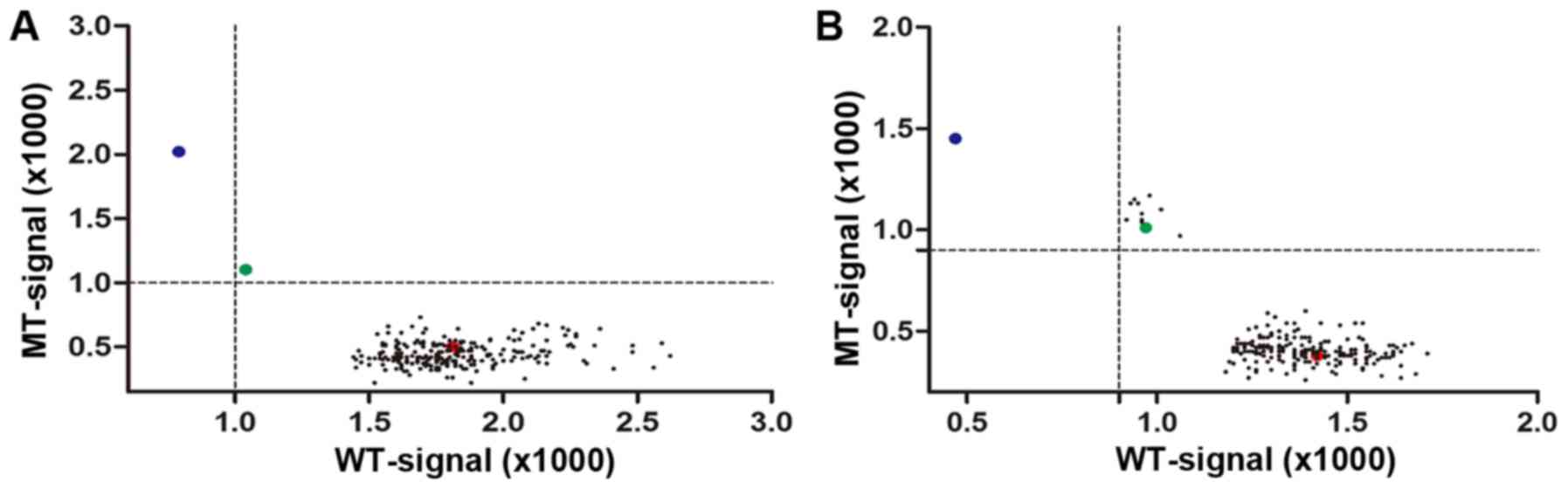
|
1
|
Appell M, Hindorf U, Almer S and Peterson
C: Thiopurines in inflammatory bowel disease-the role of
pharmacogenetics and therapeutic drug monitoring. Curr
Pharmacogenomics. 4:285–300. 2006. View Article : Google Scholar
|
|
2
|
Ford LT and Berg JD: Thiopurine
S-methyltransferase (TPMT) assessment prior to starting thiopurine
drug treatment; A pharmacogenomic test whose time has come. J Clin
Pathol. 63:288–295. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Florin THJ, Wright JD, Jambhrunkar SD,
Henman MG and Popat A: A well-tolerated and rapidly acting
thiopurine for IBD? Drug Discov Today. 24:37–41. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Saibeni S, Kohn A, Meucci G and Papi C;
Italian Group for Inflammatory Bowel Disease, : How thiopurines are
used for the treatment of inflammatory bowel diseases: An Italian
survey. Dig Liver Dis. 47:170–173. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Estlin EJ: Continuing therapy for
childhood acute lymphoblastic leukaemia: Clinical and cellular
pharmacology of methotrexate, 6-mercaptopurine and 6-thioguanine.
Cancer Treat Rev. 27:351–363. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sobiak J, Skalska-Sadowska J, Chrzanowska
M, Resztak M, Koltan S, Wysocki M and Wachowiak J: Thiopurine
methyltransferase activity in children with acute myeloid leukemia.
Oncol Lett. 16:4699–4706. 2018.PubMed/NCBI
|
|
7
|
Hollander AA, van Saase JL, Kootte AM, van
Dorp WT, van Bockel HJ, van Es LA and van der Woude FJ: Beneficial
effects of conversion from cyclosporin to azathioprine after kidney
transplantation. Lancet. 345:610–614. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hogarth LA, Redfern CP, Teodoridis JM,
Hall AG, Anderson H, Case MC and Coulthard SA: The effect of
thiopurine drugs on DNA methylation in relation to TPMT expression.
Biochem Pharmacol. 76:1024–1035. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Larussa T, Suraci E, Lentini M, Nazionale
I, Gallo L, Abenavoli L, Imeneo M, Costanzo FS, Cuda G and Luzza F:
High prevalence of polymorphism and low activity of thiopurine
methyltransferase in patients with inflammatory bowel disease. Eur
J Intern Med. 23:273–277. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Matalon ST, Ornoy A and Lishner M: Review
of the potential effects of three commonly used antineoplastic and
immunosuppressive drugs (cyclophosphamide, azathioprine,
doxorubicin on the embryo and placenta). Reprod Toxicol.
18:219–230. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dervieux T, Blanco JG, Krynetski EY, Vanin
EF, Roussel MF and Relling MV: Differing contribution of thiopurine
methyltransferase to mercaptopurine versus thioguanine effects in
human leukemic cells. Cancer Res. 61:5810–5816. 2001.PubMed/NCBI
|
|
12
|
Coulthard SA, Hogarth LA, Little M,
Matheson EC, Redfern CPF, Minto L and Hall AG: The effect of
thiopurine methyltransferase expression on sensitivity to
thiopurine drugs. Mol Pharmacol. 62:102–109. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang RS and Ratain MJ: Pharmacogenetics
and pharmacogenomics of anticancer agents. CA Cancer J Clin.
59:42–55. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lorenz M, Weise A, Prause S, Klemm M,
Eidens M, Luchi M, Forst T, Pfützner A and Weber MM: Development
and validation of a rapid and reliable method for TPMT genotyping
using real-time PCR. Clin Lab. 58:959–971. 2012.PubMed/NCBI
|
|
15
|
Maitland ML, Vasisht K and Ratain MJ:
TPMT, UGT1A1 and DPYD: Genotyping to ensure safer cancer therapy?
Trends Pharmacol Sci. 27:432–437. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nguyen CM, Mendes MAS and Ma JD:
Thiopurine methyltransferase (TPMT) genotyping to predict
myelosuppression risk. PLoS Current. 3:RRN12362011. View Article : Google Scholar
|
|
17
|
Hindorf U and Appell ML: Genotyping should
be considered the primary choice for pre-treatment evaluation of
thiopurine methyltransferase function. J Crohns Colitis. 6:655–659.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Relling MV, Gardner EE, Sandborn WJ,
Schmiegelow K, Pui CH, Yee SW, Stein CM, Carrillo M, Evans WE and
Klein TE; Clinical Pharmacogenetics Implementation Consortium, :
Clinical Pharmacogenetics Implementation Consortium guidelines for
thiopurine methyltransferase genotype and thiopurine dosing. Clin
Pharmacol Ther. 89:387–391. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen D, Yang Z, Xia H, Huang JF, Zhang Y,
Jiang TN, Wang GY, Chuai ZR, Fu WL and Huang Q: Enhanced
specificity of TPMT*2 genotyping using unidirectional wild-type and
mutant allele-specific scorpion primers in a single tube. PLoS One.
9:e918242014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sanger F, Nicklen S and Coulson AR: DNA
sequencing with chain-terminating inhibitors. Proc Natl Acad Sci
USA. 74:5463–5467. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Román M, Cabaleiro T, Ochoa D, Novalbos J,
Chaparro M, Gisbert JP and Abad-Santos F: Validation of a
genotyping method for analysis of TPMT polymorphisms. Clin Ther.
34:878–884. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shendure J, Porreca G, Reppas N, Lin X,
McCutcheon J, Rosenbaum A, Wang M, Zhang K, Mitra R and Church G:
Accurate multiplex polony sequencing of an evolved bacterial
genome. Science. 309:1728–1732. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shendure J, Mitra RD, Varma C and Church
GM: Advanced sequencing technologies: Methods and goals. Nat Rev
Genet. 5:335–344. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shendure J and Ji H: Next-generation DNA
sequencing. Nat Biotechnol. 26:1135–1145. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wangkumhang P, Chaichoompu K, Ngamphiw C,
Ruangrit U, Chanprasert J, Assawamakin A and Tongsima S: WASP: A
Web-based Allele-Specific PCR assay designing tool for detecting
SNPs and mutations. BMC Genomics. 8:2752007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Collie-Duguid ES, Pritchard SC, Powrie RH,
Sludden J, Collier DA, Li T and McLeod HL: The frequency and
distribution of thiopurine methyltransferase alleles in Caucasian
and Asian populations. Pharmacogenetics. 9:37–42. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tyagi S, Bratu DP and Kramer FR:
Multicolor molecular beacons for allele discrimination. Nat
Biotechnol. 16:49–53. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ng CT, Gilchrist CA, Lane A, Roy S, Haque
R and Houpt ER: Multiplex real-time PCR assay using Scorpion probes
and DNA capture for genotype-specific detection of Giardia lamblia
on fecal samples. J Clin Microbiol. 43:1256–1260. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang K, Niu Y, Wang Q, Liu H, Jin Y and
Zhang S: Cloning and evaluation of reference genes for quantitative
real-time PCR analysis in Amorphophallus. PeerJ. 5:e32602017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qu XM, Zhao N, Mo QY, Yao P, Su N, Wei K,
Wang L, Huang JF, Ren XD, Ren S, et al: Development of
duplex-crossed allele-specific PCR targeting of TPMT*3B and *3C
using crossed allele-specific blockers to eliminate non-specific
amplification. Anal Biochem. 575:54–62. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Burchard PR, AbouTayoun AN, Lefferts JA,
Lewis LD, Tsongalis GJ and Cervinski MA: Development of a rapid
clinical TPMT genotyping assay. Clin Biochem. 47:126–129. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang JP, Zhou SF, Chen X and Huang M:
Determination of intra-ethnic differences in the polymorphisms of
thiopurine S-methyltransferase in Chinese. Clin Chim Acta.
365:337–341. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lueders T and Friedrich MW: Evaluation of
PCR amplification bias by terminal restriction fragment length
polymorphism analysis of small-subunit rRNA and mcrA genes by using
defined template mixtures of methanogenic pure cultures and soil
DNA extracts. Appl Environ Microbiol. 69:320–326. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cabaleiro T, Roman M, Gisbert JP and
Abad-Santos F: Utility of assessing thiopurine S-methyltransferase
polymorphisms before azathioprine therapy. Curr Drug Metab.
13:1277–1293. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Robert J, Morvan VL, Smith D, Pourquier P
and Bonnet J: Predicting drug response and toxicity based on gene
polymorphisms. Crit Rev Oncol Hematol. 54:171–196. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hamdy SI, Hiratsuka M, Narahara K, Endo N,
El-Enany M, Moursi N, Ahmed MSE and Mizugaki M: Genotype and allele
frequencies of TPMT, NAT2, GST, SULT1A1 and MDR-1 in the Egyptian
population. Br J Clin Pharmacol. 55:560–569. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Teresa C, Manuel R, Javier PG and
Francisco AS: Utility of assessing Thiopurine S-methyltransferase
polymorphisms before azathioprine therapy. Curr Drug Metab.
13:1277–1293. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kham SK, Tan PL, Tay AH, Heng CK, Yeoh AE
and Quah TC: Thiopurine methyltransferase polymorphisms in a
multiracial asian population and children with acute lymphoblastic
leukemia. J Pediatr Hematol Oncol. 24:353–359. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang LR, Song DK, Zhang W, Zhao J, Jia LJ
and Xing DL: Efficient screening method of the thiopurine
methyltransferase polymorphisms for patients considering taking
thiopurine drugs in a Chinese Han population in Henan Province
(central China). Clinica Chimica Acta. 376:45–51. 2007. View Article : Google Scholar
|
|
40
|
Liu L, Yang L, Zhang YC, Ai XF, Wang JX
and Xiao ZJ: Polymorphisms of drug-metabolizing enzymes genes in a
Han Chinese population. Zhonghua Yi Xue Za Zhi. 89:2675–2681.
2009.(In Chinese). PubMed/NCBI
|