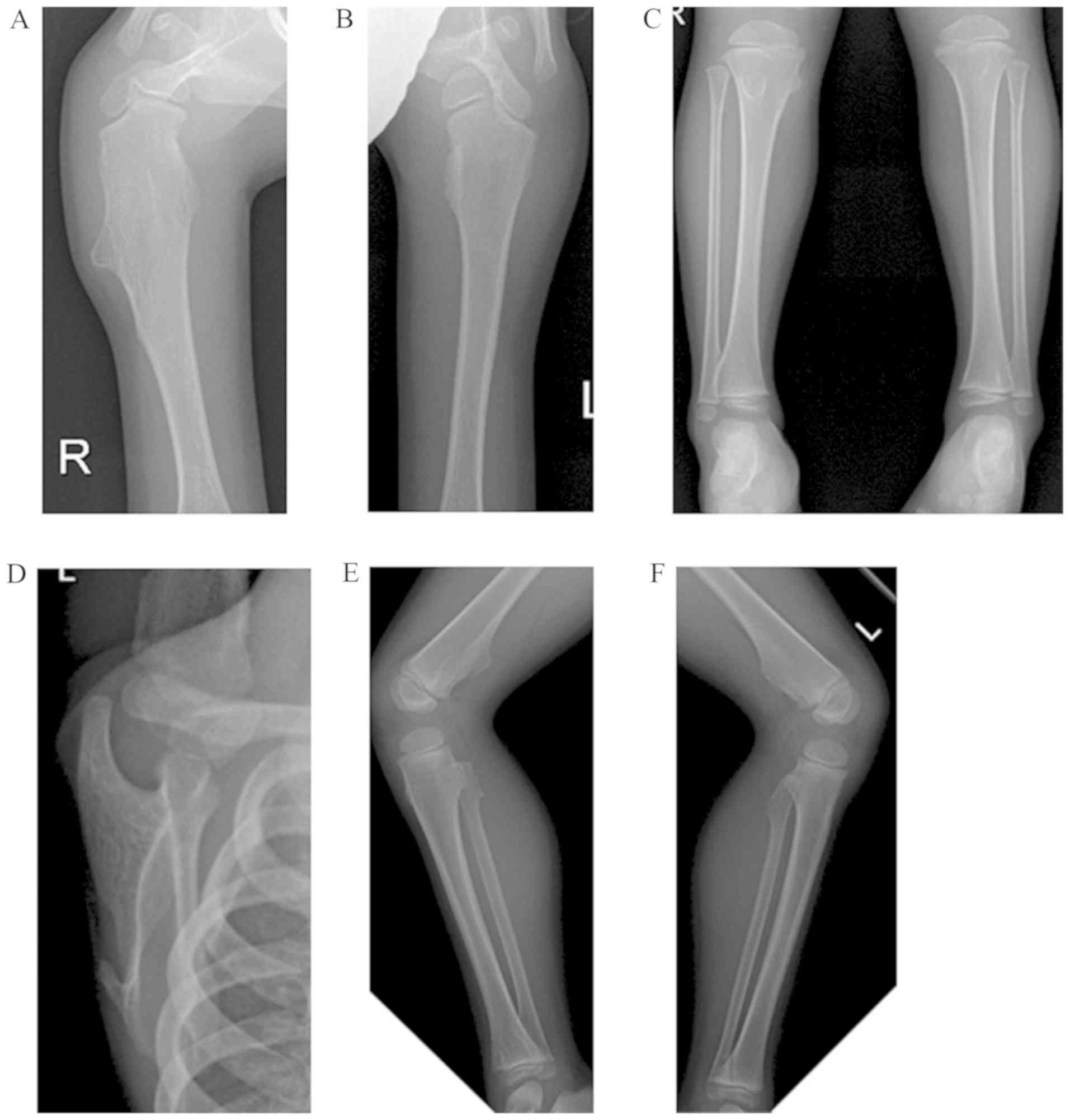
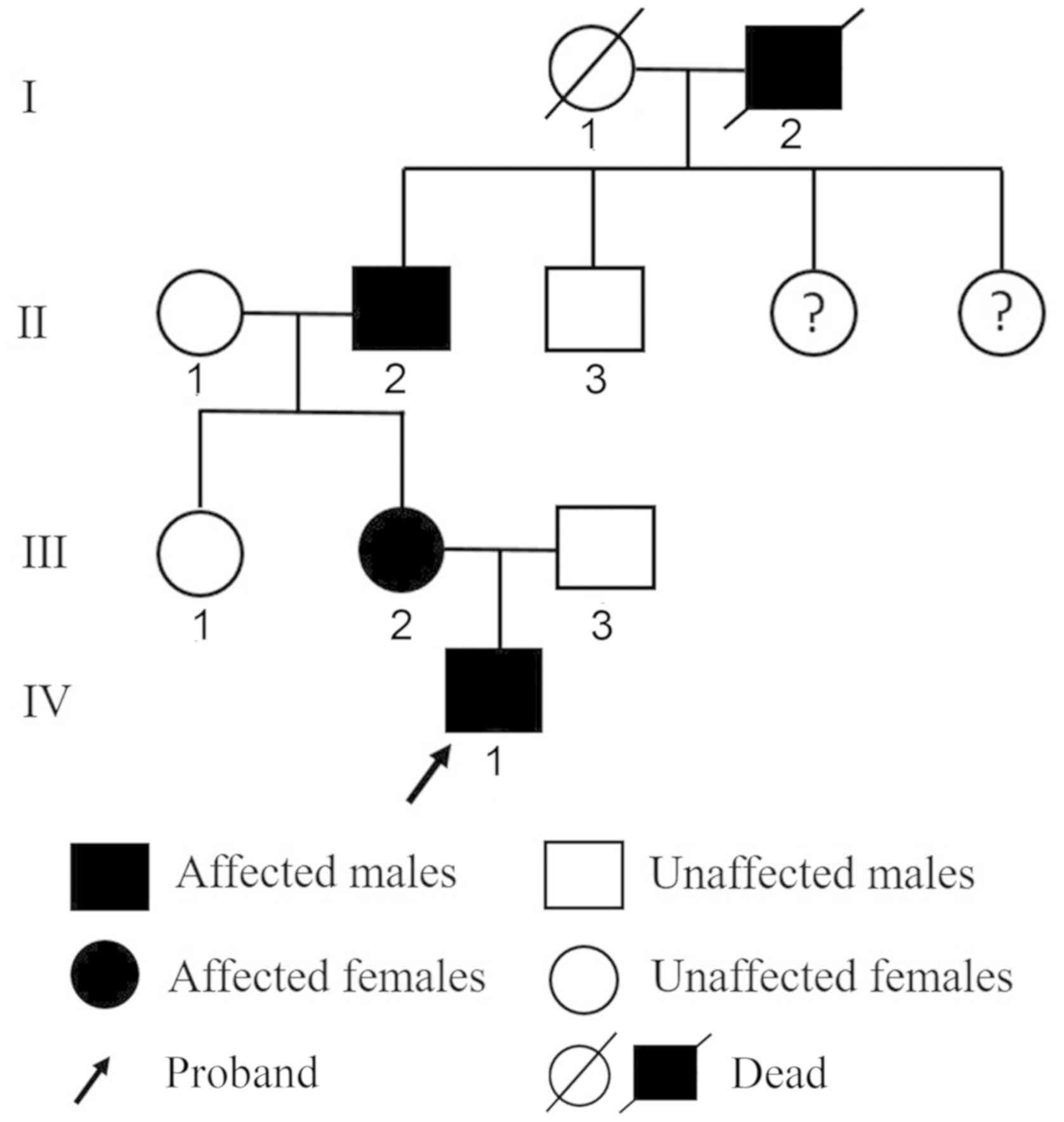
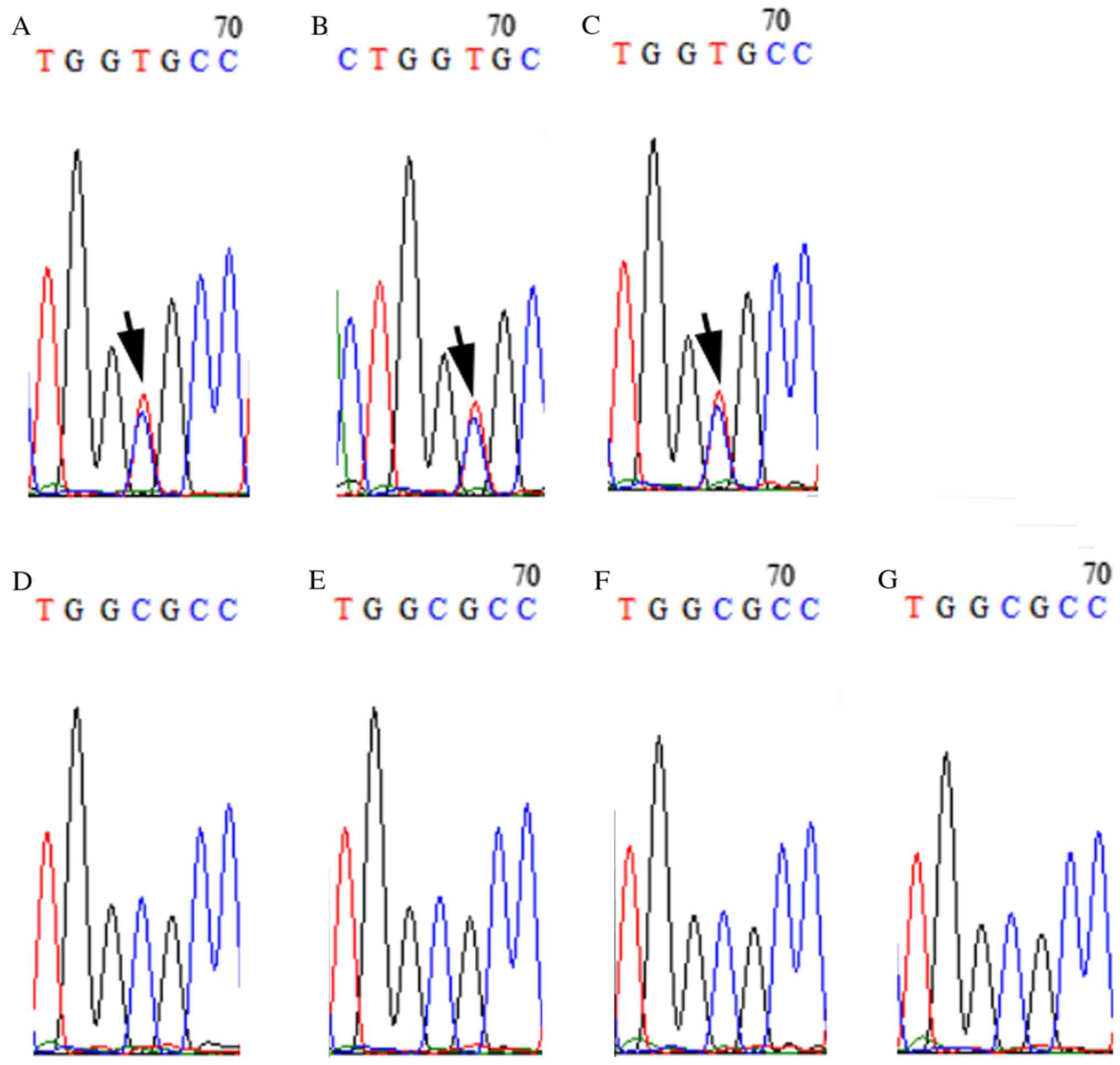
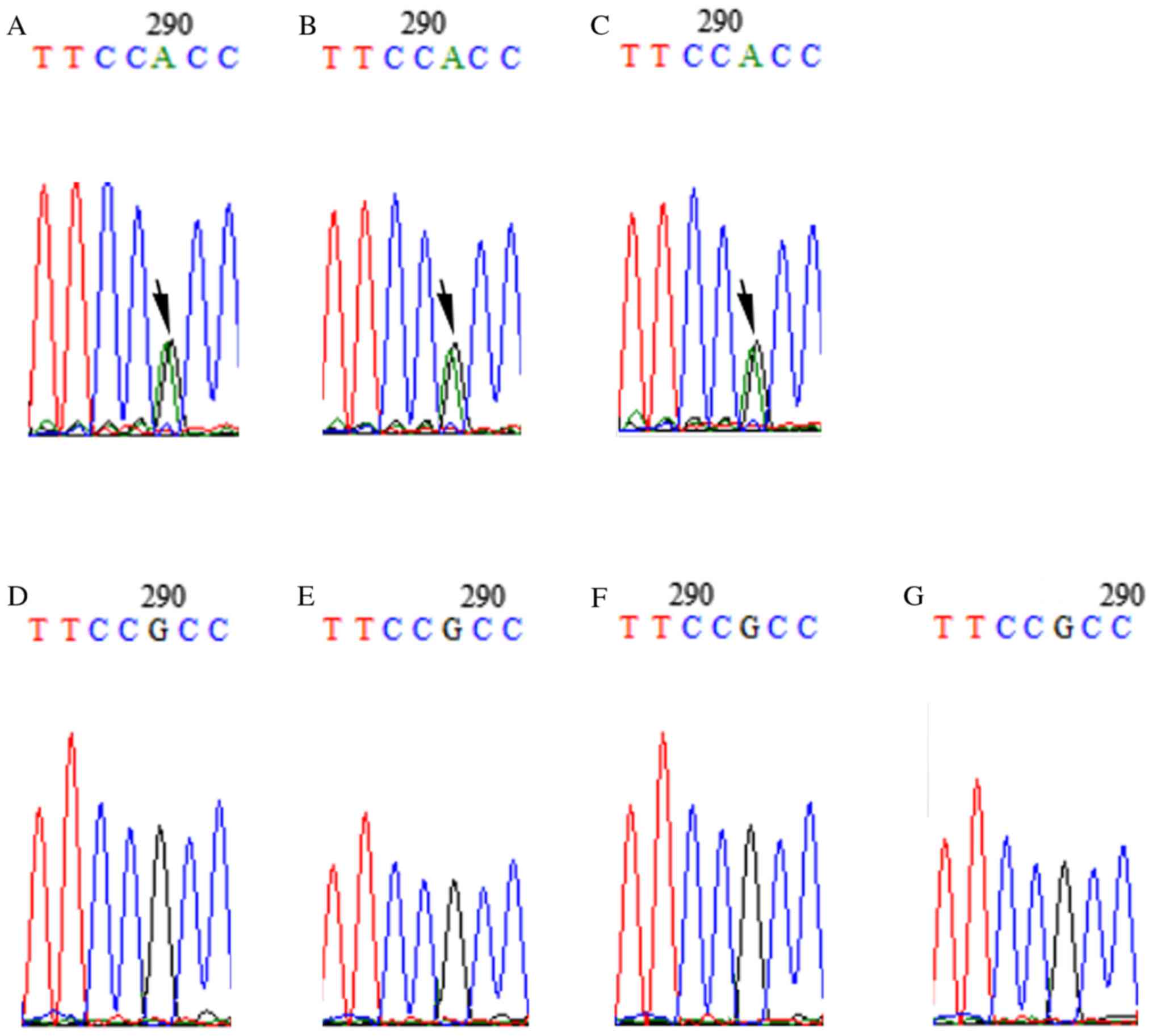
|
1
|
Beltrami G, Ristori G, Scoccianti G,
Tamburini A and Capanna R: Hereditary multiple exostoses: A review
of clinical appearance and metabolic pattern. Clin Cases Miner Bone
Metab. 13:110–118. 2016.PubMed/NCBI
|
|
2
|
McFarlane J, Knight T, Sinha A, Cole T,
Kiely N and Freeman R: Exostoses, enchondromatosis and
metachondromatosis; diagnosis and management. Acta Orthop Belg.
82:102–105. 2016.PubMed/NCBI
|
|
3
|
Pacifici M: Hereditary multiple exostoses:
New insights into pathogenesis, clinical complications, and
potential treatments. Curr Osteoporos Rep. 15:142–152. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yang C, Zhang R, Lin H and Wang H:
Insights into the molecular regulatory network of pathomechanisms
in osteochondroma. J Cell Biochem. 120:16362–16369. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jochmann K, Bachvarova V and Vortkamp A:
Heparan sulfate as a regulator of endochondral ossification and
osteochondroma development. Matrix Biol. 34:55–63. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jennes I, Pedrini E, Zuntini M, Mordenti
M, Balkassmi S, Asteggiano CG, Casey B, Bakker B, Sangiorgi L and
Wuyts W: Multiple osteochondromas: Mutation update and description
of the multiple osteochondromas mutation database (MOdb). Hum
Mutat. 30:1620–1627. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Heinritz W, Hüffmeier U, Strenge S,
Miterski B, Zweier C, Leinung S, Bohring A, Mitulla B, Peters U and
Froster UG: New mutations of EXT1 and EXT2 genes in German patients
with Multiple Osteochondromas. Ann Hum Genet. 73:283–291. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ishimaru D, Gotoh M, Takayama S, Kosaki R,
Matsumoto Y, Narimatsu H, Sato T, Kimata K, Akiyama H, Shimizu K
and Matsumoto K: Large-scale mutational analysis in the EXT1 and
EXT2 genes for Japanese patients with multiple osteochondromas. BMC
Genet. 17:522016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Santos SCL, Rizzo I, Takata RI,
Speck-Martins CE, Brum JM and Sollaci C: Analysis of mutations in
EXT1 and EXT2 in Brazilian patients with multiple osteochondromas.
Mol Genet Genomic Med. 6:382–392. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Panagopoulos I, Bjerkehagen B, Gorunova L,
Taksdal I and Heim S: Rearrangement of chromosome bands 12q14~15
causing HMGA2-SOX5 gene fusion and HMGA2 expression in
extraskeletal osteochondroma. Oncol Rep. 34:577–584. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Van Hul W, Wuyts W, Hendrickx J, Speleman
F, Wauters J, De Boulle K, Van Roy N, Bossuyt P and Willems PJ:
Identification of a third EXT-like gene (EXTL3) belonging to the
EXT gene family. Genomics. 47:230–237. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sanger F, Nicklen S and Coulson AR: DNA
sequencing with chain-terminating inhibitors. Proc Natl Acad Sci
USA. 74:5463–5467. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Richards S, Aziz N, Bale S, Bick D, Das S,
Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, et al:
Standards and guidelines for the interpretation of sequence
variants: A joint consensus recommendation of the american college
of medical genetics and genomics and the association for molecular
pathology. Genet Med. 17:405–424. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pruitt KD, Tatusova T and Maglott DR: NCBI
reference sequences (RefSeq): A curated non-redundant sequence
database of genomes, transcripts and proteins. Nucleic Acids Res 35
(Database Issue). D61–D65. 2007. View Article : Google Scholar
|
|
15
|
Hsu F, Kent WJ, Clawson H, Kuhn RM,
Diekhans M and Haussler D: The UCSC known genes. Bioinformatics.
22:1036–1046. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Flicek P, Amode MR, Barrell D, Beal K,
Billis K, Brent S, Carvalho-Silva D, Clapham P, Coates G,
Fitzgerald S, et al: Ensembl 2014. Nucleic Acids Res 42 (Database
Issue). D749–D755. 2014. View Article : Google Scholar
|
|
17
|
Cooper GM, Stone EA, Asimenos G; NISC
Comparative Sequencing Program, ; Green ED, Batzoglou S and Sidow
A: Distribution and intensity of constraint in mammalian genomic
sequence. Genome Res. 15:901–913. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Avrahami E, Cohn DF, Feibel M and Tadmor
R: MRI demonstration and CT correlation of the brain in patients
with idiopathic intracerebral calcification. J Neurol. 241:381–384.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xu L, Xia J, Jiang H, Zhou J, Li H, Wang
D, Pan Q, Long Z, Fan C and Deng HX: Mutation analysis of
hereditary multiple exostoses in the Chinese. Hum Genet. 105:45–50.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Francannet C, Cohen-Tanugi A, Le Merrer M,
Munnich A, Bonaventure J and Legeai-Mallet L: Genotype-phenotype
correlation in hereditary multiple exostoses. J Med Genet.
38:430–434. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wuyts W, Radersma R, Storm K and Vits L:
An optimized DHPLC protocol for molecular testing of the EXT1 and
EXT2 genes in hereditary multiple osteochondromas. Clin Genet.
68:542–547. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vink GR, White SJ, Gabelic S, Hogendoorn
PC, Breuning MH and Bakker E: Mutation screening of EXT1 and EXT2
by direct sequence analysis and MLPA in patients with multiple
osteochondromas: Splice site mutations and exonic deletions account
for more than half of the mutations. Eur J Hum Genet. 13:470–474.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lonie L, Porter DE, Fraser M, Cole T, Wise
C, Yates L, Wakeling E, Blair E, Morava E, Monaco AP and Ragoussis
J: Determination of the mutation spectrum of the EXT1/EXT2 genes in
British Caucasian patients with multiple osteochondromas, and
exclusion of six candidate genes in EXT negative cases. Hum Mutat.
27:11602006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kojima H, Wada T, Seki H, Kubota T, Wakui
K and Fukushima Y: One third of Japanese patients with multiple
osteochondromas may have mutations in genes other than EXT1 or
EXT2. Genet Test. 12:557–561. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jennes I, Entius MM, Van Hul E, Parra A,
Sangiorgi L and Wuyts W: Mutation screening of EXT1 and EXT2 by
denaturing high-performance liquid chromatography, direct
sequencing analysis, fluorescence in situ hybridization, and a new
multiplex ligation-dependent probe amplification probe set in
patients with multiple osteochondromas. J Mol Diagn. 10:85–92.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kang Z, Peng F and Ling T: Mutation
screening of EXT genes in Chinese patients with multiple
osteochondromas. Gene. 506:298–300. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sarrión P, Sangorrin A, Urreizti R,
Delgado A, Artuch R, Martorell L, Armstrong J, Anton J, Torner F,
Vilaseca MA, et al: Mutations in the EXT1 and EXT2 genes in Spanish
patients with multiple osteochondromas. Sci Rep. 3:13462013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jamsheer A, Socha M, Sowińska-Seidler A,
Telega K, Trzeciak T and Latos-Bieleńska A: Mutational screening of
EXT1 and EXT2 genes in Polish patients with hereditary multiple
exostoses. J Appl Genet. 55:183–188. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li Y, Wang J, Tang J, Wang Z, Han B, Li N,
Yu T, Chen Y and Fu Q: Heterogeneous spectrum of EXT gene mutations
in Chinese patients with hereditary multiple osteochondromas.
Medicine (Baltimore). 97:e128552018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Guo X, Lin M, Yan W, Chen W and Hong G: A
novel splice mutation induces exon skipping of the EXT1 gene in
patients with hereditary multiple exostoses. Int J Oncol.
54:859–868. 2019.PubMed/NCBI
|
|
31
|
Yang A, Kim J, Jang JH, Lee C, Lee JE, Cho
SY and Jin DK: Identification of a novel mutation in EXT2 in a
fourth-generation Korean family with multiple osteochondromas and
overview of mutation spectrum. Ann Hum Genet. 83:160–170. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xia P, Xu H, Shi Q and Li D:
Identification of a novel frameshift mutation of the EXT2 gene in a
family with multiple osteochondroma. Oncol Lett. 11:105–110. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Collins JF, Bai L and Ghishan FK: The
SLC20 family of proteins: Dual functions as sodium-phosphate
cotransporters and viral receptors. Pflugers Arch. 447:647–652.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Forster IC, Hernando N, Biber J and Murer
H: Phosphate transporters of the SLC20 and SLC34 families. Mol
Aspects Med. 34:386–395. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Virkki LV, Biber J, Murer H and Forster
IC: Phosphate transporters: A tale of two solute carrier families.
Am J Physiol Renal Physiol. 293:F643–F654. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Solomon DH, Wilkins RJ, Meredith D and
Browning JA: Characterisation of inorganic phosphate transport in
bovine articular chondrocytes. Cell Physiol Biochem. 20:99–108.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Orfanidou T, Malizos KN, Varitimidis S and
Tsezou A: 1,25-Dihydroxyvitamin D(3) and extracellular inorganic
phosphate activate mitogen-activated protein kinase pathway through
fibroblast growth factor 23 contributing to hypertrophy and
mineralization in osteoarthritic chondrocytes. Exp Biol Med
(Maywood). 237:241–253. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sekine SI, Nishii K, Masaka T, Kurita H,
Inden M and Hozumi I: SLC20A2 variants cause dysfunctional
phosphate transport activity in endothelial cells induced from
Idiopathic Basal Ganglia Calcification patients-derived iPSCs.
Biochem Biophys Res Commun. 510:303–308. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kuroi Y, Akagawa H, Yoneyama T, Kikuchi A,
Maegawa T, Onda H and Kasuya H: Novel SLC20A2 mutation in a
Japanese pedigree with primary familial brain calcification. J
Neurol Sci. 399:183–185. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lemos RR, Ramos EM, Legati A, Nicolas G,
Jenkinson EM, Livingston JH, Crow YJ, Campion D, Coppola G and
Oliveira JR: Update and mutational analysis of SLC20A2: A major
cause of primary familial brain calcification. Hum Mutat.
36:489–495. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Beck-Cormier S, Lelliott CJ, Logan JG,
Lafont DT, Merametdjian L, Leitch VD, Butterfield NC, Protheroe HJ,
Croucher PI, Baldock PA, et al: Slc20a2, encoding the phosphate
transporter PiT2, is an important genetic determinant of bone
quality and strength. J Bone Miner Res. 34:1101–1114. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zalutskaya AA, Cox MK and Demay MB:
Phosphate regulates embryonic endochondral bone development. J Cell
Biochem. 108:668–674. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Khoshniat S, Bourgine A, Julien M, Weiss
P, Guicheux J and Beck L: The emergence of phosphate as a specific
signaling molecule in bone and other cell types in mammals. Cell
Mol Life Sci. 68:205–218. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu ES, Zalutskaya A, Chae BT, Zhu ED,
Gori F and Demay MB: Phosphate interacts with PTHrP to regulate
endochondral bone formation. Endocrinology. 155:3750–3756. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Magne D, Bluteau G, Faucheux C, Palmer G,
Vignes-Colombeix C, Pilet P, Rouillon T, Caverzasio J, Weiss P,
Daculsi G and Guicheux J: Phosphate is a specific signal for ATDC5
chondrocyte maturation and apoptosis-associated mineralization:
Possible implication of apoptosis in the regulation of endochondral
ossification. J Bone Miner Res. 18:1430–1442. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Milgram JW: The origins of osteochondromas
and enchondromas. A histopathologic study. Clin Orthop Relat Res.
174:264–284. 1983.
|
|
47
|
Benoist-Lasselin C, de Margerie E, Gibbs
L, Cormier S, Silve C, Nicolas G, LeMerrer M, Mallet JF, Munnich A,
Bonaventure J, et al: Defective chondrocyte proliferation and
differentiation in osteochondromas of MHE patients. Bone. 39:17–26.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Durigon R, Mitchell AL, Jones AW, Manole
A, Mennuni M, Hirst EM, Houlden H, Maragni G, Lattante S, Doronzio
PN, et al: LETM1 couples mitochondrial DNA metabolism and nutrient
preference. EMBO Mol Med. 10:e85502018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
McQuibban AG, Joza N, Megighian A,
Scorzeto M, Zanini D, Reipert S, Richter C, Schweyen RJ and
Nowikovsky K: A Drosophila mutant of LETM1, a candidate gene for
seizures in Wolf-Hirschhorn syndrome. Hum Mol Genet. 19:987–1000.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Schlickum S, Moghekar A, Simpson JC,
Steglich C, O'Brien RJ, Winterpacht A and Endele SU: LETM1, a gene
deleted in Wolf-Hirschhorn syndrome, encodes an evolutionarily
conserved mitochondrial protein. Genomics. 83:254–261. 2004.
View Article : Google Scholar : PubMed/NCBI
|