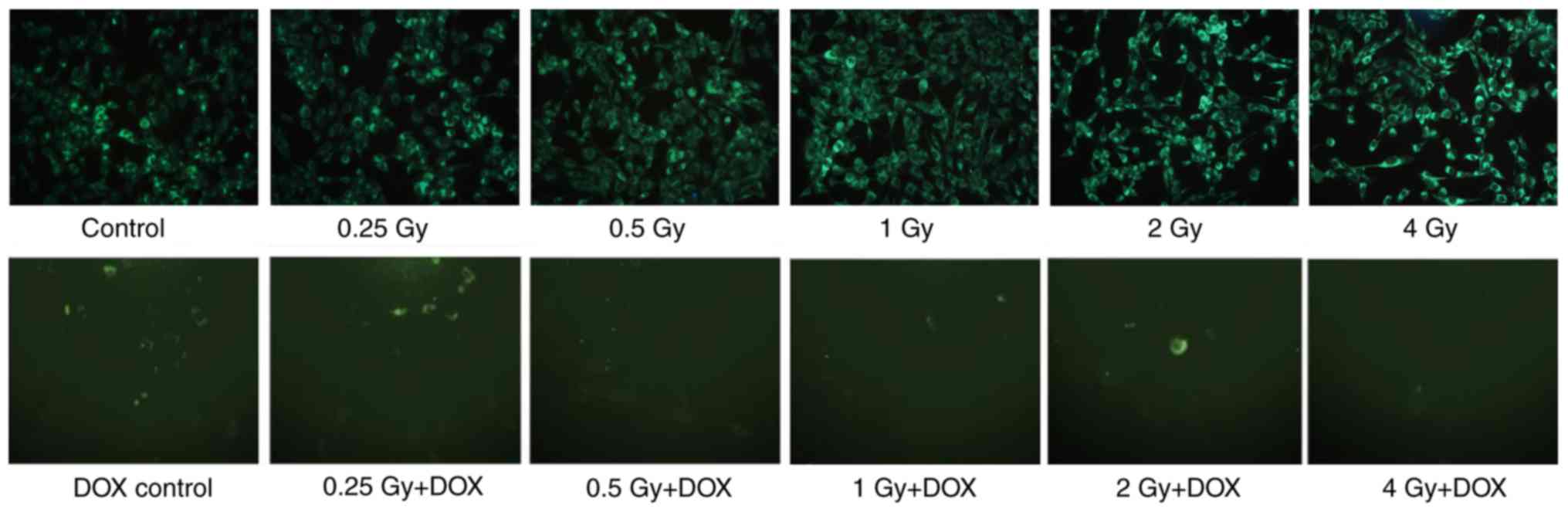
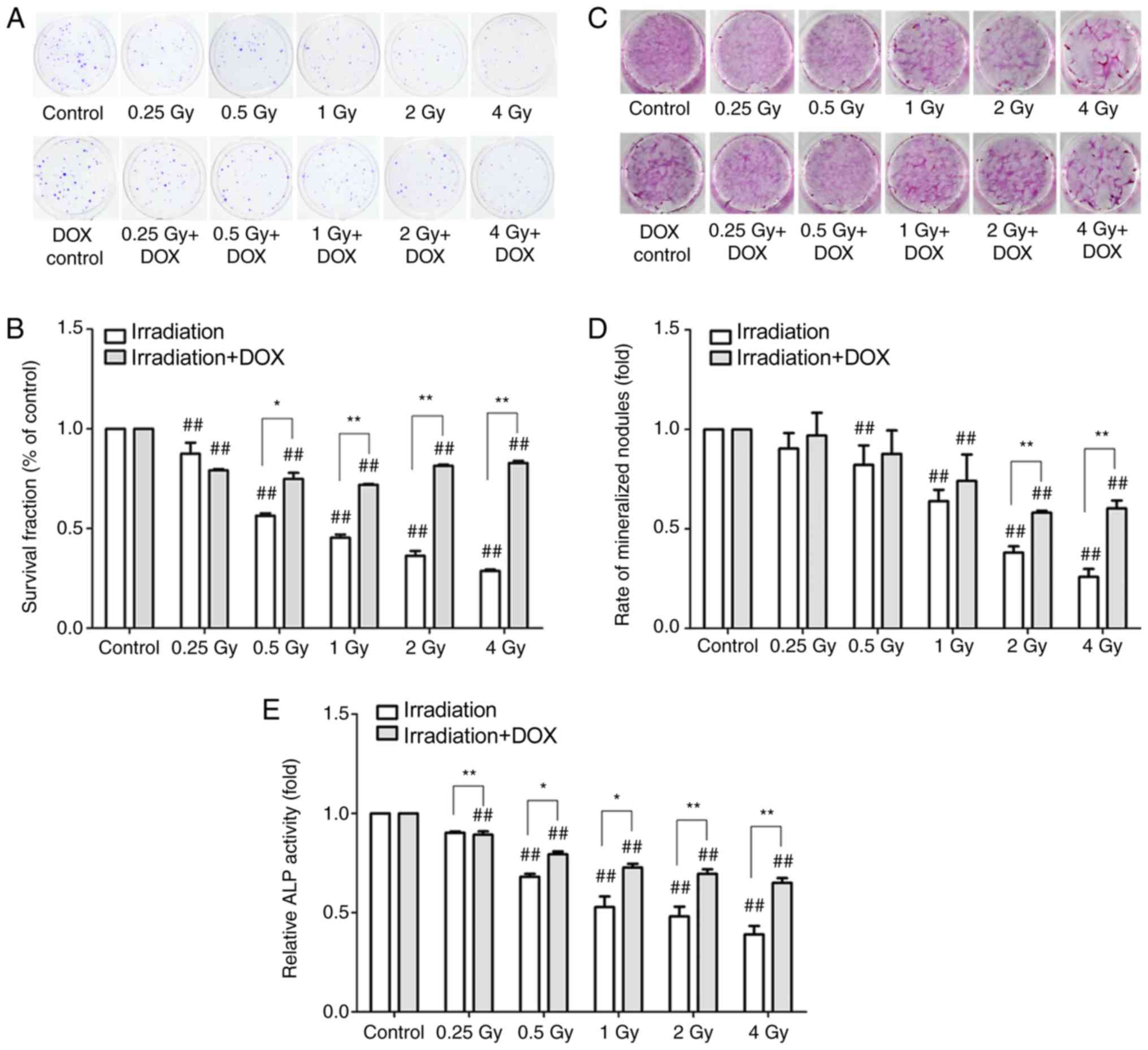
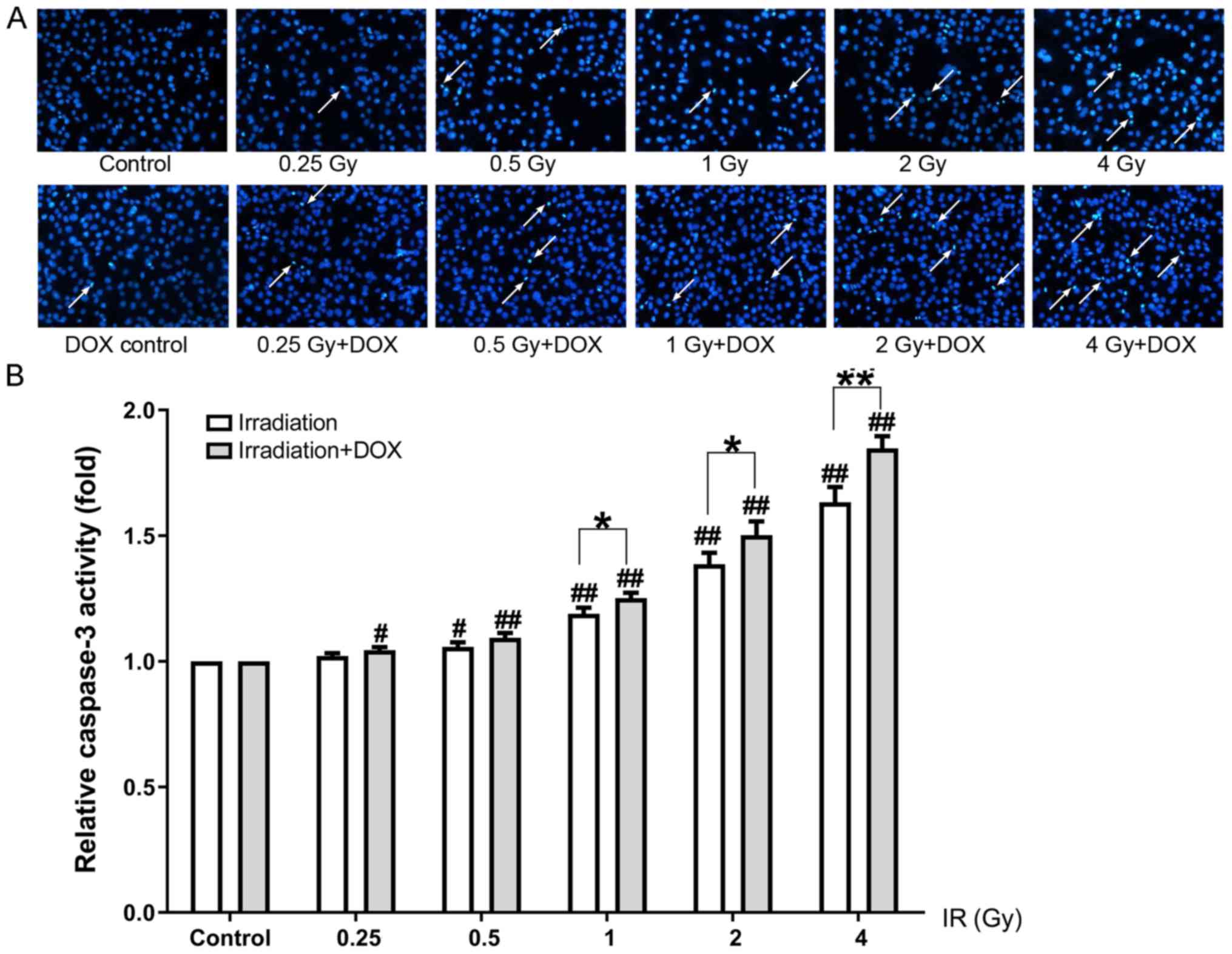
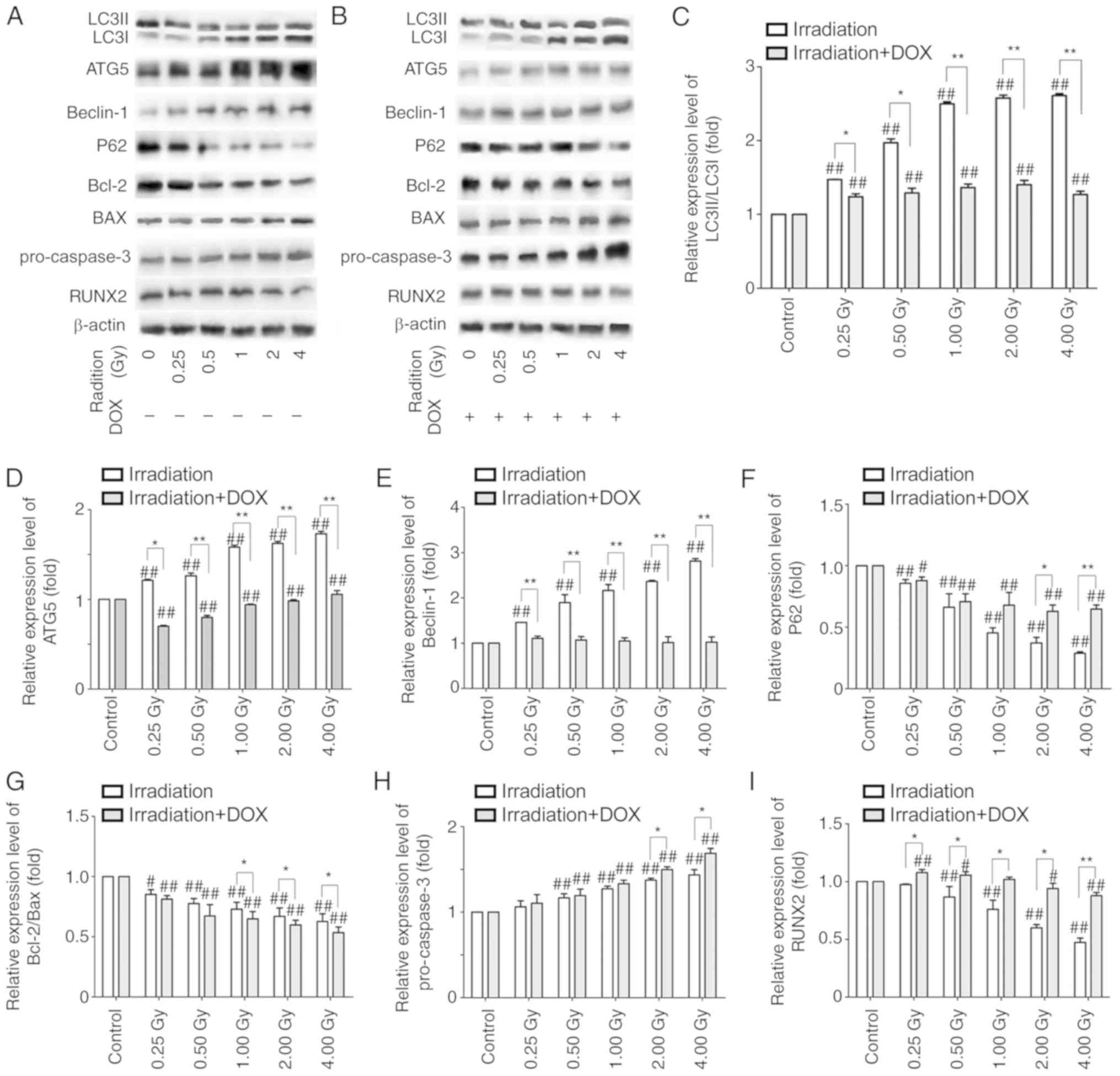
|
1
|
Moding EJ, Kastan MB and Kirsch DG:
Strategies for optimizing the response of cancer and normal tissues
to radiation. Nat Rev Drug Discov. 12:3473–542. 2013. View Article : Google Scholar
|
|
2
|
Cheriex KC, Nijhuis TH and Mureau MA:
Osteoradionecrosis of the jaws: A review of conservative and
surgical treatment options. J Reconstr Microsurg. 29:69–76. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chronopoulos A, Zarra T, Ehrenfeld M and
Otto S: Osteoradionecrosis of the jaws: Definition, epidemiology,
staging and clinical and radiological findings. A concise review.
Int Dent J. 68:22–30. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rice N, Polyzois I, Ekanayake K, Omer O
and Stassen LFA: The management of osteoradionecrosis of the jaws-a
review. Surgeon. 13:101–109. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mendenhall WM, Suárez C, Genden EM, de
Bree R, Strojan P, Langendijk JA, Mäkitie AA, Smee R, Eisbruch A,
Lee AWM, et al: Parameters associated with mandibular
osteoradionecrosis. Am J Clin Oncol. 41:1276–1280. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ktistakis NT and Tooze SA: Digesting the
expanding mechanisms of autophagy. Trends Cell Biol. 26:624–635.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tang BL: Autophagy in response to
environmental stresses: New monitoring perspectives. Ecol Indic.
60:453–459. 2016. View Article : Google Scholar
|
|
8
|
Ko A, Kanehisa A, Martins I, Senovilla L,
Chargari C, Dugue D, Mariño G, Kepp O, Michaud M, Perfettini JL, et
al: Autophagy inhibition radiosensitizes in vitro, yet reduces
radioresponses in vivo due to deficient immunogenic signalling.
Cell Death Differ. 21:92–99. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lu C and Xie C: Radiation-induced
autophagy promotes esophageal squamous cell carcinoma cell survival
via the LKB1 pathway. Oncol Rep. 35:3559–3565. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim KW, Moretti L, Mitchell LR, Jung DK
and Bo L: Combined Bcl-2/mammalian target of rapamycin inhibition
leads to enhanced radiosensitization via induction of apoptosis and
autophagy in non-small cell lung tumor xenograft model. Clin Cancer
Res. 15:6096–6105. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ondrej M, Cechakova L, Durisova K, Pejchal
J and Tichy A: To live or let die: Unclear task of autophagy in the
radiosensitization battle. Radiother Oncol. 119:265–275. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nollet M, Santucci-Darmanin S, Breuil V,
Al-Sahlanee R, Cros C, Topi M, Momier D, Samson M, Pagnotta S,
Cailleteau L, et al: Autophagy in osteoblasts is involved in
mineralization and bone homeostasis. Autophagy. 10:1965–1977. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu F, Fang F, Yuan H, Yang D, Chen Y,
Williams L, Goldstein SA, Krebsbach PH and Guan JL: Suppression of
autophagy by FIP200 deletion leads to osteopenia in mice through
the inhibition of osteoblast terminal differentiation. J Bone Miner
Res. 28:2414–2430. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Han Y, Zhang L, Xing Y, Zhang L, Chen X,
Tang P and Chen Z: Autophagy relieves the function inhibition and
apoptosis-promoting effects on osteoblast induced by
glucocorticoid. Int J Mol Med. 41:800–808. 2018.PubMed/NCBI
|
|
15
|
Li Y, Su J, Sun W, Cai L and Deng Z:
AMP-activated protein kinase stimulates osteoblast differentiation
and mineralization through autophagy induction. Int J Mol Med.
41:2535–2544. 2018.PubMed/NCBI
|
|
16
|
Hosokawa N, Hara Y and Mizushima N:
Generation of cell lines with tetracycline-regulated autophagy and
a role for autophagy in controlling cell size. FEBS Lett.
580:2623–2629. 2007. View Article : Google Scholar
|
|
17
|
Pankiv S, Clausen TH, Lamark T, Brech A,
Bruun JA, Outzen H, Øvervatn A, Bjørkøy G and Johansen T:
p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of
ubiquitinated protein aggregates by autophagy. J Biol Chem.
282:24131–24145. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li W, Wei S, Liu C, Song M, Wu H and Yang
Y: Regulation of the osteogenic and adipogenic differentiation of
bone marrow-derived stromal cells by extracellular uridine
triphosphate: The role of P2Y2 receptor and ERK1/2 signaling. Int J
Mol Med. 37:63–73. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Huang S, Wang S, Bian C, Yang Z, Zhou H,
Zeng Y, Li H, Han Q and Zhao RC: Upregulation of miR-22 promotes
osteogenic differentiation and inhibits adipogenic differentiation
of human adipose tissue-derived mesenchymal stem cells by
repressing HDAC6 protein expression. Stem Cells Dev. 21:2531–2540.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tian L, He LS, Soni B and Shang HT:
Myofibroblasts and their resistance to apoptosis: A possible
mechanism of osteoradionecrosis. Clin Cosmet Investig Dent.
4:21–27. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kook SH, Kim KA, Ji H, Lee D and Lee JC:
Irradiation inhibits the maturation and mineralization of
osteoblasts via the activation of Nrf2/HO-1 pathway. Mol Cell
Biochem. 410:255–266. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu W, Xu L, Chen M, Mao YT, Xie ZG, Wu SL
and Dong QR: The effects of low dose x-irradiation on osteoblastic
MC3T3-E1 cells in vitro. BMC Musculoskelet Disord. 13:942012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yang B, Tang Q, Post J, Zhou H, Huang XB,
Zhang XD, Wang Q, Sun YM and Fan FY: Effect of radiation on the
notch signaling pathway in osteoblasts. Int J Mol Med. 31:698–706.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pramojanee SN, Pratchayasakul W,
Chattipakorn N and Chattipakorn SC: Low-dose dental irradiation
decreases oxidative stress in osteoblastic MC3T3-E1 cells without
any changes in cell viability, cellular proliferation and cellular
apoptosis. Arch Oral Biol. 57:252–256. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang C, Blough E, Dai X, Olajide O,
Driscoll H, Leidy JW, July M, Triest WE and Wu M: Protective
effects of cerium oxide nanoparticles on MC3T3-E1 osteoblastic
cells exposed to X-ray irradiation. Cell Physiol Biochem.
38:1510–1519. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Szymczyk KH, Shapiro IM and Adams CS:
Ionizing radiation sensitizes bone cells to apoptosis. Bone.
34:148–156. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Alwood JS, Shahnazari M, Chicana B,
Schreurs AS, Kumar A, Bartolini A, Shirazi-Fard Y and Globus RK:
Ionizing radiation stimulates expression of pro-osteoclastogenic
genes in marrow and skeletal tissue. J Interferon Cytokine Res.
35:480–487. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang L, Meng H and Yang M: Autophagy
protects osteoblasts from advanced glycation end products-induced
apoptosis through intracellular reactive oxygen species. J Mol
Endocrinol. 56:291–300. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yumoto H, Hirao K, Tominaga T, Bando N,
Takahashi K and Matsuo T: Electromagnetic wave irradiation promotes
osteoblastic cell proliferation and up-regulates growth factors via
activation of the ERK1/2 and p38 MAPK pathways. Cell Physiol
Biochem. 35:601–615. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hao Y, Liu C, Huang J, Gu Y, Li H, Yang Z,
Liu J, Wang W and Li R: Ghrelin protects against depleted
uranium-induced apoptosis of MC3T3-E1 cells through oxidative
stress-mediated p38-mitogen-activated protein kinase pathway.
Toxicol Appl Pharmacol. 290:116–125. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kralova J, Dvorak M, Koc M and Kral V: p38
MAPK plays an essential role in apoptosis induced by
photoactivation of a novel ethylene glycol porphyrin derivative.
Oncogene. 27:3010–3020. 2008. View Article : Google Scholar : PubMed/NCBI
|