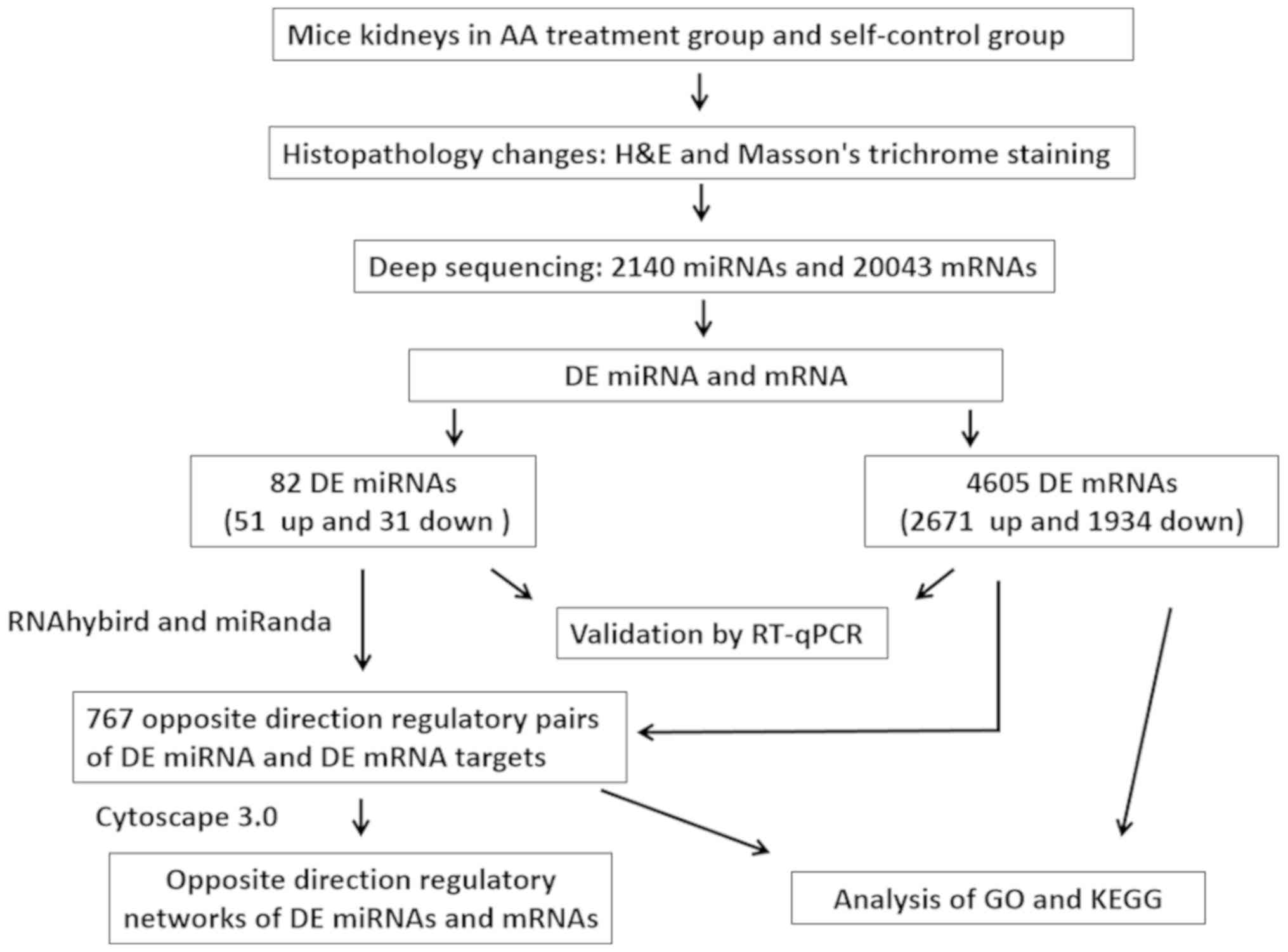
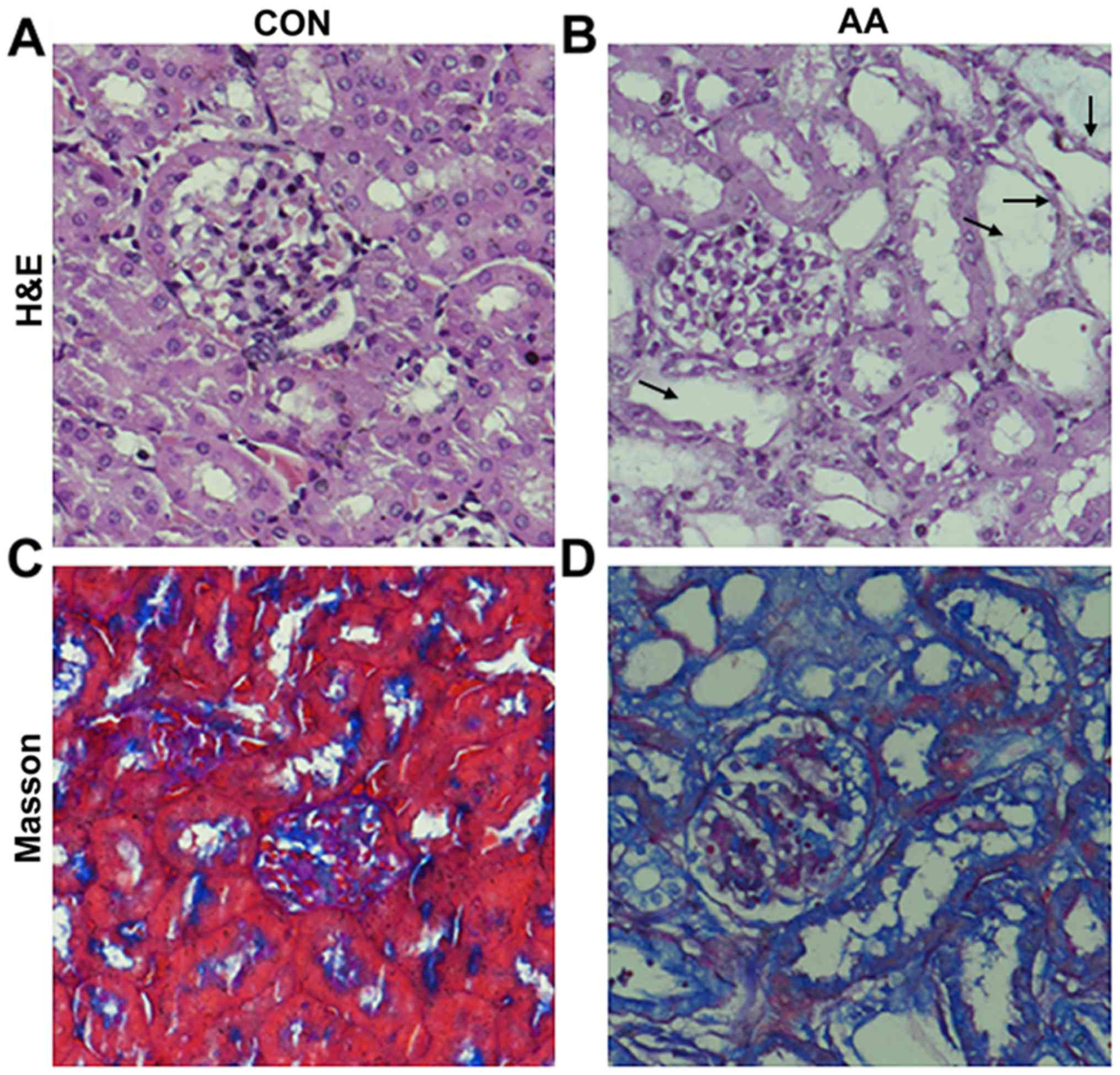
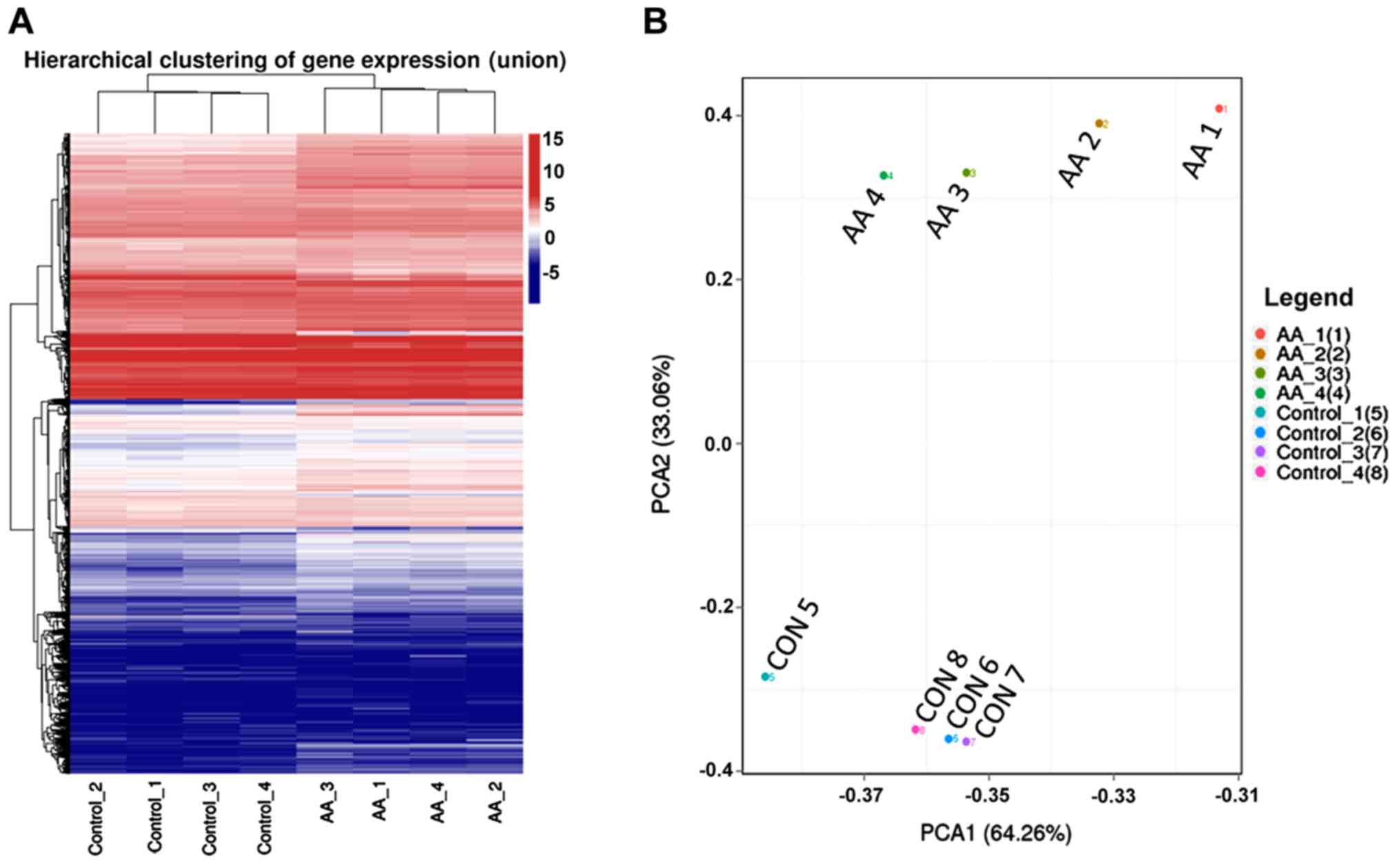
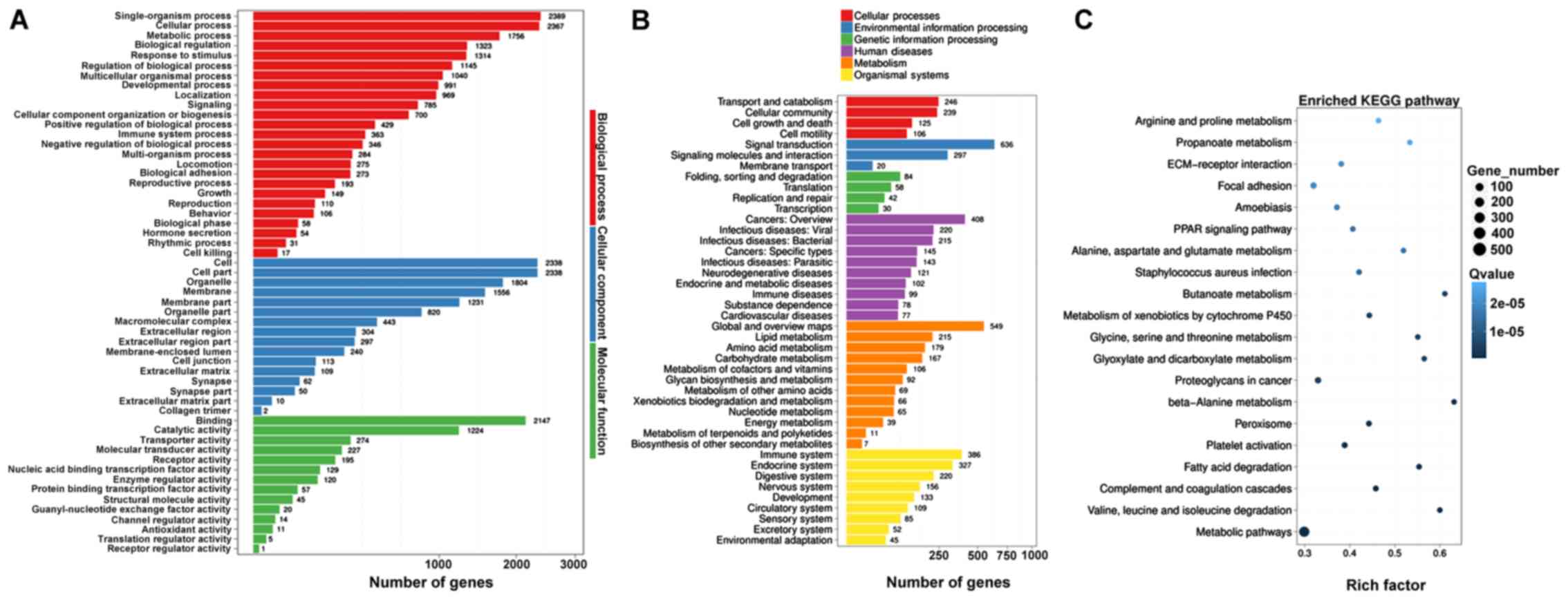
|
1
|
Schaneberg BT, Applequist WL and Khan IA:
Determination of aristolochic acid I and II in North American
species of Asarum and Aristolochia. Pharmazie.
57:3367–689. 2002.
|
|
2
|
Michl J, Jennings HM, Kite GC, Ingrouille
MJ, Simmonds MS and Heinrich M: Is aristolochic acid nephropathy a
widespread problem in developing countries? A case study of
Aristolochia indica L. in Bangladesh using an
ethnobotanical-phytochemical approach. J Ethnopharmacol.
149:235–244. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bara T Jr, Gurzu S, Sugimura H, Bara T,
Beleaua MA and Jung I: A systematic review of the possible
carcinogenic role of the aristolochic acid. Rom J Morphol Embryol.
58:41–44. 2017.PubMed/NCBI
|
|
4
|
Michl J, Ingrouille MJ, Simmonds MSJ and
Heinrich M: Naturally occurring aristolochic acid analogues and
their toxicities. Nat Prod Rep. 31:676–693. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yang L, Li X and Wang H: Possible
mechanisms explaining the tendency towards interstitial fibrosis in
aristolochic acid-induced acute tubular necrosis. Nephrol Dial
Transplant. 22:445–456. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jadot I, Decleves AE, Nortier J and Caron
N: An integrated view of aristolochic acid nephropathy: Update of
the literature. Int J Mol Sci. 18:2972017. View Article : Google Scholar
|
|
7
|
Humphreys BD: Mechanisms of renal
fibrosis. Annu Rev Physiol. 80:309–326. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rockey DC, Bell PD and Hill JA: Fibrosis-a
common pathway to organ injury and failure. N Engl J Med.
372:1138–1149. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yeh YC, Wei WC, Wang YK, Lin SC, Sung JM
and Tang MJ: Transforming growth factor-{beta}1 induces
Smad3-dependent {beta}1 integrin gene expression in
epithelial-to-mesenchymal transition during chronic
tubulointerstitial fibrosis. Am J Pathol. 177:1743–1754. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang Y, Feng XJ, Liu XY, Wang LH and Zheng
GP: The effect of transforming growth factor β(1) in the transition
of bone marrow-derived macrophages into myofibroblasts during renal
fibrosis. Zhonghua Nei Ke Za Zhi. 56:610–613. 2017.(In Chinese).
PubMed/NCBI
|
|
11
|
Gebert LFR and MacRae IJ: Regulation of
microRNA function in animals. Nat Rev Mol Cell Biol. 20:21–37.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bushati N and Cohen SM: MicroRNA
functions. Annu Rev Cell Dev Biol. 23:175–205. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chung AC and Lan HY: MicroRNAs in renal
fibrosis. Front Physiol. 6:502015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang B and Ricardo S: Role of microRNA
machinery in kidney fibrosis. Clin Exp Pharmacol Physiol.
41:543–550. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tang C, Xie Y and Yan W: 1 AASRA: An
anchor alignment-based small RNA. bioRxiv. 2017.(Epub ahead for
print).
|
|
16
|
Nawrocki EP and Eddy SR: Infernal 1.1:
100-fold faster RNA homology searches. Bioinformatics.
29:2933–2935. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim D, Langmead B and Salzberg SL: HISAT:
A fast spliced aligner with low memory requirements. Nat Methods.
12:357–360. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kruger J and Rehmsmeier M: RNAhybrid:
microRNA target prediction easy, fast and flexible. Nucleic Acids
Res. 34((Web Server Issue)): W451–W454. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
John B, Enright AJ, Aravin A, Tuschl T,
Sander C and Marks DS: Human microRNA targets. PLoS Biol.
2:e3632004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mi H, Muruganujan A, Ebert D, Huang X and
Thomas PD: PANTHER version 14: More genomes, a new PANTHER GO-slim
and improvements in enrichment analysis tools. Nucleic Acids Res.
47D:D419–D426. 2019. View Article : Google Scholar
|
|
21
|
Kanehisa M, Furumichi M, Tanabe M, Sato Y
and Morishima K: KEGG: New perspectives on genomes, pathways,
diseases and drugs. Nucleic Acids Res. 45D:D353–D361. 2017.
View Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Horowitz JC and Thannickal VJ: Mechanisms
for the resolution of organ fibrosis. Physiology (Bethesda).
34:43–55. 2019.PubMed/NCBI
|
|
24
|
Schafer S, Viswanathan S, Widjaja AA, Lim
WW, Moreno-Moral A, DeLaughter DM, Ng B, Patone G, Chow K, Khin E,
et al: IL-11 is a crucial determinant of cardiovascular fibrosis.
Nature. 552:110–115. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li Z, Qin T, Wang K, Hackenberg M, Yan J,
Gao Y, Yu LR, Shi L, Su Z and Chen T: Integrated microRNA, mRNA,
and protein expression profiling reveals microRNA regulatory
networks in rat kidney treated with a carcinogenic dose of
aristolochic acid. BMC Genomics. 16:3652015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Meng F, Li Z, Yan J, Manjanatha M, Shelton
S, Yarborough S and Chen T: Tissue-specific microRNA responses in
rats treated with mutagenic and carcinogenic doses of aristolochic
acid. Mutagenesis. 29:357–365. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li R, Chung AC, Dong Y, Yang W, Zhong X
and Lan HY: The microRNA miR-433 promotes renal fibrosis by
amplifying the TGF-β/Smad3-Azin1 pathway. Kidney Int. 84:1129–1144.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bijkerk R, de Bruin RG, van Solingen C,
van Gils JM, Duijs JM, van der Veer EP, Rabelink TJ, Humphreys BD
and van Zonneveld AJ: Silencing of microRNA-132 reduces renal
fibrosis by selectively inhibiting myofibroblast proliferation.
Kidney Int. 89:1268–1280. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhou SG, Zhang W, Ma HJ, Guo ZY and Xu Y:
Silencing of lncRNA TCONS_00088786 reduces renal fibrosis through
miR-132. Eur Rev Med Pharmacol Sci. 22:166–173. 2018.PubMed/NCBI
|
|
30
|
Muralidharan J, Ramezani A, Hubal M,
Knoblach S, Shrivastav S, Karandish S, Scott R, Maxwell N, Ozturk
S, Beddhu S, et al: Extracellular microRNA signature in chronic
kidney disease. Am J Physiol Renal Physiol. 312:F982–F991. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu Y: Cellular and molecular mechanisms
of renal fibrosis. Nat Rev Nephrol. 7:684–696. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gaugg MT, Engler A, Bregy L,
Nussbaumer-Ochsner Y, Eiffert L, Bruderer T, Zenobi R, Sinues P and
Kohler M: Molecular breath analysis supports altered amino acid
metabolism in idiopathic pulmonary fibrosis. Respirology.
24:437–444. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Park JS, Choi HI, Kim DH, Kim CS, Bae EH,
Ma SK and Kim SW: Alpha-lipoic acid attenuates p-cresyl
sulfate-induced renal tubular injury through suppression of
apoptosis and autophagy in human proximal tubular epithelial cells.
Biomed Pharmacother. 112:1086792019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Dong Q, Jie Y, Ma J, Li C, Xin T and Yang
D: Renal tubular cell death and inflammation response are regulated
by the MAPK-ERK-CREB signaling pathway under hypoxia-reoxygenation
injury. J Recept Signal Transduct Res. 39:383–391. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kramann R: Hedgehog Gli signalling in
kidney fibrosis. Nephrol Dial Transplant. 31:1989–1995. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhou D, Tan RJ and Liu Y: Sonic hedgehog
signaling in kidney fibrosis: A master communicator. Sci China Life
Sci. 59:920–929. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fabian SL, Penchev RR, St-Jacques B, Rao
AN, Sipila P, West KA, McMahon AP and Humphreys BD: Hedgehog-Gli
pathway activation during kidney fibrosis. Am J Pathol.
180:1441–1453. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kai Y, Yoneyama H, Koyama J, Hamada K,
Kimura H and Matsushima K: Treatment with chondroitinase ABC
alleviates bleomycin-induced pulmonary fibrosis. Med Mol Morphol.
40:128–140. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Vermeulen Z, Hervent AS, Dugaucquier L,
Vandekerckhove L, Rombouts M, Beyens M, Schrijvers DM, De Meyer
GRY, Maudsley S, De Keulenaer GW and Segers VFM: Inhibitory actions
of the NRG-1/ErbB4 pathway in macrophages during tissue fibrosis in
the heart, skin, and lung. Am J Physiol Heart Circ Physiol.
313:H934–H945. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Scheving LA, Zhang X, Threadgill DW and
Russell WE: Hepatocyte ERBB3 and EGFR are required for maximal
CCl4-induced liver fibrosis. Am J Physiol Gastrointest Liver
Physiol. 311:G807–G816. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Essam RM, Ahmed LA, Abdelsalam RM and
El-Khatib AS: Phosphodiestrase-1 and 4 inhibitors ameliorate liver
fibrosis in rats: Modulation of cAMP/CREB/TLR4 inflammatory and
fibrogenic pathways. Life Sci. 222:245–254. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Han K, Zhang Y and Yang Z: Cilostazol
protects rats against alcohol-induced hepatic fibrosis via
suppression of TGF-β1/CTGF activation and the cAMP/Epac1 pathway.
Exp Ther Med. 17:2381–2388. 2019.PubMed/NCBI
|
|
43
|
Kanai AJ, Konieczko EM, Bennett RG, Samuel
CS and Royce SG: Relaxin and fibrosis: Emerging targets,
challenges, and future directions. Mol Cell Endocrinol. 487:66–74.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zheng G, Cai J, Chen X, Chen L, Ge W, Zhou
X and Zhou H: Relaxin ameliorates renal fibrosis and expression of
endothelial cell transition markers in rats of
isoproterenol-induced heart failure. Biol Pharm Bull. 40:960–966.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Newbury LJ, Wang JH, Hung G, Hendry BM and
Sharpe CC: Inhibition of Kirsten-Ras reduces fibrosis and protects
against renal dysfunction in a mouse model of chronic folic acid
nephropathy. Sci Rep. 9:140102019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhou G, Li J, Zeng T, Yang P and Li A: The
regulation effect of WNT-RAS signaling in hypothalamic
paraventricular nucleus on renal fibrosis. J Nephrol. 33:289–297.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang J, Zhu H, Huang L, Zhu X, Sha J, Li
G, Ma G, Zhang W, Gu M and Guo Y: Nrf2 signaling attenuates
epithelial-to-mesenchymal transition and renal interstitial
fibrosis via PI3K/Akt signaling pathways. Exp Mol Pathol.
111:1042962019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Dou F, Liu Y, Liu L, Wang J, Sun T, Mu F,
Guo Q, Guo C, Jia N, Liu W, et al: Aloe-Emodin ameliorates renal
fibrosis via inhibiting PI3K/Akt/mTOR signaling pathway in vivo and
in vitro. Rejuvenation Res. 22:218–229. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Sun L, Xu T, Chen Y, Qu W, Sun D, Song X,
Yuan Q and Yao L: Pioglitazone attenuates kidney fibrosis via
miR-21-5p modulation. Life Sci. 232:1166092019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Tian C, Wang Y, Chang H, Li J and La X:
Spleen-kidney supplementing formula alleviates renal fibrosis in
diabetic rats via TGF-β1-miR-21-PTEN signaling pathway. Evid Based
Complement Alternat Med. 2018:38243572018. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Tang CR, Luo SG, Lin X, Wang J and Liu Y:
Silenced miR-21 inhibits renal interstitial fibrosis via targeting
ERK1/2 signaling pathway in mice. Eur Rev Med Pharmacol Sci. 23 (3
Suppl):S110–S116. 2019.
|
|
52
|
McClelland AD, Herman-Edelstein M, Komers
R, Jha JC, Winbanks CE, Hagiwara S, Gregorevic P, Kantharidis P and
Cooper ME: miR-21 promotes renal fibrosis in diabetic nephropathy
by targeting PTEN and SMAD7. Clin Sci (Lond). 129:1237–1249. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wang J, Gao Y, Ma M, Li M, Zou D, Yang J,
Zhu Z and Zhao X: Effect of miR-21 on renal fibrosis by regulating
MMP-9 and TIMP1 in kk-ay diabetic nephropathy mice. Cell Biochem
Biophys. 67:537–546. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zhong X, Chung AC, Chen HY, Meng XM and
Lan HY: Smad3-mediated upregulation of miR-21 promotes renal
fibrosis. J Am Soc Nephrol. 22:1668–1681. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhou H, Qiu ZZ, Yu ZH, Gao L, He JM, Zhang
ZW and Zheng J: Paeonol reverses promoting effect of the
HOTAIR/miR-124/Notch1 axis on renal interstitial fibrosis in a rat
model. J Cell Physiol. 234:14351–14363. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhou H, Gao L, Yu ZH, Hong SJ, Zhang ZW
and Qiu ZZ: lncRNA HOTAIR promotes renal interstitial fibrosis by
regulating Notch1 pathway via the modulation of miR-124. Nephrology
(Carlton). 24:472–480. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Disayabutr S, Kim EK, Cha SI, Green G,
Naikawadi RP, Jones KD, Golden JA, Schroeder A, Matthay MA, Kukreja
J, et al: miR-34 miRNAs regulate cellular senescence in type II
alveolar epithelial cells of patients with idiopathic pulmonary
fibrosis. PLoS One. 11:e1583672016. View Article : Google Scholar
|
|
58
|
Li WQ, Chen C, Xu MD, Guo J, Li YM, Xia
QM, Liu HM, He J, Yu HY and Zhu L: The rno-miR-34 family is
upregulated and targets ACSL1 in dimethylnitrosamine-induced
hepatic fibrosis in rats. FEBS J. 278:1522–1532. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Sun Y, Wang H, Li Y, Liu S, Chen J and
Ying H: miR-24 and miR-122 negatively regulate the transforming
growth Factor-β/smad signaling pathway in skeletal muscle fibrosis.
Mol Ther Nucleic Acids. 11:528–537. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Lou G, Yang Y, Liu F, Ye B, Chen Z, Zheng
M and Liu Y: miR-122 modification enhances the therapeutic efficacy
of adipose tissue-derived mesenchymal stem cells against liver
fibrosis. J Cell Mol Med. 21:2963–2973. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Halasz T, Horvath G, Par G, Werling K,
Kiss A, Schaff Z and Lendvai G: miR-122 negatively correlates with
liver fibrosis as detected by histology and FibroScan. World J
Gastroenterol. 21:7814–7823. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Li N, Wang LJ, Xu WL, Liu S and Yu JY:
MicroRNA3795p suppresses renal fibrosis by regulating the
LIN28/let7 axis in diabetic nephropathy. Int J Mol Med.
44:1619–1628. 2019.PubMed/NCBI
|
|
63
|
Cevikbas F, Schaefer L, Uhlig P, Robenek
H, Theilmeier G, Echtermeyer F and Bruckner P: Unilateral
nephrectomy leads to up-regulation of syndecan-2- and
TGF-beta-mediated glomerulosclerosis in syndecan-4 deficient male
mice. Matrix Biol. 27:42–52. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Xia Y, Jin X, Yan J, Entman ML and Wang Y:
CXCR6 plays a critical role in angiotensin II-induced renal injury
and fibrosis. Arterioscler Thromb Vasc Biol. 34:1422–1428. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Uil M, Scantlebery AML, Butter LM, Larsen
PWB, de Boer OJ, Leemans JC, Florquin S and Roelofs JJTH: Combining
streptozotocin and unilateral nephrectomy is an effective method
for inducing experimental diabetic nephropathy in the ‘resistant’
C57Bl/6J mouse strain. Sci Rep. 8:55422018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Esposito C, He CJ, Striker GE, Zalups RK
and Striker LJ: Nature and severity of the glomerular response to
nephron reduction is strain-dependent in mice. Am J Pathol.
154:891–897. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Liu BC, Tang TT and Lv LL: How tubular
epithelial cell injury contributes to renal fibrosis. Adv Exp Med
Biol. 1165:233–252. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Liu BC, Tang TT, Lv LL and Lan HY: Renal
tubule injury: A driving force toward chronic kidney disease.
Kidney Int. 93:568–579. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Youl ENH, Husson C, El Khattabi C, El Mere
S, Decleves AE, Pochet S, Nortier J and Antoine MH:
Characterization of cytotoxic effects of aristolochic acids on the
vascular endothelium. Toxicol In Vitro. 65:1048112020. View Article : Google Scholar : PubMed/NCBI
|