Introduction
As a serious chronic systemic bone disease,
osteoporosis (OP) involves excessive bone resorption and decreased
bone formation (1), resulting from
an imbalance induced by inhibition of osteogenic differentiation
(2), such as abnormal bone
mesenchymal stem cell (BMSC) differentiation, the precursor cells
of osteoblasts (2). Due to their
osteogenic differentiation ability, BMSCs were indicated to be a
suitable cell type for the repair and remodeling of bones (3). Therefore, it is of significance to
induce BMSC differentiation into bone tissues for OP treatment, and
the mechanism to enhance osteogenic differentiation has received
increasing attention.
Long non-coding RNAs (lncRNAs) are non-coding RNAs,
>200 nucleotides in length, that regulate different processes
via various molecular mechanisms (4). It has been reported that lncRNAs
participate in regulating osteogenic activity. For instance, KCNQ1
opposite strand/antisense transcript 1 (KCNQ1OT1) regulates
osteogenic differentiation by sponging microRNA (miRNA/miR)-214
(5), whereas MALAT1 promotes
osterix (OSX) expression to regulate osteogenic differentiation by
sponging miRNA-143 (6). Small
nucleolar RNA host gene 1 (SNHG1) is upregulated in numerous types
of cancer (7,8), and can attenuate heterogeneous
nuclear ribonucleoprotein C-p53 protein interactions to enhance the
stability of p53 (4). Besides,
elevated SNHG1 can contribute to the progression of non-small cell
lung cancer via inhibition of miR-101-3p (9). Moreover, SNHG1 inhibits BMSC
osteogenic differentiation by modulating the p38 MAPK signaling
pathway (10). However, the
molecular mechanism underlying lncRNA SNHG1-mediated regulation of
BMSC differentiation is not completely understood.
miRNAs are 18–25 base-long endogenous RNAs that are
involved in regulating multiple physiological biological processes
(11). miR-101 is significantly
upregulated during human BMSC osteogenic differentiation, and it
can target the enhancer of zeste 2 polycomb repressive complex 2
subunit/Wnt/β-catenin signaling pathway to promote human BMSC
osteogenic differentiation (12).
Bioinformatics analysis has revealed that SNHG1 has a miR-101
binding region (13); however,
whether SNHG1 regulates BMSC osteogenic differentiation by binding
with miR-101 is not completely understood.
The entire osteoblastic lineage is affected by Wnt
signaling, and Wnt/β-catenin signaling can indirectly repress
osteoclast differentiation and bone resorption (14,15).
Dickkopf 1 (DKK1) is a Wnt signaling inhibitor, which inhibits the
canonical Wnt signaling pathway by binding to the Wnt complex
receptor Low-density lipoprotein receptor-related protein 5/6
(16). By bioinformatics, it was
also predicted that there may be a binding site between miR-101 and
DKK1 (17). Moreover, to the best
of our knowledge, no previous study has focused on the regulation
of BMSC osteogenic differentiation via the miR-101/DKK1/Wnt axis.
It was hypothesized that the SNHG1/miR-101/DKK1 axis served a
regulatory role in BMSC osteogenic differentiation.
Materials and methods
Cell culture
Human BMSCs (cat. no. HUXMA-01001; Cyagen
Biosciences) were cultured in DMEM (HyClone; Cytiva) containing 10%
FBS (Gibco; Thermo Fisher Scientific, Inc.), 100 U/ml penicillin
and 100 U/ml streptomycin. Cells were cultured in a humidified
atmosphere containing 5% CO2 at 37°C.
Induction of osteogenic
differentiation
To induce osteogenic differentiation, cells were
cultured in osteogenic differentiation medium consisting of DMEM
with 10% FBS, dexamethasone (100 nM; Sigma-Aldrich; Merck KGaA),
ascorbic acid 2-phosphate (200 µM; Sigma-Aldrich; Merck KGaA) and
β-glycerophosphate (10 mM; Sigma-Aldrich; Merck KGaA) for 15 days
at 37°C. The medium was changed every 3 days.
Cell transfection
pcDNA-SNHG1and miR-101 mimics were constructed and
purchased from Shanghai GenePharma Co., Ltd.. Cells were
transfected with 200 ng/well pcDNA-SNHG1, pcDNA empty vector and 50
nM miR-101 mimics (UACAGUACUGUGAUAACUGAA) and its scramble control
(mimics-NC) (UCACAACCUCCUAGAAAGAGUAGA) using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol when the
confluence of the cells reached 70–80%. Then, cells were collected
at 48 h after transfection for further use.
Luciferase activity assay
The predicted miR-101 binding site in SNHG1 and
DKK1, as well as the mutated regions, were amplified and cloned
into the pmirGLO plasmid (Promega Corporation). Then,
1×104/well 293T cells were co-transfected with 800 ng
luciferase reporter plasmid and 50 pmol miRNA mimics or
mimics-negative control (NC) using Lipofectamine 3000 in a 24-well
plate. At 48 h post-transfection, the Dual-Luciferase Reporter
Assay system (Promega Corporation) was used to examine relative
luciferase activity. Fluorescence signals were normalized to those
of Renilla luciferase activity, and are presented as the
mean ± SD of three independent experiments.
Alkaline phosphatase (ALP) activity
detection
ALP staining was performed using an ALP activity
colorimetric assay kit (BioVision, Inc.) following the
manufacturer's instructions. Briefly, cells were fixed with 70%
ethanol at room temperature for 30 min and washed with PBST. Then,
50 µl 5 mM P-nitrophenyl phosphate (p-NPP) solution was added to
test and control samples, and 10 µl ALP enzyme solution was added
to each p-NPP Standard wells and incubated at room temperature for
15–30 min. Cells were gently washed three times with cold PBS.
Subsequently, cells were lysed with 1% Triton X-100 (Sigma-Aldrich;
Merck KGaA) and washed with deionized water. The absorbance of
p-nitrophenol at a wavelength of 405 nm was measured using a
microplate reader (Bio-Rad Laboratories, Inc.) according to the
manufacturer's instructions.
Alizarin red staining (ARS)
ARS (Cyagen Biosciences, Inc.) was conducted to
assess mineral deposition. Briefly, cells were fixed with 4%
paraformaldehyde at room temperature for 15 min and stained with 2%
Alizarin Red S (pH 4.2; Sigma-Aldrich; Merck KGaA) at room
temperature in deionized water for 20 min. Subsequently, cells were
washed with PBS and observed under a light microscope (Leica
Microsystems GmbH, magnification, ×100).
Reverse transcription-quantitative PCR (RT-qPCR).
Total RNA was extracted from cells using TRIzol®
(Invitrogen; Thermo Fisher Scientific, Inc.). Total RNA was reverse
transcribed into cDNA using the ImProm-II Reverse Transcription
system (Promega Corporation) following the manufacturer's
instructions. Briefly, the reaction was incubated at 37°C for 1 h
followed by 95°C for 5 min and 4°C for 5 min Subsequently, qPCR was
performed on days 0, 7 and 15 of osteogenic differentiation using
TransStart Eco Green qPCR SuperMix (TransGen Biotech Co., Ltd.) and
the following thermocycling conditions: Initial denaturation at
95°C for 10 min; 40 cycles of denaturation/annealing/extension at
95°C for 15 sec; 60°C for 30 sec and followed by final extension at
60°C for 35 sec. The following primer sequences were used for qPCR:
SNHG1 forward, 5′-ACTCCACTTCGTGTCTGTTCC−3′ and reverse,
5′-TGAAGAGCAAGGCCCTGAAT-3′; miR-101 forward,
5′-GCGCGCATACAGTACTGTGATA-3′ and reverse,
5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACTTCAGT-3′; DKK1
forward, 5′-TGCCCCGGGAATTACTGCAAA−3′ and reverse,
5′-CTGGAATACCCATCCAAGGTGCTA−3′; RUNX family transcription factor 2
(RUNX2) forward, 5′-CGGAATGCCTCTGCTGTTAT-3′ and reverse,
5′-TTCCCGAGGTCCATCTACTG-3′; osteopontin (OPN) forward,
5′-GATGGCCGAGGTGATAGTGT-3′ and reverse, 5′-GTGGGTTTCAGCACTCTGGT-3′;
osteocalin (OCN) forward, 5′-GGCAGCGAGGTAGTGAAGAG-3′ and reverse,
5′-CTAGACCGGGCCGTAGAAG-3′; GAPDH forward, 5′-CCAGGTGGTCTCCTCTGA−3′
and reverse, 5′-GCTGTAGCCAAATCGTTGT-3′; and U6 forward,
5′-CTCGCTTCGGCAGCACA-3′ and reverse, 5′-AACGCTTCACGAATTTGCGT-3′.
miRNA and mRNA expression levels were quantified using
2−∆∆Cq method and normalized to the internal reference
genes U6 and GAPDH, respectively (18).
Western blotting
Total protein was extracted from cells using RIPA
lysis buffer containing protease (Beyotime Institute of
Biotechnology). Total protein was quantified using a BCA protein
assay (Thermo Fisher Scientific, Inc.). A total of 100 µg
protein/lane was separated via 10% SDS-PAGE and transferred to
nitrocellulose membranes. After blocking with TBST buffer
containing 5% non-fat milk (Thermo Fisher Scientific, Inc.) for 1 h
at room temperature, the membranes were incubated with specific
primary antibodies at 4°C overnight targeted against: DKK1
(1:1,000; cat. no. ab109416, Abcam), β-catenin (1:1,000; cat. no.
ab184919; Abcam), RUNX2 (1:1,000; cat. no. ab192256; Abcam), OCN
(1:1,000; cat. no. ab133612; Abcam), OPN (1:1,000; cat. no.
ab214050; Abcam), OSX (1:500; cat. no. ab209484; Abcam), collagen
type I α1 chain (COL1a1; 1:1,000; cat. no. ab34710; Abcam) and
β-actin (1:1,000; cat. no. ab8227; Abcam). Following primary
incubation, the membranes were incubated with goat anti-Rabbit IgG
horseradish-peroxidase-conjugated secondary antibodies (1:5,000;
cat. no. G-21234; Thermo Fisher Scientific, Inc.) for 1 h at room
temperature. Protein bands were visualized using chemiluminescence
ECL detection system (EMD Millipore) and the Bio-Rad XRS
chemiluminescence detection system (Bio-Rad Laboratories, Inc.).
β-actin was used as the loading control. The proteins were
quantified using Quantity One software v4.62 (Bio-Rad Laboratories,
Inc.).
Statistical analysis
Data are presented as the mean ± standard deviation.
The unpaired Student's t-test was used to compare the difference
between two groups. One-way ANOVA followed by Tukey's post hoc test
was used to compare differences among multiple groups. Statistical
analyses were conducted using SPSS software (version 18.0; SPSS,
Inc.). P<0.05 was considered to indicate a statistically
significant difference. All experiments were performed in
triplicate and carried out at least three times.
Results
Expression levels of SNHG1, miR-101
and DKK1 during BMSC osteogenic differentiation
After induction of osteogenic differentiation, the
expression of SNHG1, miR-101, DKK1 and osteogenesis-related genes,
including RUNX2, OCN and OPN, on day 0, 7 and 15 were detected via
RT-qPCR. During the process of osteogenic differentiation, the
expression of miR-101 and the osteogenesis-related genes were
upregulated, whereas SNHG1 and DKK1 were downregulated in a
time-dependent manner (Fig. 1A-F).
Moreover, ALP activity and mineralization assays indicated an
osteoblastic phenotype (Fig. 1G and
H). Furthermore, the protein expression levels of the
osteogenesis-related genes were also upregulated in a
time-dependent manner during the process of osteogenic
differentiation, whereas DKK1 protein expression levels were
downregulated in a time-dependent manner (Fig. 1I and J). Collectively, the results
indicated that SNHG1, miR-101 and DKK1 served a regulatory role in
osteogenic differentiation.
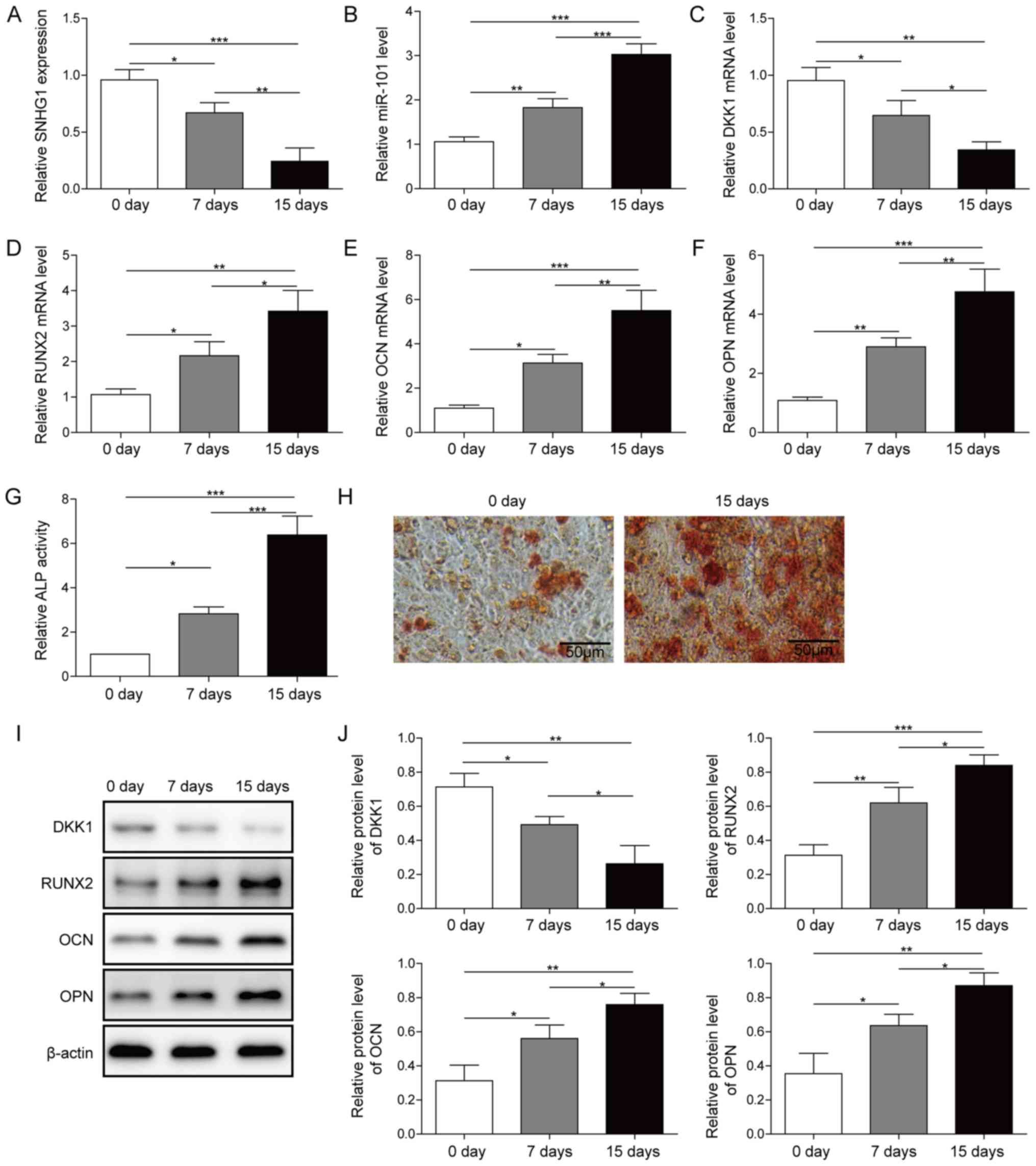 | Figure 1.Expression of SNHG1, DKK1 and miR-101
during osteogenic differentiation. Expression of (A) SNHG1, (B)
miR-101, (C) DKK1, (D) RUNX2, (E) OCN and (F) OPN in osteogenic
differentiation medium-treated BMSCs at different time points. (G)
ALP activity and (H) Alizarin Red staining in osteogenic
differentiation medium-treated BMSCs at different time points.
Protein expression levels were (I) determined by western blotting
and (J) semi-quantified for DKK1, RUNX2, OPN and OCN in osteogenic
differentiation medium-treated BMSCs at different time points.
*P<0.05, **P<0.01 and ***P<0.001. SNHG1, small nucleolar
RNA host gene 1; DKK1, dickkopf WNT signaling pathway inhibitor 1;
miR, microRNA; RUNX2, RUNX family transcription factor 2; OCN,
osteocalin; OPN, osteopontin; ALP, alkaline phosphatase; BMSC, bone
mesenchymal stem cell; d, days. |
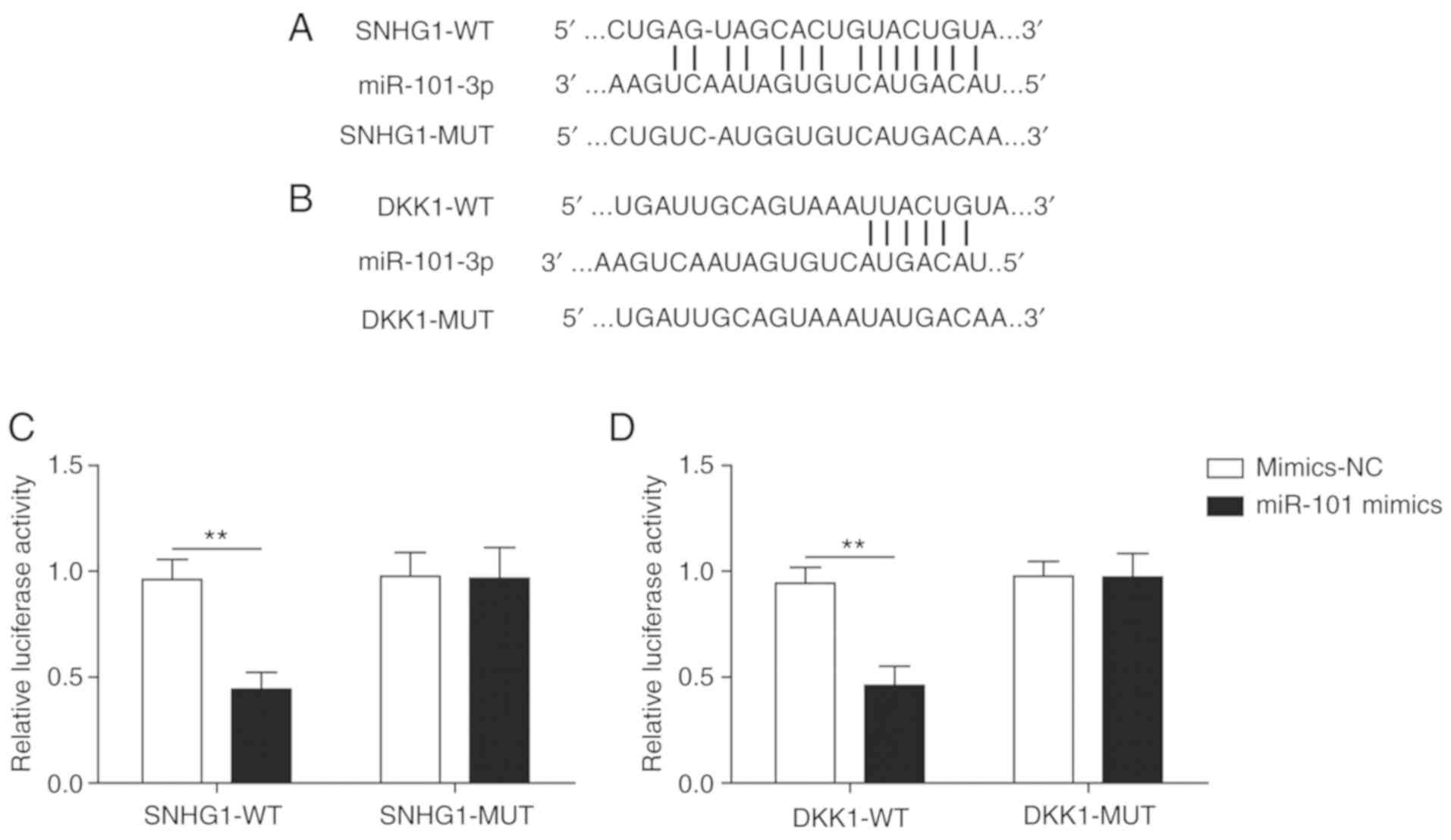
SNHG1 and DKK1 are targets of
miR-101
Firstly, the binding sites of miR-101 on SNHG1
(Fig. 2A) and DKK1 (Fig. 2B) were predicted by bioinformatics
analysis. The luciferase assay indicated that miR-101 mimics
significantly decreased the luciferase activity of SNHG1-wild-type
(WT) compared with mimics-NC, but miR-101 mimics did not
significantly alter the luciferase activity of SNHG1-mutated (MUT),
indicating a direct interaction between SNHG1 and miR-101 (Fig. 2C). Similarly, miR-101 mimics
significantly decreased the luciferase activity of DKK1-WT compared
with mimics-NC, but did not significantly alter the luciferase
activity of DKK1-MUT (Fig. 2D).
The results demonstrated that SNHG1 and DKK1 were targets of
miR-101.
SNHG1 inhibits BMSC osteogenic
differentiation
Compared with pcDNA, SNHG1 overexpression
significantly increased the expression of SNHG1 and DKK1 (Fig. 3A), but significantly decreased the
expression of miR-101 (Fig. 3A).
The expression levels of the osteogenesis-related markers were also
significantly decreased by SNHG1 overexpression compared with pcDNA
(Fig. 3B). Meanwhile, ALP activity
and matrix mineralization were both obviously decreased by SNHG1
overexpression compared with pcDNA (Fig. 3C and D). Compared with pcDNA, the
protein expression level of DKK1 was significantly increased,
whereas the protein expression levels of the osteogenesis-related
markers were significantly decreased after SNHG1 overexpression
(Fig. 3E and F), which indicated
that SNHG1 may inhibit osteogenic differentiation by regulating
miR-101 and DKK1.
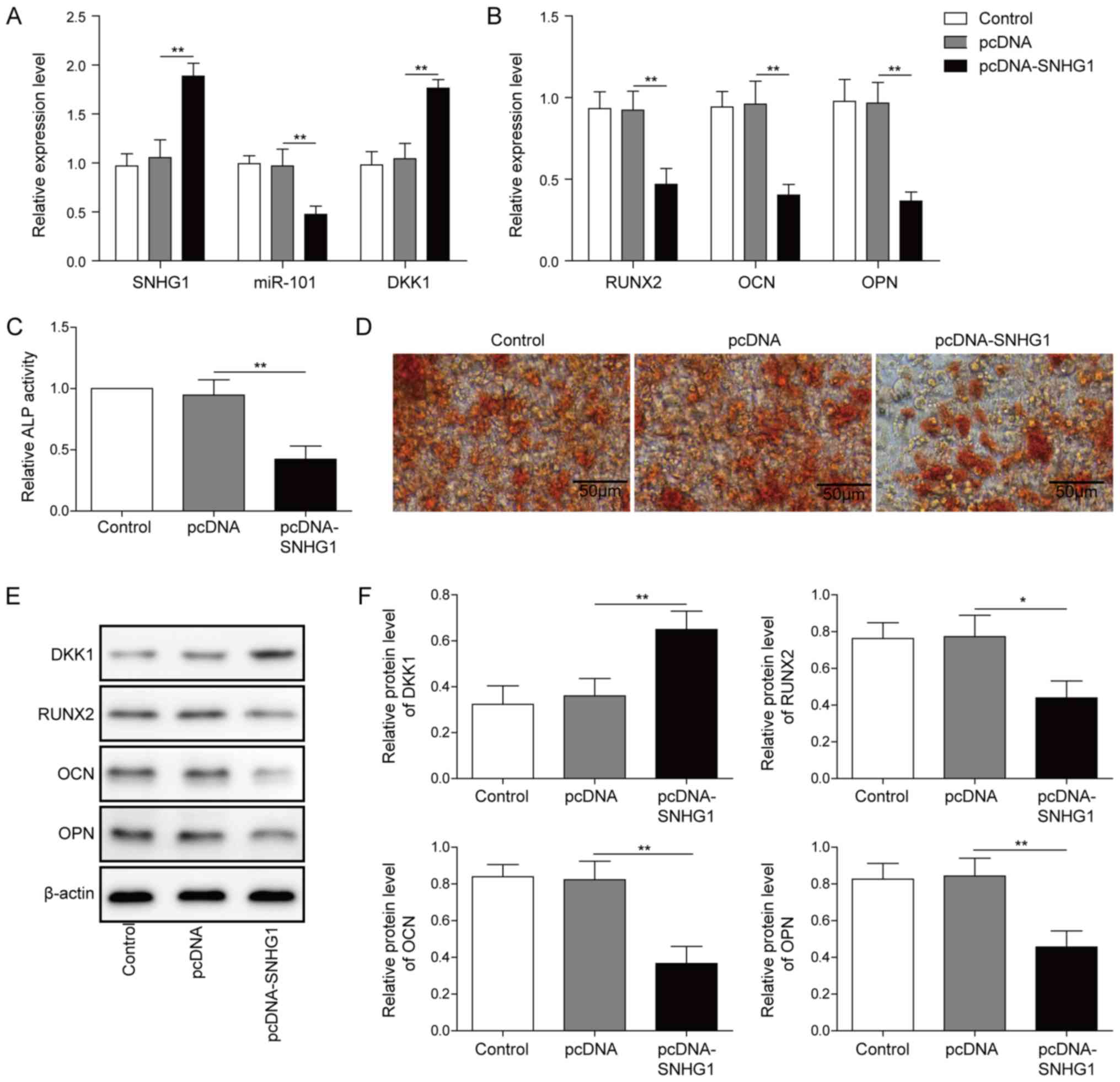 | Figure 3.SNHG1 overexpression inhibits
osteogenic differentiation. Effect of SNHG1 overexpression on the
expression of (A) SNHG1, miR-101, DKK1, (B) RUNX2, OPN and OCN in
BMSCs. Effect of SNHG1 overexpression on (C) ALP activity and (D)
Alizarin Red staining in BMSCs. Protein expression levels were (E)
determined by western blotting and (F) semi-quantified for DKK1,
RUNX2, OPN and OCN in SNHG1-overexpression BMSCs. *P<0.05 and
**P<0.01. SNHG1, small nucleolar RNA host gene 1; miR, microRNA;
DKK1, dickkopf WNT signaling pathway inhibitor 1; RUNX2, RUNX
family transcription factor 2; OPN, osteopontin; OCN, osteocalin;
BMSC, bone mesenchymal stem cell; ALP, alkaline phosphatase. |
SNHG1 inhibits BMSC osteogenic
differentiation via miR-101
To verify DKK1 was the target of miR-101, BMSCs were
transfected with miR-101 mimics and mimics-NC. Compared with the
mimics-NC group, DKK1 expression was significantly decreased and
miR-101 expression was significantly increased in the miR-101
mimics group (Fig. 4A). To assess
whether SNHG1 regulated osteogenic differentiation via miR-101,
BMSCs were co-transfected with pcDNA-SNHG1 and miR-101 mimics.
SNHG1 overexpression significantly increased DKK1 expression
compared with the pcDNA group, which was reversed by
co-transfection with miR-101 mimics (Fig. 4B). The expression levels of the
osteogenesis-related markers were significantly decreased by SNHG1
overexpression compared with the pcDNA group, whereas
co-transfection with miR-101 mimics reversed SNHG1
overexpression-induced effects (Fig.
4C). Furthermore, miR-101 mimics also reversed SNHG1
overexpression-mediated effects on ALP activity and matrix
mineralization (Fig. 4D and E).
Collectively, the results indicated that miR-101 reversed SNHG1
overexpression-mediated inhibition of osteogenic
differentiation.
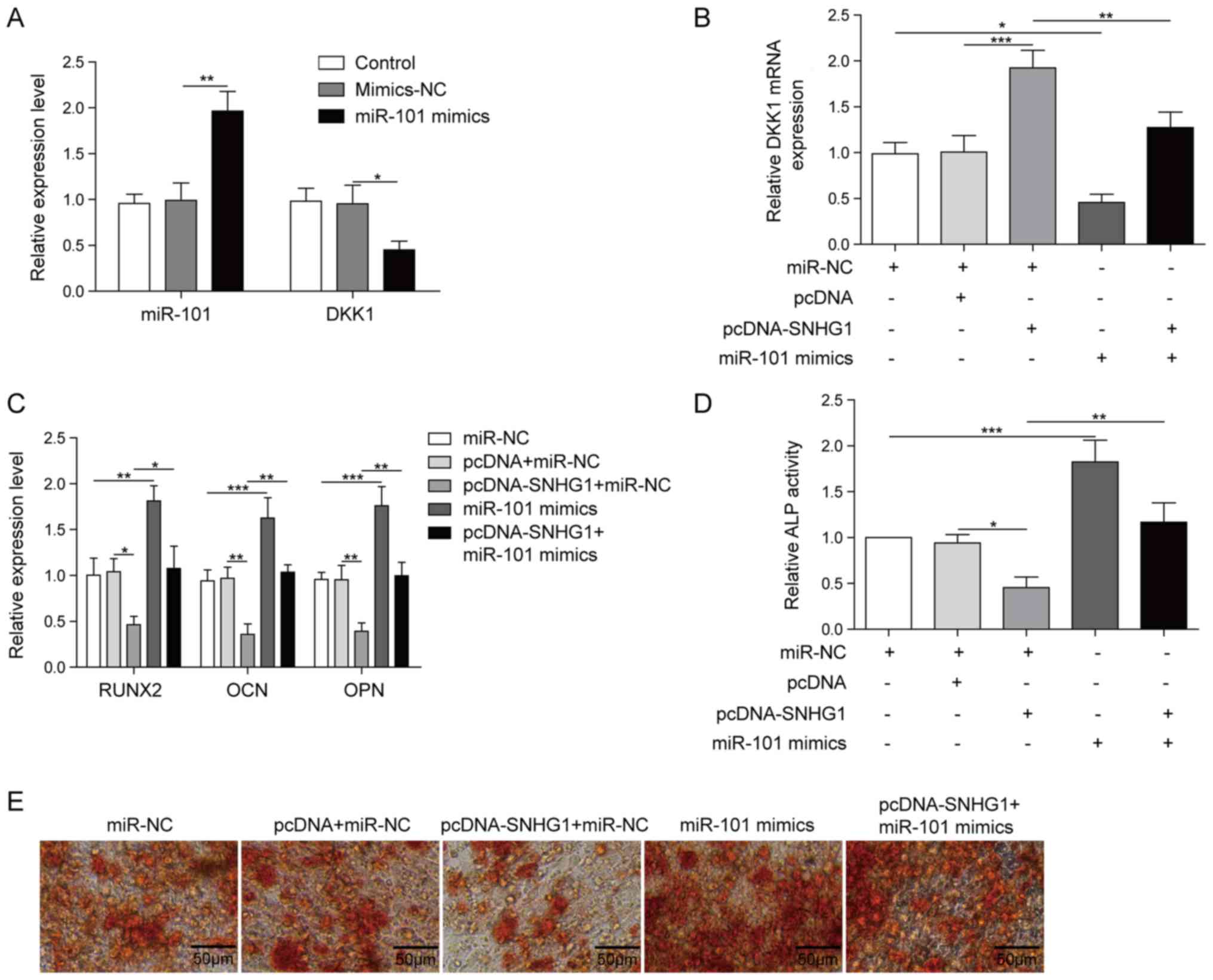 | Figure 4.SNHG1 overexpression alleviates BMSC
osteogenesis differentiation by targeting miR-101. (A) miR-101 and
DKK1 expression levels in BMSCs transfected with miR-101 mimics and
mimics-NC. (B) DKK1 expression levels in BMSCs following
co-transfection with pcDNA-SNHG1 and miR-101 mimics. (C) The
expression levels of RUNX2, OPN and OCN expression in BMSCs
co-transfected with pcDNA-SNHG1 and miR-101 mimics. (D) ALP
activity and (E) Alizarin Red staining in BMSCs co-transfected with
pcDNA-SNHG1 and miR-101 mimics. *P<0.05, **P<0.01 and
***P<0.001. SNHG1, small nucleolar RNA host gene 1; BMSC, bone
mesenchymal stem cell; miR, microRNA; DKK1, dickkopf WNT signaling
pathway inhibitor 1; NC, negative control; RUNX2, RUNX family
transcription factor 2; OPN, osteopontin; OCN, osteocalin. |
SNHG1 regulates the Wnt/β-catenin
signaling pathway to attenuate osteogenic differentiation
As DKK1 was identified as a target of miR-101,
whether SNHG1 regulated the expression of DKK1 and proteins in its
downstream signaling pathway was further examined. SNHG1
overexpression upregulated the protein expression levels of DKK1
and downregulated the protein expression levels of β-catenin,
RUNX2, OCN, OPN, OSX and COL1a1 compared with pcDNA (Fig. 5A and B). miR-101 mimics reversed
SNHG1 overexpression-mediated effects on protein expression
(Fig. 5A and B), indicating that
SNHG1 regulated the expression of DKK1 and its downstream
Wnt/β-catenin signaling pathway by sponging miR-101 to attenuate
osteogenic differentiation.
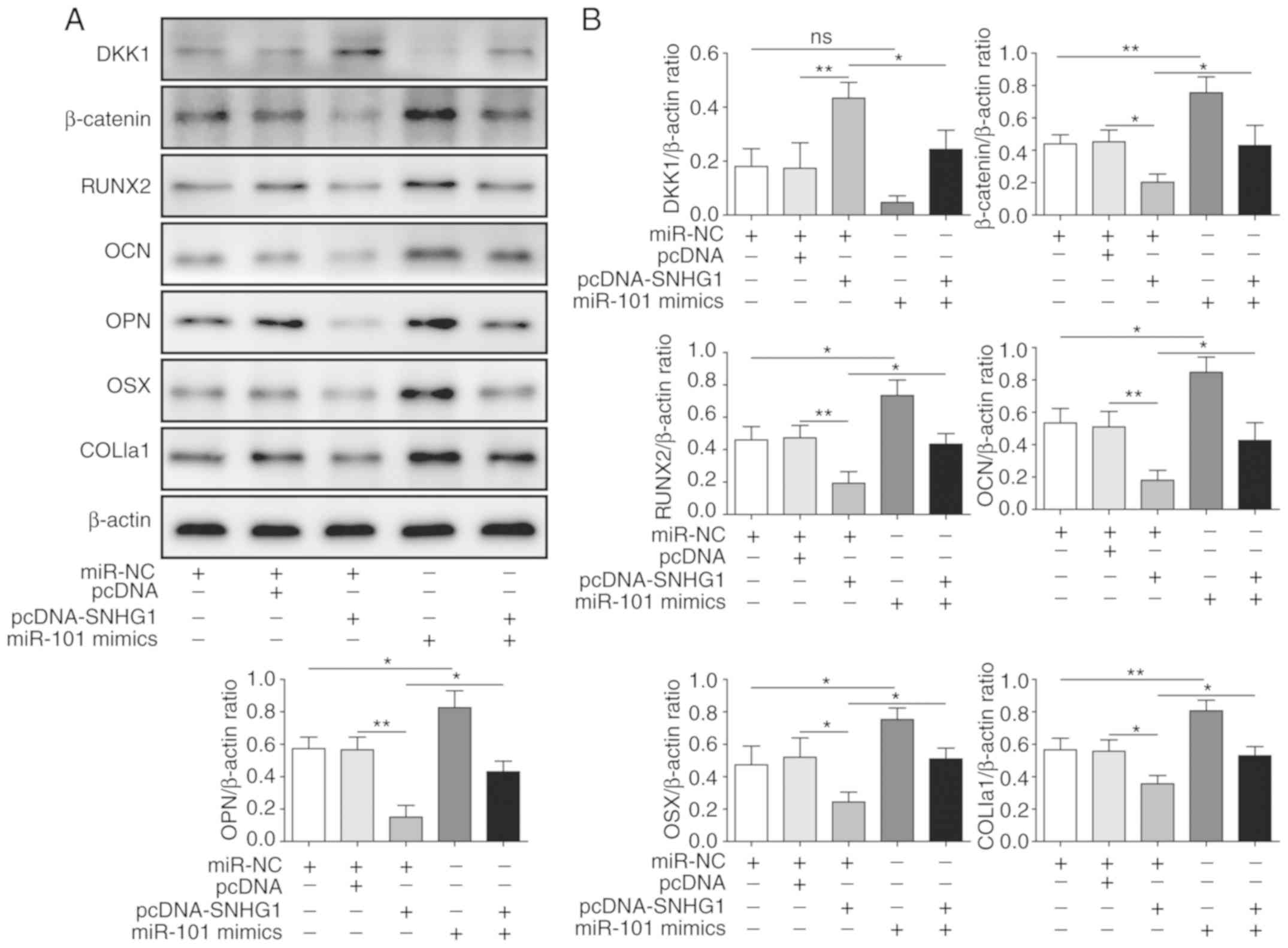 | Figure 5.SNHG1 regulates the Wnt/β-catenin
signaling pathway to inhibit osteogenic differentiation. Protein
expression levels were (A) determined by western blotting and (B)
semi-quantified for β-catenin, DKK1, RUNX2, OCN, OPN, OSX and
COL1a1 in bone mesenchymal stem cells co-transfected with
pcDNA-SNHG1 and miR-101 mimics. *P<0.05 and **P<0.01. SNHG1,
small nucleolar RNA host gene 1; DKK1, dickkopf WNT signaling
pathway inhibitor 1; RUNX2, RUNX family transcription factor 2;
OCN, osteocalin; OPN, osteopontin; OSX, osterix; COL1a1, collagen
type I α1 chain; NC, negative control. |
Discussion
As a serious and complicated bone disease, OP is
associated with a high risk of fracture (19). Previous studies have demonstrated
that abnormal BMSC osteogenic differentiation is the leading cause
of OP (2,20,21).
Therefore, regulation of BMSC osteogenic differentiation is a vital
focus in OP pathogenesis research.
lncRNAs can serve a crucial role in the regulation
of osteogenic differentiation. For example, lncRNA twist family
bHLH transcription factor (TWIST)1 can promote periodontal
mesenchymal stem cells from patients with periodontitis (PPDLSC)
and periodontal mesenchymal stem cells from healthy
microenvironment (HPDLSC) osteogenic differentiation by inhibiting
the expression of TWIST (22).
lncRNA-OG was reported to promote BMSC osteogenic differentiation
under the regulation of heterogeneous nuclear ribonucleoprotein K
(23). Moreover, H19 imprinted
maternally expressed transcript (H19) can serve as a competing
endogenous RNA (ceRNA) to promote osteogenic differentiation via
the Wnt/β-catenin signaling pathway (24). Moreover, lncRNA SNHG1 can inhibit
BMSC osteogenic differentiation by modulating the p38 MAPK
signaling pathway (10). However,
the downstream molecular mechanisms underlying the effects of SNHG1
in osteogenic differentiation are not completely understood. The
present study indicated that SNHG1 expression was downregulated in
time-dependent manner during osteogenic differentiation, and SNHG1
overexpression inhibited osteogenic differentiation, indicating a
negative regulatory role of SNHG1 in BMSC osteogenic
differentiation.
ceRNA was reported to be a new regulatory molecular
mechanism underlying lncRNAs, where lncRNAs function as miRNA
sponges to upregulate the expression of their targets (25). SNHG1 was also reported to function
as a ceRNA and serve a regulatory role with this mechanism. For
instance, SNHG1 can regulate programmed cell death 4 expression by
sponging miR-195-5p in hepatocellular carcinoma (26). SNHG1 can also antagonize the effect
of miR-145a-5p on the downregulation of NUAK family kinase 1 in
nasopharyngeal carcinoma cells (27). Besides, SNHG1 can promote
osteosarcoma tumorigenesis by sponging miR-326 (28). Therefore, the present study
investigated whether SNHG1 regulated osteogenic differentiation as
a ceRNA. As miR-101 was predicted to be the target of SNHG1, the
expression of miR-101 and its relationship with SNHG1 in BMSC
osteogenic differentiation were investigated. Moreover, the results
indicated that miR-101 overexpression reversed SNHG1-mediated
inhibition of osteogenic differentiation, indicating that SNHG1
served as a ceRNA to regulate osteogenic differentiation by
sponging miR-101. To the best of our knowledge, the present study
was the first to suggest that SNHG1 regulated BMSC osteogenic
differentiation by sponging miR-101 as a ceRNA.
The Wnt/β-catenin signaling pathway was reported to
be involved in osteogenic differentiation, which was also the
downstream molecular mechanism underlying a number of lncRNAs
(29,30). For example, lncRNA H19 could
promote rat ectomesenchymal stem cell osteogenic differentiation
via the Wnt/β-catenin signaling pathway (24), whereas lncRNA KCNQ1OT1 promoted
osteogenic differentiation to relieve osteolysis via Wnt/β-catenin
activation (23). In addition,
lncRNA-p21 was also reported to promote mesenchymal stem cell
osteogenic differentiation in the rat model of osteoporosis via the
Wnt/β-catenin signaling pathway (31). The present study indicated that the
Wnt/β-catenin signaling pathway was a downstream mechanism of the
SNHG1/miR-101 axis for the regulation of osteogenic
differentiation, which was consistent with previous reports.
The present study had a number of limitations. The
pathogenesis of OP requires further investigation. For instance, an
animal model should be applied to validate the molecular mechanism
related to the SNHG1/miR-101/DKK1 axis in regulation of BMSC
osteogenic differentiation and prove its therapeutic potential.
Therefore, the mechanism identified in the present study requires
further investigation using in vivo models of OP. In
summary, the present study identified a potential downstream
molecular mechanism of the SNHG1 axis in regulating osteogenic
differentiation, providing potential therapeutic targets for
OP.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Hunan
Provincial Health and Family Planning Research Program (grant no.
C2019121).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
BC and JX conceptualized and designed the study and
revised the manuscript. HQF collected and interpreted the data.
WJF, ZX and FL analyzed and interpreted the data. FL drafted the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Iseme RA, McEvoy M, Kelly B, Agnew L,
Walker FR and Attia J: Is osteoporosis an autoimmune mediated
disorder? Bone Rep. 7:121–131. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Phetfong J, Sanvoranart T, Nartprayut K,
Nimsanor N, Seenprachawong K, Prachayasittikul V and Supokawej A:
Osteoporosis: The current status of mesenchymal stem cell-based
therapy. Cell Mol Biol Lett. 21:122016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Oryan A, Kamali A, Moshiri A and Baghaban
Eslaminejad M: Role of mesenchymal stem cells in bone regenerative
medicine: What is the evidence? Cells Tissues Organs. 204:59–83.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shen Y, Liu S, Fan J, Jin Y, Tian B, Zheng
X and Fu H: Nuclear retention of the lncRNA SNHG1 by doxorubicin
attenuates hnRNPC-p53 protein interactions. EMBO Rep. 18:536–548.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang CG, Liao Z, Xiao H, Liu H, Hu YH,
Liao QD and Zhong D: LncRNA KCNQ1OT1 promoted BMP2 expression to
regulate osteogenic differentiation by sponging miRNA-214. Exp Mol
Pathol. 107:77–84. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gao Y, Xiao F, Wang C, Wang C, Cui P,
Zhang X and Chen X: Long noncoding RNA MALAT1 promotes osterix
expression to regulate osteogenic differentiation by targeting
miRNA-143 in human bone marrow-derived mesenchymal stem cells. J
Cell Biochem. 119:6986–6996. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sun Y, Wei G, Luo H, Wu W, Skogerbø G, Luo
J and Chen R: The long noncoding RNA SNHG1 promotes tumor growth
through regulating transcription of both local and distal genes.
Oncogene. 36:6774–6783. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xu M, Chen X, Lin K, Zeng K, Liu X, Pan B,
Xu X, Xu T, Hu X, Sun L, et al: The long noncoding RNA SNHG1
regulates colorectal cancer cell growth through interactions with
EZH2 and miR-154-5p. Mol Cancer. 17:1412018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cui Y, Zhang F, Zhu C, Geng L, Tian T and
Liu H: Upregulated lncRNA SNHG1 contributes to progression of
non-small cell lung cancer through inhibition of miR-101-3p and
activation of Wnt/β-catenin signaling pathway. Oncotarget.
8:17785–17794. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jiang Y, Wu W, Jiao G, Chen Y and Liu H:
LncRNA SNHG1 modulates p38 MAPK pathway through Nedd4 and thus
inhibits osteogenic differentiation of bone marrow mesenchymal stem
cells. Life Sci. 228:208–214. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Oliveto S, Mancino M, Manfrini N and Biffo
S: Role of microRNAs in translation regulation and cancer. World J
Biol Chem. 8:45–56. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang H, Meng Y, Cui Q, Qin F, Yang H, Chen
Y, Cheng Y, Shi J and Guo Y: MiR-101 targets the EZH2/Wnt/β-catenin
the pathway to promote the osteogenic differentiation of human bone
marrow-derived mesenchymal stem cells. Sci Rep. 6:369882016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Deng R, Zhang J and Chen J: lncRNA SNHG1
negatively regulates miRNA-101-3p to enhance the expression of
ROCK1 and promote cell proliferation, migration and invasion in
osteosarcoma. Int J Mol Med. 43:1157–1166. 2019.PubMed/NCBI
|
|
14
|
Yavropoulou MP and Yovos JG: The role of
the Wnt signaling pathway in osteoblast commitment and
differentiation. Hormones (Athens). 6:279–294. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Albers J, Keller J, Baranowsky A, Beil FT,
Catala-Lehnen P, Schulze J, Amling M and Schinke T: Canonical Wnt
signaling inhibits osteoclastogenesis independent of
osteoprotegerin. J Cell Biol. 200:537–549. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Y, Lu W, King TD, Liu CC, Bijur GN and
Bu G: Dkk1 stabilizes Wnt co-receptor LRP6: Implication for Wnt
ligand-induced LRP6 down-regulation. PLoS One. 5:e110142010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mirfazeli ES, Arefian E, Nadri S,
Rezazadeh Valojerdi R, Kehtari M and Zeynali B: DKK1 expression is
suppressed by miR-9 during induced dopaminergic differentiation of
human trabecular meshwork mesenchymal stem cells. Neurosci Lett.
707:1342502019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sözen T, Özışık L and Başaran NÇ: An
overview and management of osteoporosis. Eur J Rheumatol. 4:46–56.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhou DA, Zheng HX, Wang CW, Shi D and Li
JJ: Influence of glucocorticoids on the osteogenic differentiation
of rat bone marrow-derived mesenchymal stem cells. BMC
Musculoskelet Disord. 15:2392014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang C, Meng H, Wang X, Zhao C, Peng J and
Wang Y: Differentiation of bone marrow mesenchymal stem cells in
osteoblasts and adipocytes and its role in treatment of
osteoporosis. Med Sci Monit. 22:226–233. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu Y, Qin W, Guo D, Liu J, Zhang M and Jin
Z: LncRNA-TWIST1 promoted osteogenic differentiation both in
PPDLSCs and in HPDLSCs by inhibiting TWIST1 expression. Biomed Res
Int. 2019:87359522019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tang S, Xie Z, Wang P, Li J, Wang S, Liu
W, Li M, Wu X, Su H, Cen S, et al: LncRNA-OG promotes the
osteogenic differentiation of bone marrow-derived mesenchymal stem
cells under the regulation of hnRNPK. Stem Cells. 37:270–283. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gong YY, Peng MY, Yin DQ and Yang YF: Long
non-coding RNA H19 promotes the osteogenic differentiation of rat
ectomesenchymal stem cells via Wnt/β-catenin signaling pathway. Eur
Rev Med Pharmacol Sci. 22:8805–8813. 2018.PubMed/NCBI
|
|
25
|
Du Z, Sun T, Hacisuleyman E, Fei T, Wang
X, Brown M, Rinn JL, Lee MG, Chen Y, Kantoff PW and Liu XS:
Integrative analyses reveal a long noncoding RNA-mediated sponge
regulatory network in prostate cancer. Nat Commun. 7:109822016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Huang D, Wei Y, Zhu J and Wang F: Long
non-coding RNA SNHG1 functions as a competitive endogenous RNA to
regulate PDCD4 expression by sponging miR-195-5p in hepatocellular
carcinoma. Gene. 714:1439942019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lan X and Liu X: LncRNA SNHG1 functions as
a ceRNA to antagonize the effect of miR-145a-5p on the
down-regulation of NUAK1 in nasopharyngeal carcinoma cell. J Cell
Mol Med. 23:2351–2361. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang J, Cao L, Wu J and Wang Q: Long
non-coding RNA SNHG1 regulates NOB1 expression by sponging miR-326
and promotes tumorigenesis in osteosarcoma. Int J Oncol. 52:77–88.
2018.PubMed/NCBI
|
|
29
|
Shen JJ, Zhang CH, Chen ZW, Wang ZX, Yang
DC, Zhang FL and Feng KH: LncRNA HOTAIR inhibited osteogenic
differentiation of BMSCs by regulating Wnt/β-catenin pathway. Eur
Rev Med Pharmacol Sci. 23:7232–7246. 2019.PubMed/NCBI
|
|
30
|
Li N, Ma Y, Wang W, Yin CC, Wu W, Sun R,
Zhao G, Li S and Wang X: LOC101928834, a novel lncRNA in
Wnt/β-catenin signaling pathway, promotes cell proliferation and
predicts poor clinical outcome in myelodysplastic syndromes. Clin
Sci (Lond). 134:1279–1293. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yang K, Tian N, Liu H, Tao XZ, Wang MX and
Huang W: LncRNAp21 promotes osteogenic differentiation of
mesenchymal stem cells in the rat model of osteoporosis by the
Wnt/β-catenin signaling pathway. Eur Rev Med Pharmacol Sci.
23:4303–4309. 2019.PubMed/NCBI
|