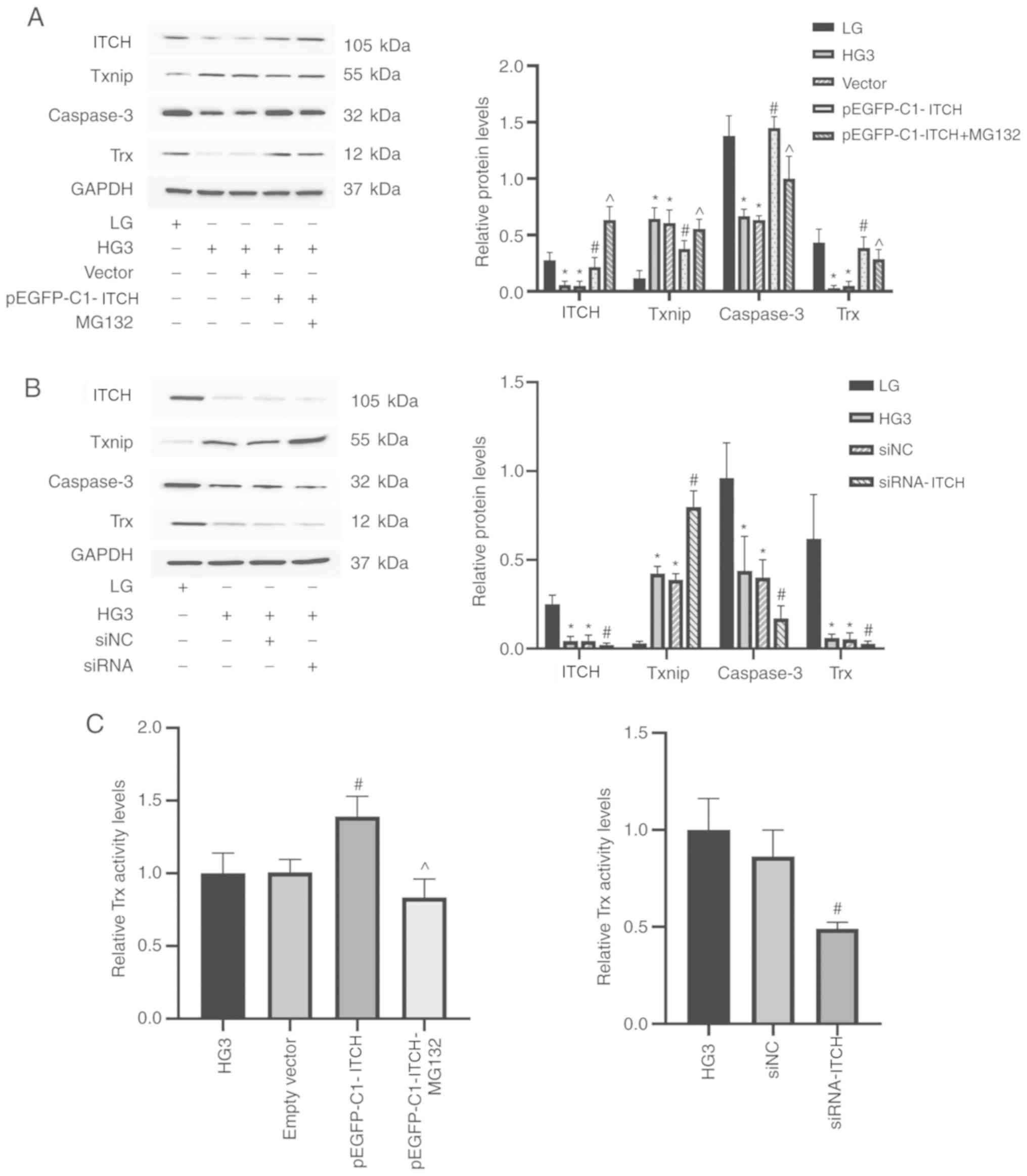
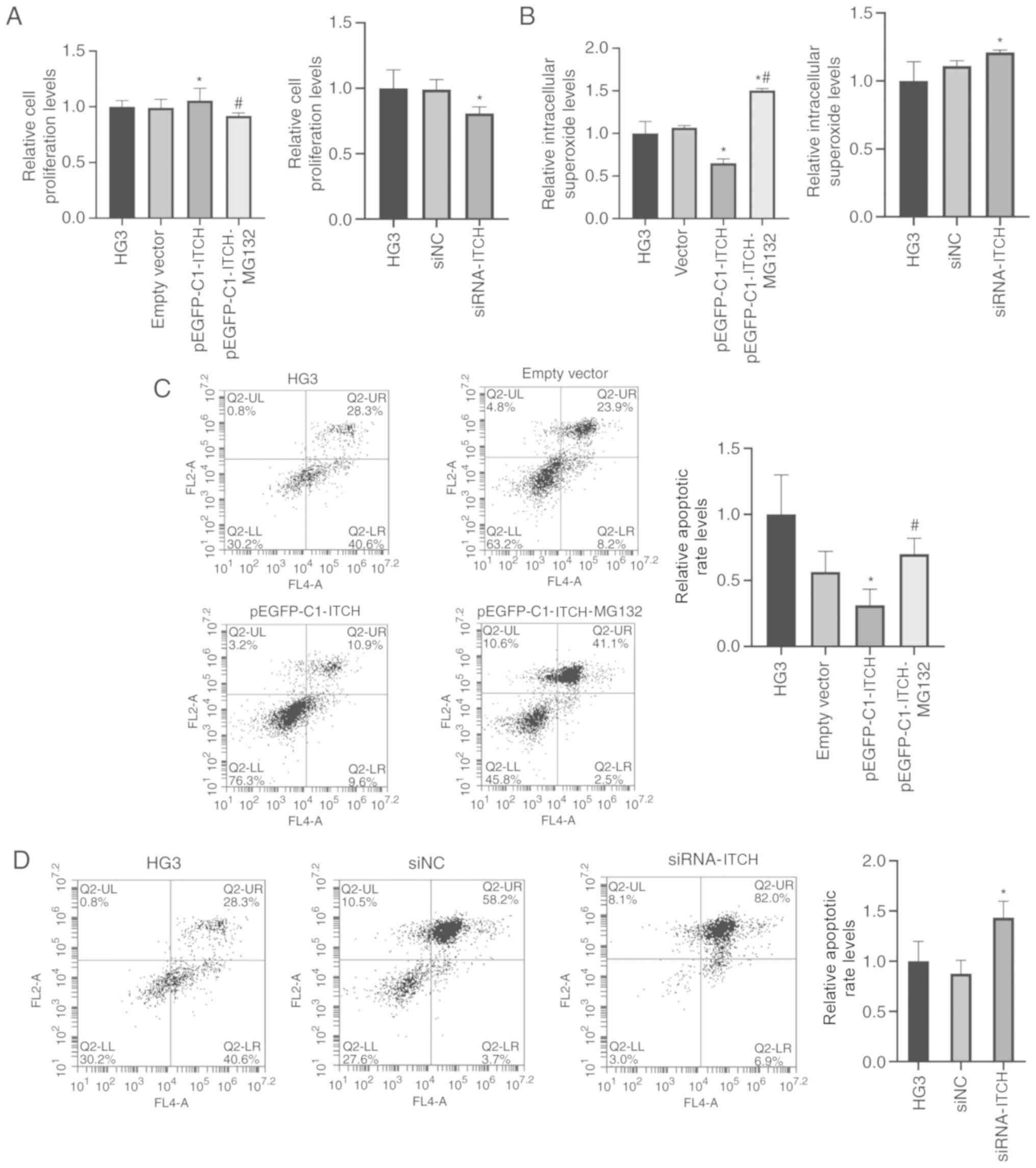
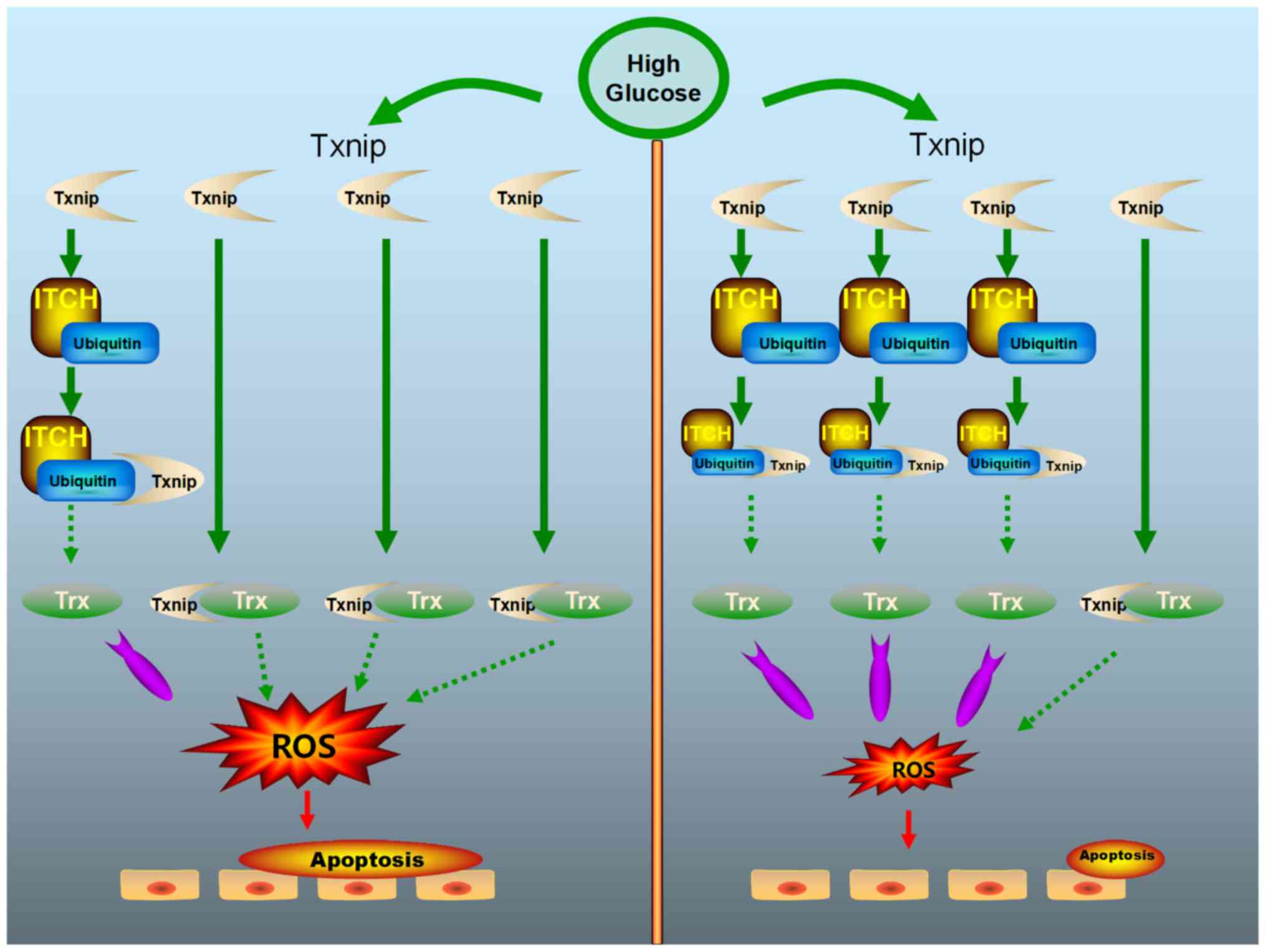
|
1
|
Fukuoka H and Afshari NA: The impact of
age-related cataract on measures of frailty in an aging global
population. Curr Opin Ophthalmol. 28:93–97. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Francis PJ and Moore AT: Genetics of
childhood cataract. Curr Opin Ophthalmol. 15:10–15. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Andley UP, Tycksen E, McGlasson-Naumann BN
and Hamilton PD: Probing the changes in gene expression due to
α-crystallin mutations in mouse models of hereditary human
cataract. PLoS One. 13:e01908172018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bahia S, Blais E, Murugkar S, Chauhan V
and Kumarathasan P: Oxidative and nitrative stress-related changes
in human lens epithelial cells following exposure to X-rays. Int J
Radiat Biol. 94:366–373. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bhagat AK, Bhardwaj H, Bhardwaj BL, Goyal
S, Jaura S and Jain P: Acute bilateral cataract in patient with
type 1 diabetes mellitus. J Assoc Physicians India.
67:832019.PubMed/NCBI
|
|
6
|
Mirsky N, Cohen R, Eliaz A and Dovrat A:
Featured article: Inhibition of diabetic cataract by glucose
tolerance factor extracted from yeast. Exp Biol Med (Maywood).
241:817–829. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tinkov AA, Bjørklund G, Skalny AV,
Holmgren A, Skalnaya MG, Chirumbolo S and Aaseth J: The role of the
thioredoxin/thioredoxin reductase system in the metabolic syndrome:
Towards a possible prognostic marker? Cell Mol Life Sci.
75:1567–1586. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Montano SJ, Lu J, Gustafsson TN and
Holmgren A: Activity assays of mammalian thioredoxin and
thioredoxin reductase: Fluorescent disulfide substrates,
mechanisms, and use with tissue samples. Anal Biochem. 449:139–146.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lu J and Holmgren A: The thioredoxin
antioxidant system. Free Radic Biol Med. 66:75–87. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen KS and DeLuca HF: Isolation and
characterization of a novel cDNA from HL-60 cells treated with
1,25-dihydroxyvitamin D-3. Biochim Biophys Acta. 1219:26–32. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Patwari P, Higgins LJ, Chutkow WA,
Yoshioka J and Lee RT: The interaction of thioredoxin with Txnip.
Evidence for formation of a mixed disulfide by disulfide exchange.
J Biol Chem. 281:21884–21891. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Abderrazak A, Syrovets T, Couchie D, El
Hadri K, Friguet B, Simmet T and Rouis M: NLRP3 inflammasome: From
a danger signal sensor to a regulatory node of oxidative stress and
inflammatory diseases. Redox Biol. 4:296–307. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nishiyama A, Matsui M, Iwata S, Hirota K,
Masutani H, Nakamura H, Takagi Y, Sono H, Gon Y and Yodoi J:
Identification of thioredoxin-binding protein-2/vitamin D(3)
up-regulated protein 1 as a negative regulator of thioredoxin
function and expression. J Biol Chem. 274:21645–21650. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pasternak Y, Ohana M, Biron-Shental T,
Cohen-Hagai K, Benchetrit S and Zitman-Gal T: Thioredoxin,
thioredoxin interacting protein and transducer and activator of
transcription 3 in gestational diabetes. Mol Biol Rep.
47:1199–1206. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gao C, Huang W, Kanasaki K and Xu Y: The
role of ubiquitination and sumoylation in diabetic nephropathy.
Biomed Res Int. 2014:1606922014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lorenz S: Structural mechanisms of
HECT-type ubiquitin ligases. Biol Chem. 399:127–145. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Michelle C, Vourc'h P, Mignon L and Andres
CR: What was the set of ubiquitin and ubiquitin-like conjugating
enzymes in the eukaryote common ancestor? J Mol Evol. 68:616–628.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li W, Bengtson MH, Ulbrich A, Matsuda A,
Reddy VA, Orth A, Chanda SK, Batalov S and Joazeiro CA: Genome-wide
and functional annotation of human E3 ubiquitin ligases identifies
MULAN, a mitochondrial E3 that regulates the organelle's dynamics
and signaling. PLoS One. 3:e14872008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aki D, Zhang W and Liu YC: The E3 ligase
Itch in immune regulation and beyond. Immunol Rev. 266:6–26. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Otaki Y, Takahashi H, Watanabe T, Funayama
A, Netsu S, Honda Y, Narumi T, Kadowaki S, Hasegawa H, Honda S, et
al: HECT-type ubiquitin E3 ligase ITCH interacts with
thioredoxin-interacting protein and ameliorates reactive oxygen
species-induced cardiotoxicity. J Am Heart Assoc. 5:e0024852016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Han X, Dong XX, Shi MY, Feng L, Wang XL,
Zhang JS and Yan QC: SUMOylation and deacetylation affect NF-κB p65
activity induced by high glucose in human lens epithelial cells.
Int J Ophthalmol. 12:1371–1379. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Park YJ, Oanh NTK, Heo J, Kim SG, Lee HS,
Lee H, Lee JH, Kang HC, Lim W, Yoo YS and Cho H: Dual targeting of
RIG-I and MAVS by MARCH5 mitochondria ubiquitin ligase in innate
immunity. Cell Signal. 67:1095202020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Junn E, Han SH, Im JY, Yang Y, Cho EW, Um
HD, Kim DK, Lee KW, Han PL, Rhee SG and Choi I: Vitamin D3
up-regulated protein 1 mediates oxidative stress via suppressing
the thioredoxin function. J Immunol. 164:6287–6295. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang Y, De Keulenaer GW and Lee RT:
Vitamin D(3)-up- regulated protein-1 is a stress-responsive gene
that regulates cardiomyocyte viability through interaction with
thioredoxin. J Biol Chem. 277:26496–26500. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Osnes-Ringen Ø, Berg KH, Moe MC,
Zetterström C, Røger M and Nicolaissen B: Cell death pattern in
lens epithelium of cataract patients. Acta Ophthalmol. 94:514–520.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bron AJ, Brown NA, Harding JJ and Ganea E:
The lens and cataract in diabetes. Int Ophthalmol Clin. 38:37–67.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Collet JF and Messens J: Structure,
function, and mechanism of thioredoxin proteins. Antioxid Redox
Signal. 13:1205–1216. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen KS and DeLuca HF: Cloning of the
human 1 alpha, 25-dihydroxyvitamin D-3 24-hydroxylase gene promoter
and identification of two vitamin D-responsive elements. Biochim
Biophys Acta. 1263:1–9. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jeanine CS, Amie DM and Walter HW:
Oxidation and nuclear localization of thioredoxin-1 in sparse cell
cultures. J Cell Biochem. 104:1879–1889. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang X, Zhang JH, Chen XY, Hu QH, Wang
MX, Jin R, Zhang QY, Wang W, Wang R, Kang LL, et al: Reactive
oxygen species-induced TXNIP drives fructose-mediated hepatic
inflammation and lipid accumulation through NLRP3 inflammasome
activation. Antioxid Redox Signal. 22:848–870. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Oslowski CM, Hara T, O'Sullivan-Murphy B,
Kanekura K, Lu S, Hara M, Ishigaki S, Zhu LJ, Hayashi E, Hui ST, et
al: Thioredoxin-interacting protein mediates ER stress-induced β
cell death through initiation of the inflammasome. Cell Metab.
16:265–273. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bernardini S, Gravina P, Croce N,
Perricone R, Knight RA, Valentini A, Melino G and Federici G: Itch
gene polymorphisms in healthy population and in patients affected
by rheumatoid arthritis and atopic dermatitis. Cell Cycle.
7:3607–3609. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang P, Wang C, Gao K, Wang D, Mao J, An
J, Xu C, Wu D, Yu H, Liu JO and Yu L: The ubiquitin ligase itch
regulates apoptosis by targeting thioredoxin-interacting protein
for ubiquitin-dependent degradation. J Biol Chem. 285:8869–8879.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mouchantaf R, Azakir BA, McPherson PS,
Millard SM, Wood SA and Angers A: The ubiquitin ligase itch is
auto-ubiquitylated in vivo and in vitro but is protected from
degradation by interacting with the deubiquitylating enzyme
FAM/USP9X. J Biol Chem. 281:38738–38747. 2006. View Article : Google Scholar : PubMed/NCBI
|