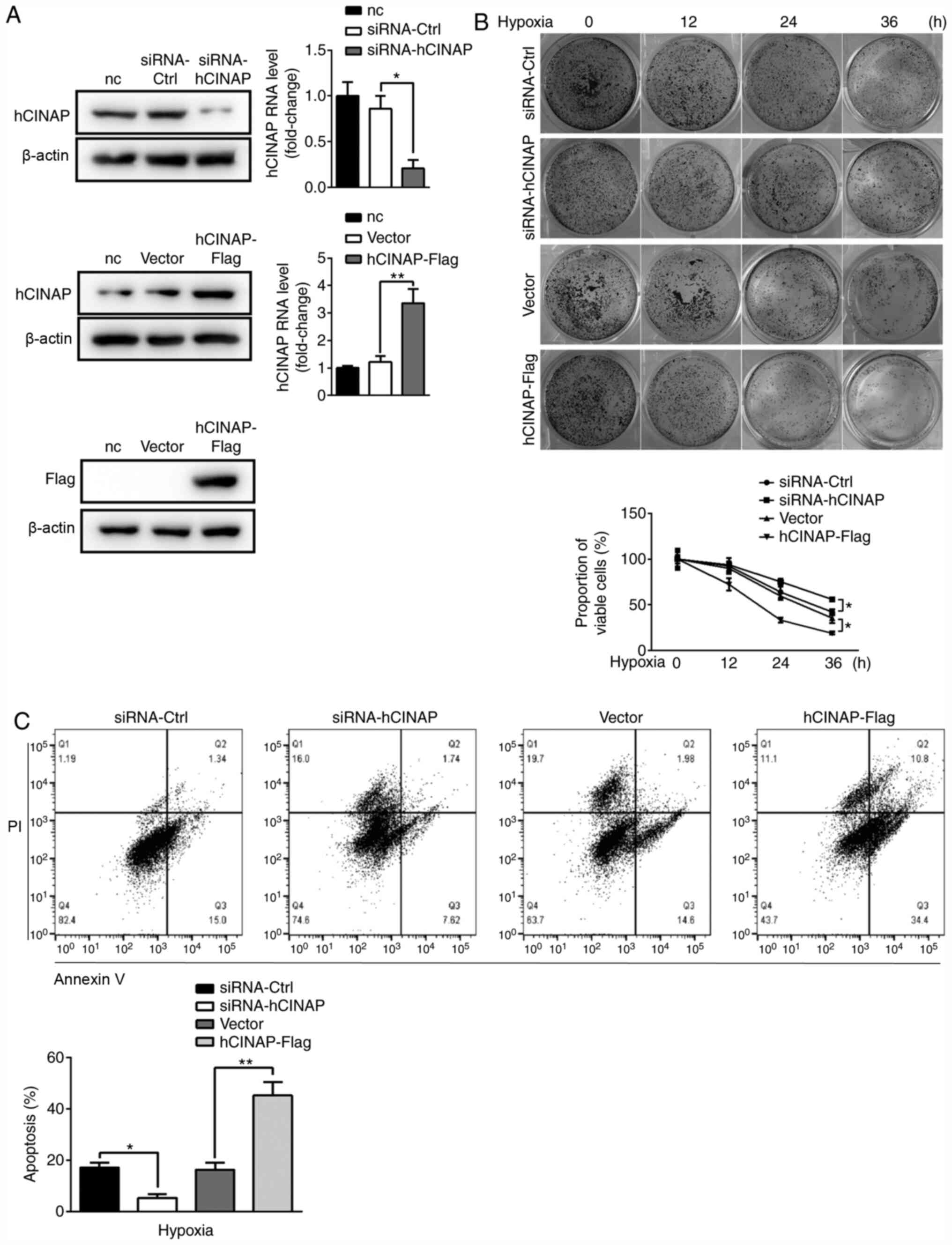
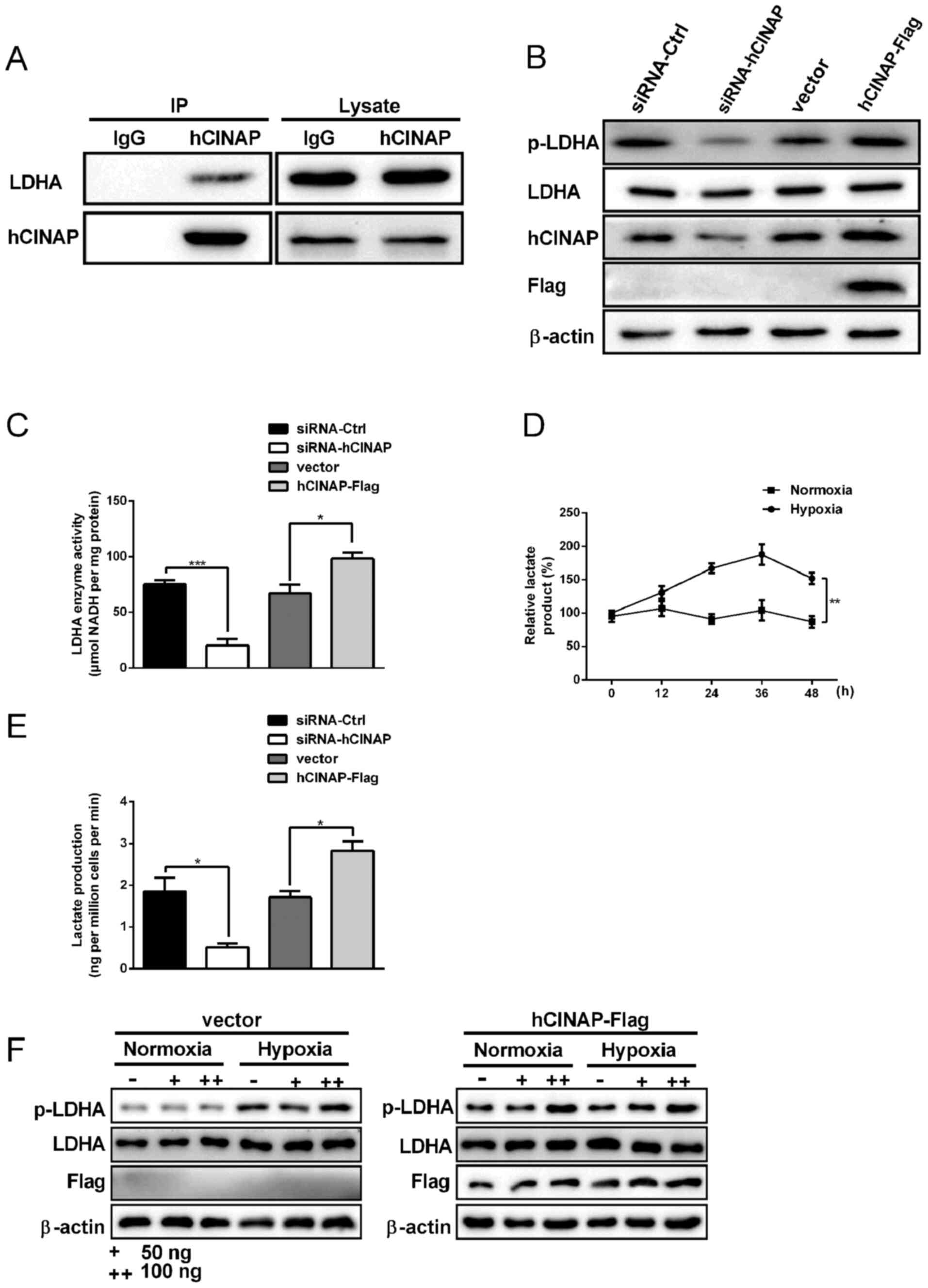
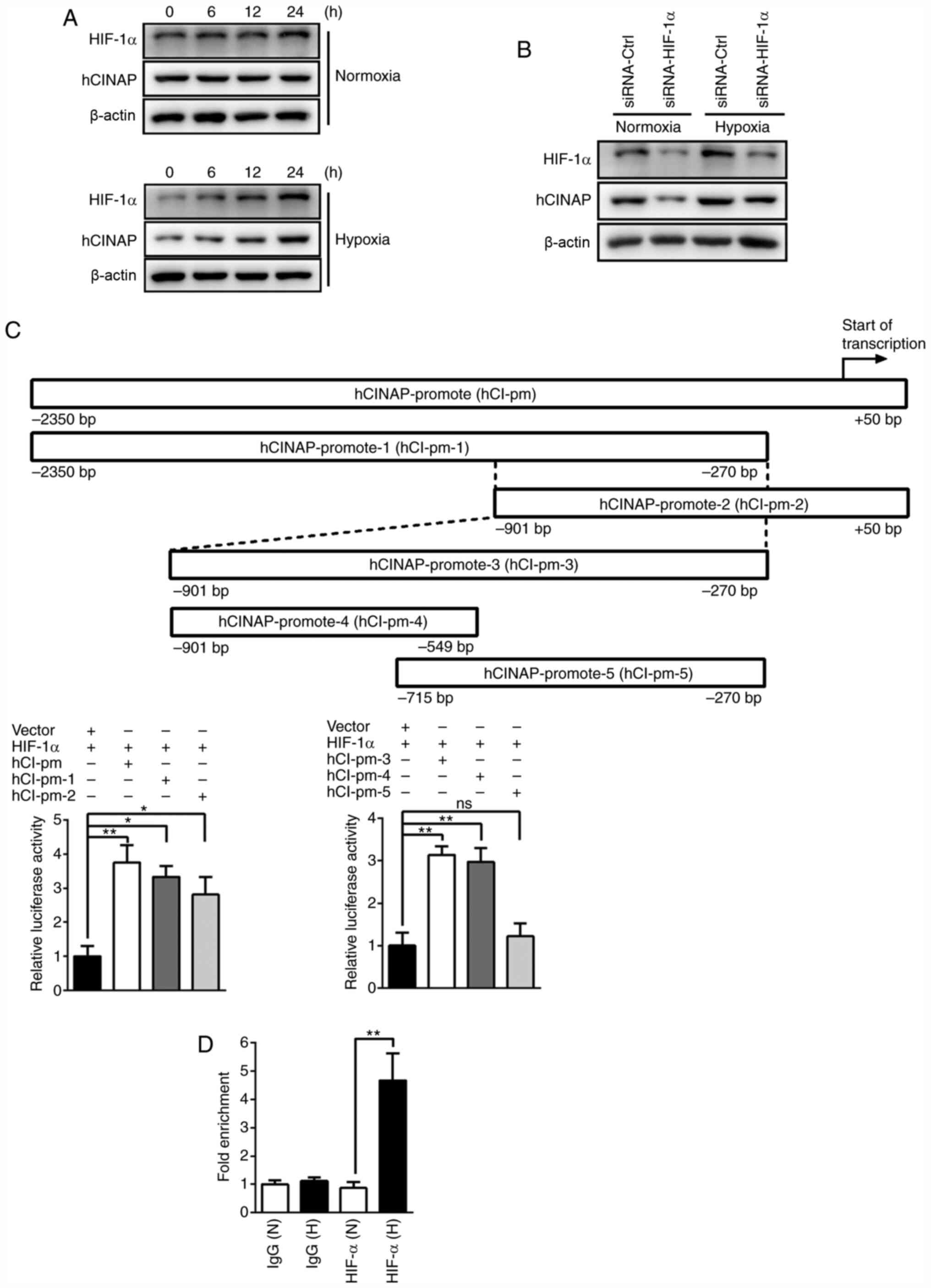
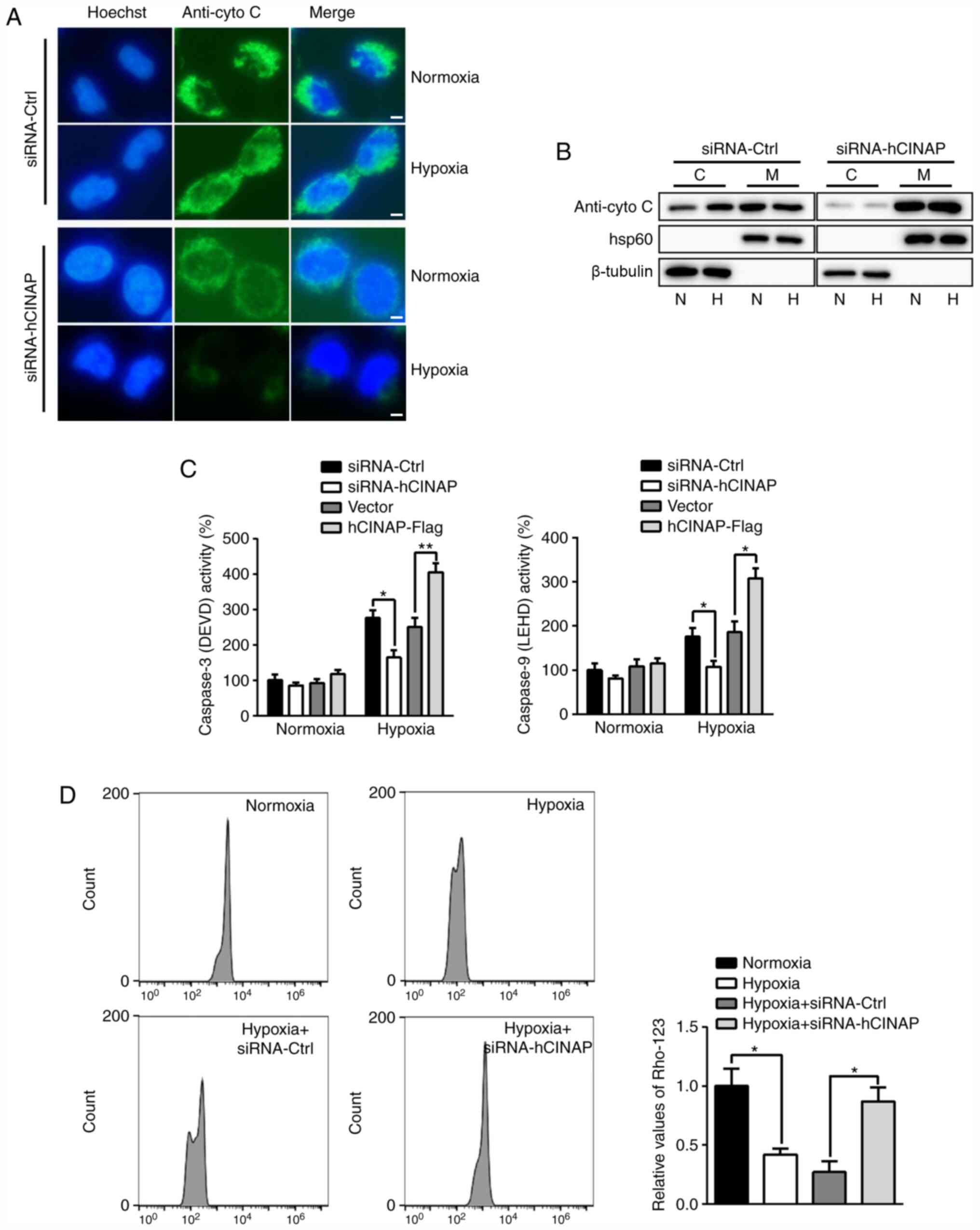
|
1
|
Benjamin EJ, Virani SS, Callaway CW,
Chamberlain AM, Chang AR, Cheng S, Chiuve SE, Cushman M, Delling
FN, Deo R, et al: Heart disease and stroke statistics-2018 update:
A report from the American heart association. Circulation.
137:e67–e492. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Halestrap AP and Wilson MC: The
monocarboxylate transporter family-role and regulation. IUBMB Life.
64:109–119. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Anderson JL and Morrow DA: Acute
myocardial infarction. N Engl J Med. 376:2053–2064. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nakada Y, Canseco DC, Thet S, Abdisalaam
S, Asaithamby A, Santos CX, Shah AM, Zhang H, Faber JE, Kinter MT,
et al: Hypoxia induces heart regeneration in adult mice. Nature.
541:222–227. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yin J, Ni B, Liao WG and Gao YQ:
Hypoxia-induced apoptosis of mouse spermatocytes is mediated by
HIF-1α through a death receptor pathway and a mitochondrial
pathway. J Cell Physiol. 233:1146–1155. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhang H, Liu B, Li T, Zhu Y, Luo G, Jiang
Y, Tang F, Jian Z and Xiao Y: AMPK activation serves a critical
role in mitochondria quality control via modulating mitophagy in
the heart under chronic hypoxia. Int J Mol Med. 41:69–76.
2018.PubMed/NCBI
|
|
7
|
Kim R, Emi M and Tanabe K: Role of
mitochondria as the gardens of cell death. Cancer Chemother
Pharmacol. 57:545–553. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Balaban RS, Nemoto S and Finkel T:
Mitochondria, oxidants, and aging. Cell. 120:483–495. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Soltani M, Moghimian M, Abtahi-Eivari SH,
Shoorei H, Khaki A and Shokoohi M: Protective effects of matricaria
chamomilla extract on torsion/detorsion-induced tissue damage and
oxidative stress in adult rat testis. Int J Fertil Steril.
12:242–248. 2018.PubMed/NCBI
|
|
10
|
Lokmic Z, Musyoka J, Hewitson TD and Darby
IA: Hypoxia and hypoxia signaling in tissue repair and fibrosis.
Int Rev Cell Mol Biol. 296:139–185. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shokoohi M, Khaki A, Shoorei H, Khaki AA,
Moghimian M and Abtahi-Eivary SH: Hesperidin attenuated
apoptotic-related genes in testicle of a male rat model of
varicocoele. Andrology. 8:249–258. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ameli M, Hashemi MS, Moghimian M and
Shokoohi M: Protective effect of tadalafil and verapamil on
testicular function and oxidative stress after torsion/detorsion in
adult male rat. Andrologia. 50:e130682018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Denko NC: Hypoxia, HIF1 and glucose
metabolism in the solid tumour. Nat Rev Cancer. 8:705–713. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mandl M and Depping R: Hypoxia-inducible
aryl hydrocarbon receptor nuclear translocator (ARNT) (HIF-1β): Is
it a rare exception? Mol Med. 20:215–220. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Depping R, Jelkmann W and Kosyna FK:
Nuclear-cytoplasmatic shuttling of proteins in control of cellular
oxygen sensing. J Mol Med (Berl). 93:599–608. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brahimi-Horn MC, Bellot G and Pouyssegur
J: Hypoxia and energetic tumour metabolism. Curr Opin Genet Dev.
21:67–72. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Loor G and Schumacker PT: Role of
hypoxia-inducible factor in cell survival during myocardial
ischemia-reperfusion. Cell Death Differ. 15:686–690. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Evans RK, Schwartz DD and Gladden LB:
Effect of myocardial volume overload and heart failure on lactate
transport into isolated cardiac myocytes. J Appl Physiol (1985).
94:1169–1176. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Johannsson E, Lunde PK, Heddle C, Sjaastad
I, Thomas MJ, Bergersen L, Halestrap AP, Blackstad TW, Ottersen OP
and Sejersted OM: Upregulation of the cardiac monocarboxylate
transporter MCT1 in a rat model of congestive heart failure.
Circulation. 104:729–734. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fiume L, Manerba M, Vettraino M and Di
Stefano G: Inhibition of lactate dehydrogenase activity as an
approach to cancer therapy. Future Med Chem. 6:429–445. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ždralević M, Brand A, Di Ianni L, Dettmer
K, Reinders J, Singer K, Peter K, Schnell A, Bruss C, Decking SM,
et al: Double genetic disruption of lactate dehydrogenases A and B
is required to ablate the ‘Warburg effect’ restricting tumor growth
to oxidative metabolism. J Biol Chem. 293:15947–15961. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Urbanska K and Orzechowski A:
Unappreciated role of LDHA and LDHB to control apoptosis and
autophagy in tumor cells. Int J Mol Sci. 20:20852019. View Article : Google Scholar
|
|
23
|
Cui XG, Han ZT, He SH, Wu XD, Chen TR,
Shao CH, Chen DL, Su N, Chen YM, Wang T, et al: HIF1/2α mediates
hypoxia-induced LDHA expression in human pancreatic cancer cells.
Oncotarget. 8:24840–24852. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Santama N, Ogg SC, Malekkou A, Zographos
SE, Weis K and Lamond AI: Characterization of hCINAP, a novel
coilin-interacting protein encoded by a transcript from the
transcription factor TAFIID32 locus. J Biol Chem. 280:36429–36441.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Granneman S, Nandineni MR and Baserga SJ:
The putative NTPase Fap7 mediates cytoplasmic 20S pre-rRNA
processing through a direct interaction with Rps14. Mol Cell Biol.
25:10352–10364. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang J, Bai D, Ma X, Guan J and Zheng X:
hCINAP is a novel regulator of ribosomal protein-HDM2-p53 pathway
by controlling NEDDylation of ribosomal protein S14. Oncogene.
33:246–254. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang J, Zhang F and Zheng X: Depletion of
hCINAP by RNA interference causes defects in Cajal body formation,
histone transcription, and cell viability. Cell Mol Life Sci.
67:1907–1918. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xu R, Yu S, Zhu D, Huang X, Xu Y, Lao Y,
Tian Y, Zhang J, Tang Z, Zhang Z, et al: hCINAP regulates the
DNA-damage response and mediates the resistance of acute myelocytic
leukemia cells to therapy. Nat Commun. 10:38122019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ji Y, Yang C, Tang Z, Yang Y, Tian Y, Yao
H, Zhu X, Zhang Z, Ji J and Zheng X: Adenylate kinase hCINAP
determines self-renewal of colorectal cancer stem cells by
facilitating LDHA phosphorylation. Nat Commun. 8:153082017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Iida A, Iwagawa T, Kuribayashi H, Satoh S,
Mochizuki Y, Baba Y, Nakauchi H, Furukawa T, Koseki H, Murakami A
and Watanabe S: Histone demethylase Jmjd3 is required for the
development of subsets of retinal bipolar cells. Proc Natl Acad Sci
USA. 111:3751–3756. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Brooks GA: Energy flux, lactate shuttling,
mitochondrial dynamics, and hypoxia. Adv Exp Med Biol. 903:439–455.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bonen A: The expression of lactate
transporters (MCT1 and MCT4) in heart and muscle. Eur J Appl
Physiol. 86:6–11. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Prech M, Marszalek A, Schröder J, Filas V,
Lesiak M, Jemielity M, Araszkiewicz A and Grajek S: Apoptosis as a
mechanism for the elimination of cardiomyocytes after acute
myocardial infarction. Am J Cardiol. 105:1240–1245. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bai D, Zhang J, Li T, Hang R, Liu Y, Tian
Y, Huang D, Qu L, Cao X, Ji J and Zheng X: The ATPase hCINAP
regulates 18S rRNA processing and is essential for embryogenesis
and tumour growth. Nat Commun. 7:123102016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Leong SP, Aktipis A and Maley C: Cancer
initiation and progression within the cancer microenvironment. Clin
Exp Metastasis. 35:361–367. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Warburg O: On the origin of cancer cells.
Science. 123:309–314. 1956. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Saddik M and Lopaschuk GD: Myocardial
triglyceride turnover and contribution to energy substrate
utilization in isolated working rat hearts. J Biol Chem.
266:8162–8170. 1991.PubMed/NCBI
|
|
39
|
Liochev SI: Reactive oxygen species and
the free radical theory of aging. Free Radic Biol Med. 60:1–4.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Martinon F: Signaling by ROS drives
inflammasome activation. Eur J Immunol. 40:616–619. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hockenbery DM, Oltvai ZN, Yin XM, Milliman
CL and Korsmeyer SJ: Bcl-2 functions in an antioxidant pathway to
prevent apoptosis. Cell. 75:241–251. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jeong D, Kim TS, Lee JW, Kim KT, Kim HJ,
Kim IH and Kim IY: Blocking of acidosis-mediated apoptosis by a
reduction of lactate dehydrogenase activity through antisense mRNA
expression. Biochem Biophys Res Commun. 289:1141–1149. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Xu J, Xu X, Si L, Xue L, Zhang S, Qin J,
Wu Y, Shao Y, Chen Y and Wang X: Intracellular lactate signaling
cascade in atrial remodeling of mitral valvular patients with
atrial fibrillation. J Cardiothorac Surg. 8:342013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Gao C, Wang F, Wang Z, Zhang J and Yang X:
Asiatic acid inhibits lactate-induced cardiomyocyte apoptosis
through the regulation of the lactate signaling cascade. Int J Mol
Med. 38:1823–1830. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Qu L, Ji Y, Zhu X and Zheng X: hCINAP
negatively regulates NF-κB signaling by recruiting the phosphatase
PP1 to deactivate IKK complex. J Mol Cell Biol. 7:529–542. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yan Y, Yuan X, Xue C and He Y: Human
coilin interacting nuclear ATPase protein in cancer: Uncovering new
insights into pathogenesis and therapy. Am J Transl Res.
12:4051–4058. 2020.PubMed/NCBI
|