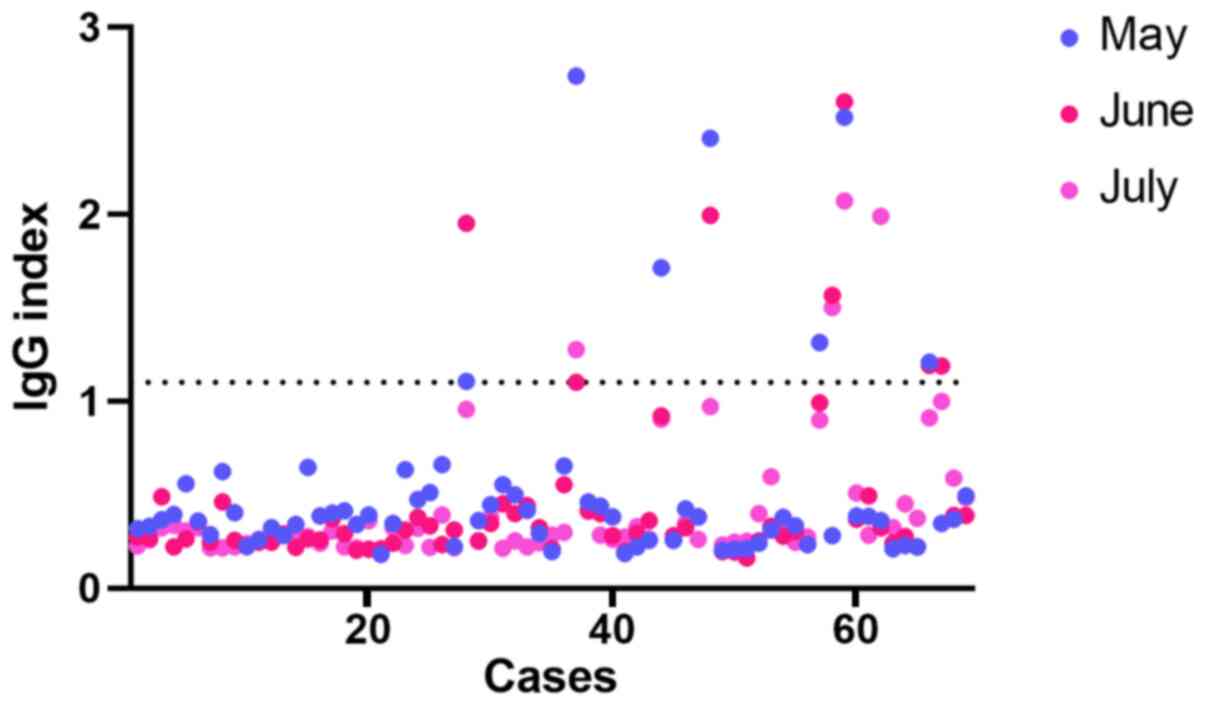
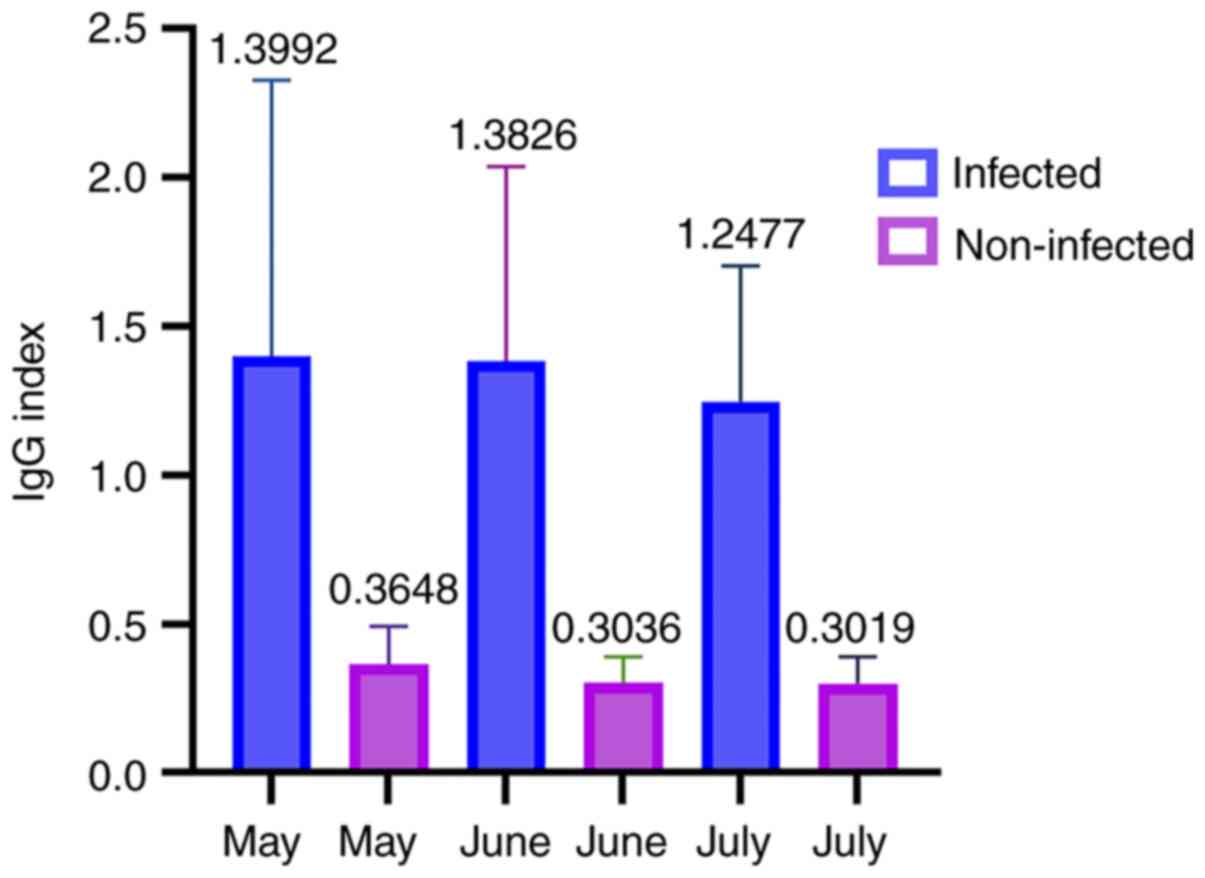
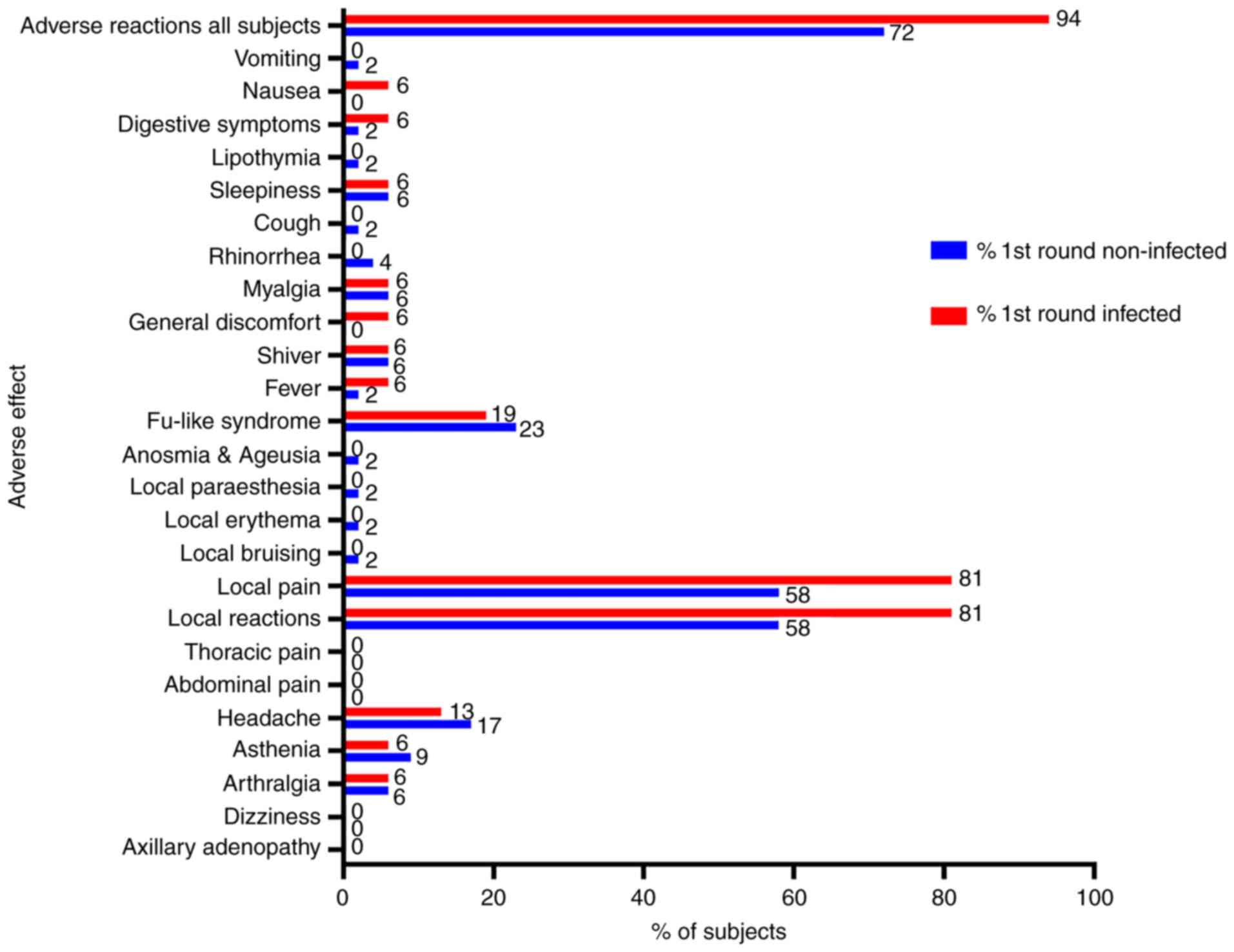
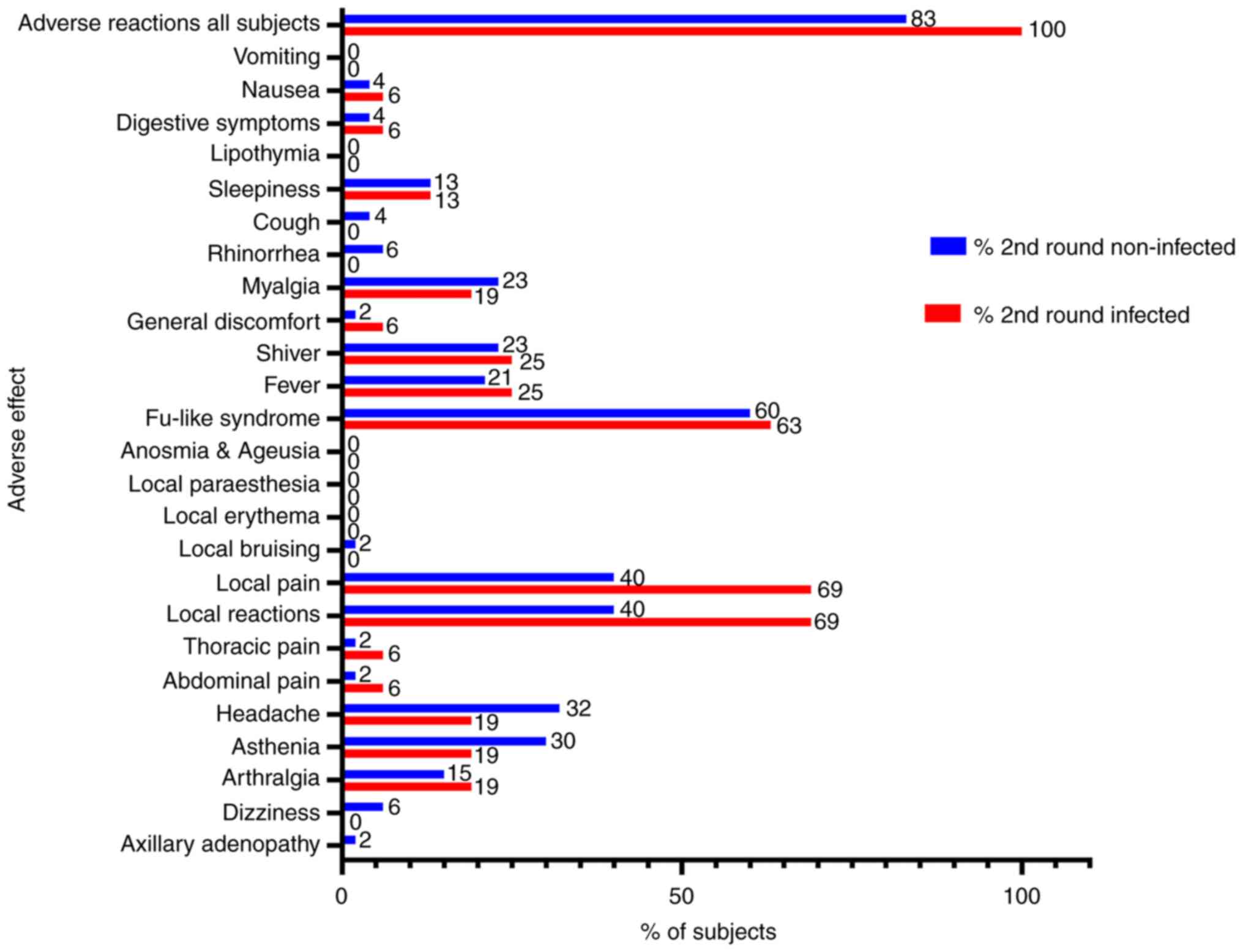
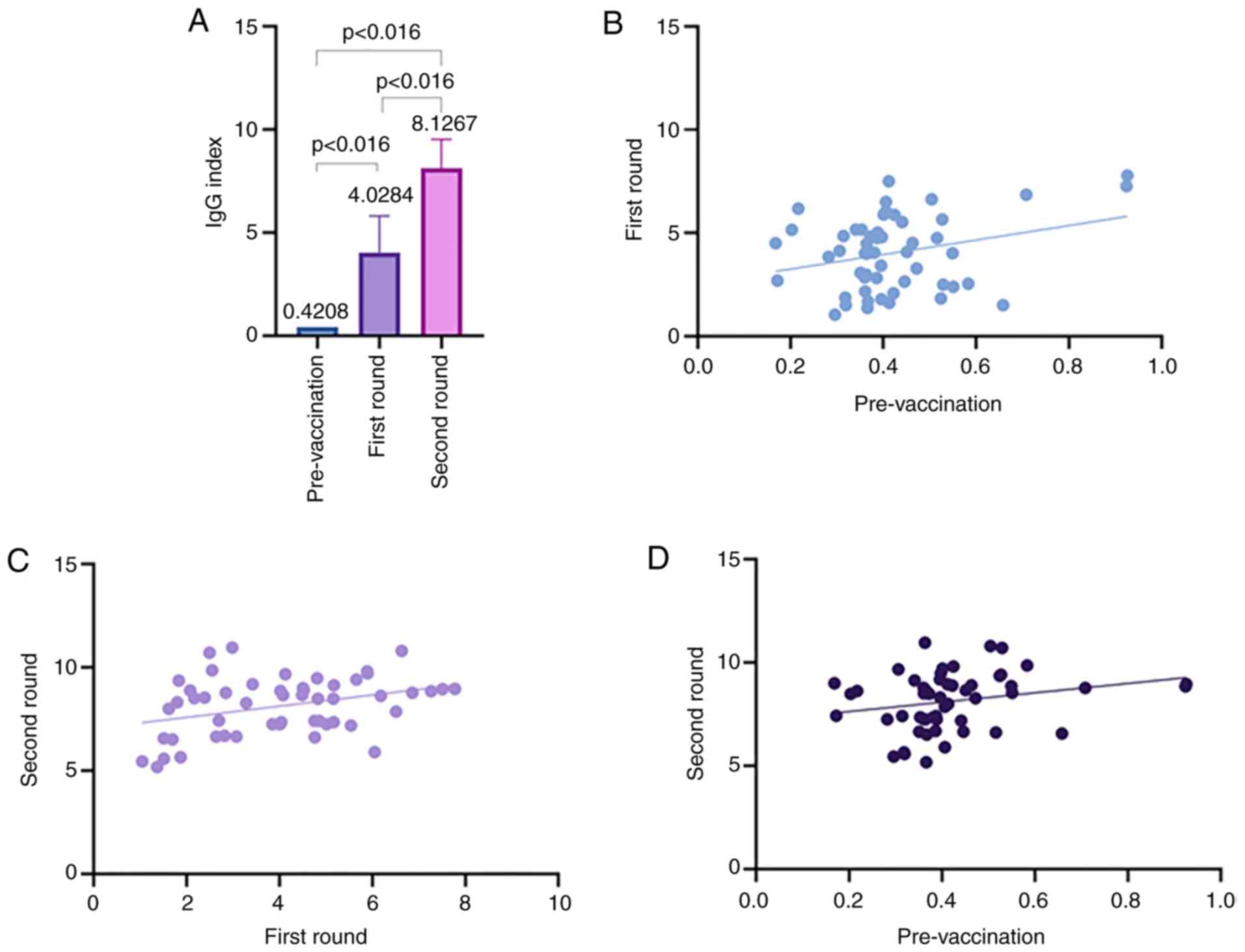
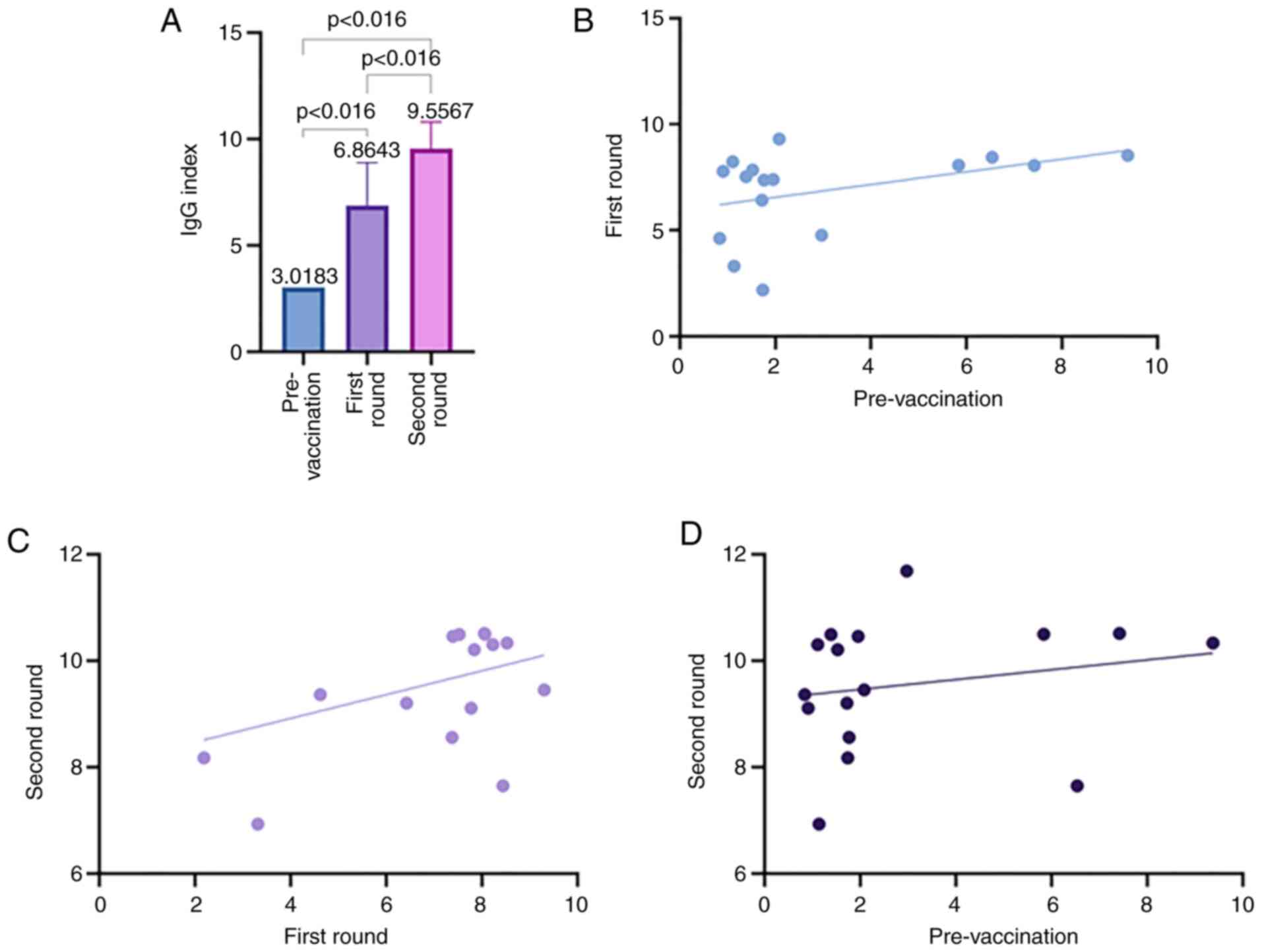
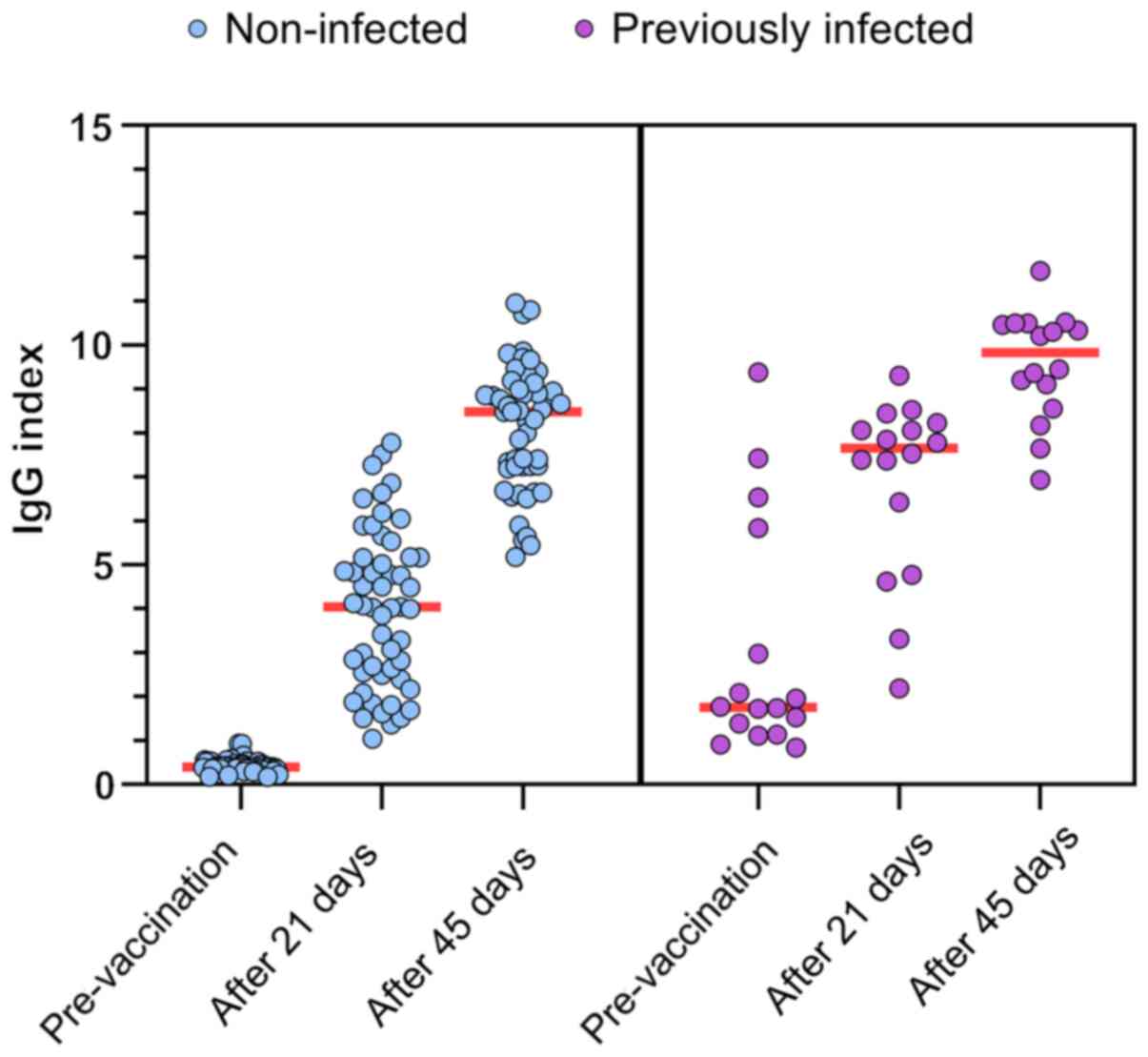
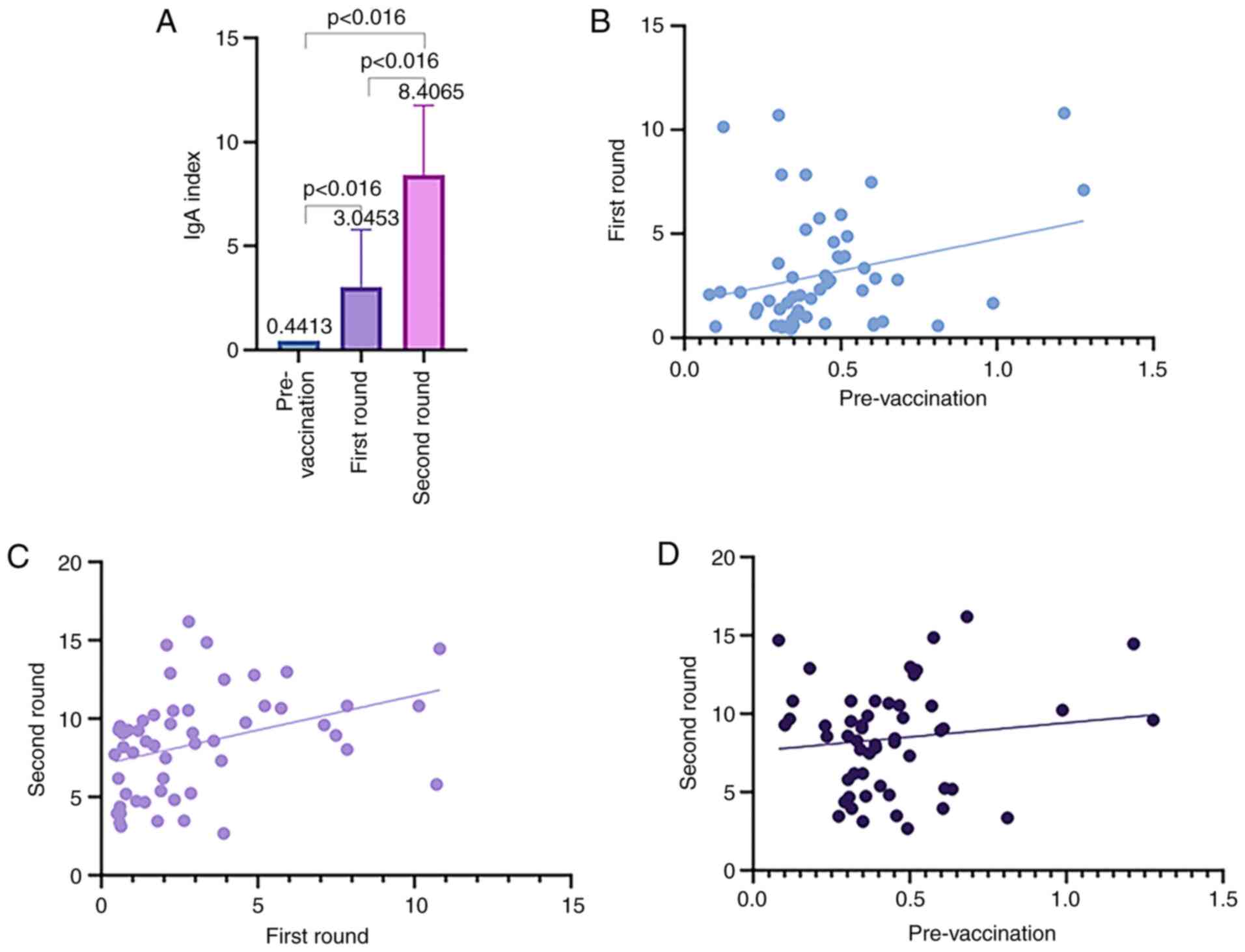
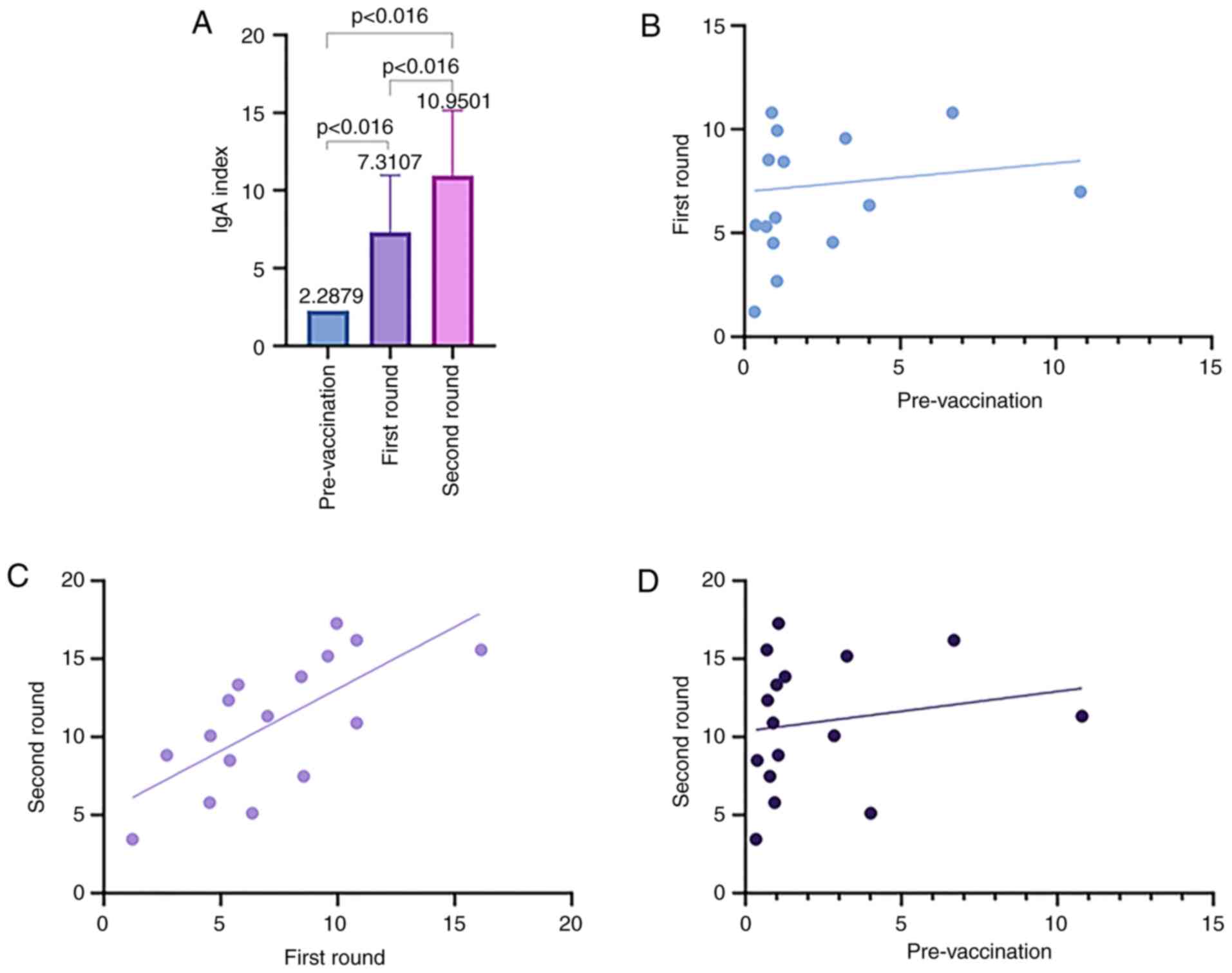
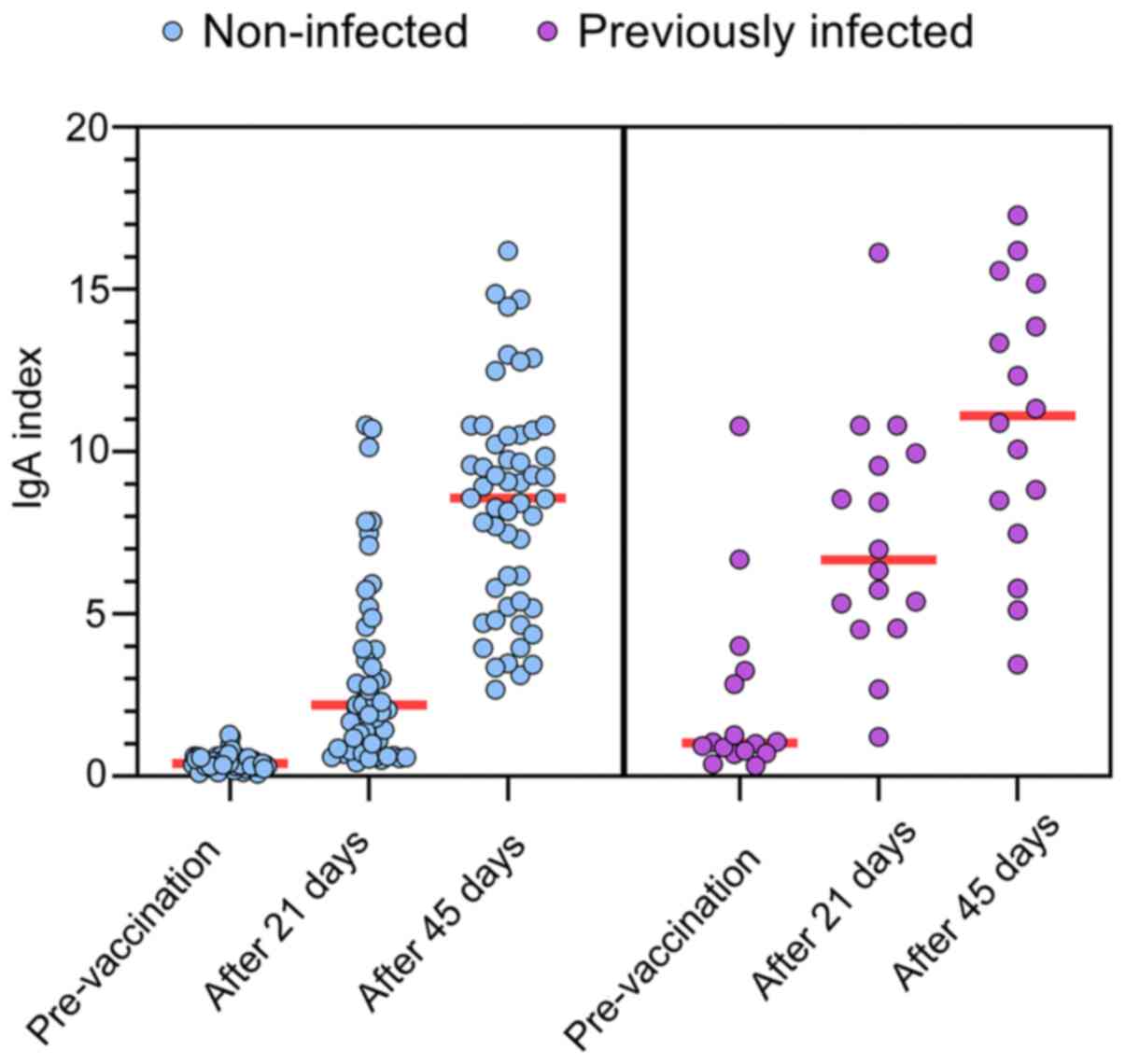
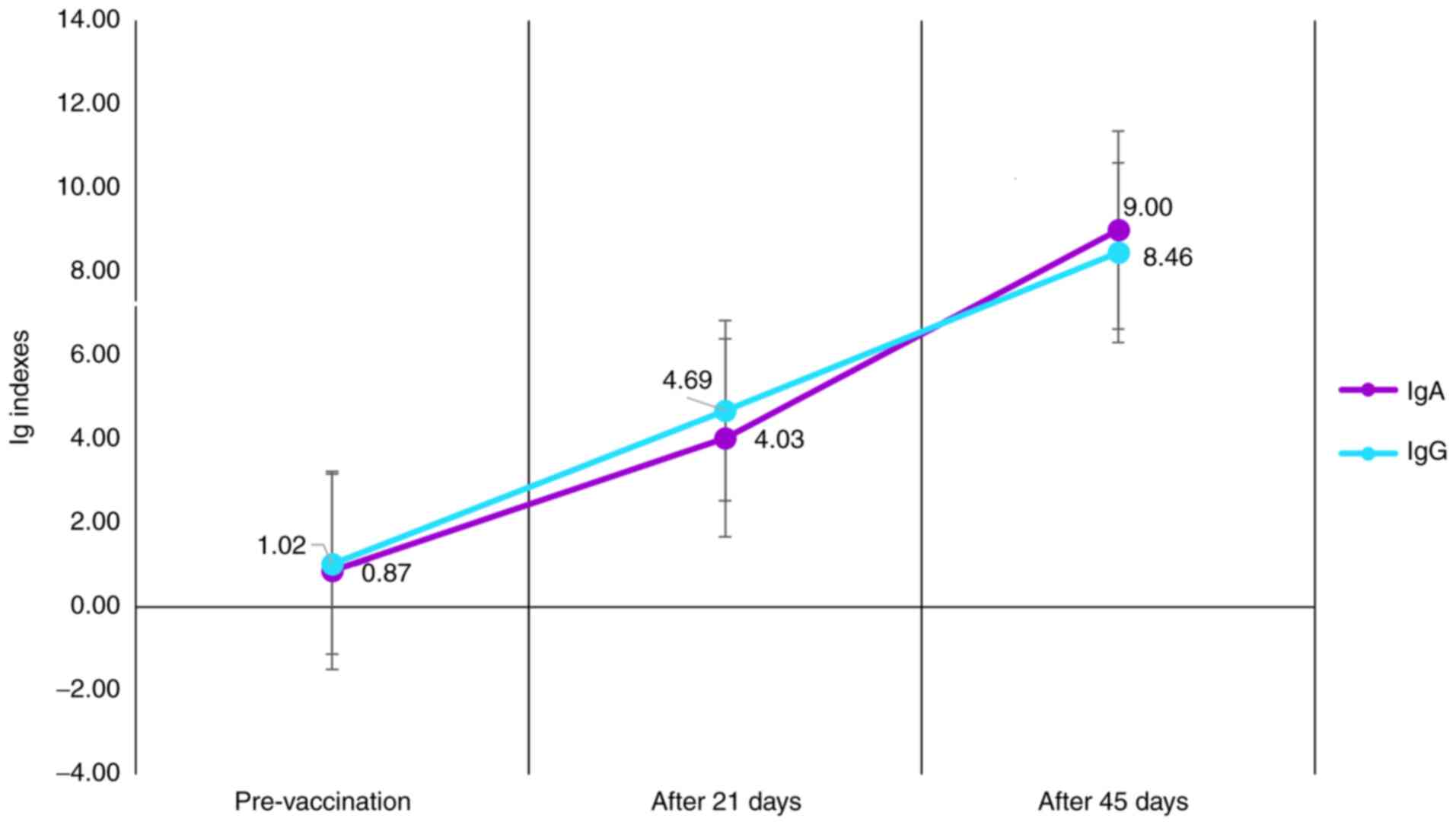
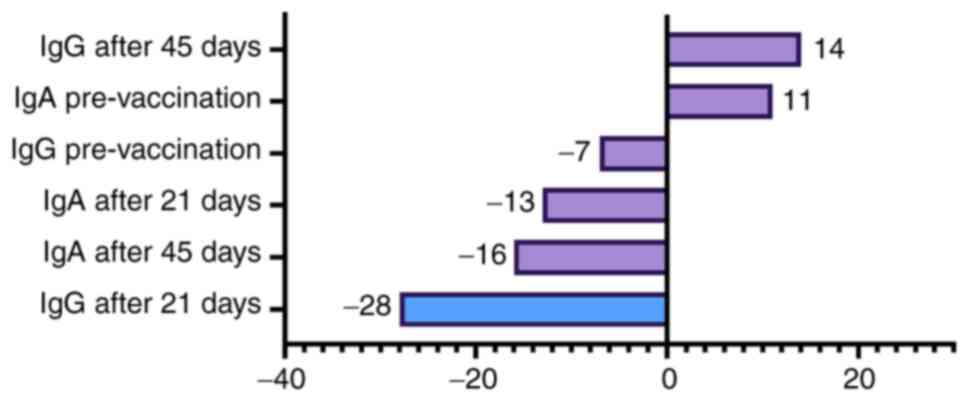
|
1
|
Constantin C, Neagu M, Diana Supeanu T,
Chiurciu V and A Spandidos D: IgY - turning the page toward passive
immunization in COVID-19 infection (Review). Exp Ther Med.
20:151–158. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Neagu M: The bumpy road to achieve herd
immunity in COVID-19. J Immunoassay Immunochem. 41:928–945. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
World Health Organization (WHO), .
COVID-19 vaccine tracker and landscape. WHO; Geneva: 2020,
https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccinesJune
8–2021
|
|
4
|
Kaur SP and Gupta V: COVID-19 vaccine: A
comprehensive status report. Virus Res. 288:1981142020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tauzin A, Nayrac M, Benlarbi M, Gong SY,
Gasser R, Beaudoin-Bussières G, Brassard N, Laumaea A, Vézina D,
Prévost J, et al: A single BNT162b2 mRNA dose elicits antibodies
with Fc-mediated effector functions and boost pre-existing humoral
and T cell responses. bioRxiv. doi: 10.1101/2021.03.18.435972
(Preprint).
|
|
6
|
Oliver SE, Gargano JW, Marin M, Wallace M,
Curran KG, Chamberland M, McClung N, Campos-Outcalt D, Morgan RL,
Mbaeyi S, et al: The Advisory Committee on Immunization Practices'
Interim Recommendation for Use of Pfizer-BioNTech COVID-19 Vaccine
- United States, December 2020. MMWR Morb Mortal Wkly Rep.
69:1922–1924. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Deva Priya SA, Kavitha S, Venugopal P,
Sriram DK and George M: Can mRNA vaccines turn the tables during
the COVID-19 pandemic? Current status and challenges. Clin Drug
Investig. Mar 23–2021.(Epub ahead of print).
|
|
8
|
Meo SA, Bukhari IA, Akram J, Meo AS and
Klonoff DC: COVID-19 vaccines: Comparison of biological,
pharmacological characteristics and adverse effects of
Pfizer/BioNTech and Moderna Vaccines. Eur Rev Med Pharmacol Sci.
25:1663–1669. 2021.PubMed/NCBI
|
|
9
|
Liu X, Wang J, Xu X, Liao G, Chen Y and Hu
CH: Patterns of IgG and IgM antibody response in COVID-19 patients.
Emerg Microbes Infect. 9:1269–1274. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Long QX, Liu BZ, Deng HJ, Wu GC, Deng K,
Chen YK, Liao P, Qiu JF, Lin Y, Cai XF, et al: Antibody responses
to SARS-CoV-2 in patients with COVID-19. Nat Med. 26:845–848. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shen L, Wang C, Zhao J, Tang X, Shen Y, Lu
M, Ding Z, Huang C, Zhang J, Li S, et al: Delayed specific IgM
antibody responses observed among COVID-19 patients with severe
progression. Emerg Microbes Infect. 9:1096–1101. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Long QX, Tang XJ, Shi QL, Li Q, Deng HJ,
Yuan J, Hu JL, Xu W, Zhang Y, Lv FJ, et al: Clinical and
immunological assessment of asymptomatic SARS-CoV-2 infections. Nat
Med. 26:1200–1204. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lou B, Li TD, Zheng SF, Su YY, Li ZY, Liu
W, Yu F, Ge SX, Zou QD, Yuan Q, et al: Serology characteristics of
SARS-CoV-2 infection after exposure and post-symptom onset. Eur
Respir J. 56:20007632020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Surcel M, Constantin C, Caruntu C, Zurac S
and Neagu M: Inflammatory cytokine pattern is sex-dependent in
mouse cutaneous melanoma experimental model. J Immunol Res.
2017:92121342017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bellenghi M, Puglisi R, Pontecorvi G, De
Feo A, Carè A and Mattia G: Sex and gender disparities in melanoma.
Cancers (Basel). 12:18192020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gebhard C, Regitz-Zagrosek V, Neuhauser
HK, Morgan R and Klein SL: Impact of sex and gender on COVID-19
outcomes in Europe. Biol Sex Differ. 11:292020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ledford H: Moderna COVID vaccine becomes
second to get US authorization. Nature. Dec 18–2020.(Epub ahead of
print). View Article : Google Scholar
|
|
18
|
Prüβ BM: Current state of the first
COVID-19 vaccines. Vaccines (Basel). 9:302021. View Article : Google Scholar
|
|
19
|
Khani E, Khiali S and Entezari-Maleki T:
Potential COVID-19 therapeutic agents and vaccines: an
evidence-based review. J Clin Pharmacol. 61:429–460. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Skelly DT, Harding AC, Gilbert-Jaramillo
J, Knight ML, Longet S, Brown A, Adele S, Adland E, Brown H;
Medawar Laboratory Team, ; Tipton T, Stafford L, Johnson SA, Amini
A; OPTIC Clinical Group, ; Tan TK, Schimanski L, Huang KYA, Rijal
P, et al: Vaccine-induced immunity provides more robust heterotypic
immunity than natural infection to emerging SARS-CoV-2 variants of
concern. Research Square. doi: 10.21203/rs.3.rs-226857/v1
(Preprint).
|
|
21
|
Hyseni I, Molesti E, Benincasa L, Piu P,
Casa E, Temperton NJ, Manenti A and Montomoli E: Characterisation
of SARS-CoV-2 lentiviral pseudotypes and correlation between
pseudotype-based neutralisation assays and live virus-based micro
neutralisation assays. Viruses. 12:10112020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ni L, Ye F, Cheng M-L, Feng Y, Deng YQ,
Zhao H, Wei P, Ge J, Gou M, Li X, et al: Detection of
SARS-CoV-2-specific humoral and cellular immunity in COVID-19
convalescent individuals. Immunity. 52:971–977.e3. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Stadlbauer D, Tan J, Jiang K, Hernandez
MM, Fabre S, Amanat F, Teo C, Arunkumar GA, McMahon M, Capuano C,
et al: Repeated cross-sectional sero-monitoring of SARS-CoV-2 in
New York City. Nature. 590:146–150. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Neagu M, Calina D, Docea AO, Constantin C,
Filippini T, Vinceti M, Drakoulis N, Poulas K, Nikolouzakis TK,
Spandidos DA, et al: Back to basics in COVID-19: Antigens and
antibodies-Completing the puzzle. J Cell Mol Med. 25:4523–4533.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Beavis KG, Matushek SM, Abeleda APF,
Bethel C, Hunt C, Gillen S, Moran A and Tesic V: Evaluation of the
EUROIMMUN Anti-SARS-CoV-2 ELISA Assay for detection of IgA and IgG
antibodies. J Clin Virol. 129:1044682020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Thakkar A, Pradhan K, Jindal S, Cui Z,
Rockwell B, Shah AP, Packer SR, Sica RA, Sparano JD, Goldstein DY,
et al: Patterns of seroconversion for SARS-CoV-2 IgG in patients
with malignant disease and association with anticancer therapy. Nat
Cancer. 2:392–399. 2021. View Article : Google Scholar
|
|
27
|
Yu HQ, Sun BQ, Fang ZF, Zhao JC, Liu XY,
Li YM, Sun XZ, Liang HF, Zhong B, Huang ZF, et al: Distinct
features of SARS-CoV-2-specific IgA response in COVID-19 patients.
Eur Respir J. 56:20015262020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ebinger JE, Fert-Bober J, Printsev I, Wu
M, Sun N, Prostko JC, Frias EC, Stewart JL, Van Eyk JE, Braun JG,
et al: Antibody responses to the BNT162b2 mRNA vaccine in
individuals previously infected with SARS-CoV-2. Nat Med. Apr
1–2021.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Walsh EE, Frenck RW Jr, Falsey AR, Kitchin
N, Absalon J, Gurtman A, Lockhart S, Neuzil K, Mulligan MJ, Bailey
R, et al: Safety and immunogenicity of two RNA-based COVID-19
vaccine candidates. N Engl J Med. 383:2439–2450. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mkaddem SB, Christou I, Rossato E,
Berthelot L, Lehuen A and Monteiro RC: IgA, IgA receptors, and
their anti-inflammatory properties. Curr Top Microbiol Immunol.
382:221–235. 2014.PubMed/NCBI
|
|
31
|
Gayet R, Michaud E, Nicoli F, Chanut B,
Paul M, Rochereau N, Guillon C, He Z, Papagno L, Bioley G, et al:
Impact of IgA isoforms on their ability to activate dendritic cells
and to prime T cells. Eur J Immunol. 50:1295–1306. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Russell MW, Moldoveanu Z, Ogra PL and
Mestecky J: Mucosal immunity in COVID-19: a neglected but critical
aspect of SARS-CoV-2 infection. Front Immunol. 11:6113372020.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Isho B, Abe KT, Zuo M, Jamal AJ, Rathod B,
Wang JH, Li Z, Chao G, Rojas OL, Bang YM, et al: Persistence of
serum and saliva antibody responses to SARS-CoV-2 spike antigens in
COVID-19 patients. Sci Immunol. 5:eabe55112020.PubMed/NCBI
|
|
34
|
Lu L, Zhang H, Zhan M, Jiang J, Yin H,
Dauphars DJ, Li SY, Li Y and He YW: Antibody response and therapy
in COVID-19 patients: What can be learned for vaccine development?
Sci China Life Sci. 63:1833–1849. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sterlin D, Mathian A, Miyara M, Mohr A,
Anna F, Claër L, Quentric P, Fadlallah J, Devilliers H, Ghillani P,
et al: IgA dominates the early neutralizing antibody response to
SARS-CoV-2. Sci Transl Med. 13:eabd22232021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Dan JM, Mateus J, Kato Y, Hastie KM, Yu
ED, Faliti CE, Grifoni A, Ramirez SI, Haupt S, Frazier A, et al:
Immunological memory to SARS-CoV-2 assessed for up to 8 months
after infection. Science. 371:eabf40632021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang Z, Lorenzi JCC, Muecksch F, Finkin S,
Viant C, Gaebler C, Cipolla M, Hoffmann HH, Oliveira TY, Oren DA,
et al: Enhanced SARS-CoV-2 neutralization by dimeric IgA. Sci
Transl Med. 13:eabf15552021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Manisty C, Otter AD, Treibel TA, McKnight
Á, Altmann DM, Brooks T, Noursadeghi M, Boyton RJ, Semper A and
Moon JC: Antibody response to first BNT162b2 dose in previously
SARS-CoV-2-infected individuals. Lancet. 397:1057–1058. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sahin U, Muik A, Derhovanessian E, Vogler
I, Kranz LM, Vormehr M, Baum A, Pascal K, Quandt J, Maurus D, et
al: COVID-19 vaccine BNT162b1 elicits human antibody and TH1 T cell
responses. Nature. 586:594–599. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lombardi A, Bozzi G, Ungaro R, Villa S,
Castelli V, Mangioni D, Muscatello A, Gori A and Bandera A: Mini
review immunological consequences of immunization with COVID-19
mRNA vaccines: preliminary results. Front Immunol. 12:6577112021.
View Article : Google Scholar : PubMed/NCBI
|