Introduction
Neurosurgery improves the survival rate and quality
of life of patients. However, the invasive nature of neurosurgery
can occasionally induce non-traumatic injury, namely, surgical
brain injury (SBI) (1). Due to
incision, electrocoagulation, bleeding and other invasive
operations in the process of brain surgery, there will be some
damage to the brain tissue surrounding the operation site, which
may lead to peripheral tissue damage, edema, apoptosis and
inflammation (2). Among these
pathologies, cerebral edema leads to postoperative neurological
dysfunction, which is one of the primary adverse reactions of SBI
(3). Thus, the reduction of
cerebral edema after SBI is beneficial for improving neural
function, postoperative recovery and patient prognosis (4,5). The
prevention and treatment of brain edema before brain surgery has
become a possible therapeutic target for reducing SBI (6). However, the mechanism and preventative
treatment of postoperative cerebral edema requires further
investigation.
As the most common complication after SBI, brain
edema is caused by the close connection of vascular endothelial
cells and the destruction of astrocytes and other components of the
blood-brain barrier (BBB), thereby damaging the BBB and
exacerbating brain injury (7).
Previous studies have shown that the WNK lysine deficient protein
kinase 3 (WNK3)/STE20/SPS1-related proline/alanine-rich kinase
(SPAK) signaling pathway serves an important role in the process of
brain edema (8–10). It has been revealed that inhibiting
the expression levels of components of this signaling pathway can
reduce BBB damage, as well as decrease the degree of brain edema
and neurobehavioral dysfunction (11). SPAK is involved in brain edema via
regulating the downstream
Na+-K+-Cl− cotransporter 1 (NKCC1)
(12). NKCC1 regulates the entry of
Na+, K+, Cl− and water into cells,
maintains concentrations of intracellular ions/water, ensures
homeostasis of the intracellular environment, contributes to
BBB-selective ion permeability and regulates cellular volume
(13,14). SPAK is a key effector in regulating
ion flow and balance (15). When
SPAK interacts with downstream NKCC1, it serves a related role in
regulating ion transport, increasing cellular volume and
influencing cellular homeostasis (11,16).
After brain injury, the expression levels of proteins in the WNK
family are upregulated, and they associate with and activate SPAK
via the phosphorylation of the T-loop at Thr233/Thr185 (17). Upon its activation through
phosphorylation by upstream kinases, phosphorylated (p)-SPAK binds
to specific peptides located in the cytosolic tail of NKCC1,
whereby it phosphorylates NKCC1 and stimulates cotransport activity
(18). Moreover, increased
expression and activity of p-SPAK/p-NKCC1 increases the entry of
extracellular ions and water into cells (19). This process induces the enlargement
of neurons, glial cells and endothelial cells, destroys the
cytoskeleton structure of cells, and leads to cell swelling and
apoptosis, which can damage the BBB, resulting in brain edema and
neurological dysfunction (11,20).
The SPAK/NKCC1 signaling pathway has been reported
to serve an important role in the pathophysiology of brain
diseases, such as stroke (21),
traumatic brain injury (22) and
subarachnoid hemorrhage (23).
However, the role and mechanism of this signaling pathway in SBI
are yet to be fully elucidated. The aim of the present study was to
investigate the influence of the SPAK/NKCC1 signaling pathway on
the BBB and neural function in a rat model of SBI. Closantel, an
anti-parasitic agent that is widely used in livestock (24), was used to inhibit SPAK. Closantel
inhibits SPAK by binding to a C-terminal domain at a highly
conserved allosteric site (25).
Furthermore, closantel has been shown to successfully inhibit SPAK,
thereby reducing p-NKCC1 expression in the aorta (26).
Materials and methods
Experimental design and groupings
The present study consisted of two separate
experiments, the designs and groupings of which are depicted in
Fig. 1A-C.
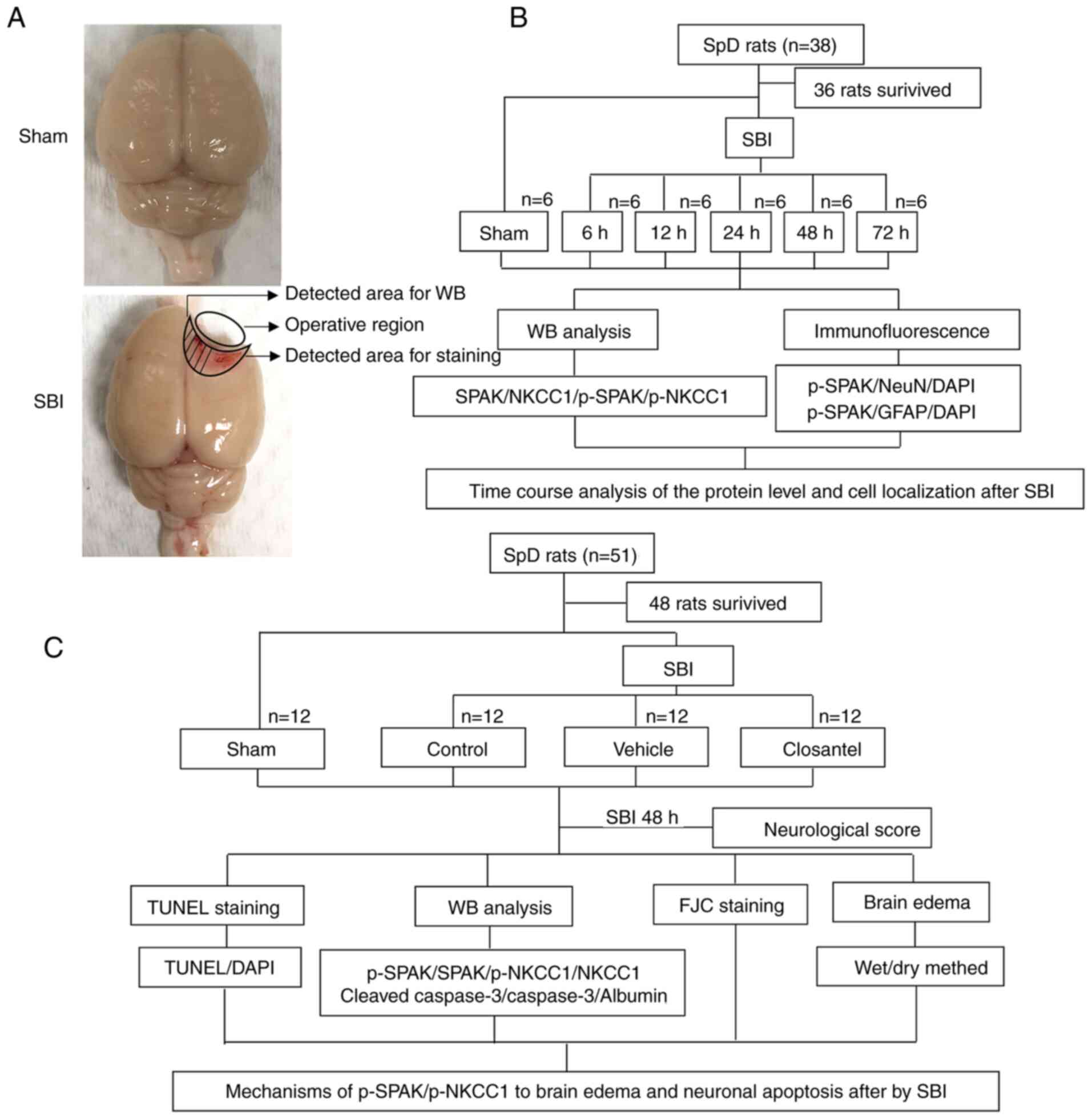 | Figure 1.SBI model and experimental design.
(A) Brain tissue from the same location in the sham group as that
injured in the SBI group was obtained for testing; some tissues
were used for WB analysis, whereas the remaining tissues were used
for staining. (B) Expression levels of SPAK, p-SPAK, NKCC1 and
p-NKCC1, as well as cellular localization of p-SPAK and p-NKCC1
were assessed after SBI to determine suitable time points for
subsequent experiments. (C) The role and mechanism of the
p-SPAK/p-NKCC1 signaling pathway in early brain injury after SBI
was investigated. SBI, surgical brain injury; p, phosphorylated;
FJC, Fluoro-Jade C; SpD, Sprague-Dawley; SPAK, STE20/SPS1-related
proline/alanine-rich kinase; NKCC1,
Na+-K+-Cl− cotransporter 1; WB,
western blot; NeuN, neuronal nuclei; GFAP, glial fibrillary acidic
protein. |
Experiment 1: There was no significant difference in
weight, food intake or exercise among the different groups. In
total there were 38 rats in experiment 1, 36 of which survived
surgeries and were randomly divided into six groups (n=6 per group)
consisting of a sham-operation group and five experimental groups
(6, 12, 24, 48 and 72 h after SBI operations). The experimental
rats were all sacrificed at their designated time points after SBI,
at which time brain tissue was harvested from around the damaged
area. Western blot (WB) analysis was performed on part of this
harvested brain tissue, whereas the rest of the brain tissue was
used for double immunofluorescence (IF) analyses.
Experiment 2: A total of 51 rats were included in
experiment 2, 48 rats survived and were randomly divided into the
following four groups (n=12 per group): i) Sham; ii) SBI; iii) SBI
+ vehicle; and iv) SBI + closantel. According to the results of
experiment 1, rats (n=6 per group) were sacrificed at 48 h after
SBI in experiment 2, and brain tissues surrounding the damaged area
were used for WB analysis, IF, TUNEL and Fluoro-Jade C (FJC)
staining assays. Brain tissue from the remaining six rats in each
group was used to measure brain edema. Neurological examinations
were performed in all groups prior to sacrifice.
Experimental animals
All experiments received approval from the Institute
of Animal Care Committee of Zhangjiagang Traditional Chinese
Medicine Hospital (approval no. 2020-10-1; Suzhou, China), and were
performed in accordance with the Chinese Association for Laboratory
Animal Sciences (27). A total of
89 male Sprague-Dawley rats (age, 8 weeks; weight, 320–350 g) were
purchased from the Zhaoyan (Suzhou) New Drug Research Center Co.,
Ltd. All rats were raised at a temperature of ~26°C, with a
humidity of ~50%, and light/dark periods identical to outdoor
conditions. Food and water were provided ad libitum.
Establishment of SBI model in
rats
A rat model of SBI was established as previously
reported (28). Sprague-Dawley rats
were anesthetized via an intraperitoneal injection of sodium
pentobarbital (10 mg/ml; 40 mg/kg). The rats were then prone and
fixed in a stereotaxic apparatus (Yuyan Instruments). An incision
was made along the midline of the skin above the brain to expose
the skull in order to identify bregma. Then, a 5×5 mm craniotomy
was performed on the right frontal skull by removing a bone flap
using a bone drill from the right skull bone 2-mm along the
sagittal suture and 1-mm along coronal suture. A durotomy was
performed and 2×3 mm brain tissue was excised via sharp dissection.
An electrocautery unit was used to stop bleeding, after which the
area was rinsed with normal saline. In the sham rats, the same
surgical method was used; a craniotomy was performed to remove the
bone flap, but part of the right frontal lobe was not removed.
Important parameters of interest were monitored during and after
surgery, including heart rate, body temperature and body weight.
According to the experimental requirements, the rats were
sacrificed via an intraperitoneal injection of sodium pentobarbital
(20 mg/ml; 150 mg/kg) at different time points (Fig. 1).
Drug injections
The experimental 2 rats were divided into the
following four groups: i) Sham; ii) SBI; iii) SBI + vehicle; and
iv) SBI + closantel. The SBI + closantel group was
intraperitoneally injected with 20 mg/kg closantel (Abcam) at 30
min before SBI, as previously described (26). The weight-matched SBI + vehicle
group was intraperitoneally injected with an equal volume of 10%
DMSO.
Neurological scoring
The neurological function of each rat was evaluated
according to the scale before sacrifice. Neurological scoring was
conducted as previously reported (29) and consisted of the following seven
components: i) Symmetry of limb movement; ii) forelimb-stretching
exercises; iii) lateral turning; iv) climbing; v) body movements;
vi) proprioception; and vii) responses to vibrissae touch. Scores
on each subtest ranged from 0–3, with a combined maximum score of
21. Higher scores were indicative of reduced neurological damage
(i.e., 21, no neurological deficits).
Tissue collection and sectioning
To isolate proteins, rats were perfused with 200 ml
0.9% normal saline (at 4°C) through the heart and cortical samples
<3 mm from the contusion edge were collected on ice (Fig. 1A). Some brain tissue samples (n=6
rats per group) were immediately frozen and stored at −80°C until
subsequent WB analysis. To obtain brain sections, brains were
harvested, immersed in 4% paraformaldehyde at 4°C for >48 h and
then embedded in paraffin. Paraffin-embedded brain sections were
sectioned using a paraffin slicing machine to a thickness of 5-µm
each. All procedures for tissue removal and selection were
performed by two pathologists who were blinded to the experimental
conditions.
WB analysis
WB analysis was performed as described previously
(30). The extracted brain tissue
was homogenized in tissue protein-extraction reagent with a
protease inhibitor cocktail (CWBio) and incubated on ice for 20
min. Subsequently, the homogenized brain tissue was centrifuged at
12,000 × g at 4°C for 20 min. The supernatant was then collected
and protein concentrations were determined using the Pierce TM BCA
protein-detection kit (Thermo Fisher Scientific, Inc.). Then, 8, 10
and 12% gels (Beyotime Institute of Biotechnology) were used
according to the different molecular weights. The extracted
proteins were loaded onto SDS-polyacrylamide gels at 3 mg per lane.
These electrophoresed proteins were then transferred to a PVDF
membrane (EMD Millipore). WB quick seal liquid (Beyotime Institute
of Biotechnology) was used to block the PVDF membrane for 30 min at
room temperature. Samples were then incubated with primary
antibodies at 4°C overnight, including: Rabbit anti-SPAK (1:1,000;
cat. no. 2281; Cell Signaling Technology, Inc.), mouse anti-NKCC1
(1:500; cat. no. sc-514774; Santa Cruz Biotechnology, Inc.), sheep
anti-p-SPAK (1:400; cat. no. S668B; MRC Protein Phosphorylation and
Ubiquitylation Unit), sheep anti-p-NKCC1 (1:400; cat. no. S763B;
MRC Protein Phosphorylation and Ubiquitylation Unit), rabbit
anti-GAPDH (1:5,000; cat. no. G9545; Sigma-Aldrich; Merck KGaA),
rabbit anti-Albumin (1:1,000; cat. no. ab207327; Abcam), rabbit
anti-cleaved-Caspase-3 (1:1,000; cat. no. 9664; Cell Signaling
Technology, Inc.) and rabbit anti-Caspase-3 (1:1,000; cat. no.
ab184787; Abcam). After washing three times with PBS, samples were
incubated with secondary antibodies for 2 h at 4°C, including goat
anti-mouse IgG-HRP (1:5,000; cat. no. 91196; Cell Signaling
Technology, Inc.), goat anti-rabbit IgG-HRP (1:5,000; cat. no.
31466; Invitrogen; Thermo Fisher Scientific, Inc.) and rabbit
anti-sheep IgG-HRP (1:2,000; cat. no. 81-8620; Thermo Fisher
Scientific, Inc.). Immunoblots were probed with the Immobilon
Western Chemiluminescent HRP substrate (EMD Millipore) and
visualized using an imaging system (Teledyne Photometrics). All
data were analyzed using ImageJ software (v 1.8.0; National
Institutes of Health). GAPDH was used as the loading control.
IF staining
Double-IF staining was conducted as previously
described (31). The
paraffin-embedded sections were heated at 70°C for 1 h in an oven,
soaked in xylene, rehydrated in anhydrous ethanol, 95, 85 and 70%
ethanol, and then repaired with sodium citrate. Following three
washes with PBS, the membranes were permeabilized using
immunostaining permeable solution (Beyotime Institute of
Biotechnology). The brain sections were covered for ≥30 min with
immunostaining-blocking solution (Beyotime Institute of
Biotechnology) at room temperature, after which samples were
incubated with primary antibodies at 4°C overnight. After three
washes in PBS, samples were incubated with secondary antibodies at
room temperature for 1 h. Finally, the samples were counterstained
with DAPI (Shanghai Yeasen Biotechnology Co., Ltd.) at room
temperature for 10 min and observed under a fluorescent microscope
(Olympus Corporation). The following antibodies were used: Sheep
anti-p-SPAK (1:100; cat. no. S668B; MRC Protein Phosphorylation and
Ubiquitylation Unit), mouse anti-glial fibrillary acidic protein
(GFAP; 1:400; cat. no. 14-9892-95; Invitrogen; Thermo Fisher
Scientific, Inc.), mouse anti-neuronal nuclei (NeuN; 1:1,000; cat.
no. ab104224; Abcam), donkey anti-Sheep IgG, Alexa Fluor 488
(1:800; cat. no. A-11015; Invitrogen; Thermo Fisher Scientific,
Inc.) and donkey anti-mouse IgG, Alexa Fluor 555 (1:800; cat. no.
A32787TR; Invitrogen; Thermo Fisher Scientific, Inc.).
TUNEL staining
Apoptosis was detected in the paraffin-embedded
sections using a TUNEL Apoptosis Detection Kit (Beyotime Institute
of Biotechnology) according to the manufacturer's protocols. The
paraffin-embedded section dewaxing steps were performed according
to the aforementioned protocol described in the IF staining
section. Sections were washed with distilled water for 2 min and
then incubated in a protease-K working solution for 30 min at 37°C.
Thereafter, samples were washed three times in PBS (5 min each
time). Brain sections were covered with TUNEL working solution and
incubated in a damp box at 37°C in the dark for 1 h. Samples were
then washed three times in PBS (5 min each time). Finally, the
mounting medium containing DAPI (Shanghai Yeasen Biotechnology Co.,
Ltd.) was applied at room temperature for 10 min and observed under
a fluorescent microscope (Olympus Corporation). Each brain tissue
was observed under a microscope in a random selection of six fields
of vision around the damaged area.
FJC staining
FJC Ready-to-Dilute Staining Kit (Biosensis Pty,
Ltd.) was used as per the manufacturer's instructions.
Paraffin-embedded sections were incubated in an oven at 70°C for 1
h. Then, samples were rehydrated in a descending alcohol series
consisting of xylene, 100% ethanol and 70% ethanol. Subsequently,
samples were washed twice with double-distilled H2O for
1 min at room temperature before being cleaned with distilled water
for 2 min at room temperature. Then, nine parts of distilled water
were mixed with one part of solution B (potassium permanganate),
which the slides were then incubated in for 10 min at room
temperature. Subsequently, slides were rinsed for 2 min in
distilled water. Then, nine parts of distilled water were mixed
with one part of solution C, and samples were incubated in the dark
for 10 min at room temperature. After washing three times with
distilled water, samples were dried in an oven at 60°C for ≥5 min.
Then, samples were soaked in xylene for 5 min at room temperature.
After drying, samples were sealed with neutral resin in liquid
(Shanghai Yeasen Biotechnology Co., Ltd.) and observed under a
fluorescent microscope.
Brain edema
Brain edema was assessed by determining the moisture
content of the brain using the wet-dry method (32,33).
After separation of rat brain tissues, the brains were divided into
ipsilateral and contralateral hemispheres, and were quickly weighed
to determine their wet weights. Then, the brain samples were dried
in an oven at 100°C for 24 h, and subsequently weighed to determine
their dry weights. The percentage of brain water content (%) was
calculated as follows: [(Wet weight-dry weight)/(wet weight)]
×100%.
Statistical analysis
The experiments were repeated six times. All data
are presented as the mean ± SD. GraphPad Prism 8.0 software
(GraphPad Software, Inc.) was used for all statistical analyses.
One-way ANOVA followed by Tukey's post hoc test was used for
multiple comparisons to determine the differences among all groups.
IF staining were analyzed using an unpaired Student's t-test.
Neurological behavioral scores among different groups were analyzed
using the Kruskal-Wallis test followed by Dunn's post hoc test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Post-SBI protein expression levels of
SPAK, p-SPAK, NKCC1 and p-NKCC1 in the brain
The expression levels of SPAK, p-SPAK, NKCC1 and
p-NKCC1 at 6, 12, 24, 48 and 72 h after SBI, as well as in sham
rats, were assessed via WB analysis. Compared with those in the
sham group, there was no significant change in SPAK or NKCC1
expression, whereas p-SPAK expression was significantly increased
beginning at 6 h after SBI and reaching a peak at 48 h post-SBI
(Fig. 2A and B). Additionally,
compared with the sham group, p-NKCC1 expression was significantly
increased beginning at 12 h after SBI and reaching a peak at 48 h
post-SBI (Fig. 2C and D).
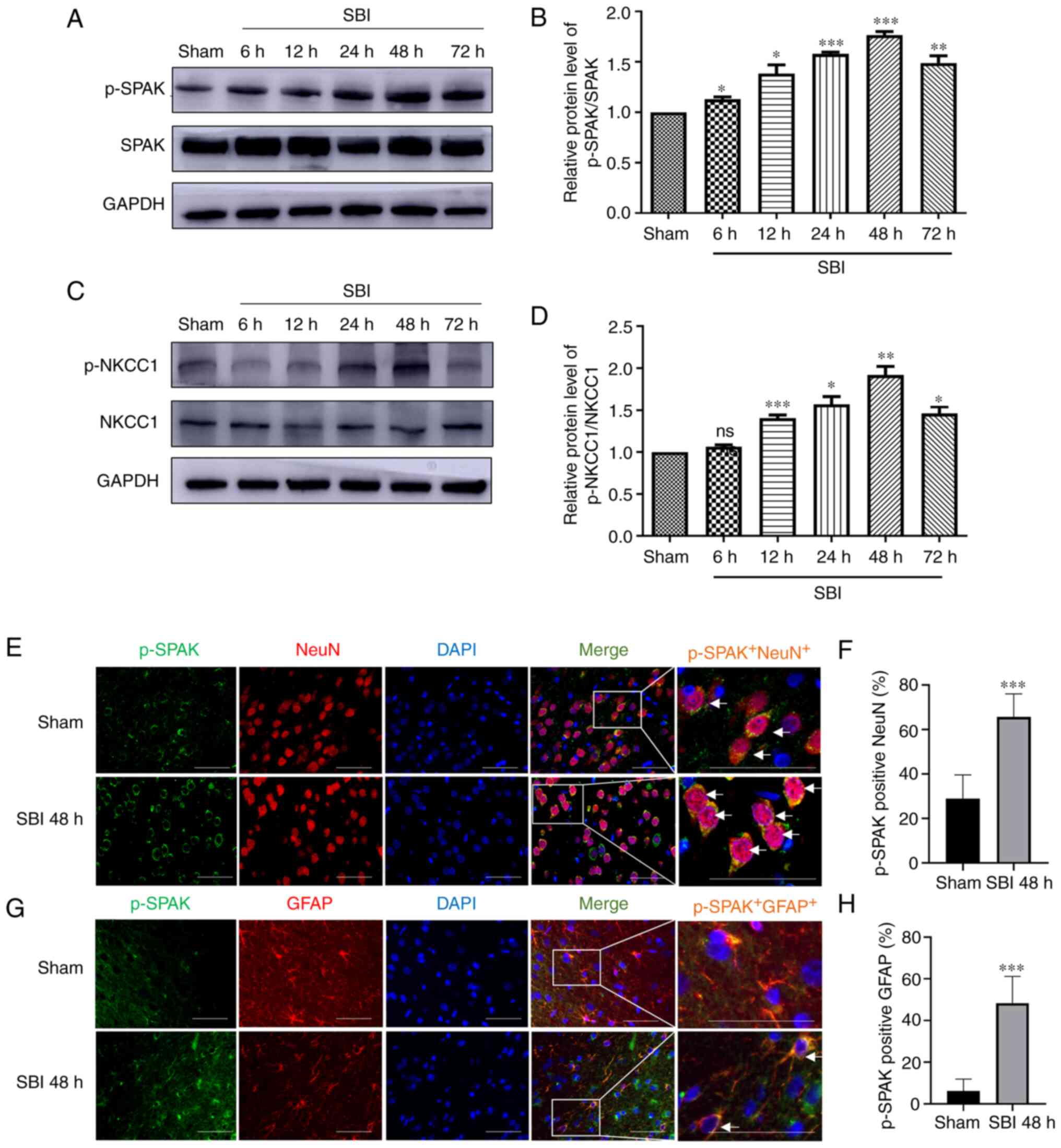 | Figure 2.Protein expression levels of SPAK,
p-SPAK, NKCC1 and p-NKCC1 in the lesion of the peripheral cortex
after SBI injury. SPAK/p-SPAK protein expression levels at 6, 12,
24, 48 and 72 h in the SBI groups and in the sham group were (A)
determined by WB analysis and (B) semi-quantified. p-NKCC1/NKCC1
protein expression levels at 6, 12, 24, 48 and 72 h in the SBI
groups and in the sham group were (C) determined by WB analysis and
(D) semi-quantified. The relative densities of each protein were
standardized to those of the sham group. ImageJ software was used
to semi-quantify the protein expression, and the mean value of the
sham group was normalized to 1. Statistical analysis was performed
using one-way ANOVA followed by Tukey's post hoc test. Double IF
staining images of p-SPAK in the peripheral cortex of the damaged
area. In the sham group and at 48 h after SBI, the expression
profiles of green-labeled p-SPAK and red-labeled (E and F) NeuN/(G
and H) GFAP were identified by performing double IF staining. The
nuclei were labeled with DAPI (blue) fluorescence. Arrows indicate
the co-localization of p-SPAK with NeuN/GFAP. Scale bar, 50 µm; The
images for p-SPAK, NeuN/GFAP, DAPI and Merge are at ×400
magnification. Statistical analyses were performed using an
unpaired Student's t-test. Data are presented as the mean ± SD (n=6
per group). *P<0.05, **P<0.01 and ***P<0.001 vs. sham.
SPAK, STE20/SPS1-related proline/alanine-rich kinase; NKCC1,
Na+-K+-Cl− cotransporter 1; NeuN,
neuronal nuclei; GFAP, glial fibrillary acidic protein; SBI,
surgical brain injury; p, phosphorylated; IF, immunofluorescence;
ns, not significant; WB, western blot. |
Post-SBI p-SPAK expression in
peri-injury cortical cells
p-SPAK expression was assessed via IF staining with
a NeuN marker and astrocytic (GFAP) marker. The expression level of
p-SPAK in NeuN+ cells is shown in Fig. 2E and F, and that in GFAP+
cells is presented in Fig. 2G and
H. The analyses revealed that the numbers of p-SPAK+
neurons and astrocytes in the 48 h post-SBI group were
significantly increased compared with those of the sham group.
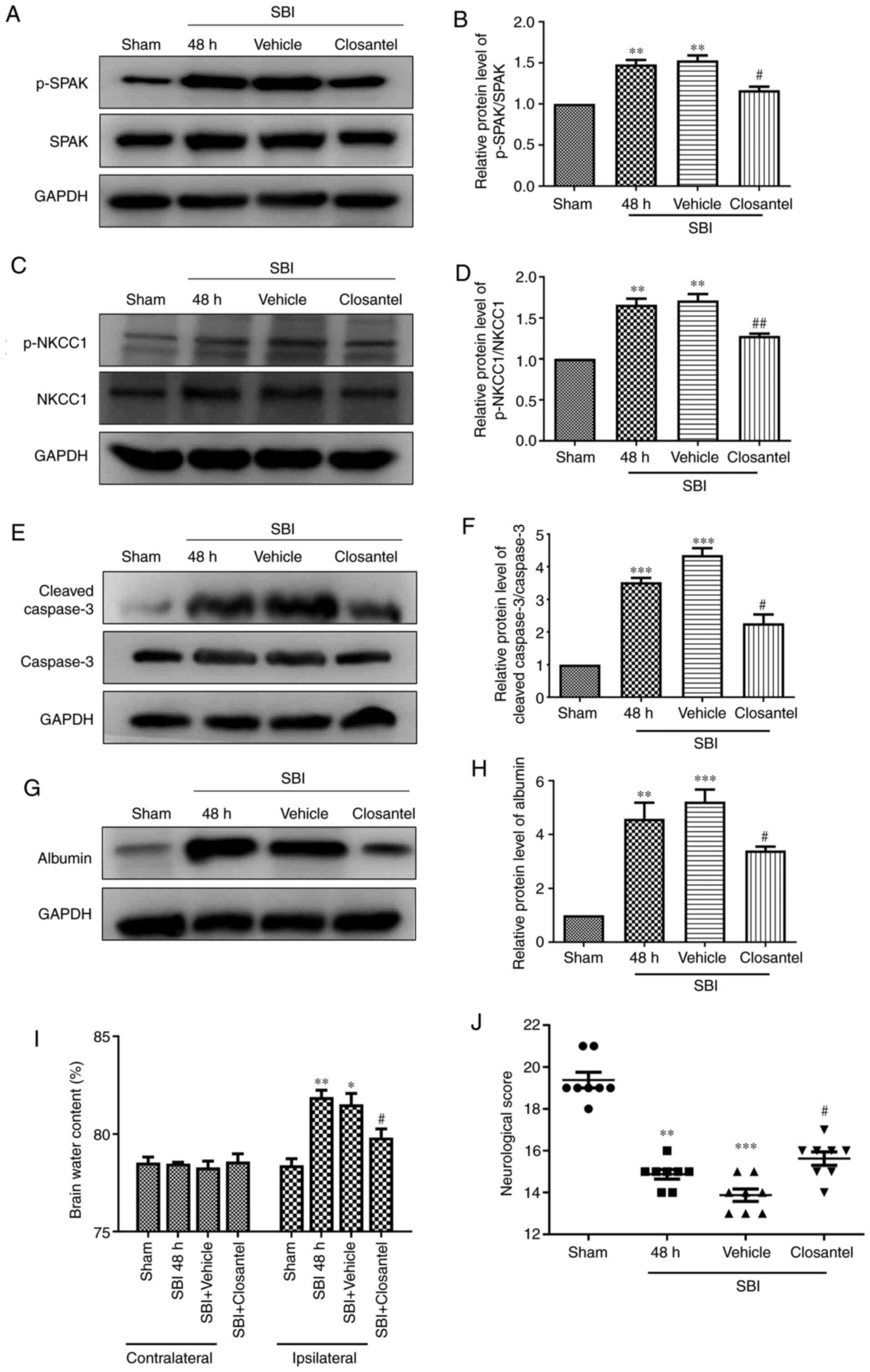
Closantel decreases p-SPAK and p-NKCC1
expression after SBI
It was found that p-SPAK expression was
significantly higher in the SBI and SBI + vehicle groups compared
with that in the sham group (Fig. 3A
and B). Moreover, after closantel intervention, p-SPAK
expression was significantly decreased in the closantel group
compared with that in the SBI + vehicle group, as demonstrated by
WB analysis. Additionally, after closantel intervention, p-NKCC1
expression was significantly decreased compared with that in the
SBI + vehicle group (Fig. 3C and
D). The trend for cleaved-caspase-3 expression was similar to
that of p-SPAK expression across these groups (Fig. 3E and F).
Closantel ameliorates the BBB and
neurobehavioral scores in SBI model rats
Then, the integrity of the BBB was assessed by
measuring albumin and brain wet-dry specific weight. Albumin
expression was significantly higher in the SBI group and SBI +
vehicle group compared with that in the sham group (Fig. 3G and H). After closantel
intervention, albumin expression was significantly decreased in the
closantel group compared with that in the SBI + vehicle group.
After SBI, brain edema in the damaged hemispheres
was significantly reduced by closantel intervention (Fig. 3I). However, there was no significant
change in brain water content in the contralateral hemispheres.
These results confirmed that there was a significant impairment of
the BBB after SBI, which was mitigated following closantel
intervention. Compared with those of the sham group, the
neurological scores of rats in the SBI group and SBI + vehicle
group were significantly lower (Fig.
3J). Furthermore, compared with those of the SBI + vehicle
group, the neurobehavioral scores of rats were significantly
improved in the SBI + closantel group. These results indicated that
the neurobehavioral scores were significantly improved after SBI +
closantel intervention.
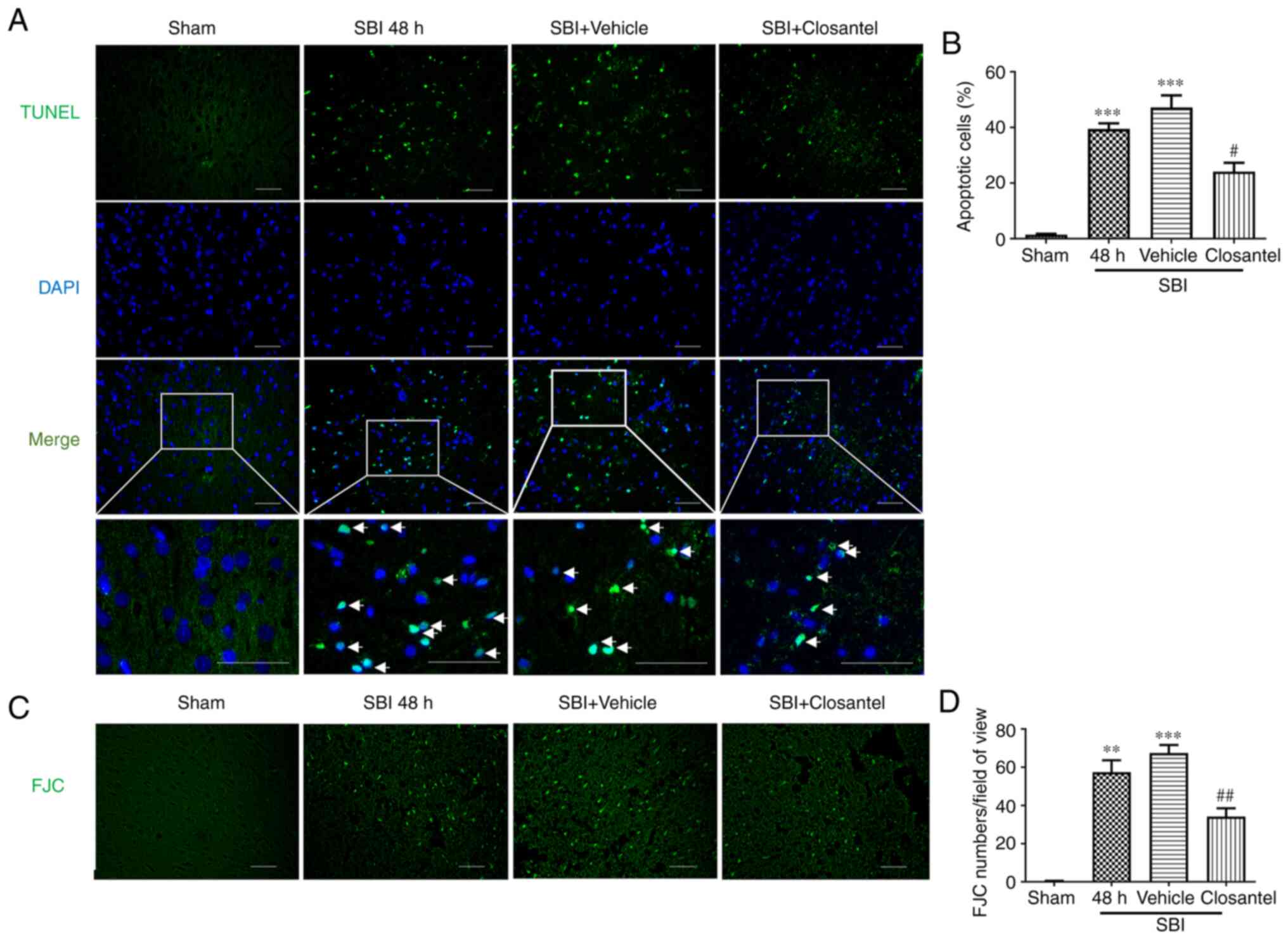
Effect of closantel intervention on
neuronal degeneration and apoptosis after SBI
TUNEL staining was used to detect apoptotic cells in
the brain tissue surrounding the injured area. The degree of
neuronal apoptosis in the SBI and SBI + vehicle groups was
significantly higher compared with that in the sham group (Fig. 4A and B). Additionally, the degree of
apoptosis in the SBI + closantel group was significantly lower
compared with that in the SBI + vehicle group. Neuronal
degeneration was detected via FJC staining. The trend for FJC
staining was similar to the TUNEL staining results across these
groups (Fig. 4C and D). These
results suggested that closantel intervention significantly
improved neuronal degeneration and apoptosis in SBI model rats.
Discussion
The present study investigated the neuroprotective
effect of inhibiting SPAK in the SBI rat model and evaluated its
potential mechanism. The current results demonstrated that,
compared with the sham group, the protein expression level of
p-SPAK began to increase at 6 h after SBI and reached a peak by 48
h, after which it began to decline. Furthermore, inhibition of
p-SPAK expression significantly reduced albumin expression, cell
apoptosis, brain edema and neurological impairment in SBI model
rats. After SBI, p-NKCC1 expression increased slightly later
compared with that of p-SPAK. Furthermore, when p-SPAK expression
was decreased, p-NKCC1 expression was also decreased. These results
indicated that, in the current SBI rat model, NKCC1 may be located
downstream of SPAK and may be regulated by SPAK. Moreover,
inhibiting p-SPAK may reduce secondary brain injury caused by
SBI.
Secondary brain edema is caused by the destruction
of the original ion homeostasis, which leads to changes to brain
tissue cell volume and homeostasis (34). Leakage of the BBB leads to albumin
extravasation, which in turn increases inflammation and edema, thus
exacerbating brain damage (35,36).
Lu et al (37) reported that
the mRNA and protein expression levels of NKCC1 were upregulated in
the neurons of animals with traumatic brain injury, and the animals
displayed severe brain edema and neuronal damage. NKCC1 belongs to
the cation-Cl− cotransporters and is an important
determinant of ion homeostasis in the brain (38). In the central nervous system,
intracellular Cl− concentrations are determined by
NKCC1, and the two chloride ion channels have been identified as
novel targets for the treatment of cerebral edema (23,39).
SPAK is the primary regulator of cation-chloride cotransporters and
stimulates NKCC1 ion influx via phosphorylation (40). While investigating the activation
mechanism of NKCC1, Thastrup et al (41) revealed that NKCC1 could not be
phosphorylated or activated during the deletion of SPAK/odd-skipped
related transcription factor 1 (OSR1), and provided genetic
evidence to confirm that SPAK/OSR1 serves an important role in
controlling NKCC1 function. SPAK/NKCC1 regulates the coordinated
transmembrane inflow and outflow of ions and water, which is
necessary for the homeostasis of the volume of neurons, glial
cells, endothelial cells and other cells, as well as for the
integrity of the BBB (38).
Furthermore, SPAK phosphorylation of NKCC1 has been associated with
a number of neurological diseases, such as intracerebral hemorrhage
(11) and schizophrenia (42). After ischemic stroke and other brain
injuries, NKCC1 regulation of cell permeability changes the levels
of extracellular fluid in inflammatory cells and blood
extravasation, resulting in brain edema, inflammation and other
secondary brain injury (43,44).
A study examining cerebral ischemia suggested that
the WNK3/SPAK/NKCC1 signaling pathway is one of the key
contributors to the formation of brain edema (45); a finding which was consistent with
the current results. The present study found that the expression
levels of p-SPAK in astrocytes and albumin exudation were increased
after SBI in rats, which indicated that the BBB was destroyed,
leading to brain edema. This phenomenon may be due to the effect of
the increase of p-NKCC1 on cell ion transport (40). Previous studies have shown that
inhibition of NKCC1 phosphorylation has a protective effect on
brain edema, the mechanism of which is by reducing astrocyte
swelling and BBB destruction (46–48).
p-NKCC1 regulates the entry of Na+, K+,
Cl− and water into cells, changes the cellular volume
and cytoskeletal structure of damaged cells, and can induce damage
to astrocytes (47,49). Astrocytes are one of the components
of BBB, and their apoptosis will lead to the destruction of BBB
(50). Thus, the present study
evaluated whether BBB function was impaired in SBI model rats using
indicators associated with brain edema. It was identified that
after SBI, albumin levels significantly rose and brain water
content significantly increased, indicating that the function of
the BBB was impaired. Furthermore, closantel inhibited SPAK
activity, thereby reducing brain edema in SBI model rats.
Previous studies have reported that there was a
significant correlation between brain edema and some neurological
dysfunction after SBI injury in rats (51,52).
The present results suggested that, compared with those in the sham
group, neurobehavioral scores were significantly reduced in rats
after SBI, which may be due to the degeneration and apoptosis of a
large number of nerve cells after SBI, as well as the aggravation
of brain edema, leading to neurological dysfunction. In a previous
study of ischemic injury, SPAK knockout mice showed a 50% reduction
in infarct size and cerebral edema, as well as significantly
improved neurological function (8).
The same result was observed when SPAK activity was inhibited in
the current study. The present study revealed that after inhibiting
p-SPAK using closantel, the expression level of p-NKCC1 was also
decreased in SBI model rats. Compared with the SBI + vehicle group,
albumin release was significantly decreased in SBI model rats
treated with closantel. In addition, the apoptosis index was
significantly decreased, the brain water content of the injured
side was significantly decreased and the neurological function
score was significantly increased in SBI model rats treated with
closantel. These results suggested that inhibiting p-SPAK may
mitigate SBI-induced secondary cerebral edema and reduce neuronal
apoptosis, as well as ameliorate neurological dysfunction.
The mechanism of SBI causing secondary brain injury
is highly complex. In the present study, p-SPAK was greatly
increased after SBI, and the phosphorylation of the downstream
p-NKCC1 signal was increased accordingly. A large number of ions
and water enter nerve cells, which increases the cell volume and
disrupts the cell structure, causing degeneration and apoptosis,
destruction of the BBB, brain edema and neurological dysfunction,
as demonstrated in the current study (Fig. 5). In the present study, it was
identified that inhibition of the SPAK/NKCC1 signaling pathway
alleviated secondary brain injury of SBI.
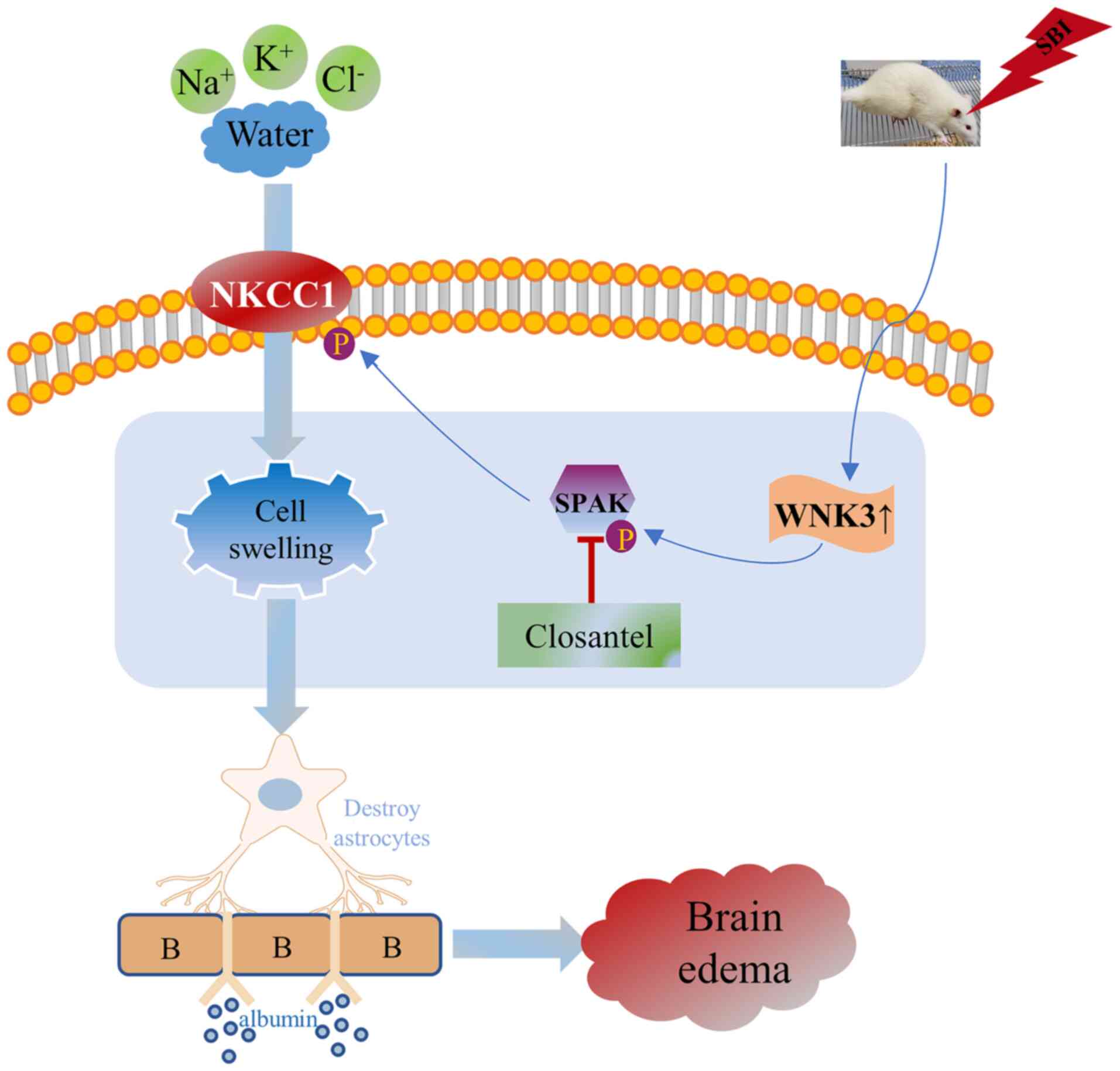 | Figure 5.Schematic diagram illustrating the
possible mechanisms underlying SPAK-mediated effects on SBI via
regulating NKCC1. The present study indicated that SBI induced the
rapid increase of p-SPAK expression, which lead to the increase of
p-NKCC1. Subsequently, inorganic substances, such as
Na+, K+, and Cl−, flow into the
astrocytes, causing the astrocytes to swell, damaging the stability
of the BBB and eventually resulting in the brain edema. SPAK,
STE20/SPS1-related proline/alanine-rich kinase; NKCC1,
Na+-K+-Cl− cotransporter 1; SBI,
surgical brain injury; p, phosphorylated; WNK, WNK lysine deficient
protein kinase 3; BBB, blood brain barrier. |
The current study has several limitations that
should be discussed. The sample size of the study was small and
only male rats were used. Therefore, it was not possible to
investigate sex-mediated differences in the expression levels of
p-SPAK and p-NKCC1 after SBI. Thus, this should be considered when
interpreting the present results. In addition, it was not examined
whether inhibition of p-NKCC1 would lead to changes in p-SPAK,
which would further corroborate the current results. Moreover, the
present study did not investigate whether p-SPAK agonists would
lead to the upregulation of p-NKCC1 and further aggravate brain
injury. Therefore, future studies are required to investigate
further.
In conclusion, the present study showed that after
SBI in rats, SPAK was activated, which could phosphorylate
downstream NKCC1, and promote ions and water to enter cells, thus
leading to cell apoptosis, which aggravated secondary brain injury.
Inhibiting the activity of SPAK induced protective effects on the
brain. These results indicated that SPAK may be potentially used as
a prevention and control target for SBI.
Acknowledgements
Not applicable.
Funding
This work was supported by the Zhangjiagang Science
and Technology Project (grant no. ZKS1914) and the Zhangjiagang
Health Youth Science and Technology Project (grant no.
ZJGQNKJ202030).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
GC and BQD conceived and designed this study. YG and
MW drafted and revised the manuscript. HG, MW and YG conducted the
experiments. RG was responsible for analyzing and interpreting the
data. FG and MS performed figure processing and data analysis. BQD
and RG agreed to be responsible for all aspects of the work. BQD
and RG confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
All experiments received approval from the Institute
of Animal Care Committee of Zhangjiagang Traditional Chinese
Medicine Hospital (approval no. 2020-10-1; Suzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zakhary G, Sherchan P, Li Q, Tang J and
Zhang JH: Modification of kynurenine pathway via inhibition of
kynurenine hydroxylase attenuates surgical brain injury
complications in a male rat model. J Neurosci Res. 98:155–167.
2020.PubMed/NCBI
|
|
2
|
Matchett G, Hahn J, Obenaus A and Zhang J:
Surgically induced brain injury in rats: The effect of
erythropoietin. J Neurosci Methods. 158:234–241. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Benggon M, Chen H, Applegate RL II and
Zhang J: Thrombin preconditioning in surgical brain injury in rats.
Acta Neurochir Suppl. 121:299–304. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Eser Ocak P, Ocak U, Sherchan P, Gamdzyk
M, Tang J and Zhang JH: Overexpression of Mfsd2a attenuates blood
brain barrier dysfunction via Cav-1/Keap-1/Nrf-2/HO-1 pathway in a
rat model of surgical brain injury. Exp Neurol. 326:1132032020.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen JH, Hsu WC, Huang KF and Hung CH:
Neuroprotective effects of collagen-glycosaminoglycan matrix
implantation following surgical brain injury. Mediators Inflamm.
2019:68489432019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim CH, McBride DW, Sherchan P, Person CE,
Gren ECK, Kelln W, Lekic T, Hayes WK, Tang J and Zhang JH: Crotalus
helleri venom preconditioning reduces postoperative cerebral edema
and improves neurological outcomes after surgical brain injury.
Neurobiol Dis. 107:66–72. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wu MY, Gao F, Yang XM, Qin X, Chen GZ, Li
D, Dang BQ and Chen G: Matrix metalloproteinase-9 regulates the
blood brain barrier via the hedgehog pathway in a rat model of
traumatic brain injury. Brain Res. 1727:1465532020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhao H, Nepomuceno R, Gao X, Foley LM,
Wang S, Begum G, Zhu W, Pigott VM, Falgoust LM, Kahle KT, et al:
Deletion of the WNK3-SPAK kinase complex in mice improves
radiographic and clinical outcomes in malignant cerebral edema
after ischemic stroke. J Cereb Blood Flow Metab. 37:550–563. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gong YT, Wu MY, Tang JF, Shen JC, Li J,
Gao R, Dang BQ and Chen G: Advances in the study of the role and
molecular mechanism of withnolysine kinase 3 in nervous system
diseases (Review). Mol Med Rep. 23:3932021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Klug NR, Chechneva OV, Hung BY and
O'Donnell ME: High glucose-induced effects on
Na+-K+−2Cl− cotransport and
Na+/H+ exchange of blood-brain barrier
endothelial cells: Involvement of SGK1, PKCβII, and SPAK/OSR1. Am J
Physiol Cell Physiol. 320:C619–C634. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wu D, Lai N, Deng R, Liang T, Pan P, Yuan
G, Li X, Li H, Shen H, Wang Z and Chen G: Activated WNK3 induced by
intracerebral hemorrhage deteriorates brain injury maybe via
WNK3/SPAK/NKCC1 pathway. Exp Neurol. 332:1133862020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Delpire E and Austin TM: Kinase regulation
of Na+-K+−2Cl− cotransport in
primary afferent neurons. J Physiol. 588:3365–3373. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang J, Gao G, Begum G, Wang J, Khanna
AR, Shmukler BE, Daubner GM, de Los Heros P, Davies P, Varghese J,
et al: Functional kinomics establishes a critical node of
volume-sensitive cation-Cl− cotransporter regulation in
the mammalian brain. Sci Rep. 6:359862016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lykke K, Assentoft M, Hørlyck S, Helms HC,
Stoica A, Toft-Bertelsen TL, Tritsaris K, Vilhardt F, Brodin B and
MacAulay N: Evaluating the involvement of cerebral microvascular
endothelial Na+/K+-ATPase and
Na+-K+−2Cl− co-transporter in
electrolyte fluxes in an in vitro blood-brain barrier model of
dehydration. J Cereb Blood Flow Metab. 39:497–512. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Watanabe M and Fukuda A: Development and
regulation of chloride homeostasis in the central nervous system.
Front Cell Neurosci. 9:3712015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Haas BR, Cuddapah VA, Watkins S, Rohn KJ,
Dy TE and Sontheimer H: With-No-Lysine Kinase 3 (WNK3) stimulates
glioma invasion by regulating cell volume. Am J Physiol Cell
Physiol. 301:C1150–C1160. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang J, Karimy JK, Delpire E and Kahle
KT: Pharmacological targeting of SPAK kinase in disorders of
impaired epithelial transport. Expert Opin Ther Targets.
21:795–804. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gagnon KB and Delpire E: Multiple pathways
for protein phosphatase 1 (PP1) regulation of Na-K-2Cl
cotransporter (NKCC1) function: The N-terminal tail of the Na-K-2Cl
cotransporter serves as a regulatory scaffold for Ste20-related
proline/alanine-rich kinase (SPAK) AND PP1. J Biol Chem.
285:14115–14121. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cao-Pham AH, Urano D, Ross-Elliott TJ and
Jones AM: Nudge-nudge, WNK-WNK (kinases), say no more? New Phytol.
220:35–48. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Alessi DR, Zhang J, Khanna A, Hochdörfer
T, Shang Y and Kahle KT: The WNK-SPAK/OSR1 pathway: Master
regulator of cation-chloride cotransporters. Sci Signal. 7:re32014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Begum G, Yuan H, Kahle KT, Li L, Wang S,
Shi Y, Shmukler BE, Yang SS, Lin SH, Alper SL and Sun D: Inhibition
of WNK3 kinase signaling reduces brain damage and accelerates
neurological recovery after stroke. Stroke. 46:1956–1965. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang M, Cui Z, Cui H, Cao Y, Zhong C and
Wang Y: Astaxanthin alleviates cerebral edema by modulating NKCC1
and AQP4 expression after traumatic brain injury in mice. BMC
Neurosci. 17:602016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tian Y, Guo SX, Li JR, Du HG, Wang CH,
Zhang JM and Wu Q: Topiramate attenuates early brain injury
following subarachnoid haemorrhage in rats via duplex protection
against inflammation and neuronal cell death. Brain Res.
1622:174–185. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
AlAmri MA, Kadri H, Alderwick LJ, Simpkins
NS and Mehellou Y: Rafoxanide and closantel inhibit SPAK and OSR1
kinases by binding to a highly conserved allosteric site on Their
C-terminal domains. ChemMedChem. 12:639–645. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lin GM, Liu PY, Wu CF, Wang WB and Han CL:
Perspective of future drugs targeting sterile 20/SPS1-related
proline/alanine-rich kinase for blood pressure control. World J
Cardiol. 7:306–310. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kikuchi E, Mori T, Zeniya M, Isobe K,
Ishigami-Yuasa M, Fujii S, Kagechika H, Ishihara T, Mizushima T,
Sasaki S, et al: Discovery of Novel SPAK inhibitors that block WNK
kinase signaling to cation chloride transporters. J Am Soc Nephrol.
26:1525–1536. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wu BJ QC, Kong Q, Tan DM and Tan Y:
Laboratory animal-Guideline for using animals in the education.
Chinese Association for Laboratory Animal Sciences. 2017.
|
|
28
|
Akyol O, Sherchan P, Yilmaz G, Reis C, Ho
WM, Wang Y, Huang L, Solaroglu I and Zhang JH: Neurotrophin-3
provides neuroprotection via TrkC receptor dependent pErk5
activation in a rat surgical brain injury model. Exp Neurol.
307:82–89. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Feng D, Wang B, Wang L, Abraham N, Tao K,
Huang L, Shi W, Dong Y and Qu Y: Pre-ischemia melatonin treatment
alleviated acute neuronal injury after ischemic stroke by
inhibiting endoplasmic reticulum stress-dependent autophagy via
PERK and IRE1 signalings. J Pineal Res. 622017.doi:
10.1111/jpi.12395.
|
|
30
|
Zhang J, Xu X, Zhou D, Li H, You W, Wang Z
and Chen G: Possible role of Raf-1 kinase in the development of
cerebral vasospasm and early brain injury after experimental
subarachnoid hemorrhage in rats. Mol Neurobiol. 52:1527–1539. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhao Y, Huang G, Chen S, Gou Y, Dong Z and
Zhang X: Homocysteine aggravates cortical neural cell injury
through neuronal autophagy overactivation following rat cerebral
ischemia-reperfusion. Int J Mol Sci. 17:11962016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang YC, Cui Y, Cui JZ, Sun LQ, Cui CM,
Zhang HA, Zhu HX, Li R, Tian YX and Gao JL: Neuroprotective effects
of brilliant blue G on the brain following traumatic brain injury
in rats. Mol Med Rep. 12:2149–2154. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Eroğlu H, Kaş HS, Oner L, Türkoğlu OF,
Akalan N, Sargon MF and Ozer N: The in-vitro and in-vivo
characterization of PLGA:L-PLA microspheres containing
dexamethasone sodium phosphate. J Microencapsul. 18:603–612. 2001.
View Article : Google Scholar
|
|
34
|
Ke K, Rui Y, Li L, Zheng H, Xu W, Tan X,
Cao J, Wu X, Cui G and Cao M: Upregulation of EHD2 after
intracerebral hemorrhage in adult rats. J Mol Neurosci. 54:171–180.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bankstahl M, Breuer H, Leiter I, Märkel M,
Bascuñana P, Michalski D, Bengel FM, Löscher W, Meier M, Bankstahl
JP and Härtig W: Blood-brain barrier leakage during early
epileptogenesis is associated with rapid remodeling of the
neurovascular unit. eNeuro. 5:ENEURO.0123–18.2018. 2018. View Article : Google Scholar
|
|
36
|
Yamazaki Y and Kanekiyo T: Blood-brain
barrier dysfunction and the pathogenesis of Alzheimer's disease.
Int J Mol Sci. 18:19652017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lu KT, Cheng NC, Wu CY and Yang YL:
NKCC1-mediated traumatic brain injury-induced brain edema and
neuron death via Raf/MEK/MAPK cascade. Crit Care Med. 36:917–922.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang J, Bhuiyan MIH, Zhang T, Karimy JK,
Wu Z, Fiesler VM, Zhang J, Huang H, Hasan MN, Skrzypiec AE, et al:
Modulation of brain cation-Cl− cotransport via the SPAK
kinase inhibitor ZT-1a. Nat Commun. 11:782020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lee HA, Jeong H, Kim EY, Nam MY, Yoo YJ,
Seo JT, Shin DM, Ohk SH and Lee SI: Bumetanide, the specific
inhibitor of Na+-K+−2Cl−
cotransport, inhibits 1alpha,25-dihydroxyvitamin D3-induced
osteoclastogenesis in a mouse co-culture system. Exp Physiol.
88:569–574. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Huang H, Song S, Banerjee S, Jiang T,
Zhang J, Kahle KT, Sun D and Zhang Z: The WNK-SPAK/OSR1 kinases and
the cation-chloride cotransporters as therapeutic targets for
neurological diseases. Aging Dis. 10:626–636. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Thastrup JO, Rafiqi FH, Vitari AC,
Pozo-Guisado E, Deak M, Mehellou Y and Alessi DR: SPAK/OSR1
regulate NKCC1 and WNK activity: Analysis of WNK isoform
interactions and activation by T-loop trans-autophosphorylation.
Biochem J. 441:325–337. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yang SS, Huang CL, Chen HE, Tung CS, Shih
HP and Liu YP: Effects of SPAK knockout on sensorimotor gating,
novelty exploration, and brain area-dependent expressions of NKCC1
and KCC2 in a mouse model of schizophrenia. Prog
Neuropsychopharmacol Biol Psychiatry. 61:30–36. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Luo WD, Min JW, Huang WX, Wang X, Peng YY,
Han S, Yin J, Liu WH, He XH and Peng BW: Vitexin reduces epilepsy
after hypoxic ischemia in the neonatal brain via inhibition of
NKCC1. J Neuroinflammation. 15:1862018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Simard JM, Kahle KT and Gerzanich V:
Molecular mechanisms of microvascular failure in central nervous
system injury-synergistic roles of NKCC1 and SUR1/TRPM4. J
Neurosurg. 113:622–629. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
LeVine SM: Albumin and multiple sclerosis.
BMC Neurol. 16:472016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Lee YC, Kao ST and Cheng CY: Acorus
tatarinowii Schott extract reduces cerebral edema caused by
ischemia-reperfusion injury in rats: Involvement in regulation of
astrocytic NKCC1/AQP4 and JNK/iNOS-mediated signaling. BMC
Complement Med Ther. 20:3742020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Taherian M, Norenberg MD, Panickar KS,
Shamaladevi N, Ahmad A, Rahman P and Jayakumar AR: Additive effect
of resveratrol on astrocyte swelling post-exposure to ammonia,
ischemia and trauma in vitro. Neurochem Res. 45:1156–1167. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zhang M, Cui Z, Cui H, Wang Y and Zhong C:
Astaxanthin protects astrocytes against trauma-induced apoptosis
through inhibition of NKCC1 expression via the NF-kappaB signaling
pathway. BMC Neurosci. 18:422017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Luo L, Guan X, Begum G, Ding D, Gayden J,
Hasan MN, Fiesler VM, Dodelson J, Kohanbash G, Hu B, et al:
Blockade of cell volume regulatory protein NKCC1 increases
TMZ-induced glioma apoptosis and reduces astrogliosis. Mol Cancer
Ther. 19:1550–1561. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Campisi M, Shin Y, Osaki T, Hajal C,
Chiono V and Kamm RD: 3D self-organized microvascular model of the
human blood-brain barrier with endothelial cells, pericytes and
astrocytes. Biomaterials. 180:117–129. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
McBride DW, Wang Y, Adam L, Oudin G, Louis
JS, Tang J and Zhang JH: Correlation between subacute sensorimotor
deficits and brain edema in rats after surgical brain injury. Acta
Neurochir Suppl. 121:317–321. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Gong Y, Wu M, Shen J, Tang J, Li J, Xu J,
Dang B and Chen G: Inhibition of the NKCC1/NF-κB signaling pathway
decreases inflammation and improves brain edema and nerve cell
apoptosis in an SBI Rat model. Front Mol Neurosci. 14:6419932021.
View Article : Google Scholar : PubMed/NCBI
|