Introduction
Sepsis is a type of disease derived from systemic
immune response to infection, which results in the death of 210,000
people annually in America as one of the main factors that
contribute to the death of critically sick patients (1). The mortality of sepsis is ~30–70%
(2). Currently, no effective
therapeutic method is available to decrease the mortality of sepsis
clinically. Therefore, it is of great significance to explore the
detailed pathological mechanism of sepsis to provide novel
therapeutic methods for the treatment of clinical sepsis.
A series of immune reactions can be induced by
sepsis, which change as the time goes on (3). It is recently reported that
pro-inflammatory reaction and anti-inflammatory reaction occur
simultaneously under sepsis pathological state, with overactive
immune response as the primary characteristic. According to the
clinical data, most patients survived at the stage of overactive
immune response, rather than at the stage of immunosuppression,
which is manifested as a tardy response to antigen and incapable of
eliminating secondary infection induced by primary infection
(4). It is widely reported that
apoptosis plays an important role in the immunosuppression
response. Severe apoptosis was found on lymphocytes, parenchymatous
cells and vascular endothelial cells isolated from sepsis animal
models (5), which induce
significant immunosuppression clinically and make patients more
susceptible to the infection by pathogenic bacteria such as
pseudomonas aeruginosa and candida. Apart from
apoptosis, the dysfunction of monocytes also plays a significant
role in immunosuppression. In the primary immune response post
bacterial infection, monocytes defense the body from infection by
phagocytizing microorganisms, detecting the metabolism of
microorganisms, response to stimulators from microorganisms and
producing numerous pro-inflammatory factors, such as interleukin
(IL)-6 and tumor necrosis factor (TNF)-α (6). Under normal physiological state,
circling monocytes are recruited back to the marrow to be
differentiated to peripheral tissues, which support the survival of
macrophages and dendritic cells (DC). The immunosuppression induced
by sepsis can be regulated by both the primary immune and secondary
immune system. In addition, the acquired immunity system can be
built by antigen presenting cells (APC) through stimulating the
response of specific T cells (7,8).
However, under the immunosuppression state induced by sepsis,
dysfunction of monocytes against bacterial infections was observed.
Furthermore the antigen-presenting ability of APC will be inhibited
by the downregulation of human leukocyte antigen DR (HLA-DR)
(9,10). Therefore, regulating the response of
monocytes to immunosuppression induced by sepsis is very important
to explore novel therapeutic routine for the treatment of
sepsis.
Programmed death 1 (PD-1) was firstly found in the
programmed dead cell lines using subtractive hybridization
technology by Ishida et al (11), which belongs to type I transmembrane
glycoproteins in immunoglobulin superfamily with a molecular weight
of 55 kDa. PD-1 is highly expressed on T cells, B cells, myeloid
cells and thymocytes, which regulates the immune system negatively
(12). It is widely reported that
PD-1/programmed cell death ligand 1 (PD-L1) signaling pathway is
involved in immunologic tolerance in peripheral tissues and plays
an important role in the development of chronic virus infection,
inflammation, tumor immunity and autoimmune disease by mediating
its downstream signal pathways, such as PI3K/AKT and SHP-2 signal
pathway (13,14). Some progress has already been made
on weakening, restricting or terminating the function of effective
T cells by interacting with the PD-1/PD-L1 signal pathway (15). Tong-fu-li-fei (TFL) decoction is a
traditional Chinese recipe that has already been proved to elicit
therapeutic effects on sepsis (16). However, the underlying mechanism
remains unknown, particularly its effects on immunosuppression
induced by sepsis. In the present study, the effects of
Tong-fu-li-fei decoction on PD-1/PD-L1 axis, as well as the
downstream signaling pathway, will be investigated to explore the
potential mechanism underlying the therapeutic property of
Tong-fu-li-fei decoction on sepsis. Supportive pre-clinical data
will be provided by the present study for the clinical treatment of
sepsis with Tong-fu-li-fei decoction.
Materials and methods
Preparation of TFL decoction
TFL prescription was obtained from a national
medical master Professor Liu Shangyi. The TFL prescription
including 8 herbs, as shown in Table
I, which were purchased from Sinopharm Group Tongjitang
Pharmaceutical Co. Ltd, provided by a pharmacy from the First
Affiliated Hospital of Guiyang College of Traditional Chinese
Medicine. The extraction process was as follows: Rheum
officinale, mirabilite, Magnolia officinalis, Forsythia,
Scutellaria, almond, Bletilla striata and notoginseng (quality
ratio of herb=2:1:1:1:1:1:1:1) were immersed in distilled water (1
g: 20 ml) and boiled gently for 1 h twice. The resulting extracts
were filtered, combined, condensed into 1 g/ml (the content of
crude drug in the decoction), and stored at 4°C before use.
 | Table I.Herbs included in the Tong-fu-li-fei
(TFL) prescription. |
Table I.
Herbs included in the Tong-fu-li-fei
(TFL) prescription.
| Latin name | Chinese name | Weight of crude
drug, g | Quality ratio |
|---|
| Rheum officinale
Baill | Dahuang | 20 | 2 |
|
Mirabilite | Mangxiao | 10 | 1 |
| Magnolia
officinalis Rehd. et Wils | Houpo | 10 | 1 |
| Forsythia
suspensa (Thunb.) Vahl | Lianqiao | 10 | 1 |
| Scutellaria
baicalensis Georgi | Huangqin | 10 | 1 |
| Prunus armeniaca
L. var. ansu Maxim | Kuxingren | 10 | 1 |
| Bletilla striata
(Thunb.) Reichb. F | Baiji | 10 | 1 |
| Panax
notoginseng (Burk.) F. H. Chen | Sanqi | 10 | 1 |
Animals
One-hundred male Sprague-Dawley rats weighing
200–220 g were procured from Vital River Laboratory Animal Co. Ltd.
and group housed in polypropylene cages. The animal room
environment was controlled (target conditions: Temperature, 20 to
26°C; relative humidity, 30 to 70%′ 12-h artificial light and 12-h
dark). Temperature and relative humidity was monitored daily. The
light cycle may have been interrupted for facility or study-related
procedures. The rats were fed by standard dry rat diet and pure
water. To minimize the animal suffering, we replaced the wood
filings in the cage every day during the experimental
procedures.
Establishment of the sepsis model by
cecal ligation and puncture (CLP) in rats and treatment
The rats were anesthetized by an abdominal injection
of 3% pentobarbital sodium (30 mg/kg). Surgery was performed under
sterile conditions. A midline abdominal incision was performed and
the cecum was exposed and the distal 1/3 was ligated with a 3-0
silk. At the end of the appendix, two holes were punctured with an
18-gauge needle. The cecum was then replaced in its original
position within the abdomen, which was closed in two layers (in
sham group, cecum was replaced). Surgery lasted for 20 min. Warm
Ringer's lactate (50 ml/kg) with buprenorphine (0.01 mg/kg) was
administered subcutaneously via abdominal wall. Immediately after
the surgical operation, animals received an oral gavage of normal
saline in the sham group (n=6) and model group (n=6), 750 mg/kg
glutamine (n=6), 7 ml/kg (n=6), 14 ml/kg (n=6), and 28 ml/kg
Tong-fu-li-fei decoction (n=6), respectively.
Symptoms and signs of rats
The symptoms of the rats were observed
postoperatively. To determine the effect of TFL on mortality from
CLP-induced sepsis, survival after CLP was assessed four times a
day for at least 7 days and the cumulative survival curve was
plotted using the Kaplan-Meier method (17).
FD4 detection
A 4-kD fluorescein isothiocyanate (FITC)-dextran
(FD4; Sigma-Aldrich; Merck KGaA) solution was used for intestinal
permeability measurement (18).
Briefly, 20.8 mM solution of FD4 was prepared with sterile
distilled water using colored Eppendorf tubes to keep away from
light. The FD4 solution was gently administered as oral gavage to
rats in each group at 10 ml/g bodyweight. Rats were held in an
upright position for about 10 sec to avoid regurgitation. Blood
(0.5–1 ml) was collected into an EDTA-loaded vacuum tube, and
plasma samples were separated immediately by spinning down in a
refrigerated centrifuge (1,000 × g for 30 min at 48°C). Then,
plasma samples were added to a black 96-well microplate. The FITC
concentration was measured using a Microplate spectrophotometer
with an excitation wavelength of 485 nm and an emission wavelength
of 535 nm. In addition, a serial dilution of FITC was prepared and
used for a standard curve. The plasma obtained from control group
was used as a negative control.
Hematoxylin and eosin (H&E)
staining
After FD4 detection, 200 mg/kg phenobarbital sodium
was administered intraperitoneally for euthanasia in rats. The
intestinal mucosa tissue of each animal was collected and washed
over by sterile water for a couple of hours. The tissue was
dehydrated by 70, 80 and 90% ethanol solution successively and
mixed with equal quantity of ethanol and xylene. After 15-min
incubation at room temperature, the tissue was mixed with equal
quantity of xylene for 15 min at room temperature. The step was
repeated until the tissue looked transparent. Subsequently, the
tissue was embedded in paraffin, sectioned (4 µm), and stained with
H&E at room temperature for 30 min. Images were captured using
an inverted light microscope (magnification, ×50 and ×100; Olympus
Corporation). The H&E staining score was evaluated according to
the colon mucosa damage index system described previously (19). Briefly, 0 point represented no
injury to the intestinal mucosa; 1 point indicated that the surface
of the intestinal mucosa was smooth, with no erosion or ulceration,
but with mild hyperemia and edema; 2 points indicated that the
intestinal mucosa had congestion and edema, and the mucosa was
coarse and granular, with erosion or intestinal adhesion; 3 points
indicated necrosis and ulcers appeared on the surface of intestinal
mucosa, which also exhibited high congestion and edema (maximum
longitudinal diameter of the ulcer, <1.0 cm), the intestinal
wall surface also had necrosis and inflammation or hyperplasia of
the intestinal wall was detected; 4 points indicated that the
maximum longitudinal diameter of the ulcer was >1.0 cm with
total intestinal wall necrosis more severe than that observed at 3
points.
Transmission electron microscope
(TEM)
Intestinal mucosa tissue was collected after the
animals were euthanized and was fixed with 2.5% glutaraldehyde for
>2 h and washed over by 0.1 M phosphoric acid solution for 3
times. Then, the tissues were fixed with 1% osmic acid for 2–3 h
and washed over by 0.1 M phosphoric acid solution for 3 times.
Ethyl alcohol (50, 70, 90 and 90%) and 90% acetone (v:v=1:1), 90%
acetone were used to wash out the tissues for 15–20 min,
successively. Subsequently, the tissues were incubated thrice with
100% acetone at room temperature for 15–20 min every time. Acetone
(100%) and the embedding solution were used to incubate with the
tissues for 3–4 h. Finally, the tissues were incubated with
embedding solution in 37°C oven overnight, 45°C oven for 12 h and
60°C oven for 48 h, successively. The solid tissues were cut into
coronal sections (30 µm) and the slides were stained with 3% uranyl
acetate and 3% lead citrate for 15 min at room temperature.
Ultrastructural changes of endometrium were observed and
photographed under transmission electron microscopy (JEM-1230;
magnification, ×20,000).
Transfection
The pc-DNA3.1-PD-1 and pc-DNA3.1-PD-L1 plasmids were
designed and constructed by Genscript Technology. DC and
CD8+ T cells were transfected with pc-DNA3.1-PD-L1 (1.5
µg/well) combined with Lipofectamine® 3000 (Thermo
Fisher Scientific, Inc.) at 37° for 48 h to establish the PD-L1
overexpressed DC and CD8+ T cells. These transfected
cells were utilized in the subsequent experiments 2 days later. The
pc-DNA3.1-NC was used as a negative control.
Enzyme-linked immunosorbent assay
(ELISA)
According to the instruction of the manufacturer,
the concentration of IL-6 (cat. no. RK00008; ABclonal Biotech Co.,
Ltd.) and TNF-α (cat. nos. RK00027; ABclonal Biotech Co., Ltd.) in
the intestine mucosa tissue of each rat or the supernatant of cells
was determined by ELISA. Basically, the operation includes: Sample
adding, enzyme adding, incubation, working solution preparing,
washing, dyeing, terminating, and detecting. Linear regression
equation was described based on the concentration of standards and
optical density (OD) value. The concentrations of the samples were
calculated according to the equation, detected OD value and
dilution factor.
Flow cytometry
After cells were counted, 2×106 cells per
sample were stained with Aqua Live/Dead viability dye (Thermo
Fisher Scientific, Inc.), according to the manufacturer's
instructions. Cells were then incubated in blocking solution
containing 5% normal mouse serum, 5% normal rat serum, and 1%
FcBlock (eBioscience; Thermo Fisher Scientific, Inc.) in PBS at 4°C
for 1 h, and then stained with a standard panel of
immunophenotyping antibodies (CD11c, 1:800, cat. no. ab254183;
CD80, 1:800, cat. no. ab134120; CD3, 1:1,000, cat. no. ab16669;
CD8, 1:500, cat. no. ab217344 and DX5, 1:200, cat. no. ab181548;
Abcam) for 30 min at room temperature. After staining, cells were
washed and fixed with 0.4% paraformaldehyde in PBS at 4°C for 24 h.
Data was acquired with a BD LSRII flow cytometer using BD FACSDiva
software (v 8.0.3; BD Biosciences). Compensation was performed on
the BD LSRII flow cytometer at the beginning of each experiment.
Data were analyzed using Flowjo v10 (FlowJo, LLC). Cell sorting for
cytospins was performed on a BD Aria II. The collected cells were
stained with a Jenner-Giemsa Stain kit (ENG Scientific Inc.) at
room temperature for 15–30 min and examined by light microscopy
(magnification, ×40).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was collected from the intestine mucosa
tissue from each rat or cells using an RNA Extraction kit (Takara
Bio, Inc.) according to the manufacturer's instructions. The
extracted RNA was quantified with a NanoDrop spectrophotometer
(NanoDrop Technologies; Thermo Fisher Scientific, Inc.). A specific
RT primer was used to reverse transcribe the complementary DNA at
50°C for 45 min. SYBR Premix Ex Taq™ (Takara Bio, Inc.) with an
Applied Bio-Rad CFX96 Sequence Detection system (Applied
Biosystems; Thermo Fisher Scientific, Inc.) was used for the PCR
procedure. PCR was conducted according to the following conditions:
95°C for 5 min, followed by 40 cycles at 95°C for 10 sec, 60°C for
15 sec and 72°C for 30 sec, and a final extension step at 72°C for
5 min. The expression levels of SHP-2, PD-1, PD-L1 and PI3K was
determined by the threshold cycle (Ct), and relative expression
levels were calculated by the 2−ΔΔCq method (20). The expression level of GAPDH in the
tissue was considered for the normalization of mRNA expression.
Three independent assays were performed. The information of the
primers is shown in Table II.
 | Table II.Sequences of primers for SHP-2, PI3K
and GAPDH. |
Table II.
Sequences of primers for SHP-2, PI3K
and GAPDH.
| Primer ID | Sequences
(5′-3′) |
|---|
| SHP-2 F |
AGACCACCAGCCAAGACAAG |
| SHP-2 R |
CTCCAGGTCCATCACCAAGT |
| PI3K F |
ACACCACGGTTTGGACTATGG |
| PI3K R |
GGCTACAGTAGTGGGCTTGG |
| GAPDH F |
CAATGACCCCTTCATTGACC |
| GAPDH R |
GAGAAGCTTCCCGTTCTCAG |
Western blotting
Total proteins were isolated from tissues or cells
using the Nuclear and Cytoplasmic Protein Extraction kit (Beyotime
Institute of Biotechnology), and were quantified using the BCA kit
(Beyotime Institute of Biotechnology). Approximately 40 µg of
protein was separated on 12% SDS-polyacrylamide gel (SDS-PAGE) and
the gel was transferred to polyvinylidene difluoride (PVDF)
membrane (EMD Millipore). The membrane was blocked with 5% nonfat
dry milk in TBST (Tris-buffered saline/0.1% Tween-20; pH 7.4) for 1
h at room temperature. The membrane was then incubated at 4°C
overnight with primary rabbit anti-human antibodies to PD-1
(1:1,000; cat. no. ab243644), PD-L1 (1:1,000; cat. no. ab205921),
SHP-2 (1:1,000; cat. no. ab32083), PI3K (1:1,000; cat. no.
ab32089), phosphorylated (p)-AKT (1:1,000; cat. no. ab8805), AKT
(1:1,000; cat. no. ab38449) and GAPDH (1:1,000; cat. no. ab9485)
(all from Abcam). A horseradish peroxidase-conjugated antibody
against rabbit IgG (1:5,000; cat. no. ab7090; Abcam) was used as a
secondary antibody. Blots were incubated with the ECL reagents
(Beyotime Institute of Biotechnology) and exposed to Tanon
5200-multi to detect protein expression. ImageJ software 1.8.0.
(National Institutes of Health) was used to semi-quantify the
bands. Three independent assays were performed.
Statistical analysis
Data are expressed as mean ± SD, with the exception
of HE score data, which are shown as median and range.
Statistically significant differences for continuous variables were
determined using a one-way analysis of variance (ANOVA) with
Tukey's test for the normally distribution data. All analyses were
performed using GraphPad Prism 5 software (GraphPad Software,
Inc.). P<0.05 was considered to indicate a statistically
significant difference.
Results
General symptom and survival rate of
CLP-induced rats are alleviated by TFL
No abnormal symptom was observed in animals in the
sham group. Apparent pathological symptoms described by Wichterman
et al (21) were found in
CLP-induced rats. Such clinical observations include listless
spirit, rolled up body, decreased movements and decreased water
intake, which were recorded for animals in the sepsis group at the
early stage post-surgery. As the time went on, the CLP rats showed
low skin temperature, weakened muscle strength, excreted watery
stool, yellow color, fishy smell, and developed shortness of
breath, no resistance to passive supine and the feces were slimy
and abundant, which were reversed significantly by the treatment of
glutamine (Gln) and TFL. The data on the survival rate for each
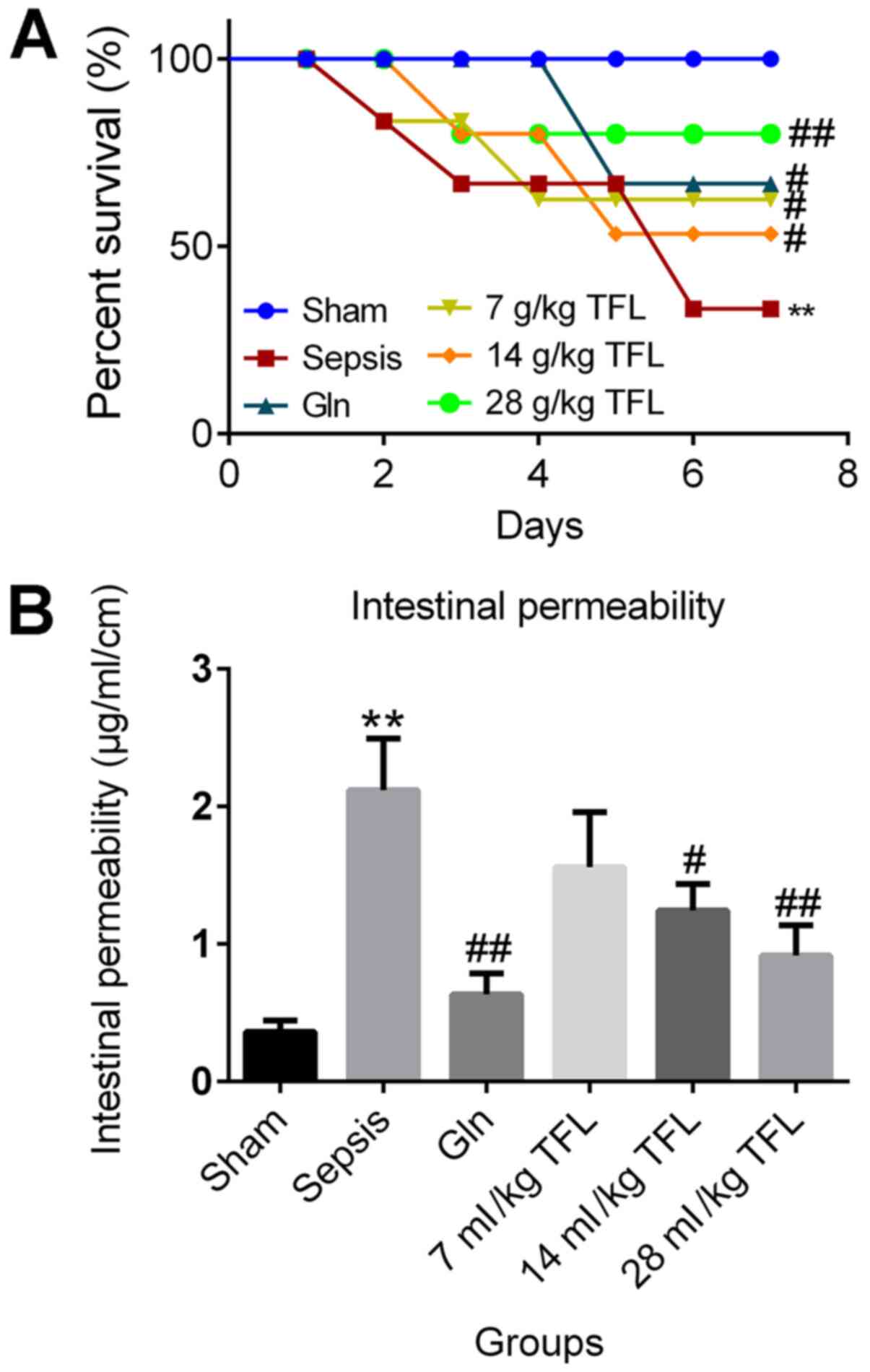
group was illustrated in Fig. 1A.
The overall survival rate of animals in the sham group was
maintained as 100% in the 7 days post-surgery. However, significant
decrease in the survival rate was observed in CLP-induced rats
(**P<0.01 vs. sham), which was greatly ameliorated by the
introduction of Gln and TFL, especially for high and moderate
dosages of TFL (#P<0.05 vs. sepsis;
##P<0.01 vs. sepsis). The intestinal permeability of
each rat was further investigated to demonstration the potential
toxicity. As shown in Fig. 1B, the
permeability of intestine was significantly promoted in CLP-induced
rats compared with the sham group (**P<0.01 vs. sham), which was
greatly suppressed by the introduction of Gln and TFL (14 and 28
ml/kg) (#P<0.05 vs. sepsis; ##P<0.01
vs. sepsis), respectively.
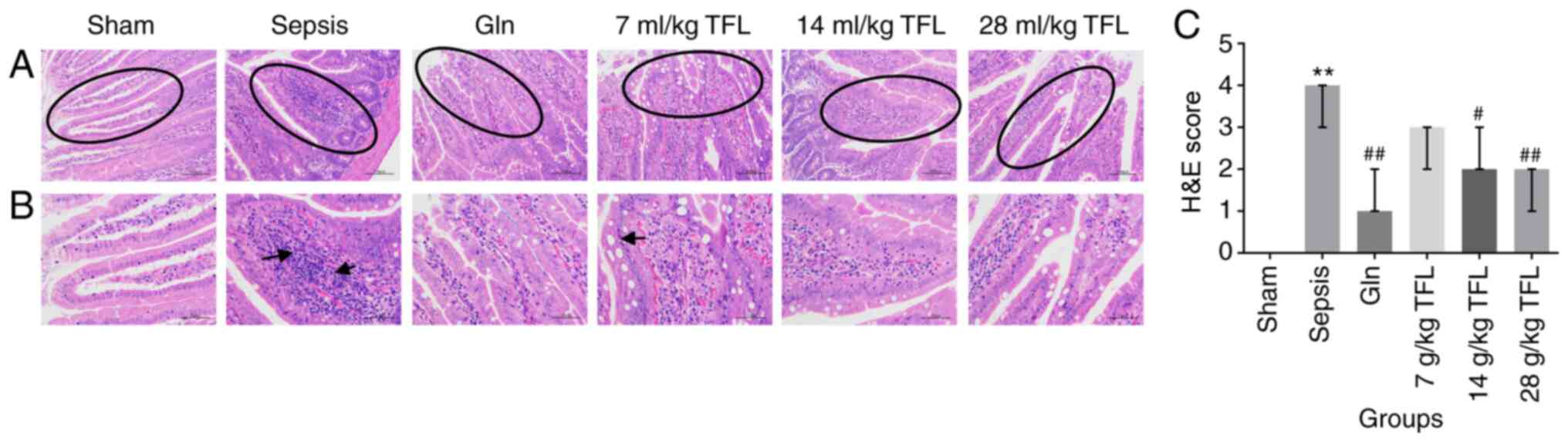
Effect of TFL on histopathology of
intestinal tissues in CLP-induced rats
H&E staining was used to evaluate the
pathological state of intestine tissue of each rat (Fig. 2). The rats in the sham group
exhibited normally structured intestines under light microscope
observation, without tissue edema, and presented with normal villus
structure and clear edge of microvilli. Regarding the animals from
sepsis group, the intestinal wall was noted to be thinner, the
mucosa was atrophied, and local shedding and villous rupture (black
arrows) were observed. The rats in the Gln, 14 and 21 ml/kg TFL
group exhibited partial intestinal mucosal necrosis and shedding,
but the intestinal mucosal lesions were relatively mild. These data
claimed potential therapeutic effects of TFL on the injured
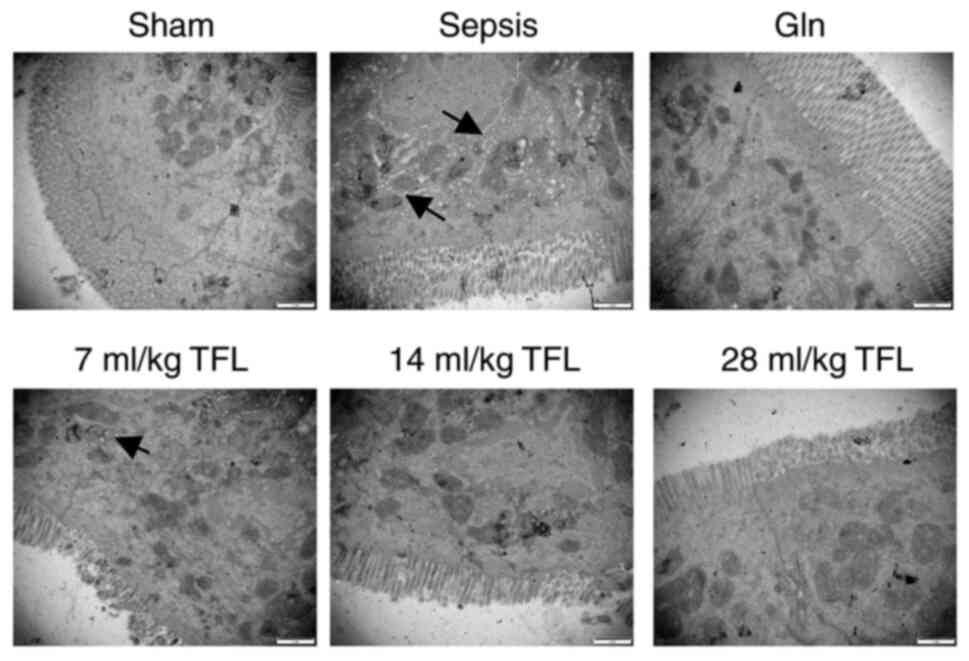
intestinal tissues induced by CLP. In order to check the
ultrastructural change in intestinal mucosa, TEM was performed on
the intestinal mucosa tissues. In the sepsis group, the
ultrastructure of intestinal epithelial cells showed decreased and
deformed microvilli, and incomplete desmosomes (Fig. 3). The edema of the villi cells was
more pronounced with the mitochondrial dropsy (black arrows) and
vacuolar change, gaps of enterocytes were sharply widened,
junctional complex among enterocytes were shortened and widened. In
contrast, the ultrastructure from Gln, 14 and 21 ml/kg TFL-treated
rats showed that the microvilli were dense and regular with a
jagged and interlocking pattern among enterocytes and the
mitochondria were clear.
TFL suppresses the inflammation and
PD-1/PD-L1 signal pathway induced by CLP
To investigate the potential mechanism underlying
the effects of TFL on the injured intestine induced by CLP, the
concentration of IL-6 and TNF-α, as well as the expression level of
key proteins involved in PD-1/PD-L1 signal pathway was determined
in the intestine tissues. As shown in Fig. 4A, compared with the sham group, the
concentration of IL-6 and TNF-α was elevated significantly in
CLP-treated rats (**P<0.01 vs. sham), which was greatly
suppressed by the treatment of Gln, 14 and 28 ml/kg TFL
(#P<0.05 vs. sepsis; ##P<0.01 vs.
sepsis), respectively. SHP-2, PI3K and p-AKT were found to be
significantly upregulated in CLP-induced rats (**P<0.01 vs.
sham; Fig. 4B-C), which was
downregulated by the introduction of Gln, 14 and 28 ml/kg TFL
(#P<0.05 vs. sepsis, ##P<0.01 vs.
sepsis), respectively. In addition, it was found that the elevated
expression level of PD-1 and PD-L1 in the intestine induced by CLP
was significantly suppressed by the treatment of TFL in a
dose-dependent manner (#P<0.05 vs. sepsis;
##P<0.01 vs. sepsis; Fig.
4B-C).
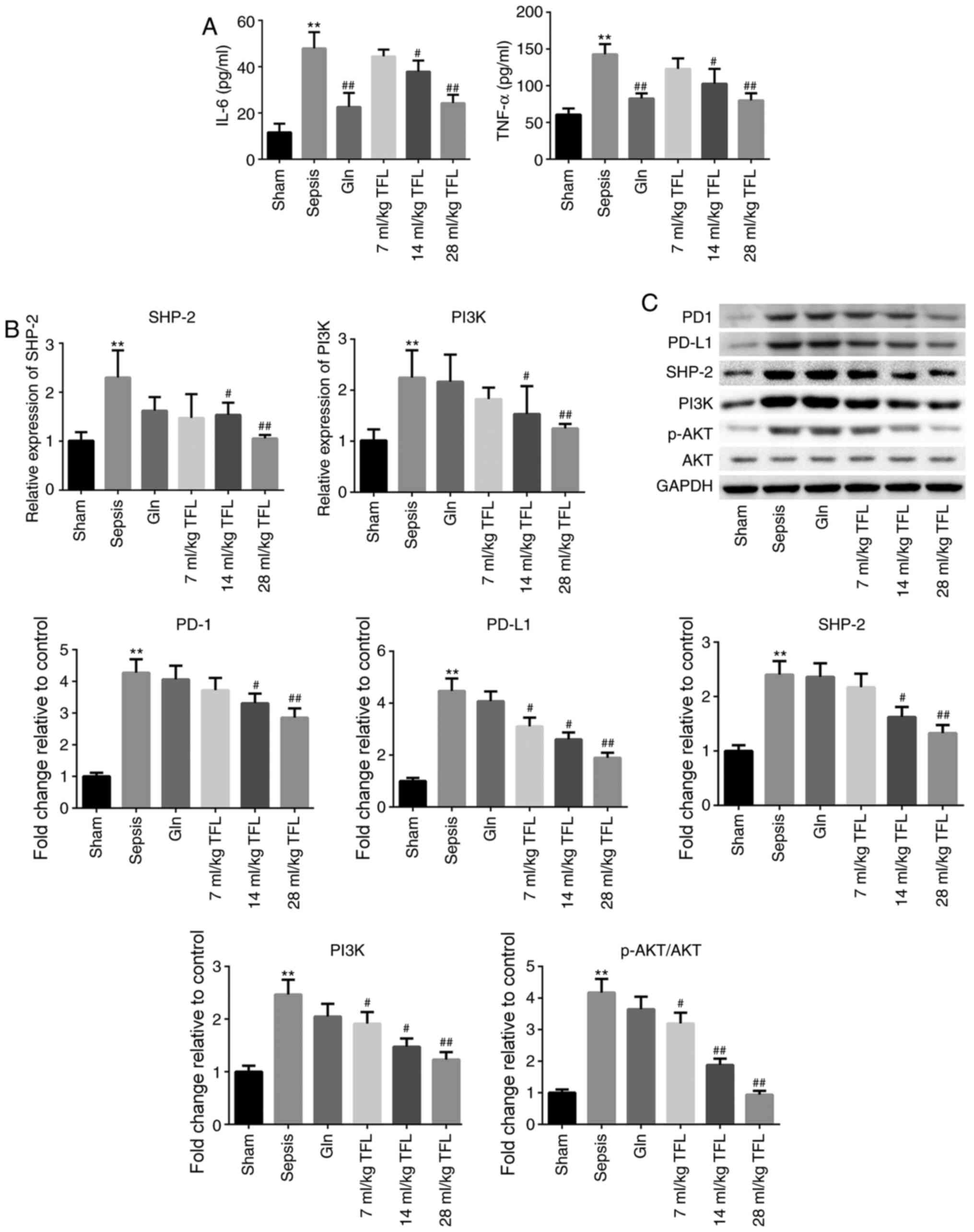 | Figure 4.TFL suppresses the inflammation and
PD-1/PD-L1 signal pathway induced by cecal ligation and puncture.
(A) The concentration of IL-6 and TNF-α was determined by
enzyme-linked immunosorbent assay. (B) The expression of SHP-2 and
PI3K was evaluated by reverse transcription-quantitative PCR. (C)
The expression level of PD-1, PD-L1, SHP-2, PI3K, p-AKT and AKT in
the intestine was detected by western blotting. **P<0.01 vs.
sham; #P<0.05 vs. sepsis; ##P<0.01 vs.
sepsis. IL-6, interleukin-6; TNF-α, tumor necrosis factor-α; PD-1,
programmed death 1; PD-L1, programmed cell death ligand 1; p-,
phosphorylated-; TFL, Tong-fu-li-fei; Gln, glutamine. |
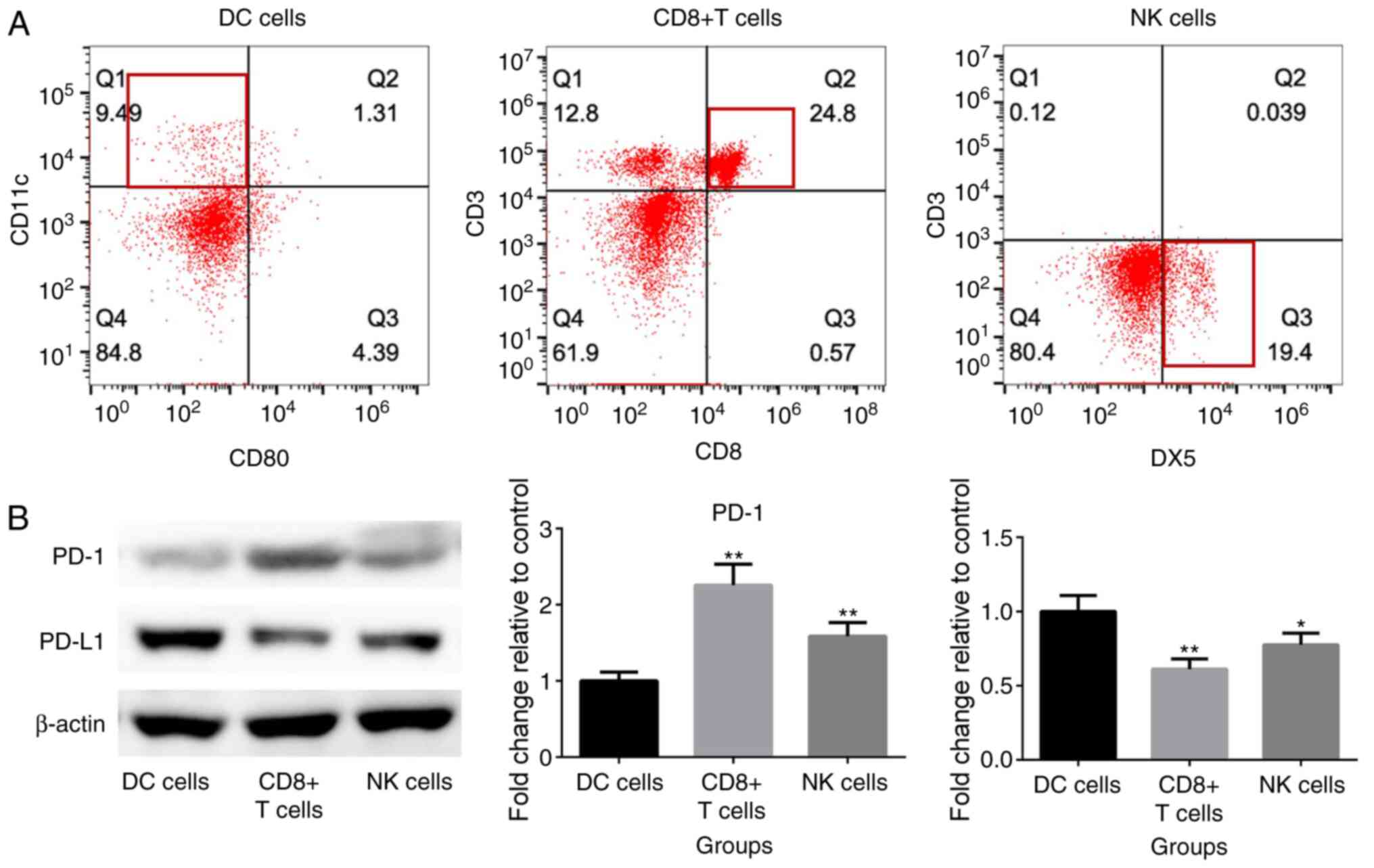
TFL inhibits the release of
inflammatory factors by blocking the PD-1/PD-L1 signal pathway
To further verify the inhibitory effects of TFL on
inflammation through regulating the PD-1/PD-L1 signal pathway, DC
cells, CD8+ T cells and NK cells were isolated from the
peripheral blood of rats, the phenotypes of which are shown in
Fig. 5A.
CD11c+CD80− cells were isolated as DC cells,
CD3+ CD8+ as CD8+ T cells and
CD3−DX5+ cells as NK cells by sorting flow
cytometry. As shown in Fig. 5B,
compared with DC cells, higher PD-1 expression level and lower
PD-L1 expression level were observed both on CD8+ T
cells and NK cells, respectively. 0.01, 0.02, and 0.04 ml/ml TFL
were incubated with the co-cultural system containing both DC cells
and CD8+ T cells. As shown in Fig. 6A, the production of IL-6 and TNF-α
was significantly elevated by the introduction of 0.02 and 0.04
ml/ml TFL in a dose-dependent manner (*P<0.05 vs. control;
**P<0.01 vs. control). Fig. 6B
showed the expression level of key proteins in the downstream of
PD-1/PD-L1 signal pathway. It was found that SHP-2, PI3K and p-AKT
were significantly downregulated by the treatment of 0.02 and 0.04
ml/ml TFL in a dose-dependent manner (*P<0.05 vs. control;
**P<0.01 vs. control).
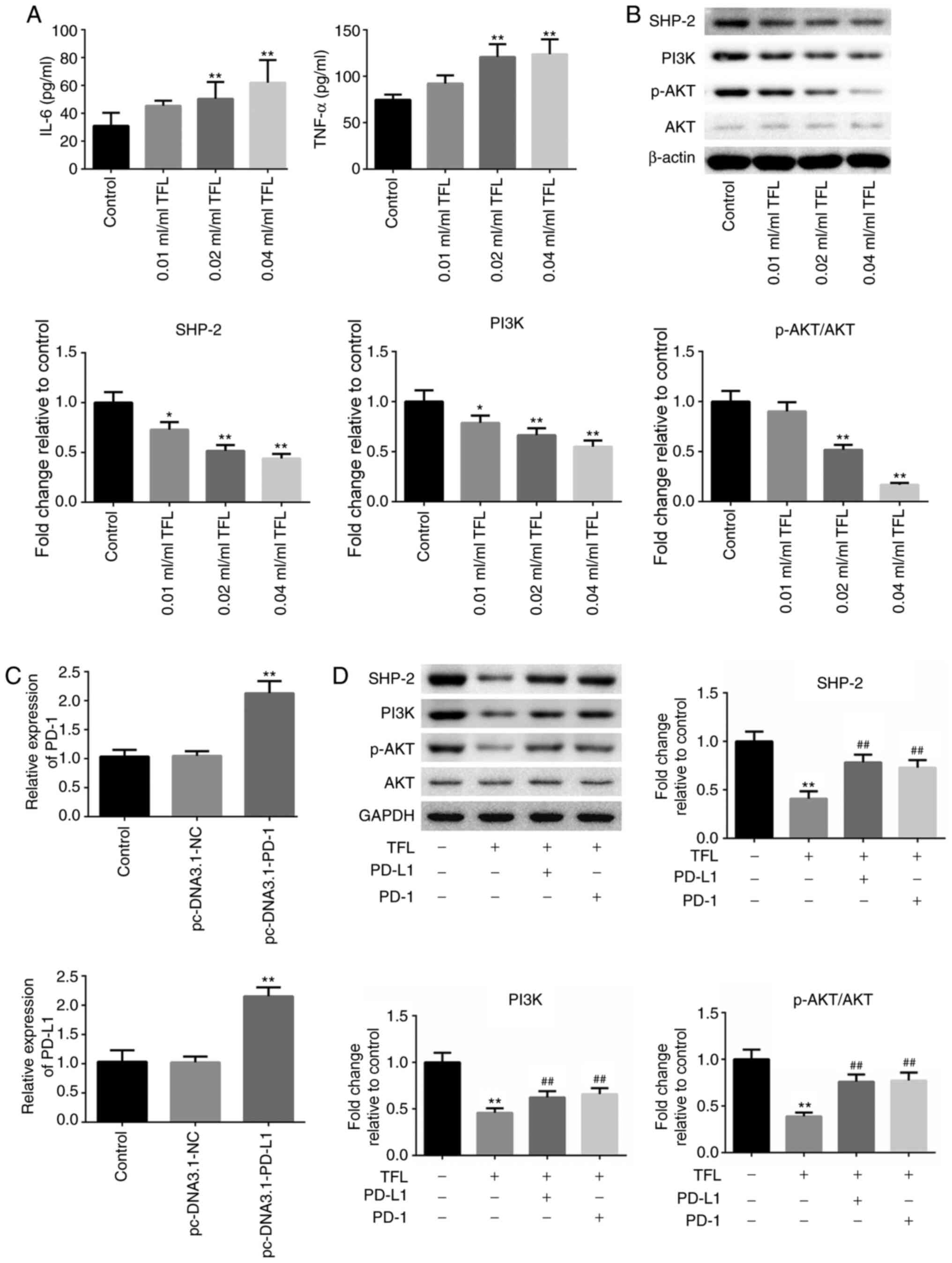 | Figure 6.TFL inhibits the release of
inflammatory factors by blocking the PD-1/PD-L1 signal pathway. (A)
The concentration of IL-6 and TNF-α was determined by enzyme-linked
immunosorbent assay. (B) The expression level of SHP-2, PI3K, p-AKT
and AKT was detected by western blotting. *P<0.05 vs. control;
**P<0.01 vs. control. (C) The expression of PD-1 in
CD8+ T cells and the expression of PD-L1 in DC cells
were determined by reverse transcription-quantitative PCR assay.
**P<0.01 vs. pc-DNA3.1-NC. (D) The expression levels of SHP-2,
PI3K, p-AKT and AKT in the co-cultural system of DC cells (wild
type or PD-L1 overexpressed) and CD8+ T cells (wild type
or PD-1 overexpressed) in the presence of TFL was determined by
western blotting. **P<0.01 vs. control; ##P<0.01
vs. TFL. IL-6, interleukin-6; TNF-α, tumor necrosis factor-α; p-,
phosphorylated-; DC, dendritic cells; NK cells; natural killer
cells; NC, negative control; PD-1, programmed death 1; PD-L1,
programmed cell death ligand 1; TFL, Tong-fu-li-fei. |
To further verify the mechanism, the PD-L1
overexpressed DC cells and the PD-1 overexpressed CD8+ T
cells were established, with DC cells transfected with pc-DNA3.1-NC
and CD8+ T cells transfected with pc-DNA3.1-NC as
negative control, respectively. As shown in Fig. 6C, compared with pc-DNA3.1-NC
transfected DC cells, the expression level of PD-L1 was
significantly elevated in pc-DNA3.1-PD-L1 transfected DC cells
(**P<0.01 vs. pc-DNA3.1-NC). In addition, compared with
pc-DNA3.1-NC transfected CD8+ T cells, the expression
level of PD-1 was notably increased in pc-DNA3.1-PD-1 transfected
CD8+ T cells (**P<0.01 vs. pc-DNA3.1-NC).
Subsequently, four groups were formed: DC cells incubated with
CD8+ T cells in the absence of 0.02 ml/ml TFL; DC cells
incubated with CD8+ T cells in the presence of 0.02
ml/ml TFL; PD-L1-overexpressed DC cells incubated with
CD8+ T cells in the presence of 0.02 ml/ml TFL; and DC
cells incubated with PD-1-overexpressed CD8+ T cells. As
shown in Fig. 6D, it was found that
the suppressed expression of SHP-2, PI3K and p-AKT (induced by the
introduction of TFL) was dramatically elevated by upregulating
PD-L1 in the DC cells or upregulating PD-1 in the CD8+ T
cells (**P<0.01 vs. control; ##P<0.01 vs. TFL).
These data indicate that the regulatory effect of TFL on SHP-2 and
PI3K/AKT signal pathway was associated with the inactivation of the
PD-1/PD-L1 signal pathway.
Discussion
Sepsis is manifested as life-threating organic
dysfunction induced by the imbalance of immune reaction following
infection (22). It is currently
suggested that inflammatory reaction and immunosuppression are
involved in the whole pathological process of sepsis (23,24).
Immunosuppression is the main factor that contributes to the
secondary infection and multiple organ dysfunction syndrome
diagnosed in patients with sepsis (25). Thus, ameliorating the
immunosuppression state may be beneficial to the treatment of
clinical sepsis. Intestinal injury is one of the main pathological
characteristics of sepsis, which significantly influences the
health of patients with sepsis (26). In the present study, a sepsis model
was established by CLP in rats to simulate the pathological state
of sepsis-induced intestinal barrier injury. The CLP-induced rat
model was confirmed by significantly low survival rate, elevated
intestinal permeability, histopathological changes in intestinal
tissues, such as thinner barrier, atrophied mucosa, local shedding
and villous rupture, and ultrastructure changes, such as sharply
decreased microvilli, mitochondrial dropsy, vacuolar changes,
widened gaps of enterocytes, and shortened junctional complex among
enterocytes. In addition, severe inflammation was observed in the
intestinal tissues of CLP-induced rats. Gln, which showed
significant effects in preventing intestinal injury caused by
external stimulations (27,28), was used as a positive control in the
present study. A high, moderate, and low dosage of TFL was
administered to the CLP-induced rats orally. It was found that the
total survival rate was promoted by both Gln and TFL, especially at
the high dosage. The enlarged permeability induced by CLP was
suppressed by both Gln and TFL, indicating a potential repair
function of TFL on injured intestine tissues caused by CLP. The
alleviating effects were verified by ameliorated pathological state
and ultrastructure changes in endothelial cells, as well as the
assuasive inflammation caused by CLP. These in-vivo data
claimed a potential therapeutic effect of TFL on intestinal injury
induced by sepsis. However, more detailed investigation will be
explored in future work to confirm the pharmacological function,
including the evaluation of immunosuppression-associated cells,
clinical chemistry of peripheral blood and the determination of an
optimized dosage.
Pro-inflammatory reaction and anti-inflammatory
reaction are reported to be simultaneously involved in the
pathological procedure of sepsis (29). Currently, systemic inflammation
caused by sepsis can be treated as the development of medicine.
However, in the advanced stage of sepsis, patients will be
experienced with the immunosuppression state, which is
characterized by low immune reaction or no response. The secondary
infection and associated complications will be induced as the
sensitivity and clearance of immune system to the pathogenic
bacteria decreases (30,31). Roquilly et al (32) reported that the antigen-presentation
ability of both macrophages and DCs was suppressed in rats with
sepsis, accompanied with increased production of Treg cells and
transforming factor-β, indicating a significant immunosuppression
state induced by sepsis. PD-1/PD-L1 is a classic signaling pathway
that mediates the immunosuppression system. The activation of
effective T cells can be suppressed by binding with PD-L1 using
PD-1 expression on T cells to maintain the balance of immune
system. It is reported that the survival rate of rats with sepsis
could be elevated as the release of interferon (IFN)-γ and the
apoptotic level are inhibited, which were achieved by blocking the
PD-1/PD-L1 signaling pathway (33).
The effects of blocking the PD-1/PD-L1 signaling pathway have also
been verified by clinical administrating anti-PD-1 or anti-PD-L1
antibodies to the patients with sepsis, with decreased apoptosis
and increased production of IFN-γ and IL-2 (34). SHP-2 and PI3K/AKT are main
downstream signal pathways of PD-1/PD-L1 that exert the
immunosuppression effects (35,36).
In the present study, accompanied with inflammation detection, it
was found that the SHP-2 and PI3K/AKT signal pathway were
significantly activated by CLP, indicating a significant
immunosuppression induced by activated PD-1/PD-L1 signal pathway.
By the introduction of TFL, especially at high and moderate dosage,
the activated SHP-2 and PI3K/AKT signaling pathway were
significantly suppressed, indicating an inhibitory effect on the
PD-1/PD-L1 signaling pathway and an effect on the activation of
immune system. Further mechanistic study showed that SHP-2 and
PI3K/AKT signal pathway in the co-cultural system of DC cells and
CD8+ T cells was significantly inhibited by the
introduction of TFL. Interestingly, the production of IL-6 and
TNF-α in the co-cultural system was found to be elevated by the
treatment of TFL, which was not consistent with the in-vivo
data. It was suspected that the inflammatory situation
in-vivo is more complicated compared with that
in-vitro and the balance of the immunity in the CLP-induced
rats might be maintained by TFL, so that the decreased production
of inflammatory factors was accompanied by improved sepsis
symptoms. In the in-vitro study, the release of the
inflammatory factors might only be regulated by the PD-1/PD-L1
signaling pathway, which was inhibited by TFL and resulting in an
immunopotentiation in CD 8+ T cells. The hypothesis was
further verified by activating the PD-1/PD-L1 signal pathway in the
presence of TFL. However, further investigations will be performed
on the direct interaction between TFL and the PD-1/PD-L1 signaling
pathway to further verify our hypothesis. In addition, these data
indicated that the PD-1/PD-L1 interaction between DC and
CD8+ T cells was blocked by TFL. However, the evidence
with these preliminary data from the present study is not
sufficient to directly claim that the anti-sepsis effect of TFL
resulted from blocking the PD-1/PD-L1 signaling pathway. Further
investigations at the molecular level will be explored to better
understand the association between the therapeutic effects of TFL
against sepsis and the immunosuppression induced by blocking the
PD-1/PD-L1 signaling pathway.
Taken together, the data from the present study
indicate that Tong-fu-li-fei decoction may attenuate
immunosuppression to protect the intestinal mucosal barrier in
sepsis via inhibiting the PD-1/PD-L1 signaling pathway.
Acknowledgements
Not applicable.
Funding
This work was supported by grants from the National
Natural Science Foundation of China (grant no. 81860846) and
Science and Technology Project of Guiyang City [grant no.
(2019)9-2-12].
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
BL conceived and designed the present study. LL, SZ
and QL performed the experiments and analyzed the data. LC
interpreted the data and wrote the manuscript. BL and LC confirm
the authenticity of all the raw data. All authors read and approved
the final manuscript, and agree to be accountable for all aspects
of the research in ensuring that the accuracy or integrity of any
part of the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
All animal experiments involved in this manuscript
were authorized by the ethical committee of First Affiliated
Hospital of Guizhou University of Chinese Medicine (approval date,
10 December 2019; approval no. GUCM-FAH-20191210) and was carried
out according to the guidelines for care and use of laboratory
animals, as well as to the principles of laboratory animal care and
protection.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Uchimido R, Schmidt EP and Shapiro NI: The
glycocalyx: A novel diagnostic and therapeutic target in sepsis.
Crit Care. 23:162019. View Article : Google Scholar
|
|
2
|
Riedemann NC, Guo RF and Ward PA: The
enigma of sepsis. J Clin Invest. 112:460–467. 2003. View Article : Google Scholar
|
|
3
|
Osuchowski MF, Welch K, Siddiqui J and
Remick DG: Circulating cytokine/inhibitor profiles reshape the
understanding of the SIRS/CARS continuum in sepsis and predict
mortality. J Immunol. 177:1967–1974. 2006. View Article : Google Scholar
|
|
4
|
Gotts JE and Matthay MA: Sepsis:
Pathophysiology and clinical management. BMJ. 353:i15852016.
View Article : Google Scholar
|
|
5
|
Hotchkiss RS, Tinsley KW, Swanson PE,
Schmieg RE Jr, Hui JJ, Chang KC, Osborne DF, Freeman BD, Cobb JP,
Buchman TG and Karl IE: Sepsis-induced apoptosis causes progressive
profound depletion of B and CD4+ T lymphocytes in
humans. J Immunol. 166:6952–6963. 2001. View Article : Google Scholar
|
|
6
|
Munoz C, Carlet J, Fitting C, Misset B,
Bleriot JP and Cavaillon JM: Dysregulation of in vitro cytokine
production by monocytes during sepsis. J Clin Invest. 88:1747–1754.
1991. View Article : Google Scholar
|
|
7
|
Wang HW, Yang W, Gao L, Kang JR, Qin JJ,
Liu YP and Lu JY: Adoptive transfer of bone marrow-derived
dendritic cells decreases inhibitory and regulatory T-cell
differentiation and improves survival in murine polymicrobial
sepsis. Immunology. 145:50–59. 2015. View Article : Google Scholar
|
|
8
|
Bouras M, Asehnoune K and Roquilly A:
Contribution of dendritic cell responses to sepsis-induced
immunosuppression and to susceptibility to secondary pneumonia.
Front Immunol. 9:25902018. View Article : Google Scholar
|
|
9
|
Manjuck J, Saha DC, Astiz M, Eales LJ and
Rackow EC: Decreased response to recall antigens is associated with
depressed costimulatory receptor expression in septic critically
ill patients. J Lab Clin Med. 135:153–160. 2000. View Article : Google Scholar
|
|
10
|
Le Tulzo Y, Pangault C, Amiot L, Guilloux
V, Tribut O, Arvieux C, Camus C, Fauchet R, Thomas R and Drénou B:
Monocyte human leukocyte antigen-DR transcriptional downregulation
by cortisol during septic shock. Am J Respir Crit Care Med.
169:1144–1151. 2004. View Article : Google Scholar
|
|
11
|
Ishida Y, Agata Y, Shibahara K and Honjo
T: Induced expression of PD-1, a novel member of the immunoglobulin
gene superfamily, upon programmed cell death. EMBO J. 11:3887–3895.
1992. View Article : Google Scholar
|
|
12
|
Keir ME, Butte MJ, Freeman GJ and Sharpe
AH: PD-1 and its ligands in tolerance and immunity. Annu Rev
Immunol. 26:677–704. 2008. View Article : Google Scholar
|
|
13
|
Naidoo J, Schindler K, Querfeld C, Busam
K, Cunningham J, Page DB, Postow MA, Weinstein A, Lucas AS,
Ciccolini KT, et al: Autoimmune bullous skin disorders with immune
checkpoint inhibitors targeting PD-1 and PD-L1. Cancer Immunol Res.
4:383–389. 2016. View Article : Google Scholar
|
|
14
|
Dai S, Jia R, Zhang X, Fang Q and Huang L:
The PD-1/PD-Ls pathway and autoimmune diseases. Cell Immunol.
290:72–79. 2014. View Article : Google Scholar
|
|
15
|
Brunner-Weinzierl MC and Rudd CE: CTLA-4
and PD-1 control of T-Cell motility and migration: Implications for
tumor immunotherapy. Front Immunol. 9:27372018. View Article : Google Scholar
|
|
16
|
Chen L, Li L, Han Y, Lv B, Zou S and Yu Q:
Tong-fu-li-fei decoction exerts a protective effect on intestinal
barrier of sepsis in rats through upregulating
ZO-1/occludin/claudin-1 expression. J Pharmacol Sci. 143:89–96.
2020. View Article : Google Scholar
|
|
17
|
Fu J, Li G, Wu X and Zang B: Sodium
butyrate ameliorates intestinal injury and improves survival in a
rat model of cecal ligation and puncture-induced sepsis.
Inflammation. 42:1276–1286. 2019. View Article : Google Scholar
|
|
18
|
Rendon JL, Li X, Akhtar S and Choudhry MA:
Interleukin-22 modulates gut epithelial and immune barrier
functions following acute alcohol exposure and burn injury. Shock.
39:11–18. 2013. View Article : Google Scholar
|
|
19
|
Ke J, Bian X, Liu H, Li B and Huo R:
Edaravone reduces oxidative stress and intestinal cell apoptosis
after burn through up-regulating miR-320 expression. Mol Med.
25:542019. View Article : Google Scholar
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
21
|
Wichterman KA, Baue AE and Chaudry IH:
Sepsis and septic shock-a review of laboratory models and a
proposal. J Surg Res. 29:189–201. 1980. View Article : Google Scholar
|
|
22
|
Singer M, Deutschman CS, Seymour CW,
Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche
JD, Coopersmith CM, et al: The Third International consensus
definitions for sepsis and septic shock (Sepsis-3). JAMA.
315:801–810. 2016. View Article : Google Scholar
|
|
23
|
Shankar-Hari M, Harrison DA, Rubenfeld GD
and Rowan K: Epidemiology of sepsis and septic shock in critical
care units: Comparison between sepsis-2 and sepsis-3 populations
using a National critical care database. Br J Anaesth. 119:626–636.
2017. View Article : Google Scholar
|
|
24
|
Thomas H: Sepsis: Bile acids promote
inflammation in cholestasis-associated sepsis. Nat Rev
Gastroenterol Hepatol. 14:324–325. 2017. View Article : Google Scholar
|
|
25
|
Rosenthal MD and Moore FA: Persistent
inflammation, immunosuppression, and catabolism: Evolution of
multiple organ dysfunction. Surg Infect (Larchmt). 17:167–172.
2016. View Article : Google Scholar
|
|
26
|
Zhu Y, Wang Y, Teng W, Shan Y, Yi S, Zhu S
and Li Y: Role of aquaporin-3 in intestinal injury induced by
sepsis. Biol Pharm Bull. 42:1641–1650. 2019. View Article : Google Scholar
|
|
27
|
Chang X, Wang LL, Lian SJ, Tang Q, Chen P
and Wang H: Effect of oral glutamine on intestinal barrier function
in young rats with endotoxemia. Zhongguo Dang Dai Er Ke Za Zhi.
12:809–811. 2010.(In Chinese).
|
|
28
|
Meena AS, Shukla PK, Sheth P and Rao R:
EGF receptor plays a role in the mechanism of glutamine-mediated
prevention of alcohol-induced gut barrier dysfunction and liver
injury. J Nutr Biochem. 64:128–143. 2019. View Article : Google Scholar
|
|
29
|
Chao CY, Sung PJ, Wang WH and Kuo YH:
Anti-inflammatory effect of Momordica charantia in sepsis mice.
Molecules. 19:12777–12788. 2014. View Article : Google Scholar
|
|
30
|
Venet F, Rimmele T and Monneret G:
Management of sepsis-induced immunosuppression. Crit Care Clin.
34:97–106. 2018. View Article : Google Scholar
|
|
31
|
Venet F and Monneret G: Advances in the
understanding and treatment of sepsis-induced immunosuppression.
Nat Rev Nephrol. 14:121–137. 2018. View Article : Google Scholar
|
|
32
|
Roquilly A, McWilliam HEG, Jacqueline C,
Tian Z, Cinotti R, Rimbert M, Wakim L, Caminschi I, Lahoud MH, Belz
GT, et al: Local modulation of antigen-presenting cell development
after resolution of pneumonia induces long-term susceptibility to
secondary infections. Immunity. 47:135–147 e5. 2017. View Article : Google Scholar
|
|
33
|
Chang KC, Burnham CA, Compton SM, Rasche
DP, Mazuski RJ, McDonough JS, Unsinger J, Korman AJ, Green JM and
Hotchkiss RS: Blockade of the negative co-stimulatory molecules
PD-1 and CTLA-4 improves survival in primary and secondary fungal
sepsis. Crit Care. 17:R852013. View
Article : Google Scholar
|
|
34
|
Chang K, Svabek C, Vazquez-Guillamet C,
Sato B, Rasche D, Wilson S, Robbins P, Ulbrandt N, Suzich J, Green
J, et al: Targeting the programmed cell death 1: Programmed cell
death ligand 1 pathway reverses T cell exhaustion in patients with
sepsis. Crit Care. 18:R32014. View
Article : Google Scholar
|
|
35
|
Li J, Jie HB, Lei Y, Gildener-Leapman N,
Trivedi S, Green T, Kane LP and Ferris RL: PD-1/SHP-2 inhibits
Tc1/Th1 phenotypic responses and the activation of T cells in the
tumor microenvironment. Cancer Res. 75:508–518. 2015. View Article : Google Scholar
|
|
36
|
Zhao R, Song Y, Wang Y, Huang Y, Li Z, Cui
Y, Yi M, Xia L, Zhuang W, Wu X and Zhou Y: PD-1/PD-L1 blockade
rescue exhausted CD8+ T cells in gastrointestinal
stromal tumours via the PI3K/Akt/mTOR signalling pathway. Cell
Prolif. 52:e125712019. View Article : Google Scholar
|