Introduction
Ulcerative colitis (UC) is a chronic disease that is
difficult to cure using conventional treatments. It is often
accompanied by diarrhea, weight loss, abdominal pain and other
clinical symptoms, such as blood in the stool, vomiting and nausea.
The occurrence of UC is related to a variety of factors, including
intestinal flora, intestinal mucosal immunity, external
environments and genetic susceptibility. However, the pathogenesis
of UC is not fully understood (1). Until now, anti-inflammatory drugs
and immune modulators, including aminosalicylates, corticosteroids,
monoclonal antibodies and immunosuppressants, have been used in the
clinic to control severe symptoms (2,3).
Although enteritis can be improved to some extent, these
traditional medicines usually result in side effects, such as
osteopenia and growth failure (4), thus are not suitable for long-term
treatment. The cure rate of enteritis is very low (5). Therefore, safe therapeutic
approaches with improved efficiency are needed.
Functional oligosaccharides are carbohydrates
composed of 2–10 monosaccharide units linked together via
O-glycosidic or N-glycosidic bonds (6). They are associated with numerous
health benefits, including regulating the immune response and
alleviating allergic inflammation, and can reduce intestinal
inflammation and modulate the composition of the intestinal
microbiota, particularly in UC, to improve the host response to
pathogens. Studies have shown that the most common functional types
of oligosaccharide primarily include galacto-oligosaccharide (GOS),
chitosan-oligosaccharide (COS), pectin-oligosaccharide (POS),
fructo-oligosaccharide and isomalto-oligosaccharide (IMO) (7–9).
IMO is a commercial mixture of glucose monomers that can improve
intestinal absorption by modulating epithelial functions and
stimulating enteric motor neurons (10). POS is composed of rhamnose and
galacturonic acid (11), and it
can promote the generation of short-chain fatty acids to inhibit
pathogenic bacteria in the intestine and decrease the incidence of
colon cancer (12).
Weilan gum (WLG) polysaccharide is a macromolecular
exopolysaccharide secreted by Alcaligenes (13) that has broad commercial
applications in the food industry as a thickener, suspending agent,
binder and emulsifier (14). For
example, WLG functions as a protective colloid and plays an
important role in eliminating protein flocculation in dairy
products (15,16). WLG can be used as a food additive
in baked products to replace oils, fats, margarine and other fatty
acids to improve the taste (17),
and as a thickener in ice cream products (18). Therefore, WLG may be a safe
approach to alleviate UC. According to a previous report,
commercialized WLG is an acidic hetero-polysaccharide composed of
glucose, rhamnose, glucuronic acid and mannose, with a molecular
weight (Mw) of ~1.0×106 (19). Due to its complex structure and
high Mw, it is difficult for WLG to permeate into epithelial cells
and be utilized by intestinal microorganisms (13). Thus, trifluoroacetic acid (TFA)
has been used to degrade WLG into oligosaccharides (20). Studies have shown that WLG has no
side effects on clinical signs and body weight (21,22). Additionally, WLG is composed of
glucose, rhamnose, glucuronic acid and mannose (23). Glucose is widely used in
confectionery and medicine (24),
rhamnose is used as a sweetener in the food industry (25), and glucuronic acid and mannose can
be used directly to synthesize glycoproteins to participate in
immune regulation and be used as glyconutrients in clinical
medicine (26). All these data
indicate that both WLG and WLG oligosaccharide (WLGO) are safe for
use in humans. Thus, it was hypothesized in the present study that
WLGO may display similar effects on intestinal health as POS and
IMO.
The objectives of this study were to explore the
therapeutic effects of WLGO on UC. WLGO structural and physical
properties were characterized by electrospray mass spectrometry
(ESI) and Fourier transform infrared spectroscopy (FTIR), and its
effects on UC were assessed in vitro by using
lipopolysaccharide (LPS)-induced Caco-2 cells and in vivo by
using dextran sulfate sodium (DSS)-induced UC model mice. Body
weight loss, disease activity index (DAI) scores, colon
histopathological changes and inflammatory cytokine interleukin-1β
(IL-1β), interleukin-6 (IL-6) and tumor necrosis factor α (TNF-α)
levels were measured to evaluate the therapeutic effect of WLGO on
UC.
Materials and methods
Chemicals and reagents
WLG (purity >90%; Mw 1.0×106) was
obtained from Shandong Academy of Sciences. TFA (cat. no. 76-0501;
purity >99%; Mw 114.02 g/mol) (76-05-1; http://www.sigmaaldrich.cn) was purchased from
Shanghai Macklin Biochemical Co., Ltd. DSS (cat. no. 9011-18-1.
MW40000) (https://www.aladdin-e.com) was
purchased from Shanghai Aladdin Biochemical Technology Co., Ltd.
LPS (purity >99%; Escherichia coli 055:B5; Sigma-Aldrich;
Merck KGaA) was dissolved in ddH2O to make a 1 mg/ml
stock solution. Human IL-1β (cat. no. 1110122), human IL-6 (cat.
no.1110602), human TNF-α (cat. no. 1117202), mouse IL-1β (cat. no.
1210122), mouse IL-6 (cat. no. 1210602) and mouse TNF-α (cat. no.
1317202) ELISA kits purchased from Beijing Dakowei Biotechnology
Co., Ltd. were used. CYN (cat. no. Z36020518) was purchased from
Zhejiang Jinhua Conba Biopharm Co., Ltd.
Preparation of WLGO
A total of 2 mg WLG was dissolved in 4 ml TFA (2
mol/l) in a hydrolysis tube and hydrolyzed at 110°C for 3 h in an
autoclave. WLGO solution was mixed with 5 ml methanol, and the
solvent was evaporated in a rotary evaporator. The procedure was
repeated four times, and the obtained WLGO was analyzed with ESI
and FTIR. For the FTIR, WLGO was prepared as Kbr tablets and
examined using a Bruker Tensor 27 model infrared spectrometer (scan
range, 400-4,000 cm−1; resolution, 4.0 cm−1).
The obtained spectra were the results of averaging 64 scans.
Cell culture and treatment
Caco-2 cells (presented by Professor Yanqing Li from
Qilu Hospital of China) were used as the epithelial cell model for
this study. Cells were cultured in high-glucose DMEM (DMEM-H; cat.
no. 12800-017; Gibco; Thermo Fisher Scientific, Inc.) supplemented
with 10% v/v FBS (cat. no. SV30087.02; HyClone; Cytiva) in a
humidified incubator with 5% CO2 at 37°C. The cells were
divided into the following five groups: i) Normal group (Nor); ii)
LPS group; iii) LPS + 50 µg/ml WLGO group; iv) LPS + 100 µg/ml WLGO
group; and v) LPS + 200 µg/ml WLGO group. For the induction of
inflammatory conditions, cells in the LPS and LPS + WLGO groups
were treated with 50 µg/ml LPS for 12 h in a humidified incubator
with 5% CO2 at 37°C. After LPS treatment for 12 h, cells
were cultured in the presence of WLGO for 12, 24 or 48 h. At the
end of cell culture experiments, the supernatants were collected by
centrifugation at 1,000 × g at 4°C for 10 min. The samples stored
at −80°C for subsequent analyses.
Animals and treatment
A total of 30 male C57BL/6J mice (age, 6 weeks;
weight, 22–25 g) were purchased from Beijing Vital River Laboratory
Animal Technology Co., Ltd. and used in the animal experiments. All
mice were adapted to the environment for 7 days before the
experiments. All mice were housed in Makrolon cages, maintained
under an air-conditioned atmosphere at 22±2°C with 50–60% humidity,
12 h light/dark cycles, and free access to tap water and standard
rodent diet. These mice were randomly divided into the following
five groups: i) Normal control group (Nor); ii) DSS group; iii)
Chang Yan Ning group (CYN, 2.24 g/kg/day); iv) high WLGO group
(4.48 g/kg/day); and v) low WLGO group (2.24 g/kg/day). C57BL/6J
mice received an oral dose of 200 µl 2.5% DSS solution for 7 days
to establish the UC mouse model. Mice in the Nor group were
received sterile water via oral administration. For induction of
UC, mice in the DSS, CYN and WLGO groups were treated with drinking
water containing 2.5% (w/v) DSS once per day for 7 days.
Subsequently, mice in the CYN and WLGO groups were treated with
drinking water containing CYN or WLGO, respectively, dissolved in
sterile water for 7 days. Body weight was recorded every day during
the experiment (27). At the end
of the experiment, all mice were anesthetized and euthanized by
cervical dislocation. The organs were removed and examined. Colon
and blood samples were collected. Colons were measured to determine
their length, then divided into three segments and stored at −80°C
for hematoxylin and eosin (H&E) staining, ELISA and reverse
transcription-quantitative PCR (RT-qPCR) (28). All animal experiments were
performed in compliance with the Animal Research: Reporting of
In Vivo Experiments guidelines, the UK Animals (Scientific
Procedures) Act (1986) and the associated guidelines, the EU
Directive 2010/63/EU for Animal experiments, the National
Institutes of Health Guide for the Care and Use of Laboratory
Animals (NIH Publications no. 8023; revised 1978) and the Animal
Management Rules of the Chinese Ministry of Health (http://www.gov.cn/gongbao/content/2017/content_5219148.htm)
(29). This study was approved by
the Animal Experiment Ethics Committee of Qilu University of
Technology (Jinan, China) (30).
Cell viability assay
The
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltertrazolium bromide (MTT)
assay was used to assess cell viability in response to WLGO. Caco-2
cells were seeded (5×104 cells/100 µl) in 96-well plates
and treated with different concentrations (50–200 µg/ml) of WLGO.
After 24 and 48 h, 20 µl MTT reagent (0.5 mg/ml) was added into
each well for another 4 h at 37°C. The medium was removed and 100
µl DMSO (0.04 M) was added. The optical density was measured at a
wavelength of 570 nm using a SpectraMax ABS microplate
spectrophotometer (Molecular Devices, LLC) (31).
DAI and histological assessment
The DAI was calculated and recorded in accordance
with stool consistency, body weight loss and blood in feces. Colon
samples were fixed in optimal cutting temperature embedding medium
and stained with H&E. Each colon was assessed using four
consecutive 8-µm sections taken every 40 µm to cover the entire
colon. Histopathological changes were estimated according to a
previously established scoring system. In brief, the scoring system
comprised two parameters, tissue damage and infiltration of lamina
propria by inflammatory cells, and scored from 0 (no changes) to 6
(widespread cellular infiltration and extensive tissue damage). The
intestinal inflammation was graded blindly by two observers under
the guidance of previously established scoring system using a light
microscope (32,33).
ELISA
The colon tissues were weighed, and homogenized with
NP40 lysis buffer (Beyotime Institute of Biotechnology) on ice. The
mixture was centrifuged at 5,000 × g and 4°C for 15 min, and the
supernatants were collected and quantified using a BCA assay
(Beyotime Institute of Biotechnology) as reported previously
(34). The serum was prepared by
centrifugation at 3,000 × g for 20 min at 4°C and stored at −80°C
for biochemical analysis. ELISA kits were used to measure IL-1β,
IL-6 and TNF-α levels in mouse colon tissue and blood serum
samples.
RT-qPCR
Total RNA was extracted from colon tissues using
TRIzol® (Invitrogen; Thermo Fisher Scientific, Inc.).
The purity and concentration of the isolated RNA were determined
using NanoDrop 1000 (NanoDrop Technologies; Thermo Fisher
Scientific, Inc.), RNA samples were reverse transcribed into cDNAs
using an ABScript II RT Master Mix (ABclonal Biotech Co., Ltd.)
according to the manufacturer's protocol. The cDNAs were subjected
to qPCR using the following primers (purchased from Sangon Biotech
Co., Ltd.): IL-1β forward, 5′-GCCACCTTTTGACAGTGATGAG-3′ and
reverse, 5′-ATGTGCTGCTGCGAGATTTG-3′; IL-6 forward,
5′-ACCCCAATTTCCAATGCTCTCC-3′ and reverse, 5′-GCATAACGCACTAGGTTTGCC
−3′; TNF-α forward, 5′-GGACTAGCCAGGAGGGAGAACAG-3′ and reverse,
5′-GCCAGTGAGTGAAAGGGACAGAAC −3′; and GAPDH forward,
5′-TGTGTCCGTCGTGGATCTGA-3′ and reverse, 5′-TTGCTGTTGAAGTCGCAGGAG
−3′. The mRNA expression levels were examined on a Rotor-Gene Q
instrument (Qiagen China Co., Ltd.) using a SYBR Green Fast qPCR
Mix (ABclonal Biotech Co., Ltd.) to evaluate the amount of double
stranded DNA. The primer amplification efficiencies were measured
in cDNA dilutions from 1×10 to 1×105 copies, and the
amplification was linear over the range of 1×10 to 1×105
copies. The efficiencies of the primers in the standard curves
ranged from 97 to 102%. The following thermocycling conditions were
used for qPCR: 95°C for 30 sec; 40 cycles at 58°C for 30 sec and at
72°C for 30 sec; and a final cycle at 72°C for 5 min. IL-1β, IL-6
and TNF-α gene expression levels were normalized against GAPDH
using the 2−ΔΔCq method (35).
Statistical analysis
All experiments were repeated at least three times
independently. Data were recorded and analyzed using Excel (version
2010; Microsoft Corporation) and GraphPad Prism software (version
8.0.2; GraphPad Software, Inc.) Data are presented as the mean ±
SEM. Differences among Nor, DSS, CYN and WLGO groups were analyzed
by Kruskal-Wallis followed by Dunn's post hoc test or one-way ANOVA
followed by Tukey's post hoc test using SPSS software (version
11.5; SPSS, Inc.). P<0.05 was considered to indicate a
statistically significant difference.
Results
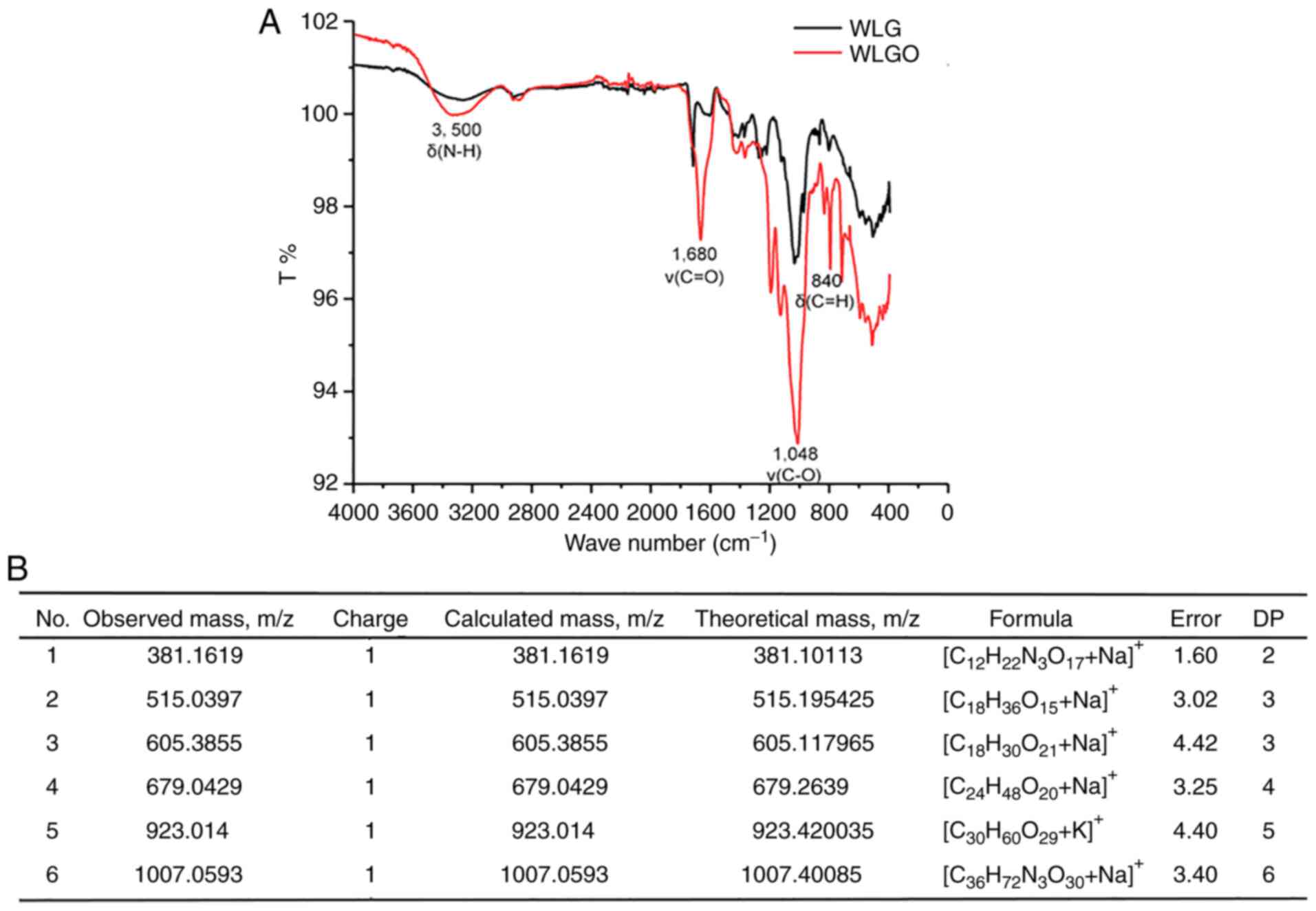
Chemical profile of WLGO
The molecular mass and structure of WLGO were
characterized using ESI and FTIR analysis. FTIR analysis showed the
bands of 840 cm−1 (C-H deformation vibration), 1,048
cm−1 (C-O deformation vibration), 1,680 cm−1
(C=O stretching vibration) and 3,500 cm−1 (N-H
deformation vibration) in WLGO (Fig.
1A), which was consistent with a previous study reporting that
WLG is composed of repeating units with glucose, rhamnose,
glucuronic acid and mannose (19). ESI analysis showed that the
polymerization degrees of WLGO were mainly between 2 and 6
(Fig. 1B). WLGO with a
polymerization degree of 2 consists of glucuronic acid and rhamnose
units. WLGO with a polymerization degree of 3 consists of three
rhamnose units. WLGO with a polymerization degree of 4 consists of
four rhamnose units. WLGO with a polymerization degree of 5
contains four mannoses units and one rhamnose unit. WLGO with a
polymerization degree of 6 contains six rhamnose units. The m/z of
WLGO was mainly concentrated in 381.10113 and 1007.40085. Overall,
the ESI and FTIR results confirmed that the WLG was successfully
degraded into WLGO.
Effect of WLGO on Caco-2 cell
viability
To evaluate the non-toxic concentration of WLGO,
Caco-2 cells were incubated with 50, 100 and 200 µg/ml WLGO for 24
and 48 h. The results showed that WLGO had no cytotoxic effects on
the Caco-2 cells after treatment for 24 h (Fig. 2A).
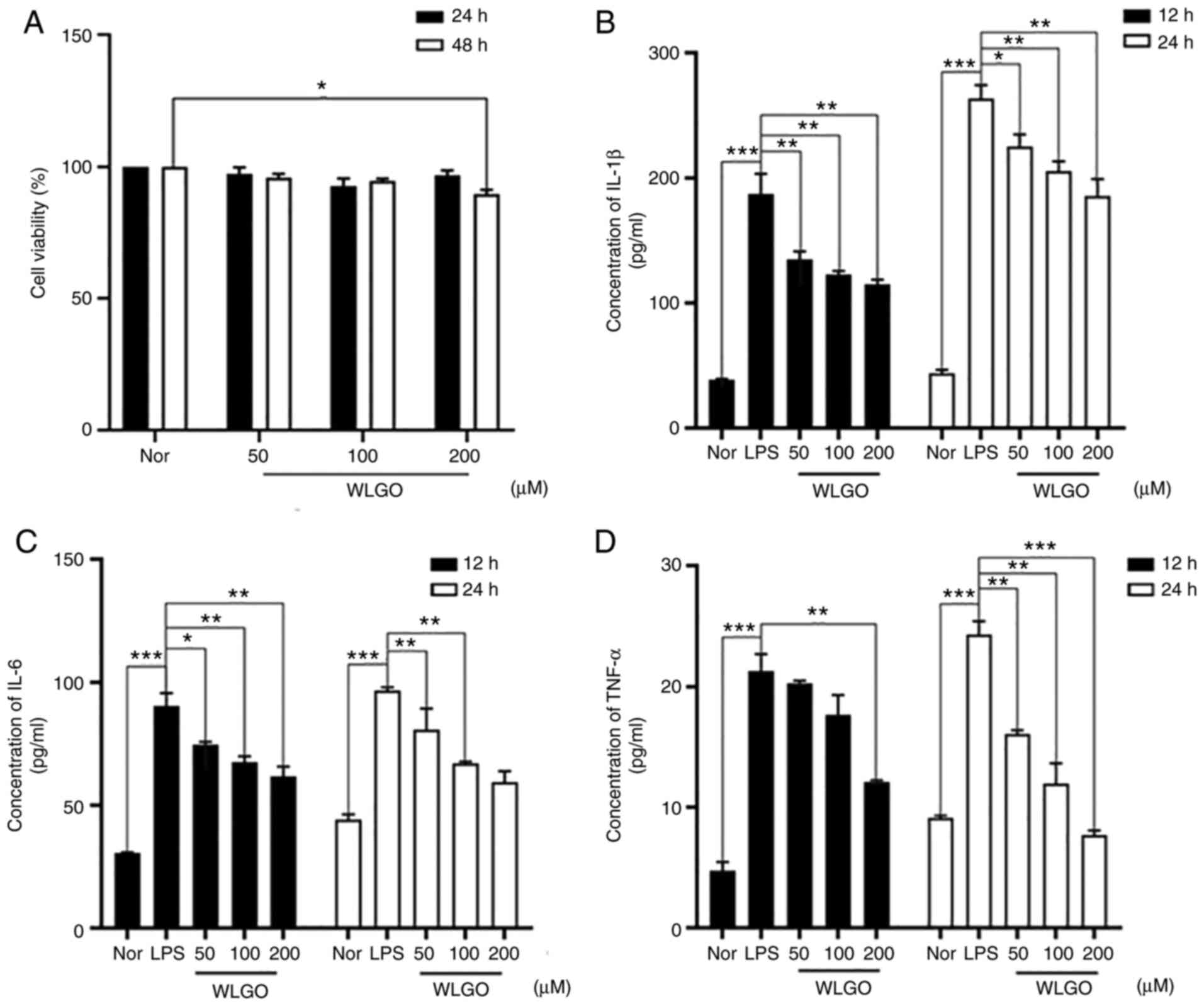 | Figure 2.Caco-2 cell viability and IL-1β, IL-6
and TNF-α levels in Caco-2 cells after treatment with WLGO at
different doses. (A) Caco-2 cells were treated with different
concentrations (50–200 µg/ml) of WLGO for 24 and 48 h. (B) IL-1β,
(C) IL-6 and (D) TNF-α levels were examined in the supernatant of
Caco-2 cells that were pretreated with 50 µg/ml LPS for 12 h and
then treated with different concentrations of WLGO for 12 or 24 h.
The following groups were assessed: i) Nor, normal group; ii) LPS,
Caco-2 cells stimulated with LPS; and iii) WLGO, Caco-2 cells
stimulated with LPS and 50, 100 or 200 µg/ml WLGO, as indicated.
Data are expressed as the mean ± SEM. All experiments were
performed in triplicate. *P<0.05, **P<0.01, ***P<0.001;
IL, interleukin; TNF-α, tumor necrosis factor α; WLGO, weilan gum
oligosaccharide; LPS, lipopolysaccharide; Nor, normal. |
WLGO decreases LPS-induced
upregulation of TNF-α, IL-6 and IL-1β
To explore the anti-inflammatory effects of WLGO
in vitro, LPS-induced Caco-2 cells were used (19). Compared with those in the Nor
group, LPS significantly increased the concentrations of IL-1β,
IL-6 and TNF-α (Fig. 2B-D). WLGO
treatment significantly decreased IL-1β levels in a
concentration-dependent manner in LPS-induced Caco-2 cells
(Fig. 2B). In addition, 50, 100
and 200 µg/ml WLGO treatment significantly decreased IL-6 levels in
LPS-induced Caco-2 cells (Fig.
2C). Moreover, 200 µg/ml WLGO treatment significantly decreased
TNF-α levels both at 12 and 24 h (Fig. 2D), and 50 and 100 µg/ml WLGO
treatment significantly decreased TNF-α levels at 24 h. These
results suggested that WLGO had an anti-inflammatory effect on
LPS-induced cells.
WLGO alleviates the clinical symptoms
of DSS-induced colitis
To investigate the anti-inflammatory role of WLGO,
C57BL/6J mice received an oral dose of 200 µl 2.5% DSS solution for
7 days to establish the UC mouse model. During DSS treatment, body
weight was measured. Compared with that in the Nor group, the body
weight of mice in the DSS group reduced significantly, reaching the
lowest value at day 10 (Fig.
S1). The percentage of body weight loss was reduced by 10.9% in
the DSS group on day 14 compared with that in the Nor group.
However, the percentage of body weight loss was reduced by 7.9% in
the WLGO-High group at the 14 day time point, indicating that WLGO
could relieve the reduction in body weight in mice with DSS-induced
colitis (P<0.05; Figs. 3A and
S1).
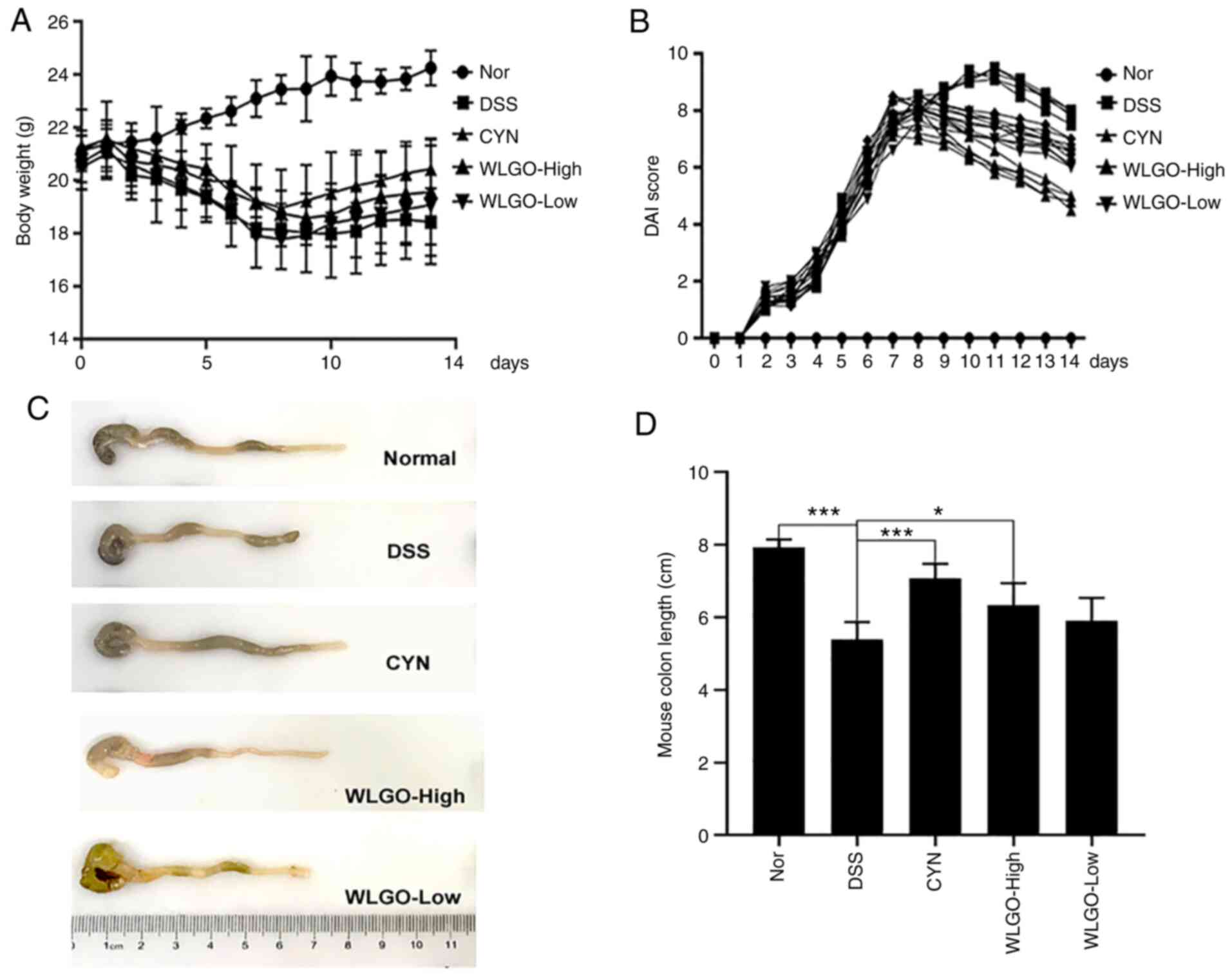 | Figure 3.Effects of WLGO treatment on
DSS-induced ulcerative colitis model mice. (A) Body weight of mice
was measured every day. (B) DAI score of mice was examined every
day. (C) Images of colons. (D) Calculated colon lengths based on
images from C. The following groups were assessed: i) Nor, mice
treated with saline; ii) DSS, mice treated with drinking water
containing 2.5% DSS; iii) CYN, mice treated with 2.24 g/kg/day CYN;
iv) WLGO-High, mice treated with 4.48 g/kg/day WLGO; and v)
WLGO-Low, mice treated with 2.24 g/kg/day WLGO. Data are expressed
as the mean ± SEM. All experiments were performed in triplicate.
*P<0.05, ***P<0.001. WLGO, weilan gum oligosaccharide; DSS,
dextran sulfate sodium; DAI, disease activity index; CYN, Chang Yan
Ning; Nor, normal. |
Mice in the DSS group exhibited apparent rectal
bleeding and diarrhea after DSS treatment. As the treatment
continued, the severity of inflammation was evident in the mice, as
measured by the DAI score. Compared with that in the Nor group, the
DAI score in the DSS group increased (Figs. 3B and S2). Moreover, compared with the DSS
group, the DAI scores of DSS-treated mice were significantly
reduced in the WLGO treatment groups (Fig. S2). These results indicated that
WLGO could relieve the symptoms of DSS-induced UC. The organs
(heart, spleen, kidney, lung and liver) of mice in each treatment
group were examined. There were no notable changes in any of the
organs among the different groups (Fig. S3).
WLGO protects mucosal integrity
against injury
The colon length of mice in the DSS group was
significantly shorter compared with that in the Nor group
(P<0.05; Fig. 3C and D). The
colon length of mice in all three treatment groups was notably
longer compared with that in the DSS group. Moreover, the colon
length of mice in the CYN group almost returned to the level
observed in the Nor group, which significantly relieved the colon
length loss in DSS-induced UC.
DSS-induced intestinal inflammation was accompanied
by mucosal infiltration of inflammatory cells. Histological
examination of frozen colon tissue sections showed the degree of
inflammation and epithelial damage. Multifocal erosion areas and
severe inflammatory cell infiltration were observed on the colonic
surface epithelium of mice in the DSS group (Fig. 4A). By contrast, WLGO-High
treatment notably ameliorated histological alterations and
decreased histological scores compared with those in the DSS group
(Fig. 4B). These results
indicated that WLGO at a high dose exhibited a protective effect
against mucosal injury.
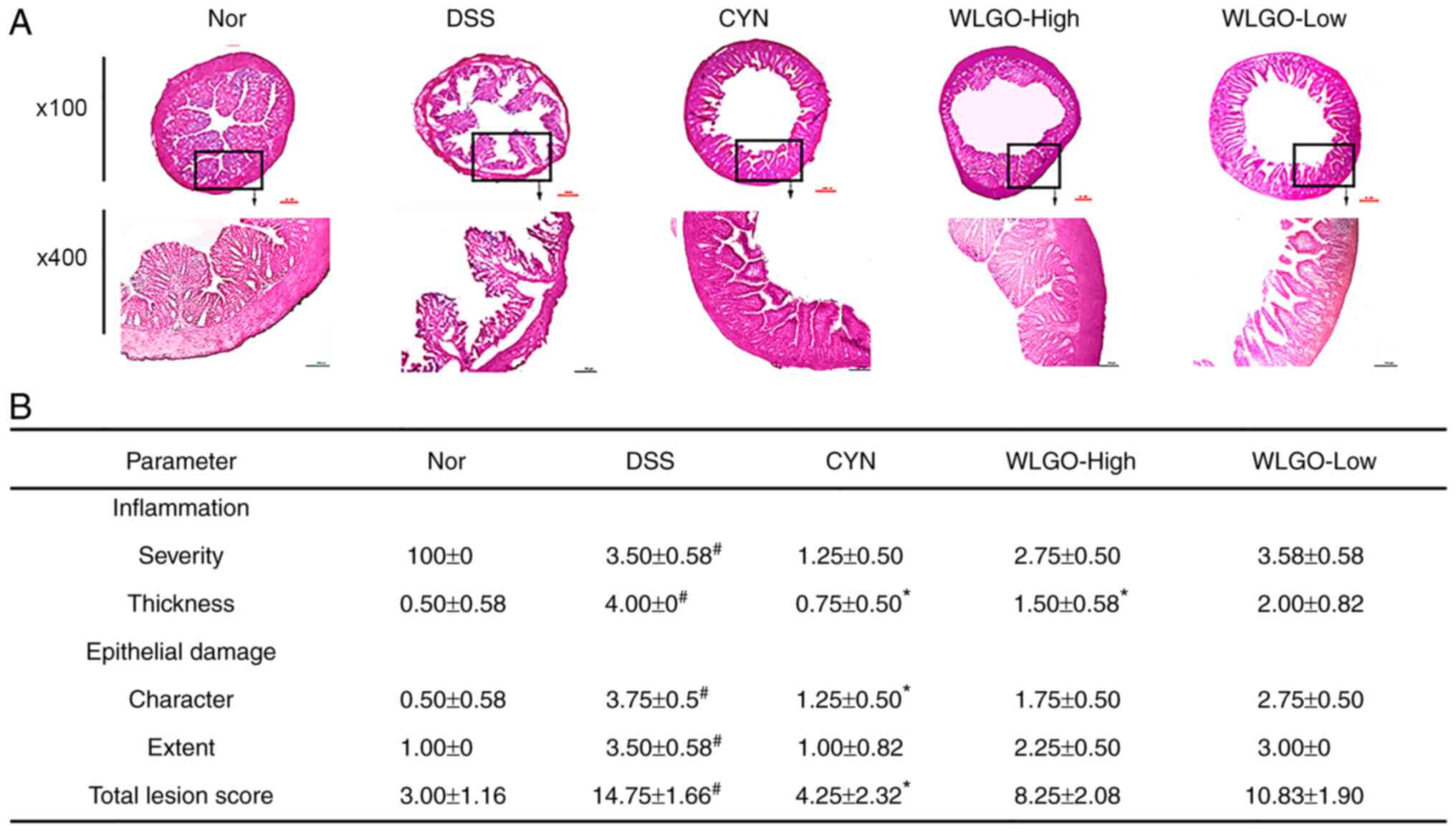 | Figure 4.Histological sections of colonic
tissues stained with H&E. (A) Histological sections of colonic
tissues stained with H&E under a microscope. (B) Effects of
WLGO on colon pathology of DSS-induced ulcerative colitis model
mice. The following groups were assessed: i) Nor, mice treated with
saline; ii) DSS, mice treated with drinking water containing 2.5%
DSS; iii) CYN, mice treated with 2.24 g/kg/day CYN; iv) WLGO-High,
mice treated with 4.48 g/kg/day WLGO; and v) WLGO-Low, mice treated
with 2.24 g/kg/day WLGO. Data are expressed as the mean ± SEM. All
experiments were performed in triplicate. #P<0.001
vs. Nor; *P<0.05 vs. DSS. H&E, hematoxylin and eosin; WLGO,
weilan gum oligosaccharide; DSS, dextran sulfate sodium; CYN, Chang
Yan Ning; Nor, normal. |
WLGO decreases proinflammatory
cytokines in DSS-induced mice
To investigate the treatment effect of WLGO on
DSS-induced UC, the levels of proinflammatory cytokines (IL-1β,
IL-6 and TNF-α) in serum and colon tissues were measured using
ELISAs. The results showed that the levels of IL-1β, IL-6 and TNF-α
were significantly increased in mice in the DSS group compared with
those in the Nor group (Fig.
5A-C). In addition, compared with those in the DSS group,
levels of IL-1β, IL-6 and TNF-α in both serum and colon tissues
were significantly decreased in the WLGO-High group (Figs. 5A-C and 6A-C). These results suggested that all
three proinflammatory cytokines were decreased after WLGO
treatment, and the most significant changes were observed with a
high dose of WLGO.
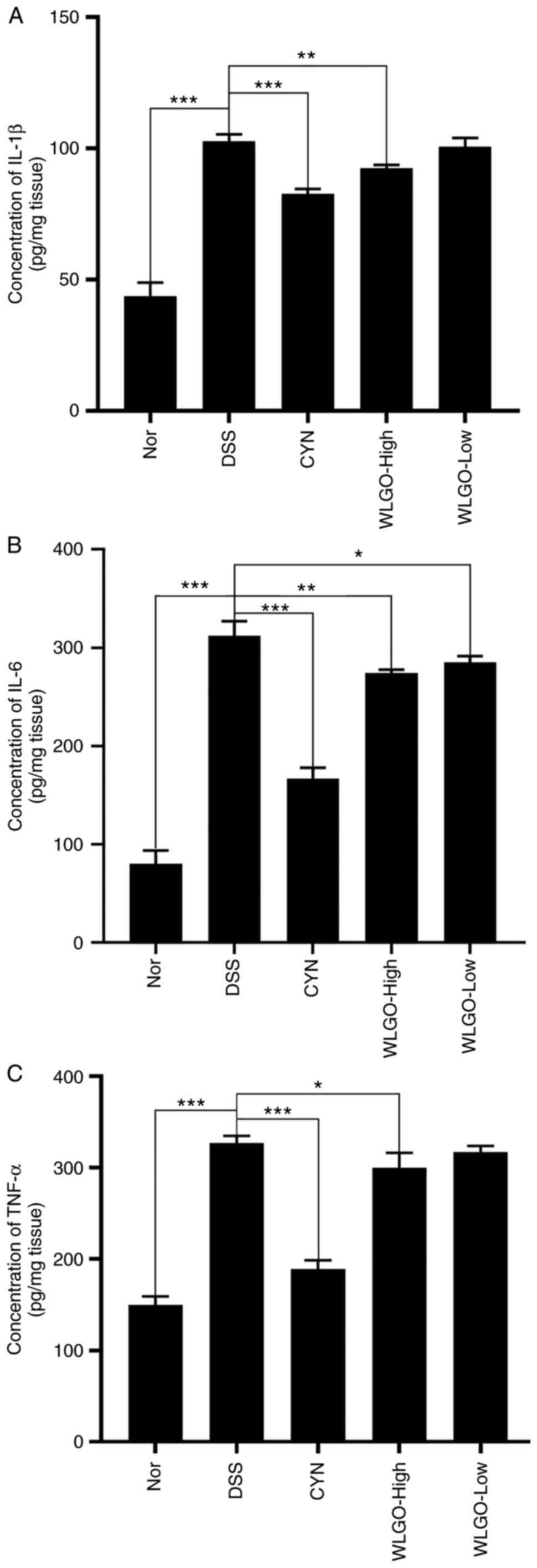 | Figure 5.Effects of WLGO on the levels of
inflammatory cytokines in colonic tissues of mice. Levels of (A)
IL-1β, (B) IL-6 and (C) TNF-α in the colonic homogenates of
DSS-induced ulcerative colitis model mice were detected using ELISA
kits. The following groups were assessed: i) Nor, mice treated with
saline; ii) DSS, mice treated with drinking water containing 2.5%
DSS; iii) CYN, mice treated with 2.24 g/kg/day CYN; iv) WLGO-High,
mice treated with 4.48 g/kg/day WLGO; and v) WLGO-Low, mice treated
with 2.24 g/kg/day WLGO. Data are expressed as the mean ± SEM. All
experiments were performed in triplicate. *P<0.05, **P<0.01,
***P<0.001. WLGO, weilan gum oligosaccharide; DSS, dextran
sulfate sodium; CYN, Chang Yan Ning; IL, interleukin; TNF-α, tumor
necrosis factor α; Nor, normal. |
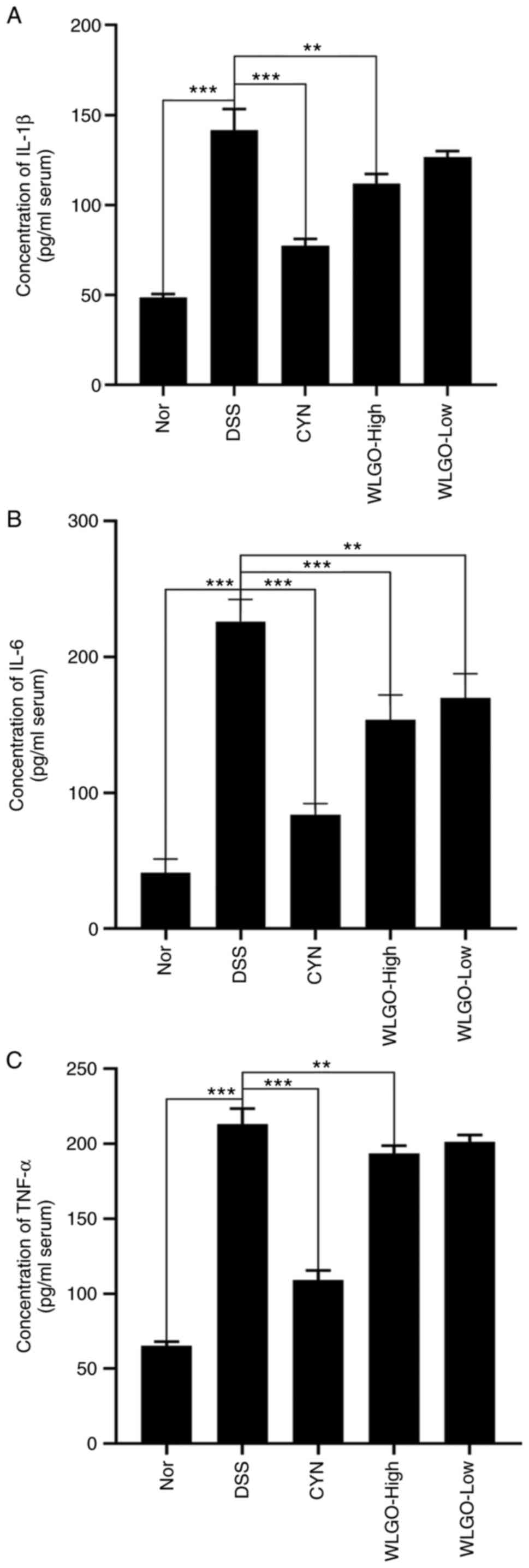 | Figure 6.Effects of WLGO on the serum levels
of inflammatory cytokines. Levels of (A) IL-1β, (B) IL-6 and (C)
TNF-α in the blood serum of DSS-induced ulcerative colitis model
mice were detected using ELISA kits. The following groups were
assessed: i) Nor, mice treated with saline; ii) DSS, mice treated
with drinking water containing 2.5% DSS; iii) CYN, mice treated
with 2.24 g/kg/day CYN; iv) WLGO-High, mice treated with 4.48
g/kg/day WLGO; v) WLGO-Low, mice treated with 2.24 g/kg/day WLGO.
Data are expressed as the mean ± SEM. All experiments were
performed in triplicate. **P<0.01, ***P<0.001. WLGO, weilan
gum oligosaccharide; DSS, dextran sulfate sodium; CYN, Chang Yan
Ning; IL, interleukin; TNF-α, tumor necrosis factor α; Nor,
normal. |
To explore the anti-inflammatory effect of WLGO on
mRNA expression levels, the mRNA expression levels of IL-1β, IL-6
and TNF-α in the colons of mice with DSS-induced UC were measured
via RT-qPCR. The results showed that, compared with those in the
Nor group, the mRNA expression levels of IL-1β, IL-6 and TNF-α
increased significantly in the DSS group, whereas WLGO-High
treatment significantly reversed these increases (Fig. 7A-C). The results demonstrated that
WLGO inhibited the expression of proinflammatory cytokines (IL-1β,
IL-6 and TNF-α) in mice with DSS-induced UC, and WLGO at a high
dose exhibited stronger effects than WLGO at a low dose.
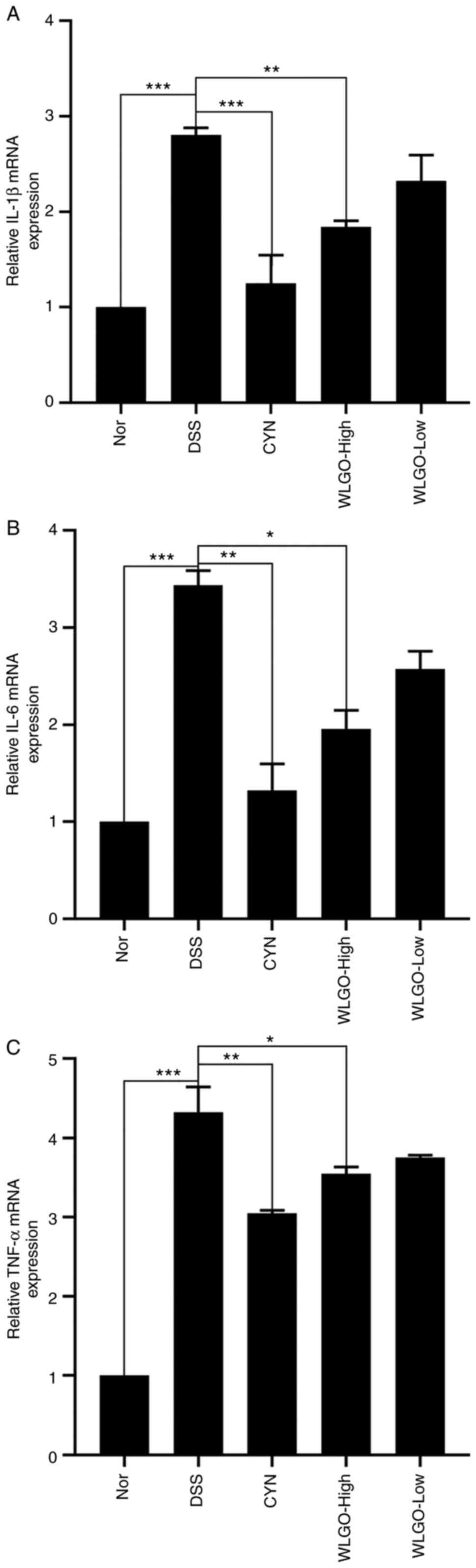 | Figure 7.Effects of WLGO on the mRNA
expression levels of inflammatory cytokines in the colons of mice.
mRNA expression levels of (A) IL-1β, (B) IL-6 and (C) TNF-α in the
colons of DSS-induced ulcerative colitis model mice were detected.
The following groups were assessed: i) Nor, mice treated with
saline; ii) DSS, mice treated with drinking water containing 2.5%
DSS; iii) CYN, mice treated with 2.24 g/kg/day CYN; iv) WLGO-High,
mice treated with 4.48 g/kg/day WLGO; and v) WLGO-Low, mice treated
with 2.24 g/kg/day WLGO. Data are expressed as the mean ± SEM. All
experiments were performed in triplicate. *P<0.05, **P<0.01,
***P<0.001. WLGO, weilan gum oligosaccharide; DSS, dextran
sulfate sodium; CYN, Chang Yan Ning; IL, interleukin; TNF-α, tumor
necrosis factor α; Nor, normal. |
Discussion
WLGO is composed of glucose, rhamnose, glucuronic
acid and mannose, and its structure is similar to that of POS
(36). Compared with other
oligosaccharides, WLGO is more diverse in its monosaccharide
composition (37). Among its
components, glucuronic acid has a wide variety of pharmacological
activities, including anti-inflammatory, cell protective and immune
regulatory effects (38–40). Mannose can inhibit tumor growth
and prolong the survival of patients with cancer (41). Therefore, it was proposed that
WLGO may display more biological activities than other
oligosaccharides.
Although UC can be triggered by bacteria, viruses
and other environmental factors, the inflammatory process of the
intestinal mucosa is ultimately induced by soluble inflammatory
mediators (42). The inflammatory
mediators IL-1β, IL-6 and TNF-α play leading roles in the
development of UC (43).
Therefore, the effective reduction of IL-1β, IL-6 and TNF-α in the
serum and colon tissues is a reasonable modality for UC treatment
(44,45). The present data suggested that
IL-1β, IL-6 and TNF-α levels were decreased by WLGO in the colon
tissues and serum samples of DSS-induced UC model mice. These
results demonstrated that WLGO alleviated DSS-induced UC in mice by
reducing the levels of inflammatory cytokines.
The present study showed that WLGO treatment reduced
LPS-induced secretion of cytokines (IL-6, IL-β and TNF-α) in Caco-2
cells. Moreover, the MTT assay demonstrated that treatment with 200
µM WLGO for 48 h reduced the percentage of viable cells. A previous
study reported that oligosaccharides at various concentrations have
different effects, exhibiting adverse effects at high dosages
(46). For example, COS at ≥600
µM markedly decreases cell viability, whereas COS at low doses
shows a protective effect on intestinal porcine epithelial cells
(IPEC-J2 cells) (47). Acute and
chronic toxicology tests are required to verify the safe
concentration of WLGO. Therefore, the therapeutic WLGO dose in UC
should be carefully evaluated in clinical settings.
CYN is the most common drug to relieve chronic
enteritis in China (48). It is a
mixture of extracts from Euphorbia humifusa Willd, Herba hedyotids
chrysotrichae and Cinnamomum camphora (48). It has been shown that Euphorbia
humifusa Willd can significantly reduce the production of
inflammatory mediators nitric oxide and TNF-α in LPS-treated RAW
264.7 cells (49). Herba
hedyotids chrysotrichae has broad pharmacological effects as an
antioxidant, antibacterial and antitumor agent (50). It has been shown that Euphorbia
humifusa Willd can protect against DSS-induced experimental UC in
mice (51). Additionally, it has
been reported that a variety of natural ingredients in CYN, such as
flavones and organic acid, can protect epithelial cells (52). Thus, CYN was chosen as the
positive control in the present study.
The present data showed that both CYN and WLGO could
attenuate the symptoms of DSS-induced UC, and the therapeutic
effect of WLGO on UC was less potent than CYN. It has been
highlighted that patients with UC usually show complications, such
as nausea, vomiting and stomach pain, after receiving CYN treatment
(53). Thus, CYN cannot be used
for an extended period. Moreover, WLGO is an oligosaccharide
composed of four monosaccharide units (25), glucose, rhamnose, glucuronic acid
and mannose. These units are widely used in the food industry and
clinical settings (25),
indicating that WLGO is stable and does not result in adverse
reactions. Additionally, the present study aimed to identify
potential treatment strategies for UC through lowering inflammatory
cytokines and alleviating UC symptoms. The current results showed
that WLGO had similar beneficial effects to CYN on colon length
recovery, DAI score and anti-inflammatory activities (IL-1β, IL-6
and TNF-α) in mice with DSS-induced UC.
It has been pointed that the reduction of
inflammatory cytokines in the LPS-induced Caco-2 cells represents a
logical target for UC therapy (44). The inflammatory factors need to be
expressed before they can be secreted from the cells. It has been
shown that the accumulation of inflammatory factors induces cell
apoptosis and alters cell survival (54). Thus, changes in the levels of
inflammatory cytokines precede the changes in cell survival. Cell
viability was measured during the cell cycle at 24 and 48 h. The
results showed that WLGO had no cytotoxic effects on the Caco-2
cells after treatment for 24 h. For subsequent inflammatory factor
assays, time points within 24 h were chosen. WLGO treatment could
decrease the levels of cytokines in LPS-induced Caco-2 cells.
It has been suggested that lowering the levels of
inflammatory factors is a reasonable target for UC therapy, and the
intestinal inflammatory response is mediated by a complex network
of cytokines (proinflammatory cytokines, chemokines, growth factors
and adhesion molecules) released from epithelial cells within the
lamina propria (55). TNF-α is an
essential factor in the inflammatory cytokine network and is the
target of numerous novel biological therapies to attenuate the
symptoms of UC in patients (56).
It has become apparent that the progressive release of cytokines,
such as IL-6 and IL-1β, from T cells and macrophages also plays
critical roles in the development of UC (57). Therefore, proinflammatory
cytokines (TNF-α, IL-6 and IL-1β) could be inflammatory biomarkers
for UC. Reducing the production of these biomarkers is critical to
explore the efficacy of medicine to treat UC. In the present study,
WLGO inhibited the production of proinflammatory cytokines in
LPS-treated Caco-2 cells and the intestine of DSS-induced UC model
mice, suggesting that this effect was the primary response of the
anti-UC activity of WLGO. According to previous reports, rhamnose
and glucose are the monomer units of WLGO (23). It has been demonstrated that
rhamnose exhibits anti-inflammatory effects via inhibiting the
toll-like receptor 4 (TLR4)/NF-κB signaling pathway (58). We speculated that the TLR4/NF-κB
signaling pathway may be involved in the suppressive effect of WLGO
on the production of proinflammatory cytokines in UC. Further
studies are required to confirm this hypothesis.
In summary, WLG was degraded into WLGO using TFA,
and the effects of WLGO on LPS-induced Caco-2 cells and DSS-induced
UC mouse model were explored. The results showed that WLGO markedly
attenuated the inflammatory responses to DSS-induced UC, increased
mouse body weight and colon length, improved DAI score and
microscopic damages, and decreased mRNA expression levels of IL-1β,
IL-6 and TNF-α in mice. Moreover, WLGO treatment also decreased the
levels of cytokines in LPS-induced Caco-2 cells (Fig. 8). This study revealed that WLGO
may be an effective drug for the treatment of UC and laid a solid
foundation for future studies on the underlying mechanisms of WLGO
in the treatment of UC.
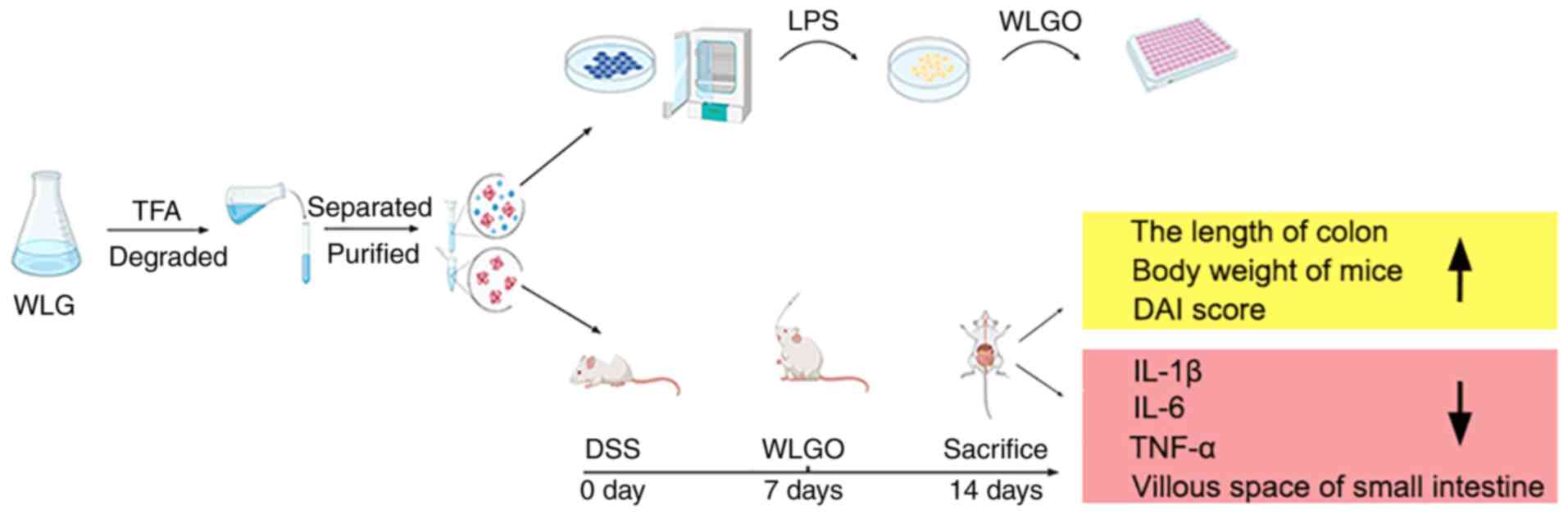 | Figure 8.Diagram of experimental procedures
and results. WLG was degraded by TFA into WLGO. The effects of WLGO
on UC were investigated in LPS-induced Caco-2 cells and DSS-induced
mouse UC model. WLG, weilan gum; WLGO, weilan gum oligosaccharide;
TFA, trifluoroacetic acid; UC, ulcerative colitis; LPS,
lipopolysaccharide; DSS, dextran sulfate sodium; DAI, disease
activity index; IL, interleukin; TNF-α, tumor necrosis factor α;
Nor, normal. |
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This work was supported by the Spring Industry Leader Talent
Support Plan (grant nos. 2017035 and 2019042), Shandong Taishan
Leading Talent Project (grant no. LJNY202015), Key R&D Program
of Shandong Province (grant nos. 2018YYSP022, 2019YYSP019 and
2019QYTPY024), Science, Education and Industry Integration and
Innovation Pilot Project of Qilu University of Technology, Shandong
Academy of Sciences (grant nos. 2020KJC-YJ01 and 2020KJC-GH10),
National Key Plan ‘Science and Technology to help the economy’
Special Project and University, Government, Industry, research
Collaborative Innovation Fund Project (grant no. 2020-CXY45) and
Yantai Development Zone Science and Technology Leading Talents
Project (grant no. 2020CXRC4).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
PZ designed the work, performed the experiments,
analyzed the data and wrote the manuscript. LS performed the
experiments and analyzed the data. FM performed the experiments. XJ
performed the experiments. YS analyzed the data. QY analyzed the
data. CZ acquired the data. SZ revised the manuscript and analyzed
the data. XS revised the manuscript and designed the study. LZ
supplied the funds, analyzed the data, drafted the manuscript,
revised the manuscript and provided final approval of the version
to be published and supplied the funds. LZ and LS confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
All animal experiments were performed in compliance
with the ARRIVE guidelines and in accordance with the UK Animals
(Scientific Procedures) Act (1986) and the associated guidelines,
the EU Directive 2010/63/EU for Animal experiments, the National
Institutes of Health Guide for the Care and Use of Laboratory
Animals (NIH Publications no. 8023, revised 1978) and the Animal
Management Rules of the Chinese Ministry of Health (no. 55, 2001).
This study was approved by the Animal Experiment Ethics Committee
of Qilu University of Technology (Jinan, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wehkamp J, Götz M, Herrlinger K, Steurer W
and Stange EF: Inflammatory bowel disease. Dtsch Arztebl Int.
113:72–82. 2016.PubMed/NCBI
|
|
2
|
Ananthakrishnan AN, Bernstein CN,
Iliopoulos D, Macpherson A, Neurath MF, Ali RAR, Vavricka SR and
Fiocchi C: Environmental triggers in IBD: A review of progress and
evidence. Nat Rev Gastroenterol Hepatol. 15:39–49. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cleynen I, Boucher G, Jostins L, Schumm
LP, Zeissig S, Ahmad T, Andersen V, Andrews JM, Annese V, Brand S,
et al: Inherited determinants of Crohn's disease and ulcerative
colitis phenotypes: A genetic association study. Lancet.
387:156–167. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nielsen OH and Munck LK: Drug insight:
Aminosalicylates for the treatment of IBD. Nat Clin Pract
Gastroenterol Hepatol. 4:160–170. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Darb Emamie A, Rajabpour M, Ghanavati R,
Asadolahi P, Farzi S, Sobouti B and Darbandi A: The effects of
probiotics, prebiotics and synbiotics on the reduction of IBD
complications, a periodic review during 2009-2020. J Appl
Microbiol. 130:1823–1838. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu J, Yang R, Gao M, Zhang H and Zhan X:
Synthesis of functional oligosaccharides and their derivatives
through cocultivation and cellular NTP regeneration. Adv Appl
Microbiol. 115:35–63. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang L, Liu YS, Wu YF and Fu QY: Effects
of chitosan oligosaccharide on alveolar bone resorption, Th17/Treg
balance and OPG/RANKL/RANK pathway in periodontitis rats. Shanghai
Kou Qiang Yi Xue. 30:237–242. 2021.(In Chinese). PubMed/NCBI
|
|
8
|
Liu SH, Chen RY and Chiang MT: Effects of
chitosan oligosaccharide on plasma and hepatic lipid metabolism and
liver histomorphology in normal sprague-dawley rats. Mar Drugs.
18:4082020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Huang Y and Wang T: Pectin
oligosaccharides enhance α2,6-sialylation modification that
promotes apoptosis of bladder cancer cells by targeting the
hedgehog pathway. Cell Biochem Biophys. 79:719–728. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yu X, Fu C, Cui Z, Chen G, Xu Y and Yang
C: Inulin and isomalto-oligosaccharide alleviate constipation and
improve reproductive performance by modulating motility-related
hormones, short-chain fatty acids, and feces microflora in pregnant
sows. J Anim Sci. 99:skab2572021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rastall RA: Functional oligosaccharides:
Application and manufacture. Annu Rev Food Sci Technol. 1:305–339.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Babbar N, Dejonghe W, Gatti M, Sforza S
and Elst K: Pectic oligosaccharides from agricultural by-products:
Production, characterization and health benefits. Crit Rev
Biotechnol. 36:594–606. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhao M, Zhang H, Xu X, Li S and Xu H: A
strategy for the synthesis of low-molecular-weight welan gum by
eliminating capsule form of Sphingomonas strains. Int J Biol
Macromol. 178:11–18. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ke C, Wei L, Wang M, Li Q, Liu X, Guo Y
and Li S: Effect of NaCl addition on the production of welan gum
with the UV mutant of Sphingomonas sp. Carbohydr Polym.
265:1181102021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li H, Xu H, Xu H, Li S, Ying HJ and Ouyang
PK: Enhanced welan gum production using a two-stage agitation speed
control strategy in Alcaligenes sp. CGMCC2428. Bioprocess
Biosyst Eng. 34:95–102. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Martin-Piñero MJ, García MC, Muñoz J and
Alfaro-Rodriguez MC: Influence of the welan gum biopolymer
concentration on the rheological properties, droplet size
distribution and physical stability of thyme oil/W emulsions. Int J
Biol Macromol. 133:270–277. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Berninger T, Dietz N and González López Ó:
Water-soluble polymers in agriculture: Xanthan gum as eco-friendly
alternative to synthetics. Microb Biotechnol. 14:1881–1896. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhu H, Sun SW, Li H, Chang A, Liu YC, Qian
J and Shen YL: Significantly improved production of Welan gum by
Sphingomonas sp. WG through a novel quorum-sensing-interfering
dipeptide cyclo(L-Pro-L-Phe). Int J Biol Macromol. 126:118–122.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kaur V, Bera MB, Panesar PS, Kumar H and
Kennedy JF: Welan gum: Microbial production, characterization, and
applications. Int J Biol Macromol. 65:454–461. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Martis BS, Droux M, Deboudard F, Nasser W,
Meyer S and Reverchon S: Separation and quantification of
2-keto-3-deoxy-gluconate (KDG) a major metabolite in pectin and
alginate degradation pathways. Anal Biochem. 619:1140612021.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hagiwara A, Imai N, Doi Y, Sano M, Tamano
S, Omoto T, Asai I, Yasuhara K and Hayashi SM: Ninety-day oral
toxicity study of rhamsan gum, a natural food thickener produced
from Sphingomonas ATCC 31961, in Crl:CD(SD)IGS rats. J Toxicol Sci.
35:493–501. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li Q, Zhou Y, Ke C, Bai Y, Liu X and Li S:
Production of welan gum from cane molasses by Sphingomonas sp.
FM01. Carbohydr Polym. 244:1164852020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhu P, Zhan Y, Wang C, Liu X, Liu L and Xu
H: Efficient biosynthesis of polysaccharide welan gum in heat shock
protein-overproducing Sphingomonas sp. via temperature-dependent
strategy. Bioprocess Biosyst Eng. 44:247–257. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gupta L, Khandelwal D and Kalra S: Applied
carbohydrate counting. J Pak Med Assoc. 67:1456–1457.
2017.PubMed/NCBI
|
|
25
|
Slámová K, Kapešová J and Valentová K:
‘Sweet flavonoids’: Glycosidase-catalyzed modifications. Int J Mol
Sci. 19:21262018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Martinez-Pomares L: The mannose receptor.
J Leukoc Biol. 92:1177–1186. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jang J, Kim SM, Yee SM, Kim EM, Lee EH,
Choi HR, Lee YS, Yang WK, Kim HY, Kim KH, et al: Daucosterol
suppresses dextran sulfate sodium (DSS)-induced colitis in mice.
Int Immunopharmacol. 72:124–130. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cao H, Liu J, Shen P, Cai J, Han Y, Zhu K,
Fu Y, Zhang N, Zhang Z and Cao Y: Protective effect of naringin on
DSS-induced ulcerative colitis in mice. J Agric Food Chem.
66:13133–13140. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Morton DB: The animals (scientific
procedures) Act 1986 and research into anaesthesia. Br J Anaesth.
65:303–305. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
National Research Council (US) Committee
for the Update of the Guide for the Care and Use of Laboratory
Animals: Guide for the Care and Use of Laboratory Animals. 8th
edition. National Academies Press (US); Washington, DC: 2011
|
|
31
|
Pacheco MT, Vezza T, Diez-Echave P,
Utrilla P, Villamiel M and Moreno FJ: Anti-inflammatory bowel
effect of industrial orange by-products in DSS-treated mice. Food
Funct. 9:4888–4896. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen Y, Zhang M and Ren F: A role of
exopolysaccharide produced by streptococcus thermophilus in the
intestinal inflammation and mucosal barrier in Caco-2 monolayer and
dextran sulphate sodium-induced experimental murine colitis.
Molecules. 24:5132019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ling X, Linglong P, Weixia D and Hong W:
Protective effects of bifidobacterium on intestinal barrier
function in LPS-induced enterocyte barrier injury of Caco-2
monolayers and in a rat NEC model. PLoS One. 11:e01616352016.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Konstantinou GN: Enzyme-linked
immunosorbent assay (ELISA). Methods Mol Biol. 1592:79–94. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhu R, Wang C, Zhang L, Wang Y, Chen G,
Fan J, Jia Y, Yan F and Ning C: Pectin oligosaccharides from fruit
of Actinidia arguta: Structure-activity relationship of prebiotic
and antiglycation potentials. Carbohydr Polym. 217:90–97. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chumpitazi BP: The gut microbiome as a
predictor of low fermentable oligosaccharides disaccharides
monosaccharides and polyols diet efficacy in functional bowel
disorders. Curr Opin Gastroenterol. 36:147–154. 2020.PubMed/NCBI
|
|
38
|
Krasiński R and Tchórzewski H:
Hyaluronan-mediated regulation of inflammation. Postepy Hig Med
Dosw (Online). 61:683–689. 2007.(In Polish). PubMed/NCBI
|
|
39
|
el-Nezhawy AO, Adly FG, Eweas AF, Hanna
AG, el-Kholy YM, el-Syed SH and el-Naggar TB: Design, synthesis and
antitumor activity of novel D-glucuronic acid derivatives. Med
Chem. 7:624–638. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Dinda B, Dinda S, DasSharma S, Banik R,
Chakraborty A and Dinda M: Therapeutic potentials of baicalin and
its aglycone, baicalein against inflammatory disorders. Eur J Med
Chem. 131:68–80. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Cheng PW, Davidson S and Bhat G: Markers
of malignant prostate cancer cells: Golgi localization of
α-mannosidase 1A at GM130-GRASP65 site and appearance of high
mannose N-glycans on cell surface. Biochem Biophys Res Commun.
527:406–410. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wei YY, Fan YM, Ga Y, Zhang YN, Han JC and
Hao ZH: Shaoyao decoction attenuates DSS-induced ulcerative
colitis, macrophage and NLRP3 inflammasome activation through the
MKP1/NF-κB pathway. Phytomedicine. 92:1537432021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Guazelli CFS, Fattori V, Ferraz CR, Borghi
SM, Casagrande R, Baracat MM and Verri WA Jr: Antioxidant and
anti-inflammatory effects of hesperidin methyl chalcone in
experimental ulcerative colitis. Chem Biol Interact.
333:1093152021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Peng Y, Yan Y, Wan P, Chen D, Ding Y, Ran
L, Mi J, Lu L, Zhang Z, Li X, et al: Gut microbiota modulation and
anti-inflammatory properties of anthocyanins from the fruits of
Lycium ruthenicum Murray in dextran sodium sulfate-induced colitis
in mice. Free Radic Biol Med. 136:96–108. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lamb CA, Kennedy NA, Raine T, Hendy PA,
Smith PJ, Limdi JK, Hayee B, Lomer MCE, Parkes GC, Selinger C, et
al: British society of gastroenterology consensus guidelines on the
management of inflammatory bowel disease in adults. Gut. 68 (Suppl
3):s1–s106. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Casellas F, Borruel N, Torrejón A, Varela
E, Antolin M, Guarner F and Malagelada JR: Oral
oligofructose-enriched inulin supplementation in acute ulcerative
colitis is well tolerated and associated with lowered faecal
calprotectin. Aliment Pharmacol Ther. 25:1061–1067. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Fang T, Yao Y, Tian G, Chen D, Wu A, He J,
Zheng P, Mao X, Yu J, Luo Y, et al: Chitosan oligosaccharide
attenuates endoplasmic reticulum stress-associated intestinal
apoptosis via the Akt/mTOR pathway. Food Funct. 12:8647–8658. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yang X, Yang SP, Zhang X, Yu XD, He QY and
Wang BC: Study on the multi-marker components quantitative HPLC
fingerprint of the compound Chinese medicine Wuwei changyanning
granule. Iran J Pharm Res. 13:1191–1201. 2014.PubMed/NCBI
|
|
49
|
Luyen BT, Tai BH, Thao NP, Eun KJ, Cha JY,
Xin MJ, Lee YM and Kim YH: Anti-inflammatory components of
euphorbia humifusa Willd. Bioorg Med Chem Lett. 24:1895–1900. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wang LL, Fu H, Li WW, Song FJ, Song YX, Yu
Q, Liu GX and Wang XM: Study of effect of humifuse euphorbia herb
on alleviating insulin resistance in type 2 diabetic model KK-Ay
mice. Zhongguo Zhong Yao Za Zhi. 40:1994–1998. 2015.(In Chinese).
PubMed/NCBI
|
|
51
|
Sann H, Erichsen JV, Hessmann M, Pahl A
and Hoffmeyer A: Efficacy of drugs used in the treatment of IBD and
combinations thereof in acute DSS-induced colitis in mice. Life
Sci. 92:708–718. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Shin SY, Kim CG, Jung YJ, Jung Y, Jung H,
Im J, Lim Y and Lee YH: Euphorbia humifusa Willd exerts inhibition
of breast cancer cell invasion and metastasis through inhibition of
TNFα-induced MMP-9 expression. BMC Complement Altern Med.
16:4132016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Cao SY, Ye SJ, Wang WW, Wang B, Zhang T
and Pu YQ: Progress in active compounds effective on ulcerative
colitis from Chinese medicines. Chin J Nat Med. 17:81–102.
2019.PubMed/NCBI
|
|
54
|
Zhuang YT, Xu DY, Wang GY, Sun JL, Huang Y
and Wang SZ: IL-6 induced lncRNA MALAT1 enhances TNF-α expression
in LPS-induced septic cardiomyocytes via activation of SAA3. Eur
Rev Med Pharmacol Sci. 21:302–309. 2017.PubMed/NCBI
|
|
55
|
Baumgart DC and Carding SR: Inflammatory
bowel disease: Cause and immunobiology. Lancet. 369:1627–1640.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Atreya R and Neurath MF: Chemokines in
inflammatory bowel diseases. Dig Dis. 28:386–394. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zhang H, Kovacs-Nolan J, Kodera T, Eto Y
and Mine Y: γ-Glutamyl cysteine and γ-glutamyl valine inhibit TNF-α
signaling in intestinal epithelial cells and reduce inflammation in
a mouse model of colitis via allosteric activation of the
calcium-sensing receptor. Biochim Biophys Acta. 1852:792–804. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Singh DP, Khare P, Zhu J, Kondepudi KK,
Singh J, Baboota RK, Boparai RK, Khardori R, Chopra K and Bishnoi
M: A novel cobiotic-based preventive approach against high-fat
diet-induced adiposity, nonalcoholic fatty liver and gut
derangement in mice. Int J Obes (Lond). 40:487–496. 2016.
View Article : Google Scholar : PubMed/NCBI
|