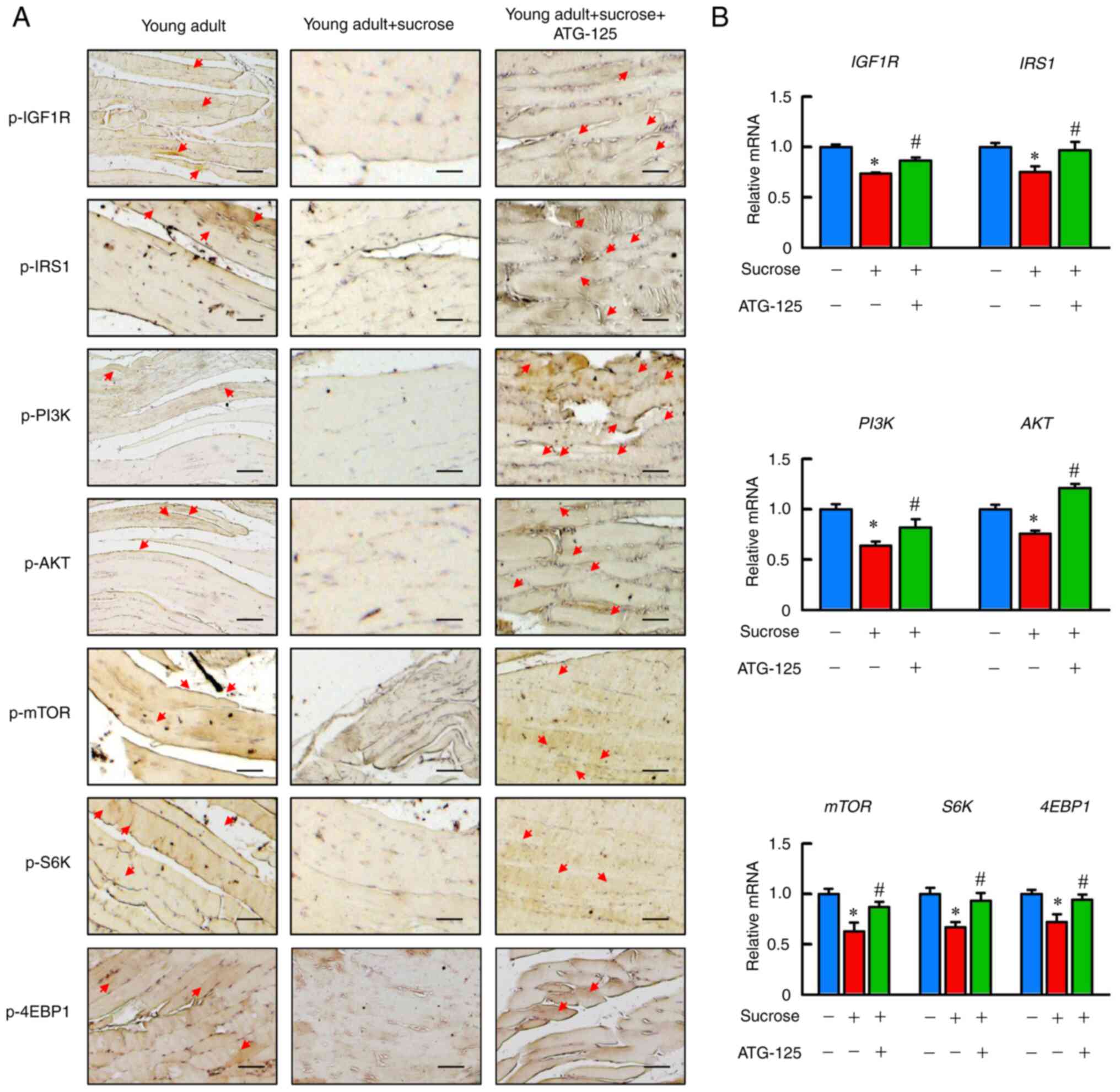
|
1
|
Lynch GS, Schertzer JD and Ryall JG:
Therapeutic approaches for muscle wasting disorders. Pharmacol
Ther. 113:461–487. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bonaldo P and Sandri M: Cellular and
molecular mechanisms of muscle atrophy. Dis Model Mech. 6:25–39.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ferrucci L and Guralnik JM: Inflammation,
hormones, and body composition at a crossroad. Am J Med.
115:501–502. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pedersen BK and Bruunsgaard H: Possible
beneficial role of exercise in modulating low-grade inflammation in
the elderly. Scand J Med Sci Sports. 13:56–62. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Franceschi C, Bonafè M, Valensin S,
Olivieri F, De Luca M, Ottaviani E and De Benedictis G:
Inflamm-aging. An evolutionary perspective on immunosenescence. Ann
N Y Acad Sci. 908:244–254. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ali S and Garcia JM: Sarcopenia, cachexia
and aging: Diagnosis, mechanisms and therapeutic options-a
mini-review. Gerontology. 60:294–305. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Costamagna D, Costelli P, Sampaolesi M and
Penna F: Role of Inflammation in muscle homeostasis and myogenesis.
Mediators Inflamm. 2015:8051722015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Milan G, Romanello V, Pescatore F, Armani
A, Paik JH, Frasson L, Seydel A, Zhao J, Abraham R, Goldberg AL, et
al: Regulation of autophagy and the ubiquitin-proteasome system by
the FoxO transcriptional network during muscle atrophy. Nat Commun.
6:66702015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gregor MF and Hotamisligil GS:
Inflammatory mechanisms in obesity. Annu Rev Immunol. 29:415–445.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Glass CK and Olefsky JM: Inflammation and
lipid signaling in the etiology of insulin resistance. Cell Metab.
15:635–645. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Olefsky JM and Glass CK: Macrophages,
inflammation, and insulin resistance. Annu Rev Physiol. 72:219–246.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Perry BD, Caldow MK, Brennan-Speranza TC,
Sbaraglia M, Jerums G, Garnham A, Wong C, Levinger P, Asrar Ul Haq
M, Hare DL, et al: Muscle atrophy in patients with Type 2 Diabetes
Mellitus: Roles of inflammatory pathways, physical activity and
exercise. Exerc Immunol Rev. 22:94–109. 2016.PubMed/NCBI
|
|
13
|
Maliszewska K, Adamska-Patruno E and
Krętowski A: The interplay between muscle mass decline, obesity,
and type 2 diabetes. Pol Arch Intern Med. 129:809–816.
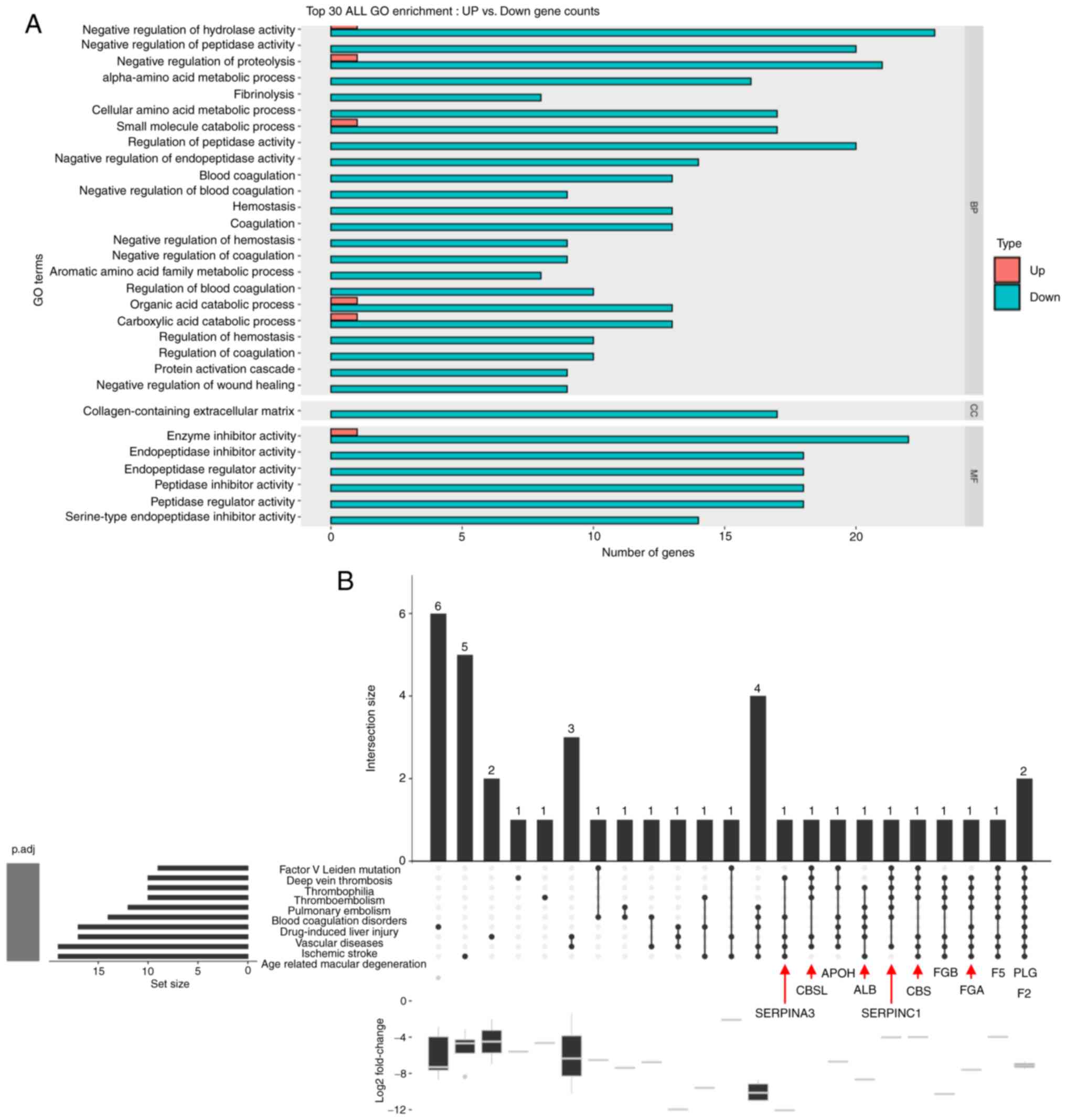
2019.PubMed/NCBI
|
|
14
|
Monier S, Le Cam A and Le Marchand-Brustel
Y: Insulin and insulin-like growth factor I. Effects on protein
synthesis in isolated muscles from lean and goldthioglucose-obese
mice. Diabetes. 32:392–397. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rommel C, Bodine SC, Clarke BA, Rossman R,
Nunez L, Stitt TN, Yancopoulos GD and Glass DJ: Mediation of
IGF-1-induced skeletal myotube hypertrophy by PI(3)K/Akt/mTOR and
PI(3)K/Akt/GSK3 pathways. Nat Cell Biol. 3:1009–1013. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sacheck JM, Ohtsuka A, McLary SC and
Goldberg AL: IGF-I stimulates muscle growth by suppressing protein
breakdown and expression of atrophy-related ubiquitin ligases,
atrogin-1 and MuRF1. Am J Physiol Endocrinol Metab. 287:E591–E601.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
de Alvaro C, Teruel T, Hernandez R and
Lorenzo M: Tumor necrosis factor alpha produces insulin resistance
in skeletal muscle by activation of inhibitor kappaB kinase in a
p38 MAPK-dependent manner. J Biol Chem. 279:17070–17078. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dogra C, Changotra H, Wedhas N, Qin X,
Wergedal JE and Kumar A: TNF-related weak inducer of apoptosis
(TWEAK) is a potent skeletal muscle-wasting cytokine. FASEB J.
21:1857–1869. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hirosumi J, Tuncman G, Chang L, Görgün CZ,
Uysal KT, Maeda K, Karin M and Hotamisligil GS: A central role for
JNK in obesity and insulin resistance. Nature. 420:333–336. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Peterson JM, Bakkar N and Guttridge DC:
NF-κB signaling in skeletal muscle health and disease. Curr Top Dev
Biol. 96:85–119. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Törrönen R, Kolehmainen M, Sarkkinen E,
Mykkänen H and Niskanen L: Postprandial glucose, insulin, and free
fatty acid responses to sucrose consumed with blackcurrants and
lingonberries in healthy women. Am J Clin Nutr. 96:527–533. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Abdullah A, Peeters A, de Courten M and
Stoelwinder J: The magnitude of association between overweight and
obesity and the risk of diabetes: A meta-analysis of prospective
cohort studies. Diabetes Res Clin Pract. 89:309–319. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Boden G: Obesity and free fatty acids.
Endocrinol Metab Clin North Am. 37:635–646, viii-ix. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
K S, Senthilkumar GP, Sankar P and Bobby
Z: Attenuation of oxidative stress, inflammation and insulin
resistance by allium sativum in fructose-fed male rats. J Clin
Diagn Res. 7:1860–1862. 2013.PubMed/NCBI
|
|
25
|
Hwang IS, Ho H, Hoffman BB and Reaven GM:
Fructose-induced insulin resistance and hypertension in rats.
Hypertension. 10:512–516. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gatineau E, Savary-Auzeloux I, Migné C,
Polakof S, Dardevet D and Mosoni L: Chronic intake of sucrose
accelerates sarcopenia in older male rats through alterations in
insulin sensitivity and muscle protein synthesis. J Nutr.
145:923–930. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Shin MS, Lee J, Lee JW, Park SH, Lee IK,
Choi JA, Lee JS and Kang KS: Anti-Inflammatory effect of
Artemisia argyi on Ethanol-Induced gastric ulcer: Analytical
in vitro and in vivo studies for the identification of action
mechanism and active compounds. Plants (Basel). 10:3322021.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gao XH, Zhang SD, Wang LT, Yu L, Zhao XL,
Ni HY, Wang YQ, Wang JD, Shan CH and Fu YJ: Anti-Inflammatory
effects of neochlorogenic acid extract from mulberry leaf (Morus
alba L.) against LPS-Stimulated inflammatory response through
mediating the AMPK/Nrf2 signaling pathway in A549 Cells. Molecules.
25:13852020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hu PF, Sun FF and Qian J: Leonurine exerts
Anti-Catabolic and Anti-apoptotic effects via nuclear factor kappa
B (NF-κB) and mitogen-activated protein kinase (MAPK) signaling
pathways in chondrocytes. Med Sci Monit. 25:6271–6280. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cho SY, Kim HW, Lee MK, Kim HJ, Kim JB,
Choe JS, Lee YM and Jang HH: Antioxidant and Anti-Inflammatory
activities in relation to the flavonoids composition of pepper
(Capsicum annuum L.). Antioxidants (Basel). 9:9862020.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hernández-Ortega M, Ortiz-Moreno A,
Hernández-Navarro MD, Chamorro-Cevallos G, Dorantes-Alvarez L and
Necoechea-Mondragón H: Antioxidant, antinociceptive, and
anti-inflammatory effects of carotenoids extracted from dried
pepper (Capsicum annuum L.). J Biomed Biotechnol.
2012:5240192012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lai KH, Chen PJ, Chen CC, Yang SH,
El-Shazly M, Chang YC, Wu YH, Wu YH, Wang YH, Hsieh HL and Hwang
TL: Lophatherum gracile Brongn. attenuates neutrophilic
inflammation through inhibition of JNK and calcium. J
Ethnopharmacol. 264:1132242021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ti H, Mai Z, Wang Z, Zhang W, Xiao M, Yang
Z and Shaw P: Bisabolane-type sesquiterpenoids from Curcuma
longa L. exert anti-influenza and anti-inflammatory activities
through NF-κB/MAPK and RIG-1/STAT1/2 signaling pathways. Food
Funct. 12:6697–6711. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Memarzia A, Khazdair MR, Behrouz S,
Gholamnezhad Z, Jafarnezhad M, Saadat S and Boskabady MH:
Experimental and clinical reports on anti-inflammatory,
antioxidant, and immunomodulatory effects of Curcuma longa
and curcumin, an updated and comprehensive review. Biofactors.
47:311–350. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yue SJ, Qin YF, Kang A, Tao HJ, Zhou GS,
Chen YY, Jiang JQ, Tang YP and Duan JA: Total Flavonoids of
Glycyrrhiza uralensis Alleviates irinotecan-induced colitis
via modification of gut microbiota and fecal metabolism. Front
Immunol. 12:6283582021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li M, Chai X, Wang L, Yang J and Wang Y:
Study of the variation of phenolic acid and flavonoid content from
fresh artemisiae argyi folium to moxa wool. Molecules. 24:46032019.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhao L, Wang D, Liu J, Yu X, Wang R, Wei
Y, Wen C and Ouyang Z: Transcriptomic analysis of key genes
involved in chlorogenic acid biosynthetic pathway and
characterization of MaHCT from Morus alba L. Protein Expr
Purif. 156:25–35. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chang Y, Zhang D, Yang G, Zheng Y and Guo
L: Screening of Anti-Lipase components of Artemisia argyi
leaves based on Spectrum-Effect relationships and HPLC-MS/MS. Front
Pharmacol. 12:6753962021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wen YQ, Gong LY, Wang L, Zhao N, Sun Q,
Kamara MO, Ma HY and Meng FH: Comparative pharmacokinetics study of
leonurine and stachydrine in normal rats and rats with
Cold-Stagnation and Blood-Stasis primary dysmenorrhoea after the
administration of Leonurus japonicus houtt electuary. J Sep
Sci. 42:1725–1732. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Dan-Dan L, Hua-Sheng P, Yuan Z, Xian-Zhang
H, Zhi-Lai Z, Da-Hui L, Li-Ping K and Lu-Qi H: Comparison of
chemical components between Artemisia stolonifera and Artemisia
argyi using UPLC-Q-TOF-MS. Zhongguo Zhong Yao Za Zhi.
45:4057–4064. 2020.(In Chinese). PubMed/NCBI
|
|
41
|
Wang ZL, Gao HM, Wang S, Zhang M, Chen K,
Zhang YQ, Wang HD, Han BY, Xu LL, Song TQ, et al: Dissection of the
general two-step di-C-glycosylation pathway for the biosynthesis of
(iso)schaftosides in higher plants. Proc Natl Acad Sci USA.
117:30816–30823. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Suntornsuk L, Kasemsook S and Wongyai S:
Quantitative analysis of aglycone quercetin in mulberry leaves
(Morus alba L.) by capillary zone electrophoresis.
Electrophoresis. 24:1236–1241. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kim GD, Lee YS, Cho JY, Lee YH, Choi KJ,
Lee Y, Han TH, Lee SH, Park KH and Moon JH: Comparison of the
content of bioactive substances and the inhibitory effects against
rat plasma oxidation of conventional and organic hot peppers
(Capsicum annuum L.). J Agric Food Chem. 58:12300–12306.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li S, Zhou S, Yang W and Meng D:
Gastro-protective effect of edible plant Artemisia argyi in
ethanol-induced rats via normalizing inflammatory responses and
oxidative stress. J Ethnopharmacol. 214:207–217. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chuang TY, Lien CY, Tsai YC, Lin KF, Hsu
CH, Wu WJ, Su LY, Lu CW and Wu CH: Oral treatment with the Chinese
herbal supplements B307 enhances muscle endurance of ICR mice after
exhaustive swimming via suppressing fatigue, oxidative stress, and
inflammation. Food Sci Nutr. 8:3682–3691. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Lien CY, Lu CW, Lin YH, Wu WJ, Hsu CH,
Chuang TY, Lin KF, Chuang WC, Lee MC and Wu CH: Chinese herbal
medicine, guilu erxian glue, as alternative medicine for adverse
side effects of chemotherapy in Doxorubicin-Treated cell and mouse
models. Evid Based Complement Alternat Med. 2021:55489682021.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kato T, Iizuka K, Takao K, Horikawa Y,
Kitamura T and Takeda J: ChREBP-Knockout mice show sucrose
intolerance and fructose malabsorption. Nutrients. 10:3402018.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Burke SJ, Batdorf HM, Martin TM, Burk DH,
Noland RC, Cooley CR, Karlstad MD, Johnson WD and Collier JJ:
Liquid sucrose consumption promotes obesity and impairs glucose
tolerance without altering circulating insulin levels. Obesity
(Silver Spring). 26:1188–1196. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen YS, Liu HM and Lee TY:
Ursodeoxycholic acid regulates hepatic energy homeostasis and white
adipose tissue macrophages polarization in Leptin-Deficiency obese
mice. Cells. 8:2532019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Moylan JS, Smith JD, Chambers MA,
McLoughlin TJ and Reid MB: TNF induction of atrogin-1/MAFbx mRNA
depends on Foxo4 expression but not AKT-Foxo1/3 signaling. Am J
Physiol Cell Physiol. 295:C986–C993. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Qian Y, Guo X, Che L, Guan X, Wu B, Lu R,
Zhu M, Pang H, Yan Y, Ni Z and Gu L: Klotho reduces necroptosis by
targeting oxidative stress involved in renal Ischemic-Reperfusion
injury. Cell Physiol Biochem. 45:2268–2282. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Omede F, Zhang S, Johnson C, Daniel E,
Zhang Y, Fields TA, Boulanger J, Liu S, Ahmed I, Umar S, et al:
Dietary phosphate restriction attenuates polycystic kidney disease
in mice. Am J Physiol Renal Physiol. 318:F35–F42. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Murdoch CE, Alom-Ruiz SP, Wang M, Zhang M,
Walker S, Yu B, Brewer A and Shah AM: Role of endothelial Nox2
NADPH oxidase in angiotensin II-induced hypertension and vasomotor
dysfunction. Basic Res Cardiol. 106:527–538. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Sag CM, Schnelle M, Zhang J, Murdoch CE,
Kossmann S, Protti A, Santos CXC, Sawyer G, Zhang X, Mongue-Din H,
et al: Distinct regulatory effects of myeloid cell and endothelial
cell NAPDH oxidase 2 on blood pressure. Circulation. 135:2163–2177.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Yates Z, Tarling EJ, Langley-Evans SC and
Salter AM: Maternal undernutrition programmes atherosclerosis in
the ApoE*3-Leiden mouse. Br J Nutr. 101:1185–1194. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wang CH, Liu HM, Chang ZY, Huang TH and
Lee TY: Losartan prevents hepatic steatosis and macrophage
polarization by inhibiting HIF-1α in a murine model of NAFLD. Int J
Mol Sci. 22:78412021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Huang Y, Wang J, Qua G, Wang X, Yang L and
Zhong L: Lactobacillus acidophilus ATCC 4356 prevents
atherosclerosis via inhibition of intestinal cholesterol absorption
in apolipoprotein E-knockout mice. Appl Environ Microbiol.
80:7496–7504. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Jin YN, Hwang WY, Jo C and Johnson GV:
Metabolic state determines sensitivity to cellular stress in
Huntington disease: Normalization by activation of PPARγ. PLoS One.
7:e304062012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Shimano H, Yahagi N, Amemiya-Kudo M, Hasty
AH, Osuga J, Tamura Y, Shionoiri F, Iizuka Y, Ohashi K, Harada K,
et al: Sterol regulatory element-binding protein-1 as a key
transcription factor for nutritional induction of lipogenic enzyme
genes. J Biol Chem. 274:35832–35839. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Sung YY, Yoon T, Jang S and Kim HK:
Forsythia suspensa suppresses house dust mite
Extract-Induced atopic dermatitis in NC/Nga Mice. PLoS One.
11:e01676872016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Saba E, Lee YS, Yang WK, Lee YY, Kim M,
Woo SM, Kim K, Kwon YS, Kim TH, Kwak D, et al: Effects of a herbal
formulation, KGC3P, and its individual component, nepetin, on coal
fly dust-induced airway inflammation. Sci Rep. 10:140362020.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kagari T, Doi H and Shimozato T: The
importance of IL-1 beta and TNF-alpha, and the noninvolvement of
IL-6, in the development of monoclonal antibody-induced arthritis.
J Immunol. 169:1459–1466. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Choi BH, Ahn IS, Kim YH, Park JW, Lee SY,
Hyun CK and Do MS: Berberine reduces the expression of adipogenic
enzymes and inflammatory molecules of 3T3-L1 adipocyte. Exp Mol
Med. 38:599–605. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Yang H, Liao D, Tong L, Zhong L and Wu K:
MiR-373 exacerbates renal injury and fibrosis via
NF-κB/MatrixMetalloproteinase-9 signaling by targeting Sirtuin1.
Genomics. 111:786–792. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Shin H, Ma Y, Chanturiya T, Cao Q, Wang Y,
Kadegowda AKG, Jackson R, Rumore D, Xue B, Shi H, et al: Lipolysis
in brown adipocytes is not essential for Cold-Induced thermogenesis
in mice. Cell Metab. 26:764–777.e5. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Thornton SM, Krolopp JE and Abbott MJ:
IL-15 mediates mitochondrial activity through a PPAR
δ-Dependent-PPAR α-Independent mechanism in skeletal muscle cells.
PPAR Res. 2016:54658042016. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Luan G, Li G, Ma X, Jin Y, Hu N, Li J,
Wang Z and Wang H: Dexamethasone-Induced mitochondrial dysfunction
and insulin Resistance-Study in 3T3-L1 adipocytes and mitochondria
isolated from mouse liver. Molecules. 24:19822019. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Dusaulcy R, Rancoule C, Grès S, Wanecq E,
Colom A, Guigné C, van Meeteren LA, Moolenaar WH, Valet P and
Saulnier-Blache JS: Adipose-specific disruption of autotaxin
enhances nutritional fattening and reduces plasma lysophosphatidic
acid. J Lipid Res. 52:1247–1255. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Bernardi S, Toffoli B, Tisato V, Bossi F,
Biffi S, Lorenzon A, Zauli G, Secchiero P and Fabris B: TRAIL
reduces impaired glucose tolerance and NAFLD in the high-fat diet
fed mouse. Clin Sci (Lond). 132:69–83. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Bermúdez-Muñoz JM, Celaya AM,
Hijazo-Pechero S, Wang J, Serrano M and Varela-Nieto I: G6PD
overexpression protects from oxidative stress and age-related
hearing loss. Aging Cell. 19:e132752020. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Wang Y, Sakata T, Elalieh HZ, Munson SJ,
Burghardt A, Majumdar S, Halloran BP and Bikle DD: Gender
differences in the response of CD-1 mouse bone to parathyroid
hormone: Potential role of IGF-I. J Endocrinol. 189:279–287. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Al-Dwairi A, Brown AR, Pabona JM, Van TH,
Hamdan H, Mercado CP, Quick CM, Wight PA, Simmen RC and Simmen FA:
Enhanced gastrointestinal expression of cytosolic malic enzyme
(ME1) induces intestinal and liver lipogenic gene expression and
intestinal cell proliferation in mice. PLoS One. 9:e1130582014.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhao L, Li C, Liu F, Zhao Y, Liu J, Hua Y,
Liu J, Huang J and Ge C: A blockade of PD-L1 produced antitumor and
antimetastatic effects in an orthotopic mouse pancreatic cancer
model via the PI3K/Akt/mTOR signaling pathway. Onco Targets Ther.
10:2115–2126. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Deng L, Huang L, Sun Y, Heath JM, Wu H and
Chen Y: Inhibition of FOXO1/3 promotes vascular calcification.
Arterioscler Thromb Vasc Biol. 35:175–183. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Choi DH, Yang J and Kim YS: Rapamycin
suppresses postnatal muscle hypertrophy induced by
myostatin-inhibition accompanied by transcriptional suppression of
the Akt/mTOR pathway. Biochem Biophys Rep. 17:182–190.
2019.PubMed/NCBI
|
|
77
|
Roselló-Díez A, Madisen L, Bastide S, Zeng
H and Joyner AL: Cell-nonautonomous local and systemic responses to
cell arrest enable long-bone catch-up growth in developing mice.
PLoS Biol. 16:e20050862018. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Prieto C and Barrios D: RaNA-Seq:
Interactive RNA-Seq analysis from FASTQ files to functional
analysis. Bioinformatics. Nov 15–2019.(Epub ahead of print).
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Bolger AM, Lohse M and Usadel B:
Trimmomatic: A flexible trimmer for Illumina sequence data.
Bioinformatics. 30:2114–2120. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Sahraeian SME, Mohiyuddin M, Sebra R,
Tilgner H, Afshar PT, Au KF, Bani Asadi N, Gerstein MB, Wong WH,
Snyder MP, et al: Gaining comprehensive biological insight into the
transcriptome by performing a broad-spectrum RNA-seq analysis. Nat
Commun. 8:592017. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wang L, Wang S and Li W: RSeQC: Quality
control of RNA-seq experiments. Bioinformatics. 28:2184–2185. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Okonechnikov K, Conesa A and
García-Alcalde F: Qualimap 2: Advanced multi-sample quality control
for high-throughput sequencing data. Bioinformatics. 32:292–294.
2046.PubMed/NCBI
|
|
83
|
Liao Y, Smyth GK and Shi W: FeatureCounts:
An efficient general purpose program for assigning sequence reads
to genomic features. Bioinformatics. 30:923–930. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Varet H, Brillet-Guéguen L, Coppée JY and
Dillies MA: SARTools: A DESeq2- and EdgeR-Based R pipeline for
comprehensive differential analysis of RNA-Seq data. PLoS One.
11:e01570222016. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Holm L and Sander C: Removing
near-neighbour redundancy from large protein sequence collections.
Bioinformatics. 14:423–429. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Benjamini Y and Hochberg Y: Controlling
the false discovery rate: A practical and powerful approach to
multiple testing. J R Stat Soc Ser C Appl Stat. 57:289–300.
1995.
|
|
87
|
Liu S, Cai Y, Changyong E, Sheng J and
Zhang X: Screening and validation of independent predictors of poor
survival in pancreatic cancer. Pathol Oncol Res. 27:16098682021.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Gene Ontology Consortium: The Gene
Ontology project in 2008. Nucleic Acids Res. 36:D440–D444. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Zhu HM, Fei Q, Qian LX, Liu BL, He X and
Yin L: Identification of key pathways and genes in nasopharyngeal
carcinoma using bioinformatics analysis. Oncol Lett. 17:4683–4694.
2019.PubMed/NCBI
|
|
90
|
Deng YJ, Ren FH, Yuan WH, Zhang GZ, Wu ZL
and Xie QQ: GRB10 and E2F3 as diagnostic markers of osteoarthritis
and their correlation with immune infiltration. Diagnostics
(Basel). 10:1712020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Wu T, Hu E, Xu S, Chen M, Guo P, Dai J,
Feng T, Zhou L, Tang W, Zhan L, et al: clusterProfiler 4.0: A
universal enrichment tool for interpreting omics data. Innovation
(N Y). 2:1001412021.PubMed/NCBI
|
|
92
|
Bagul PK, Middela H, Matapally S, Padiya
R, Bastia T, Madhusudana K, Reddy BR, Chakravarty S and Banerjee
SK: Attenuation of insulin resistance, metabolic syndrome and
hepatic oxidative stress by resveratrol in fructose-fed rats.
Pharmacol Res. 66:260–268. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Phuwamongkolwiwat P, Suzuki T, Hira T and
Hara H: Fructooligosaccharide augments benefits of
quercetin-3-O-β-glucoside on insulin sensitivity and plasma total
cholesterol with promotion of flavonoid absorption in sucrose-fed
rats. Eur J Nutr. 53:457–468. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Marzani B, Balage M, Vénien A, Astruc T,
Papet I, Dardevet D and Mosoni L: Antioxidant supplementation
restores defective leucine stimulation of protein synthesis in
skeletal muscle from old rats. J Nutr. 138:2205–2211. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Slowik A, Borratynska A, Turaj W, Pera J,
Dziedzic T, Figlewicz DA, Betlej M, Krzyszkowski T, Czepko R and
Szczudlik A: Alpha1-antichymotrypsin gene (SERPINA3) A/T
polymorphism as a risk factor for aneurysmal subarachnoid
hemorrhage. Stroke. 36:737–740. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
O'Brien M: The reciprocal relationship
between inflammation and coagulation. Top Companion Anim Med.
27:46–52. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Levi M: The coagulant response in sepsis
and inflammation. Hamostaseologie. 30:10–12. 14–16. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Levi M and van der Poll T: Inflammation
and coagulation. Crit Care Med. 38 (Suppl 2):S26–S34. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Sohn EH, Khanna A, Tucker BA, Abràmoff MD,
Stone EM and Mullins RF: Structural and biochemical analyses of
choroidal thickness in human donor eyes. Invest Ophthalmol Vis Sci.
55:1352–1360. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Nazir S, Jankowski V, Bender G, Zewinger
S, Rye KA and van der Vorst EPC: Interaction between high-density
lipoproteins and inflammation: Function matters more than
concentration! Adv Drug Deliv Rev. 159:94–119. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Maïga SF, Kalopissis AD and Chabert M:
Apolipoprotein A-II is a key regulatory factor of HDL metabolism as
appears from studies with transgenic animals and clinical outcomes.
Biochimie. 96:56–66. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
da Silva IT, de Almeida-Pititto B and
Ferreira SRG: Reassessing lipid metabolism and its potentialities
in the prediction of cardiovascular risk. Arch Endocrinol Metab.
59:171–180. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Lehman JJ, Barger PM, Kovacs A, Saffitz
JE, Medeiros DM and Kelly DP: Peroxisome proliferator-activated
receptor gamma coactivator-1 promotes cardiac mitochondrial
biogenesis. J Clin Invest. 106:847–856. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Price NL, Gomes AP, Ling AJ, Duarte FV,
Martin-Montalvo A, North BJ, Agarwal B, Ye L, Ramadori G, Teodoro
JS, et al: SIRT1 is required for AMPK activation and the beneficial
effects of resveratrol on mitochondrial function. Cell Metab.
15:675–690. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Vazquez F, Lim JH, Chim H, Bhalla K,
Girnun G, Pierce K, Clish CB, Granter SR, Widlund HR, Spiegelman BM
and Puigserver P: PGC1α expression defines a subset of human
melanoma tumors with increased mitochondrial capacity and
resistance to oxidative stress. Cancer Cell. 23:287–301. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Zhang L, Zhou Y, Wu W, Hou L, Chen H, Zuo
B, Xiong Y and Yang J: Skeletal muscle-specific overexpression of
PGC-1α induces Fiber-type conversion through enhanced mitochondrial
respiration and fatty acid oxidation in mice and pigs. Int J Biol
Sci. 13:1152–1162. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Lin J, Wu H, Tarr PT, Zhang CY, Wu Z, Boss
O, Michael LF, Puigserver P, Isotani E, Olson EN, et al:
Transcriptional co-activator PGC-1 alpha drives the formation of
slow-twitch muscle fibres. Nature. 418:797–801. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Puigserver P, Wu Z, Park CW, Graves R,
Wright M and Spiegelman BM: A cold-inducible coactivator of nuclear
receptors linked to adaptive thermogenesis. Cell. 92:829–839. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Eisele PS, Salatino S, Sobek J, Hottiger
MO and Handschin C: The peroxisome proliferator-activated receptor
γ coactivator 1α/β (PGC-1) coactivators repress the transcriptional
activity of NF-κB in skeletal muscle cells. J Biol Chem.
288:2246–2260. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Lira VA, Benton CR, Yan Z and Bonen A:
PGC-1alpha regulation by exercise training and its influences on
muscle function and insulin sensitivity. Am J Physiol Endocrinol
Metab. 299:E145–E161. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Sandri M, Sandri C, Gilbert A, Skurk C,
Calabria E, Picard A, Walsh K, Schiaffino S, Lecker SH and Goldberg
AL: Foxo transcription factors induce the atrophy-related ubiquitin
ligase atrogin-1 and cause skeletal muscle atrophy. Cell.
117:399–412. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Stitt TN, Drujan D, Clarke BA, Panaro F,
Timofeyva Y, Kline WO, Gonzalez M, Yancopoulos GD and Glass DJ: The
IGF-1/PI3K/Akt pathway prevents expression of muscle
atrophy-induced ubiquitin ligases by inhibiting FOXO transcription
factors. Mol Cell. 14:395–403. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Guttridge DC, Mayo MW, Madrid LV, Wang CY
and Baldwin AS Jr: NF-kappaB-induced loss of MyoD messenger RNA:
possible role in muscle decay and cachexia. Science. 289:2363–2366.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Kang C, Shin WS, Yeo D, Lim W, Zhang T and
Ji LL: Anti-inflammatory effect of avenanthramides via NF-κB
pathways in C2C12 skeletal muscle cells. Free Radic Biol Med.
117:30–36. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Solomon V and Goldberg AL: Importance of
the ATP-ubiquitin-proteasome pathway in the degradation of soluble
and myofibrillar proteins in rabbit muscle extracts. J Biol Chem.
271:26690–26697. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Scott SV and Klionsky DJ: Delivery of
proteins and organelles to the vacuole from the cytoplasm. Curr
Opin Cell Biol. 10:523–529. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Lecker SH, Goldberg AL and Mitch WE:
Protein degradation by the ubiquitin-proteasome pathway in normal
and disease states. J Am Soc Nephrol. 17:1807–1819. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Papanicolaou KN, Izumiya Y and Walsh K:
Forkhead transcription factors and cardiovascular biology. Circ
Res. 102:16–31. 2008. View Article : Google Scholar : PubMed/NCBI
|