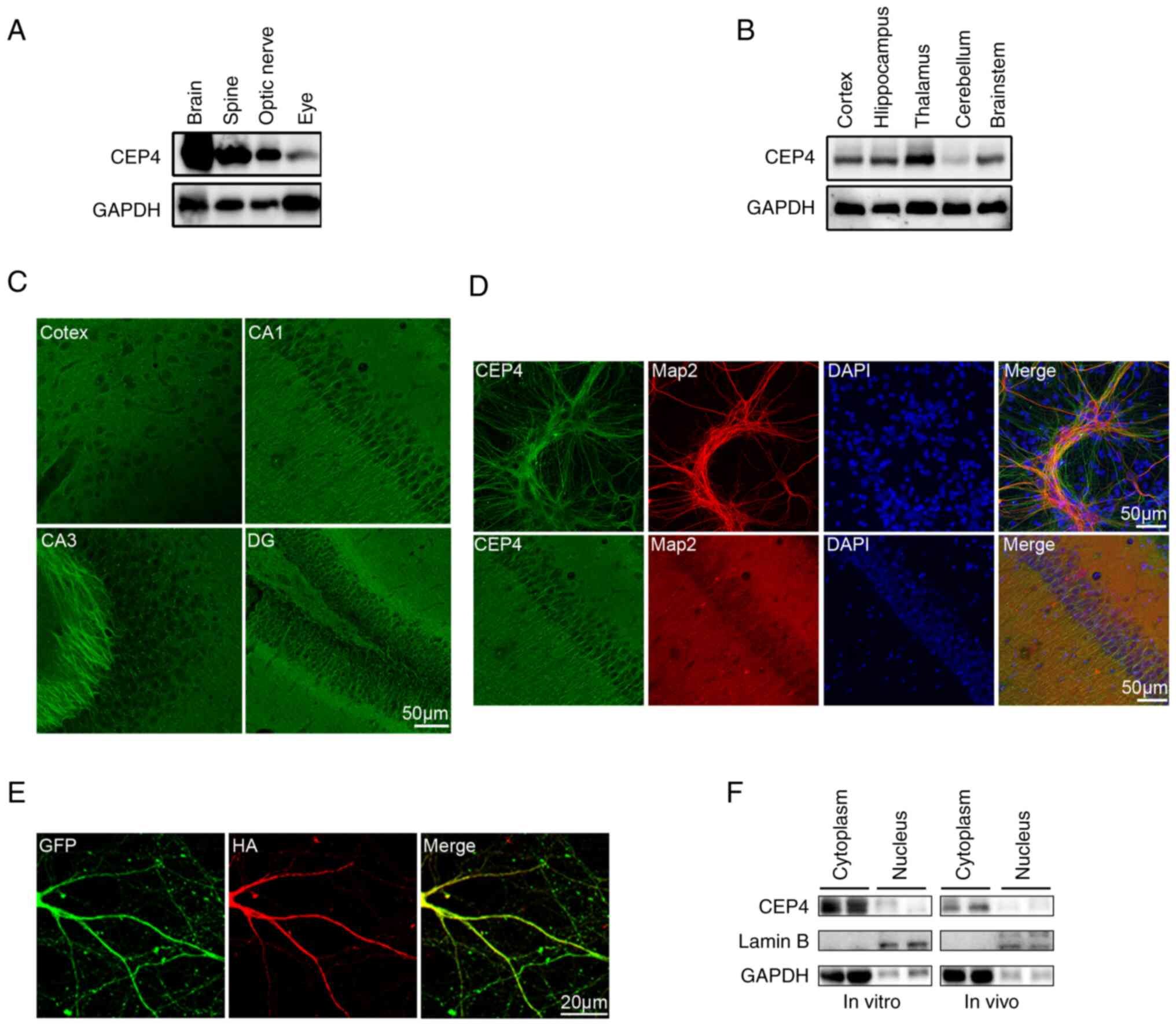
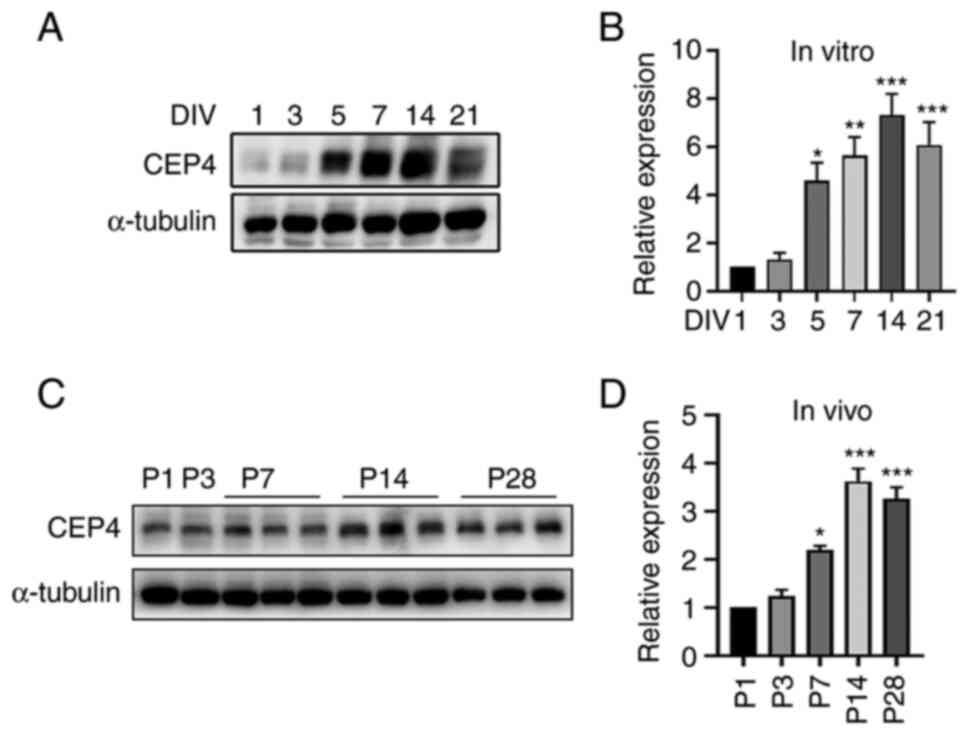
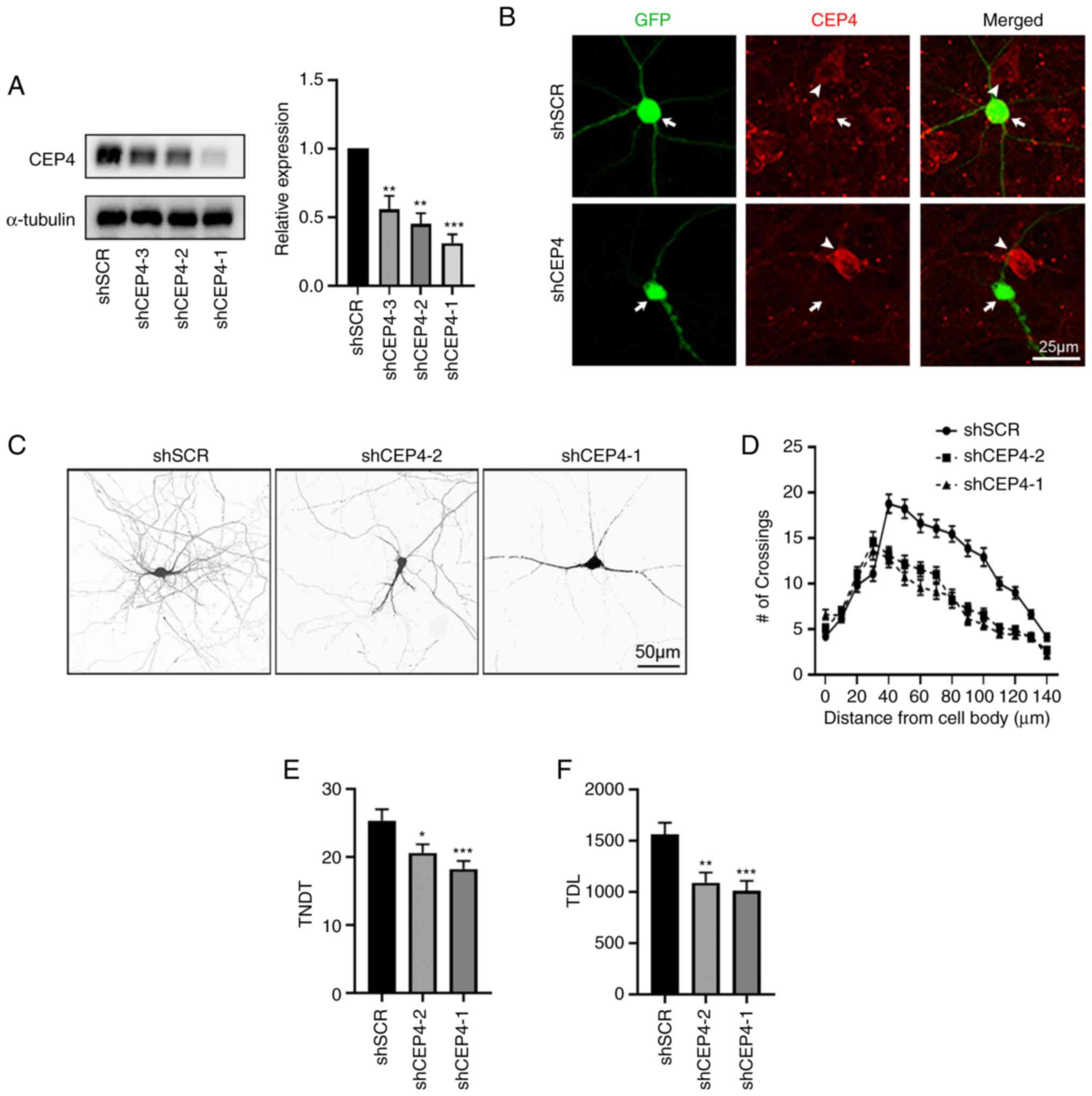
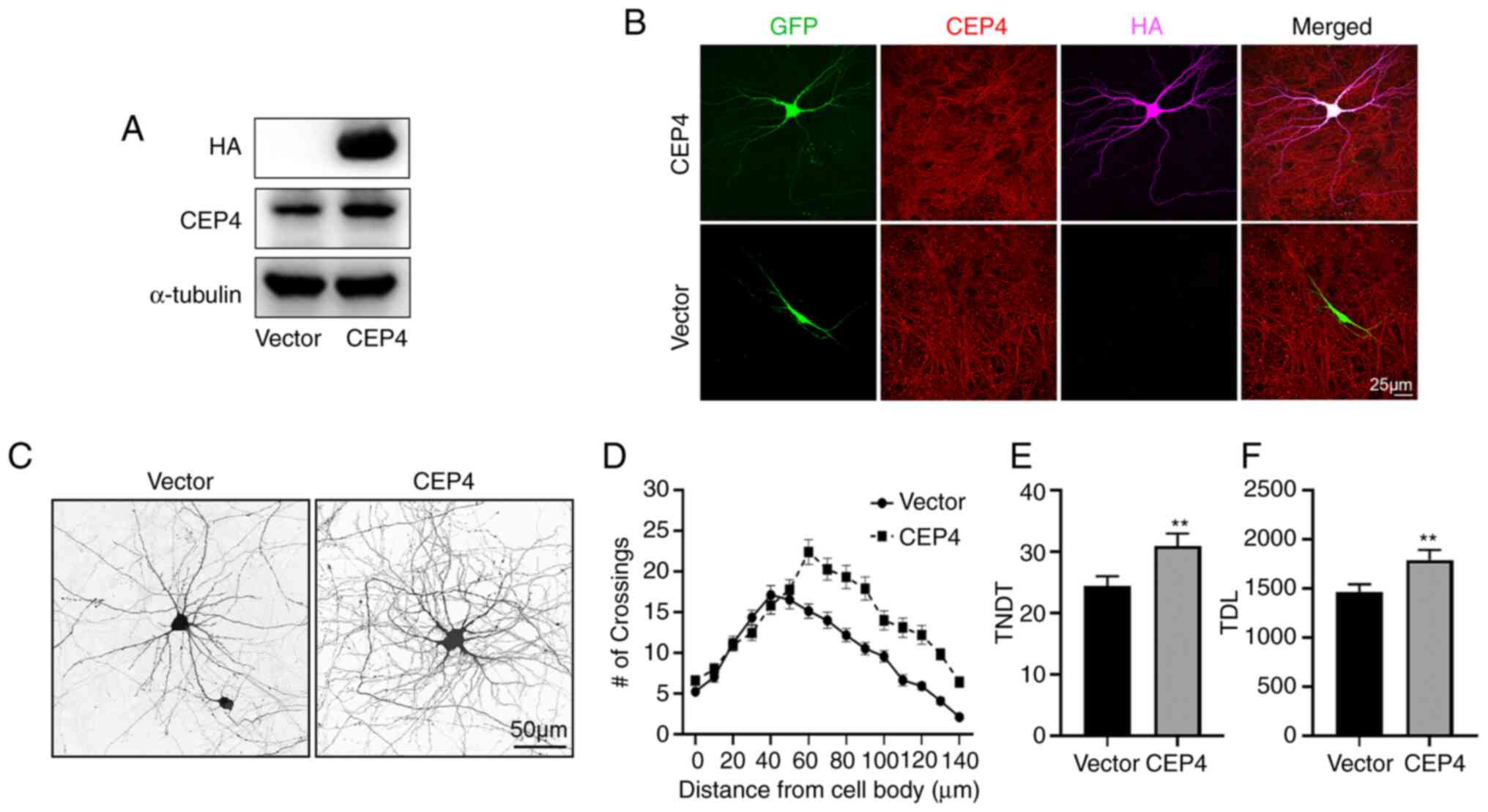
|
1
|
Takano T, Xu C, Funahashi Y, Namba T and
Kaibuchi K: Neuronal polarization. Development. 142:2088–2093.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Muñoz-Lasso DC, Romá-Mateo C, Pallardó FV
and Gonzalez-Cabo P: Much more than a scaffold: Cytoskeletal
proteins in neurological disorders. Cells. 9:3582020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ledda F and Paratcha G: Mechanisms
regulating dendritic arbor patterning. Cell Mol Life Sci.
74:4511–4537. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hodge RG and Ridley AJ: Regulating Rho
GTPases and their regulators. Nat Rev Mol Cell Biol. 17:496–510.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen H and Firestein BL: RhoA regulates
dendrite branching in hippocampal neurons by decreasing cypin
protein levels. J Neurosci. 27:8378–8386. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Leemhuis J, Boutillier S, Barth H,
Feuerstein TJ, Brock C, Nürnberg B, Aktories K and Meyer DK: Rho
GTPases and phosphoinositide 3-kinase organize formation of
branched dendrites. J Biol Chem. 279:585–596. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stankiewicz TR and Linseman DA: Rho family
GTPases: Key players in neuronal development, neuronal survival,
and neurodegeneration. Front Cell Neurosci. 8:3142014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen C, Wirth A and Ponimaskin E: Cdc42:
An important regulator of neuronal morphology. Int J Biochem Cell
Biol. 44:447–451. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Scott GA and Cassidy L: Rac1 mediates
dendrite formation in response to melanocyte stimulating hormone
and ultraviolet light in a murine melanoma model. J Invest
Dermatol. 111:243–250. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Threadgill R, Bobb K and Ghosh A:
Regulation of dendritic growth and remodeling by Rho, Rac, and
Cdc42. Neuron. 19:625–634. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kalpachidou T, Spiecker L, Kress M and
Quarta S: Rho GTPases in the physiology and pathophysiology of
peripheral sensory neurons. Cells. 8:5912019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kamiyama D and Chiba A: Endogenous
activation patterns of Cdc42 GTPase within Drosophila
embryos. Science. 324:1338–1340. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kreis P, Thévenot E, Rousseau V, Boda B,
Muller D and Barnier JV: The p21-activated kinase 3 implicated in
mental retardation regulates spine morphogenesis through a
Cdc42-dependent pathway. J Biol Chem. 282:21497–21506. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Luo L, Liao YJ, Jan LY and Jan YN:
Distinct morphogenetic functions of similar small GTPases:
Drosophila Drac1 is involved in axonal outgrowth and
myoblast fusion. Genes Dev. 8:1787–1802. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Scott EK, Reuter JE and Luo L: Small
GTPase Cdc42 is required for multiple aspects of dendritic
morphogenesis. J Neurosci. 23:3118–3123. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Z, Van Aelst L and Cline HT: Rho
GTPases regulate distinct aspects of dendritic arbor growth in
Xenopus central neurons in vivo. Nat Neurosci. 3:217–225.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ruchhoeft ML, Ohnuma S, McNeill L, Holt CE
and Harris WA: The neuronal architecture of Xenopus retinal
ganglion cells is sculpted by rho-family GTPases in vivo. J
Neurosci. 19:8454–8463. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Franke K, Otto W, Johannes S, Baumgart J,
Nitsch R and Schumacher S: miR-124-regulated RhoG reduces neuronal
process complexity via ELMO/Dock180/Rac1 and Cdc42 signalling. EMBO
J. 31:2908–2921. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rosário M, Schuster S, Jüttner R,
Parthasarathy S, Tarabykin V and Birchmeier W: Neocortical
dendritic complexity is controlled during development by
NOMA-GAP-dependent inhibition of Cdc42 and activation of cofilin.
Genes Dev. 26:1743–1757. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Joberty G, Perlungher RR and Macara IG:
The Borgs, a new family of Cdc42 and TC10 GTPase-interacting
proteins. Mol Cell Biol. 19:6585–6597. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Osada N, Kusuda J, Suzuki Y, Sugano S and
Hashimoto K: Sequence analysis, gene expression, and chromosomal
assignment of mouse Borg4 gene and its human orthologue. J Hum
Genet. 45:374–377. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhao X and Rotenberg SA: Phosphorylation
of Cdc42 effector protein-4 (CEP4) by protein kinase C promotes
motility of human breast cells. J Biol Chem. 289:25844–25854. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen X, Zhao X, Abeyweera TP and Rotenberg
SA: Analysis of substrates of protein kinase C isoforms in human
breast cells by the traceable kinase method. Biochemistry.
51:7087–7097. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wangsa D, Chowdhury SA, Ryott M, Gertz EM,
Elmberger G, Auer G, Lundqvist EÅ, Küffer S, Ströbel P, Schäffer
AA, et al: Phylogenetic analysis of multiple FISH markers in oral
tongue squamous cell carcinoma suggests that a diverse distribution
of copy number changes is associated with poor prognosis. Int J
Cancer. 138:98–109. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hirsch DS, Pirone DM and Burbelo PD: A new
family of Cdc42 effector proteins, CEPs, function in fibroblast and
epithelial cell shape changes. J Biol Chem. 276:875–883. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Passafaro M, Piëch V and Sheng M:
Subunit-specific temporal and spatial patterns of AMPA receptor
exocytosis in hippocampal neurons. Nat Neurosci. 4:917–926. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen K, Zhu L, Guo L, Pan YB and Feng DF:
Maf1 regulates dendritic morphogenesis and influences learning and
memory. Cell Death Dis. 11:6062020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Peng Y, Lee J, Rowland K, Wen Y, Hua H,
Carlson N, Lavania S, Parrish JZ and Kim MD: Regulation of dendrite
growth and maintenance by exocytosis. J Cell Sci. 128:4279–4292.
2015.PubMed/NCBI
|
|
29
|
Jaworski J, Spangler S, Seeburg DP,
Hoogenraad CC and Sheng M: Control of dendritic arborization by the
phosphoinositide-3′-kinase-Akt-mammalian target of rapamycin
pathway. J Neurosci. 25:11300–11312. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Swiech L, Blazejczyk M, Urbanska M,
Pietruszka P, Dortland BR, Malik AR, Wulf PS, Hoogenraad CC and
Jaworski J: CLIP-170 and IQGAP1 cooperatively regulate dendrite
morphology. J Neurosci. 31:4555–4568. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Van Aelst L and Cline HT: Rho GTPases and
activity-dependent dendrite development. Curr Opin Neurobiol.
14:297–304. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chan WWR, Li W, Chang RCC and Lau KF:
ARF6-Rac1 signaling-mediated neurite outgrowth is potentiated by
the neuronal adaptor FE65 through orchestrating ARF6 and ELMO1.
FASEB J. 34:16397–16413. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Narayanan R, Pham L, Kerimoglu C, Watanabe
T, Hernandez RC, Sokpor G, Ulmke PA, Kiszka KA, Tonchev AB,
Rosenbusch J, et al: Chromatin remodeling BAF155 subunit regulates
the genesis of basal progenitors in developing cortex. iScience.
4:109–126. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rao DD, Vorhies JS, Senzer N and
Nemunaitis J: siRNA vs. shRNA: Similarities and differences. Adv
Drug Deliv Rev. 61:746–759. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Moazed D: Small RNAs in transcriptional
gene silencing and genome defence. Nature. 457:413–420. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Boettcher M and McManus MT: Choosing the
right tool for the job: RNAi, TALEN, or CRISPR. Mol Cell.
58:575–585. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Williams DW and Truman JW: Mechanisms of
dendritic elaboration of sensory neurons in Drosophila:
Insights from in vivo time lapse. J Neurosci. 24:1541–1550. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li Y, Li X, Wang H, Gao Q, Zhang J, Zhang
W, Zhang Z, Li L, Yu Y and Shuai L: CRISPR/Cas9-edited Pax6-GFP
reporter system facilitates the generation of mouse neural
progenitor cells during differentiation. J Genet Genomics.
45:277–280. 2018. View Article : Google Scholar : PubMed/NCBI
|